Convalescent plasma therapy: A promising solution for SARS-CoV-2 outbreak
Abstract
Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2) has caused more than 18 million confirmed cases and 700000 deaths worldwide after the outbreak in November 2019 (COVID-19). It has been considered as most pathogenic infection under this category. Till date none of the therapeutics or prophylaxis measures have been claimed by any researcher which could cure the patient suffering from the SARS-CoV-2 infection. There is an urgent need for any alternative and effective way of disease management for COVID-19. However, convalescent plasma therapy (CPT) has gain attention of researchers with significant improvement of patient survival rate. Interestingly, there are numerous examples where CPT has proven its potential upon post-exposure prophylaxis and/or treatment in various diseases including COVID-19. This review summarizes the essential elements related to CPT, its past clinical evidences and application of CPT for the management of SARS-CoV-2 infection during COVID-19. We used the published literatures from PubMed, EMBASE and Medline databases until 31st May 2020.
1Introduction
The outbreak of novel coronavirus SARS-CoV-2 (nCoV) infection was associated with acute respiratory infection started in December 2019 from Wuhan city, Hubei province of China. Till 15 June 2020 the total number of 50, 54, 222 SARS-CoV-2 infected patients have been confirmed including 3, 02, 597 death globally [1]. Similar to past coronavirus outbreaks of SARS and Middle East respiratory syndrome (MERS), the nCoV also caused respiratory infection [2, 3]. Later it started crossing the boundaries of China and became a pandemic of the year 2019, named as COVID-19. Currently the COVID-19 has drawn a well deserved attention and became a global threat due to its spread in more than 200 countries, as reported by Worldometer (a private statistical data company). Zhou et al has discussed genomic comparison of SARS-CoV, MERS-CoV and novel SARS-CoV-2 with bat coronaviruses [4]. The study suggested that SARS-CoV-2 has long genome (∼30 kb) and belong to β coronavirus type. However, the genome sequences were found 96%similar to bat coronavirus RaTG13 and 79%similar to SARS-CoV [4]. SARS-CoV-2 uses its S protein to interact with host receptor called “human angiotensin-converting enzyme 2 (hACE2)”, majorly present on lung epithelial cells. Binding affinity of the receptor-binding domain (RBD) of SARS-CoV-2, S protein is nearly 20-fold higher than that of SARS-CoV, causing higher infectivity and transmissibility of SARS-CoV-2 compared to SARS-CoV [5].
2Convalescent plasma therapy (CPT)
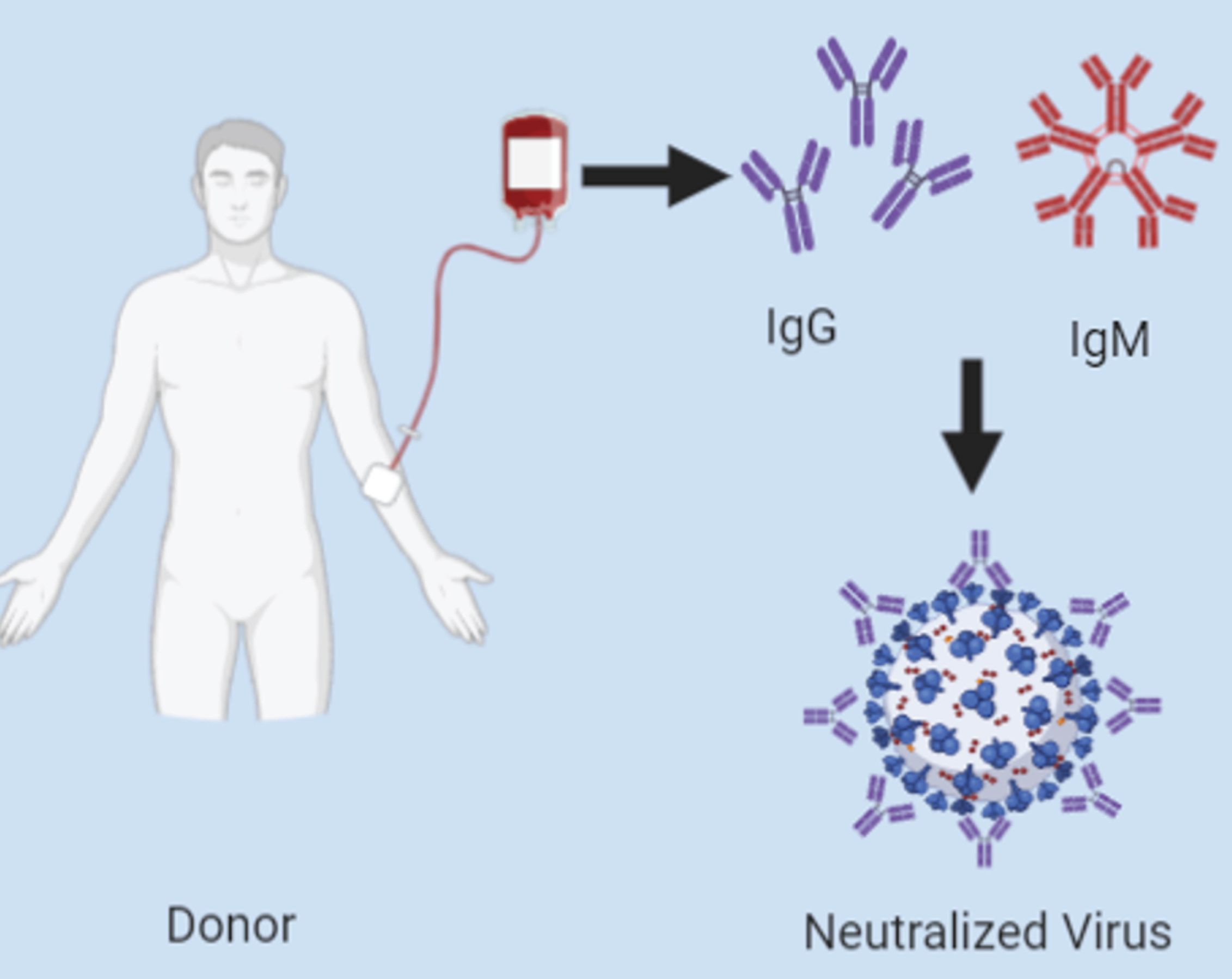
Currently, the treatment options for nCoV through antiviral drugs are limited and the vaccines are still under clinical trials [6]. Thus Convalescent plasma therapy (CPT) is now considered as the best treatment option for critical patients since it has been successfully used in other viral disease outbreaks earlier [7, 8]. The possible mechanism of the CPT and its outcome in the pathogenesis of SARS-CoV-2 includes neutralizing antibodies (Nabs) response or direct neutralization of the virus (Fig. 1) Nabs of the transfused plasma can suppresses the viruses or helps in virus clearance [9]. In case of COVID-19, the peak of viraemia in a patient is prominent in the first week of infection and the primary immune response of the host generally develops from day 10 to day 14 from the infection [10]. Thus administration of convalescent plasma would be more effective if administered at the early stage of the disease but currently it is not possible due to the treatment guideline provided by FDA [11]. However, the efficacy of CPT depends on the concentration of the NAbs in plasma from recovered donors. It was discovered that Nabs plays a role by binding to the spike proteins of the coronavirus, and hence inhibiting viral entry and replication [12]. Moreover, CP also helps in control of overactive immune response by providing passive immunomodulatory effect which helps the recipient to control the excessive inflammatory cascade induced by COVID-19 [13].
Fig. 1
Schematic representation of antiviral activity of Nabs of convalescent plasma by neutralizing spike protein of SARS-CoV-2.
3Significance of CPT in past pandemics
The therapy has been used since it was first adopted in 1918-1920 for Spanish flu and proven its importance in many viral pandemics like Ebola, MERS as well as other SARS associated outbreaks [40–43]. However it was first tested in diphtheria, a bacterial disease, in late 1890 for which Emil Behring was awarded Novel prize in 1901. The COID-19 pandemic necessitates the step towards the CPT until there is no proper medicine or vaccine available sufficiently. The evidences for past success of the plasma therapy are listed in Table 1.
Table 1
Use of CPT in past pandemics
| Disease | Infections | References |
| Coronavirus infection | ||
| Severe acute respiratory syndrome | Corona virus | [17] |
| (SARS) outbreak (2002) | [18] | |
| Middle East respiratory | Corona virus | [8] |
| syndrome (MERS) (2015) | [19] | |
| Influenza virus infection | ||
| H5N1 avian flu | Influenza A (H5N1) virus | [20] |
| H1N1 influenza A (2009) | Influenza A (H1N1) virus | [21] |
| Spanish flu (1918) | H1N1 influenza virus | [22] |
| Other viral infections | ||
| Argentine haemorrhagic fever (1958) | Junin virus | [23] |
| Bolivian hemorrhagic fever (1959) | Machupo virus | [24] |
| Lassa fever (1984) | Lassa virus | [25] |
| West Nile encephalitis (2002) | West Nile virus | [26] |
| Ebola virus disease (2015) | Ebola virus | [27, 28] |
| Other infections | ||
| Pertussis | Bordetella pertussis | [29] |
| Diptheria | Corynebacterium diphtheria | [30] |
4Regulatory aspects of CPT
Recently FDA expedites the process of clinical trial approval through e-filling. FDA released a set of guidelines for CPT which majorly covers the route to start the investigation in US, fixed the eligibility criteria of patient recruitment, collection of plasma from the eligible donors with proper labelling and maintenance of record [36]. According to these guidelines, investigator should submit application under the traditional IND regulatory pathway.
In continuation, the ICMR also released their guidelines and approved 34 clinical trials recently for CPT [31]. Since, China was the first country to report COVID-19 they started clinical trials for every possible treatment options including CPT. Currently, most of the clinical studies for any COVID-19 research are from China or the prior affected countries like Korea and Italy.
5Application of convalescent plasma therapy (CPT) in COVID-19
The CPT is based on the passive immunity (or antibody) developed among the recovered patient. The therapy is among one of the major option during COVID-19 pandemic [32]. However, the significance of the therapy has not been clinically proven. Since there are no vaccines or antibodies available for the treatmentof SARS-CoV-2 patients’ CPT has proven its efficacy at significant level [39]. Recently CPT was adopted for the treatment of hepatitis E virus infection in an immunocompromised elderly patient [44]. Although some studies has been published before FDA guidelines for the convalescent plasma for investigators and healthcare professionals [44].
6Clinical practices
Currently, the clinical evidences for the solicitation of CPT belong to China and Korea. In addition, a letter to the editor was submitted by an Italian team Franchini et al to suggest how CP should be collected from a recovered patients [45]. Till date there are seven reports on CPT where the studies were done with a very small sample size and critical patients (Table 2). Since the CPT was adopted after realizing that the patient’s condition did not improve with the all available treatment options. Among the describedreports,one study showed 5 patients died out of 6 after receiving the CPT but the result was similar to other successful studiesin terms of clearing theviral loads in all 6 patients [36]. Herethe test subjects in the study received the CP after ∼21.5 days of virus detection and critically damaged respiratory system. Overall the study emphases that CP must be provided within 14 days of infection [37]. In all the studies, before the CP transfusion ABO compatibility and confirmatory testsfor other viral infections such as HIV, Syphilis, HBV and HCV were precarious [36–39]. However, single 200–250 ml dose were effectively cleared the viral loadfrom patients but in some cases the number of doses were increased up to 8 in order to cure the critical patients [37, 40, 41]. In another study,the CPT was found effective in 5 critical patients with two doses of∼200 to 250 ml and the other life support with antiviral drugs [42]. The improved medical conditions of the recipients were observed through the increased leucocytes (0.65×109→ 0.75×109 per L), decrease in C-protein level (55.98 →18.13 mg/L), increased oxyhemoglobin saturation (93%→ 96%) apart from the fever, cough and other common symptoms [38]. In addition, the chest CT scan was the major concern which was associated with the patients’respiratory issues such as shortness of breath, chest pain and abnormal ground-glass opacity (GGO)of the nCoV-2 infected person [37, 40, 41]. Interestingly, the improved CT report was observed in most of the cases after CP transfusion except the Zeng Q-L et al case study [38, 43, 44].
Table 2
Case study with CPT
| Sl.No. | Location of study | No. of patients’ (n) | Dose of CP (ml) | Additional drug &support | Effect of CPT | Ref. |
| 1. | Wuhan, China | 10 | 200 | Antiviral therapy and other life support | •No death | [38] |
| •7 improved | ||||||
| •3 discharged | ||||||
| 2. | Nanjing, China | 1 | 200 | Invasive mechanical ventilation | No ventilation needed after 11 days of transfusion (improved) | [45] |
| 3. | Wuhan, China | 6 | 200(1 to 3 time) | No information | All patients’ showed radiological and symptomatic improvement | [37] |
| 4. | Zhengzhou, China | 6 | 300 (3 times) | ICU + other | •Virus cleared in all patients | [44] |
| •5 died & 1 Survived | ||||||
| 5. | Guangdong, China | 4 | 200 to 400 (1 to 8 doses) | ICU + other | All 4 recovered | [40] |
| 6. | Seoul, Korea | 2 | 250 (2doses) | Hydroxychloroquine, lopinavir/ritonavir &other life support | Both patients recovered | [41] |
| 7. | Shenzhen, China | 5 | 400 mL(Total) | Mechanical ventilation + antiviral agents | All five recovered | [39] |
Recently, in a study the characterization and analysis of the CP donor was done where the team emphasize on donor rather than recipients [45]. However most of the studies were checked for donors IgM and IgG titer, confirmation of negative result of severe viral disease including nCoV-2, HIV, HBV etc. as well as respiratory issues [38, 41, 44]. In the study of Zhang L et al included C-reactive protein, IL-6, CD3/CD4, creatine kinase andlactate dehydrogenase level apart from the above. It helped the CP recipient to be provided additional care if accepting abnormal plasma [45].
7Conclusion
Convalescent plasma therapy has been useful in treatment of many infectious diseases, including viral infections. However, very few clinical evidences are available so far related to its application in SARS-CoV-2 treatment but all reports are in support that the therapy can be effectively used. In the situation where there are no available drugs and vaccines, convalescent plasma therapy have shown potent application in the treatment of -SARS-CoV-2 infections. For more evidence and data, large multi-centric clinical study should be performed.
Conflict of interest
The authors have no conflict of interest to report.
References
[1] | WHO Situation Report. WHO website (https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200428-sitrep-99-covid-19.pdf?sfvrsn=119fc381_2). |
[2] | Zaki AM , van Boheemen S , Bestebroer TM , Osterhaus ADME , Fouchier RAM . Isolation of a Novel Coronavirus from a Man with Pneumonia in Saudi Arabia. N Engl J Med. (2012) ;367: (19):1814–20. |
[3] | Chan-Yeung M , Xu R-H . SARS: epidemiology. Respirology. (2003) ;8: (s1):S9–14. |
[4] | Zhou P , Yang X-L , Wang X-G , Hu B , Zhang L , Zhang W , et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. (2020) ;579: (7798):270–3. |
[5] | Wrapp D , Wang N , Corbett KS , Goldsmith JA , Hsieh C-L , Abiona O , et al. Cryo-EM structure of the 2019 -nCoV spike in the prefusion conformation. Science. (2020) ;367: (6483):1260–3. |
[6] | Zhu Y , Li J , Pang Z . Recent insights for the emerging COVID-19: drug discovery, therapeutic options and vaccine development. Asian J Pharm Sci [Internet]. 2020 Jul 4 [cited 2020 Aug 19]; Available from: http://www.sciencedirect.com/science/article/pii/S181808762030564X. |
[7] | Winkler AM , Koepsell SA . The use of convalescent plasma to treat emerging infectious diseases: focus on Ebola virus disease. Curr Opin Hematol. (2015) ;22: (6):521–6. |
[8] | Arabi YM , Hajeer AH , Luke T , Raviprakash K , Balkhy H , Johani S , et al. Feasibility of Using Convalescent Plasma Immunotherapy for MERS-CoV Infection, Saudi Arabia. Emerg Infect Dis. (2016) ;22: (9):1554–61. |
[9] | Casadevall A , Pirofski L . The convalescent sera option for containing COVID-19. J Clin Invest. (2020) ;130: (4):1545–8. |
[10] | Chowdhury MA , Hossain N , Kashem MA , Shahid MdA , Alam A . Immune response in COVID-19: A review. J Infect Public Health [Internet]. 2020 Jul 14 [cited 2020 Aug 19]; Available from: http://www.sciencedirect.com/science/article/pii/S1876034120305670. |
[11] | Research C for BE and. Recommendations for Investigational COVID-19 Convalescent Plasma. FDA [Internet]. 2020 Jan 5 [cited 2020 May 9];Available from: https://www.fda.gov/vaccines-blood-biologics/investigational-new-drug-ind-or-device-exemption-ide-process-cber/recommendations-investigational-covid-19-convalescent-plasma. |
[12] | Rojas M , Rodríguez Y , Monsalve DM , Acosta-Ampudia Y , Camacho B , Gallo JE , et al. Convalescent plasma in Covid-19 Possible mechanisms of action. Autoimmun Rev. (2020) ;19: (7):102554. |
[13] | Rizk JG , Kalantar-Zadeh K , Mehra MR , Lavie CJ , Rizk Y , Forthal DN . Pharmaco-Immunomodulatory Therapy in COVID-19. Drugs. (2020) :1–26. |
[14] | Luke TC , Kilbane EM , Jackson JL , Hoffman SL . Meta-analysis: convalescent blood products for Spanish influenza pneumonia: a future H5N1 treatment? Ann Intern Med. (2006) ;145: (8):599–609. |
[15] | Stockman LJ , Bellamy R , Garner P . SARS: systematic review of treatment effects. PLoS Med. (2006) ;3: (9):e343. |
[16] | Arabi YM , Hajeer AH , Luke T , Raviprakash K , Balkhy H , Johani S , et al. Feasibility of Using Convalescent Plasma Immunotherapy for MERS-CoV Infection, Saudi Arabia. Emerg Infect Dis. (2016) ;22: (9):1554–61. |
[17] | Wong VWS , Dai D , Wu AKL , Sung JJY . Treatment of severe acute respiratory syndrome with convalescent plasma. Hong Kong Med J Xianggang Yi Xue Za Zhi. (2003) ;9: (3):199–201. |
[18] | Cheng Y , Wong R , Soo YOY , Wong WS , Lee CK , Ng MHL , et al. Use of convalescent plasma therapy in SARS patients in Hong Kong. Eur J Clin Microbiol Infect Dis Off Publ Eur Soc Clin Microbiol. (2005) ;24: (1):44–6. |
[19] | Ko J-H , Seok H , Cho SY , Ha YE , Baek JY , Kim SH , et al. Challenges of convalescent plasma infusion therapy in Middle East respiratory coronavirus infection: a single centre experience. Antivir Ther. (2018) ;23: (7):617–22. |
[20] | Kong LK , Zhou BP . Successful treatment of avian influenza with convalescent plasma. Hong Kong Med J Xianggang Yi Xue Za Zhi. (2006) ;12: (6):489. |
[21] | Hung IF , To KK , Lee C-K , Lee K-L , Chan K , Yan W-W , et al. Convalescent Plasma Treatment Reduced Mortality in Patients With Severe Pandemic Influenza A (H1N1) 2009 Virus Infection. Clin Infect Dis. (2011) ;52: (4):447–56. |
[22] | Marano G , Vaglio S , Pupella S , Facco G , Catalano L , Liumbruno GM , et al. Convalescent plasma: new evidence for an old therapeutic tool? Blood Transfus. (2016) ;14: (2):152–7. |
[23] | Ruggiero HA , Pérez Isquierdo F , Milani HA , Barri A , Val A , Maglio F , et al. [Treatment of Argentine hemorrhagic fever with convalescent’s plasma. 4433 cases]. Presse Medicale Paris Fr 1983. (1986) ;15: (45):2239–42. |
[24] | Stinebaugh BJ , Schloeder FX , Johnson KM , Mackenzie RB , Entwisle G , De Alba E . Bolivian hemorrhagic fever. A report of four cases. Am J Med. (1966) ;40: (2):217–30. |
[25] | Jahrling PB , Niklasson BS , McCormick JB . Early diagnosis of human Lassa fever by ELISA detection of antigen and antibody. Lancet Lond Engl. (1985) ;1: (8423):250–2. |
[26] | Planitzer CB , Modrof J , Kreil TR . West Nile Virus Neutralization by US Plasma-Derived Immunoglobulin Products. J Infect Dis. (2007) ;196: (3):435–40. |
[27] | Gutfraind A , Meyers LA . Evaluating Large-scale Blood Transfusion Therapy for the Current Ebola Epidemic in Liberia. J Infect Dis. (2015) ;211: (8):1262–7. |
[28] | Kraft CS , Hewlett AL , Koepsell S , Winkler AM , Kratochvil CJ , Larson L , et al. The Use of TKM-100802 and Convalescent Plasma in 2 Patients With Ebola Virus Disease in the United States. Clin Infect Dis Off Publ Infect Dis Soc Am. (2015) ;61: (4):496–502. |
[29] | Saida K , Ogura M , Kano Y , Ishimori S , Yoshikawa T , Nagata H , et al. Treatment of hemolytic uremic syndrome related to Bordetella pertussis infection -is plasma exchange or eculizumab use necessary? BMC Nephrol. (2018) 17;19: (1):365. |
[30] | Hall PD , Sinha D , Frankel AE . Fresh frozen plasma and platelet concentrates may increase plasma anti-diphtheria toxin IgG concentrations: Implications for diphtheria fusion protein therapy. Cancer Immunol Immunother CII. (2006) ;55: (8):928–32. |
[31] | COVID-19 | Indian Council of Medical Research | Government of India [Internet]. [cited 2020 May 14]. Available from: https://main.icmr.nic.in/content/covid-19. |
[32] | Bloch EM , Shoham S , Casadevall A , Sachais BS , Shaz B , Winters JL , et al. Deployment of convalescent plasma for the prevention and treatment of COVID-19. J Clin Invest [Internet]. 2020 Apr 7 [cited 2020 May 9]; Available from: https://www.jci.org/articles/view/138745. |
[33] | Brown BL , McCullough J . Treatment for emerging viruses: Convalescent plasma and COVID-19. Transfus Apher Sci Off J World Apher Assoc Off J Eur Soc Haemapheresis. (2020) ;102790. |
[34] | Ankcorn M , Gallacher J , Ijaz S , Taha Y , Harvala H , Maclennan S , et al. Convalescent plasma therapy for persistent hepatitis E virus infection. J Hepatol. (2019) ;71: (2):434–8. |
[35] | Franchini M , Marano G , Velati C , Pati I , Pupella S , Liumbruno GM . Operational protocol for donation of anti-COVID-19 convalescent plasma in Italy. Vox Sang. 2020; |
[36] | Zeng Q-L , Yu Z-J , Gou J-J , Li G-M , Ma S-H , Zhang G-F , et al. Effect of Convalescent Plasma Therapy on Viral Shedding and Survival in Patients With Coronavirus Disease 2019. J Infect Dis. (2020) ;222: (1):38–43. |
[37] | Ye M , Fu D , Ren Y , Wang F , Wang D , Zhang F , et al. Treatment with convalescent plasma for COVID-19 patients in Wuhan, China. J Med Virol. 2020 Apr 15; |
[38] | Duan K , Liu B , Li C , Zhang H , Yu T , Qu J , et al. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc Natl Acad Sci U S A. (2020) ;117: (17):9490–6. |
[39] | Shen C , Wang Z , Zhao F , Yang Y , Li J , Yuan J , et al. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA. 2020 Mar 27; |
[40] | Zhang B , Liu S , Tan T , Huang W , Dong Y , Chen L , et al. Treatment With Convalescent Plasma for Critically Ill Patients With SARS-CoV-2 Infection. Chest [Internet]. [cited 2020 May 16]; Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7195335/. |
[41] | Ahn JY , Sohn Y , Lee SH , Cho Y , Hyun JH , Baek YJ , et al. Use of Convalescent Plasma Therapy in Two COVID-19 Patients with Acute Respiratory Distress Syndrome in Korea. J Korean Med Sci. (2020) ;35: (14):e149. |
[42] | Amanat F , Stadlbauer D , Strohmeier S , Nguyen THO , Chromikova V , McMahon M , et al. A serological assay to detect SARS-CoV-2 seroconversion in humans. Nat Med. (2020) ;1–4. |
[43] | Zhang L , Pang R , Xue X , Bao J , Ye S , Dai Y , et al. Anti-SARS-CoV-2 virus antibody levels in convalescent plasma of six donors who have recovered from COVID-19. Aging. (2020) ;12: (8):6536–42. |
[44] | Zeng Q-L , Yu Z-J , Gou J-J , Li G-M , Ma S-H , Zhang G-F , et al. Effect of Convalescent Plasma Therapy on Viral Shedding and Survival in COVID-19 Patients. J Infect Dis. 2020 Apr 29; |
[45] | Zhang L , Pang R , Xue X , Bao J , Ye S , Dai Y , et al. Anti-SARS-CoV-2 virus antibody levels in convalescent plasma of six donors who have recovered from COVID-19. Aging. (2020) ;12: (8):6536–42. |




