Autonomic dysfunction in adults following mild traumatic brain injury: A systematic review
Abstract
BACKGROUND:
Increasing evidence suggests autonomic nervous system (ANS) dysfunction may occur following mild traumatic brain injury (mTBI). Measures of heart rate, heart rate variability, blood pressure and baroreceptor sensitivity can be used to evaluate ANS dysfunction following mTBI.
OBJECTIVE:
Summarize the evidence for ANS dysfunction in adults following mTBI.
METHODS:
A search of Embase, MEDLINE, Cochrane Central Register, PsycINFO, CINAHL and SPORTDiscus databases was conducted. Search topics included: mTBI and ANS. Identified abstracts were independently reviewed by 2 reviewers followed by full text screening. Risk of bias was assessed using a modified SIGN checklist. A structured synthesis was performed.
RESULTS:
Thirty-nine studies (combined 1,467 participants diagnosed with mTBI) evaluating ANS function were included. ANS function was evaluated under various conditions including: rest, during exertion, cold pressor test, Valsalva maneuver, using face cooling and eyeball pressure paradigms. Short-term or ultra-short-term recordings were most common. The majority of studies (28/39) were rated as “unacceptable” for quality of evidence.
CONCLUSIONS:
Altered parameters of ANS function have been reported in multiple conditions following mTBI, both acutely and in the post-acute/chronic stages of recovery. However, due to methodological limitations, conclusions regarding the severity and timing of ANS dysfunction following mTBI cannot be drawn.
1Introduction
Mild traumatic brain injury (mTBI), often referred to as concussion, can occur following a biomechanical force to the head or body resultant in a neurometabolic cascade and energy crisis (Giza & Hodva, 2014). While diagnostic criteria differ based on a variety of definitions of mTBI and concussion, it is an injury often characterized by a loss of consciousness less than 30 minutes, post-traumatic amnesia less than 24 hours and Glasgow Coma Scale score of 13–15 (Mild Traumatic Brain Injury Committee of the Head Injury Interdisciplinary Special Interest Group, 1993). There is an annual estimated incidence of mTBI of 1,153/100,000 in Canada (Langer et al., 2020). Adults generally recover in the 10–14 day period following injury (McCrory et al., 2017), however up to 30% will go on to develop persistent post-concussive symptoms, including: headache (Lucas et al., 2013), dizziness (Theadom et al., 2016), fatigue (Stulemeijer et al., 2006) and exercise intolerance (Kozlowski et al., 2013). Currently, the pathophysiology underpinning mTBI recovery and symptom resolution is not fully understood; however, it is hypothesized several of the aforementioned post-concussive symptoms may be associated with autonomic dysfunction. Therefore, evaluation of autonomic function post-mTBI may provide insight into physiologic mechanisms underpinning mTBI recovery and symptom presentation.
The autonomic nervous system (ANS), composed of the parasympathetic and sympathetic branches, functions to maintain bodily homeostasis (Wehrwein et al., 2016). Sympathetic preganglionic neurons exit the ventral horns of the spinal cord, synapsing on sympathetic postganglionic neurons in the paravertebral sympathetic chain before terminating on effector organs, including the heart and lungs (Wehrwein et al., 2016). Activation of the sympathetic nervous system relaxes tracheal and bronchial smooth muscle and increases atrial muscle contractility, conduction velocity of the atrial ventricular node and heart rate (HR) (through innervation of the sinoatrial node) (Wehrwein et al., 2016). The parasympathetic branch of the ANS includes cranial nerves III, VII, IX and X (Wehrwein et al., 2016). Cranial nerve X, the vagus nerve, regulates several organ systems, including the heart and lungs. Increased vagal activation of the sinoatrial node further decreases its intrinsic firing rate, thus decreasing HR (Wehrwein et al., 2016).
Several measures can be used to evaluate ANS function. The ones most commonly employed are: heart rate variability (HRV) and cardiac baroreceptor sensitivity (BRS). HRV is normally monitored either at rest (i.e., supine, seated or upright quiet-stance) or during physiologic challenge (i.e., sit-stand protocol, isometric hand grip). HRV describes the variation in time intervals between consecutive heart beats and provides a measure of the ability of the ANS to adapt to external stimuli/challenge (Shaffer & Ginsberg, 2017). Higher time-domain HRV is generally considered to be an indicator of cardiovascular health, especially in terms of cardiovascular-related mortality (Task Force of the European Society of Cardiology and The North American Society of Pacing and Electrophysiology, 1996). Due to the exponential decay-like relationship between time-domain HRV parameters and HR (Monfredi et al., 2014), an exertional task is expected to produce an increase in HR and corresponding decrease in HRV. However, altered HR/HRV responses to physiologic challenge have been observed in individuals with mTBI compared to healthy control groups (Pertab et al., 2018). Measures of BRS provide another method of evaluating autonomic function. The baroreflex serves to regulate HR in response to blood pressure (BP) changes sensed by baroreceptors in the carotid arteries and aortic arch (Duschek et al., 2013). Research suggests BRS may be blunted post-mTBI (Pertab et al., 2018). Whether altered autonomic function post-mTBI is due to deconditioning (in those with persistent symptoms), altered central integration or another mechanism has not yet been well characterized.
Given the potential for metrics of autonomic function to both help understand the pathophysiology of post-concussive symptoms and provide an objective measure of recovery, a review of the literature including adults with all mechanisms of mTBI is warranted. While there are previous reviews of the literature in this field (Blake et al., 2016; Callaway & Kosofsky, 2019; Pertab et al., 2018; Purkayastha, Stokes et al., 2019) they have several limitations. Namely, the prior reviews either were not systematic in nature or they did not sufficiently address/discuss the methodological limitations of the included studies. Further, as this is a rapidly advancing field of research, this review includes recently published studies that were not available to be included in prior reviews. Therefore, the objective of this review was to systematically characterize measures of autonomic function, including HRV, BP and BRS, in patients with mTBI compared to controls or in mTBI groups over time.
2Materials and methods
2.1Protocol and registration
The review was first registered in November 2019 (updated April 2020 and January 2021) with the National Institute for Health Research’s International Prospective Register of Systematic Reviews. The registered protocol (PROSPERO CRD42020157698) can be accessed at: https://www.crd.york.ac.uk/prospero/ display_record.php?RecordID = 157698. Institutional ethics approval was not required as no primary data collection occurred. The methods of this review followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Liberati et al., 2009).
2.2Information sources
Embase (1974 –January 21, 2021), Ovid MEDLINE (1946 –January 22, 2021), Cochrane Central Register of Clinical Trials (December 2020), PsycINFO (1803 –January Week 3, 2021), CINAHL (1973 –January 2021) and SPORTDiscus (1930 –January 2021) were searched. In databases that allowed, results were restricted to human studies. Search results were limited to English language studies. Filters were applied to select for adult and young adult populations as appropriate within the specific database searches. The final search was completed on January 24, 2021. No study authors were contacted to identify investigations beyond published work. Authors were encouraged to share additional references meeting inclusion criteria.
2.3Search
Development of the search strategy was done with assistance of a University of Calgary health sciences librarian. Search terms were related to 1) mTBI and 2) ANS. An example search strategy is provided for Embase in Table 1 and search strategies for additional databases are also available (Supplementary Tables 1-5). Records were exported directly from databases and imported into EndnoteX9 (EndNote Team, 2013). All results were exported from EndnoteX9 (EndNote Team, 2013) and imported into Covidence systematic review software where duplicates were removed.
Table 1
Example search strategy
| Embase search strategy |
| 1. exp brain concussion |
| 2. ((brain or cerebral or craniocerebral or cranio-cerebral or intra-cranial or intracranial) adj2 (concuss* or hematoma* or haematoma* or injur* or contus*)).tw,kf. |
| 3. mild traumatic brain injur*.tw,kf. |
| 4. mtbi.tw,kf. |
| 5. ((mild or minor or minimal) adj head injur*).tw,kf. |
| 6. (concussion* or contusion*).tw,kf. |
| 7. Post-Concussion Syndrome |
| 8. or/1-7 |
| 9. exp Autonomic Nervous System |
| 10. (autonomic or parasympathetic or sympathetic) adj3 nervous system adj3 (disorder* or dysfunction* or disease*)).tw,kf. |
| 11. Heart Rate |
| 12. heart rate variability.tw,kf. |
| 13. heart rate control.tw,kf. |
| 14. pulse rate.tw,kf. |
| 15. Blood Pressure |
| 16. blood pressure.tw,kf. |
| 17. Syncope/ or syncope, vasovagal |
| 18. syncope.tw,kf. |
| 19. tilt-table test |
| 20. tilt-table test.tw,kf. |
| 21. Postural orthostatic tachycardia syndrome |
| 22. Postural orthostatic tachycardia syndrome.tw,kf. |
| 23. valsalva maneuver |
| 24. valsalva maneuver.tw,kf. |
| 25. homeostasis/ or feedback, physiological |
| 26. homeostasis.tw,kf. |
| 27. cerebral autoregulation.tw,kf. |
| 28. Baroreflex |
| 29. Baroreflex.tw,kf. |
| 30. or/9-29 |
| 31. 8 and 30 |
| 32. limit 31 to (“all adult (19 plus years)” or “adolescent (13 to 18 years)”) |
| 33. limit 32 to English |
2.4Eligibility criteria
Inclusion and exclusion criteria for study design, participants, outcome measures and comparators are provided in Table 2.
Table 2
Eligibility criteria
| Eligibility criteria | Inclusion | Exclusion |
| Participants | Mild traumatic brain injury group with mean age≥18 | Pediatric participants, pre-clinical studies, GCS < 13, heart conditions, neurosurgical interventions, cohorts of only moderate-severe traumatic brain injury |
| Measures | Resting heart rate, heart rate variability, systolic blood pressure, diastolic blood pressure, mean arterial pressure, pupillary response, baroreceptor sensitivity, autonomic function questionnaires | Intracranial pressure, cerebrovascular resistance, neurofeedback, cerebral perfusion pressure, cerebral blood flow |
| Comparators | Control group without acute mild traumatic brain injury | |
| Study types | Cohort studies, randomized control trials, case series, case-control trials, meta-analysis, cross-sectional studies | Case studies, reviews, expert opinions, conference abstracts, economic evaluations, pre-clinical studies |
2.5Study selection
Following duplicate removal, all abstracts were screened by two independent reviewers based on eligibility criteria. Conflicts regarding inclusion/exclusion of studies were resolved by a third independent reviewer. Full text screening of included abstracts was completed by two independent reviewers and the same process was applied for conflict resolution.
2.6Data extraction
Data extraction and risk of bias (RoB) were performed by two reviewers independently for all studies meeting the inclusion criteria following full text screening. The following data was collected: participant characteristics (age, sex/gender), mTBI diagnosis and classification, control group, assessment timepoint(s), time since injury, physiologic measure with units and duration, test condition(s) and summarized results.
Each study was assessed for RoB by two independent reviewers using modified checklists evaluated and adapted to meet Scottish Intercollegiate Guidelines Network (SIGN) criteria (Healthcare Improvement Scotland, n.d.). A modified version of the SIGN Cohort checklist was used for cross-sectional and cohort study designs. A modified version of the SIGN Case-control checklist was used for case-control study design. The injury (mTBI) was considered as the “exposure” or “case” (for case-control studies) and metrics of ANS dysfunction as the “outcome”. The SIGN checklists contain 13–15 questions with four response categories: “yes”, “no”, “not applicable” and “can’t say”. In the cohort study checklist: A) Item 1.5 regarding drop outs was used to assess whether or not drop outs were reported; B) Item 1.11 was divided into two items, 1.11 and 1.12, which respectively assessed, “the outcome measure(s) is/are reliable” and “the outcome measure(s) is/are valid”; C) For item 1.13, “exposure level of prognostic factor is assessed more than once”, symptomatology/presence of persistent symptoms was considered the “exposure” and for studies with a single timepoint symptomatology had to be assessed once for the item to be rated as “yes”. In the case-control study checklist: the items, “the outcome measure(s) is/are reliable” (1.10) and “the outcome measure(s) is/are valid” (1.11) were added. Evaluation is based on reliability and validity of subject selection, assessment, confounding factors and statistical analysis. The overall assessment of the study was based on the ability to minimize RoB. Discrepancies were discussed and if a consensus was not achieved by two reviewers, conflicts were resolved by a third independent reviewer.
The corresponding authors for three included studies were contacted. The first researcher was contacted to provide supplementary tables referenced in the manuscript (Ding et al., 2020) (replied), the second researcher was contacted to clarify results (Huang et al., 2019) (did not reply) and a third author was contacted regarding result discrepancies within the manuscript (Kozlowski et al., 2013) (replied).
2.7Data synthesis
Due to heterogeneity of study methods, participant demographics, outcome measures and mTBI diagnostic criteria, data was not pooled for a meta-analysis. Demographics and results were summarized.
2.8Data availability
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
3Results
3.1Study selection
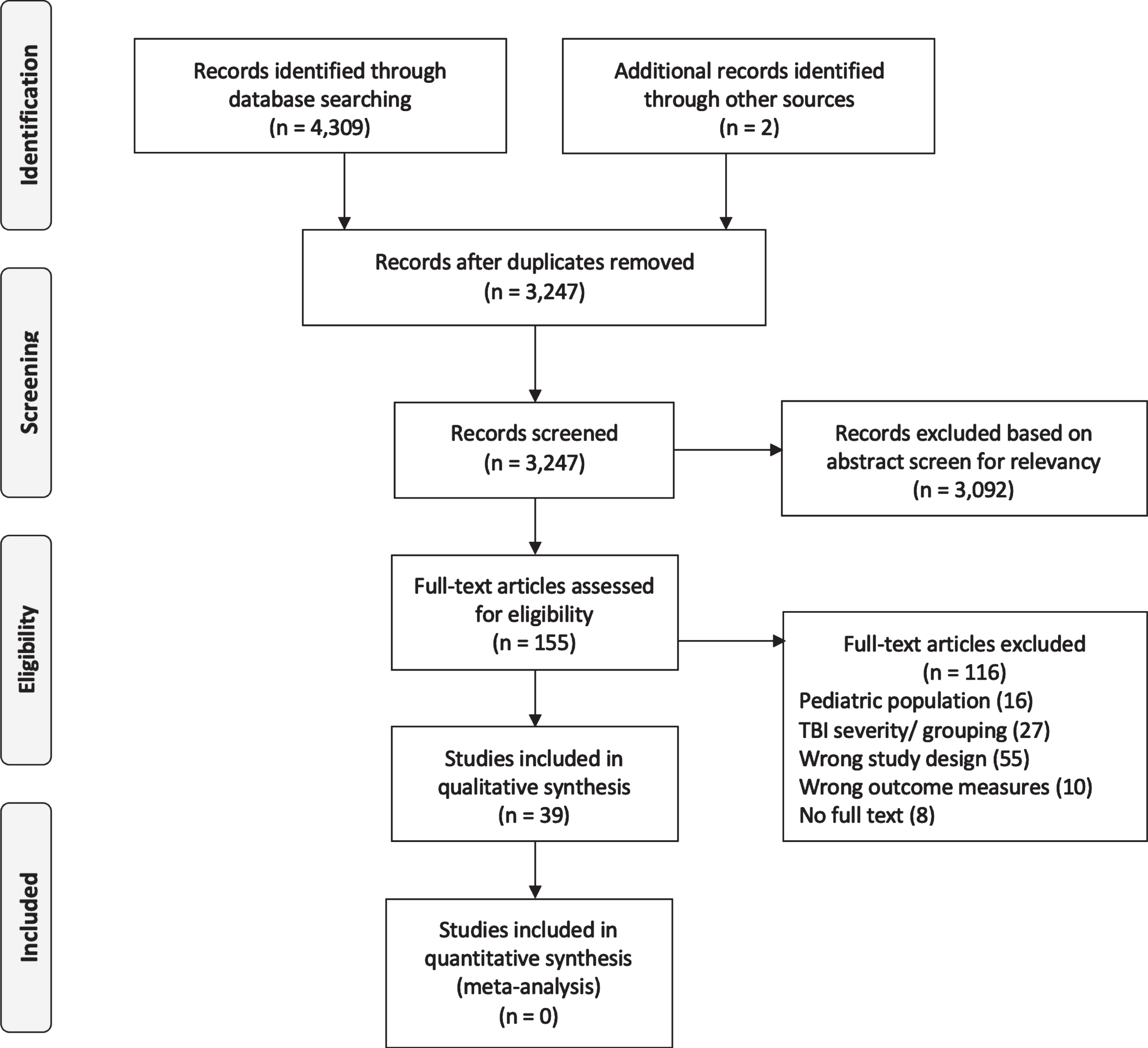
A total of 4,311 studies were identified, of which, 1,064 duplicates were removed. Two references were shared by a study author (Haider et al., 2020; Pyndiura et al., 2020). The title and abstract of 3,247 studies were screened with 3,092 not meeting inclusion criteria (see Table 2). Full texts of 155 studies were assessed for eligibility and 116 were excluded. The most common reason for exclusion was incorrect study design. Ultimately, 39 studies were included in the qualitative synthesis (see Fig. 1 for PRISMA flowchart).
Fig. 1
PRISMA flowchart.
3.2Study characteristics
Study characteristics, participant demographics and RoB grade are summarized in Table 3.
Table 3
Study characteristics
| Author (year) | Sample description age ± SD (% female) | mTBI diagnosis, classification | Time point(s) (time since injury mean ± SD [range]) | Test condition(s) (sample length analyzed) | Main finding(s) | RoB rating |
| Abaji (2016) | n = 12 mTBI asymptomatic athletes21yrs±1.1 (0%)n = 12 age/height/weight/sport/ education matched controls22yrs±1.6 (0%) | On field diagnosis by MD, verified by neuropsychologist, 3rd CISG consensus statement | 1 assessment (95d±63 [29–138]) | A) Rest, seated (3mins)B) Exertion, 30% IHGC (3mins)s | Significantly lower HFms2 and higher LF/HF in mTBI vs controls during IHGC. Significant correlations between time since injury and RMSSD, HFms2 and ApEN at rest. | Unacceptable |
| Bishop (2017) | n = 12 mTBI symptomatic athletes19.92yrs±3.06 (0%)n = 89 controls17.78yrs±2.33 (0%) | Diagnosis by athletic therapist, physiotherapist or MD | 1 assessment (2.17d±0.72)n = 3≤24hrsn = 5≤48hrsn = 4≤72hrs | A) Rest, seated (5mins)B) 10SS (x14, averaged) | Significantly lower pNN50 in mTBI vs controls during 10SS. Differences in HRSD and change-DBP/SBP/MAP in mTBI vs controls during squat/standing phases of 10SS. | Unacceptable |
| Brandt (2020) | n = 52 mTBI symptomatic28.96yrs±10.25 (37%)n = 32 age/sex matched controls29.59yrs±10.60 (50%) | Glasgow Coma Scale 13–15, LOC < 24hrs | 1) 3–14d (n = 50 mTBI, n = 27 controls)2) 1-3mos (n = 39 mTBI, n = 23 controls)3) 3–5mos (n = 37 mTBI, n = 24 controls) | Rest, seated (1min, x4) | Significant negative correlations between lnHF and depressive symptoms at 3-14d and 1-3mos post-mTBI. | Unacceptable |
| Clausen (2016) | n = 9 mTBI symptomatic (pre-intervention)23yrs±6 (100%)n = 13 age matched controls21yrs±3 (100%) athletes | World Health Organization criteria for post-concussion syndrome | 1) Pre-intervention (9wks [6–12])2) Post-12wk aerobic exercise intervention (n = 6 mTBI) | A) Exercise onset†B) Max exercise tolerance†(pre & post-intervention) | Significantly higher HR at exercise onset in mTBI vs controls pre and post-intervention. Significantly lower HR and SBP at max exercise tolerance in mTBI vs controls (exercise intolerance) pre-intervention. Significantly higher HR and SBP at max exercise tolerance in mTBI post vs pre-intervention. | Unacceptable |
| Ding (2020) | n = 13 symptomatic mTBI | American Congress of Rehabilitative Medicine criteria | 1 assessment | A) Rest, seated (5mins) | Significantly lower resting DBP-LF and higher sit-stand DBP-LF in TBI vs controls. | Acceptable |
| 45.7yrs±14.3 (62%)n = 13 age/sex/fitness level/BMI matched controls47.0yrs±14.7 (58%) | TBI (all severity): 18mos±16 [6–72] | B) Sit-stands (5mins) | ||||
| Dobney (2018) | n = 35 mTBI hx athletesn = 65 no mTBI hx athletesasymptomatic at time of testingFemales (all): 19.4yrs±1.7 (49%)Males (all): 20.8yrs±1.9 (51%) | Self-report | 1 assessment | A) Rest, supine (5mins)B) Exertion, max IHGC (x3, max score used) | Significantly higher resting DBP in those with hx of≥2 mTBI vs no hx of mTBI. | Unacceptable |
| Dobson (2017) | n = 12 mTBI20.1yrs±0.9 (50%)n = 11 sex/height/weight matched controls21.7yrs±0.9 (55%) | Diagnosed by athletic therapist, confirmed by MD, 4th CISG consensus statement | 1)≤48hrs2) 24h later3) 1wk4) 2wks | A) Rest, supine (30s)B) Forced breathing, supine (x8, averaged)C) Supine-to-standingD) VM | Significantly altered HR response to standing in mTBI at≤48hrs vs controls. Significantly higher resting SBP, altered SBP response to standing and longer VM 90% -SBP- normalization-time in mTBI at≤48hrs vs controls. | Unacceptable |
| Gall (2004) | n = 14 mTBI asymptomatic & symptomatic athletes18.1yrs±0.4 (0%)n = 14 age/height/weight/ position/play time matched controls18.8yrs±0.4 (0%) | Suspected mTBI by team trainers/game observers, Canadian Hockey Association concussion symptomatology | 1) 2d‡Resting (1.8d±0.2)n = 12≤48hrs, n = 2≤72hrsExertion (5.0±1.4d)n = 6≤72hrs, n = 8 > 72hrs2) 5d later (7d) | A) Rest, seated (5mins)B) Exertion, cycling (5mins) | Significantly higher HR, LFms2 & HFms2 during exertion in mTBI at both timepoints (2d/7d) vs controls. | Unacceptable |
| Haider (2020) | n = 9 mTBI asymptomatic athletes18.3yrs±2 (56%)n = 21 age/sex/height/weight matched controls16.7yrs±3 (33%) | Diagnosed by MD (or relevant clinician) | 1 assessment (median = 2yrs [1–8]) | A) Rest, supine (2mins)B) FC (3mins, analyzed as 1min segments) | Significantly lower resting HR in mTBI vs controls. At min 2 of FC, mTBI had greater % change in HR and lesser % change in RMSSD from rest vs controls. | Unacceptable |
| Hanna-Pladdy (2001) | a) n = 22 mTBI symptomatic22.77yrs±4.27 (68%)b) n = 22 mTBI asymptomatic23.87yrs±7.34 (41%)c) n = 22 control symptomatic20.41yrs±4.79 (82%)d) n = 22 control asymptomatic21.64yrs±3.01 (64%) | Self-report, criterion established by Kay (1993) | 1 assessment [≤1–15yrs] | A) Rest (pre-test), seated(2mins)B) Experimental conditions 4mins×4 of either: i) stress (n = 11 of each group a-d) OR ii) relaxation (n = 11 of each group a-d) before each block of neuropsychological testsC) Rest (post-test) | Significantly higher HR in mTBI during stress condition vs relaxation condition. Significantly higher HR in symptomatic mTBI group during stress vs groups b-d. | Unacceptable |
| Hilz (2011) | n = 20 mTBI asymptomatic37.0yrs±13.3 (15%)n = 20 controls25.6yrs±8.8 (25%) | World Health Organization criteria | 1 assessment (20mos±11 [5–43]) | A) Rest, supine (2mins)B) Supine-to-standingC) Rest, standing (2mins) | Significantly higher resting (supine) HR, SDNN, RMSSD, HFms2, HFnu, LFnu and LF/HF in mTBI vs controls. Significantly lower resting (standing) SDNN and LFms2 in mTBI vs controls. Significantly altered HR and HRV response to standing in mTBI vs controls. Significantly lower BRS at rest and in response to standing in mTBI vs controls. | Unacceptable |
| Hilz (2015) | n = 24 Mtbi33.7yrs±12.7 (29%)n = 27 controls30.1yrs±11.7 (37%) | World Health Organization criteria | 1 assessment (32.0mos±24.4 [5–86]) | A) Rest, seated (1min)B) EP, 30mmHg (1min) | Significant decrease in LFms2, LFnu and increase in HFnu with EP in controls, but not in mTBI. Significant increase in SBP and DBP with EP in mTBI, but not in controls. Significantly different SBP-LF and DBP-LF at rest and in response to EP in mTBI vs controls. | Unacceptable |
| Hilz (2016) | n = 25 mTBI asymptomatic35.0yrs±13.2 (28%)n = 29 controls31.2yrs±12.2 (31%) | World Health Organization criteria | 1 assessment (34mos±29 [4–98]) | A) Rest (1min)B) VM (x3, analyzed least abnormal response) | Significantly lower resting SDNN, RMSSD, LFms2, HFms2, TPms2, SBP-LF and BRS in mTBI vs controls. Significantly longer VM 90% -SBP-normalization-time in mTBI vs controls. | Unacceptable |
| Hilz (2017) | n = 20 mTBI33.1yrs±13.5 (30%)n = 20 controls29yrs±10 (30%) | mTBI diagnosed if≥1 of the following criteria applied:1) LOC < 30mins2) PTA < 24hrs3) depressed, basilar or linear skull fracture (dura intact) | 1 assessment (25.2mos±20.5 [6–78]) | A) Rest, supine (2mins)B) Supine-to-standing | Significantly higher resting LFnu in mTBI vs controls. Significantly lower resting HFnu in mTBI vs controls. | Unacceptable |
| Hilz (2020) | n = 17 mTBI37.1yrs±2.9 (29%)n = 17 controls29.9yrs±2.8 (41%) | World Health Organization criteria | 1 assessment (32.4mos±6.8 [4–98]) | A) Rest (1min)B) Pleasant odorous stimulation (102s)C) Unpleasant odorous stimulation (102s) | Significant decrease in SBP and DBP with pleasant odour stimulation in controls, but not in mTBI. Significant increase in HR with unpleasant odour stimulation in controls, but not in mTBI. | Unacceptable |
| Howard (2018) | n = 56 mTBI symptomatic 37.9yrs±10.7 (32%)n = 30 migraine non-mTBI40.9yrs±10.7 (77%)n = 36 controls37.6yrs±9.5 (47%) | All headache (mTBI & migraine) diagnosed by certified headache specialist using ICHD-3β criteria | 1 assessment (10.56yrs±8.7) | COMPASS-31 questionnaire | Significantly higher COMPASS-31 total scores in mTBI vs migraine and controls. Significantly higher orthostatic intolerance and bladder subscale scores in mTBI vs migraine. Significant correlation between number of lifetime mTBIs and total COMPASS-31 score in mTBI. | Acceptable |
| Huang (2019) | n = 23 mTBI symptomatic athletes 20yrs±1.5 (39%)n = 23 sex/sport matched controls20yrs±1.2 (35%) | Diagnosed by MD, National College Athletic Association diagnostic criteria | 1 assessment (4d±1) | A) Rest, seated (3mins)B) Cognitive task, 2-Back (3mins) | Significantly lower resting % HF in mTBI vs controls. During 2-Back, % HF increased significantly from rest in mTBI, but not in controls. | Unacceptable |
| Hutchison (2017) | n = 26 mTBI acutely symptomatic athletesn = 26 age/sex/sport/yr of study matched controlsAll: 21.0yrs±2.5 (38%) | Diagnosed by MD based on: 1) observed or reported acceleration/deceleration of the head AND 2) any observed alteration in mental status AND/OR 3) signs such as confusion, vacant stare, poor coordination, difficulty concentrating, poor balance AND/OR 4) any self-reported symptoms such as headache, LOC, nausea, balance problems, or difficulty reading/concentrating | 1) “symptomatic phase” [2-7d]2) “asymptomatic exercise progression phase” = initiation of exercise3) 1wk post-return-to-play clearance | Rest, seated (5mins x2, averaged) | Significantly lower SDNN and HFs2 in mTBI vs controls at all 3 timepoints. Significantly lower LFs2 in mTBI vs controls at initiation of exercise. Significantly higher LF/HF in mTBI vs controls 1wk post-return-to-play clearance. | Acceptable |
| Johnson (2018) | n = 11 mTBI symptomatic athletes 20yrs±2 (45%)n = 10 controls24yrs±4 (50%) | Glasgow Coma Scale = 15 | 1 assessment (≤10d) | A) Rest, supine (5mins)B) FC (3mins, analyzed as 1min segments) | Significantly altered HR, RMSSD and HFms2 response to FC in mTBI vs controls. Significantly greater increase in SBP, DBP and MAP in response to FC in controls vs mTBI. | Unacceptable |
| Johnson (2020) | n = 10 mTBI symptomatic athletes 20yrs±2 (50%)n = 10 controls24yrs±4 (50%) | Glasgow Coma Scale = 15 | 1 assessment (5d±2) | A) Pre-test rest, supine (5mins)B) CPT (120s, analyzed as 30s segments)C) Post-test rest, supine (60s) | Significantly blunted and delayed HR and BP response to CPT in mTBI vs controls. Cardiovascular response to CPT not correlated with symptom severity. | Acceptable |
| Kozlowski (2013) | n = 34 mTBI symptomatic27.5yrs±12.9 (50%)n = 22 age/sex/sport participation hx matched controls23.3yrs±6.2 (50%) | Diagnosed by MD, World Health Organization criteria for post-concussion syndrome | 1 assessment (226.2d±219.3) | A) RestB) Symptom-limited treadmill test | Significantly lower HRmax, SBPmax and higher DBPmax at termination of symptom-limited treadmill test (indicative of exercise intolerance) in mTBI vs controls. | Acceptable |
| La Fountaine (2009)§ | n = 3 mTBI symptomatic athletes19yrs±2 (33%)n = 3 age/sex/height/weight matched controls19yrs±2 (33%) | Suspected mTBI identified by sports medicine staff | 1)≤48hrs2) 2wks later | A) Rest, seated (5mins)B) Exertion, 30% IHGC (3mins) | Significantly lower ApEN during IHGC in mTBI vs controls at≤48hrs. Between group difference in ApEn did not persist 2wks later. | Unacceptable |
| La Fountaine (2011)§ | n = 3 mTBI acutely symptomatic athletes 19yrs±2 (33%)n = 3 age/sex/height/weight/ sport position matched controls19yrs±2 (33%) | Confirmed diagnosis by clinical staff, “accepted practices of concussion assessment” | 1) n = 1 2d, n = 1 3d, n = 1 5d2) 1wk later3) 2wks later | Rest, seated (60s) | Significantly higher QTVI in mTBI vs controls at 1st assessment. Between group difference in QTVI did not persist at follow-up assessments. | Unacceptable |
| La Fountaine (2016) | n = 10 mTBI athletes19.4yrs±1.1 (20%)n = 7 age/sex/height/weight matched controls20.0yrs±1.4 (14%) | Suspected mTBI identified by sports medicine staff, “accepted practices of concussion assessment” | 1)≤48hrs2) 1wk later | A) Rest, seated (5mins)B) Exertion, 30% IHGC (60s) | Significantly lower SysSlope¶ in mTBI vs controls at rest and with exertion at both timepoints. Significant correlation between resting pulse pressure and SysSlope¶. Return-to-play correlated with a couple arterial pulse contour variables. | Unacceptable |
| La Fountaine (2018) | n = 10 mTBI athletes19.6yrs±1.0 (0%)n = 10 demographically matched controls19.8yrs±1.0 (0%) | Clinically accepted practices of concussion assessment on-field and in subsequent office-based follow-up | 1)≤48hrs2) 1wk later | Rest, seated (5mins) | Significantly lower measures of BRS in mTBI vs controls; no group specific differences from≤48hrs to 1wk. | Unacceptable |
| Leddy (2010) | n = 12 mTBI symptomatic (pre-intervention) | Diagnosed by MD, World Health Organization criteria for post-concussion syndrome | 1) Pre exercise intervention (19wk [6–40]) | Symptom-limited treadmill test | Post progressive exercise intervention, participants able to exercise to significantly higher HRmax and SBPmax without symptom exacerbation on treadmill test. | Acceptable |
| 27.9yrs±14.3 (42%) | 2) 2-3wks later (also pre- exercise intervention) | |||||
| 3) every 3wks during exercise intervention until resolution of exercise intolerance (treadmill test) | ||||||
| (47.6d±31.8) | ||||||
| Liao (2016) | n = 165 mTBI40.08yrs±11.08 (61%)n = 82 controls30.71yrs±12.09 (71%) | Diagnosed by specialist based on: 1) LOC < 30mins; 2) PTA < 24hrs; Glasgow Coma Scale 13–15 upon acute medical evaluation; 3) negative-CT scan | 1)≤1wk (n = 165)2) 6wks (n = 97)3) 12wks (n = 46) | Rest (5mins, x2) | Significantly lower SDNN, lnLF, lnHF, lnTP, lnLF/HF in mTBI with and without anxiety vs controls at≤1wk. Significant increase in % LF from 6wks to 12wks in mTBI with anxiety. Significant negative correlations between anxiety scores and HRV at 6 and 12wks in mTBI with anxiety. | Unacceptable |
| Mirow (2016) | n = 61 mTBI symptomatic active-duty service members/veterans33.3yrs±7.5 (0%) | Ohio State University Traumatic Brain Injury Identification | 1 assessment (23.9mos±15.8 [4–60]) | A) Aerobic exercise (≥10mins)B) Rest, seated (5mins)C) Rest, standing (2mins)24hr recording, including:D) WakefulnessE) Sleep | 24hr recording:Abnormal SRT|| associated with decreased LF/HF, LFnu and increased HFnu.Sleep duration < 7hrs associated with decreased LF/HF and increased HFnu. | Unacceptable |
| Purkayastha (2019) | n = 31 mTBI acutely symptomatic athletes20yrs±1.5 (29%)n = 31 controls20yrs±1.2 (39%) | Diagnosed by MD, 5th CISG consensus statement | 1) 4d±1 (n = 31)2) 22d±4 (n = 28)3) 95d±11 (n = 21) | Rest, seated (6mins) | Significantly lower RMSSD, pNN50 & HFms2 in mTBI vs controls at 4d±1. Significant increase in RMSSD, pNN50 and HFms2 in mTBI across timepoints. | Acceptable |
| Pyndiura (2020) | n = 41 mTBI asymptomatic athletes median = 21yrs (51%)n = 72 control athletesmedian = 20.3yrs (57%) | Self-report | 1 assessment (median = 3.2yrs [3.5mos-13yrs]) | A) Rest, seated (5mins)B) Rest, supine (5mins) | Significantly higher seated HR in males mTBI vs male controls. | Unacceptable |
| Russell (2020) | n = 31 mTBI23.8yrs±4.6 (10%)n = 32 age/sex matched controls24.0yrs±4.8 (13%) | “Medically diagnosed” | 1 assessment (≤72hrs) | A) Seated (3mins)-to-standing (3mins)B) Supine on floor (3mins)-to-standing (3mins)C) Seated cross-legged (3mins)-to-standing (3mins) | Altered HR response during seated-to-standing and supine-to-standing postural changes in mTBI vs controls. | Unacceptable |
| Senthinathan (2017) | n = 11 mTBI acutely symptomatic athletes19.9yrs±0.8 (64%)n = 11 sex/sport matched controls20.3yrs±0.6 (64%) | Diagnosed by MD based on: (1) observed or reported acceleration/deceleration of the head AND (2) any observed alteration in mental status AND/OR (3) observed signs such as confusion, vacant stare, poor coordination, difficulty concentrating, poor balance, AND/OR (4) any self-reported symptoms such as headache, LOC, nausea, balance problems, or difficulty reading or concentrating | 1) “symptomatic phase” (4.7d±2.1 [2–7])2) “asymptomatic exercise progression phase” (18.1d±7.9 [8–30])3) 1wk post-return-to-play clearance (25.5d±8.2 [17–38]) | A) Rest, seated (5mins, x2)B) Rest, standing (5mins) | Lower resting standing SampEN in mTBI vs controls across timepoints. Changes in resting seated LFnu, HFnu and LF/HF across recovery timepoints. Significant negative correlation between number of previous mTBIs and seated SampEN at 4.7d. | Unacceptable |
| Solbakk (2005) | n = 20 symptomatic mTBI41.2yrs±11.2 (30%)n = 20 age/education matched controls36.4yrs±12.7 (35%) | American Congress of Rehabilitative Medicine criteria, uncomplicated mTBI# | 1 assessment(5.7yrs±3.1 [1–12]) | A) Pleasant visual stimulusB) Unpleasant visual stimulusC) Neutral visual stimulus | No group differences. | Acceptable |
| Sung (2016)a | n = 181 mTBI38.47yrs±9.27 (60%)n = 83 controls30.52yrs±10.44 | Diagnosed by specialists, Glasgow Coma Scale 13–15 & negative head CT | ≤1wk2) 1.5mos3) 3mos4) 6mos5) 12mos6) 18mos | Rest, seated (5mins, 2x) | Significantly lower SDNN, lnLF, lnHF, lnLF/HF in mTBI vs controls at≤1wk. Significantly higher % HF mTBI vs controls at≤1wk. In female mTBI participants, % HF and LF/HF at≤1wk were significantly correlated with BDI at 18mos. | Unacceptable |
| Sung (2016)b | n = 331 mTBImedian = 40.0yrs (60%)n = 152 controlsmedian = 27.5yrs (69%) | Diagnosed by specialists, Glasgow Coma Scale 13–15 & negative head CT | 1)≤1wk2) 6wks (n = 125) | Rest (5mins, 2x) | Significantly lower SDNN, lnLF, lnLF/HF lnTP and higher % HF in mTBI vs controls at≤1wk and 6wks. Correlations between anxiety and depression scores at 6wks with frequency-domain measures at≤1wk. | Unacceptable |
| Tan (2009) | n = 11 mTBI veterans with PTSD [22–49yrs] (0%)n = 240 normative control data†† | Diagnosis obtained via review of comprehensive evaluation by the polytrauma centre | 1 assessment | Rest, seated (10mins) | Significantly lower SDNN in the study participants (with mTBI, PTSD and/or pain) vs normative control data. | Unacceptable |
| Truong (2016) | n = 32 symptomatic mTBI37yrs±11n = 40 controls33yrs±12 | Diagnosed by licensed practitioners using standard clinical criteria | 1 assessment (>45d) | Pupillary light reflex to (each x3, averaged):A) Dim white pulseB) Bright white pulseC) Dim white stepD) Bright white stepE) Bright red stepF) Bright blue step | For conditions A-F, 5–8 (out of 9) pupillary parameters were statistically different in mTBI vs controls. | Acceptable |
| Wright (2017)‡‡ | n = 18 (acute) mTBI athletes | Diagnosed by MD, 4th CISG consensus statement | Acute mTBI group: | A) Rest, seated (1min) | No difference in resting HR or MAP between hx3 + vs hx-. No difference in HR or MAP in the acute mTBI group across timepoints. | Acceptable |
| 18.6yrs±1.5 (0%) | 1) Pre-season | B) visual stimulation paradigm | ||||
| n = 31 hx≥3 mTBI athletes (hx3 +) | 2)≤72hrs3) 2wks4) 1mo (n = 16) | |||||
| 19.7yrs±1.7 (0%) | ||||||
| n = 42 no mTBI hx athletes (hx-) | ||||||
| 18.9yrs±2.1 (0%) | ||||||
| Wright (2018)‡‡ | n = 18 (acute) mTBI athletes | Diagnosed by MD, 4th CISG consensus statement | Acute mTBI group: | C) Rest, standing (5mins) | No difference in resting HR or MAP between hx3 + vs hx-. No difference in HR or MAP in the acute mTBI group across timepoints. | Acceptable |
| 18.6yrs±1.5 (0%) | 1) Pre-season | D) 5SS (5mins) | ||||
| n = 31 hx≥3 mTBI athletes (hx3 +) 19.6yrs±1.9 (0%)n = 42 no mTBI hx athletes (hx-) 19.0yrs±1.4 (0%) | 2)≤72hrs3) 2wks4) 1mo (n = 16) | E) 10SS (5mins) |
Abbreviations: ApEN, approximate entropy; BDI, Beck Depression Inventory; BMI, body mass index; BP, blood pressure; BRS, baroreceptor sensitivity; CISG, Concussion in Sport Group; COMPASS-31 = Composite Autonomic Symptom Score 31 questionnaire; CPT, cold pressor test; CT, computerized tomography; DBP, diastolic blood pressure; EP, eyeball pressure stimulation; FC, face cooling; HF = high frequency; HR, heart rate; HRV, heart rate variability; HRSD, standard deviation of heart rate; hx = history; hx-, no mTBI history; hx3 +, history of≥3 mTBI; ICHD, International Classification of Headache Disorders; IHGC, isometric hand grip contraction; LF, low frequency; LOC, loss of consciousness; MAP, mean arterial pressure; MD, medical doctor; mTBI, mild traumatic brain injury; pNN50, percentage of successive RR intervals that differ by more than 50ms; PTA, post-traumatic amnesia; PTSD, Post-traumatic stress disorder; QVTI, QT interval variability index; RMSSD, root mean square of successive RR interval differences; RoB, risk of bias; SampEN, sample entropy; SBP, systolic blood pressure; SDNN, standard deviation of NN intervals; SysSlope, systolic slope; SRT, Sharpened Romberg Test (balance test); TBI, traumatic brain injury; TP, total power; VM, valsalva maneuver; 5SS, 5s squat, 5s stand (squat-stands); 10SS, 10s squat, 10s stand (squat-stands). †using progressive treadmill test. ‡exercise testing only completed if symptoms had returned to baseline; if participants were symptomatic follow-ups were conducted at 3d intervals until athlete was asymptomatic at rest/had returned to play. §data from same cohort. ¶systolic slope (SysSlope) of the arterial pressure wave = calculated from the rate of rise (change in pressure divided by change in time) of the systolic upstroke. ||abnormal SRT = loss of balance in first 30s of 60s balance test (across 4 trials) where participant stands with one foot in front of the other, toes touching heel, arms crossed across chest, eyes closed. #uncomplicated mTBI (meeting diagnostic criteria for mTBI with no intercranial lesions on conventional imaging). ††normative data was taken from the average of each 5min segment over 24hrs recorded from 240 controls; mTBI did not follow the same experimental procedure. ‡‡data from same cohort. aSung et al. Psychophysiol. bSung et al. Clin Neuropsychol.
3.2.1Methods
Of the included studies, twenty-one were cross-sectional studies (Abaji et al., 2016; Bishop et al., 2017; Ding et al., 2020; Dobney et al., 2018; Hanna-Pladdy et al. 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Howard et al., 2018; Huang et al., 2019; Johnson et al., 2018, 2020; Kozlowski et al., 2013; Mirow et al., 2016; Pyndiura et al., 2020; Russell et al., 2020; Solbakk et al., 2005; Tan et al., 2009; Truong & Ciuffredia, 2016), sixteen were cohort studies (Brandt et al., 2020; Clausen et al., 2016; Dobson et al., 2017; Gall et al., 2004; La Fountaine et al., 2009, 2011, 2016, 2018; Leddy et al., 2010; Liao et al., 2016; Purkayastha, Williams et al., 2019; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Wright et al. 2017, 2018), while two (Haider et al., 2020; Hutchison et al., 2017) were case-control studies. All studies, with the exception of two (Leddy et al., 2010; Mirow et al., 2016), compared mTBI participants to non-head injured control groups. Of these, five studies (Gall et al., 2004; Huang et al., 2019; Hutchison et al., 2017; Purkayastha, Williams et al., 2019; Senthinathan et al., 2017) included controls with a remote history of mTBI, three studies (Johnson et al., 2018, 2020; Russell et al., 2020) included controls with no mTBI in the last twelve months and eleven studies (Bishop et al., 2017; Clausen et al., 2016; Hilz et al., 2011, 2015, 2016, 2017, 2020; La Fountaine et al., 2016, 2018; Solbakk et al., 2005; Tan et al., 2009) did not report on mTBI history of the control group. Two studies (Wright et al., 2017, 2018) compared post-mTBI values to preseason values.
In twenty-two studies (Abaji et al., 2016; Bishop et al., 2017; Ding et al., 2020; Dobney et al., 2018; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Howard et al., 2018; Huang et al., 2019; Johnson et al., 2018, 2020; Kozlowski et al., 2013; Mirow et al., 2016; Pyndiura et al., 2020; Russell et al., 2020; Solbakk et al., 2005; Tan et al., 2009; Truong & Ciuffreda, 2016) autonomic dysfunction was assessed at a single timepoint (within 72 hours to greater than 10 years post-injury). Multiple assessments were conducted in fifteen studies (Brandt et al., 2020; Dobson et al., 2017; Gall et al., 2004; Hutchison et al., 2017; La Fountaine et al., 2009, 2011, 2016, 2018; Liao et al., 2016; Purkayastha, Williams, et al., 2019; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sung, Lee, et al., 2016; Wright et al., 2017, 2018). Timing of follow up assessments ranged from within 24 hours up to 18 months after the initial assessment. Outcomes were assessed pre and post sub-symptom threshold exercise interventions in two studies (Clausen et al., 2016; Leddy et al., 2010).
3.2.2Demographics
A total of 1,467 mTBI participants were included in the thirty-nine studies. The number of mTBI participants across studies ranged from 3 –331 with a mean of 38±60. Eight studies included only male participants (Abaji et al., 2016; Bishop et al., 2017; Gall et al., 2004; La Fountaine et al., 2018; Mirow et al., 2016; Tan et al., 2009; Wright et al., 2017; Wright et al., 2018) and in most studies reporting sex/gender, males outnumbered female mTBI participants with a male:female ratio of 768:619. Two studies (Dobney et al., 2018; Truong & Ciuffreda, 2016) did not specify the sex/gender of mTBI participants; one study (Truong & Ciuffreda, 2016) did not include sex/gender of mTBI or control participants. Twenty studies (Brandt et al., 2020; Dobney et al., 2018; Dobson et al., 2017; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017; Howard et al., 2018; Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2018, 2020; Kozlowski et al., 2013; La Fountaine et al., 2011; Pyndiura et al. 2020; Russell et al., 2020; Senthinathan et al., 2017; Solbakk et al., 2005) reported participant sex and eight studies (Hilz et al., 2020; La Fountaine et al., 2009, 2016; Leddy et al., 2010; Liao et al., 2016; Mirow et al., 2016; Sung, Chen et al., 2016; Tan et al., 2009) reported participant gender. Ten studies (Abaji et al., 2016; Bishop et al., 2017; Clausen et al., 2016; Ding et al., 2020; Gall et al., 2004; La Fountaine et al., 2018; Purkayastha, Williams et al., 2019; Sung, Lee et al., 2016; Wright et al., 2017, 2018) did not specify if reporting sex/gender or interchanged the two terms.
The mean age of mTBI participants across studies ranged from 18.1 to 41.2 years with an overall mean age of 26.9±8.4 years. Hutchison et al. (Hutchison et al., 2017) did not separately report the age of mTBI and control groups, stating that the average age was not significantly different.
3.2.3Injury characteristics
Time since injury varied greatly between studies, with nine studies assessing participants within 72 hours of mTBI (Bishop et al., 2017; Dobson et al., 2017; Gall et al., 2004; La Fountaine et al. 2009, 2016, 2018; Russell et al., 2020; Wright et al., 2017, 2018), nine studies within the first 7 days (Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2020; La Fountaine et al., 2011; Liao et al., 2016; Purkayastha, Williams et al., 2019; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sung, Lee et al., 2016), one within 10 days (Johnson et al., 2018), one within the first 2 weeks (Brandt et al., 2020) and the remaining seventeen studies (Abaji et al., 2016; Clausen et al., 2016; Ding et al., 2020; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Howard et al., 2018; Kozlowski et al., 2013; Leddy et al., 2010; Mirow et al., 2016; Pyndiura et al., 2020; Solbakk et al., 2005; Truong & Ciuffreda, 2016) ranged from > 45 days to > 10 years post-mTBI. Time since injury was not reported in two studies (Dobney et al., 2018; Tan et al., 2009).
Seventeen studies (Abaji et al., 2016; Bishop et al., 2017; Dobson et al., 2017; Gall et al., 2004; Haider et al., 2020; Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2018, 2020; La Fountaine et al., 2009, 2011, 2016, 2018; Purkayastha, Williams et al., 2019; Senthinathan et al., 2017; Wright et al., 2017, 2018) included participants with sport-related mTBI, thirteen (Brandt et al., 2020; Ding et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2015, 2020; Howard et al., 2018; Kozlowski et al., 2013; Leddy et al., 2010; Liao et al., 2016; Solbakk et al., 2005; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Truong & Ciuffreda, 2016) included multiple mechanisms of injury for mTBI (including motor vehicle collision, fall, sport-related, assault or other) and three studies with participants who sustained an mTBI in military combat (Mirow et al., 2016; Russell et al., 2020; Tan et al., 2009). Six studies (Clausen et al., 2016; Dobney et al., 2018; Hilz et al., 2011, 2016, 2017; Pyndiura et al., 2020) did not specify the mechanism of injury.
Fourteen studies (Bishop et al., 2017; Brandt et al., 2020; Clausen et al., 2016; Ding et al., 2020; Gall et al., 2004; Howard et al., 2018; Hung et al., 2019; Johnson et al., 2018, 2020; Kozlowski et al., 2013; La Fountaine et al., 2009; Mirow et al., 2016; Solbakk et al., 2005; Truong & Ciuffreda, 2016) included mTBI participants who were symptomatic. Participants (mTBI) were asymptomatic in six studies (Abaji et al., 2016; Dobney et al., 2018; Haider et al., 2020; Hilz et al., 2011, 2016; Pyndiura et al., 2020). One study (Hanna-Pladdy et al., 2001) compared symptomatic and asymptomatic mTBI participants and seven studies followed participants across timepoints (acutely symptomatic until they were asymptomatic) (Leddy et al., 2010; Hutchison et al., 2017; La Fountaine et al., 2011; Purkayastha, Williams et al., 2019; Senthinathan et al., 2017; Wright et al., 2017, 2018). Eleven studies did not report on symptoms at the time of assessment(s) (Dobson et al., 2017; Hilz et al., 2015, 2017, 2020; La Fountaine et al., 2016, 2018; Liao et al., 2016; Russell et al., 2020; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Tan et al., 2009).
3.2.4Assessment of autonomic dysfunction
Autonomic dysfunction was assessed at rest in thirty-four of the thirty-nine studies (Abaji et al., 2016; Bishop et al., 2017; Brandt et al., 2020; Ding et al., 2020; Dobney et al., 2018; Dobson et al., 2017; Gall et al., 2004; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2018, 2020; Kozlowski et al., 2013; La Fountaine et al., 2009, 2011, 2016, 2018; Liao et al., 2016; Mirow et al., 2016; Purkayastha, Williams et al., 2019; Pyndiura et al., 2020; Russell et al., 2020; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Tan et al., 2009; Wright et al., 2017, 2018). Nine studies assessed autonomic dysfunction during a postural change (sit-to-stand, squat-stands) (Bishop et al., 2017; Ding et al., 2020; Dobson et al., 2017; Hilz et al., 2011, 2017; Pyndiura et al., 2020; Russell et al., 2020; Senthinathan et al., 2017; Wright et al., 2018), eight studies during exertion (treadmill, cycling or isometric hand grip contraction) (Abaji et al., 2016; Clausen et al., 2016; Dobney et al., 2018; Gall et al., 2004; Kozlowski et al., 2013; La Fountaine et al., 2009, 2016, Leddy et al., 2010), two studies during Valsalva maneuver (Dobson et al., 2017; Hilz et al., 2016), two studies during face cooling (Haider et al., 2020; Johnson et al., 2018) and two studies during a cognitive task (Hanna-Pladdy et al., 2001; Huang et al., 2019). Other conditions included eyeball pressure (Hilz et al., 2015), olfactory (Hilz et al., 2020) or visual (Solbakk et al., 2005) stimuli, cold pressor test (Johnson et al., 2020) and various light conditions (for assessment of pupillary light reflex) (Truong & Ciuffreda, 2016).
The most commonly assessed measures of autonomic function were: HR (assessed in 87% of studies (Abaji et al., 2016; Bishop et al., 2017; Clausen et al., 2016; Ding et al., 2020; Dobney et al., 2018; Dobson et al., 2017; Gall et al., 2004; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2018, 2020; Kozlowski et al., 2013; La Fountaine et al., 2009, 2011, 2016, 2018; Leddy et al., 2010; Liao et al., 2016; Purkayastha, Williams et al., 2019; Pyndiura et al., 2020; Russell et al., 2020; Senthinathan et al., 2017; Solbakk et al., 2005; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Wright et al., 2017, 2018), frequency-domain HRV metrics (62% of studies (Abaji et al., 2016; Bishop et al., 2017; Brandt et al., 2020; Ding et al., 2020; Dobney et al., 2018; Gall et al., 2004; Haider et al., 2020; Hilz et al., 2011, 2015, 2016, 2017; Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2018; La Fountaine et al., 2009, 2018; Liao et al., 2016; Mirow et al., 2016; Purkayastha, Williams et al., 2019; Pyndiura et al., 2020; Russell et al., 2020; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sun, Lee et al., 2016)), BP (49% of studies (Bishop et al., 2017; Clausen et al., 2016; Ding et al., 2020; Dobney et al., 2018; Dobson et al., 2017; Hilz et al., 2011, 2015, 2016, 2017, 2020; Johnson et al., 2018, 2020; Kozlowski et al., 2013; La Fountaine et al., 2016, 2018; Leddy et al., 2010; Purkayastha, Williams et al., 2019; Wright et al., 2017, 2018)), time-domain HRV metrics (46% of studies (Abaji et al., 2016; Bishop et al., 2017; Dobney et al., 2018; Gall et al., 2004; Haider et al., 2020; Hilz et al., 2011, 2016, 2017; Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2018; La Fountaine et al., 2011; Liao et al., 2016; Purkayastha, Williams et al., 2019; Pyndiura et al., 2020; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Tan et al., 2009)), BRS (15% of studies (Ding et al., 2020; Dobson et al., 2017; Hilz et al., 2011, 2016, 2017; La Fountaine et al., 2018)), non-linear HRV metrics (10% of studies (Abaji et al., 2016; Bishop et al., 2017; La Fountaine et al., 2009; Senthinathan et al., 2017)) and frequency-domain BP variability (10% of studies (Ding et al., 2020; Hilz et al., 2015, 2016, 2017)). Other metrics included measures of pupillary reflex (Truong & Ciuffreda, 2016) and BP and/or HR response to Valsalva maneuver (Dobson et al., 2017; Hilz et al., 2016). The Composite Autonomic Symptom Score 31 (COMPASS-31) questionnaire was evaluated in one study (Howard et al., 2018).
3.4Risk of Bias (RoB)
Results from the RoB assessment are reported in Table 4. No studies were graded as “high-quality”. Eleven studies (Ding et al., 2020; Howard et al., 2018; Hutchison et al., 2017; Johnson et al., 2020; Kozlowski et al., 2013; Leddy et al., 2010; Purkayastha, Williams et al., 2019; Solbakk et al., 2005; Truong & Ciuffreda, 2016; Wright et al., 2017, 2018) were of “acceptable” quality, with the remaining 28 (Abaji et al., 2016; Bishop et al., 2017; Brandt et al., 2020; Clausen et al., 2016; Dobney et al., 2018; Dobson et al., 2017; Gall et al., 2004; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Huang et al., 2019; Johnson et al., 2018; La Fountaine et al., 2009, 2011, 2016, 2018; Liao et al., 2016; Mirow et al., 2016; Pyndiura et al., 2020; Russell et al., 2020; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Tan et al., 2009) graded as “unacceptable”. The most common biases were a lack of reporting how many people were approached to participate, not specifying whether the assessment of outcomes was blinded to exposure status, unreliable measures of exposure (not specifying mTBI diagnostic criteria), unreliable or invalid outcome measure and not addressing potential confounders.
Table 4
Risk of bias of included studies
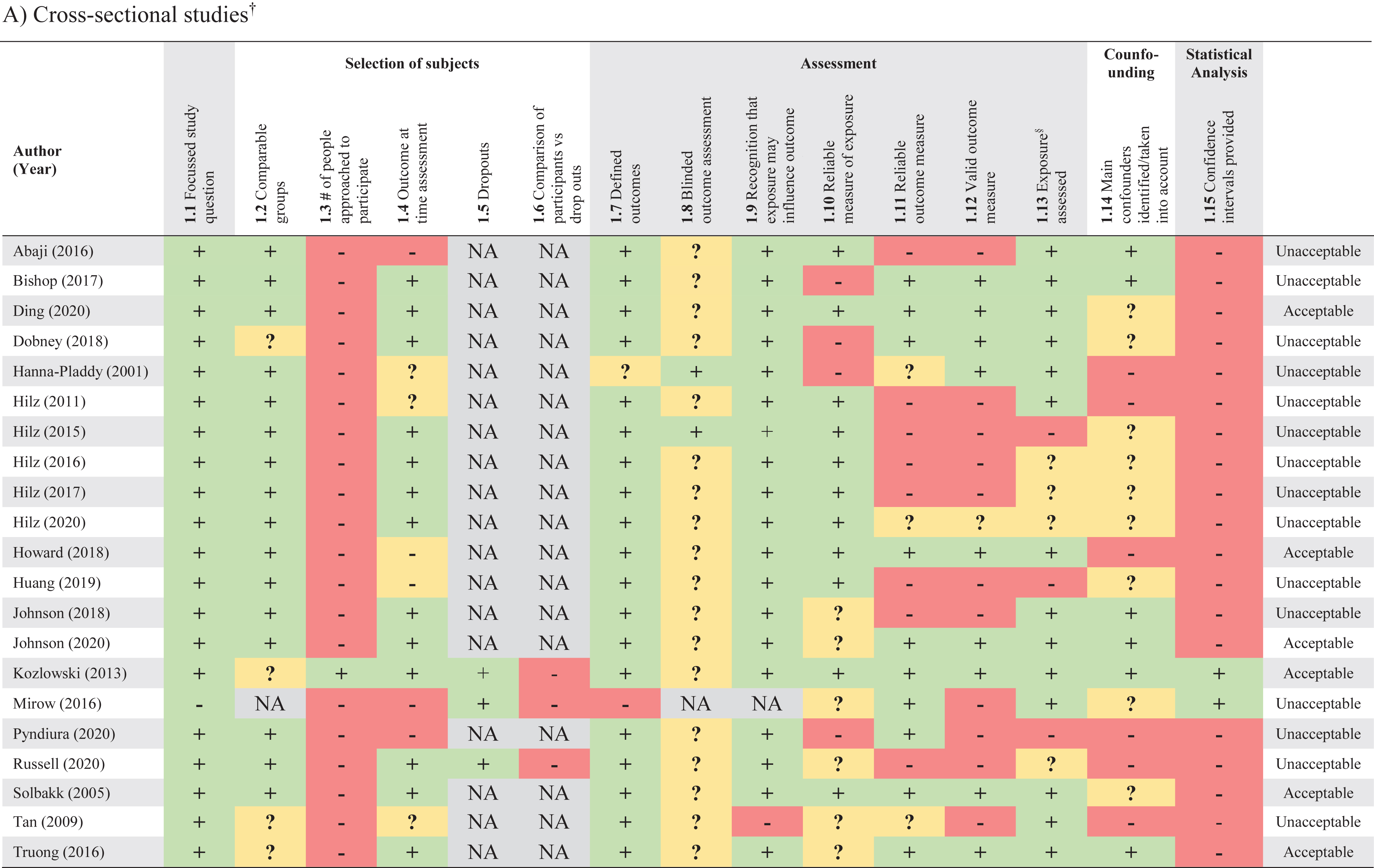 |
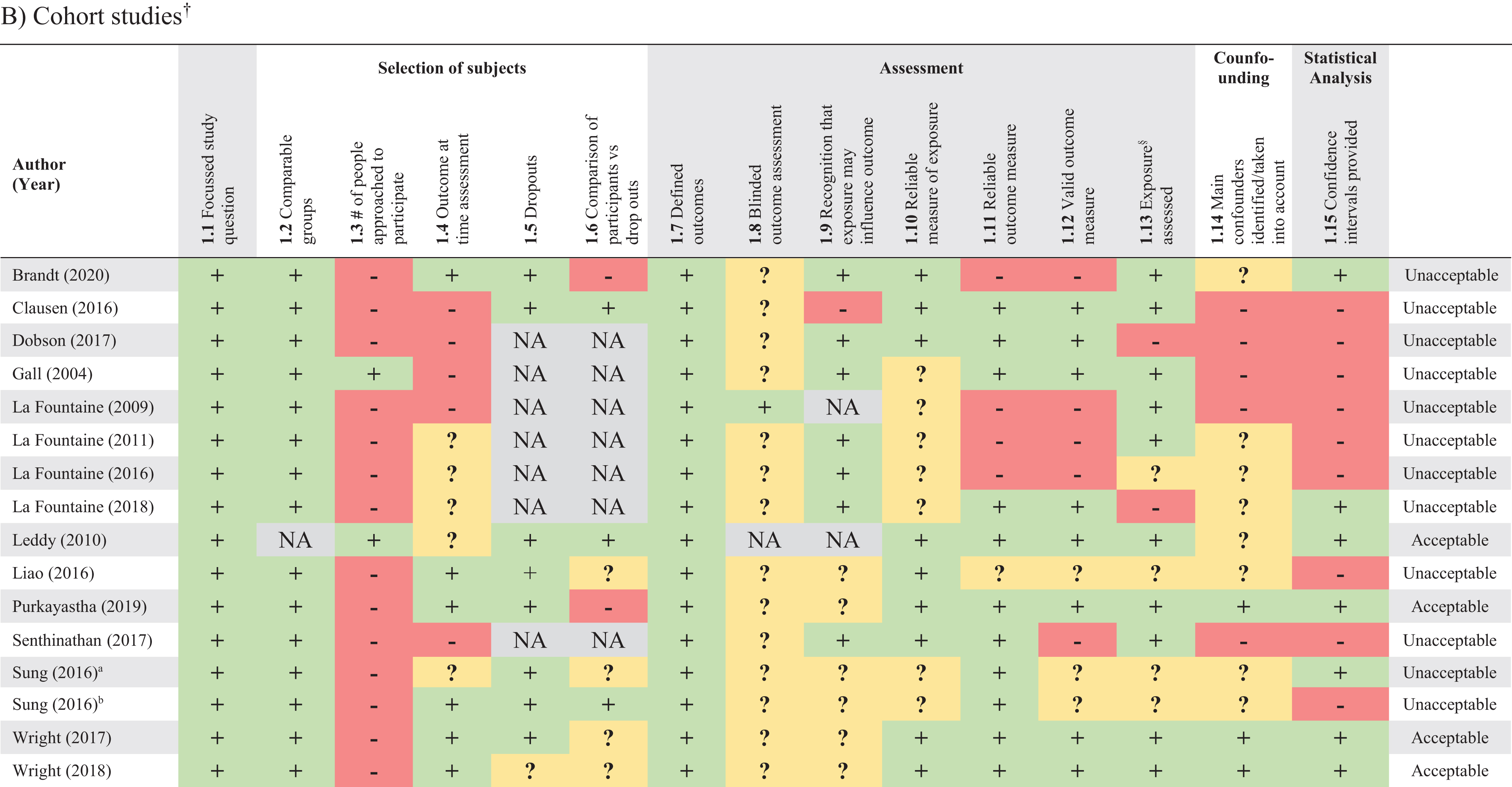 |
 |
Table 5
Reporting of study confounders
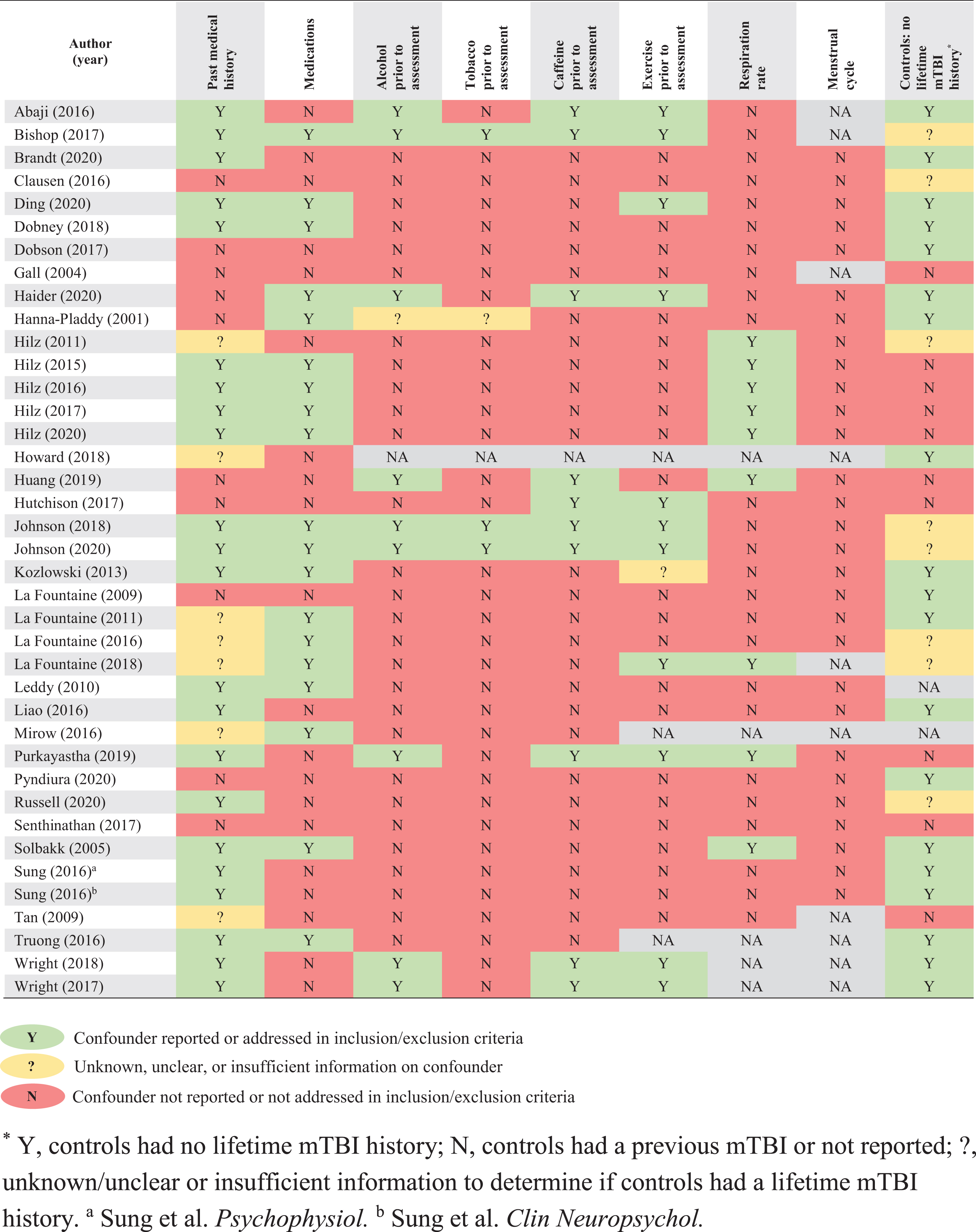 |
3.5Study results
A summary of HR and HRV results is presented in Supplementary Table 6. BP, BRS and alternative autonomic measure results are presented in Supplementary Table 7. Correlations between metrics of autonomic function and injury characteristics are reported in Supplementary Table 7.
4Discussion
In recent years, measurement of physiologic biomarkers of recovery has become a growing field of mTBI research. This includes assessment of autonomic parameters, such as those included in this review. These measures have the potential to provide an objective assessment of recovery and are therefore of great interest and clinically relevant. Evaluation of measures of autonomic function may prove especially important in the context of return-to-sport and allow for evaluation of physiologic changes that may persist beyond symptom resolution. The presented results offer evidence that a degree of ANS dysfunction may occur as a sequela of mTBI in adult cohorts. However, interpretation of the data is complicated by the wide variety of methods, reported variables, timepoints and patient populations evaluated in the literature. Additionally, significant covariates that may potentially confound or modify the relationship between mTBI and measures of autonomic function were not evaluated in many cases. While there is a paucity of literature with low RoB, the key findings of this review can be summarized as follows:
1. It remains unclear if there are differences in HR, HRV and BP parameters at rest (in a range of supine, seated and upright quiet-stance postures) in individuals with mTBI compared to controls, although most studies suggest there is not a significant difference at rest.
2. A wide range of (non-rest) test conditions have revealed differences in measures of autonomic function between mTBI and control groups. These include exertional tasks (i.e., isometric hand grip, squat-stands, submaximal treadmill test), Valsalva maneuver, face cooling, eyeball pressure, postural change (i.e., supine-to-stand, sit-to-stand), odorous and visual stimulations, cold pressor test, and measures of pupillary light reflex, among others. Findings suggest employing a physiologic challenge may be needed to reveal subtle differences in autonomic function between groups with and without a history of mTBI. A wide range of timepoints have been assessed in the literature from within 48 hours of injury (Dobson et al., 2017; La Fountaine et al., 2009, 2016, 2018) up to 15 years post-injury (Hanna-Pladdy et al., 2001). The majority of studies included were cross-sectional (one timepoint) (Abaji et al., 2016; Bishop et al., 2017; Ding et al., 2020; Dobney et al., 2018; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Howard et al., 2018; Huang et al., 2019; Johnson et al., 2018, 2020; Kozlowski et al., 2013; Mirow et al., 2016; Pyndiura et al., 2020; Russell et al., 2020; Solbakk et al., 2005; Tan et al., 2009; Truong & Ciuffreda, 2016), whereas others assessed temporal changes (Brandt et al., 2020; Clausen et al., 2016; Dobson et al., 2017; Gall et al., 2004; Hutchison et al., 2017; La Fountaine et al., 2009, 2011, 2016, 2018; Leddy et al., 2010; Liao et al., 2016; Purkayastha, Williams et al., 2019; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Wright et al., 2017, 2018). While several studies found that between group differences noted acutely post-injury resolved at repeat assessments (Dobson et al., 2017; La Fountaine et al., 2009, 2011; Purkayastha,Williams et al., 2019), other studies evaluating one timepoint in the post-acute/chronic stage reported between group differences.
3. The relationship between ANS dysfunction and post-concussive symptoms requires further investigation. None of the papers included in this review had a primary aim of characterizing this relationship. Especially in studies completing a single assessment in the post-acute/chronic stage of injury, clear symptom reporting is necessary to understand generalizability. Whether the participants (or a subset of the participants) are experiencing persistent symptoms should be described.
4.1Proposed pathophysiology of ANS dysfunction following mTBI
The underlying pathophysiology of ANS dysfunction following mTBI remains unknown; however, pre-clinical and clinical work suggests this may result from either: a) diffuse axonal injury during the initial mTBI; b) reduced cerebral blood flow (CBF) post-mTBI or c) deconditioning following the injury. Higher brain centers, including the prefrontal cortex (anterior cingulate, insula, orbitofrontal and ventromedial cortex) and limbic structures (amygdala and hypothalamus) in addition to structures in the brainstem (periaqueductal gray matter, nucleus ambiguus, medulla) form the Central Autonomic Network (Benarroch, 1993). Therefore, depending on the mechanism of injury and whether there is diffuse axonal injury, centers responsible for control of HRV and other autonomic function may be affected.
CBF has been increasingly investigated following mTBI using a variety of methods including transcranial Doppler and arterial spin labeling (ASL) magnetic resonance imaging (MRI). Using ASL MRI, Meier et al. reported reduced CBF in the right dorsal midinsular cortex in collegiate athletes in the first week post-mTBI compared to controls (Meier et al., 2015). This difference did not persist at 1 month post-injury. Athletes who were slow to recover, characterized as > 14 days until clearance for return-to-play, had lower CBF in the right dorsal midinsular cortex at 1 month compared to those with a more rapid return-to-play (Meier et al., 2015). As noted previously, the insula is part of the Central Autonomic Network, playing an important role in cardiovascular ANS integration (Bernaroch, 1993). While preliminary studies suggest a link between ANS dysfunction and altered CBF, additional research is required.
Following mTBI, individuals may struggle with exercise intolerance (Kozlowski et al., 2013), characterized as the inability to return to one’s premorbid level of physical activity due to symptom exacerbation. This may result in reduced physical activity and subsequent deconditioning. It is understood that deconditioning (reduced exercise capacity) is associated with decreased parasympathetic (vagal) activity and thus an increased resting HR (Gourine & Ackland, 2019). For example, improvement in HR recovery, a measure of parasympathetic activity, observed following a 6 week training program is lost after only 4 weeks of detraining (Heffernan et al., 2007). Individuals with higher resting HR and lower percentage predicted peak oxygen-consumption have been shown to have slower HR recovery following exercise, a measure of an individual’s ability to recruit cardiac vagal tone (Machhada et al., 2017). Unfortunately, the link between physical activity/exercise intolerance and ANS function in individuals with mTBI has not been well characterized.
4.2Methodological confounders
Previous reviews of the ANS literature (Blake et al., 2016; Callaway & Kosofsky, 2019; Pertab et al., 2018; Purkayastha, Stokes et al., 2019) have failed to highlight the significant methodological confounders present in the included studies. Additionally, several of these were not systematic reviews, nor did they include participants will all mechanisms of injury. The current systematic review addressed these gaps in the literature by including a wide range of autonomic function outcomes, completing an RoB assessment, providing a comprehensive review of confounders for each study and outlining future directions for ANS research in the mTBI context. Advances in this field will be dependent on the methodological rigor of future research. Additional studies addressing confounders and with larger sample sizes will be necessary before these measures may be implemented clinically.
Providing a clear operational definition of the “exposure” (mTBI) is important for reproducibility and generalizability of the research. In the case of eighteen studies reliability and/or validity of exposure status could not be ascertained based on presented information. Future research should use accepted mTBI diagnostic criteria, such as the American Congress of Rehabilitation Medicine criteria (Mild Traumatic Brain Injury Committee of the Head Injury Interdisciplinary Special Interest Group, 1993) or the 5th Consensus statement on concussion in sport criteria (McCrory et al., 2017). Injury was self-reported with no additional detail or diagnostic criteria in three studies (Dobney et al., 2018; Hanna-Pladdy et al., 2001; Pyndiura et al., 2020). It is also important to consider the mTBI history of control groups. In eighteen studies (Bishop et al., 2017; Clausen et al., 2016; Gall et al., 2004; Hilz et al., 2011, 2015, 2016, 2017, 2020; Huang et al., 2019; Hutchison et al., 2017; Johnson et al., 2018, 2020; La Fountaine et al., 2016, 2018; Purkayastha, Williams et al., 2019; Russell et al., 2020; Senthinathan et al., 2017; Tan et al., 2009), controls either did have a reported lifetime history of mTBI or insufficient information on mTBI history was presented. Particularly when evaluating ANS function in the chronic stage of injury or in those with persistent symptoms, control groups should be free of lifetime mTBI history to confidently report between group differences.
Many studies failed to report confounders that could affect ANS function data, such as other medical conditions, medications, cardiorespiratory fitness and stage of menstrual cycle. Despite many of the cohorts being youth athletes, screening and recording of past medical history and medications affecting ANS function should be clearly reported. Unfortunately, this was not the case in many studies and limits the internal validity. This is especially important in older cohorts and those with persistent symptoms where polypharmacy may be more likely. Cardiorespiratory fitness should also be considered, especially in athletic populations. As mentioned previously, resting HR, and thus HRV (Monfredi et al., 2014), is affected by cardiorespiratory fitness (Machhada et al., 2017). Several studies (Clausen et al., 2016; Haider et al., 2020; Johnson et al., 2018) compared varsity athletes to recreational control athletes. In the absence of either a direct or indirect index of cardiorespiratory fitness completed by both groups, interpretation of study findings is limited. Those with higher cardiorespiratory fitness, and thus lower resting HR, would have higher time-domain HRV measures, compared to their peers with lower cardiorespiratory fitness, thus producing between group differences unrelated to mTBI history. Stage of the menstrual cycle is also known to affect HRV. A recent meta-analysis highlighted fluctuation in cardiac vagal activity across the menstrual cycle with a significant decrease from the follicular to luteal phase (Schmalenberger et al., 2019). Analysis comparing cycle sub-phases (i.e., menstrual, mid-to-late follicular, ovulatory, early-to-mid luteal, premenstrual) revealed a decrease in measures of cardiac vagal activity from menstrual to premenstrual sub-phases and mid-to-late follicular to premenstrual sub-phases, indicating that the change from follicular to luteal phases is likely driven by a decline in the premenstrual sub-phase (of the luteal phase) (Schmalenberger et al., 2019). The meta-analysis pooled multiple autonomic measures (HF, RMSSD, BRS) and separate analyses for each variable were not completed (Schmalenberger et al., 2019). Unfortunately, none of the included studies in our review reported if participants were eumenorrheic or menstrual cycle stage, which should be included in future studies.
Multiple studies did not specify if reporting sex/gender or interchanged the two terms and no studies explicitly reported both sex and gender. Sex refers to a set of biological attributes (primarily associated with physical and physiological features including chromosomes, gene expression, hormone levels and function, and reproductive/sexual anatomy), whereas gender refers to the socially constructed roles, behaviours, expressions and identities of girls, women, boys, men, and gender diverse people (Canadian Institutes of Health Research, 2020). Future research should consider reporting both sex and gender. There is a lack of evaluation of both sex and gender differences in autonomic function following mTBI.
Length of recording periods are generally divided as ultra-short-term (<3 minutes), short-term (5-20 minutes) or 24 hour recordings. It is inappropriate to compare HRV metrics when collected from epochs of different lengths, as longer recordings are associated with increased HRV (Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology, 1996). Unfortunately, one study (Tan et al., 2009) did compare data from short-term recordings (mTBI participants) to 24 hour control data. Ultra-short-term recordings for analysis of HRV remain controversial, yet these were used in the majority of included studies (Abaji et al., 2016; Brandt et al., 2020; Haider et al., 2020; Hilz et al., 2011, 2015, 2016, 2017; Huang et al., 2019; Johnson et al., 2018; La Fountaine 2009, 2011; Russell et al., 2020). Recent research suggests that recordings of at least 4 minutes should be used to approximate short-term data (Burma et al., 2021). Using 1 minute recordings, metrics can vary by ≥30% when compared to a 5 minute recording (Burma et al., 2021). Based on these findings, analysis using epochs < 4 minutes were rated as unreliable/invalid in the RoB assessment.
Recording methods should also be carefully considered. While higher-end HR monitors may obtain fairly close values to an electrocardiogram (ECG) at rest and during exertion (pNN50 being an exception) (Vanderlei et al., 2008), ECG remains the gold-standard for measurement of HRV. For specific variables, such as QTVI, a 12-lead ECG should be used in place of a 3-lead ECG (which was used in an included study (La Fountaine et al., 2011)) as electrode placement can cause variation in QT time of 25-30 ms (Salvi et al., 2012). Respiratory rate is also known to affect certain HRV variables, specifically HF. Respiratory rate was measured in nine studies (Hilz et al., 2011, 2015, 2016, 2017, 2020; Huang et al., 2019; La Fountaine et al., 2018; Purkayastha, Williams et al., 2019; Solbakk et al., 2005). HF is commonly referred to as the respiratory frequency as it includes frequencies between 0.15–0.4 Hz, corresponding to physiologic events occurring 9–24 times per minute. At rest, respirations occur ∼12–20 times per minute, thus falling into the HF range (Singh et al., 2018); however, altered breathing faster than the normal range, potentially due to test anxiety, would elevate this rate above the HF band and thus skew interpretation of results. Therefore, future studies investigating HRV following mTBI should ensure there is a steady and consistent rate and depth of breathing for all participants at all time points. Additional considerations should be taken for certain test conditions. For example, studies using face cooling (Haider et al., 2020; Johnson et al., 2018) or cold pressor test (Johnson et al., 2020) conditions should address whether participants (in the case of the included studies, athletes) use ice baths in their training regimen as this may affect response.
Small sample size is another limitation of the included research given there is large between-subject variability for many HRV variables (Nunan et al., 2010). Of the included studies, only twelve had a sample size ≥25 in both groups (mTBI, controls) (Brandt et al., 2020; Dobney et al., 2018; Hanna-Pladdy et al., 2011; Hilz et al., 2016; Howard et al., 2018; Hutchison et al., 2017; Liao et al., 2016; Pyndiura et al., 2020l; Russell et al., 2020; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Truong & Ciuffreda, 2016). The n = 25 sample size as been used as a cut-off for small sample size papers in other systematic reviews of this population (Iverson et al., 2020). Notably, only three of the studies (Howard et al., 2018; Hutchison et al., 2017; Truong & Ciuffreda, 2016) with a larger sample (≥25 in both groups) were rated as “acceptable” on the risk of bisk assessment. Ability to detect between-group differences may be affected by these small sample sizes with potentially great inter-subject variability. A lack of evaluation for confounding/modification by factors that may have affected test results in addition to the use of unreliable/invalid autonomic function outcome measures (i.e., ultra-short-term recordings) are reflected in the RoB assessment where 72% (28 out of 39) of studies (Abaji et al., 2016; Bishop et al., 2017; Brandt et al., 2020; Clausen et al., 2016; Dobney et al., 2018; Dobson et al., 2017; Gall et al., 2004; Haider et al., 2020; Hanna-Pladdy et al., 2001; Hilz et al., 2011, 2015, 2016, 2017, 2020; Huang et al., 2019; Johnson et al., 2018; La Fountaine et al., 2009, 2011, 2016, 2018; Liao et al., 2016; Mirow et al., 2016; Pyndiura et al., 2020; Russell et al., 2020; Senthinathan et al., 2017; Sung, Chen et al., 2016; Sung, Lee et al., 2016; Tan et al., 2009) were rated as “unacceptable”.
4.3Participant symptomatology
Whether a degree of ANS dysfunction persists in some or all cases following symptom resolution remains a question of great clinical interest. The relationship between specific post-concussive symptoms and autonomic dysfunction also requires further study. With return to sport protocols currently based on symptom exacerbation with increasing exertion/physical contact (McCrory et al., 2017), assessment of specific autonomic parameters could offer an objective measure of recovery. Preliminary data suggests certain autonomic measures may be altered in asymptomatic individuals in the post-acute stage of injury (Abaji et al., 2016; Haider et al., 2020), although these studies are limited by methodological confounders (see Table 5). In general, symptomatology is poorly characterized in the included literature, with sixteen studies (Abaji et al., 2016; Dobson et al., 2017; Gall et al., 2004; Hilz et al. 2011, 2015, 2016, 2017; Johnson et al., 2020; La Fountaine et al., 2009, 2016, 2018; Pyndiura et al., 2020; Senthinathan et al., 2017; Solbakk et al., 2005; Tan et al., 2009; Truong & Ciuffreda, 2016) not reporting results of a specific symptom questionnaire nor a general symptom battery. Unfortunately, several studies noted a symptom checklist had been completed, but results were not reported in the manuscript (Gall et al., 2004; Johnson et al., 2020; La Fountaine et al., 2009; Senthinathan et al., 2017; Tan et al., 2009). Additionally, the heterogeneity of questionnaires does not allow for easy comparison between studies and standardization should be considered. There is a great need for a study with the primary aim of evaluating change in multiple post-concussive symptoms and autonomic metrics across recovery timepoints in a large cohort. Finally, study of ANS dysfunction in cohorts with persistent post-concussive symptoms is limited. While several studies did complete assessments in the chronic stage of injury (>3 months post), symptoms were poorly characterized in many cases (Hilz et al., 2011, 2015, 2016, 2017; Pyndiura et al., 2020; Solbakk et al., 2005). Research in cohorts with persistent post-concussive symptoms, diagnosed using standardized criteria, is necessary to understand potential underlying physiologic mechanisms contributing to persistent symptoms, such as exercise intolerance, dizziness and headache.
4.4Limitations
There are several limitations based on the methods of this review. Only published work was included in the review, thus the findings are subject to publication bias. Additionally, only English language papers were included. Our results and conclusions are therefore subject to language-of-publication bias. The majority of studies included in this review had a high RoB due to methodological shortcomings. As pre-clinical studies were not included, this review does not address translational research on this topic. It is understood that different critical appraisal tools uniquely assess the methodological quality of a publication and different RoB tools have been shown to lead to different conclusions (ratings of study quality) (Losilla et al., 2018). The SIGN checklist was chosen for this review as it is a comprehensive, easy to use RoB assessment with checklists tailored to different study designs (i.e., cohort, case-control studies). The heterogeneity of methods, outcomes and samples precluded a quantitative review or meta-analysis.
4.5Future directions
There is great opportunity for the use of ANS function parameters, such as HRV, to understand recovery following mTBI and potentially guide return to play; however, the significant limitations of the presented literature limit its clinical applicability. We propose the following recommendations to build upon the current literature and ensure methodological rigor in the field.
1. Additional research is needed to evaluate change in autonomic parameters across time points. While several studies have assessed metrics across time, due to a lack of control for confounders, small sample sizes and lack of strong methodological designs, future research is needed to understand the relationship between mTBI and ANS changes that may occur following injury. Completing both resting and exertional tasks at all timepoints should be considered.
2. Symptomatology of study cohorts should be characterized using commonly used symptom batteries (i.e., symptom evolution score in SCAT5, or PCSS in ImPACT). Symptom data should be reported alongside all autonomic data. This is necessary to understand the link between specific post-concussive symptoms and autonomic function.
3. Recording duration must be considered in the interpretation of HRV results. Ultra-short-term recordings (<3 minutes) remain controversial and recent data suggest that a recording of at least 4 minutes is required to approximate short-term recordings (>5 minutes) (Burma et al., 2021).
4. Increasing methodological rigor in the field requires addressing confounders in the study design. These include reporting on past medical history and medications of participants, consumption of caffeine/tobacco and participation in exercise prior to data collection.
5. There is a lack of investigation of sex/gender differences across recovery trajectory and test conditions. We know that females may be more likely to develop persistent post-concussive symptoms (Iverson et al., 2017), however this has not been investigated in the context of ANS dysfunction. Multiple studies included only male participants (Abaji et al., 2016; Bishop et al., 2017; Gall et al., 2004; La Fountaine et al., 2018; Mirow et al., 2016; Tan et al., 2009; Wright et al., 2017, 2018). Furthermore, none of the included studies reported on stage of the menstrual cycle, although this is known to influence HRV (Schmalenberger et al., 2020). Additionally, for certain test conditions, such as the isometric hand grip, there are known sexually dimorphic differences in force steadiness (Jakobi et al., 2018) that should be accounted for. This is also a reflection of the relatively small sample sizes of most studies that are not adequately powered to investigate these questions.
5Conclusion
Measures of autonomic function, such as HRV and BRS, are objective, quantitative measures with the potential to be used to assess recovery trajectory following mTBI. This review summarized a broad scope of methods and variables to examine changes in autonomic function following mTBI. Currently, recovery is largely assessed by subjective metrics, such as symptom questionnaires. While objective measures of recovery continue to be investigated, there is significant opportunity for the use of these novel ANS provocation methods to evaluate both recovery across timepoints and the underlying pathophysiology of persistent symptoms. However, additional research using more rigorous methodology to enable evaluation of multiple covariates will be required before these tests can be used clinically. The potential for evaluation of these metrics across timepoints to inform return-to-sport, in addition to traditional symptom ratings, is of particular interest in the field.
Acknowledgments
The authors would like to acknowledge the help of Lorraine Toews, a health science librarian, for providing her expertise in the development of the search strategy for this review.
Conflict of interest
The authors have no conflict of interest to disclose.
Supplementary materials
[1] The supplementary tables are available from https://dx.doi.org/10.3233/NRE-210243.
References
1 | Abaji, J. P. , Curnier, D. , Moore, R. D. , & Ellemberg, D. ((2016) ). Persisting effects of concussion on heart rate variability during physical exertion. Journal of Neurotrauma, 33: , 811–817. |
2 | Benarroch, E. E. ((1993) ). The central autonomic network: functional organization, dysfunction, and perspective. Mayo Clinic Proceedings, 68: , 988–1001. |
3 | Bishop, S. , Dech, R. , Baker, T. , Butz, M. , Aravinthan, K. , & Neary, J. P. ((2017) ). Parasympathetic baroreflexes and heart rate variability during acute stage of sport concussion recovery. Brain Injury, 31: , 247–259. |
4 | Blake, T. A. , McKay, C. D. , Meeuwisse, W. H. , & Emery, C. A. ((2016) ). The impact of concussion on cardiac autonomic function: A systematic review. Brain Injury, 30: , 132–145. |
5 | Brandt, E. , Wilson, J. K. , Rieger, R. E. , Gill, D. , Mayer, A. R. , & Cavanagh, J. F. ((2020) ). Respiratory sinus arrhythmia correlates with depressive symptoms following mild traumatic brain enjury. Journal of Psychophysiology, 35: , 1–13. |
6 | Burma, J. S. , Graver, S. , Miutz, L. N. , Macaulay, A. , Copeland, P. V. , & Smirl, J. D. ((2021) ). The validity and reliability of ultra-short-term heart rate variability parameters and the influence of physiological covariates. Journal of Applied Physiology, 130: , 1848–1867. |
7 | Callaway, C. C. M. , & Kosofsky, B. E. ((2019) ). Autonomic dysfunction following mild traumatic brain injury. Current Opinion in Neurology, 32: , 802–807. |
8 | Canadian Institutes of Health Research. (2020, April 28). What is gender? What is sex?. https://cihr-irsc.gc.ca/e/.html |
9 | Clausen, M. , Pendergast, D. R. , Willer, B. , & Leddy, J. ((2010) ). Cerebral blood flow during treadmill exercise is a marker of physiological postconcussion syndrome in female athletes. Journal of Head Trauma Rehabilitation, 31: , 215–224. |
10 | Ding, K. , Tarumi, T. , Tomoto, T. , Mccolloster, M. , Le, T. , Dieppa, M. , Diaz-Arrastia, R. , Bell, K. , Madden, C. , Cullum, C. M. , & Zhang, R. ((2020) ). Impaired cerebral blood flow regulation in chronic traumatic brain injury. Brain Researach, 1743: , 146924. |
11 | Dobney, D. M. , Thomas, S. G. , Taha, T. , & Keightley, M. ((2018) ). Physiological and performance measures for baseline concussion assessment. Journal of Sport Rehabilitation, 27: , 312–318. |
12 | Dobson, J. L. , Yarbrough, M. B. , Perez, J. , Evans, K. , & Buckley, T. ((2017) ). Sport-related concussion induces transient cardiovascular autonomic dysfunction. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 312: , R575–R584. |
13 | Duschek, S. , Werner, N. S. , & Reyes Del Paso, G. A. ((2013) ). The behavioral impact of baroreflex function: a review. Psychophysiology, 50: , 1183–1193. |
14 | EndNote Team. ((2013) ) EndNote. EndNote X9 ed. Philadelphia, PA: Clarivate. |
15 | Gall, B. , Parkhouse, W. , & Goodman, D. ((2004) ). Heart rate variability of recently concussed athletes at rest and exercise. Medicine & Science in Sports & Exercise, 36: , 1269–1274. |
16 | Giza, C. C. , & Hovda, D. A. ((2014) ). The new neurometabolic cascade of concussion. Neurosurgery, 75: , S24–33. |
17 | Gourine, A. V. , & Ackland, G. L. ((2019) ). Cardiac vagus and exercise. Physiology, 34: , 71–80. |
18 | Haider, M. N. , Johnson, B. D. , Horn, E. C. , Leddy, J. J. , Wilber, C. G. , Reed, E. L. , O’Leary, M. , Bloomfield, A. , Decezaro, L. L. , & Willer, B. S. ((2020) ). Blunted cardiac parasympathetic activation in student athletes with a remote history of concussion: A pilot study. Frontiers in Neurology, 11: , 547126. |
19 | Hanna-Pladdy, B. , Berry, Z. M. , Bennett, T. , Phillips, H. L. , & Gouvier, W. D. ((2001) ). Stress as a diagnostic challenge for postconcussive symptoms: Sequelae of mild traumatic brain injury or physiological stress response. The Clinical Neuropsychologist, 3: , 289–304. |
20 | Healthcare Improvement Scotland. (n.d.). Checklists. https://www.sign.ac.uk/what-we-do/methodology/checklists/ |
21 | Heffernan, K. S. , Fahs, C. A, , Shinsako, K. K. , Jae, S. Y. , & Fernhall, B. ((2007) ). Heart rate recovery and heart rate complexity following resistance exercise training and detraining in young men. American Journal of Physiology-Heart Circulatory Physiology, 293: , H3180–3186. |
22 | Hilz, M. J. , DeFina, P. A. , Anders, S. , Koehn, J. , Lang, C. L. , Pauli, E. , Flanagan, S. R. , Schwab, S. , & Marthol, H. ((2011) ). Frequency analysis unveils cardiac autonomic dysfunction after mild traumatic brain injury. Journal of Neurotrauma, 28: , 1727–1738. |
23 | Hilz, M. J. , Aurnhammer, F. , Flanagan, S. R. , Intravooth, T. , Wang, R. , Hösl, K. M. , Pauli, E. , & Koehn, J. ((2015) ). Eyeball pressure stimulation unveils subtle autonomic cardiovascular dysfunction in persons with a history of mild traumatic brain injury. Journal of Neurotrauma, 32: , 1796–1804. |
24 | Hilz, M. J. , Liu, M. , Koehn, J. , Wang, R. , Ammon, F. , Flanagan, S. R. , & Hösl, K. M. ((2016) ). Valsalva maneuver unveils central baroreflex dysfunction with altered blood pressure control in persons with a history of mild traumatic brain injury. BMC Neurology, 16: , 61. |
25 | Hilz, M. J. , Wang, R. , Markus, J. , Ammon, F. , Hösl, K. M. , Flanagan, S. R. , Winder, K. , & Koehn, J. ((2017) ). Severity of traumatic brain injury correlates with long-term cardiovascular autonomic dysfunction. Journal of Neurology, 264: , 1956–1967. |
26 | Hilz, M. J. , Wang, R. , Liu, M. , Muresanu, D. , Flanagan, S. , Winder, K. , Hösl, K. , & Hummel, T. ((2020) ). Emotional and autonomic processing of olfactory stimuli is compromised in patients with a history of mild traumatic brain injury. Journal of Neurotrauma, 37: , 125–138. |
27 | Howard, L. , Dumkrieger, G. , Chong, C. D. , Ross, K. , Berisha, V. , & Schwedt, T. J. ((2018) ). Symptoms of autonomic dysfunction among thosewith persistent posttraumatic headache attributed to mild traumaticbrain injury: A comparison to migraine and healthy controls. Headache, 58: , 1397–1407. |
28 | Huang, M. , Frantz, J. , Moralez, G. , Sabo, T. , Davis, P. F. , Davis, S. L. , Bell, K. R. , & Pukayastha, S. ((2019) ). Reduced resting and increased elevation of heart rate variability with cognitive task performance in concussed athletes. Journal of Head Trauma Rehabilitation, 34: , 45–51. |
29 | Hutchison, M. G. , Mainwaring, L. , Senthinathan, A. , Churchill, N. , Thomas, S. , & Richards, D. ((2017) ). Psychological and physiological markers of stress in concussed athletes across recovery milestones. Journal of Head Trauma Rehabilitation, 32: , E38–E48. |
30 | Iverson, G. L. , Gardner, A. J. , Terry, D. P. , Ponsford, J. L. , Sills, A. K. , Broshek, D. K. , & Solomon, G. S. ((2017) ). Predictors of clinical recovery from concussion: A systematic review. British Journal of Sports Medicine, 51: , 941–948. |
31 | Iverson, G. L. , Williams, M. W. , Gardner, A. J. , & Terry, D. P. ((2020) ). Systematic review of preinjury mental health problems as a vulnerability factor for worse outcome after sport-related concussion. The Orthopaedic Journal of Sports Medicine, 8: , 2325967120950682. |
32 | Jakobi, J. M. , Haynes, E. M. K. , & Smart, R. R. ((2018) ). Is there sufficient evidence to explain the cause of sexually dimorphic behaviour in force steadiness? Applied Physiology, Nutrition, and Metabolism, 43: , 1207–1214. |
33 | Johnson, B. D. , O’Leary, M. C. , McBryde, M. , Sackett, J. R. , Schlader, Z. J. , & Leddy, J. J. ((2018) ). Face cooling exposes cardiac parasympathetic and sympathetic dysfunction in recently concussed college athletes. Physiological Reports, 6: , e13694. |
34 | Johnson, B. D. , Sackett, J. R. , Schlader, Z. J. , & Leddy, J. J. ((2020) ). Attenuated cardiovascular responses to the cold pressor test in concussed collegiate athletes. Journal of Athletic Training, 55: , 124–131. |
35 | Kozlowski, K. F. , Graham, J. , Leddy, J. J. , Devinney-Boymel, L. , & Willer, B. S. ((2013) ). Exercise intolerance in individuals with postconcussion syndrome. Journal of Athletic Training, 48: , 627–635. |
36 | La Fountaine, M. F. , Heffernan, K. S. , Gossett, J. D. , Bauman, W. A. , & De Meersman, R. E. ((2009) ). Transient suppression of heart rate complexity in concussed athletes. Autonomic Neuroscience, 148: , 101–103. |
37 | La Fountaine, M. F. , Gossett, J. D. , De Meersman, R. E. , & Bauman, W. A. ((2011) ). Increased QT interval variability in recently concussed athletes: An exploratory observation. Journal of Athletic Training, 36: , 230–233. |
38 | La Fountaine, M. F. , Toda, M. , Testa, A. J. , & Hill-Lombardi, V. ((2016) ). Autonomic nervous system responses to concussion: Arterial pulse contour analysis. Frontiers in Neurology, 7: , 13. |
39 | La Fountaine, M. F. , Hohn, A. N. , Testa, A. J. , & Weir, J. P. ((2018) ). Attenuation of spontaneous baroreceptor sensitivity after concussion. Medicine & Science in Sports Exercise, 51: , 792–797. |
40 | Langer, L. , Levy, C. , & Bayley, M. ((2020) ). Increasing incidence of concussion: True epidemic or better recognition? Journal of Head Trauma Rehabilitation, 35: , E60–E66. |
41 | Leddy, J. J. , Kozlowski, K. , Donnelly, J. P. , Pendergast, D. R. , Epstein, L. H. , & Willer, B. ((2010) ). A preliminary study of subsymptom threshold exercise training for refractory post-concussion syndrome. Clinical Journal of Sport Medicine, 20: , 21–27. |
42 | Liao, K. H. , Sung, C. W. , Chu, S. F. , Chiu, W. T. , Chiang, Y. H. , Hoffer, B. , Ou, J. C. , Chen, K. Y. , Tsai, S. H. , Lin, C. M. , Chen, G. S. , Li, W. J. , & Wang, J. Y. ((2016) ). Reduced power spectra of heart rate variability are correlated with anxiety in patients with mild traumatic brain injury. Psychiatry Research, 243: , 349–356. |
43 | Liberati, A. , Altman, D. G. , Tetzlaff, J. , Mulrow, C. , Gøtzsche, P. C. , Ioannidis, J. P. A. , Clarke, M. , Devereaux, P. J. , Kleijnen, J. , & Moher, D. ((2009) ). The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLOS Medicine, 6: , e1000100. |
44 | Losilla, J. M. , Oliveras, I. , Marin-Garcia, J. A. , & Vives, J. ((2018) ). Three risk of bias tools lead to opposite conclusions in observational research synthesis. Journal of Clinical Epidemiology, 101: , 61–72. |
45 | Lucas, S. , Hoffman, J. M. , Bell, K. R. , & Dikmen, S. ((2013) ). A prospective study of prevalence and characterization of headache following mild traumatic brain injury. Cephalalgia, 34: , 93–102. |
46 | Machhada, A. , Trapp, S. , Marina, N. , Stephens, R. C. M. , Whittle, J. , Lythgoe, M. F. , Kasparov, S. , Ackland, G. L. , & Gourine, A. V. ((2017) ). Vagal determinants of exercise capacity. Nature Communications, 8: , 15097. |
47 | McCrory, P. , Meeuwisse, W. , Dvorak, J. , Aubry, M. , Bailes, J. , Broglio, S. , Cantu, R. C. , Cassidy, D. , Echemendia, R. J. , Castellani, R. J. , Davis, G. A. , Ellenbogen, R. , Emery, C. , Engebretsen, L. , Feddermann-Dermont, N. , Giza, C. C. , Guskiewicz, K. M. , Herring, S. , Iverson, G. L. , . . .& Vos, P. E. ((2017) ). Consensus statement on concussion in sport-the th international conference on concussion in sport held in Berlin, October 2016. British Journal of Sports Medicine, 51: , 838–847. |
48 | Meier, T. B. , Bellgowan, P. S. , Singh, R. , Kuplicki, R. , Polanski, D. W. , & Mayer, A. R. ((2015) ). Recovery of cerebral blood flow following sports-related concussion. JAMA Neurology, 72: , 530–538. |
49 | Mild Traumatic Brain Injury Committee of the Head Injury Interdisciplinary Special Interest Group of the American Congree of Rehabilitation Medicine. ((1993) ). Definition of mild traumatic brain injury. Journal of Head Trauma Rehabilitation, 8: , 86–87. |
50 | Mirow, S. , Wilson, S. H. , Weaver, L. K. , Churchill, S. , Deru, K. , & Lindblad, A. S. ((2016) ). Linear analysis of heart rate variability in post-concussive syndrome. Undersea & Hyperbaric Medicine, 43: , 531–547. |
51 | Monfredi, O. , Lyashkov, A. E. , Johnsen, A. B. , Inada, S. , Schneider, H. , Wang, R. , Nirmalan, M. , Wisloff, U. , Maltsev, V. A. , Lakatta, E. G. , Zhang, H. , & Boyett, M. R. ((2014) ). Biophysical characterization of the underappreciated and important relationship between heart rate variability and heart rate. Hypertension, 64: , 1334–1343. |
52 | Nunan, D. , Sandercock, G. R. , & Brodie, D. A. ((2010) ). A quantitative systematic review of normal values for short-term heart rate variability in healthy adults. Pacing and Clinical Electrophysiology, 33: , 1407–1417. |
53 | Pertab, J. L. , Merkley, T. L. , Cramond, A. J. , Cramond, K. , Paxton, H , & Wu, T. ((2018) ). Concussion and the autonomic nervous system-An introduction to the field and the results of a systematic review. NeuroRehabilitation, 42: , 397–427. |
54 | Pyndiura, K. L. , Di Battista, A. P. , & Hutchison, M. G. ((2020) ). A history of concussion is associated with minimal perturbations to heart rate variability in athletes. Brain Injury, 34: , 1416–1421. |
55 | Purkayastha, S. , Stokes, M. , & Bell, K. R. ((2019) ). Autonomic nervous system dysfunction in mild traumatic brain injury: A review of related pathophysiology and symptoms. Brain Injury, 33: , 1129–1136. |
56 | Purkayastha, S. , Williams, B. , Murphy, M. , Lyng, S. , Sabo, T. , & Bell, K. R. ((2019) ). Reduced heart rate variability and lower cerebral blood flow associated with poor cognition during recovery following concussion. Autonomic Neuroscience: Basic and Clinical, 220: , 102548. |
57 | Russell, K. N. , Preble, E. A. , Hegarty-Craver, M. , Arrieux, J. P. , Cole, W. R. , Choi, S. , Grego, S. , Olmsted, K. R. , & Gilchrist, K. H. ((2020) ). Feasibility of mild traumatic brain injury assessment based on cardiovascular response to postural change. Journal of Head Trauma Rehabilitation, 35: , E422–E428. |
58 | Salvi, V. , Karnad, D. R. , Kerkar, V. , Panicker, G. K. , Manohar, D. , Natekar, M. , Kothari, S. , Narula, D. , & Lokhandwala, Y. ((2012) ). Choice of an alternative lead for QT interval measurement in serial ECGs when lead II is not suitable for analysis. Indian Heart Journal, 64: , 535–540. |
59 | Schmalenberger, K. M. , Eisenlohr-Moul, T. A. , Würth, L. , Schneider, E. , Thayer, J. F. , Ditzen, B. , & Jarczok, M. N. ((2019) ). A systematic review and meta-analysis of within-person changes in cardiac vagal activity across the menstrual cycle: Implications for female health and future studies. Journal of Clinical Medicine, 8: , 1946. |
60 | Schmalenberger, K. M. , Eisenlohr-Moul, T. A. , Jarczok, M. N. , Eckstein, M. , Schneider, E. , Brenner, I. G. , Duffy, K. , Schweizer, S. , Kiesner, J. , Thayer, J. F. , & Ditzen, B. ((2020) ). Menstrual cycle changes in vagally-mediated heart rate variability are associated with progesterone: Evidence from two within-person studies. Journal of Clinical Medicine, 9: , 617. |
61 | Senthinathan, A. , Mainwaring, L. M. , & Hutchinson, M. ((2017) ). Heart rate variability of athletes across concussion recovery milestones: A preliminary study. Clinical Journal of Sport Medicine, 27: , 288–295. |
62 | Shaffer, F. , & Ginsberg, J. P. ((2017) ). An overview of heart rate variability metrics and norms. Frontiers in Public Health, 5: , 258. |
63 | Singh, N. , Moneghetti, K. J. , Christle, J. W. , Hadley, D. , Plews, D. , & Froelicher, V. ((2018) ). Heart rate variability: An old metric with new meaning in the era of using mHealth technologies for health and exercise training guidance. Part One: Physiology and methods. Arrhythmia & Electrophysiology Review, 7: , 193–198. |
64 | Solbakk, A. K. , Reinvang, I. , Svebak, S. , Nielsen, C. S. , & Sundet, K. ((2005) ). Attention to affective pictures in closed head injury: event-related brain potentials and cardiac responses. Journal of Clinical and Experimental Neuropsychology, 27: , 205–223. |
65 | Stulemeijer, M. , van der Werf, S. , Bleijenberg, G. , Biert, J. , Brauer, J. , & Vos, P. E. ((2006) ). Recovery from mild traumatic brain injury: A focus on fatigue. Journal of Neurology, 253: , 1041–1047. |
66 | Sung, C. W. , Chen, K. Y. , Chiang, Y. H. , Chiu, W. T. , Ou, J. C. , Lee, H. C. , Tsai, S. H. , Lin, J. W. , Yang, C. M. , Tsai, Y. R. , Liao, K. H. , Chen, G. S. , Li, W. J. , & Wang, J. Y. ((2016) ). Heart rate variability and serum level of insulin-like growth factor- are correlated with symptoms of emotional disorders in patients suffering a mild traumatic brain injury. Clinical Neurophysiology, 127: , 1629–1638. |
67 | Sung, C. W. , Lee, H. C. , Chiang, Y. H. , Chiu, W. T. , Chu, S. F. , Ou, J. C. , Tsai, S. H. , Liao, K. H. , Lin, C. M. , Lin, J. W. , Chen, G. S. , Li, W. J. , & Wang, J. Y. ((2016) ). Early dysautonomia detected by heart rate variability predicts late depression in female patients following mild traumatic brain injury. Psychophysiology, 53: , 455–464. |
68 | Tan, G. , Fink, B. , Dao, T. K. , Hebert, R. , Farmer, L. S. , Sanders, A. , Pastorek, N. , & Gevirtz, R. ((2009) ). Associations among pain, PTSD, mTBI, and heart rate variability in veterans of Operation Enduring and Iraqi Freedom: a pilot study. Pain Medicine, 10: , 1237–1245. |
69 | Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. ((1996) ). Heart rate variability. Standards of measurement, physiological interpretation, and clinical use. European Heart Journal, 17: , 354–381. |
70 | Theadom. A. , Parag, V. , Dowell, T. , McPherson, K. , Starkey, N. , Barker-Collo, S. , Jones, K. , Ameratunga, S. , & Feigin, V. L. , BIONIC Research Group ((2016) ). Persistent problems year after mild traumatic brain injury: a longitudinal population study in New Zealand. British Journal of General Practice, 66: , e16–23. |
71 | Truong, J. Q. , & Ciuffreda, K. J. ((2016) ). Comparison of pupillary dynamics to light in the mild traumatic brain injury (mTBI) and normal populations. Brain Injury, 30: , 1378–1389. |
72 | Vanderlei, L. C. M. , Silva, R. A. , Pastre, C. M. , Azevedo, F. M. , & Godoy, M. F. ((2008) ). Comparison of the Polar Si monitor and the ECG for the analysis for heart rate variability in the time and frequency domains. Brazilian Journal of Medical and Biological Research, 41: , 854–859. |
73 | Wehrwein, E. A. , Orer, H. S. , & Barman, S. M. ((2016) ). Overview of the anatomy, physiology, and pharmacology of the autonomic nervous system. Comprehensive Physiology, 6: , 1239–1278. |
74 | Wright, A. D. , Smirl, J. D. , Bryk, K. , & van Donkelaar, P. ((2017) ). A prospective transcranial doppler ultrasound-based evaluation of the acute and cumulative effects of sport-related concussion on neurovascular coupling response dynamics. Journal of Neurotrauma, 34: , 3097–3106. |
75 | Wright, A. D. , Smirl, J. D. , Bryk, K. , Fraser, S. , Jakovac, M. , & van Donkelaar, P. ((2018) ). Sport-related concussion alters indices of dynamic cerebral autoregulation. Frontiers in Neurology, 9: , 196. |




