Characterization of neuropathic component of back pain in patients with osteoporotic vertebral fractures
Abstract
BACKGROUND:
Osteoporotic vertebral fractures (OVFs) are often followed by chronic back pain which may have a nociceptive, neuropathic, or mixed component. However, literature on this topic is lacking.
OBJECTIVE:
The objective of this cross-sectional study is to characterize the neuropathic component of chronic back pain in patients with OVFs.
METHODS:
Spine fractures were detected by morphometric examination. Pain severity and its impact on activities of daily living (ADL) were evaluated through the Brief Pain Inventory (BPI). Neuropathic pain was investigated through the Italian versions of the Leeds Assessment of Neuropathic Symptoms and Signs pain scale (LANSS) and the painDETECT questionnaire (PD-Q).
RESULTS:
We included 72 patients, mainly women (88.8%), with mean age of 69.2 years. The 70.8% of patients had multiple OVFs, of which 47% located at the thoracic spine, 43.1% at the thoracic and at lumbar spine, and 9.8% at the lumbar spine. The BPI showed moderate back pain in 23.6% of cases and severe in 8.3% of cases, with high interference with ADL in 38.9% of patients. The PD-Q revealed the presence of neuropathic pain in 5.5% of cases, while the LANSS in 23.6% of cases.
CONCLUSIONS:
In our study, the prevalence of neuropathic component of chronic back pain ranged from 5.5% to 23.6%, according to PD-Q and LANSS respectively, in patients with OVFs. Further studies should investigate if the characterization of chronic back pain might contribute to appropriateness of interventions for this population.
1Introduction
Osteoporosis (OP) is a systemic skeletal disease characterized by poor bone strength that predisposes to increased risk for fracture (Tella & Gallagher, 2014). Typically, fragility fractures of appendicular skeleton are a consequence of low energy trauma due to mechanical forces equivalent to a fall from upright or lower position, and which normally should not cause a fracture (National Institute for Health and Care Excellence, 2017). On the other hand, vertebral fractures, the most common osteoporotic fractures, are rarely related to a fall or trauma in general and often go undiagnosed due to little or no acute pain; therefore, their incidence in the general population is underestimated. However osteoporotic vertebral fractures (OVFs) can lead to chronic back pain, disability, and increased risk for new vertebral and non-vertebral fragility fractures (Tarantino et al., 2010).
The pathophysiology of bone pain in patients with OVFs is poorly understood and often assimilated to the pain experienced in other diseases or present in experimental models (Mediati et al., 2014). Nociceptors of the vertebral body are mainly located in the trabecular tissue and at the center of the endplate (Antonacci et al., 1998; Fagan et al., 2003), and contain specific peptide, such as the Transient Receptor Potential Vanilloid 1 (TRPV1), an acid-sensing receptor that are activated by acidic and inflammatory microenvironment resulting from osteoclastic bone resorption that is responsible for increased responsiveness of nociceptors (peripheral sensitization) (Nagae et al., 2006; Orita et al., 2010). In osteoporotic animal models, also dorsal root ganglia (DRG) neurons showed increased expression of TRPV1 and in addition of calcitonin gene-related peptide (CGRP), key mediators for the development of neuropathic pain as well as of peripheral and central sensitization (Orita et al., 2010). Therefore, it has been hypothesized that also neuropathic mechanisms play a key role in the genesis of back pain following OVFs (Suzuki et al., 2013). Neuropathic pain affects 7–10% of the general population and 37% of patients with chronic back pain presents clinical features of both neuropathic and nociceptive pain (Freynhagen et al., 2006).
Literature about characterization of the neuropathic component of chronic back pain in patients with OVFs is lacking. The aim of this study is therefore to characterize the prevalence of neuropathic component of chronic back pain in patients with OVFs.
2Materials and methods
2.1Participants
We conducted a cross-sectional study, satisfying the STROBE checklist criteria specific for this kind of study. Participants were consecutively recruited at a rehabilitation outpatient service dedicated to the management of osteoporosis (Fracture Liaison Service, FLS). They were informed about the study protocol and provided informed consent to the processing of personal data. Patients who satisfied the following criteria were included: (1) presence of chronic back pain (persisting for more than 3 months) (2) history of at least one OVFs documented by a radiographic imaging or vertebral morphometry performed from at least 3 to 12 months from the enrollment. We excluded patients with (1) acute back pain or (2) a clinical history of other causes of neuropathic pain such as disc herniation, spinal stenosis or by (3) chronic widespread pain, including fibromyalgia. The current study was performed in accordance with the Declaration of Helsinki and its later amendments.
2.2Vertebral fractures and pain evaluation
Vertebral fractures were identified by morphometric analysis performed through dual-energy x-ray absorptiometry (DXA). Vertebral morphometry is a semi-quantitative method to detect OVFs through the measurement of the different body vertebral heights (Diacinti, & Guglielmi, 2010). The presence of a OVFs was defined as the reduction of at least one vertebral height over 20% (Genant et al., 1993), whose severity of OVFs was by the following grades: 0 = normal; 1 = mild (a decrease in a height of a vertebra of 20–25%); grade 2 = moderate (a decrease of 25–40%); and grade 3 = severe (a decrease of 40% or more) fracture. We calculated the Spine Deformity Index (SDI), by summing the grade of the severity for each vertebra from T4 to L4.
Back pain severity and its impact on activities of daily living (ADL) were assessed through the Brief Pain Inventory (BPI) (Caraceni et al., 1996). The pain intensity section of the BPI includes four items (worst pain in last 24 hours, least pain in last 24 hours, pain on overage, pain right now) with a score ranging from 0 (no pain) to 10 (worst possible pain). Pain severity is calculated from the mean of the four BPI intensity items: absence of pain (score 0), mild pain (score 1–4) moderate pain (score 5–6) severe pain (score 7–10) (Deandrea et al., 2008; Li et al., 2007). BPI interference assessment is composed of seven items (general activity, mood, walking ability, normal work, relations with other people, sleep, enjoyment of life) with a score ranging from 1 (no interference) to 10 (complete interference). Pain interference with ADL is calculated from the mean of the BPI interference items as low interference (score 1–4) or high interference (score 5–10) (Cleeland & Ryan, 1994).
To identify neuropathic component of back pain in our population, we used the Leeds Assessment of Neuropathic Symptoms and Signs scale (LANSS) and the painDETECT questionnaire (PD-Q) (Migliore et al., 2021). The LANSS is an assess-ment tool used to estimate sensory dysfunction and the probable presence of a neuropathic mechanism underlying the pain. The tool consists of two parts, one of an interview with patient and the other one is a specific clinical examination. It includes 7 items: first 5 questions concern pain-related symptoms experienced by the patient, and the last 2 questions consist of clinical tests of non-painful (i.e., light touch) and pin-prick stimulations to detect allodynia or hyperalgesia, respectively. A score is assigned to each of the 7 items. For each item, the score is 0 in the absence of symptoms, while it ranged from 1 to 5 in the presence of symptoms or signs with a total score from 0 to 24. A score of 12 or more suggests the presence of a neuropathic component.
PD-Q is a simple screening tool to identify neuropathic pain in patients suffering from back pain. It includes 9 items of which 7 sensory symptom items for pain (score from 0 = never to 5 = strongly), one temporal item on pain-course pattern (score from –1 to +1), and one spatial item on pain radiation (score 0 = no radiation or +2 = radiating pain). The total score ranges from –1 to 38. The presence of neuropathic component is considered ambiguous with a score ranging from 12 to 18, while it is likely if the score is≥19.
2.3Statistical analysis
Statistical analysis was conducted using STATA 11.0. Study data were collected in a dedicated database. Data for continuous variables are given as means±standard deviations (SD) and categorical variables as counts (percentages). The normal distribution was investigated through the Shapiro-Wilk test for all the outcomes data. If data observed had a normal distribution, the Student’s t-test was used to compare continuous variables across groups; if not the two-sample Wilcoxon rank-sum (Mann–Whitney) test and Chi-square exact test were used when appropriate. To compare the means of three or more independent groups, one-way ANOVA test was performed. Correlation between SDI and type of pain was performed using Pearson’s correlation coefficient or Spearman’s rank correlation, in case of non-parametric variables. All tests were judged statistically significant if two-sided p-values were < 0.05.
3Results
Seventy-two patients were included (mainly women, 88.8%). The mean age of the participants was 69.2±8.9 years. Most individuals had multiple vertebral fractures (n = 51, 70.8%) (Table 1). All patients taking pain killers (25%) received paracetamol “as needed”, and none were on antidepressants or anticonvulsants. The most common site of OVFs was thoracic spine, both as single (n = 13, 61.9%) or multiple (n = 24, 47%) vertebral fractures (Figs. 1, 2). Considering pain intensity, our patients showed mild pain at BPI severity index (S.I.) (mean score 4.0±1.8) and a low interference with ADL (mean BPI interference score 4.3±2.1) (Table 2). According to the PD-Q, 82% (n = 59) of patients reported nociceptive pain, while neuropathic component was present only in 5.5% (n = 4) of cases. On the other side, the LANSS detected probable neuropathic mechanism in 23.6% (n = 17) of cases (Table 3). No significant differences were found about mean values of BPI S.I., BPI I.I., PD-Q and LANSS in patients with single OVFs versus those with multiple OVFs (Table 3). Among individuals with single fracture, neuropathic pain was significantly more prevalent in those with thoracic OVFs compared to patients with lumbar OVFs (Table 4). In participants with multiple fractures, only PD-Q identified a neuropathic component of back pain that was significantly more prevalent in patients with lumbar OVFs (Table 5). Finally no correlation among the SDI and the type of pain was found in our population (p = 0.551 and p = 0.863 for PD-Q and LANSS, respectively).
Table 1
Demographic and clinical data of study population
| Total | |
| n | 72 |
| Age (years) | 69.2±8.9 |
| Female sex | 64 (88.9%) |
| Male sex | 8 (11.1%) |
| Usage of pain medication | 18 (25%) |
| Anti-osteoporotic therapy | 62 (86.1%) |
| Bisphosphonates | 20 (27.8%) |
| Denosumab | 32 (44.4%) |
| Teriparatide | 10 (13.9%) |
| Single vertebral fracture | 21 (29.2%) |
| Multiple vertebral fractures | 51 (70.8%) |
Note: The values are expressed as means±standard deviations (SD) for continuous data and counts (percentages) for categorical data.
Fig. 1
Sites of single vertebral fragility fracture in our population.
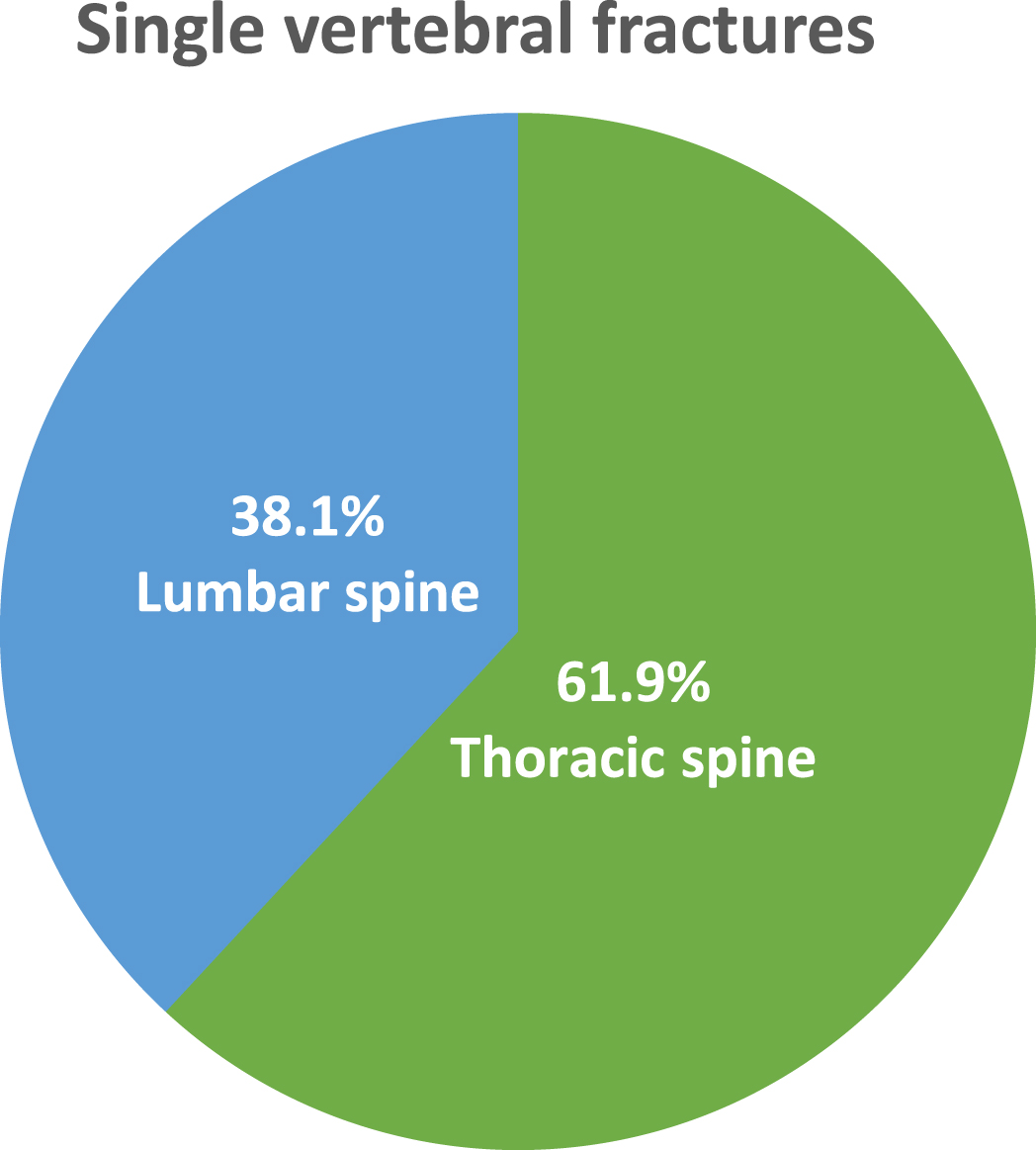
Fig. 2
Sites of multiple vertebral fragility fractures in our population.
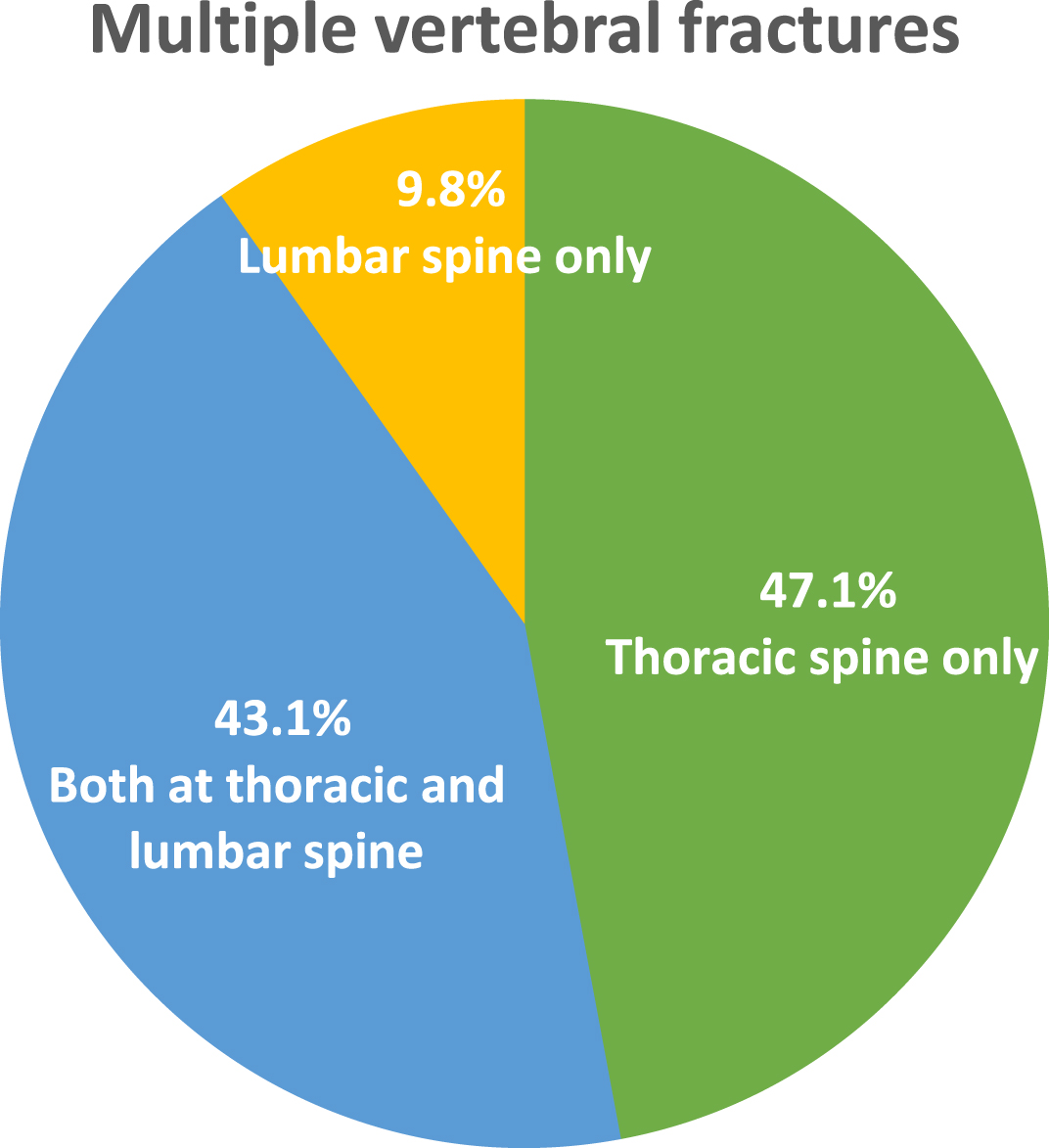
Table 2
Pain severity and interference with daily activities
| Assessment tool | No.% | Score (SD) |
| BPI severity index | ||
| Mild (1–4) | 49 (68.1) | 3.0±1.0 |
| Moderate (5–6) | 17 (23.6) | 5.9±0.4 |
| Severe (7–10) | 6 (8.3) | 7.2±0.3 |
| BPI interference index | ||
| Low (1–4) | 44 (61.1) | 2.9±1.2 |
| High (5–10) | 28 (38.9) | 6.6±0.8 |
Note: % =percentage; SD = standard deviation. BPI = Brief Pain Inventory.
Table 3
Pain classification through BPI and its indexes, painDETECT questionnaire and LANSS scale and its characterization according to the number of vertebral fragility fractures
| Assessment tool | n (%) | Score | Single vertebral fractures (n = 21) | Multiple vertebralfractures (n = 51) | p-values |
| BPI S.I. | 72 (100%) | 4.0±1.8 | 3.9±1.7 | 4.1±1.8 | 0.58* |
| BPI I.I. | 72 (100%) | 4.3±2.1 | 4.5±2.1 | 4.3±2.1 | 0.72** |
| PainDETECT questionnaire | 7.7±5.5 | 7.1±5.9 | 0.51** | ||
| Nociceptive (0–12) | 59 (82.0) | 5.1±3.6 | |||
| Mixed (13–18) | 9 (12.5) | 14.7±1.2 | |||
| Neuropathic (19–38) | 4 (5.5) | 22.2±2.0 | |||
| LANSS scale | 5.8±5.6 | 5.9±6.6 | 0.74** | ||
| Likely neuropathic pain (≥12) | 17 (23.6) | 15.9±2.1 | |||
| Unlikely neuropathic pain (< 12) | 55 (76.4) | 2.8±3.2 |
Note: % = percentage; SD = standard deviation. BPI = Brief Pain Inventory. S.I. = Severity Index. I.I. = Interference Index. LANSS = Leeds Assessment of Neuropathic Symptoms and Signs. *Student t-test. **Two-sample Wilcoxon rank-sum (Mann–Whitney) test.
Table 4
Relationship between vertebral fracture site and neuropathic pain in individuals with single fractures
| Thoracic spine (n = 13) | Lumbar spine (n = 8) | p-Value | |
| pain DETECT≥19 | 10.4±5.2 | 3.3±2.1 | 0.002* |
| LANSS≥12 | 7.8±5.8 | 2.6±3.0 | < 0.05** |
Note: Values are expressed as means±standard deviations (SD). LANSS = Leeds Assessment of Neuropathic Symptoms and Signs. *Student t-test. **Two-sample Wilcoxon rank-sum (Mann–Whitney) test.
Table 5
Relationship between vertebral fracture site and neuropathic pain in individuals with multiple fractures
| Thoracic spine (n = 24) | Lumbar spine (n = 5) | Thoracic-lumbar spine (n = 22) | p-Value | |
| pain DETECT≥19 | 5.0±4.3 | 12.4±7.0 | 8.1±6.3 | *0.022 |
| LANSS≥12 | 4.0±5.5 | 7.8±4.8 | 7.5±7.4 | *0.16 |
Note: Values are expressed as means±standard deviations (SD). LANSS = Leeds Assessment of Neuropathic Symptoms and Signs. *One-way ANOVA test.
4Discussion
To the best of our knowledge, this is the first study evaluating neuropathic component of chronic back pain in patients with OVFs.
Pain in osteoporotic patients is mostly nociceptive. Persistence of pathogenic mechanisms (e.g., inflammation or mechanical) contributes to sensitization of peripheral and central nervous system resulting in development of chronic pain (Mediati et al., 2014). Although the incidence of neuropathic component in patients with non-specific chronic back pain has been reported (Orita et al., 2016), data about neuropathic component in OVFs-related pain are lacking.
In our population, the percentage of patients experiencing neuropathic pain was 23.6% when neuropathic component was evaluated through LANSS, while this percentage decreased to 5.5% when it was assessed by the PD-Q.
The comparison of these assessment tools for defining the presence of the neuropathic component of pain has been performed in other studies. In particular, the greater sensitivity of LANSS has already been found in a study investigating pain type in individuals with carpal tunnel syndrome (Ceceli et al., 2018), while this finding has not been confirmed in patients with knee osteoarthritis (Moreton et al., 2015). In our opinion, the higher percentage of neuropathic pain detected by LANSS in our study might be related to the physical examination section included in this tool, performed by a physician able to investigate for example allodynia and hyperalgesia by skin stimulation. Moreover, while PD-Q scores provide a definition of three categories (i.e., “nociceptive”, “mixed” and “neuropathic” pain), in the LANSS we have only a distinction between likely or unlikely neuropathic pain mechanism. Therefore, it should be considered that the decreased percentage of cases reporting neuropathic pain might be explained by the fact that some patients had “mixed” pain (nociceptive with a neuropathic component) and so they have been included in the mixed pain category.
Regarding the prevalence of neuropathic component in osteoporotic patients, Fujimoto et al. conducted a study in 113 patients with back pain, reporting similar percentage (84.8% nociceptive, 11.6% mixed and 3.6% neuropathic pain) using PD-Q, although only 23% of their population had OVFs (Fujimoto et al., 2017).
In our study, no significant differences were found about pain intensity and interference with ADL as well as pain type in patients with single vertebral fracture versus those with multiple vertebral fractures. However, it has been reported that it is not so much the number but the severity of OVFs to be relevant for back pain-related loss of function and quality of life (Moretti et al., 2015).
According to our data, it seems that a different location of OVFs would be a main role in determining neuropathic component of back pain, since single thoracic fractures were significantly associated with higher prevalence of this type of pain. A possible hypothesis to explain this finding is that, compared to vertebral fractures in other sites, thoracic fractures had worse prognosis in terms of pain relief (Gerdhem, 2013). If back pain becomes chronic it is more probable the establishment of neuropathic mechanisms (Baron et al., 2016).
Our data showed that the association of fracture location and neuropathic component was not significant in patients with thoracic multiple OVFs, while it became significant, with higher prevalence in those with lumbar vertebral fractures. This is surprising considering that lumbar spine is less commonly involved by osteoporotic fractures.
In our study, these fractures affected only 9.8% of participants with multiple OVFs. It should be underlined that OVFs at lumbar spine have a significant higher incidence of radiculopathy that might explain the occurrence of neuropathic pain (Kim et al., 2015M; Ploumis, Transfledt, & Denis, 2007).
Back pain associated to OVFs may occur as a consequence of fracture per se (e.g., due to trauma and/or inflammation that activate nociceptors in acute fractures) as well as because of biomechanical changes of the spine (e.g., in persistent back pain progressively evolving in increased thoracic kyphosis) (Lindsay et al., 2001; Fechtenbaum et al., 2005). Moreover, persistent back pain may be associated to bone marrow edema that might occur either in the fractured or in the adjacent non-fractured vertebral bodies (Wang et al., 2013; Nakamura T, 2003). The transition from acute to chronic back pain may be due to both peripheral and central sensitizations, depending on nociceptive inputs from damaged bone tissue or from involvement of peripheral nerves that triggers central pain mechanisms (e.g., maladaptive neuroplasticity), respectively (Francis et al., 2008).
Unexpectedly, our patients showed mild pain and low interference with ADL, independently from number and site of OVFs. These findings can be attributable to the fact that most patients started anti-osteoporotic drug therapy immediately after the diagnosis of OVF, as expected by the specialized setting of study participants’ recruitment (i.e., FLS). Indeed, some analgesic effects have been described for anti-osteoporotic drugs (Ahn, Shin & Kim, 2017). Bisphosphonates improve pain acting with an antiresorptive mechanism on osteoclasts as well as inhibiting macrophages activation thus limiting neuropeptides and inflammatory cytokines production (Paolucci, Saraceni, & Piccinini, 2016). Teriparatide seems to reduce back pain not only by minimizing the risk of new OVFs, but also through healing and stabilization of pre-existing fractures (Nakajima et al., 2002; Andreassen, Ejersted & Oxlund, 1999). Denosumab seems to exert analgesic effects by suppressing production, differentiation, activation, and survival of osteoclasts, thus reducing osteoclast-mediated acidification and consequent acid-sensitive nociceptor stimulation, and by modulating NF-κB production, via RANK/RANKL inhibition that lead to reduced neuroinflammation and chronic pain (Moretti et al., 2019; Moretti et al., 2015).
Our study presents some limitations, including small sample size, the cross-sectional study design and no data about the mean time of back pain duration. Moreover, no tool for the assessment of risk of bias had been used. Finally, among questionnaires that we used to conduct this study, only the PD-Q was designed specifically for patients with chronic low back pain.
5Conclusions
Our study pointed out for the first-time the characterization of chronic back pain in patients with OVFs, investigating how much the neuropathic component is involved in this population. Our data demonstrated that neuropathic pain is present in over 20% of cases. This type of pain is more prevalent in patients with single OVF at the thoracic spine and in those with multiple OVFs at the lumbar spine.
In our experience, LANSS identified higher percentage of patients with neuropathic component. Further studies should better characterize chronic back pain in terms of nociceptive and neuropathic contribution in people with OVFs, also to define an appropriate therapeutic strategy for optimal pain relief in this population.
Conflict of interest
None to report.
Acknowledgments
None to report.
References
1 | Ahn, D. K. , Shin, W. S. , & Kim, G. W. ((2017) ). Analgesic Effects of Antiosteoporotic Drugs. Journal of Korean Society of Spine Surgery, 24: (1), 59–64. |
2 | Andreassen, T. T. , Ejersted, C. , & Oxlund, H. ((1999) ). Intermittent parathyroid hormone (1–34) treatment increases callus formation and mechanical strength of healing rat fractures. J Bone Miner Res, 14: , 960–968. |
3 | Antonacci, M. D. , Mody, D. R. , & Heggeness, M. H. ((1998) ). Innervation of the human vertebral body: a histologic study. Journal of Spinal Disorders, 11: (6), 526–531. |
4 | Baron, R. , Binder, A. , Attal, N. , Casale, R. , Dickenson, A. H. , & Treede, R. D. ((2016) ). Neuropathic low back pain in clinical practice. European Journal of Pain (London, England), 20: (6), 861–873. https://doi.org/10.1002/ejp.838 |
5 | Caraceni, A. , Mendoza, T. R. , Mencaglia, E. , Baratella, C. , Edwards, K. , Forjaz, M. J. , Martini, C. , Serlin, R. C. , de Conno, F. , & Cleeland, C. S. ((1996) ). A validation study of an Italian version of the Brief Pain Inventory (Breve Questionario per la Valutazione del Dolore). Pain, 65: (1), 87–92. https://doi.org/10.1016/0304-3959(95)00156-5 |
6 | Ceceli, E. , Gumruk, S. , Okumus, M. , Kocaoglu, S. , Goksu, H. , & Karagoz, A. ((2018) ). Comparison of 2 methods of neuropathic pain assessment in carpal tunnel syndrome and hand functions. Neurosciences (Riyadh, Saudi Arabia), 23: (1), 23–28. https://doi.org/10.17712/nsj.2018.1.20170345 |
7 | Cleeland, C. S. , & Ryan, K. M. ((1994) ). Pain assessment: global use of the Brief Pain Inventory. Annals of the Academy of Medicine. Singapore, 23: (2), 129–138. |
8 | Deandrea, S. , Montanari, M. , Moja, L. , & Apolone, G. ((2008) ). Prevalence of undertreatment in cancer pain. A review of published literature. Annals of Oncology: Official Journal of the European Society for Medical Oncology, 19: (12), 1985–1991. https://doi.org/10.1093/annonc/mdn419 |
9 | Diacinti, D. , & Guglielmi, G. ((2010) ). Vertebral morphometry. Radiologic Clinics of North America, 48: (3), 561–575. https://doi.org/10.1016/j.rcl.2010.02.018 |
10 | Fagan, A. , Moore, R. , Vernon Roberts, B. , Blumbergs, P. , & Fraser, R. ((2003) ). ISSLS prize winner: The innervation of the intervertebral disc: a quantitative analysis. Spine, 28: (23), 2570–2576. https://doi.org/10.1097/01.BRS.0000096942.29660.B1 |
11 | Fechtenbaum, J. , Cropet, C. , Kolta, S. , Verdoncq, B. , Orcel, P. , & Roux, C. ((2005) ). Reporting of vertebral fractures on spine X-rays. Osteoporosis International: A Journal Established as Result of Cooperation Between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA, 16: (12), 1823–1826. https://doi.org/10.1007/s00198-005-1939-8 |
12 | Francis, R. M. , Aspray, T. J. , Hide, G. , Sutcliffe, A. M. , & Wilkinson, P. ((2008) ). Back pain in osteoporotic vertebral fractures. Osteoporosis International: A Journal Established as Result of Cooperation Between the European Foundation for Osteoporosis and the National Osteoporosis Foundation of the USA, 19: (7), 895–903. https://doi.org/10.1007/s00198-007-0530-x |
13 | Freynhagen, R. , Baron, R. , Gockel, U. , & Tölle, T. R. ((2006) ). painDETECT: a new screening questionnaire to identify neuropathic components in patients with back pain. Current Medical Research and Opinion, 22: (10), 1911–1920. https://doi.org/10.1185/030079906X132488 |
14 | Fujimoto, K. , Inage, K. , Orita, S. , Yamashita, M. , Abe, K. , Yamagata, M. , Sainoh, T. , Akazawa, T. , Kinoshita, T. , Nemoto, T. , Hirayama, J. , Murata, Y. , Kotani, T. , Aoki, Y. , Eguchi, Y. , Sakuma, T. , Aihara, T. , Ishikawa, T. , Suseki, K. , Hanaoka, E. , ... Ohtori, S. ((2017) ). The nature of osteoporotic low back pain without acute vertebral fracture: A prospective multicenter study on the analgesic effect of monthly minodronic acid hydrate. Journal of Orthopaedic Science: Official Journal of the Japanese Orthopaedic Association, 22: (4), 613–617. https://doi.org/10.1016/j.jos.2017.01.022 |
15 | Genant, H. K. , Wu, C. Y. , van Kuijk, C. , & Nevitt, M. C. ((1993) ). Vertebral fracture assessment using a semiquantitative technique. Journal of Bone and Mineral Research: The Official Journal of the American Society for Bone and Mineral Research, 8: (9), 1137–1148. https://doi.org/10.1002/jbmr.5650080915 |
16 | Gerdhem P. ((2013) ). Osteoporosis and fragility fractures: Vertebral fractures. Best practice & research. Clinical Rheumatology, 27: (6), 743–755. https://doi.org/10.1016/j.berh.2014.01.002 |
17 | Kim, D. E. , Kim, H. S. , Kim, S. W. , & Kim, H. S. ((2015) ). Clinical analysis of acute radiculopathy after osteoporotic lumbar compression fracture. Journal of Korean Neurosurgical Society, 57: (1), 32–35. https://doi.org/10.3340/jkns.2015.57.1.32 |
18 | Li, K. K. , Harris, K. , Hadi, S. , & Chow, E. ((2007) ). What should be the optimal cut points for mild, moderate, and severe pain? Journal of Palliative Medicine, 10: (6), 1338–1346. https://doi.org/10.1089/jpm.2007.0087 |
19 | Lindsay, R. , Silverman, S. L. , Cooper, C. , Hanley, D. A. , Barton, I. , Broy, S. B. , Licata, A. , Benhamou, L. , Geusens, P. , Flowers, K. , Stracke, H. , & Seeman, E. ((2001) ). Risk of new vertebral fracture in the year following a fracture. JAMA, 285: (3), 320–323. https://doi.org/10.1001/jama.285.3.320 |
20 | Mediati, R. D. , Vellucci, R. , & Dodaro, L. ((2014) ). Pathogenesis and clinical aspects of pain in patients with osteoporosis. Clinical Cases in Mineral and Bone Metabolism: The Official Journal of the Italian Society of Osteoporosis, Mineral Metabolism, and Skeletal Diseases, 11: (3), 169–172. |
21 | Migliore, A. , Gigliucci, G. , Moretti, A. , Pietrella, A. , Peresson, M. , Atzeni, F. , Sarzi-Puttini, P. , Bazzichi, L. , Liguori, S. , & Iolascon, G. ((2021) ). Cross Cultural Adaptation and Validation of Italian Version of the Leeds Assessment of Neuropathic Symptoms and Signs Scale and Pain DETECT Questionnaire for the Distinction between Nociceptive and Neuropathic Pain. Pain Research & Management, 2021, 6623651. https://doi.org/10.1155/2021/6623651 |
22 | Moreton, B. J. , Tew, V. , das Nair, R. , Wheeler, M. , Walsh, D. A. , & Lincoln, N. B. ((2015) ). Pain phenotype in patients with knee osteoarthritis: classification and measurement properties of painDETECT and self-report Leeds assessment of neuropathic symptoms and signs scale in a cross-sectional study. Arthritis Care & Research, 67: (4), 519–528. https://doi.org/10.1002/acr.22431 |
23 | Moretti, A. , de Sire, A. , Curci, C. , Toro, G. , Gimigliano, F. , & Iolascon, G. ((2019) ). Effectiveness of denosumab on back pain-related disability and quality-of-life in patients with vertebral fragility fractures. Current Medical Research and Opinion, 35: (1), 151–155. https://doi.org/10.1080/03007995.2018.1545636 |
24 | Moretti, A. , Gimigliano, F. , Di Pietro, G. , Gimigliano, R. , & Iolascon, G. ((2015) ). Back pain-related disability and quality of life in patients affected by vertebral fractures: data from baseline characteristics of population enrolled in Denosumab In Real Practice (DIRP). Aging Clinical and Experimental Research, 27: (Suppl 1), S3–S9. https://doi.org/10.1007/s40520-015-0428-y |
25 | Nagae, M. , Hiraga, T. , Wakabayashi, H. , Wang, L. , Iwata, K. , & Yoneda, T. ((2006) ). Osteoclasts play a part in pain due to the inflammation adjacent to bone. Bone, 39: (5), 1107–1115. https://doi.org/10.1016/j.bone.2006.04.033 |
26 | Nakajima, A. , Shimoji, N. , Shiomi, K. , Shimizu, S. , Moriya, H. , Einhorn, T. A. , & Yamazaki, M. ((2002) ). Mechanisms for the enhancement of fracture healing in rats treated with intermittent low-dose human parathyroid hormone (1-34). Journal of Bone and Mineral Research: The Official Journal of the American Society for Bone and Mineral Research, 17: (11), 2038–2047. https://doi.org/10.1359/jbmr.2002.17.11.2038 |
27 | Nakamura T. ((2003) ). Low back pain accompanying osteoporosis. Japan Med Assoc J, 46: (10), 445–451. |
28 | National Institute for Health and Care Excellence (NICE). ((2017) ). Osteoporosis: assessing the risk of fragility fracture. |
29 | Orita, S. , Ohtori, S. , Koshi, T. , Yamashita, M. , Yamauchi, K. , Inoue, G. , Suzuki, M. , Eguchi, Y. , Kamoda, H. , Arai, G. , Ishikawa, T. , Miyagi, M. , Ochiai, N. , Kishida, S. , Takaso, M. , Aoki, Y. , Toyone, T. , & Takahashi, K. ((2010) ). The effects of risedronate and exercise on osteoporotic lumbar rat vertebrae and their sensory innervation. Spine, 35: (22), 1974–1982. https://doi.org/10.1097/BRS.0b013e3181d5959e |
30 | Orita, S. , Yamashita, T. , Ohtori, S. , Yonenobu, K. , Kawakami, M. , Taguchi, T. , Kikuchi, S. I. , Ushida, T. , Konno, S. I. , Nakamura, M. , Fujino, K. , Matsuda, S. , Yone, K. , & Takahashi, K. ((2016) ). Prevalence and Location of Neuropathic Pain in Lumbar Spinal Disorders: Analysis of 1804 Consecutive Patients With Primary Lower Back Pain. Spine, 41: (15), 1224–1231. https://doi.org/10.1097/BRS.0000000000001553 |
31 | Paolucci, T. , Saraceni, V. M. , & Piccinini, G. ((2016) ). Management of chronic pain in osteoporosis: challenges and solutions. Journal of Pain Research, 9: , 177–186. https://doi.org/10.2147/JPR.S83574 |
32 | Ploumis, A. , Transfledt, E. E. , & Denis, F. ((2007) ). Degenerative lumbar scoliosis associated with spinal stenosis. The Spine Journal: Official Journal of the North American Spine Society, 7: (4), 428–436. https://doi.org/10.1016/j.spinee.2006.07.015 |
33 | Suzuki, M. , Orita, S. , Miyagi, M. , Ishikawa, T. , Kamoda, H. , Eguchi, Y. , Arai, G. , Yamauchi, K. , Sakuma, Y. , Oikawa, Y. , Kubota, G. , Inage, K. , Sainoh, T. , Kawarai, Y. , Yoshino, K. , Ozawa, T. , Aoki, Y. , Toyone, T. , Takahashi, K. , Kawakami, M. , ... Inoue, G. ((2013) ). Vertebral compression exacerbates osteoporotic pain in an ovariectomy-induced osteoporosis rat model. Spine, 38: (24), 2085–2091. https://doi.org/10.1097/BRS.0000000000000001 |
34 | Tarantino, U. , Capone, A. , Planta, M. , D’Arienzo, M. , Letizia Mauro, G. , Impagliazzo, A. , Formica, A. , Pallotta, F. , Patella, V. , Spinarelli, A. , Pazzaglia, U. , Zarattini, G. , Roselli, M. , Montanari, G. , Sessa, G. , Privitera, M. , Verdoia, C. , Corradini, C. , Feola, M. , Padolino, A. , ... Piscitelli, P. ((2010) ). The incidence of hip, forearm, humeral, ankle, and vertebral fragility fractures in Italy: results from a 3-year multicenter study. Arthritis Research & Therapy, 12: (6), R226. https://doi.org/10.1186/ar3213 |
35 | Tella, S. H. , & Gallagher, J. C. ((2014) ). Prevention and treatment of postmenopausal osteoporosis. The Journal of Steroid Biochemistry and Molecular Biology, 142: , 155–170. |
36 | Wang, C. K. , Tsai, J. M. , Chuang, M. T. , Wang, M. T. , Huang, K. Y. , & Lin, R. M. ((2013) ). Bone marrow edema in vertebral compression fractures: detection with dual-energy CT. Radiology, 269: (2), 525–533. https://doi.org/10.1148/radiology.13122577 |




