Peripheral neuropathic pain
Abstract
Neuropathic pain (NP) can have either central nervous system causes or ones from the peripheral nervous system. This article will focus on the epidemiology, classifications, pathology, non-invasive treatments and invasive treatments as a general review of NP involving the peripheral nervous system. NP has characteristic symptomatology such as burning and electrical sensations. It occurs in up to 10% of the general population. Its frequency can be attributed to its occurrence in neck and back pain, diabetes and patients receiving chemotherapy. There are a wide range of pharmacologic options to control this type of pain and when such measures fail, numerous interventional methods can be employed such as nerve blocks and implanted stimulators. NP has a cost to the patient and society in terms of emotional consequences, quality of life, lost wages and the cost of assistance from the medical system and thus deserves serious consideration for prevention, treatment and control.
1Introduction
Neuropathic pain (NP) can result from injury to either the central or peripheral nervous system (Colloca et al., 2017). Thus, it can be associated with a wide variety of conditions and syndromes. Examples of central nervous system conditions that can have NP include stroke spinal cord injury and multiple sclerosis. Chemotherapy induced neuropathy and diabetic neuropathy are two examples of disorders that can be associated with NP from peripheral nerve injury. For central NP lesions can occur anywhere along the path from the dorsal horn of the spinal cord to the cortex in the somatosensory system (Scholz et al., 2019) which is concerned with nociception, mechanoception, thermoreception, chemoreception and proprioception. For the peripheral nervous system lesions range from the dorsal root ganglion and its spinal cord connections to the periphery. Generally, the smaller fibers such as the myelinated A beta and delta fibers and the unmyelinated C fibers are affected (Colloca et al., 2017). These lesions in the somatosensory system result in maladaptive responses from the nervous system that can cause pain that is either spontaneous or provoked by sensory stimuli. NP comprises heterogeneous clinical presentations with a variety of etiologies and pathophysiologies.
The symptoms that these pathologies can generate are diverse and include sensations of burning, tingling, lightning bolts of pain, sharp sensations, unpleasant cold sensations, and electric-like sensations (Colloca et al., 2017). Touching of the skin can cause either allodynia or hyperalgesia (Derry, Bell, Straube, Wiffen, Aldington, & Moore, 2019). Allodynia is the experience of pain from a stimulus that normally is not painful whereas hyperalgesia is the exaggerated pain response to a painful stimulus. Secondary symptoms and problems can include anxiety, depression, sleep disturbances as well as impairment of quality of life.
The overall prevalence of NP in the general population is from 7 to 10% (Derry et al., 2019). There have been difficulties in determining these figures due to issues with available diagnostic criteria that can be applied to large populations. Chronic NP is slightly more frequent in women (8%) than men (5.7%) in those older than 50 years (8.9%) versus those under 50 years (5.6%). One common cause of chronic NP is diabetic neuropathy (Abbott, Malik, van Ross, Kulkarni, & Boulton, 2011). One large study offered a figure of 47% for the prevalence of neuropathy in diabetics. A study of diabetics receiving community-based health care in northwest England found that about a third of these individuals had painful neuropathic symptoms (Abbott et al., 2011). Another article stated the prevalence of NP as 11% of patients with diabetic peripheral neuropathy (Argoff, Cole, Fishbain, & Irving, 2006).
Radiculopathies comprise one of the most frequent causes of NP (Colloca et al., 2017). One study in Rochester, Minnesota showed an annual incidence of cervical radiculopathy of 107.3/100,000 for men and 63.5/100,000 for women (Kim, Nemani, Piyaskulkaew, Vargas, & Riew, 2016). Peak incidence was at 50 to 54 years of age. Motor vehicle accidents can cause cervical radiculopathy, and the relationship is established in 3% to 23% of cases (Rodine, & Vernon, 2012). The most frequent complaints in cervical radiculopathy are neck pain, paresthesias and radicular pain (Rodine, & Vernon, 2012). Although pain more often follows a myotomal pattern, dermatomal pain patterns are most frequent at C4 (60%) followed by C7 (34.2%) and C6 (35%) (Rodine, & Vernon, 2012; Magnus, Viswanath, Viswanathan, & Mesfin, 2020).
For lumbosacral radiculopathy the prevalence rate is about 3 to 5% (Dydyk, & Das, 2020). The incidence of low back pain ranges from 13% to 31% and among those with low back pain, radicular symptoms present in 12% to 40%. About 80% to 90% of all health care expenditures are accounted for by treatment of patients with chronic low back pain (Alexander, & Varacallo, 2020). Radiculopathy can present with many different symptoms such as 63% to 72% with paresthesia, 35% with lower limb radiation of pain and 27% with numbness (Dydyk, & Das, 2020). On exam 37% will have muscle weakness, 40% will have absent ankle reflexes while 18% will have absent knee reflexes (Dydyk, & Das, 2020).
Chemotherapy can also be associated with the development of painful neuropathies (Zaja̧czkowska et al., 2019). The prevalence ranges from 19% to 85%. Although the antineoplastic agents generally affect the sensory nerves, the autonomic nervous system and the motor nerves can be affected as well. Six categories of chemotherapeutic agents can cause these impairments and these are immunomodulatory agents, taxanes, proteasome inhibitors, epothilones, platinum-based antineoplastic agents and vinca alkaloids (Zaja̧czkowska et al., 2019). Efforts at prevention so far have met with minimal success and treatment also lacks any effective measures.
This article will focus on the peripheral neuropathic pain syndromes. An overview of NP will be provided that will include a discussion of the various syndromes and the available treatments. Treatments will range from non-pharmacologic, to various medications and finally to some of the more invasive options. New modalities for treatment have arisen and what can be a difficult condition to manage is gradually yielding to these new modalities when the older ones are not effective. The goals are diminution of pain, restoration of function and improvement of quality of life.
2Peripheral neuropathy classification, causes, prognosis
The nervous system is commonly differentiated into central and peripheral nervous systems. The central nervous system is the brain and spinal cord, and the peripheral system includes all nerve structures outside of the brain and spinal cord. The peripheral nervous system can be further divided into somatic and autonomic nervous systems. The autonomic nervous system is comprised of sympathetic and parasympathetic nerves that control involuntary functions such as heart rate, blood pressure, blood flow to different organs, gastrointestinal function, respiration, and sexual function (McCorry, 2007). The somatic nervous system includes efferent motor control of muscles and afferent sensory nerves. From an anatomic perspective, the somatic peripheral nervous system begins where nerve roots branch off the spinal cord. Anterior nerve roots are afferent motor nerve fibers with cell bodies in the anterior horn of the spinal cord that supply innervation to somatic structures. Posterior nerve roots are efferent nerve fibers with cell bodies in the dorsal root ganglion (Drake, Vogl, Mitchell, & Gray, 2010; Ropper, Samuels, & Klein, 2014). These roots combine to form a spinal nerve at a certain vertebral level. The spinal nerve branches into dorsal rami that innervate the back and anterior rami. For nerves running to the limbs, the nerve fibers pass through an intricate plexus before branching into separate peripheral nerves that innervate specific muscles and skin (Drake et al., 2010). Damage to the peripheral nervous system can occur at any of these anatomic levels, and it is important to classify nerve injuries by which level is affected.
2.1Classification
One classification system stratifies nerve injuries by anatomic level: neuronopathy, radiculopathy, plexopathy, mononeuropathy, mononeuropathy multiplex and, and polyneuropathy (Ropper et al., 2014). Neuronopathy is disease of the neurons or cell body of the nerve. For motor nerves this occurs in the anterior horn cell and includes amyotrophic lateral sclerosis, which classically presents as painless weakness with upper and lower motor signs (Barohn, & Amato, 2013). Sensory nerves have their cell bodies in the dorsal root ganglion just outside of the spinal cord. Ganglionopathies present as sensory changes and can be caused by paraneoplastic, autoimmune, and nutritional disorders (Hlubocky & Smith, 2014). Radiculopathy describes injuries to nerve roots. This can occur with root compression from a herniated disc or narrowed foramina due to osteoarthritis, Herpes Zoster reactivation or in diabetic radiculopathy. Radiculopathies often present with pain and weakness in distribution of the nerve root[s] affected but can be difficult to differentiate from mononeuropathies (Ropper et al., 2014).
Moving distally along the anatomic nerve pathway, there are plexopathies, most commonly involving the brachial or lumbosacral plexuses. Plexus injuries often present with asymmetric proximal and distal weakness in a limb (Abbott et al., 2011), thus not correlating with a nerve root or peripheral nerve pattern of involvement (Ropper et al., 2014). Examples of this pathology include Parsonage-Turner syndrome of the brachial plexus, traumatic injury, compression or infiltration by tumor, and post-radiation plexopathy (Suarez et al., 1996; Bowen, Seidenwurm, & Expert Panel on Neurologic Imaging, 2008; Barohn & Amato, 2013). Peripheral nerves emerge from the plexus, and there are multiple pathologies causing mononeuropathies. A mononeuropathy can be recognized by pain or weakness involving only dermatomes and myotomes of only one nerve. Entrapment syndromes are possible, common examples being median neuropathy at the carpal tunnel, ulnar neuropathy at the elbow and common fibular nerve injury at the fibular head (Hobson-Webb, & Juel, 2017). These injuries occur at locations where the peripheral nerve passes through a narrow space and can be subjected to chronic compression over time. Mononeuropathy multiplex, as indicated by its name, is an acute or subacute disease process that causes complete or nearly complete paralysis in several peripheral nerves (Ropper et al., 2014). Diseases that cause mononeuropathy multiplex include vasculitis, Leprosy, Lyme disease, sarcoid, HIV, and multifocal acquired demyelinating sensory and motor neuropathy (MADSAM) (Barohn, & Amato, 2013; Ropper et al., 2014).
The most distal classification is polyneuropathy, which is a generalized disease of the nerves and usually has symmetric involvement affecting the most distal nerves first (Ropper et al., 2014). This can occur in an acute process such as Guillain-Barre Syndrome (also known as acute inflammatory demyelinating polyneuropathy). There are also many chronic processes that cause polyneuropathies. The most common causes include diabetes mellitus, drug toxicity, hereditary neuropathies (also called Charcot-Marie-Tooth disease), HIV, inflammatory neuropathies (chronic inflammatory demyelinating polyradiculoneuropathy), alcohol abuse, and leprosy (Martyn, & Hughes, 1997; Hanewinckel, van Oijen, Ikram, & van Doorn, 2016). Some estimates of the prevalence of polyneuropathy were 1% to 3% in the general population (Hanewinckel et al., 2016). However the prevalence of peripheral neuropathy increases with age. One study used data from the National Health and Nutrition Examination Survey in the United States and found that the prevalence of peripheral neuropathy in adults age 40 and older was 14.8% (Gregg et al., 2004).
2.2Causes and prognosis
The many causes for peripheral neuropathy can be grouped into hereditary, infectious, immunologic, metabolic, mechanical, cancer related, toxic, and ideopathic (Barohn, & Amato, 2013).
2.3Hereditary neuropathies
Hereditary neuropathies are grouped into Charcot-Marie-Tooth type 1-4. Charcot-Marie-Tooth type 1 (CMT1) is passed by autosomal dominance pattern and is demyelinating. CMT2 is also autosomal dominant but affects the axons. CMT3 has onset during infancy, and CMT4 has an autosomal recessive heritance pattern (Ropper et al., 2014). The hereditary neuropathies are associated with pes cavus, hammertoes and talipes equinus due to imbalances in muscle strength or intrinsic foot muscle weakness during growth and development of bony structures that occurs in infancy or childhood (Ropper et al., 2014). Hereditary neuropathies that are not classified under CMT include hereditary sensory and autonomic neuropathies (HSAN) and giant axonal neuropathy (GAN) among others (Kang, Shefner, Nordi, & Goddeau, 2020). Severity of neuropathy and disability ranges among the subtypes of hereditary neuropathies, but as a whole they are progressive, and treatment is mostly supportive (Ropper et al., 2014).
2.4Infectious neuropathies
Infectious neuropathies include herpes zoster, leprosy, Lyme disease and human immunodeficiency virus (HIV) (Barohn, & Amato, 2013). Herpes zoster infection is caused by the reactivation of varicella zoster virus from the dorsal root ganglion, causing an often severe pain syndrome and vesicular rash in a dermatomal distribution (Seo, Lee, Hyun, & Kim, 2012). However there are reports of herpes zoster causing a polyneuropathy with improvement after antiviral treatment of the underlying infection (Seo et al., 2012; Teo, Chawla, & Kaushik, 2016). Leprosy is caused by Mycobacterium leprae, which directly attacks peripheral nerves (Wan, Rivadeneira, Jouvin, & Dellon, 2016). Due to antibiotics, leprosy is relatively uncommon in developed countries, but in 2006 there were 4 million cases estimated worldwide (Wan et al., 2016). Lyme disease can cause an inflammatory mononeuropathy multiplex or painful radiculoneuritis (Halperin, 2020). Treatment of Lyme neuropathy is with appropriate antibiotics with improvement in most patients (Hu, 2020). HIV is associated with distal symmetric polyneuropathy in up to 50% of infections. The pathology can be either direct neural damage by the virus or neurotoxicity by medications used to treat HIV (Hu, 2020).
2.5Immunologic neuropathies
Immune-mediated diseases are another cause of peripheral neuropathy (Barohn, & Amato, 2013). Guillain-Barre Syndrome (GBS) is an acute, ascending paralysis affecting motor and sensory nerves. This disease is often preceded by a viral illness and thought to be an immunologic attack on peripheral nerves. In most cases, this is inflammatory process that demyelinates the peripheral nerves, but there is an acute axonal form (Ropper et al., 2014). Treatments of IVIg or plasma exchange are effective, but about 20% of patients are still unable to walk 6 months after onset and many patients suffer from chronic pain and fatigue after the acute episode is concluded (van Doorn, 2013). Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is another immune-mediated neuropathy that is differentiated from GBS by a slower onset and progression in symptoms over months. Treatments include steroids, IVIg and plasma exchange (Ropper et al., 2014). One study of 38 patients with CIDP at 5 years showed 26% with complete remission, 61% with partial remission and ability to walk, and 13% with either severe disability or relapses requiring treatment (Kuwabara, Misawa, Mori, Tamura, Kubota, & Hattori, 2006). Another likely immune-mediated neuropathy is Parsonage-Turner Syndrome (brachial neuritis). This syndrome presents with acute pain in one or both upper limbs, followed shortly by weakness and even muscle atrophy (Tsairis, Dyck, & Mulder, 1972). The etiology is still not well understood, but in one study biopsies were taken from 4 patients which showed inflammatory cells in the brachial plexus. Steroids are often used in treatment and the overall prognosis for recovery is good with one study showing 89% of patients recovering in 3 years (Tsairis et al., 1972).
Inflammation of the blood supply to nerves, called vasa nervorum, can occur in isolation or in systemic vasculitis. This can cause asymmetric neuropathy or mononeuropathy multiplex via ischemic damage (Blaes, 2015). Prevalence of neuropathies associated with systemic vasculitis vary widely with some studies finding between 30–50% of vasculitis patients having symptoms of peripheral neuropathy (Blaes, 2015). There are many vasculitic diseases that can cause neuropathy, and prognosis for the neuropathy depends on the treatment of the underlying vasculitis. Sarcoidosis can also cause neuropathies with non-caseating granulomas seen on nerve biopsy. Corticosteroids can stabilize or even reduce symptoms of neuropathy in these cases (Said et al., 2002). Yet another immune-mediated neuropathy is associated with monoclonal gammopathy of undetermined significance. This has an onset over weeks to months and is due to antibodies against myelin or other nerve components. There is often transient improvement with plasma exchange, IVIG or immunosuppressive medications (Ropper et al., 2014).
2.6Metabolic neuropathies
Metabolic causes of peripheral neuropathy include diabetes mellitus, renal disease and vitamin deficiency (Barohn, & Amato, 2013). Damage to peripheral nerves is thought to be due to multiple factors, including release of cytokines, increase in oxidative stress, nerve tissues with increased glycation, and a metabolic cascade triggered by increased polyol flux (Yagihashi, Mizukami, & Sugimoto, 2011). The peripheral neuropathy associated with diabetes is usually irreversible, but tight glycemic control is needed to prevent neuropathy or slow the progression of disease (Hicks, & Selvin, 2019). Symmetric, progressive polyneuropathy affects approximately 70% of patients with end-stage renal disease and is worse in patients who also have diabetes (Ropper et al., 2014). The etiology is not well understood but may be due to buildup of toxins causing axonal degeneration. Hemodialysis does not improve this neuropathy, but kidney transplantation usually results in neurologic recovery (Ropper et al., 2014). Nutritional deficiency can also cause neuropathy. The most common vitamin deficiencies causing peripheral neuropathy (as well as central nervous system complications) are vitamin B12, copper, thiamine, vitamin E, and vitamin B6 (Hammond, Wang, Dimachkie, & Barohn, 2013). Patients with alcohol use disorder are at high risk for nutritional polyneuropathy (Ropper et al., 2014). Treatment is to replace the vitamin deficiency to halt or reverse the progression of neuropathy (Hammond et al., 2013).
2.7Mechanical neuropathies
Mechanical neuropathies, also called entrapment neuropathies, occur at anatomic locations where nerves are vulnerable to compression. As described above this includes radiculopathies due to stenosis and common mononeuropathies such as median neuropathy at the wrist, ulnar neuropathy at the elbow or peroneal neuropathy at the fibular head (Hobson-Webb, & Juel, 2017). For entrapment neuropathies, the treatment is releasing the compression, often with surgery.
2.8Neuropathies associated with neoplasm
Cancer can cause neuropathies by compression or infiltration of nervous tissues. Non-Hodgkin’s lymphoma can infiltrate peripheral nerves causing plexopathy, mononeuropathy or polyneuropathy (Kelly, & Karcher, 2005). Paraneoplastic neuropathy can present over weeks to months, often causing a symmetric distal sensorimotor polyneuropathy. Anti-Hu antibodies trigger inflammatory damage to the nerves, and small cell lung cancer is the most common malignancy associated with paraneoplastic neuropathies (Ropper et al., 2014). Treatment focuses on the underlying malignancy, but prognosis is poor.
2.9Toxic and idiopathic neuropathies
Several toxins cause subacute neuropathies. These include arsenic, lead, lithium and gold. Platinum is used in chemotherapy drugs such as cisplatin and causes sensorimotor polyneuropathy. There are a number of medications such as isoniazid and amiodarone that cause peripheral neuropathy (Ropper et al., 2014). The final classification of neuropathies is idiopathic. Estimates of the prevalence of idiopathic neuropathy vary. One retrospective study of 205 cases of neuropathies referred to an academic center had 49 cases (24%) that remained undiagnosed after extensive evaluation (Dyck, Oviatt, & Lambert, 1981).
3Treatment
3.1Pharmacological
Neuropathic pain is a common symptom of peripheral neuropathies and continues to be a challenge to manage and often requires a multidisciplinary approach. Up to 50% of patients with diabetic peripheral neuropathy (DPN) may complain of pain which is often a frequent reason for seeking medical attention (Tesfaye et al., 2011). The management of neuropathic pain focuses on treating the symptoms and in certain conditions treating etiological cause can relieve pain (Cavalli, Mammana, Nicoletti, Bramanti, & Mazzon, 2019). It is speculated that poor glycemic control contributes to the genesis of DPN and therefore there is a consensus that strict blood glucose control should be the initial treatment in diabetic neuropathy (Boulton, Malik, Arezzo, & Sosenko, 2004; Tesfaye et al., 2011). Numbness and coldness, also known as negative symptoms of peripheral neuropathy, do not respond to medications used to treat neuropathic pain (Doughty, & Seyedsadjadi, 2018). The updated guidelines from the IASP’s Neuropathic Pain Special Interest Group (NeuPSIG) recommend tricyclic antidepressants, gabapentin or pregabalin, and the SNRI’s venlafaxine or duloxetine as first line treatment for neuropathic pain. Second-line treatments include tramadol, topical lidocaine or high-concentration capsaicin. Finally, strong opioids (Morphine and Oxycodone) and botulinum toxin-A (BTX-A) were included as third-line treatments for peripheral neuropathic pain. Cannabinoids and valproate had weak recommendations against their use in neuropathic pain (Table 1) (Finnerup et al., 2015).
Table 1
Drug treatment for neuropathic pain –updated recommendations from the International Association for the Study of Pain (Finnerup, Attal, & Haroutounian, 2015)
| First-line | Gabapentin, Pregabalin |
| Tricyclic Antidepressants | |
| SNRI- Duloxetine, Venlafaxine | |
| Second-line | Capsaicin 8% patches |
| Lidocaine patches | |
| Tramadol | |
| Third-line | Strong opioids |
| Botulinum toxin-A (BTX-A) | |
| Weak recommendations | Cannabinoids |
| against use | Valproate |
| Strong recommendation | Levetiracetam |
| against use | Mexiletine |
3.1.1Antidepressants
Studies have shown that antidepressants can be used in alleviating neuropathic pain as they share similar neurotransmitters in neuronal analgesic processing pathways (Mika, Zychowska, Makuch, Rojewska, & Przewlocka, 2013; Iqbal, Azmi, Yadav, Ferdousi, Kumar, Cuthbertson, Lim, Malik, & Alam, 2018). In particular TCA’s and SNRI’s have shown to be effective in reducing pain.
Tricyclics: TCAs are thought to indirectly modulate serotonergic, noradrenergic and opioidergic systems in the brain and have been shown to be effective in treatment of painful neuropathy including DPN (Botney, & Fields, 1983; Finnerup, Sindrup, & Jensen, 2010; Mika et al., 2013). Amitriptyline is the most studied TCA with recommended dosage of (25–150 mg daily) (Finnerup et al., 2015). Providers need to practice caution when prescribing TCA’s due to frequency and severity of dose-related side effects including anti-cholinergic properties and adverse cardiovascular events. TCAs are contraindicated in patients with certain cardiac conduction disturbances due to possible QTc prolongation and risk of sudden death at high doses (Ray, Meredith, Thapa, Hall, & Murray, 2004; Cavalli, et al., 2019).
Serotonin and noradrenalin re-uptake inhibitors: SNRI’s relieve neuropathic pain by inhibiting the reuptake of serotonin and norepinephrine (NE) at the synaptic level, with stronger affiliation for NE. Compared to tricyclics, SNRI’s are more selective for serotonin and NE re-uptake transporters and therefore less likely to have adverse side-effects. Duloxetine (dosage 60–120 mg/day) is the most effective in reducing neuropathic pain and is FDA approved for treatment of DPN (Finnerup et al., 2015). Another SNRI, venlafaxine (dosages 150–225 mg/day), has also been well studied and shown to be efficacious in treating painful neuropathy, however, cardiovascular adverse events limit its use (Finnerup, Sindrup, & Jensen, 2010; Tesfaye et al., 2011).
3.1.2Anticonvulsants
There have been many studies done on anticonvulsants in managing neuropathic pain. At this time gabapentin (1200–3600 mg, in three divided doses) and pregabalin (300–600 mg, in two divided doses) are the two main anticonvulsants used to treat peripheral neuropathy (Table 2) (Finnerup et al., 2015; Doughty, & Seyedsadjadi, 2018). These anticonvulsants, which are similar in structure to gamma-aminobutyric acid neurotransmitter (GABA), inhibit the alpha 2 delta subunit receptor on presynaptic voltage-gated calcium channels and subsequently reduce the release of stimulatory neurotransmitters (Eisenberg, River, Shifrin, & Krivoy, 2007; Fudin, & Jeffrey 2017). Gabapentin and pregabalin are both approved by Food and Drug Administration (FDA) for treating neuropathic pain and considered first-line by NeuPSIG (Finnerup et al., 2015; Cavalli et al., 2019). Based on the 2015 systemic review and meta-analysis, NeuPSIG also recommends extended release gabapentin (1200–3600 mg, in two divided doses) as first-line treatment of neuropathic pain (Finnerup et al., 2015). Gabapentin and pregabalin are both renally excreted, therefore, need to be used with caution in patients with renal disease (Eisenberg et al., 2007). Some other common side-effects include dizziness, somnolence, confusion, and peripheral edema (Doughty, & Seyedsadjadi, 2018). Topiramate, carbamazepine and oxcarbazepine haven shown to have poor safety profile and other anticonvulsant drugs had minimal evidence of efficacy in treating neuropathic pain (Finnerup et al., 2015).
Table 2
First-line pharmacologic agents for the management of neuropathic pain in patients with neuropathy (Doughty, & Seyedsadjadi, 2018)
| Name | Starting dose | Goal dose | Maximum dose | Side effects |
| Gabapentin* | 100 mg TID or 300 mg bedtime | 300 mg TID | 3600 mg/d | Dizziness, sedation, confusion, peripheral edema |
| Pregabalin | 75 mg BID | 150 mg BID | 600 mg/d | Dizziness, sedation, confusion, peripheral edema |
| Amitriptyline/Nortriptyline | 10–25 mg bedtime | 50–100 mg bedtime | 150 mg/day | Dry mouth, sedation QTc prolongation. Amitriptyline has more anticholinergic effects |
| Duloxetine | 30 mg/d | 60 mg/d | 120 mg/day | Nausea, dyspepsia, constipation, sedation, dry mouth, sexual dysfunction |
| Venlafaxine | 37.5 mg/d (XR) | 150 mg/d (XR) | 225 mg/d | Nausea, dyspepsia, constipation, sedation, dry mouth, sexual dysfunction |
XR- extended release. *Gabapentin XR can be given 1200–3600 mg, in two divided doses.
3.1.3Topical treatments
Topical agents are not as potent as systemic treatments but often preferred due to tolerability and safety profile. Lidocaine patches and high-concentration capsaicin are recommended as second-line agents in patients with peripheral neuropathic pain (Finnerup et al., 2010; Cavalli et al., 2019). By local blockage of voltage-gated sodium channels, Lidocaine patches stabilize neuronal membranes and therefore inhibit nociceptive impulses (Cheville et al., 2009; Cavalli et al., 2019). Capsaicin is a potent selective agonist for the transient receptor potential vanilloid 1 receptor (TRPV1). This receptor is usually found on nociceptive neurons with small-diameter (Babbar, Marier, Mouksassi, Beliveau, Vanhove, Chanda, & Bley, 2009; Finnerup et al., 2010). Initial application of capsaicin activates these small fibers, however, repeated topical application of capsaicin leads to desensitization and degeneration of epidermal nerve fibers (ENFs), mostly C-fiber nociceptors. This effect on ENFs makes capsaicin effective in treating neuropathic pain syndromes (Nolano, Simone, Wendelschafer-Crabb, Johnson, Hazen, & Kennedy, 1999; Babbar et al., 2009; Finnerup et al., 2010; Kennedy et al., 2010). Although low-concentration capsaicin can be effective in pain relief, high-concentration dosing allows for longer duration of action with just one administration (Table 3) (Low, Opfer-Gehrking, Dyck, Litchy, & O’Brien, 1995; Kennedy et al., 2010). Topical ketamine, which can block N-methyl-D-aspartate (NMDA) receptors, has also been evaluated in treatment of neuropathic pain (Lodge, 2009). Mahoney et al. studied ketamine 5% in treatment of DPN and found that it was no more effective than placebo (Mahoney, Vardaxis, Moore, Hall, Haffner, & Peterson, 2012). In a retrospective study of patients with post-herpetic neuralgia (PHN), topical ketamine lead to pain relief in 15 of 23 subjects (Quan, Wellish, & Gilden, 2003). In a recent systemic review, compounded topical amitriptyline 4% with ketamine 2% (AmiKet) demonstrated benefits in treating PHN and DPN (Sawynok, & Zinger, 2016). There is still need for large randomized trials to further study the potential benefits of both compounded agents and topical ketamine in treatment of peripheral neuropathy.
Table 3
Pharmacologic agents for the management of neuropathic pain - IASP weak recommendation (Finnerup et al., 2015)
| Capsaicin 8% patches | One to four patches to the painful area for 30–60 min every 3 months |
| Lidocaine 5% patch | One to three patches to the region of pain once a day for up to 12h |
| Tramadol* | 200–400 mg, three divided doses |
| Botulinum toxin A (subcutaneous) | 50–200 units to the painful area every 3 months |
*Extended release tramadol can be given in two divided doses.
3.1.4Opioids
Opioids can relieve pain in both central and peripheral nervous system by agonizing mu-opioid receptors (Williams, 2008). Tramadol is a weak mu-opioid agonist and a serotonin and NE reuptake inhibitor. By inhibiting the reuptake of serotonin and NE, Tramadol demonstrate similar properties as SNRIs in treating neuropathic pain. Tapentadol, which is the only opioid FDA approved to treat neuropathic pain, has a greater affinity for mu-opioid receptors compared to tramadol (Smith, 2012). NeuPSIG considers tramadol a second-line agent while recommendations for Tapentadol are inconclusive due to discrepancies in clinical data. Strong opioids such as oxycodone, morphine, fentanyl, and methadone are now recommended as third-line agents per NeuSIG guidelines, which is a revision from previous second-line designation (Finnerup et al., 2015). This change is mainly due to risk for abuse and other adverse side-effects particularly in administration of higher doses of these medications (Finnerup et al., 2015).
3.1.5Botulinum toxin
Botulinum toxin (BTX), derived from bacterium Clostridium botulinum, is a neurotoxin which is commonly used for the treatment of focal muscle hyperactivity in particular dystonia and spasticity. BTX has seven antigenically variant serotypes (A-G) of which Botulinum toxin type A (BTX-A) is the most well-known and studied (Oh, & Chung, 2015). BTX is thought to inhibit neural transmission by inhibiting synaptic exocytosis of acetylcholine, however, there are some studies that suggest BTX-A may have analgesic properties possibly by inhibiting neurogenic inflammation (Handwerker, 2006; Ranoux, Attal, Morain, & Bouhassira, 2008). Subcutaneous, local injection of BTX-A has been shown to be effective in patients with focal peripheral neuropathic pain and allodynia (Ranoux et al., 2008) and painful diabetic neuropathy (Table 3) (Yuan, Sheu, Yu, Chen, Tseng, Chang, & Hu, 2009). Given overall weak quality of evidence of efficacy, BTX-A is currently included as a third-line treatment of peripheral neuropathic pain by NeuPSIG, and could be considered in refractory cases (Finnerup et al., 2015). Based on literature database search, BTX serotypes (B-G) have not been studied in treating neuropathic pain.
3.1.6Cannabinoids
There are number of studies that suggest cannabis may be effective in treatment of peripheral neuropathy (Wilsey, Marcotte, Tsodikov, Millman, Bentley, Gouaux, & Fishman, 2008; Ellis, Toperoff, Vaida, van den Brande, Gonzales, Gouaux, Bentley, & Atkinson, 2009; Ware, Wang, Shapiro, Robinson, Ducruet, Huynh, Gamsa, Bennett, & Collet, 2010). The endogenous mammalian cannabinoid system is also involved in modulating pain transmission in the nociceptive pathway in addition to effects on central nervous system (Modesto-Lowe, Bojka, & Alvarado, 2018). Cannabinoid receptors CB1 and CB2 are located throughout the central and peripheral nervous system and in organs and tissues (Grotenhermen, 2006). Cannabinoids are thought to reduce pain by modulating CB1 receptors to impede pain conduction, while activation of CB2 receptors to decreases the release of nociceptive agents (Modesto-Lowe et al., 2018). Wallace, Marcotte, Umlauf, Gouaux, & Atkinson (2015) noted improvement in diabetic peripheral neuropathy with use of THC cannabis, with higher doses having greater analgesic effect. They did, however, notice increase adverse side-effects such as decline in attention and working memory and poorer scores in quick task-switching (Wallace et al., 2015). At this time there still needs to be further investigation to better evaluate the long-term benefits and adverse effects of cannabis use for neuropathic pain. NeuPSIG provides weak recommendation against the use of cannabinoids in neuropathic pain mostly due to long-term negative results of possible abuse and mental health risk in the susceptible users (Finnerup et al., 2015).
3.2Interventional and neuromodulatory techniques for neuropathic pain management
Neuropathic pain (NP) is often a debilitating disease refractory to pharmacologic and noninterventional treatment requiring alternative treatment approaches.
Interventional treatments, are defined as “invasive procedures involving delivery of drugs into targeted areas, or ablation/modulation of targeted structures for the treatment of persistent pain.”
The recent opioid epidemic has triggered a resurgence of interest in injection therapies for pain.
Interventional injections can be categorized into three groups 1) diagnostic, 2) therapeutic and 3) palliative. Injections in the diagnostic group are used to help identify the source of pain to assist with treatment planning. Differential diagnostic nerve blocks can help differentiate pathologic ongoing pain processes involving somatic pain, visceral pain, sympathetic mediated pain, central pain and even placebo-responsive pain.
They can be used to decide which nerves to focus additional interventional/surgical interventions and to predict the level of side effects or complications prior to a permanent intervention. An example would be performing a temporary diagnostic celiac plexus block–prior to performing the neurolytic permanent celiac plexus block in order to ensure no permanent life-threatening hypotension. Diagnostic blocks while adding value to the treatment plan have limitations and always need to be viewed in the context of the patients clinical, radiological, laboratory, physical examination and psychosocial status. Limitation examples include missing the pain contribution of the dorsal root ganglia (DRG) when only injecting the peripheral nerve, and missing individual anatomical nerve variations despite image assistance, The second and third groups are the most utilized and involves using injections of substances, or electricity to treat or palliate pain.
Since 1964 a wide variety of techniques have been used to perform differential diagnostic nerve blockade (Table 4) in patients who have elusive pain diagnoses (Winnie, & Candido, 2007). It aids in identifying the relative contributions of somatic, visceral, sympathetic and other types of pain for focused treatment. Initially it involved dosing the epidural space with varying concentrations of local anesthetic over time and determining at which concentration painful responses subsided. Subsequent techniques include the “anatomical” approach (blocking specific nerves sequentially) and the Opioid approach that hoped to remove bias by eliminating the patients awareness of the signs of local anesthetic during the trial (Cherry, Gourlay, McLachlan, & Cousins, 1985). There is no data to suggest that any of these techniques are superior. The limitations have not precluded continued use of diagnostic differential blockade (Table 5).
Table 4
Differential diagnostic nerve blocks
| Epidural | Placebo responsive pain | Sympathetic pain | Somatic pain | Visceral pain | Central pain |
| Normal saline | Pain relief | No relief | No relief | No relief | No relief |
| Lidocaine 0.5% | Pain relief | Sustained relief | No relief | Pain relief | No relief |
| Lidocaine 1% | Pain relief | Sustained relief | Pain relief | Pain relief | No relief |
| Lidocaine 2% | Pain relief | Sustained relief | Pain relief | Pain relief | No relief |
Our clinic typically uses the anatomic approach, but we typically begin with sympathetic blockade and end with somatic blockade and without a placebo trial.
Table 5
Anatomical approach for differential diagnosis
| Painful area | Placebo block | Sympathetic block | Somatic block |
| Headache/iodiopathic facial pain | Normal saline | Sphenolantine ganglion | |
| Head/Neck | Normal saline | Stellate ganglion | Trigeminal nerves, C2 blocks |
| Neck | Normal saline | Stellate ganglion | Cervical plexus |
| Upper extremity | Normal saline | Stellate ganglion | Brachial plexus and peripheral nerves |
| Lower extremity | Normal saline | Lumbar sympathetic ganglion | Lumbar plexus and peripheral nerves |
| Thoracic | Normal saline | Thoracic sympathetic ganglia | Thoracic paravertebral, intercostals, erector spinae |
| Abdominal (esophagus to desc colon, liver, pancreas, gallbladder, stomach, spleen, kidneys, small intestine, adrenals) | Normal saline | Celiac plexus | Thoracic paravertebral, intercostals, erector spinae transversus abdominis plane block |
| Pelvic (lower sigmoid colon, rectum, testicles, ovaries, uterus) | Normal saline | Superior hypogastric plexus | Lumbar paravertebral |
| Pelvic (perineum) | Normal saline | Inferior hypogastric plexus (ganglion of impar) | Caudal epidural pudendal |
4Therapeutics
Our clinic typically uses the anatomic approach, but we typically begin with sympathetic blockade and end with somatic blockade and without a placebo trial.
Head and neck injections target a variety of nerves and ganglia. Neuropathic painful conditions warranting these injections include trigeminal autonomic cephalgias, migraines, cluster headaches, and intractable orofacial pain. A sphenoplantine ganglion block (SPG) is the first choice for intractable headaches and can be performed transnasally, transorally, and endoscopically. Subsequently a radiofrequency ablation (RFA) can be performed for prolonged relief. A retrospective review of patients with cluster headache treated by RFA 60% experienced complete pain relief (Sanders, & Zuurmond, 1997). An initial trigeminal nerve block for diagnosis and management of intractable trigeminal of the SPG, neuralgia is indicated after failure of pharmacologic management. This procedure can also predict prognosis for percutaneous Gasserian ganglion neuroloysis, trigeminal electrical stimulation and surgical neurolysis. Flouroscopically guided neurolysis of the Gasserian ganglion can be performed in a variety of methods including an interventionalist approach (Percutaneous Glycerol Rhizolysis, Percutaneous Radiofrequency Thermocoagulation, and Pulsed Radiofrequency Ablation of the Gasserian Ganglion). In a series review Radiofrequency Thermocoagulation has the least post procedure dysthetic complication rates (Peng, Wiley, Liang, & Bellingham, 2010).
Diagnosing orofacial pain syndromes requires more specific selective blockade (Table 7). Maxillary and Mandibular nerve blocks are performed with a similar technique but with goals of fluoroscopically entering the foramen rotundum for the maxillary and entering mandibular nerve near the ptyergoid plate or via the mandibular notch. The mandibular block stays outside of the foramen in order to differentiate results from the maxillary. Electrical stimulation causing “jaw jerk” can assist the mandibular nerve block localization.
The sympathetic innervation to the head and neck involves the cervical sympathetic ganglia. Blocking these fibers can assist with determining what component of head and neck pain is sympathetically maintained pain versus sympathetically independent pain. Diagnoses include Complex Regional Pain Syndromes, Herpetic Zoster and Post Herpetic Neuralgia, Phantom Limb Pain, Hyperhydrosis, Chronic Intractable Angina, a multitude of Peripheral Neuropathies and more recently Post Traumatic Stress Disorder and excessive Menopausal related “hot flashes”. The stellate block is the most common performed and involves placing injectate at the middle cervical ganglion using fluoroscopy, ultrasound or both. The location is at the anterolateral space above the longus coli muscle at the level of C6. Ultrasound has demonstrated larger safety profile than fluoroscopy alone. This is secondary to direct visualization of vessels, nerve roots, esophagus and thyroid. When performing neurolysis with chemicals or radiofrequency a posterior approach may be considered which is further away from the cervical plexus and less likely to lead to a permanent Horner’s syndrome.
The upper and lower peripheral nerves and the nerves of the trunk that are commonly treated for neuropathic pain by interventional pain providers include suprascapular, axillary, intercostal, the “border nerves” (genitofemoral (GH), ilioinguinal (IL), iliohypogastric (IH), lateral femoral cutaneous, piriformis, pudendal and branches of the sciatic and femoral nerves.
The suprascapular nerve block is used in a variety of nociceptive syndromes (bursitis, capsular tear, glenohumeral arthritis, etc.) but its neuropathic pain indication is primarily due to traumatic or compressive suprascapular neuropathy. The ideal injection target is the floor of the scapular spine between the suprascapular notch and spinoglenoid notch. This technique avoids pneumothorax and can be performed in the 8% of population who have no notch. It is a smaller space than the notch and thus the medication is contained more easily around the nerve with less volume when compared to entering the notch proper and escaping into the surrounding brachial plexus (Grotenhermen, 2006). Although most peripheral nerve blocks can use fluoroscopy, CT or peripheral nerve stimulation to guide the needle - the common assistive tool is ultrasound.
The intercostal nerve can be used to diagnose and/or treat neuropathic pain of the chest wall and abdomen. It can also predict success for neuroablative procedures such as cryoablation for post thoracotomy and post mastectomy pain. Fluoroscopy does not affect pneumothorax risk when compared to direct visualization of the pleura with ultrasound guidance (Shanti, Carlin, & Tyburski, 2001).
The border nerves (GH, IL, IH), are injected for diagnosis of pain involving the abdomen and thigh. This includes neuropathic painful diseases such as post herniorrhapy pain, post appendectomy pain, abdominal wall/pelvic wall trauma, and after low transverse incisions. Patients may complain of groin pain that extends into the testicle in men and the labia in women and travelling into the medial thigh.
Lower limb blocks can be used to diagnose and treat neuropathic pain states including traumatic and compressive neuropathies of the sciatic, obturator, femoral, fascia iliaca, saphenous and ankle. Ultrasound increases the success and performance time of lower limb injections (Marhofer, Schrögendorfer, Koinig, Kapral, Weinstabl, & Mayer, 1997).
The lumbar plexus injection blocks the anterior rami from T12 through L4 and affects pain in the anterior lower extremity. The large volume of local anesthetic required in lumbar plexus injections has important risks and unintended epidural spread, total spinal anesthetics, intravascular injections, seizures, cardiac arrests and deaths have been reported post procedure (Gadsden, Lindenmuth, Hadzic, Xu, Somasundarum, & Flisinski, 2008).
The femoral nerve is used in mononeuropathies involving any of the branches of the femoral nerve and can be used for treatment or to predict beneficial locations for pulsed radiofrequency or peripheral nerve stimulating implants. Of note diagnostic blockade does not necessarily predict outcome for pulsed radiofrequency or peripheral nerve simulation but is more important as a relative marker for where to begin targeting treatment efforts. Ultrasound guidance reduced the risk of vascular puncture by 80% (Abrahams, Aziz, Fu, & Horn, 2009).
Selectively attempting to block the obturator nerve for obturator neuropathy is difficult and painful according to most providers. If it must be performed ultrasound improves the process. The saphenous nerve block is the final branch of the femoral nerve and it is blocked for saphenous compressive/traumatic mononeuropathies for treatment and for planning cryotherapy or peripheral nerve stimulation. It treats pain in the anteromedial lower extremity ending in the distal medial malleolus. Ultrasound guidance has popularized this injection because of improved success rates.
The sciatic nerve block (including at the piriformis muscle location) is commonly used in interventional pain clinics. The most commonly used techniques for chronic sciatic neuropathic pain are located at the piriformis, the infragluteal, popliteal and the distal branches of the sciatic (tibial and peroneal nerves). Sciatic nerves are identified via landmark, with or without peripheral nerve stimulation, fluoroscopy or ultrasound assistance. Ultrasound guidance has been shown to decrease total volume of local anesthetic required with decreased incidence of the life -threatening syndrome Local Anesthetic Systemic Toxicity (LAST) (Barrington, & Kluger, 2013).
The ankle block (sural, superficial peroneal (SP), deep peroneal (DP), saphenous(Saph) and tibial nerves) is a landmark based block that is typically assisted by ultrasound used for evaluating neuropathic pain of the foot.
Lumbar sympathetic blockade (LSB) is used to diagnose and treat sympathetically maintained pain of the lower extremity. It can also predict the response to sympathetic denervation surgically, chemically or with radiofrequency ablation. Lumbar sympathetic blockade interrupts sympathetic efferent fibers and spares somatic nerves. Disease states receiving this injection include all peripheral neuropathies of the lower extremity, Complex Regional Pain Syndromes, and ischemic neuropathic emergencies (i.e. compartment syndromes).
Visceral pain is one of the most difficult chronic neuropathic pains to manage. Since viscera contain a multitude of innervation sources it has very diffuse and poorly localized pain which makes diagnosis more challenging and time consuming. Visceral pain is often secondary to organ trauma such as ischemia, torsion, contraction or traction. The groups of nerves we target for visceral pain are the Sphlanchnic Plexi, Celiac Plexus, Superior Hypogastric Plexus, and Inferior Hypogastric Plexus (Ganglion Impar/Ganglion of Walther). These injections can be solely therapeutic, and they also can predict response to neurolysis chemically or surgically. Patient selection is controversial in the non-malignant patient with visceral pain. The celiac plexus can be accessed via anatomical landmarks, ultrasound assistance, fluoroscopy, CT guidance or through the gastric wall with using endoscopy. Aggressive fluid management is provided throughout the periprocedure period as orthostatic hypotension approaches 50% occurrence and can continue for up to 5 days post procedure. Other complications include pneumothorax, backache, intractable diarrhea, abdominal aortic dissection, paraplegia (secondary to arterial vasospasm). Hematuria, pleurisy and intractable hiccups. Meta analyses revealed that 90% of patients at 3-month interval preceding cancer death had complete pain relief. Studies have shown that the neurolysis of the celiac plexus block was also associated with fewer opioids, better immune functions, and less nausea and vomiting. As an alternative to celiac plexus blockade many providers choose the Greater, Lesser or Least Sphlanchnic nerves for blocking abdominal visceral pain. Lillemoe et al. (Lillemoe, Cameron, Kaufman, Yeo, Pitt, & Sauter, 1993) showed that patients with nonresectable pancreatic cancer lived longer if they received splanchnic neurolysis.
The superior hypogastric block is used for patients with pelvic neuropathic pain involving the lower sigmoid colon, testicles, ovaries, uterus, rectum, bladder and fallopian tubes. The plexus is at the lower border of the L5 vertebral body extending caudally to the upper one third on the sacral first vertebral body of the junction of L5 and S1.
The last visceral pain target is the inferior hypogastric plexus (Ganglion of Impar). The plexi are located where the two sympathetic chains join retroperitoneally at the anterior surface of the sacrococcygeal junction. The ganglia innervate pain in the distal urethra, vulva, distal rectum, and distal third of the vagina as well as parasympathetic innervation to the bladder.
Sympathetic blocks typically use local anesthetics but can also use Botox A.
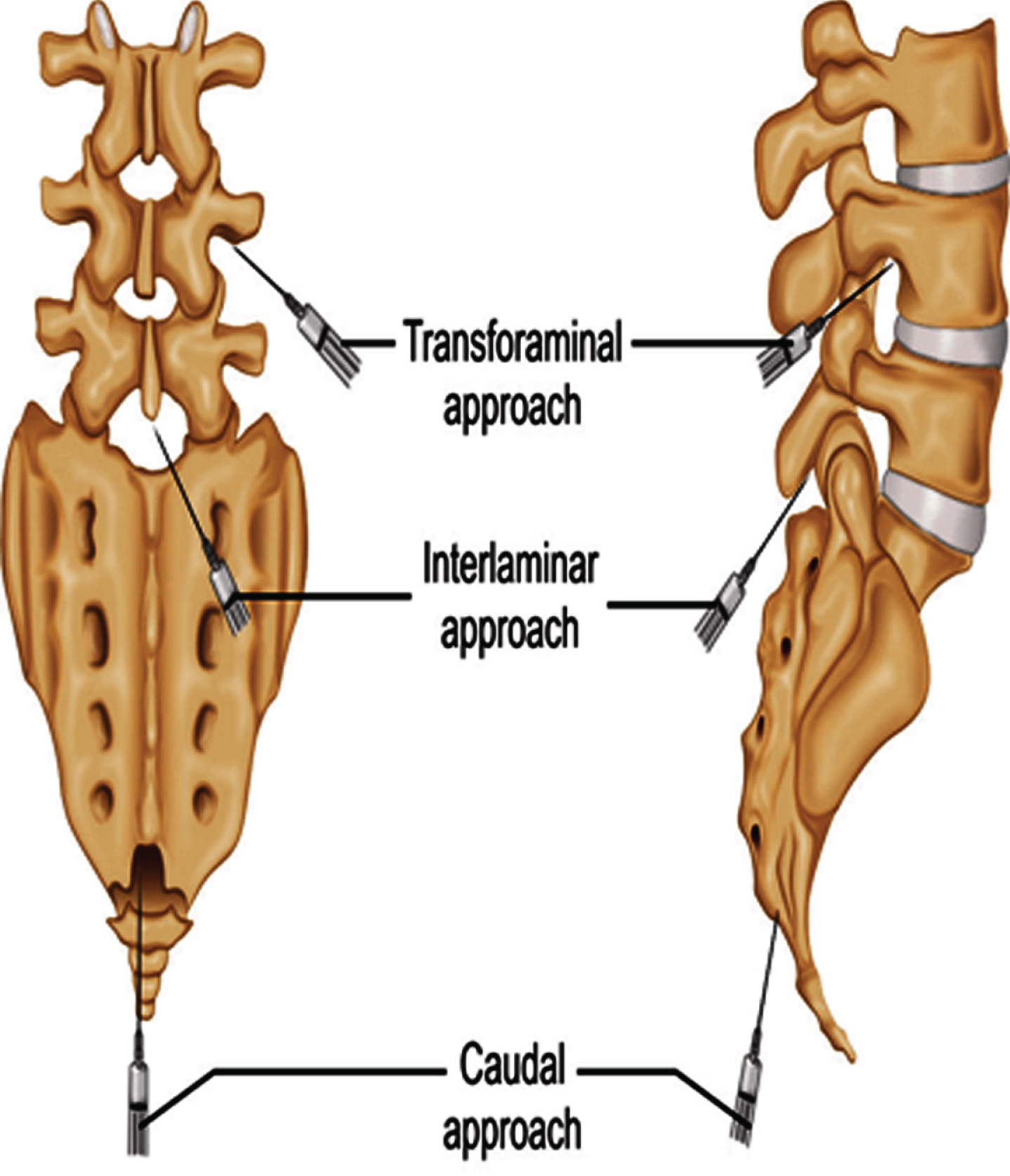
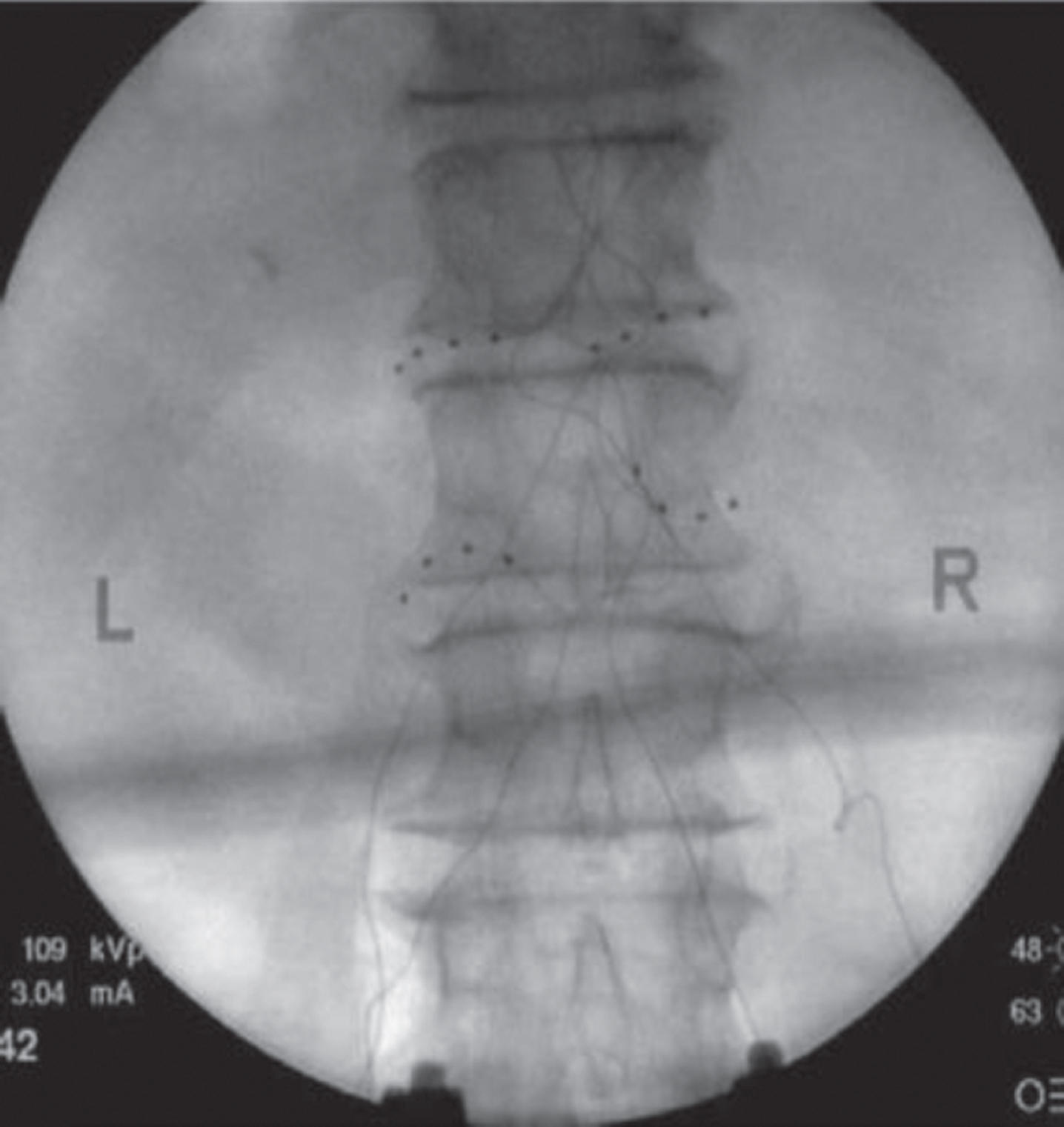
Spinal injections are the most common reason for referrals to interventional pain clinics. Therapeutic epidural injections have historically been the mainstay of treatment utilizing local anesthetics and steroid. More recently the use of injections for neuropathic radiculopathic pain has been refined to address not only the mechanical impingement sources of pain but also the cytokine-mediated radiculitits. Mechanical impingement induces inadequate oxygen and nutritional transfer to spinal nerve and the pro-inflammatory cytokines (i.e. Tumor Necrosis Factor, Interleukins, Chemokines, etc.) Painful diagnoses for spinal epidural corticosteroid injections include radiculopathic pain associated with disc herniation, protrusion, extrusion, spinal stenosis with neurogenic claudication, postsurgical back pain and leg pain secondary to disc material, scarring and/or granulation compressive and chemical intraspinal pathologies. Intraspinal cancer may also respond to epidural steroid. The epidural injections are delivered with fluoroscopic guidance either between the lamina “Interlaminar” (posterior epidural space -greater coverage) (Fig. 1) or within the neuroforamen “transforaminal” (anterior epidural space closer to nerve root) (Figs 2 and 3). The transforaminal epidural steroid injection (TFESI) is more commonly provided as it delivers the medication more closely to the nerve root in comparison to the interlaminar approach. Studies show favor towards TFESI for short term pain reduction in patients with disc herniation and spinal stenosis. More recently some providers use ultrasound guidance as an alternative to fluoroscopic guidance. Similar rates of back pain and symptom resolution has been reported when comparing this treatment with lumbar spine surgery (Atlas, Spine 2005). Guidelines for the use of epidural steroids have been published by several organizations (American Pain Society Clinical Practice Guidelines (APS 2009), American Society of Interventional Pain Physicians (ASIPP 2009), American Society of Anesthesiologists (2010), North American Spine society (NASS 2020) American Society of Regional Anesthesia and Pain Management International Working Group (ASRA 2020) but the available medical literature is overall inconsistent and difficult to interpret. Although most organizations advocate some manner of use in chronic neuropathic pain there are some specialties such as Neurology that have published guidelines that deter the use of intraspinal injections for chronic pain. A cervical intraspinal steroid injection algorithm was published by the World Institute of Pain and includes epidural, cervical nerve root injection and if fails cervical dorsal root ganglion injection. Overall the evidence supports benefit of radicular pain secondary to spinal stenosis or discogenic disease from 2 weeks to 3 months on average. Risks include infections, ischemia and needle injury. Intraspinal bleeding risk can be minimized by adhering to the ASRA guidelines “Interventional Spine and Pain Procedures in Patients on Antiplatelet and Anticoagulant Medications”.
Fig. 1
Transforamenal epidural dye study.
Fig. 2
Interlaminar epidural dye study.
Fig. 3
Needle aproaches.
A variety of substances are instilled epidurally (Table 6) to assist radicular neuropathic pain. Local anesthetics provides improved perfusion to ischemic nerve roots, suppression of ectopic discharges from damaged nerves, and interruption of nociception. In 2019 ASIPP guidelines on Disease Modifying Antirheumatic Drugs (DMARDS) -also coined “biologics”- were produced on their safe and effective use in back pain management. Biologics are used to assist in the repair, and to potentially replace or restore damaged tissue using of autologous or allogenic biologics (Navani et al., 2019). Examples of allogenic substances include 4 drugs Etanercept, Infliximab, Adalimumab, Tocilizumab. Several studies reviewed epidural administration for radicular low back pain. Autologous biologics such as lumbar intradiscal platelet rich plasma (PRP) and mesenchymal stem cell (MSC) has also been studied and has level III evidence supporting its use. Studies of PRP thus far in the epidural space has only level IV evidence. Clonidine an alpha blocker that exhibits some anticytokine activity and has equally effective pain relief in sciatica when compared to triamcinolone but shorter duration effect.
Table 6
Types of intraspinal drugs for neuropathic pain
| Intraspinal location | Inflammation pathway | Biologics | Local anesthetics | Saline/other | Opioid |
| Epidural | Non-particulate steroids Dexamethasone Betamethasone | Etanercept | Bupivicaine Ropivicaine Lidocaine | Clonidine | Rare |
| Subarachnoid Implanted pump | Bupivicaine | Ziconitide Clonidine | (Not first line) Morphine | ||
| Intraventricular (mid brain-brainstem) | Clonidine | Morphine |
The treatment algorithm for neuropathic pain may use neurolysis prior to electrical stimulation or vice versa. The neurolytic options for many of the above listed therapies are induced either chemically, thermally or mechanically. The chemicals used include alcohol, phenol, glycerol and hypertonic saline. Thermal radiofrequency lesioning is performed with specialized needles lesioning at temperatures at 60 degrees Celsius and above. Cryoneuroablative thermal lesions are at temperatures beginning at negative 30 degrees have been successful. Radiofrequency (RFA) at lower temperatures (42 degrees Celsius) is termed Pulsed Radiofrequency and can relieve some forms of neuropathic pain by modulating rather than destroying the nervous tissue. This is often used in areas where neurolysis of a nerve would lead to painful deafferentation pain. It typically has a shorter duration of effect compared to heated RFA and probably works by modulating mRNA and sensitized neural tissues.
Electrical stimulation for neuropathic pain includes three groups of therapies Spinal Cord Stimulation (SCS) (Fig. 4), Peripheral Nerve Stimulation (PNS) (Figs 5 and 6) and Dorsal Root Ganglion Stimulation (DRGS) (Fig. 7). SCS involves the percutaneous placement of an electrode into the posterior epidural space to electrically stimulate the dorsal columns of the spinal cord. The lead is the connected to an external generator that the patient uses for up to seven days as a trial for pain control. After the trial the patient can decide if a permanent device is warranted and an internalized device is placed surgically. The patient can manipulate the devices electrical output via wireless remote for maximum pain control. Recently systems have been designed with external generator devices that are worn on the body therefore not requiring surgical implantation. Certain types of neuropathic pain are challenging to manage via SCS and DRGS is a better choice. These diagnoses include truncal pain, pain of the feet and pelvic pain. At this time DRGS is not approved above the thoracic spinal level. Patients can also have trials of PNS with or without SCS. There are several types of PNS available. One type is a temporary 60-day implant that is placed percutaneously and has an external generator secured to the skin. These have been used in all peripheral nerves with exception of head and neck and have not had complications (no infections/bleeding reported) except a low rate of benign lead fracture. This 60-day trial is intended to provide pain relief both at the time of the trial and for sustained months post implant. Another type of PNS is a totally implanted permanent lead as well as a generator that is surgically placed much like the described permanent SCS. Lastly, another group of PNS is a percutaneous non-surgical permanent lead with an external generator.
Fig. 4
SCS leads.
Fig. 5
PNS leads and ultrasound appearance.
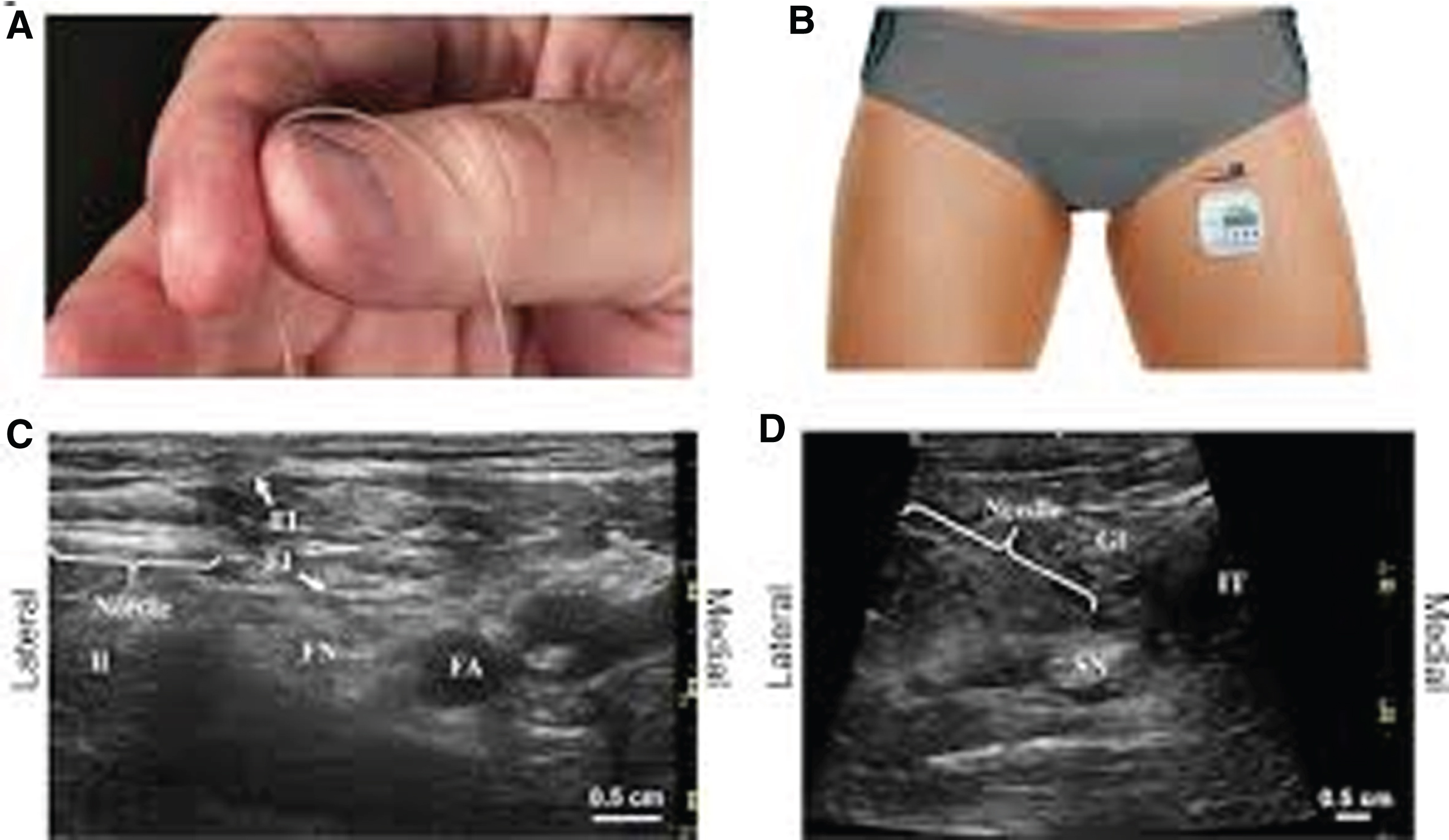
Fig. 6
DRG leads.
The PNS, SCS and DRG stimulation perceived by the patient can be a traditional low frequency (obvious paresthesia to the patient) or bursts of stimulation or high frequency (non-parasthesia) settings. Outcomes in each of these settings have demonstrated increased efficacy with limited complications in SCS and PNS. For Failed Back Surgery Syndrome, Complex Regional Pain Syndrome and peripheral ischemia and angina pain. DRG stimulation outcome is still controversial and requires further study given the increased risk of placing the device. Percutaneous stimulation studies are difficult to design for a variety of reasons and the mechanism of action is felt to multifactorial including suppression of central nervous system excitability, vasodilation and inhibition of sympathetic outflow.
The last technique for neuropathic pain (Table 7) in an interventional pain clinic is typically the Implanted Drug Delivery System (IDDS). This involves and implantable pump that delivers medications into the cerebrospinal fluid. Its goal is to provide a similar drug effect as compared to oral agents but without the dose dependent side effects. This is a permanent device and patient selection is paramount in identifying patients who may benefit. It is typically initiated with a 7-day trial of the drug via either single doses via needle over one to three day or an inpatient hospitalization with an indwelling spinal fluid catheter. Published reports show increased validity of the trial if it includes a placebo and is double -blinded and random. Once the patient has completed the trial and has satisfactory pain relief without extensive side effects he may consider being implanted surgically with permanent device. Drugs for neuropathic pain include Ziconitide and/or local anesthetics. Clonidine has been used in combination with opioids but has profound hypotensive side effects. Opioids are rarely used as a first line for neuropathic pain via the pump (Hamza et al., 2012).
Table 7
Types of pain procedure techniques
| Source of pain | Procedure summary |
| Orofacial neuropathic pain syndrome | Sphenopalantine ganglion injection with pledget transnasally, or via greater palantine foramen orally or infrazygomatic percutaneously with fluoroscopic and/or ultrasound assistance. |
| Maxillary nerve injection fluoroscopically entering the foramen rotundum. | |
| Mandibular nerve injection fluoroscopically with or without ultrasound assistance entering near the Ptyergoid plate or enter via the mandibular notch. Peripheral nerve stimulation with visible “jaw jerk” can assist injection. | |
| Head and neck neuropathic pain syndromes | Stellate ganglion injection with fluoroscopy or ultrasound or a combination at the C6 transverse process anteriorly or using a posterior approach for permanent neurolysis at T1-2. |
| Upper extremity peripheral nerve neuropathic pain syndromes | Stellate ganglion (see above). |
| Suprascapular nerve injection using ultrasound or fluoroscopic assistance within the floor of the scapular spine between the suprascapular notch and spinoglenoid notch (avoids pneumothorax and can be performed in the 8% of patients who have no notch). | |
| Truncal neuropathic pain syndromes | Intercostal nerves (ICN) injected at costal angle of rib 7 cm from midline at upper margin of the rib that is one level below the targeted ICN. Ultrasound guidance in-plane or out of plane with small amounts of hydrodissection to the intercostal muscle planes decreases the risk of pneumothorax. |
| Border nerves (GH, IL, IH) are blocked via ultrasound lateral and superior to the anterior superior iliac spine and superficial to the transversus abdominis muscle. Without ultrasound guidance reports have shown colonic puncture (Jöhr, & Sossai, 1999), vascular injury (Vaisman, 2001) and unintended femoral nerve blockade. Risks include entering the peritoneum, spermatic cord, and testicular artery. Some providers use CT guidance to avoid the ureters and intestines. | |
| Lower extremity peripheral nerve neuropathic pain syndromes | Sciatic subgluteal via ultrasound with or without nerve stimulator in a plane between greater trochanter and ischial tuberosity; Sciatic popliteal via ultrasound at 8 cm proximal and 3 cm lateral to popliteal crease. |
| Obturator nerve visualized with ultrasound guidance medial to the femoral artery at inguinal crease - first dose between adductor longus and brevis then second dose after advancing between adductor brevis and magnus. | |
| Lumbar plexus is accessed posteriorly using peripheral nerve stimulation, fluoroscopy, or ultrasound at the level of the L4 transverse process ending in the psoas compartment. | |
| Femoral nerve is accessed via ultrasound assistance below the level of the inguinal ligament near the groin crease. | |
| Fascia Iliaca is performed after visualizing ultrasonic view of fascia lata, iliaca, and iliacus muscle in one view-injection performed beneath fascia iliaca plane | |
| Saphenous nerve via ultrasound guidance with color doppler can distinguish the descending genicular artery from the saphenous nerve and the injectate is placed between the sartorius and vastus medialis muscles | |
| Ankle block is typically assisted by ultrasound when performing the sural and the posterior tibial injections at the malleoli because those are the only 2 ankle block nerves that reported as improved success with ultrasound assistance and then scanning posteriorly toward the Achilles tendon. The remaining 3 (SP, Saph, DP) are blocked via landmark approach from fanning from medial to lateral malleoli. | |
| Sympathetic blockade | The lumbar sympathetic ganglia are most commonly aggregated at the anterolateral border of the L2 lower third vertebra, L2L3 interspace and the upper third of L3 vertebra. Performed with fluoroscopy or ultrasound guidance the needle traverses the posterior lumbar paraspinals to end at the front of the L3 vertebral body. Blockade verified by demonstration of a 2-3degree Celsius warmer foot in comparison to the control limb. Careful fluid status management is required in all sympathetic blockade procedures as a loss in sympathetic tone can shift effective blood volume away from the heart and great vessels and into the limbs especially when returning to standing after the procedure. Complications: intravascular injection (LAST), subarachnoid injection, Discitis, Psoas Necrosis, Infection, Genitofemoral Neuralgia (especially if performed high at L2), Ureteral stricture, Renal injury, Ejaculatory failure (bilateral lumbar sympathetic blockade). |
| The celiac plexus block is a fluoroscopic, ultrasound or CT guided injection that is accessed at the front of the L1 vertebral body either behind or in front of the abdominal aorta. | |
| The splanchnic nerves are alternatives to the traditional celiac plexus blocks and avoids needlework near the Aorta. Needles are placed behind the diaphragm and at the lower thoracic levels. | |
| The superior hypogastric plexus is at the lower border of the L5 vertebral body extending caudally to the upper one third on the sacral first vertebral body of the junction of L5 and S1. It is approached in similar trajectory as the LSB (above) but has to navigate around the iliac crest. A transdiscal technique can lead to a theoretical discitis but it is not commonly reported. | |
| The inferior hypogastric plexus is injected either via traversing the anococcygeal ligament or the sacrococcygeal joint ending at the lower anterior sacrum. | |
| Spinal injections for neuropathic pain states | Interlaminar Epidural (ILEI) and Transforamenal Epidural (TFESI) injections are typically performed with fluoroscopic guidance and more recently using ultrasound guidance. The needles in both injections are percutaneous with drug entering the central spinal canal but the ILESI is from directly posterior in the interlaminar spaces and the TFESI travels into the neuroforamen of the respective target nerve. TFESI are more helpful in diagnostics as the drug delivered more selectively targets the predicted pain source. |
| Electrical stimulation for neuropathic pain states | Traditional placement of spinal cord stimulation leads involves using fluoroscopy to enter the epidural space percutaneously as described in ILESI and then the lead is advanced within the central spinal canal to the targeted spinal segment (cervical, thoracic lumbar). After a 7–10 day trial of stimulation the patient can be considered for permanent percutaneous or surgical implantation. |
| Dorsal root ganglion stimulation involves placement of the electrical leads into the spinal canal and then advancing the lead such that it begins to exit the neuroforaminal canal providing electrical current to the target nerves dorsal root ganglion. | |
| Peripheral nerve stimulation has several options including percutaneous 60-day implants and surgical permanent implants. The temporary peripheral nerve stimulator leads are typically placed with ultrasound guidance and are secured with biooclusive dressings for 60 days and then explanted for continued pain-relieving effect. The surgical implants involve a diagnostic peripheral nerve block initially and then a surgical implant of the permanent device is placed. | |
| Implanted Drug Delivery System (IDDS) for neuropathic pain states | The most common location of an intrathecal pump is below thoracolumbar but they have also been placed in cervical spine less commonly and very rarely intraventricular (brainstem and mid brain) for maximal supraspinal analgesia especially for head and neck cancer pain. The percutaneous trial is placed into the subarachnoid space either using landmark approach or with the assistance of fluoroscopy or ultrasound guidance. Some providers prefer to dose the epidural space as a trial rather than the subarachnoid space as they are more likely to have fewer unmanageable side effects with epidural dosing. The trial of medications can vary from a single injection or a series of injections or an indwelling catheter for several days. After the trial is successful with appropriate improvement in pain, function and low side effect profile the patient undergoes permanent surgical implantation of the device and the medication pump reservoir. |
In summary, there is are a multitude of interventional techniques available to patients for neuropathic pain and it is an evolving therapeutic arena. Physicians providing these therapies need to use diligent patient selection processes and a multimodal, individualized pain program that supports a strong risk/benefit ratio. The field of interventional pain medicine is still in the infancy stages of published Level 1 data which is mainly secondary to the challenges of designing double blinded randomized controlled protocols in this at risk subject group.
5Conclusion
This article has provided an overview of peripheral neuropathic pain including its prevalence, pathophysiology and treatment. NP affects large segments of patients with a wide variety of disorders and consequently this issue acquires a substantial medical importance. Quality of life for these individuals can suffer (McCarberg, & Billington, 2006). Quality of life parameters adversely affected can include work productivity of both the patient and his or her spouse, ability to interact socially, and impaired mobility. There are substantial costs to the individual and to society from lost work productivity and from the costs of accessing the medical system. Currently there is an expansion of efforts to control these consequences through advances in non-invasive and invasive treatments as detailed above.
Conflict of interest
None to report.
References
1 | Abbott, C. A., Malik, R. A., van Ross, E. R., Kulkarni, J., & Boulton, A. J. ((2011) ). Prevalence and characteristics of painful diabetic neuropathy in a large community-based diabetic population in the U.K. Diabetes care, 34: (10), 2220–2224. https://doi.org/10.2337/dc11-1108 |
2 | Abrahams, M. S., Aziz, M. F., Fu, R. F., & Horn, J. L. ((2009) ). Ultrasound guidance compared with electrical neurostimulation for peripheral nerve block: a systematic review and meta-analysis of randomized controlled trials. British Journal of Anaesthesia, 102: (3), 408–417. https://doi.org/10.1093/bja/aen384 |
3 | Alexander, C. E., & Varacallo, M. ((2020) ). Lumbosacral Radiculopathy. In StatPearls. StatPearls Publishing. |
4 | Argoff, C. E., Cole, B. E., Fishbain, D. A., & Irving, G. A. ((2006) ). Diabetic peripheral neuropathic pain: clinical and quality-of-life issues. Mayo Clin Proc, 81: (4 Suppl), S3–S11. doi: 10.1016/s0025-6196(11)61474-2. |
5 | Babbar, S., Marier, J. F., Mouksassi, M. S., Beliveau, M., Vanhove, G. F., Chanda, S., & Bley, K. ((2009) ). Pharmacokinetic analysis of capsaicin after topical administration of a high-concentration capsaicin patch to patients with peripheral neuropathic pain. Therapeutic Drug Monitoring, 31: (4), 502–510. https://doi.org/10.1097/FTD.0b013e3181a8b200 |
6 | Barohn, R. J., & Amato, A. A. ((2013) ). Pattern-recognition approach to neuropathy and neuronopathy. Neurologic clinics, 31: (2), 343–361. https://doi.org/10.1016/j.ncl.2013.02.001 |
7 | Barrington, M. J., & Kluger, R. ((2013) ). Ultrasound guidance reduces the risk of local anesthetic systemic toxicity following peripheral nerve blockade. Regional Anesthesia and Pain Medicine, 38: (4), 289–299. https://doi.org/10.1097/AAP.0b013e318292669b |
8 | Blaes F. ((2015) ). Diagnosis and therapeutic options for peripheral vasculitic neuropathy. Therapeutic Advances in Musculoskeletal Disease, 7: (2), 45–55. https://doi.org/10.1177/1759720X14566617 |
9 | Botney, M., & Fields, H. L. ((1983) ). Amitriptyline potentiates morphine analgesia by a direct action on the central nervous system. Annals of Neurology, 13: (2), 160–164. https://doi.org/10.1002/ana.410130209 |
10 | Boulton, A. J., Malik, R. A., Arezzo, J. C., & Sosenko, J. M. ((2004) ). Diabetic somatic neuropathies. Diabetes Care, 27: (6), 1458–1486. https://doi.org/10.2337/diacare.27.6.1458 |
11 | Bowen, B. C., Seidenwurm, D. J., Expert Panel on Neurologic Imaging ((2008) ). Plexopathy. AJNR American Journal of Neuroradiology, 29: (2), 400–402. |
12 | Cavalli, E., Mammana, S., Nicoletti, F., Bramanti, P., & Mazzon, E. ((2019) ). The neuropathic pain: An overview of the current treatment and future therapeutic approaches. International Journal of Immunopathology and Pharmacology, 33: , 20587 38419838383. https://doi.org/10.1177/2058738419838383 |
13 | Chapter 43. disorders of the nervous system caused by drugs, toxins, and chemical agents. Ropper A.H., & Samuels M.A., & Klein J.P.(Eds.), (2014). Adams & Victor’s Principles of Neurology, 10e. McGraw-Hill. https://neurology.mhmedical.com/content.aspx?bookid=690§ionid=50910894 |
14 | Cherry, D. A., Gourlay, G. K., McLachlan, M., & Cousins, M. J. ((1985) ). Diagnostic epidural opioid blockade and chronic pain: preliminary report. Pain, 21: (2), 143–152. https://doi.org/10.1016/0304-3959(85)90284-2 |
15 | Cheville, A. L., Sloan, J. A., Northfelt, D. W., Jillella, A. P., Wong, G. Y., Bearden Iii, J. D., Liu, H., Schaefer, P. L., Marchello, B. T., Christensen, B. J., & Loprinzi, C. L. ((2009) ). Use of a lidocaine patch in the management of postsurgical neuropathic pain in patients with cancer: a phase III double-blind crossover study (N01CB). Supportive Care in Cancer: Official Journal of the Multinational Association of Supportive Care in Cancer, 17: (4), 451–460. https://doi.org/10.1007/s00520-008-0542-x |
16 | Colloca, L., Ludman, T., Bouhassira, D., Baron, R., Dickenson, A. H., Yarnitsky, D., Freeman, R., Truini, A., Attal, N., Finnerup, N. B., Eccleston, C., Kalso, E., Bennett, D. L., Dworkin, R. H., & Raja, S. N. ((2017) ). Neuropathic pain. Nature reviews. Disease primers, 3: , 17002. https://doi.org/10.1038/nrdp.2017.2 |
17 | Derry, S., Bell, R. F., Straube, S., Wiffen, P. J., Aldington, D., & Moore, R. A. ((2019) ). Pregabalin for neuropathic pain in adults. The Cochrane Database of Systematic Reviews, 1: (1), CD007076. https://doi.org/10.1002/14651858.CD007076.pub3 |
18 | Doughty, C. T., & Seyedsadjadi, R. ((2018) ). Approach to Peripheral Neuropathy for the Primary Care Clinician. The American Journal of Medicine, 131: (9), 1010–1016. https://doi.org/10.1016/j.amjmed.2017.12.042 |
19 | Drake, R. L., Vogl, W., Mitchell, A.W.M., & Gray, H. ((2010) ). Gray’s Anatomy for students. Philadelphia PA: Churchill Livingstone/Elsevier. |
20 | Dyck, P. J., Oviatt, K. F., & Lambert, E. H. ((1981) ). Intensive evaluation of referred unclassified neuropathies yields improved diagnosis. Annals of Neurology, 10: (3), 222–226. https://doi.org/10.1002/ana.410100304 |
21 | Dydyk, A. M., & M Das, J. ((2020) ). Radicular Back Pain. In StatPearls. StatPearls Publishing. |
22 | Eisenberg, E., River, Y., Shifrin, A., & Krivoy, N. ((2007) ). Antiepileptic drugs in the treatment of neuropathic pain. Drugs, 67: (9), 1265–1289. https://doi.org/10.2165/00003495-200767090-00003 |
23 | Ellis, R. J., Toperoff, W., Vaida, F., van den Brande, G., Gonzales, J., Gouaux, B., Bentley, H., & Atkinson, J. H. ((2009) ). Smoked medicinal cannabis for neuropathic pain in HIV: a randomized, crossover clinical trial. Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology, 34: (3), 672–680. https://doi.org/10.1038/npp.2008.120 |
24 | Finnerup, N. B., Sindrup, S. H., & Jensen, T. S. ((2010) ). The evidence for pharmacological treatment of neuropathic pain. Pain, 150: (3), 573–581. https://doi.org/10.1016/j.pain.2010.06.019 |
25 | Finnerup, N., Attal, N., & Haroutounian, S. ((2015) ). Pharmacotherapy for Neuropathic Pain in Adults: A Systematic Review and Meta-Analysis. Journal of Vascular Surgery, 62: (4), 1091. doi: 10.1016/j.jvs.2015.08.01 |
26 | Fudin, Jeffrey. ((2017) ). “Treatment of Neuropathic Pain.” https://Www.pharmacytimes.com/, www.pharmacytimes.com/publications/health-system-edition/2017/march2017/treatment-of-neuropathic-pain. |
27 | Gadsden, J. C., Lindenmuth, D. M., Hadzic, A., Xu, D., Somasundarum, L., & Flisinski, K. A. ((2008) ). Lumbar plexus block using high-pressure injection leads to contralateral and epidural spread. Anesthesiology, 109: (4), 683–688. https://doi.org/10.1097/ALN.0b013e31818631a7 |
28 | Gregg, E. W., Sorlie, P., Paulose-Ram, R., Gu, Q., Eberhardt, M. S., Wolz, M., Burt, V., Curtin, L., Engelgau, M., Geiss, L., 1999-2000 national health and nutrition examination survey ((2004) ). Prevalence of lower-extremity disease in the US adult population> = 40 years of age with and without diabetes: national health and nutrition examination survey. Diabetes care, 27: (7), 1591–1597. https://doi.org/10.2337/diacare.27.7.1591 |
29 | Grotenhermen, Franjo. (2006). Cannabinoids and the endocannabinoid system. Cannabinoids. 1. |
30 | Halperin, J. J. ((2020) ). Nervous system lyme disease. UpToDate. https://www.uptodate.com/contents/nervous-system-lyme-disease?search=nervous%20system%20lyme%20disease&source=search_result&selectedTitle=1˜150&usage_type=default&display rank=1. Accessed April 28, 2020. |
31 | Hammond, N., Wang, Y., Dimachkie, M. M., & Barohn, R. J. ((2013) ). Nutritional neuropathies. Neurologic Clinics, 31: (2), 477–489. https://doi.org/10.1016/j.ncl.2013.02.002 |
32 | Hamza, M., Doleys, D., Wells, M., Weisbein, J., Hoff, J., Martin, M., Soteropoulos, C., Barreto, J., Deschner, S., & Ketchum, J. ((2012) ). Prospective study of 3-year follow-up of low-dose intrathecal opioids in the management of chronic nonmalignant pain. Pain medicine (Malden, Mass.), 13: (10), 1304–1313. https://doi.org/10.1111/j.1526-4637.2012.01451.x |
33 | Handwerker, H. O. ((2006) ). Nociceptors: neurogenic inflammation. In Cervero F, Jensen TS, eds. Handbook of clinical neurology. Vol 81. Pain. Amsterdam: Elsevier, 23-33. |
34 | Hanewinckel, R., van Oijen, M., Ikram, M. A., & van Doorn, P. A. ((2016) ). The epidemiology and risk factors of chronic polyneuropathy. European Journal of Epidemiology, 31: (1), 5–20. https://doi.org/10.1007/s10654-015-0094-6 |
35 | Hicks, C. W., & Selvin, E. ((2019) ). Epidemiology of Peripheral Neuropathy and Lower Extremity Disease in Diabetes. Current Diabetes Reports, 19: (10), 86. https://doi.org/10.1007/s11892-019-1212-8 |
36 | Hlubocky, A., & Smith, B. E. ((2014) ). Dorsal Root Ganglion Disorders. In: Katirji B., Kaminski H., Ruff R. (eds) Neuromuscular Disorders in Clinical Practice. Springer, New York, NY. https://doi.org/10.1007/978-1-4614-6567-6 23 |
37 | Hobson-Webb, L. D., & Juel, V. C. ((2017) ). Common Entrapment Neuropathies. Continuum (Minneapolis, Minn.), 23(2, Selected Topics in Outpatient Neurology), 487-511. https://doi.org/10.1212/CON.0000000000000452 |
38 | Hu, L. ((2020) ). Treatment of lyme disease. UpToDate. https://www.uptodate.com/contents/treatment-of-lyme-disease?search=treatment%20of%20lyme%20disease&source=search_result&selectedTitle=1˜150%usage_type=default%display_rank=1. Accessed April 28, 2020. |
39 | Iqbal, Z., Azmi, S., Yadav, R., Ferdousi, M., Kumar, M., Cuthbertson, D. J., Lim, J., Malik, R. A., & Alam, U. ((2018) ). Diabetic Peripheral Neuropathy: Epidemiology, Diagnosis, and Pharmacotherapy. Clinical Therapeutics, 40: (6), 828–849. https://doi.org/10.1016/j.clinthera.2018.04.001 |
40 | Jöhr, M., & Sossai, R. ((1999) ). Colonic puncture during ilioinguinal nerve block in a child. Anesthesia and Analgesia, 88: (5), 1051–1052. https://doi.org/10.1097/00000539-199905000-00015 |
41 | Kang, P. B., Shefner, J. M., Nordi, D. R., & Goddeau, R. P. ((2020) ). Overview of hereditary neuropathies. UpToDate. https://www.uptodate.com/contents/overview-of-hereditary-neuropathies#! |
42 | Kelly, J. J., & Karcher, D. S. ((2005) ). Lymphoma and peripheral neuropathy: a clinical review. Muscle & Nerve, 31: (3), 301–313. https://doi.org/10.1002/mus.20163 |
43 | Kennedy, W. R., Vanhove, G. F., Lu, S. P., Tobias, J., Bley, K. R., Walk, D., Wendelschafer-Crabb, G., Simone, D. A., & Selim, M. M. ((2010) ). A randomized, controlled, open-label study of the long-term effects of NGX-4010, a high-concentration capsaicin patch, on epidermal nerve fiber density and sensory function in healthy volunteers. The Journal of Pain: Official Journal of the American Pain Society, 11: (6), 579–587. https://doi.org/10.1016/j.jpain.2009.09.019 |
44 | Kim, H. J., Nemani, V. M., Piyaskulkaew, C., Vargas, S. R., & Riew, K. D. ((2016) ). Cervical Radiculopathy: Incidence and Treatment of 1,420 Consecutive Cases. Asian Spine Journal, 10: (2), 231–237. https://doi.org/10.4184/asj.2016.10.2.231 |
45 | Kuwabara, S., Misawa, S., Mori, M., Tamura, N., Kubota, M., & Hattori, T. ((2006) ). Long term prognosis of chronic inflammatory demyelinating polyneuropathy: a five year follow up of 38 cases. Journal of Neurology, Neurosurgery, and Psychiatry, 77: (1), 66–70. https://doi.org/10.1136/jnnp.2005.065441 |
46 | Lillemoe, K. D., Cameron, J. L., Kaufman, H. S., Yeo, C. J., Pitt, H. A., & Sauter, P. K. ((1993) ). Chemical splanchnicectomy in patients with unresectable pancreatic cancer. A prospective randomized trial. Annals of Surgery, 217: (5), 447–457. https://doi.org/10.1097/00000658-199305010-00004 |
47 | Lodge D. ((2009) ). The history of the pharmacology and cloning of ionotropic glutamate receptors and the development of idiosyncratic nomenclature. Neuropharmacology, 56: (1), 6–21. https://doi.org/10.1016/j.neuropharm.2008.08.006 |
48 | Low, P. A., Opfer-Gehrking, T. L., Dyck, P. J., Litchy, W. J., & O’Brien, P. C. ((1995) ). Double-blind, placebo-controlled study of the application of capsaicin cream in chronic distal painful polyneuropathy. Pain, 62: (2), 163–168. https://doi.org/10.1016/0304-3959(94)00261-c |
49 | Magnus, W., Viswanath, O., Viswanathan, V. K., & Mesfin, F. B. ((2020) ). Cervical Radiculopathy. In StatPearls. StatPearls Publishing. |
50 | Mahoney, J. M., Vardaxis, V., Moore, J. L., Hall, A. M., Haffner, K. E., & Peterson, M. C. ((2012) ). Topical ketamine cream in the treatment of painful diabetic neuropathy: a randomized, placebo-controlled, double-blind initial study. Journal of the American Podiatric Medical Association, 102: (3), 178–183. https://doi.org/10.7547/1020178 |
51 | Marhofer, P., Schrögendorfer, K., Koinig, H., Kapral, S., Weinstabl, C., & Mayer, N. ((1997) ). Ultrasonographic guidance improves sensory block and onset time of three-in-one blocks. Anesthesia and Analgesia, 85: (4), 854–857. https://doi.org/10.1097/00000539-199710000-00026 |
52 | Martyn, C. N., & Hughes, R. A. ((1997) ). Epidemiology of peripheral neuropathy. Journal of Neurology, Neurosurgery, and Psychiatry, 62: (4), 310–318. https://doi.org/10.1136/jnnp.62.4.310 |
53 | McCarberg, B. H., & Billington, R. ((2006) ). Consequences of neuropathic pain: quality-of-life issues and associated costs. The American Journal of Managed Care, 12: (9 Suppl), S263–S268. |
54 | McCorry L. K. ((2007) ). Physiology of the autonomic nervous system. American Journal of Pharmaceutical Education, 71: (4), 78. https://doi.org/10.5688/aj710478 |
55 | Mika, J., Zychowska, M., Makuch, W., Rojewska, E., & Przewlocka, B. ((2013) ). Neuronal and immunological basis of action of antidepressants in chronic pain - clinical and experimental studies. Pharmacological reports: PR, 65: (6), 1611–1621. https://doi.org/10.1016/s1734-1140(13)71522-6 |
56 | Modesto-Lowe, V., Bojka, R., & Alvarado, C. ((2018) ). Cannabis for peripheral neuropathy: The good, the bad, and the unknown. Cleveland Clinic Journal of Medicine, 85: (12), 943–949. https://doi.org/10.3949/ccjm.85a.17115 |
57 | Navani, A., Manchikanti, L., Albers, S. L., Latchaw, R. E., Sanapati, J., Kaye, A. D., Atluri, S., Jordan, S., Gupta, A., Cedeno, D., Vallejo, A., Fellows, B., Knezevic, N. N., Pappolla, M., Diwan, S., Trescot, A. M., Soin, A., Kaye, A. M., Aydin, S. M., Calodney, A. K.,... & Hirsch, J. A. ((2019) ). Responsible, Safe, and Effective Use of Biologics in the Management of Low Back Pain: American Society of Interventional Pain Physicians (ASIPP) Guidelines. Pain Physician, 22: (1S), S1–S74. |
58 | Nolano, M., Simone, D. A., Wendelschafer-Crabb, G., Johnson, T., Hazen, E., & Kennedy, W. R. ((1999) ). Topical capsaicin in humans: parallel loss of epidermal nerve fibers and pain sensation. Pain, 81: (1-2), 135–145. https://doi.org/10.1016/s0304-3959(99)00007-x |
59 | Oh, H. M., & Chung, M. E. ((2015) ). Botulinum Toxin for Neuropathic Pain: A Review of the Literature. Toxins, 7: (8), 3127–3154. https://doi.org/10.3390/toxins7083127 |
60 | Peng, P. W., Wiley, M. J., Liang, J., & Bellingham, G. A. ((2010) ). Ultrasound-guided suprascapular nerve block: a correlation with fluoroscopic and cadaveric findings. Canadian Journal of Anaesthesia = Journal canadien d’anesthesie, 57: (2), 143–148. https://doi.org/10.1007/s12630-009-9234-3 |
61 | Quan, D., Wellish, M., & Gilden, D. H. ((2003) ). Topical ketamine treatment of postherpetic neuralgia. Neurology, 60: (8), 1391–1392. https://doi.org/10.1212/01.wnl.0000055848.00032.39 |
62 | Ranoux, D., Attal, N., Morain, F., & Bouhassira, D. ((2008) ). Botulinum toxin type A induces direct analgesic effects in chronic neuropathic pain. Annals of Neurology, 64: (3), 274–283. https://doi.org/10.1002/ana.21427 |
63 | Ray, W. A., Meredith, S., Thapa, P. B., Hall, K., & Murray, K. T. ((2004) ). Cyclic antidepressants and the risk of sudden cardiac death. Clinical Pharmacology and Therapeutics, 75: (3), 234–241. https://doi.org/10.1016/j.clpt.2003.09.019 |
64 | Rodine, R. J., & Vernon, H. ((2012) ). Cervical radiculopathy: a systematic review on treatment by spinal manipulation and measurement with the Neck Disability Index. The Journal of the Canadian Chiropractic Association, 56: (1), 18–28. |
65 | Said, G., Lacroix, C., Planté-Bordeneuve, V., Le Page, L., Pico, F., Presles, O., Senant, J., Remy, P., Rondepierre, P., & Mallecourt, J. ((2002) ). Nerve granulomas and vasculitis in sarcoid peripheral neuropathy: a clinicopathological study of patients. Brain: A Journal of Neurology, 125: (Pt 2), 264–275. https://doi.org/10.1093/brain/awf027 |
66 | Sanders, M., & Zuurmond, W. W. ((1997) ). Efficacy of sphenopalatine ganglion blockade in 66 patients suffering from cluster headache: a 12- to 70-month follow-up evaluation. Journal of Neurosurgery, 87: (6), 876–880. https://doi.org/10.3171/jns.1997.87.6.0876 |
67 | Sawynok, J., & Zinger, C. ((2016) ). Topical amitriptyline and ketamine for post-herpetic neuralgia and other forms of neuropathic pain. Expert opinion on Pharmacotherapy, 17: (4), 601–609. https://doi.org/10.1517/14656566.2016.1146691 |
68 | Schütz, S. G., & Robinson-Papp, J. ((2013) ). HIV-related neuropathy: current perspectives. HIV/AIDS (Auckland, N.Z.), 5: , 243–251. https://doi.org/10.2147/HIV.S36674 |
69 | Scholz, J., Finnerup, N. B., Attal, N., Aziz, Q., Baron, R., Bennett, M. I., Benoliel, R., Cohen, M., Cruccu, G., Davis, K. D., Evers, S., First, M., Giamberardino, M. A., Hansson, P., Kaasa, S., Korwisi, B., Kosek, E., Lavand’homme, P., Nicholas, M., Nurmikko, T.,... & Classification Committee of the Neuropathic Pain Special Interest Group (NeuPSIG) ((2019) ). The IASP classification of chronic pain for ICD-: chronic neuropathic pain. Pain, 160: (1), 53–59. https://doi.org/10.1097/j.pain.0000000000001365 |
70 | Seo, D. H., Lee, S. J., Hyun, J. K., & Kim, T. U. ((2012) ). A case of herpes zoster peripheral polyneuropathy manifested by foot drop in chronic myeloid leukemia. Annals of Rehabilitation Medicine, 36: (5), 724–728. https://doi.org/10.5535/arm.2012.36.5.724 |
71 | Shanti, C. M., Carlin, A. M., & Tyburski, J. G. ((2001) ). Incidence of pneumothorax from intercostal nerve block for analgesia in rib fractures. The Journal of Trauma, 51: (3), 536–539. https://doi.org/10.1097/00005373-200109000-00019 |
72 | Smith H. S. ((2012) ). Opioids and neuropathic pain. Pain physician, 15: (3 Suppl), ES93–ES110. |
73 | Suarez, G. A., Giannini, C., Bosch, E. P., Barohn, R. J., Wodak, J., Ebeling, P., Anderson, R., McKeever, P. E., Bromberg, M. B., & Dyck, P. J. ((1996) ). Immune brachial plexus neuropathy: suggestive evidence for an inflammatory-immune pathogenesis. Neurology, 46: (2), 559–561. https://doi.org/10.1212/wnl.46.2.559 |
74 | Teo, H. K., Chawla, M., & Kaushik, M. ((2016) ). A Rare Complication of Herpes Zoster: Segmental Zoster Paresis. Case Reports in Medicine, 2016: , 7827140. https://doi.org/10.1155/2016/7827140 |
75 | Tesfaye, S., Vileikyte, L., Rayman, G., Sindrup, S. H., Perkins, B. A., Baconja, M., Vinik, A. I., Boulton, A. J., Toronto Expert Panel on Diabetic Neuropathy ((2011) ). Painful diabetic peripheral neuropathy: consensus recommendations on diagnosis, assessment and management. Diabetes/metabolism Research and Reviews, 27: (7), 629–638. https://doi.org/10.1002/dmrr.1225 |
76 | Tsairis, P., Dyck, P. J., & Mulder, D. W. ((1972) ). Natural history of brachial plexus neuropathy. Report on 99 patients. Archives of Neurology, 27: (2), 109–117. https://doi.org/10.1001/archneur.1972.00490140013004 |
77 | Vaisman J. ((2001) ). Pelvic hematoma after an ilioinguinal nerve block for orchialgia. Anesthesia and Analgesia, 92: (4), 1048–1049. https://doi.org/10.1097/00000539-200104000-00045 |
78 | van Doorn, P. A. ((2013) ). Diagnosis, treatment and prognosis of Guillain-Barré syndrome (GBS). Presse medicale (Paris, France: 1983), 42: (6 Pt 2), e193–e201. https://doi.org/10.1016/j.lpm.2013.02.328 |
79 | Wallace, M. S., Marcotte, T. D., Umlauf, A., Gouaux, B., & Atkinson, J. H. ((2015) ). Efficacy of Inhaled Cannabis on Painful Diabetic Neuropathy. The Journal of Pain: Official Journal of the American Pain Society, 16: (7), 616–627. https://doi.org/10.1016/j.jpain.2015.03.008 |
80 | Wan, E. L., Rivadeneira, A. F., Jouvin, R. M., & Dellon, A. L. ((2016) ). Treatment of Peripheral Neuropathy in Leprosy: The Case for Nerve Decompression. Plastic and Reconstructive Surgery. Global open, 4: (3), e637. https://doi.org/10.1097/GOX.0000000000000641 |
81 | Ware, M. A., Wang, T., Shapiro, S., Robinson, A., Ducruet, T., Huynh, T., Gamsa, A., Bennett, G. J., & Collet, J. P. ((2010) ). Smoked cannabis for chronic neuropathic pain: a randomized controlled trial. CMAJ: Canadian Medical Association journal=journal de l’Association medicale canadienne, 182: (14), E694–E701. https://doi.org/10.1503/cmaj.091414 |
82 | Williams J. ((2008) ). Basic Opioid Pharmacology. Reviews in pain, 1: (2), 2–5. https://doi.org/10.1177/204946370800100202 |
83 | Wilsey, B., Marcotte, T., Tsodikov, A., Millman, J., Bentley, H., Gouaux, B., & Fishman, S. ((2008) ). A randomized, placebo-controlled, crossover trial of cannabis cigarettes in neuropathic pain. The Journal of Pain: Official Journal of the American Pain Society, 9: (6), 506–521. https://doi.org/10.1016/j.jpain.2007.12.010 |
84 | Winnie, A. P., & Candido, K. D. ((2007) ). Differential neural blockade for the diagnosis of pain. In: Waldman SD, editor. Pain management. Philadelphia: Saunders;: 155-166 |
85 | Yagihashi, S., Mizukami, H., & Sugimoto, K. ((2011) ). Mechanism of diabetic neuropathy: Where are we now and where to go? Journal of Diabetes Investigation, 2: (1), 18–32. https://doi.org/10.1111/j.2040-1124.2010.00070.x |
86 | Yuan, R. Y., Sheu, J. J., Yu, J. M., Chen, W. T., Tseng, I. J., Chang, H. H., & Hu, C. J. ((2009) ). Botulinum toxin for diabetic neuropathic pain: a randomized double-blind crossover trial. Neurology, 72: (17), 1473–1478. https://doi.org/10.1212/01.wnl.0000345968.05959.cf |
87 | Zaja̧czkowska, R., Kocot-Kępska, M., Leppert, W., Wrzosek, A., Mika, J., & Wordliczek, J. ((2019) ). Mechanisms of Chemotherapy-Induced Peripheral Neuropathy. International Journal of Molecular Sciences, 20: (6), 1451. https://doi.org/10.3390/ijms20061451 |




