Endothelial glycocalyx damage in patients with severe COVID-19 on mechanical ventilation – A prospective observational pilot study
Abstract
BACKGROUND:
Coronavirus disease (COVID-19) associated endotheliopathy and microvascular dysfunction are of concern.
OBJECTIVE:
The objective of the present single-center observational pilot study was to compare endothelial glycocalyx (EG) damage and endotheliopathy in patients with severe COVID-19 (COVID-19 group) with patients with bacterial pneumonia with septic shock (non-COVID group).
METHODS:
Biomarkers of EG damage (syndecan-1), endothelial cells (EC) damage (thrombomodulin), and activation (P-selectin) were measured in blood on three consecutive days from admission to the intensive care unit (ICU). The sublingual microcirculation was studied by Side-stream Dark Field (SDF) imaging with automatic assessment.
RESULTS:
We enrolled 13 patients in the non-COVID group (mean age 70 years, 6 women), and 15 in the COVID-19 group (64 years old, 3 women). The plasma concentrations of syndecan-1 were significantly higher in the COVID-19 group during all three days. Differences regarding other biomarkers were not statistically significant. The assessment of the sublingual microcirculation showed improvement on Day 2 in the COVID-19 group. Plasma levels of C-reactive protein (CRP) were significantly higher on the first two days in the COVID-19 group. Plasma syndecan-1 and CRP were higher in patients suffering from severe COVID-19 pneumonia compared to bacterial pneumonia patients.
CONCLUSIONS:
These findings support the role of EG injury in the microvascular dysfunction in COVID-19 patients who require ICU.
1Introduction
The worldwide spread of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus has prompted changes to almost every aspect of human life. The coronavirus disease induced by SARS-CoV-2 first diagnosed in 2019 (COVID-19) presents usually as a respiratory disorder sometimes requiring high-flow oxygen therapy. A subgroup of patients needs mechanical ventilatory support [1]. SARS-CoV-2 enters the cells through binding to the angiotensin-converting enzyme (ACE)2 receptor in epithelial cells. Its subsequent target is endothelial cells (EC), and the resulting clinical syndrome appears to be aggravated by damage to the endothelial glycocalyx (EG) layer [2–6]. Thus, a link between COVID-19 and endotheliopathy has been established [4, 7, 8]. EG plays an essential role in the proper functioning of microcirculation and thus in tissue metabolism [9].
The microcirculation in COVID-19 patients has been shown dysfunctional by direct light stroboscopic microscopy of the sublingual mucosa as well as in terms of increased plasma concentrations of biomarkers of endothelial dysfunction and EG shedding [10]. Thrombotic diathesis associated with EG dysfunction and pulmonary embolism aggravates the pulmonary dysfunction [11]. This phenomenon of obstructive microthrombosis of small and medium-sized vessels might lead to multiorgan failure as the disease progresses [12, 13]. Besides specific antiviral and antibody therapy for COVID-19, patients were assigned to receive a combination of supportive therapy with therapeutic doses of heparin mostly via administration of low molecular weight heparin (LMWH) [14]. Heparin is supposed to have a protective effect on EG [15] and thus in COVID-19, this approach could be protective for the microcirculation, although it has been previously shown that therapeutic doses of anticoagulation did not benefit patients with COVID-19 and elevated D-dimers levels [16].
Side-stream dark field imaging (SDF) of the sublingual microcirculation is a tool for assessment of the microcirculation [17] that gives insight also into the status of other microcirculatory beds within different body organ systems [18, 19]. In COVID-19 patients it represents an easy-to-use technique with quick automatic assessment. On the one side there is a lack on studies about microcirculation in COVID-19, on the other side this approach would provide us with a unique insight into the pathophysiology of this disease [20].
2Objective
The aim of the present study was to examine the difference in the EG damage, microcirculatory parameters in the sublingual area, and endotheliopathy between COVID-19 patients and non-COVID patients with bacterial pneumonia and septic shock. We hypothesize that COVID-19 patients have more severe EG damage, a disorder of sublingual microcirculation and endotheliopathy.
3Materials and methods
We carried out a prospective non-randomized single-center observational pilot clinical study of adult patients with septic shock associated with COVID-19 pneumonia or bacterial pneumonia. The protocol was approved by the local ethics committee (Ethics Committee, University Hospital Hradec Kralove, Czech Republic, approval number 72195/2020-684800). The study was conducted in accordance with the Helsinki Declaration and good clinical practice. Informed consent was required and signed with patients’ relatives and an independent physician without relation to the study. The study has been registered at ClinicalTrial.gov with trial registration number NCT04644302.
3.1Selection of participants
Inclusion criteria were age over 18 years, septic shock caused by community-acquired bacterial pneumonia [21], or SARS-CoV-2 viral pneumonia (septic shock de-fined according to the latest Survival Sepsis Campaign: hypotension requiring vasopressors despite adequate fluid resuscitation [22]), treatment with mechanical ventilation, radiological signs of pneumonia with pulmonary dysfunction (oxygenation abnormalities with Horrowitz index below 200), positive microbiological culture from tracheal aspirate with Gram+ cocci and in COVID-19 patients positive antigen test (PanbioTM COVID-19 Ag Rapid Test Device, Abbott, Jena, Germany) confirmed by positive PCR test.
Exclusion criteria were initiation of palliative care within three days from admission and patient’s relative’s refusal to study participation, absence of symptoms and signs of cardiac or obstructive shock, in patients with COVID-19 another source of sepsis, a preexisting known bacterial disease, presence of other EG damage related disease (autoimmune disease, major trauma, cardiac arrest, renal failure treated by hemodialysis, radiotherapy and/or chemotherapy in the 6 months history) and COVID-19 in patient’s history.
Patients were recruited from February to November 2020 and divided into two groups: non-COVID pneumonia vs. COVID-19.
3.2Patients’ specific and supportive treatment
The therapeutic approach was the same in all COVID-19 patients. The pharmacotherapy consisted of cefotaxime from the day of mechanical ventilation initiation (Taximed, Medochemie Bohemia, Prague, Czech Republic), azithromycin (Sumamed, Teva Pharmaceuticals CR, Prague, Czech Republic), two units of convalescent plasma, dexamethasone before deterioration requiring mechanical ventilation (8 mg per day for 14 days, Dexamed, Medochemie, Limassol, Cyprus) and after intubation methylprednisolone (80 mg per day, Pfizer, Prague, Czech Republic), ascorbic acid (16 g per day, Acidum ascorbicum, BB Pharma, Prague, Czech Republic), thiamin (400 mg per day, Thiamin, Zentiva, Prague, Czech Republic), LMWH (nadroparin, Fraxiparine, Aspen Pharma, Dublin, Ireland, 0,1 ml/10 kg twice a day up to maximum dose of 1 ml/12 hours). In patients with D-dimers over 20 mg/l, 50 mg of alteplase (Actilyse, Boehringer, Ingelheim am Rhein, Germany) was given over 24 hours as rescue therapy in patients with Horrowitz index below 100. All patients received a continuous infusion of atracurium (Tracrium, Aspen Pharma, Dublin, Ireland) up to the level of relaxation with two twitches of the thumb of the upper arm in a train of four ratio. In this group, patients also received antihistamine agents (20 mg/day of cetirizine, Zyrtec, UCB, Prague, Czech Republic; 40 mg/day of famotidine, Famosan, PROMEDCS, Prague, Czech Republic) and melatonin (10 mg per day, Circadin, RAD Neurim Pharmaceuticals EEC SARL, Paris, France). All the COVID-19 patients had initially been in the prone position for 18 hours.
In non-COVID patients with bacterial pneumonia, first-choice empiric antibiotic therapy was cefotaxime and clarithromycin (Klacid, Mylan IRE Healthcare, Dublin, Ireland) together with hydrocortisone (200 mg per day, Hydrocortison, VUAB Pharma, Roztoky, Czech Republic), ascorbic acid (6 g per day as a standardized therapy), thiamine (400 mg per day, Thiamin, Zentiva, Prague, Czech Republic) and gastric stress ulcers prophylaxis by pantoprazole (80 mg per day, Nolpaza, Krka, Novo mesto, Slovenia). Fluid therapy was done by balanced crystalloid solution (Ringerfundin, BBraun, Melsungen, Germany).
3.3Study parameters
Besides demographic data and comorbidities obtained from patient’s records, a variety of relevant clinical, physiological, and laboratory data were recorded immediately on admission (Day 1) and on two following days of the ICU stay (Day 2 and 3). Blood samples for measurement of biomarkers of EG damage (syndecan-1; Human Syndecan AB Clone ELISA kit, Woburn, MA, USA), endothelial cell activation (P-selectin; ELISA kit MyBioSource, San Diego, CA, USA), and damage (thrombomodulin; ELISA kit, MyBioSource, San Diego, CA, USA) were taken at the same time on admission and in three consecutive days.
3.4Microcirculation and endothelial glycocalyx assessment
Videos of the sublingual microcirculation were recorded using a handheld MicroScan microscopy camera (image size: 720×480 pixels; field view: 1044×758μm; software: AVA 4.3 C; MicroVision Medical, Amsterdam, The Netherlands; see Fig. 1). A detailed description of the utilized SDF technology has been published elsewhere [23]. Recordings were done on admission (Day 1) and during three consecutive days. The assessment was performed according to the second international consensus on the assessment of the sublingual microcirculation in the critically ill [24]. For each time point, 4 recordings were obtained from different places underneath the patient’s tongue (two on each side in the sublingual lateral sulcus). The video acquisition process was guided by the AVA 4.3 C software to ensure adequate brightness, contrast, and stability. The recording was finished after the acquisition of 60 frames (30 frames per second) of microcirculation of vessels of diameter from 5 to 25μm. Care was taken to avoid pressure on the tissue under the probe which can decrease blood flow in the capillaries. Recordings were then analyzed offline using the same software AVA 4.3 C providing parameters according to the consensus (Fig. 2) [25]:
Fig. 1
Image of the sublingual microcirculation by Side-stream Dark Field imaging. Area of 1044×758μm.

Fig. 2
Software processing of a sublingual microcirculation recording performed by Side-stream Dark Field imaging. The same area is visualized as in Figure 1.

1. Number of vessel crossings –the total number of vessels crossing three arbitrary horizontal and three vertical equidistant lines (drawn on the screen, Fig. 2),
2. De Backer score –given in mm- 1, calculated as the number of crossings divided by the total length of the lines,
3. Proportion of Perfused Vessels (PPV) –a percentage of perfused vessels out of all visible vessels. PPV small stands for capillaries up to 25μm.
3.5Outcome measures
The primary study outcome was the plasma concentration of syndecan-1 which served as an indicator of EG damage. Secondary outcomes were plasma levels of thrombomodulin, P-selectin, and three variables of the sublingual microcirculation described above.
3.6Statistical analysis
Statistical software Prism 9 for macOS was used (version 9.0.2, GraphPad Software, San Diego, CA, USA). All data were tested for normality by the D’Agostino & Pearson test and basic descriptive statistics were done. Data are expressed as mean (±standard deviation (SD)) in the case of normal distribution, or as median (with interquartile range) for non-normal distributed parameters. For contingency table analysis, Fisher’s exact test was used. For single data set comparison, the unpaired t-test or Mann-Whitney test were used when appropriate. For multiple comparisons, repeated measures two-way analysis of variance (ANOVA) or Friedman test was used based on the distribution of the data. A p-value of 0.05 was accepted as statistically significant.
4Results
In our study, we enrolled 28 patients: 13 in the non-COVID pneumonia group (6 women) and 15 in the COVID-19 group (3 women). Data on demography and daily medications are given in Table 1. The average age was 67 (±9.6) years. Arterial hypertension, type two diabetes mellitus, and obesity were the most common comorbidities.
Table 1
Baseline demographics and patient history
| Characteristics | All patients (n = 28) | non-COVID (n = 13) | COVID-19 (n = 15) | p value |
| Gender [male/female] | 19/9 | 7/6 | 12/3 | ns |
| Age [years] | 67 (±9.6) | 70 (±11) | 64 (±7.1) | ns |
| BMI [kg/m2] | 28 (26–33) | 28 (24–35) | 28 (26–33) | ns |
| COPD [n (%)] | 5 (18%) | 5 (38%) | 0 | 0.05 |
| Asthma bronchiale [n (%)] | 3 (11%) | 1 (8%) | 2 (13%) | ns |
| Arterial hypertension [n (%)] | 11 (39%) | 3 (23%) | 8 (53%) | ns |
| Diabetes mellitus [n (%)] | 11 (39%) | 4 (30%) | 7 (46%) | ns |
| Obesity [n (%)] | 11 (39%) | 4 (30%) | 7 (46%) | ns |
| Coronary artery disease [n (%)] | 8 (29%) | 5 (38%) | 3 (20%) | ns |
| Hyperlipidemia [n (%)] | 4 (14%) | 0 | 4 (27%) | ns |
| acetylsalicylic acid [n (%)] | 4 (14%) | 2 (1%) | 2 (13%) | ns |
| atorvastatin [n (%)] | 4 (14%) | 1 (8%) | 3 (20%) | ns |
| ACEi [n (%)] | 4 (14%) | 1 (8%) | 3 (20%) | ns |
| metformin [n (%)] | 4 (14%) | 1 (8%) | 3 (20%) | ns |
BMI –body mass index, COPD –chronic obstructive pulmonary disease, ACEi –angiotensin-converting enzyme inhibitors, data are presented as mean (±standard deviation), median (interquartile range) or percentage, ns –not significant for Fisher’s exact test.
The average ICU length of stay was 13 days in the non-COVID group and 17 days in the COVID-19 group. Mortality within 30 days was 23 % in the non-COVID group (3 out of 13 patients) and 27% in the COVID-19 group (4 out of 15 patients).
COVID-19 patients had statistically higher SOFA scores on days 2 and 3 of the ICU stay (Table 2). They also had higher levels of C-reactive protein (CRP) on days 1 and 2 (Table 3).
Table 2
Clinical data
| Variable | non-COVID | COVID-19 | p values | ||||||
| Day on ICU | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 |
| SOFA score | 11 (±3.3) | 9.2 (±3.1) | 8.6 (±2.5) | 12 (±1.4) | 12 (±1.4) | 12 (±2.5) | ns | 0.004 | 0.0009 |
| MAP [mmHg] | 83 (±12) | 79 (±5.3) | 83 (±9.3) | 78 (±15) | 80 (±6.6) | 83 (±10) | ns | ns | ns |
| HR [bpm] | 89 (±35) | 78 (±21) | 81 (±15) | 103 (±23) | 82 (±21) | 76 (±20) | 0.04 | ns | ns |
| SpO2 [% ] | 95 (±3.5) | 94 (±2.7) | 94 (±3.3) | 90 (±7.7) | 94 (±1.9) | 94 (±2.2) | 0.04 | ns | ns |
| Temperature [°C] | 35 (±2.4) | 37 (±1.6) | 37 (±1.1) | 37 (±1.2) | 37 (±1) | 37 (±1) | 0.02 | ns | ns |
| RR [bpm] | 18 (±6.7) | 18 (±2.6) | 17 (±2.2) | 25 (±6.5) | 22 (±4) | 22 (±4.5) | 0.0002 | 0.003 | 0.001 |
| Pinsp [cmH2O] | 25 (±6.9) | 25 (±6.2) | 23 (±5.8) | 27 (±3.1) | 26 (±3.4) | 26 (±3.1) | ns | ns | ns |
| PEEP [cmH2O] | 7.8 (±2.8) | 8.1 (±3.1) | 8.4 (±3.2) | 12 (±3) | 11 (±2.8) | 10 (±3.1) | 0.002 | 0.02 | ns |
| EtCO2 [mmHg] | 36 (±5.1) | 39 (±4.9) | 40 (±4.5) | 45 (±8.9) | 44 (±6.4) | 42 (±4.9) | 0.004 | 0.03 | ns |
| NORA [μg/kg/min] | 0.1 (0.07–0.19) | 0.05 (0–0.1) | 0.05 (0–0.09) | 0.1 (0.05–0.5)) | 0.06 (0.04–0.3) | 0.05 (0–0.4) | ns | ns | ns |
| Cumulative fluid | 3298 (1185–4283) | 4100 (75–5750) | 2500 (0–6661) | 1350 (500–3610) | 2700 (750–4800) | 2000 (–100–5500) | ns | ns | ns |
| balance [ml] | |||||||||
SOFA –sequential organ failure assessment, MAP –mean arterial pressure, HR –heart rate, SpO2 saturation of blood by oxygen, RR –respiratory rate, Pinsp in-spiratory pressure, PEEP –positive end expiratory pressure, EtCO2 end-tidal concentration of carbon dioxide, NORA –noradrenaline, data are presented as mean (±standard deviation) or median (interquartile range), p values represent day-matched data comparison between the groups (eighter unpaired t-test or Mann-Whitney test).
Table 3
Laboratory data
| Variable | non-COVID | COVID-19 | p values | ||||||
| Day on ICU | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 |
| pH | 7.3 (±0.1) | 7.4 (±0.1) | 7.4 (±0.07) | 7.3 (±0.1) | 7.4 (±0.13) | 7.4 (±0.12) | ns | ns | ns |
| pO2 [kPa] | 12 (±3.3) | 11 (±2.6) | 12 (±3.1) | 12 (±4) | 11 (±2.4) | 11 (±3.1) | ns | ns | ns |
| HI | 184 (±78) | 182 (±71) | 228 (±118) | 136 (±46) | 143 (±34) | 151 (±72) | ns | ns | ns |
| pCO2 [kPa] | 6.8 (±2.1) | 6.5 (±2.4) | 7 (±1.6) | 7.9 (±2.5) | 7.5 (±2) | 7.2 (±1.9) | ns | ns | ns |
| lactate [mmol/l] | 5.1 (±4.6) | 2.8 (±1.7) | 2.8 (±2.7) | 3 (±1.4) | 2.6 (±0.98) | 2.5 (±0.59) | ns | ns | ns |
| HCO3- [mmol/l] | 23 (±3.7) | 25 (±3.4) | 27 (±2.6) | 25 (±6) | 28 (±5.2) | 29 (±5.5) | ns | 0.03 | ns |
| Base excess | –2.4 (±4.7) | 0.52 (±3.9) | 2.9 (±3) | 0.25 (±6.8) | 4.1 (±5.7) | 5.5 (±5.9) | ns | ns | ns |
| Leukocytes [x109/mL] | 19 (±12) | 16 (±10) | 16 (±7.4) | 16 (±5.5) | 14 (±6) | 15 (±6.9) | ns | ns | ns |
| Thrombocytes [x109/mL] | 231 (±88) | 191 (±77) | 175 (±72) | 269 (±126) | 252 (±117) | 266 (±128) | ns | ns | 0.02 |
| Hemoglobin [g/L] | 132 (±33) | 123 (±26) | 117 (±23) | 116 (±19) | 111 (±18) | 106 (±20) | ns | ns | ns |
| Hematocrit [ratio] | 0.4 (±0.1) | 0.38 (±0.11) | 0.35 (±0.07) | 0.34 (±0.05) | 0.33 (±0.06) | 0.32 (±0.06) | ns | ns | ns |
| Glucose [mmol/L] | 13 (±4.8) | 9 (±1.9) | 8.6 (±2) | 13 (±7) | 11 (±4.7) | 10 (±3.2) | ns | ns | ns |
| Sodium [mmol/L] | 143 (±5) | 142 (±4.5) | 142 (±4.4) | 143 (±3) | 144 (±3.5) | 146 (±3.9) | ns | ns | 0.04 |
| Chloride [mmol/L] | 105 (±6.3) | 106 (±6.6) | 106 (±5) | 104 (±2.8) | 104 (±3.8) | 104 (±4.8) | ns | ns | ns |
| Urea [mmol/L] | 14 (±11) | 14 (±11) | 13 (±8.5) | 11 (±7.3) | 11 (±5.8) | 11 (±4.5) | ns | ns | ns |
| Bilirubin [mmol/L] | 14 (±9.6) | 13 (±6.2) | 12 (±6) | 14 (±7.9) | 10 (±6.4) | 7.8 (±3.4) | ns | ns | 0.02 |
| Creatinine [μmol/L] | 136 (±85) | 149 (±104) | 155 (±111) | 110 (±46) | 120 (±39) | 118 (±54) | ns | ns | ns |
| ACR [g/mol] | 27 (14–82) | 7.1 (3.6–39) | 9.7 (3.5–23) | 6 (3.3–20) | 7.2 (4.1–26) | 3.4 (2.4–13) | 0.02 | ns | ns |
| CRP [mmol/L] | 63 (9.7–136) | 85 (49–101) | 94 (50–124) | 181 (80–270) | 174 (95–213) | 108 (60–180) | 0.01 | 0.04 | ns |
| PCT [μmol/L] | 1.5 (0.12–47) | 6.6 (0.49–46) | 4.4 (0.2–31) | 1.1 (0.4–1.4) | 0.87 (0.49–1.4) | 0.5 (0.37–0.76) | ns | ns | ns |
| IL-6 [nmol/L] | 78 (30–245) | 65 (26–159) | 34 (15–81) | 107 (24–218) | 32 (11–112) | 13 (6.7–83) | ns | ns | ns |
pO2 –arterial partial pressure of oxygen, HI –Horrowitz index, pCO2 –arterial partial pressure of carbon dioxide, HCO3- –bicarbonate, CRP –C-reactive protein, PCT –procalcitonin, IL-6 interleukin 6, ACR –urine albumin-creatinine ratio, data are presented as mean (±standard deviation) or median (interquartile range). P values represent day-matched data comparison between the groups (eighter unpaired t-test or Mann-Whitney test).
The assessments of sublingual microcirculation are illustrated in Graphs 1–3. These measurements revealed statistically higher values of all parameters on day 2 of the ICU stay in the COVID-19 group: the number of crossings were 35 (±7.1) vs. 4.2 (±9.8), p = 0.05; DeBacker score 7.5 (±1.6) mm- 1 vs. 8.9 (±2) mm- 1, p = 0.05; PPV 68 (±20) % vs. 82 (±14) %,p = 0.05.
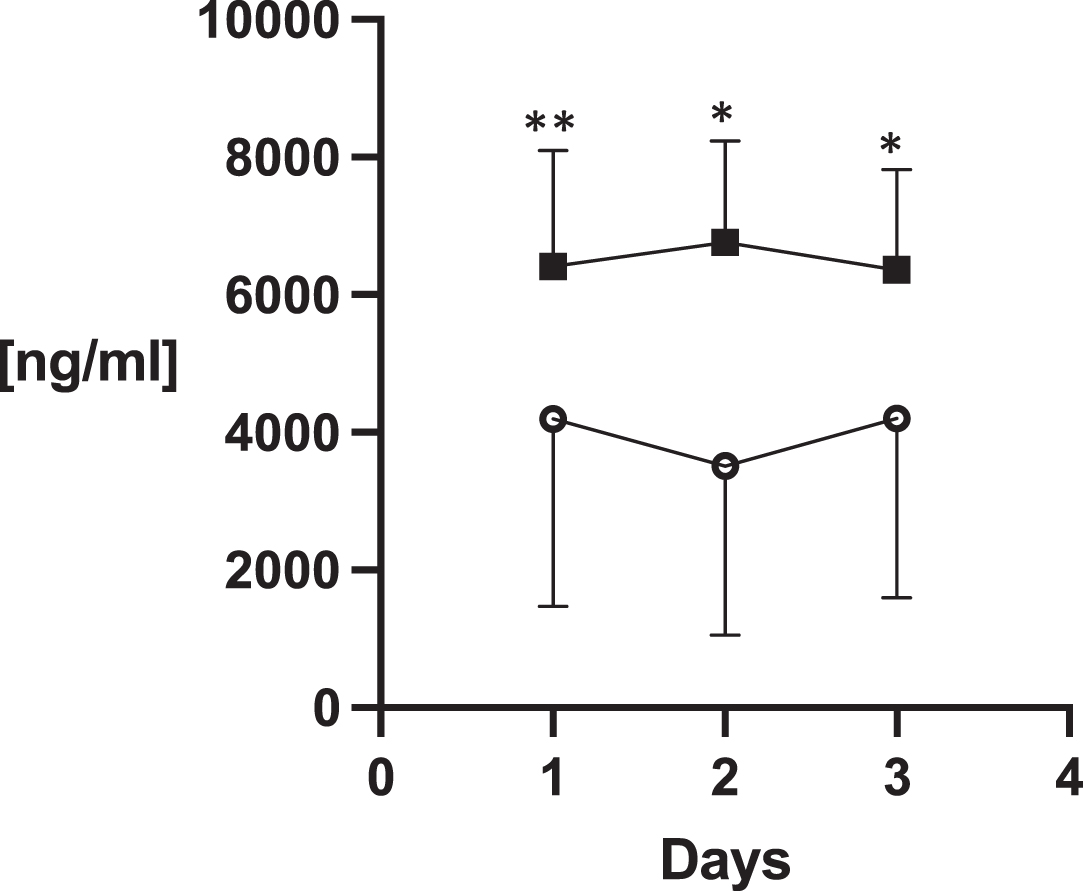
Syndecan-1 differed significantly between the groups over the monitored days of the ICU stay (ANOVA p < 0.0001; Graph 4). In other analyzed biomarkers there were no statistically significant group differences (Table 4).
Table 4
Microcirculation assessment and endothelial glycocalyx damage
| Variable | non-COVID | COVID-19 | p values | ||||||
| Day on ICU | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 | Day 1 | Day 2 | Day 3 |
| Laboratory data | |||||||||
| Thrombomodulin [ng/mL] | 5.4 (±4.3) | 5.5 (±4.4) | 5.3 (±4) | 5.5 (±4.4) | 5.4 (±4.1) | 4.8 (±3.4) | ns | ns | ns |
| P-selectin [ng/mL] | 75 (±34) | 61 (±36) | 63 (±44) | 61 (±36) | 65 (±36) | 59 (±35) | ns | ns | ns |
| Microcirculatory data | |||||||||
| PPV small [% ] | 64 (±19) | 58 (±19) | 65 (±19) | 67 (±16) | 73 (±18) | 74 (±13) | ns | 0.04 | ns |
PPV –proportion of perfused vessels, PPV small –proportion of perfused vessels with diameter up to 25μm. ICU –intensive care unit, data are presented as mean (±standard deviation) or median (interquartile range). P values represent day-matched data comparison between the groups (eighter unpaired t-test or Mann-Whitney test).
Fig. 3
DeBacker score.
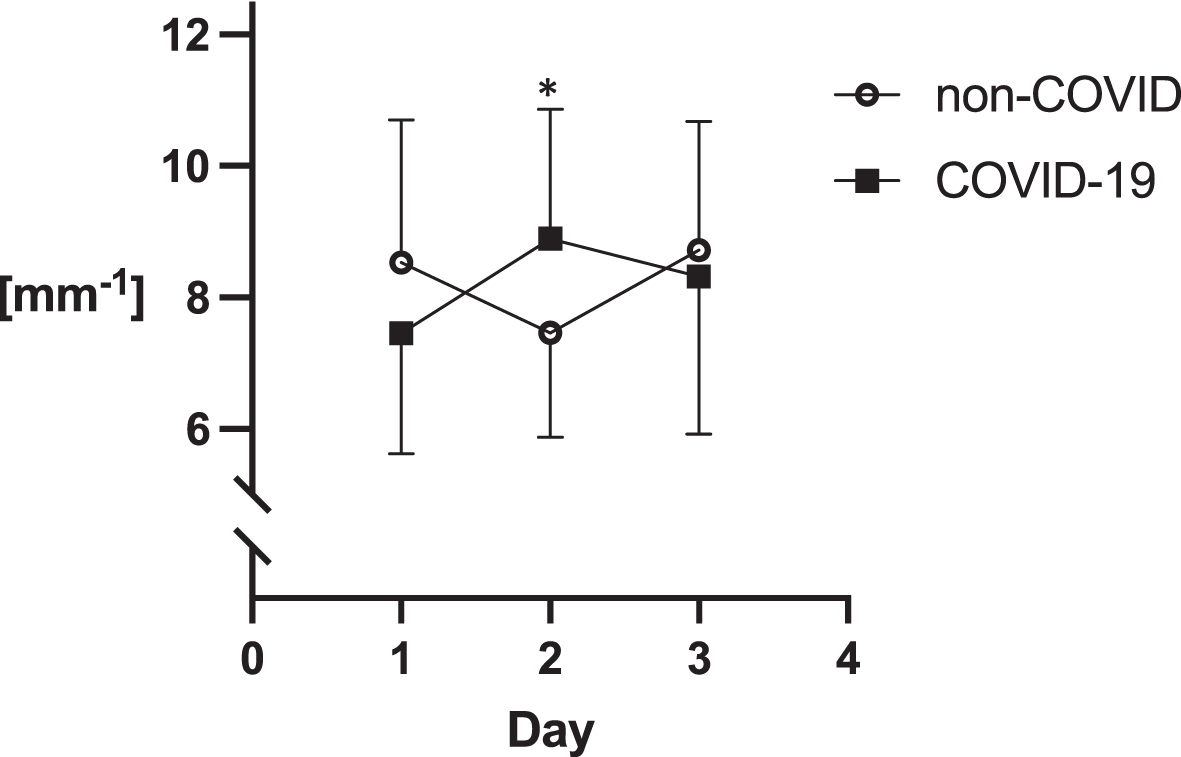
Fig. 4
Proportion of perfused vessels.
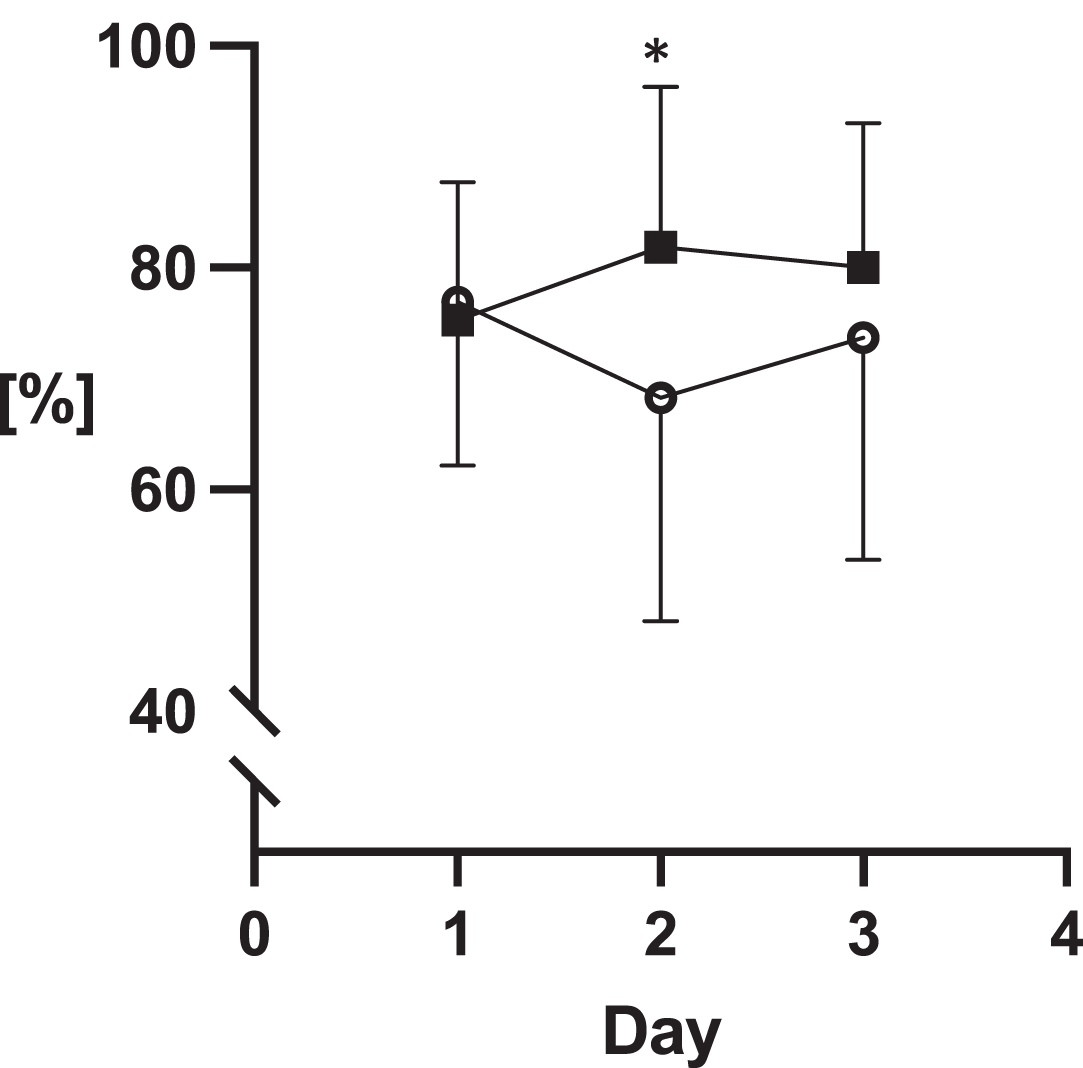
Fig. 5
Number of crossings.
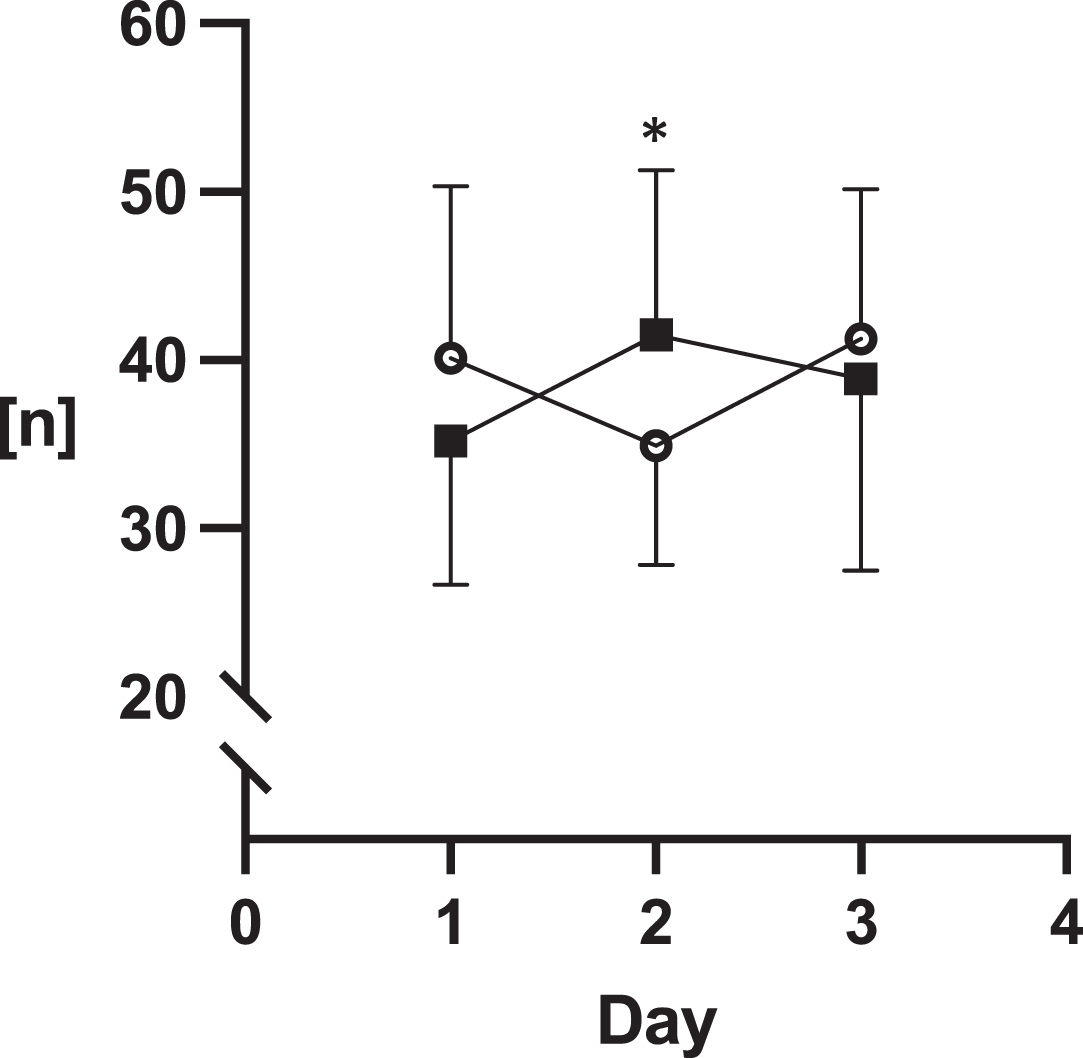
Fig. 6
Syndecan-1. Legend to the graphs: the asterisks stand for p values of unpaired t-tests of Mann-Whitney test. *P < 0.05, **P < 0.001.
5Discussion
The main finding of the presented study is that COVID-19 is associated with marked EG damage that is, in some respects, more severe than in bacterial sepsis. Syndecan-1, which is the key marker of EG damage, was significantly higher during three consecutive days of the ICU stay in the COVID-19 group compared to the non-COVID group. This finding supports our study hypothesis.
We believed that the EG damage would be more severe in the COVID-19 group due to a presumably stronger inflammatory reaction, which receives support from the higher CRP levels on all three days. We also supposed that the SARS-CoV-2 virus has an affinity to endothelial cells [26] but the markers of EC damage and activation did not show any statistically significant difference between the groups. However, all measured micro-circulatory variables showed better values in the COVID-19 group on Day 2 of ICU stay compared to the non-COVID group. This finding was surprising as we expected a better microcirculatory profile in the non-COVID group, where the EG damage was less severe. Finally, there were no significant differences between the two groups with regard to mortality and ICU length of stay.
Measurement of plasma biomarkers represents a gold standard in EG damage assessment. Although they are quantified by a precise ELISA method, they have some limitations [27]. Their plasma concentrations are short-lived and dependent on renal functions, especially heparan sulfate and hyaluronan. Syndecan-1, the core proteo-glycan in the EG structure seems to be the biomarker most suitable for the assessment of EC health [28].
Microvascular dysfunction in COVID-19 patients was confirmed also in the MYSTIC study by Rovas et al. where similar patterns of EG damage were found [29]. By contrast, markers of EC damage and activation in our study were inconclusive although we did not use healthy controls as the third cohort for comparison. We suppose that these markers will also be higher in bacterial pneumonia compared to healthy controls. Moreover, the EG damage can occur without dysfunction of the microcirculation [30]. A similar comparative study on a larger cohort of patients with hypertensive controls and healthy controls was done by Lambadiari et al. [31]. They focused on cardiac performance and measurement of Perfused Boundary Region (PBR) together with oxidative stress and endothelial biomarkers four months after COVID-19 illness. They found decreased cardiac performance in COVID-19 patients and higher PBR and thrombomodulin suggesting persisting EG damage and endothelial and vascular dysfunction 4 months after infection. As for thrombomodulin, in our study, we had higher values in both cohorts. Values in healthy controls in cited study were twenty-times lower. The EG in healthy volunteers was mapped by our research group [32]. Biomarker of EG damage, syndecan-1 in healthy people with no renal dysfunction has three-folds lower value than in present study cohorts. Regarding microcirculatory parameters, PPV is comparable with results from the study by Edul et al. [33]. In their study, they included healthy controls where the mean PPV was 100 %.
The differences in microcirculation parameters between our study groups can probably be explained by the differences in treatment approach, particularly regarding medication given to all COVID-19 patients admitted to ICU. COVID-19-associated coagulopathy and the risk for deep venous thrombosis were of serious concern [34]. Our patients received therapeutic dosing of LMWH. Heparin has been proved to protect EG and EC [35] in COVID-19 patients and our results support this concept, despite the fact that anticoagulation therapy with LMWH is not standard of care in severe COVID-19 patients [36]. Another factor affecting our results could be the corticosteroid therapy that was administered to all our COVID-19 patients. In our study, the COVID-19 group received 80 mg of methylprednisolone which was double the equivalent dose that the non-COVID group received (200 mg of hydrocortisone). Steroids have been shown to have EG protective potency [37] by several mechanisms, they are believed to stabilize endothelial barrier function and they also have strong anti-inflammatory effects ameliorating the COVID-19 vascular “endothelitis” [38]. This positive effect of corticosteroids is supported by evidence from many observational studies and randomized controlled trials [39]. Guidelines proposing corticosteroid treatment in COVID-19 patients with a strong recommendation and high level of evidence [40]. Last but not least, the COVID-19 patients were treated with statistically significant higher PEEP during the first two days in the ICU and higher respiratory rates on all three days, which was due to greater pulmonary damage. Whether or not this ventilatory setting had an impact on the microcirculation and the EG damage in COVID-19 patients is unclear.
The measurements of inflammatory biomarkers showed higher CRP levels in the COVID-19 patients, but IL-6 and procalcitonin concentrations were not significantly different. COVID-19 has been linked to high levels of cytokines [41] but, on the other hand, there are other diseases where the levels of inflammatory cytokines are even higher [42, 43]. This variation in inflammatory response results in a unique clinical phenotype of COVID-19 septic shock featuring a clear tendency towards a very intensive inflammatory response. These variations might be of importance to the EC and EG damage and microvascular dysfunction. A link to the multisystem inflammatory syndrome in COVID-19 has been established especially in children [44, 45].
Our results support the concept of the role of endothelial dysfunction as one of the mechanisms contributing to organ damage in COVID-19 patients and the higher incidence of thromboembolic events in this population [46–48]. The current level of evidence, together with our data, in our opinion, allows us to propose a new septic shock phenotype –COVID-19, characterized by a profound inflammatory reaction, concomitant damage of the endothelium accompanied by EG degradation, and subsequent microcirculatory dysfunction. In addition, this concept of immune-mediated glycocalyx remodeling in COVID-19 patients has already been published [49]. Early anti-endotheliopathy therapy in critically ill COVID-19 patients might be considered to target features of the COVID-19 inflammatory reaction phenotype. Moreover, longitudinal respiratory subphenotypes in COVID-19 related acute distress syndrome have already been identified [50].
Our study has several limitations. First, is the lack of a healthy cohort to compare the results of microcirculation assessment, EG damage, and the levels of laboratory parameters. Second, the window for our cytokines and markers of endotheliopathy was quite narrow in terms of their capability to describe entirely all key pathways responsible for EC damage and related activation. Third, for practical reasons, we conducted the study for three consecutive days only. Observing the microcirculation and evolution of EC damage parameters for a longer time might bring further important insights into the pathophysiology of COVID-19 endotheliopathy. The main reason for this was a clinical one, as previous studies have shown that the average time period between admission and development of significant changes in the clinical course (either clinical improvement or disease progression) is around 3 days in the majority of patients with sepsis. Also, the Microvision® camera and laptop needed to be cleaned precisely which was sometimes demanding in terms of work and space organization (superficial disinfection and 20 minutes of ultraviolet light exposure) and data recording were sometimes difficult due to the physical constraints with extra personal protective equipment. And finally, the study cohorts were disproportionate with regard to gender. This is due to the prospective and observational nature of the study with limited time for patient consecutive enrollment. Also, for this reason, the power analysis was not performed, and the study was designated as a pilot.
6Conclusions
EG damage was more severe in COVID-19 patients compared to the bacterial pneumonia patients. The CRP levels were higher, but the microcirculatory profile in the sublingual mucosa was still better in COVID-19 patients on day 2 of the ICU stay as compared to those treated for bacterial pneumonia. Our results might point toward the role of EG damage in the pathogenesis in organ dysfunction in COVID-19 patients.
Acknowledgments
Declaration
Ethics approval and consent to participate
The study was approved by the local ethical committee of University Hospital Hradec Kralove (approval number 72195/2020-684800). Consent to participate was signed with patients’ relatives and physicians not related to the study.
Consent for publication
All patients or their relatives gave consent for publication.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare that they have no competing interests.
Funding
Supported by MZ ČZ –RVO (UHHK, 00179906), PROGRES Q40/N8514002, and PROGRES Q40/06, all rights reserved.
Authors’ contributions
DA –designed the clinical study, wrote a grant proposal, collected the microcirculation and clinical data, did a statistical analysis of the results, wrote most of the manuscript.
AdT –conducted the laboratory investigation.
AT –conducted the laboratory investigation.
RH –conducted the laboratory investigation.
SCh –analyzed the microcirculation recordings.
MM –analyzed the microcirculation recordings.
RS –helped to write the manuscript.
ChL –helped to write the manuscript, did the language check.
RbH –helped to write the manuscript.
MaM –helped to write the manuscript.
VC –helped to design the clinical study, helped to write the manuscript.
All authors read and agree with the final manuscript. The study was not published elsewhere in whole or in a part.
Trial registration
Clinicaltrials.gov, reg. no. NCT04644302, registered 25th November 2020.
References
[1] | Wunsch H . Mechanical Ventilation in COVID-19: Interpreting the Current Epidemiology. Am J Respir Crit Care Med. (2020) ;202: (1):1–4. |
[2] | Stahl K , Gronski PA , Kiyan Y , Seeliger B , Bertram A , Pape T , et al. Injury to the Endothelial Glycocalyx in Critically Ill Patients with COVID-19. Am J Respir Crit Care Med. (2020) ;2028: ):1178–81. |
[3] | Okada H , Yoshida S , Hara A , Ogura S , Tomita H . Vascular endothelial injury exacerbates coronavirus disease The role of endothelial glycocalyx protection. Microcirculation. (2021) ;28: (3):e12654. |
[4] | Yamaoka-Tojo M . Endothelial glycocalyx damage as a systemic inflammatory microvascular endotheliopathy in COVID-19. Biomed J Elsevier B.V. (2020) ;43: (5):399–413. |
[5] | Nicosia RF , Ligresti G , Caporarello N , Akilesh S , Ribatti D . COVID-19 Vasculopathy: Mounting Evidence for an Indirect Mechanism of Endothelial Injury. Am J Pathol Elsevier Inc. (2021) ;191: (8):1374–84. |
[6] | Maiuolo J , Mollace R , Gliozzi M , Musolino V , Carresi C , Paone S , et al. The Contribution of Endothelial Dysfunction in Systemic Injury Subsequent to SARS-Cov-2 Infection. Int J Mol Sci. (2020) ;21: (23):9309. |
[7] | Varga Z , Flammer AJ , Steiger P , Haberecker M , Andermatt R , Zinkernagel AS , et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. (2020) ;395: (10234):1417–18 |
[8] | Jung F , Krüger-Genge A , Franke RP , Hufert F , Küpper JH . COVID-19 and the endothelium. Clin Hemorheol Microcirc. (2020) ;75: (1):7–11. |
[9] | Cerny V , Astapenko D , Brettner F , Benes J , Hyspler R , Lehmann C , et al. Targeting the endothelial glycocalyx in acute critical illness as a challenge for clinical and laboratory medicine. Crit Rev Clin Lab Sci. (2017) ;54: (5):343–57. |
[10] | Rovas A , Osiaevi I , Buscher K , Sackarnd J , Tepasse PR , Fobker M , et al. Microvascular dysfunction in COVID-19: the MYSTIC study. Angiogenesis. (2021) ;24: (1):145–57. |
[11] | Gómez-Mesa JE , Galindo-Coral S , Montes MC , Muñoz Martin AJ . Thrombosis and Coagulopathy in COVID-19. Curr Probl Cardiol. (2021) ;46: (3):100742. |
[12] | Ackermann M , Verleden SE , Kuehnel M , Haverich A , Welte T , Laenger F , et al. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N Engl J Med. (2020) ;383: (2):120–8. |
[13] | Teuwen LA , Geldhof V , Pasut A , Carmeliet P . COVID-19: the vasculature unleashed. Nat Rev Immunol. (2020) ;20: (7):389–91. |
[14] | Kory P , Meduri GU , Iglesias J , Varon J , Marik PE . Clinical and Scientific Rationale for the “MATH+” Hospital Treatment Protocol for COVID-19. J Intensive Care Med. (2021) ;36: (2):135–56. |
[15] | Lipowsky HH , Lescanic A . Inhibition of inflammation induced shedding of the endothelial glycocalyx with low molecular weight heparin. Microvasc Res. (2017) ;112: :72–8. |
[16] | Neil M , Ampel M . Therapeutic Doses of Anticoagulation Did Not Benefit Patients with COVID-19 and Elevated. NEJM J Watch Journal Watch 2021. |
[17] | Goedhart PT , Khalilzada M , Bezemer R , Merza J , Ince C . Sidestream Dark Field (SDF) imaging: a novel stroboscopic LED ring-based imaging modality for clinical assessment of the microcirculation. Opt Express. (2007) ;15: (23):15101–14. |
[18] | Sekino M , Funaoka H , Sato S , Egashira T , Inoue H , Yano R , et al. Association between endotoxemia and enterocyte injury and clinical course in patients with gram-positive septic shock: A posthoc analysis of a prospective observational study. Medicine (Baltimore). (2019) ;98: (28):e16452. |
[19] | Verdant CL , De Backer D , Bruhn A , Clausi CM , Su F , Wang Z , et al. Evaluation of sublingual and gut mucosal microcirculation in sepsis: a quantitative analysis. Crit Care Med. (2009) ;37: (11):2875–81. |
[20] | Martini R . The compelling arguments for the need of microvascular investigation in COVID-19 critical patients. Clin Hemorheol Microcirc. (2020) ;75: (1):27–34. |
[21] | Metlay JP , Waterer GW , Long AC , Anzueto A , Brozek J , Crothers K , et al. Diagnosis and Treatment of Adults with Community-acquired Pneumonia. An Official Clinical Practice Guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med. (2019) ;200: (7):e45–e67. |
[22] | Singer M , Deutschman CS , Seymour CW , Shankar-Hari M , Annane D , Bauer M , et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. (2016) ;315: (8):801–10. |
[23] | Cerny V , Turek Z , Parizkova R . Orthogonal polarization spectral imaging. Physiol Res. (2007) ;56: (2):141–47. |
[24] | Ince C , Boerma EC , Cecconi M , De Backer D , Shapiro NI , Duranteau J , et al. Second consensus on the assessment of sublingual microcirculation in critically ill patients: results from a task force of the European Society of Intensive Care Medicine. Intensive Care Med Springer Berlin Heidelberg. (2018) ;1–19. |
[25] | Goshua G , Pine AB , Meizlish ML , Chang CH , Zhang H , Bahel P , et al. Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study. Lancet Haematol Lancet Haematol. (2020) ;7: (8):e575–82. |
[26] | Cerny V , Astapenko D , Burkovskiy I , Hyspler R , Ticha A , Trevors MA , et al. Glycocalyx in vivo measurement. Clin Hemorheol Microcirc. (2017) ;67: (3-4):1–5. |
[27] | Hahn RG , Hasselgren E , Björne H , Zdolsek M , Zdolsek J . Biomarkers of endothelial injury in plasma are dependent on kidney function. Clin Hemorheol Microcirc. 2019;1-8. |
[28] | Rovas A , Osiaevi I , Buscher K , Sackarnd J , Tepasse PR , Fobker M , et al. Microvascular dysfunction in COVID-19: the MYSTIC study. Angiogenesis. (2021) ;24: (1):145–57. |
[29] | Rovas A , Seidel LM , Vink H , Pohlkötter T , Pavenstädt H , Ertmer C , et al. Association of sublingual microcirculation parameters and endothelial glycocalyx dimensions in resuscitated sepsis. Crit Care. (2019) ;23: (1):260. |
[30] | Lambadiari V , Mitrakou A , Kountouri A , Thymis J , Katogiannis K , Korakas E , et al. Association of COVID-19 with impaired endothelial glycocalyx, vascular function and myocardial deformation 4 months after infection. Eur J Heart Fail. (2021) ;23: (11):1916–26. |
[31] | Astapenko D , Ticha A , Tomasova A , Hyspler R , Zadak Z , Lehmann C , et al. Evaluation of endothelial glycocalyx in healthy volunteers - An observational study. Clin Hemorheol Microcirc. (2020) ;75: (3):257–65. |
[32] | Edul VS , Enrico C , Laviolle B , Vazquez AR , Ince C , Dubin A . Quantitative assessment of the microcirculation in healthy volunteers and in patients with septic shock. Crit Care Med. (2012) ;40: (5):1443–8. |
[33] | Iba T , Connors JM , Levy JH . The coagulopathy, endotheliopathy, and vasculitis of COVID-19. Inflamm Res. (2020) ;69: (12):1181–9. |
[34] | Potje SR , Costa TJ , Fraga-Silva TFC , Martins RB , Benatti MN , Almado CEL , et al. Heparin prevents in vitro glycocalyx shedding induced by plasma from COVID-19 patients. Life Sci. (2021) ;276: :119376. |
[35] | Cuker A , Tseng EK , Nieuwlaat R , Angchaisuksiri P , Blair C , Dane K , et al. American Society of Hematology guidelines on the use of anticoagulation for thromboprophylaxis in patients with COVID-19. Blood Adv The American Society of Hematology. (2021) ;5: (3):872–88. |
[36] | Kim WY , Kweon OJ , Cha MJ , Baek MS , Choi SH . Dexamethasone may improve severe COVID-19 via ameliorating endothelial injury and inflammation: A preliminary pilot study. PLoS One. (2021) ;16: (7):e0254167. |
[37] | Ferrara F , Vitiello A . Efficacy of synthetic glucocorticoids in COVID-19 endothelites. Naunyn Schmiedebergs Arch Pharmacol Springer Science and Business Media Deutschland GmbH. (2021) ;394: (5):1003–07. |
[38] | van Paassen J , Vos JS , Hoekstra EM , Neumann KMI , Boot PC , Arbous SM . Corticosteroid use in COVID-19 patients: a systematic review andmeta-analysis on clinical outcomes. Crit Care. (2020) ;24: (1):696. |
[39] | Bhimraj A , Morgan RL , Shumaker AH , Lavergne V , Baden L , Cheng VC , et al. Infectious Diseases Society of America Guidelines on the Treatment and Management of Patients with COVID-19. Clin Infect Dis. 2020:ciaa478. |
[40] | Hu B , Huang S , Yin L . The cytokine storm and COVID-19. J Med Virol. (2021) ;93: (1):250–6. |
[41] | Olbei M , Hautefort I , Modos D , Treveil A , Poletti M , Gul L , et al. SARS-CoV-2 Causes a Different Cytokine Response Compared to Other Cytokine Storm-Causing Respiratory Viruses in Severely Ill Patients. Front Immunol. (2021) ;12: :629193. |
[42] | England JT , Abdulla A , Biggs CM , Lee AYY , Hay KA , Hoiland RL , et al. Weathering the COVID-19 storm: Lessons from hematologic cytokine syndromes. Blood Rev. (2021) ;45: :100707. |
[43] | Fernández-Sarmiento J , Flórez S , Alarcón-Forero LC , Salazar-Peláez LM , Garcia-Casallas J , Mulett H , et al. CaseReport: Endothelial Glycocalyx Damage in Critically ill PatientsWith SARS-CoV-2-Related Multisystem Inflammatory Syndrome (MIS-C). Front Pediatr. (2021) ;9: :726949. |
[44] | Borgel D , Chocron R , Grimaud M , Philippe A , Chareyre J , Brakta C , et al. Endothelial Dysfunction as a Component of Severe Acute Respiratory Syndrome Coronavirus 2-Related Multisystem Inflammatory Syndrome in Children With Shock. Crit Care Med. (2021) ;49: (11):e1151–e1156. |
[45] | Bonaventura A , Vecchié A , Dagna L , Martinod K , Dixon DL , VanTassell BW , et al. Endothelial dysfunction and immunothrombosis askey pathogenic mechanisms in COVID-19. Nat Rev Immunol. (2021) ;21: (5):319–29. |
[46] | Oudkerk M , Büller HR , Kuijpers D , van Es N , Oudkerk SF , McLoud T , et al. Diagnosis, Prevention, and Treatment of Thromboembolic Complications in COVID- Report of the National Institute for Public Health of the Netherlands. Radiology. (2020) ;297: (1):E216–E222. |
[47] | Jung EM , Stroszczynski C , Jung F . Contrast enhanced ultrasonography (CEUS) to detect abdominal microcirculatory disorders in severe cases of COVID-19 infection: First experience. Clin Hemorheol Microcirc. (2020) ;74: (4):353–61. |
[48] | Jung EM , Stroszczynski C , Jung F . Contrast enhanced ultrasound (CEUS) to assess pleural pulmonal changes in severe COVID-19 infection: First results. Clin Hemorheol Microcirc. (2020) ;75: (1):19–26. |
[49] | Goonewardena SN , Grushko OG , Wells J , Herty L , Rosenson RS , Haus JM , et al. Immune-Mediated Glycocalyx Remodeling in Hospitalized COVID-19 Patients. Cardiovasc Drugs Ther. (2021) ;18: :1–7. |
[50] | Bos LDJ , Sjoding M , Sinha P , Bhavani SV , Lyons PG , Bewley AF , et al. PRoVENT-COVID collaborative grouLongitudinal respiratory subphenotypes in patients with COVID-19-related acute respiratory distress syndrome: results from three observational cohorts. Lancet Respir Med. (2021) ;9: (12):1377–86. Erratum in: Lancet Respir Med. 2021;9(12):e114. |




