Enzyme controlled release of celecoxib from inulin based nanomicelles
Abstract
Celecoxib (CLX) delivery and its enzyme-sensitive release from Inulin-d-alfa-tocopherol succinate (INVITE) nanomicelles are the main goals of this paper. CLX is a highly hydrophobic drug belonging to BCS class II (low solubility, high permeability). For this reason its formulation is problematic and its biopharmaceutical performances strongly depend from the applied delivery system. In the last years, INVITE nanomicelles has been shown their potential in the delivery of highly hydrophobic drugs such as curcumin and for this reason have been chosen as a good candidate for CLX delivery. Furthermore, due to the presence of ester bonds between INU and VITE it has been supposed that the drug release could show enzyme-sensitive (esterase) behaviors. Thus CLX was loaded in INVITE nanomicelles, the loading was quantified and the physical stability was evaluated up to 90 days, finally, drug release studies in the presence or in the absence of the specific enzyme esterase were performed.
1Introduction
The physical-chemical properties of a drug strictly influence its biopharmaceutical performance. Among these, the aqueous solubility is a critical issue that often affects the bioavailability of oral or parenteral administered drugs. In fact, numerous orally administered pharmaceutical agents present various drawbacks such as low solubility, poor absorption and/or poor stability in the intestinal tract; furthermore for the parenteral administration, hydrophobic drugs often show a short-half life and lead to formulation issues despite their good permeation ability.
To overcome the above reported issues, administration of high doses are often necessary to obtain a therapeutic effect, but involving several side effects as a consequence. It has been estimated that about 40% of the drugs presently in clinical use and up to 75% of those currently under development could be considered poorly water-soluble drugs [1] as occurs for those belonging to class II of the Biopharmaceutic Classification System (BCS). One example of drugs belonging to the BCS class II (low solubility, high permeability) is CLX (4-[5-(4-methylphenyl)-3-(trifluoromethylpyrazol-1-yl]-benzenesulfonamide) (CLX).
CLX is a nonsteroidal anti-inflammatory drug (NSAID), approved by the U.S. Food and Drug Administration (FDA)and the European Medicines Agency (EMA) for the treatment of rheumatoid arthritis and osteoarthritis [2, 3] thank to its direct effects on cartilage, bone and synoviocytes [4]. It is an NSAID that inhibits cyclooxygenase (COX), the enzyme responsible for conversion of arachidonic acid to prostaglandins. In particular, CLX belong to the class of Coxib, the non–steroidal anti-inflammatory drugs that, unlike most NSAIDs, are selective for inhibiting COX-2, the induced isoform of the two existing COX [5], which is almost absent in normal healthy tissues and cells but it increase during inflammation process, induced by various proinflammatory, catabolic and stress mediators such as cytokines and growth factors.
Furthermore, Coxibs leave the activity of COX 1 almost unaltered, hence causing less ulceration of the stomach and intestines with better gastrointestinal tolerability and reduction of other clinically important toxic effects [6, 7]. Although CLX is already used for musculoskeletal disorders and is under investigation for its distinct anticancer activities [8], it exhibits other major toxicities and there have been controversial reports regarding increased risk of heart attack and stroke.
From the physical-chemical and pharmacokinetic point of view, CLX is characterized by a low aqueous solubility (7 μg/mL) and high hydrophobicity (Log P = 3.683) [9] and it is rapidly eliminated from plasma [10]. These properties cause a variable absorption and limit the use of CLX that actually, to the best of our knowledge, is formulated as capsules under the brand name of Celebrex TM, CelebraTM or ArtilogTM and, often, it requires administration of high doses to produce a therapeutic effect, which may lead to severe toxicity [11].
Many examples of drug delivery systems for hydrophobic drugs are reported in the literature, including micro- and nano-particles [12–14], hydrogels, [15–22] polymer-drug conjugates [23–25], cyclodextrins [26–28].
In literature, there are few examples of delivery systems for CLX. For examples, Venkatesan et al. prepared composite nanoparticles from hydroxyapatite and chitosan containing CLX for the treatment of colon cancer. They stated that the incorporation of CLX into nanocomposite materials may circumvent toxicity and solubility problems of the drug and permit its targeted delivery to the tumor site by EPR effect [29].
In another study, Bachar et al. prepared â-casein micelles for oral delivery of hydrophobic drugs and they chosen CLX as a model. The obtained spherical micelles shown a mean particle diameter of 22 nm, a critical concentration association of 0.021–0.083 mM and resulted effective in incorporating CLX with 25% of encapsulation efficiency [30].
Recently, we reported the synthesis and characterization of new polymeric micelles, showing a great potential for drug delivery applications [31].
Our micelles were prepared by a simple procedure starting from two well-established “natural” molecules that exert beneficial effect on human health, inulin (INU) and vitamin E (VITE).As known INU is a hydrophilic natural polysaccharide from fructose that, in the last years, has been used for different pharmaceutical applications [32–34] and VITE is a hydrophobic vitamin, known as one of the most powerful anti-oxidant found in nature [35, 36] By combining the physical-chemical properties of INU and VITE succinate the amphiphilic polymer called INVITE was synthesized and recently proposed for the therapy of urinary tract infections [31] and already tested as carrier of the hydrophobic drug curcumin for its i.v. administration and for the formation of a carrier-in-carrier system in which curcumin-loaded-INVITE was further loaded in mesenchymal stromal cells for the dual drug delivery in the therapy central nervous system diseases [37]. Furthermore, it has been demonstrated that in the INVITE systems, the drug loading of curcumin is a result of Π-Π stacking interaction between the drug and the aromatic region of VITE so addressing their use for the preferential loading of aromatic groups bearing molecules as CLX is [38].
The purpose of the present study was to demonstrate the effectiveness of the “natural” polymeric INVITE nanomicelles as carrier of CLX, with particular emphasis on the ability of INVITE micelles to solubilize CLX in aqueous media and release it in a controlled manner and in particular as an enzyme-sensitive system due to the presence of ester bonds between INU and VITE. The obtained CLX loaded nanomicelles could be proposed for their oral or parenteral administration
2Experimental part
2.1Materials
All reagents were of analytical grade, unless otherwise stated. N,N-dimethylformamide (DMF), triethylamine (TEA), diciclohexylcarbodiimide (DCC), pyrene, D-á-tocopherol succinate, poly(acrylic acid), NaCl, KCl, Na2HPO4, KH2PO4, DMSO-d6, acetone, methanol, esterase from porcine liver, were purchased from Sigma-Aldrich (Milano, Italy). Inulin from dahlia tubers (INU, approx. 5000 Da),N-Hydroxysulfosuccinimide sodium salt (NHSS), Tween 20 were purchased from Fluka (Milano, Italy). DMSO was purchased from Carlo Erba Reagents (Milano, Italy). Dialysis tubes with a MWCO 3.500 Da (Spectra/Por ® 6) were purchased from Spectrum Labs.
2.2Apparatus
FT-IR spectra (KBr pellets) were recorded in the range 4000–400 cm–1 using a Perkin–Elmer 1600 IR Fourier Transform Spectrophotometer (Monza, Italy). The resolution was 1 cm–1.
UV-Vis analyses were performed using a Perkin–Elmer Spectrometer Lambda 25, Perkin-Elmer, (Monza, Italy).
1H-NMR spectra were recorded using a Varian Mercury 300 MHz instrument.
Centrifugations were performed with Eppendorf Centrifuge 5810 R (Milano, Italy) equipped with a temperature control.
The mean size and polydispersity index (PDI) of the INVITE micelles with or without CLX were measured using a ZetasizerNano ZS (Malvern Instruments Ltd., Worcestershire, UK).
High-performance liquid chromatography (HPLC) analysis of CLX was performed with a Waters (Waters Corp., MA) Model 600 pump equipped with a Waters 2996 photodiode array detector, a 20 μl loop injection auto sampler (Waters 717 plus), and processed by Empowertrademark Software.
2.3Synthesis of Inulin-d-alfa-tocopherol succinate (INVITE) conjugates
An Inulin-α-tocopherol succinate conjugate, called INVITE was synthesized following a published procedure [30].
Briefly, to a solution of VITE succinate (1 eq) in anhydrous DMF (4 mL) under nitrogen, DCC (2 eq) and NHSS (2 eq) were added. The amount of VITE was added according to the following molar ratio Y = 0.2, where Y indicates the molar ratio VITE/INU-repeating-units.
The activation reaction was carried out under stirring and nitrogen for 3 h at room temperature. Then, a solution of INU in anhydrous DMF and TEA (according to the following molar ratio: Z = 0.10 where Z indicates the molar ratio TEA/INU-repeating-units) was added. Then, the INU solution with TEA was added drop-wise to the VITE-NHSS ester solution and the reaction was carried out under nitrogen at 25°C for 12 h.
The INVITE derivative at 18% mol/mol in VITE groups was obtained and characterized by FT-IR and1H-NMR spectroscopy.
2.4Chromatographic conditions
HPLC analysis of CLX was carried out using a Waters (Waters Corp., MA) Model 600 pump equipped with a Waters 2996 photodiode array detector, an UV detector at a wavelength of 250 nm and a C18 Eclypse column (4.6×250 mm, 5 mm) from Agilent preceded by a C18 guard column. The column was maintained at room temperature. A mixture of methanol:H2O (75:25 v/v) was used as the mobile phase. The flow rate of 20 μl sample injection was maintained at 0.8 mL/min. A calibration curve was constructed using a CLX concentration range from 0.05 to 1 mg/mL (R2 > 0.999). The retention time of CLX was 9 min.
2.5CAC evaluation
The critical aggregation concentration (CAC) of the INVITE micelles was determined by fluorescence spectroscopy using pyrene as a hydrophobic fluorescent probe following a previous published procedure (Nota CP). Briefly, Aliquots (100 μL) of 2·10 −5M pyrene aqueous solutions were added to the 0.9 mL of aqueous INVITE solutions at different concentrations ranging from 1·10 −7 to 2.5 mg/mL. Samples were left to equilibrate overnight before fluorescence measurements were carried out. The fluorescence spectra were recorded at room temperature. Excitation was fixed at 334 nm and the emission spectra were recorded over the range 350–450 nm. Excitation and emission bandwidths were 5 nm.
2.6Preparation of CLX-loaded or empty INVITE micelles by direct dialysis from dimethylsulfoxide (DMSO)
Micelles from INVITE conjugate with or without CLX, here called INVITEMCLX and INVITEM respectively, were prepared at concentration above their critical association concentration (9.1·10 −3 mM).
100 mg of INVITE were dissolved in 10 mL of dimethylsulfoxide with or without CLX (10% w/w) and left under constant stirring for 2 h. The micellar dispersions were poured into a dialysis tubes (Spectra/Por ® 6) with a MWCO 3.500 Da and sealed. Dialyses were carried out for 4 days against distilled water which was exchanged every 3 h.
The colloidal suspensions inside the dialysis tubes appeared both transparent and uncolored for the whole dialysis time without evident precipitate formation.
At the end of the dialysis process, the drug loaded or not colloidal suspensions were filtered (0.45 μm RC membrane filter), lyophilized and recovered with a 86–90% w/w yields with respect to the starting polymer plus drug.
2.7Evaluation of drug loading and encapsulation efficiency on CLX-loaded INVITE micelles
For the evaluation of INVITE micelles drug loading, 4 mg of lyophilized CLX containing INVITE micelles (INVITE MCLX), have been dissolved in 1 mL of DMSO. The amount of drug loaded CLX was determined by HPLC analysis and the loaded amount of CLX was calculated by a calibration curve of CLX in DMSO. Each measurement was performed in triplicate. The drug loading (DL) and the encapsulation efficiency (EE) of CLX micelles were calculated according to the equations (1) and (2):
(1)
(2)
2.8Physico-chemical analysis and stability studies on INVITE micelles by size measurements
INVITE micelles empty or loaded with CLX were prepared by the above described direct dialysis method. Then, lyophilized micelles were dispersed in bi-distilled water and in PBS pH 7.4 at concentration of 1 mg/ml and the obtained clear solutions were filtered by 0.45 μm nylon filters and analyzed by ZetasizerNano ZS instrument. The measurements were performed in triplicate for each sample. The solutions were further kept at 25°C for pre-defined storage time up to 90 days to verify their physical stability, evaluated by measuring the size of the micelles after these periods of storage. The measurements were carried out in triplicate and reported as the mean ± SD.
2.9In vitro drug release studies of CLX from INVITE micelles
50 mg of CLX loaded INVITE micelles, prepared by direct dialysis, have been solubilized in 3 ml of double distilled water in the presence or not of esterase (40 U) and the solutions poured in a 3500 Da MWCO dialysis membrane. The dialysis tubes were incubated in 5 mL of PBS (pH 7.4) containing polysorbate 20 (3% wt) at 37 °C.
At scheduled time points and up to 7 days, the entire release medium was removed and replaced by pre-warmed fresh release media. The amount of released CLX has been valued by HPLC measurements and was calculated by a calibration curve of CLX in PBS (pH 7.4) containing Tween 20 (3% wt) in the concentration range 0.05–1 mg/mL (correlation coefficient r2 > 0.99). All results were the mean of three test runs and all data were expressed as the mean ± SD.
3Results and discussions
Improving the efficacy of drugs with problematic biopharmaceutical profiles means acting at a pharmaceutical-technological level with the aim to deliver them in a specific site and/or to release them only after a specific stimulus and/or to permit the use of molecules that are very powerful but with a low biopharmaceutical profile.
In this paper we prepared micellar systems able to bring in aqueous solution the drug CLX and to release it in an enzyme-controlled manner in simulated physiological conditions.
3.1Synthesis and characterization of INVITE bioconjugates
The synthesis of INVITE polymers has been performed by a previous published method [30] Briefly, α-tocopherol succinate was linked to INU by carbodiimide chemistry by activating the carboxyl group of VITE-succinate as a reactive ester. Then, the activated molecule reacted with the hydroxyl groups of INU, forming the INVITE bioconjugate (Scheme 1). The reaction was carried out in bulk to strongly reduce the use of solvent and energy also in consideration of possible scaling-up and industrial applications.
Derivatization degree (DD% mol/mol) was calculated by 1H-NMR as ratio between the peak integral at δ 0.8 ppm (VITE, m, 12 H) and the peak at 3.5–4.0 ppm (fructose ring, m, 7 H). DD values for the INVITE conjugate resulted 18 % mol/mol in agreement with the theoretic amount of VITE (Y values of reaction condition).
The Critical aggregation concentration (CAC) of INVITE amphiphilic polymer was determined by using pirene as a fluorescent probe as reported in previous published work (nota biomacromolecules). The INVITE/M showed a CAC value of 9.1·10 −3 mM that is in accordance, or slightly lower, with respect to INU-based amphiphilic polymers reported in literature such as those from Srinarong et al. that presented a CAC value of 0.009 % w/v for hydrophobized INU [39], or those from Licciardi et al. that found for their INU-based amphiphilic derivatives a CAC ranging 6·10 −2–5·10 −2 mg/mL depending on the synthesized derivative [40].
From the CAC values found for INVITE amphiphilic polymer it could be expected a good physical stability of the micelles as also previously confirmed [31, 38].
3.2Preparation and characterization of CLX-loaded or empty INVITE micelles
The preparation of INVITE micelles and their drug loading has been performed by direct dialysis. Following previous published data where loading of the hydrophobic drug curcumin by dialysis gave more reproducible results and higher DL% values with respect to O/W emulsion method, we chosen the same technique to prepare CLX loaded INVITE micelles (INVITE MCLX).
It should be noted that the colloidal dispersions obtained after reconstitution in water of the lyophilized micelles, resulted free from insolubilized material, so demonstrating that incorporation of CLX in INVITE micelles brought CLX in water solution (Fig. 1).
The comparison of FT-IR spectra of the CLX, INVITE conjugate and INVITE MCLX, reported in Fig. 3, confirmed the presence of CLX into INVITE micelles. In particular, INVITE MCLX spectrum shows the presence of band at 1643 cm −1 referred to the NH2 bending of primary sulfonamide of the CLX, that is absent in the spectrum of INVITE conjugate, (Fig. 2).
DL% of CLX into INVITE micelles was 5% w/w, higher that DL% of curcumin in the same micellar systems that reached maximum value of 3.5% w/w as previously reported [31, 38]. This is an interesting result that could be attributed to the specific structure of CLX which, as shown in Fig. 3, is provided with aromatic groups that could be able to interact via the formation of π - π stacking interactions with the benzopyran-6-yl moiety of VITE [38]. Probably the aromatic structure of CLX provided a better interaction of the drug with the VITE residues with respect to curcumin at least in terms of drug loading.
In order to verify the physical stability of the INVITE micelles with or without CLX, the drug loaded or not micelles were prepared by direct dialysis method and the size measurements were carried out on the micelle systems immediately after the preparation and also after pre-defined storage time at 25°C. The number-weighted size distribution for empty INVITE and loaded micelles has been measured by dynamic light scattering (DLS) and the results are reported in Fig. 4.
In particular, the stability study was performed by measuring the size of the micelles in ultrapure water and in PBS pH 7.4 at designated time points of 0 day, 7 days, 15 days, 60 days and 90 days. As it can be seen, in aqueous environment the INVITE micelles resulted still stable after 90 days, in fact no formation of aggregates was detected.
Moreover, Fig. 4 shows that the INVITE particle size was lower than 15 nm with no difference in size after CLX loading, confirming that incorporation of CLX does not influence the micelles structure. The slight increase in the micelles size in PBS pH 7.4 can be due to the interaction of buffer salts with the hydrophilic groups within the micelles structures, however the stabilities resulted comparable in water or PBS. Furthermore, all samples showed a narrow size distribution (PDI = 0.3–0.4).
3.3In-vitro CLX release studies
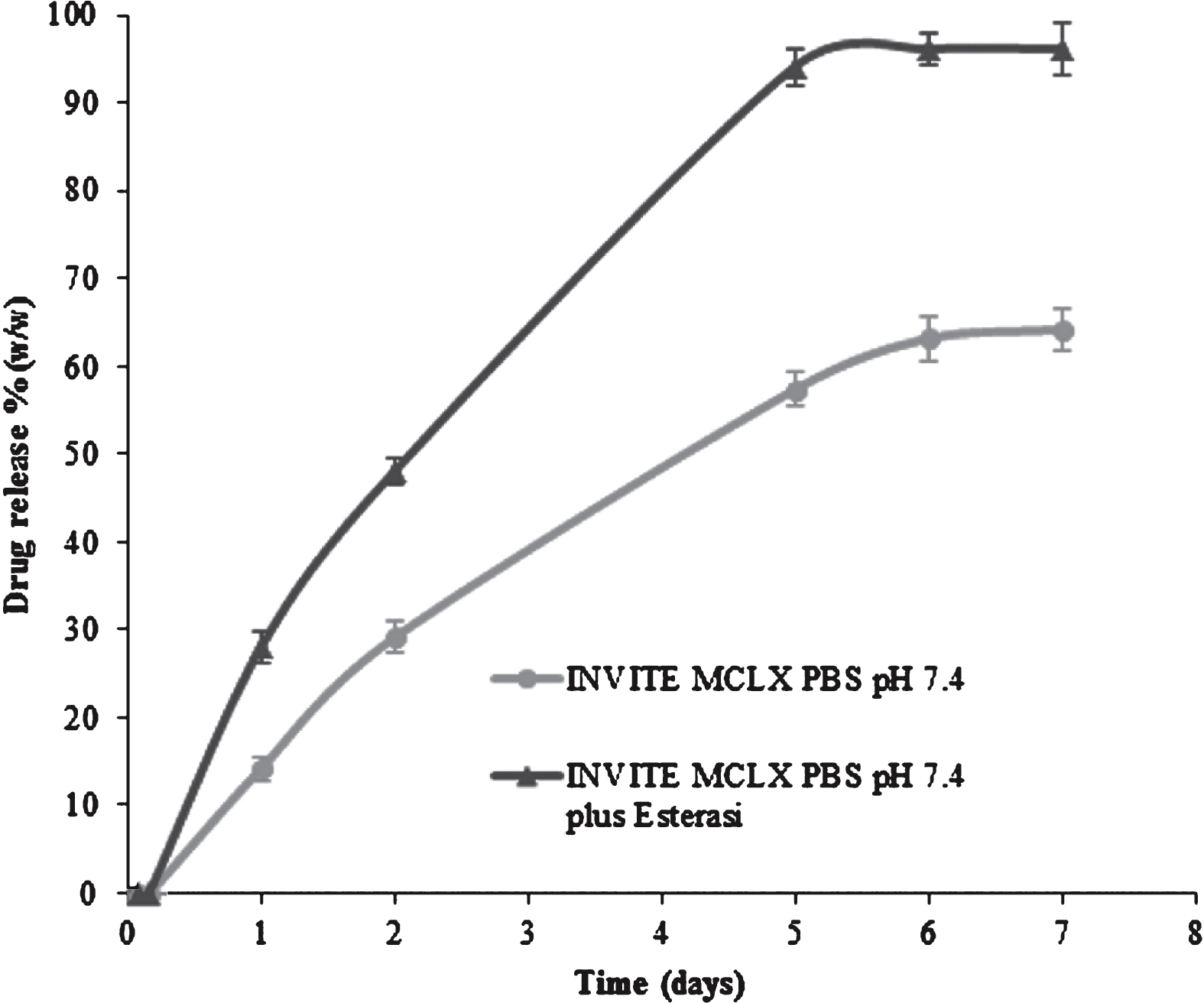
In order to evaluate the release performance of the CLX-loaded INVITE nanomicelles in simulated physiologic fluids, release studies were performed in PBS pH 7.4 in the presence or not of esterase. Furthermore, polysorbate 80 was added to the release medium to assure the sink conditions and the experiment was performed up to 7 days, Fig. 5.
As from Fig. 5, the release curves from micelle systems clearly indicate a controlled release of CLX which is significantly increased in the presence of esterase, furthermore no evident burst effect could be detected. In the same release conditions, free CLX is promptly dissolved (sink conditions reached).
The absence of a burst effect in the early stage of the experiment is indicative of the CLX localization inside the micelles, in fact, if it was adsorbed on the micelle surface a possibly evident burst effect due to its fast dissolution should appear. The absence of a burst effect, thus, could indicate that CLX is intimately embedded within the hydrophobic core by the formation of specific (hydrophobic and π - π stacking) interactions.
In particular, only 14 % of CLX from INVITE was released after 24 h of incubation at pH 7.4 while it was 28% when the INVITE MCLX was incubated in the presence of esterase. This CLX release trend was maintained throughout the experiment until 7 days in fact at day 7 the CLX release was 96 % in the presence of esterase while was 64 % without esterase. This suggests that esterase slowly degrades the polymer and permits a higher and complete drug release.
Furthermore, it should be noted that the release rate resulted almost constant with no time-related fluctuations so confirming the high physical stability of the proposed drug delivery system. The enzyme-sensitive behaviors of INVITE MCLX could be exploited for a modified and sustained release of the drug during either oral or parenteral administration of the drug delivery system.
4Conclusions
The effectiveness of INVITE systems for the aqueous solubilization and controlled release of CLX, chosen as model hydrophobic drug, has been clearly demonstrated by different experimental evidences.
It has been shown that CLX-loaded INVITE micelles are able to provide a sustained release of the entrapped drug without any evident burst effect. Furthermore, the nanometric dimensions of the INVITE systems should be favorable to escape the capture by the RES, so prolonging the time of residence of micelles in the blood when intravenously administered. Moreover, the good stability profile of the micelles suggests the possibility of their administration and storage in aqueous solution.
On the basis of presented results we postulate that INVITE systems are promising for the direct administration at the site of action of CLX which should be released in a sustained manner so providing a reduction of administered doses and, at the same time, producing less adverse side effects.
Acknowledgments
Adriana Trapani, Delia Mandracchia and Giuseppe Trapani would like to thank PONa300369 “Laboratorio per lo Sviluppo Integrato delle Scienze e delle Tecnologie dei Materiali Avanzati e per dispositive innovativi-LABORATORIO SISTEMA” financed by the Italian MIUR (Ministry of Education, University and Research).
References
1 | Williams HD, Trevaskis NL, Charman SA, Shanker RM, Charman WN, Pouton CW, Porter CJH(2013) Strategies to Address Low Drug Solubility in Discovery and DevelopmentPharmacological Reviews65: 315499 |
2 | Gaucher G, Dufresne MH, Sant VP, Kang N, Maysinger D, Leroux JC(2005) Block copolymer micelles: Preparation, characterization and application in drug deliveryJournal of Controlled Release109: 169188 |
3 | Bingham CO(2002) 3rd Development and clinical application of COX-2-selective inhibitors for the treatment of osteoarthritis and rheumatoid arthritisCleveland Clinic Journal of Medicine69: Suppl 1SI5SI12 |
4 | Zweers MC, de Boer TN, van Roon J, Bijlsma JWJ, Lafeber FPJG, Mastbergen SC(2011) Celecoxib: Considerations regardingits potential disease-modifying properties in osteoarthritisArthritis Research & Therapy13: 239 |
5 | Ullm S, Laube M, Bechmann N, Kniess T, Pietzsch J(2014) Organotypical vascular model for characterization of radioprotective compounds: Studies on antioxidant 2,3-diaryl-substituted indole-based cyclooxygenase-2 inhibitorsClin Hemorheol Microcirc58: 281295 |
6 | Tacconelli S, Capone ML, Sciulli MG, Ricciotti E, Patrignani P(2002) The biochemical selectivity of novel COX-2 inhibitors in whole blood assays of COX-isozyme activityCurrent Medical Research and Opinion18: 503511 |
7 | Silverstein FE, Faich G, Goldstein JL, Simon LS, Pincus T, Whelton A, Makuch R, Eisen G, Agarwal NM, Stenson WF, Burr AM, Zhao WW, Kent JD, Lefkowith JB, Verburg KM, Geis GS(2000) Gastrointestinal toxicity with celecoxib vs nonsteroidal anti-inflammatory drugs for osteoarthritis and rheumatoid arthritis - The CLASS study: A randomized controlled trialJama-Journal of the American Medical Association284: 12471255 |
8 | Chuang H-C, Kardosh A, Gaffney KJ, Petasis NA, Schoenthal AH(2008) COX-2 inhibition is neither necessary norsufficient for celecoxib to suppress tumor cell proliferation and focus formation in vitro MolecularCancer7: 38 |
9 | Seedher N, Bhatia S(2003) Solubility enhancement of Cox-2 inhibitors using various solvent systemsAAPS PharmSciTech4: E33E33 |
10 | Paulson SK, Zhang JY, Breau AP, Hribar JD, Liu NWK, Jessen SM, Lawal YM, Cogburn JN, Gresk CJ, Markos CS, Maziasz TJ, Schoenhard GL, Burton EG(2000) Pharmacokinetics, tissue distribution, metabolism, and excretion of celecoxib in ratsDrug Metabolism and Disposition28: 514521 |
11 | Kearney PM, Baigent C, Godwin J, Halls H, Emberson JR, Patrono C(2006) Do selective cyclo-oxygenase-2 inhibitors and traditional non-steroidal anti-inflammatory drugs increase the risk of atherothrombosis? Meta-analysis of randomised trialsBritish Medical Journal332: 13021305 |
12 | Li F, Zhu A, Song X, Ji L(2014) Novel surfactant for preparation of poly(L-lactic acid) nanoparticles with controllable release profile and cytocompatibility for drug deliveryColloids and Surfaces B-Biointerfaces115: 377383 |
13 | Licciardi M, Di Stefano M, Craparo EF, Amato G, Fontana G, Cavallaro G, Giammona G(2012) PHEA-graft-polybutylmethacrylate copolymer microparticles for delivery of hydrophobic drugsInternational Journal of Pharmaceutics433: 1624 |
14 | De Giglio E, Trapani A, Cafagna D, Sabbatini L, Cometa S(2011) Dopamine-loaded chitosan nanoparticles: Formulation and analytical characterizationAnalytical and Bioanalytical Chemistry400: 19972002 |
15 | Pitarresi G, Tripodo G, Triolo D, Fiorica C, Giammona G(2009) Inulin vinyl sulfone derivative cross-linked with bis-amino PEG: New materials for biomedical applicationsJournal of Drug Delivery Science and Technology19: 419423 |
16 | Pitarresi G, Tripodo G, Calabrese R, Craparo EF, Licciardi M, Giarnmona G(2008) Hydrogels for potential colon drug release by thiol-ene conjugate addition of a new inulin derivativeMacromolecular Bioscience8: 891902 |
17 | Pitarresi G, Palumbo FS, Tripodo G, Cavallaro G, Giammona G(2007) Preparation and characterization of new hydrogels based on hyaluronic acid and α,β-polyaspartylhydrazideEuropean Polymer Journal43: 39533962 |
18 | Tripodo G, Pitarresi G, Palumbo FS, Craparo EF, Giammona G(2005) UV-photocrosslinking of inulin derivatives to produce hydrogels for drug delivery applicationMacromolecular Bioscience5: 10741084 |
19 | Castelli F, Sarpietro MG, Micieli D, Ottimo S, Pitarresi G, Tripodo G, Carlisi B, Giammona G(2008) Differential scanning calorimetry study on drug release from an inulin-based hydrogel and its interaction with a biomembrane model: pH and loading effectEuropean Journal of Pharmaceutical Sciences35: 7685 |
20 | LoPresti C, Vetri V, Ricca M, Fodera V, Tripodo G, Spadaro G, Dispenza C(2011) Pulsatile protein release and protection using radiation-crosslinked polypeptide hydrogel delivery devicesReactive & Functional Polymers71: 155167 |
21 | Mandracchia D, Pitarresi G, Palumbo FS, Carlisi B, Giammona G(2004) PH-Sensitive hydrogel based on a novel photocross-linkable copolymerBiomacromolecules5: 19731982 |
22 | Pitarresi G, Saiano F, Cavallaro G, Mandracchia D, Palumbo FS(2007) A new biodegradable and biocompatible hydrogel with polyaminoacid structureInternational Journal of Pharmaceutics335: 130137 |
23 | Tripodo G, Mandracchia D, Collina S, Rui M, Rossi D(2014) New perspectives in cancer therapy: The biotin-antitumormolecule conjugatesMedicinal ChemistryS1S004 |
24 | Yu Y, Chen C-K, Law W-C, Weinheimer E, Sengupta S, Prasad PN, Cheng C(2014) Polylactide-graft-doxorubicin Nanoparticles with Precisely Controlled Drug Loading for pH-Triggered Drug DeliveryBiomacromolecules15: 524532 |
25 | Clementi C, Miller K, Mero A, Satchi-Fainaro R, Pasut G(2011) Dendritic Poly(ethylene glycol) Bearing Paclitaxel and Alendronate for Targeting Bone NeoplasmsMolecular Pharmaceutics8: 10631072 |
26 | Trapani A, Laquintana V, Lopedota A, Franco M, Latrofa A, Talani G, Sanna E, Trapani G, Liso G(2004) Evaluation of new propofol aqueous solutions for intravenous anesthesiaInternational Journal of Pharmaceutics278: 9198 |
27 | Aresta A, Calvano C, Trapani A, Cellamare S, Zambonin C, De Giglio E(2013) Development and analytical characterizationof vitamin(s)-loaded chitosan nanoparticles for potential food packaging applicationsJournal of NanoparticleResearch C7 - 159215: 112 |
28 | Montenegro L, Trapani A, Fini P, Mandracchia D, Latrofa A, Cioffi N, Chiarantini L, Picceri GG, Brundu S, Puglisi G(2014) Chitosan Nanoparticles for Topical Co-administration of the Antioxidants Glutathione and Idebenone: Characterization and In vitro ReleaseBritish. Journal of Pharmaceutical Research4: 23872406 |
29 | Venkatesan P, Puvvada N, Dash R, Kumar BNP, Sarkar D, Azab B, Pathak A, Kundu SC, Fisher PB, Mandal M(2011) The potential of celecoxib-loaded hydroxyapatite-chitosan nanocomposite for the treatment of colon cancerBiomaterials32: 37943806 |
30 | Bachar M, Mandelbaum A, Portnaya I, Perlstein H, Even-Chen S, Barenholz Y, Danino D(2012) Development and characterization of a novel drug nanocarrier for oral delivery, based on self-assembled beta-casein micellesJournal of Controlled Release160: 164171 |
31 | Mandracchia D, Tripodo G, Latrofa A, Dorati R(2014) Amphiphilic inulin-d-alpha-tocopherol succinate (INVITE) bioconjugates for biomedical applicationsCarbohydrate Polymers103: 4654 |
32 | Pitarresi G, Tripodo G, Cavallaro G, Palumbo FS, Giammona G(2008) Inulin-iron complexes: A potential treatment of iron deficiency anaemiaEuropean Journal of Pharmaceutics and Biopharmaceutics68: 267276 |
33 | Tripodo G, Pitarresi G, Cavallaro G, Palumbo FS, Giammona G(2009) Controlled release of IgG by novel UV induced polysaccharide/poly(aminoacid) hydrogelsMacromolecular Bioscience9: 393401 |
34 | Mandracchia D, Denora N, Franco M, Pitarresi G, Giammona G, Trapani G(2011) New Biodegradable Hydrogels Based on Inulin and alpha,beta-Polyaspartylhydrazide Designed for Colonic Drug Delivery: In Vitro Release of Glutathione and OxytocinJournal of Biomaterials Science-Polymer Edition22: 313328 |
35 | Tucker JM, Townsend DM(2005) Alpha-tocopherol: Roles in prevention and therapy of human diseaseBiomedicine & Pharmacotherapy59: 380387 |
36 | Fryer MJ(2000) Vitamin E as a protective antioxidant in progressive renal failureNephrology5: 17 |
37 | Tripodo G, Chlapanidas T, Perteghella S, Vigani B, Mandracchia D, Trapani A, Galuzzi M, Tosca MC, Antonioli B, Gaetani P, Marazzi M, Torre ML(2015) Mesenchymal Stromal Cells Loading Curcumin-INVITE-Micelles: A Drug Delivery System for Neurodegenerative DiseasesColloids and Surfaces B: Biointerfaces10.1016/j.colsurfb.2014.1011.1034 |
38 | Catenacci L, Mandracchia D, Sorrenti M, Colombo L, Serra M, Tripodo G(2014) In-Solution Structural Considerations by 1H NMR and Solid-State Thermal Properties of Inulin-d-α-Tocopherol Succinate (INVITE) Micelles as Drug Delivery Systems for Hydrophobic DrugsMacromolecular Chemistry and Physics215: 20842096 |
39 | Srinarong P, Hamalainen S, Visser MR, Hinrichs WLJ, Ketolainen J, Frijlink HW(2011) Surface-Active Derivative of Inulin (Inutec (R) SP1) Is a Superior Carrier for Solid Dispersions with a High Drug LoadJournal of Pharmaceutical Sciences100: 23332342 |
40 | Licciardi M, Scialabba C, Sardo C, Cavallaro G, Giammona G(2014) Amphiphilic inulin graft co-polymers as self-assembling micelles for doxorubicin deliveryJournal of Materials Chemistry B2: 42624271 |
Figures and Tables
Scheme1
Schematic representation of the INVITE bioconjugate chemical structure.
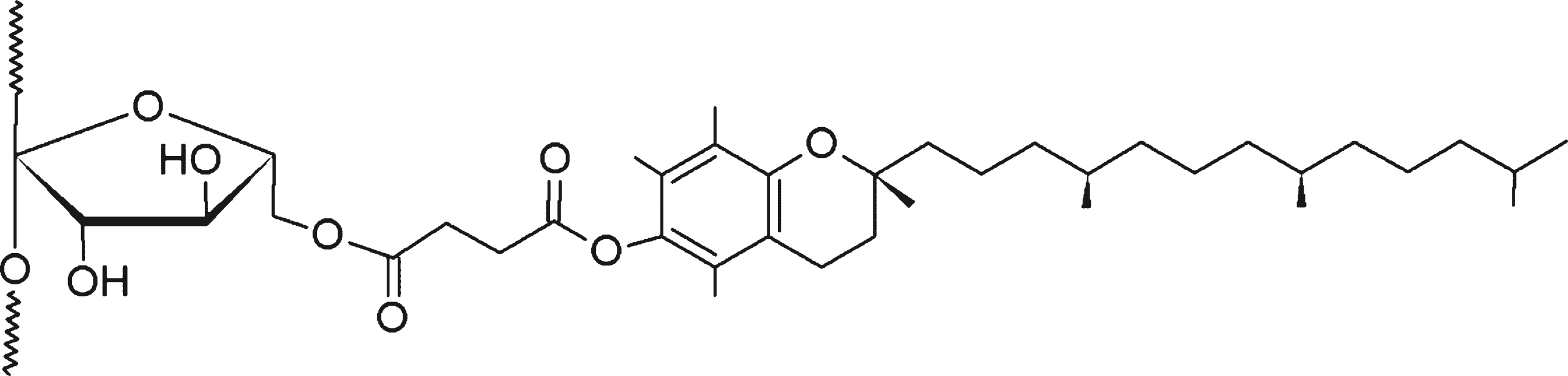
Fig.1
Reconstitution of INVITE MCLX micelles after freeze-drying.

Fig.2
FT-IR spectra of: a) CLX; b) INVITE conjugate; c) CLX loaded INVITE micelles (INVITE MCLX).
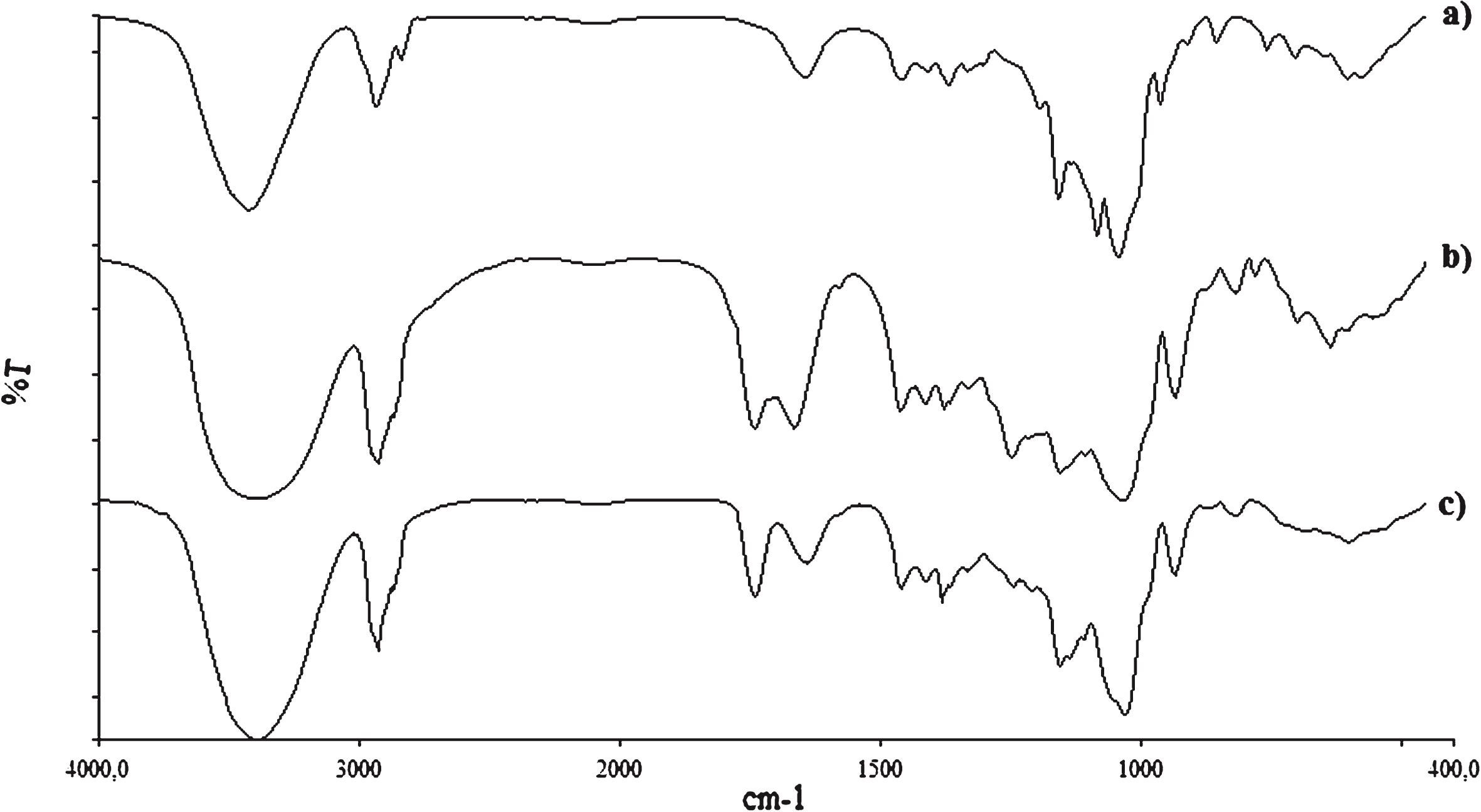
Fig.3
Chemical structures of: A) CLX; B) curcumin.
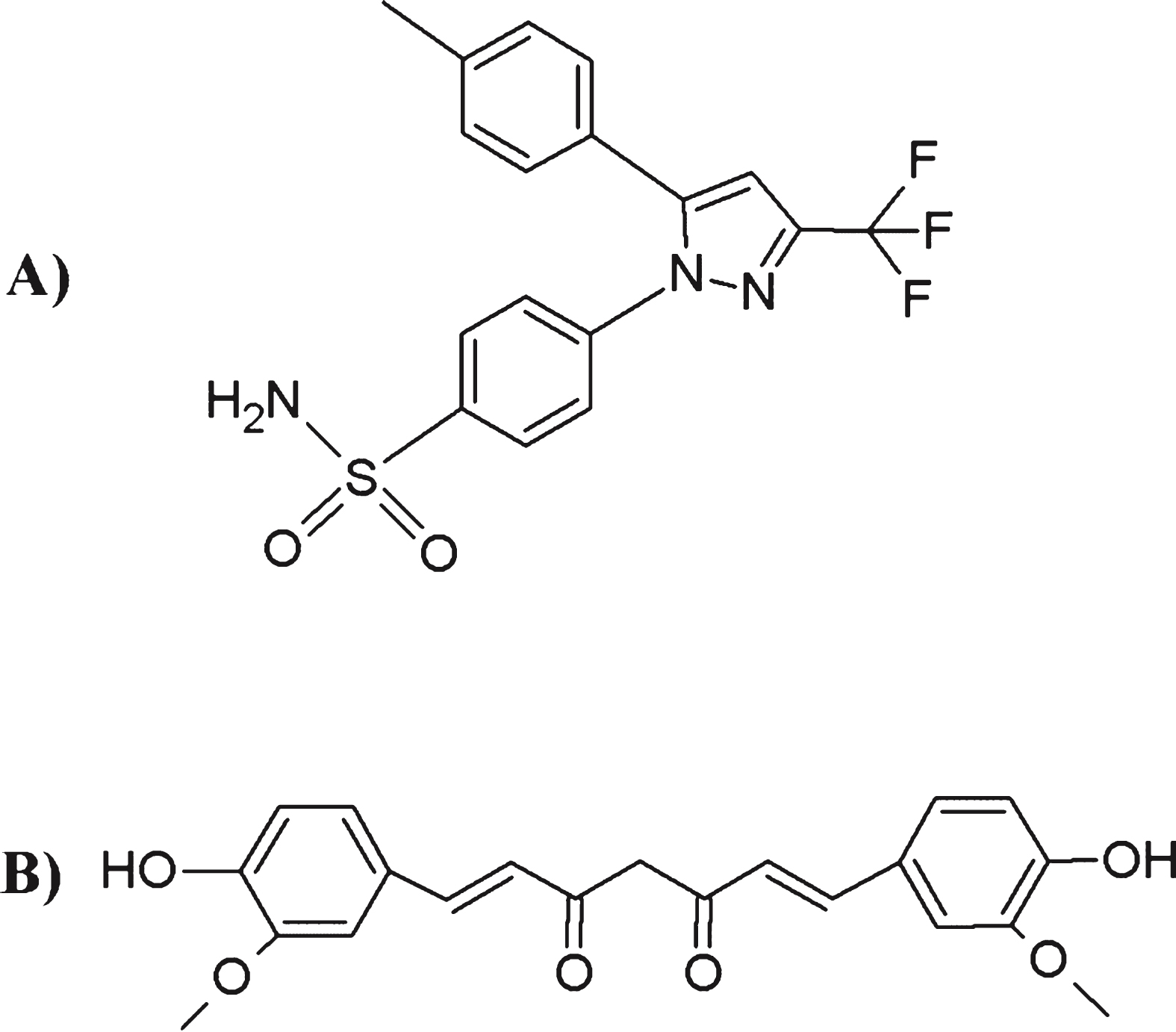
Fig.4
Stability study of empty INVITE M micelles and INVITE MCLX micelles loaded with CLX at 25°C up to 90 days in: A) ultrapure water; B) PBS pH 7.4.
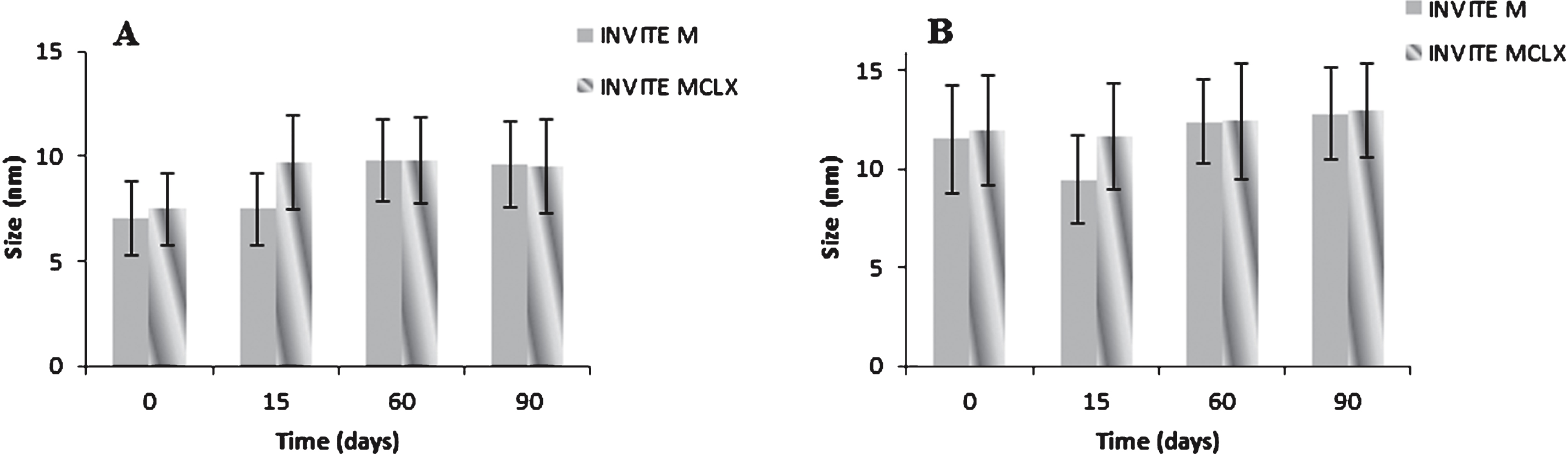
Fig.5
Cumulative release of CLX from INVITE MCLX at 37°C in PBS pH 7.4 or in PBS pH 7.4 plus esterase.