Derealization and motion-perception related to repeated exposure to 3T Magnetic Resonance Image scanner in healthy adults
Abstract
BACKGROUND:
Magnetic Resonance Imaging (MRI) scanning can induce psychological effects. No studies have investigated the role of magnetic vestibular stimulation (MVS) in 3TMRI scanner-induced psychological reactions.
OBJECTIVE:
To assess depersonalization/derealization (DD), state anxiety and motion-perception in a 3TMRI scanner, acutely and long-term.
PARTICIPANTS:
48 healthcare professionals and students were included, after preliminary rejection of claustrophobes and neuro-otology and psychiatry assessments.
PROCEDURES:
Participants completed questionnaires on personal habits, dissociation, anxiety/depression and motion sickness susceptibility. Validated DD and state anxiety questionnaires were administered before and after magnetic exposure twice, entering the bore head and feet first in random order, one week apart. During the following week, dizziness/disorientation was reported daily. One month later, 11 subjects repeated the procedure to assess reproducibility.
RESULTS:
Considerable individual susceptibility was observed, circa 40% of the subjects reported self-motion perception related to the exposure, with variable increase on DD symptoms. Multivariate analysis showed that DD scores after any exposure were influenced by entering the bore “feet first”, motion-perception, and the mean sleep hours/week (MANCOVA, R = 0.58, p = 0.00001). There was no clear effect of scanner exposure on state anxiety, which was related to trait anxiey but not to DD scores. During repeated exposures, about half of all subjects re-entering the scan reported motion-perception, but DD or anxiety symptoms were not consistent.
CONCLUSION:
Psychological effects during 3TMRI scanning result from multiple, interacting factors, including novelty of the procedure (first-exposure effect), motion-perception due to MVS, head/body orientation, sleeping habits and individual susceptibility. Forewarning subjects of these predisposing factors may increase tolerance to MRI scanning.
1Introduction
Magnetic Resonance Imaging (MRI) scans are routinely performed around the globe. The procedure provokes emotional and psychological reactions; anxiety has been reported in up to one third of the subjects [1], and claustrophobia in 2 to 10% [2]. These reactions could be attributed to a complex phenomenon that includes fears of “the unknown”, what the scan might reveal and the enclosed space(3), plus the individuals’ predisposition (trait anxiety) [4] and transitory arousal and apprehension (state anxiety) evoked by the exposure.
MRI scanners use cryogenic superconducting ma-gnets in the range of 0.5 to 7 Teslas (T); by comparison, the Earth’s magnetic field is measured in nanoteslas [5]. Incident symptoms related to 1.5T and 3T MRI scanners have been reported during occupational exposure, related to the number of hours per week working with MRI, shift length, and stress [6]. Associations between scanner strength (1.5T–7T) and transient symptoms mainly relate to vertigo [7], in agreement with recent evidence that static magnetic field≥1.5T interact with endolymphatic electrical currents in the inner ear, provoking sustained pressure onto the cupula of the semicircular canals by Lorentz force [8]. The pattern of vestibular activation is dependent on the vestibular organs orientation with respect to the polarity of the magnetic field; in a resting supine position mainly the horizontal and anterior semicircular canals of each side may be either excited or inhibited (9-10), depending if the subject is introduced to the bore “feet first” or “head first”. In hospitals, 3T magnets are of common use, and those of 7T are being increasingly implemented. Then, unintended magnetic vestibular stimulation (MVS) is becoming more potent and frequent.
Altered perceptions of the self and the environment are termed “dissociation phenomena”. Depersonalization refers to experiences of unreality, detachment, or being an outside observer with respect to one's thoughts, feelings, sensations, body or actions; while derealization refers to experiences of unreality or detachment with respect to the surrounding [11].
Perception of orientation in the environment depends on the availability of sensory inputs. In the absence of helpful visual cues (as in a scanner), distorted vestibular signals may create a misleading frame of reference, giving rise to derealization. Due to the widespread central projections of the vestibular system and the sensory conflict induced by its activation, unilateral vestibular stimulation by caloric stimulus provokes mental phenomena [12, 13]. About half of healthy subjects exposed to caloric vestibular stimulation report derealization, while vestibular patients report replication of their symptoms [12], which can also be provoked by substance intake and sensory or sleep deprivation [13]. On the other hand, after vestibular disease, deficient adaptation often induces chronic dizziness and subjective unsteadiness, accompanied by psychological/cognitive dysfunction [14]. Research on the mental symptoms related to MVS may be of value in understanding the psychological symptoms reported by patients undergoing MRI scanning, as well as those related to sensory conflict in patients with chronic dizziness/unsteadiness.
This study was mainly designed to assess depersonalization/derealization (DD) symptoms and state anxiety during exposure to the static magnetic field of a 3T MRI scanner in normal subjects. We specifically examine if such symptoms relate to self-motion sensations induced by MVS. Additional aims of the study were to explore the reproducibility of these symptoms on repeated magnetic exposure and, finally, whether there are persistent feelings of dizziness/disorientation during the week following scanner exposure.
2Methods
The research protocol was performed according to the declaration of Helsinki and its amendments. It was approved by the Institutional Research and Ethics Committees (R-2019-3601-008); and written informed consent was obtained from all the participants.
2.1Participants
Participants were selected at a Medical Center, among healthcare related students/professionals who were acquainted with the use of an MRI scanner. The first screening to participate included safety screening for MRI procedures [15], and claustrophobia assessment (Claustrophobia Questionnaire, cut-off points: <0.16 for men and <0.56 for women) [2, 16]; subjects with any safety risk for MRI exposure or claustrophobia were not invited to participate in the study.
All the participants denied clinical history of oto-logy/neuro-otology/neurology (including migraine) or psychiatric disease, confirmed through medical interview, neuro-otology evaluation and structured psychiatric interview (Mini-International Neuropsychiatric Interview) [17], with self-administered questionnaires to assess: personal habits by an in-house questionnaire (including alcohol/tobacco consumption and sleeping hours/week), burnout (short-version Burnout Measure) [18]; dissociative experiences (Dissociative Experiences Scale) [19], anxiety and depression symptoms (Hospital Anxiety and Depression Scale) [20] and motion sickness (short-form Motion Sickness Susceptibility Questionnaire) [21].
Although 51 subjects accepted to participate, 3 of them did not complete the study assessments described above, due to personal and work commitments. Thus, 48 subjects completed the study protocol (27 women/21 men; aged 19–34 years); their general characteristics are described in Table 1. Since there was a lack of studies on DD symptoms in a 3T MRI scanner, the sample size was calculated according to a previous study on DD symptoms evoked by caloric vestibular stimulation [12], to assess an increase on the DD score [22] after vestibular stimulation, assuming a standardized size effect of Δ=0.7 and ρ= 0.5; for repeated measures where v = 1 and w = 1, type I error of 0.05 and type II error of 0.2.
Table 1
General characteristics, questionnaire scores and results of vestibular test of the 48 young adults participating in the study
| VARIABLES | |
| Percentage (number) | |
| Handedness | |
| Right | 81% (39) |
| Left | 2% (1) |
| Ambidextrous | 16% (8) |
| Education | |
| Bachelor student | 70% (34) |
| Postgraduate student | 27% (13) |
| University graduate | 2% (1) |
| Tobacco and alcohol | |
| Tobacco smokers | 6% (3) |
| Alcohol use | 47% (23) |
| Corrected Myopia /Astigmatism | 60% (29) |
| Mean | |
| (Standard deviation) | |
| Age (years) | 23.89 (3.87) |
| Sleep hours per week | 41.20 (8.82) |
| Hospital Anxiety and Depression Scale (range 0–42) | |
| Depression score | 0.56 (0.89) |
| Anxiety score | 2.25 (1.97) |
| Total score | 2.81 (2.28) |
| Dissociative Experiences Scale (range 0–100) | 3.52 (3.2) |
| Burnout Measure (range 1–7) | 1.68 (0.52) |
| Claustrophobia Questionnaire (range 0–4) | 0.19 (0.12) |
| Motion Sickness Susceptibility Questionnaire short form (range 0–54) | |
| As a child (before age 12) | 4.39 (4.99) |
| Over the last 10 years | 3.06 (4.14) |
| Total | 7.45 (8.50) |
| Vestibulo-ocular reflex in darkness | |
| Gain at 0.16 Hz | 0.52 (0.12) |
| Gain at 1.28 Hz | 0.95 (0.08) |
| Visual Vertical | |
| Static (°) | 0.03 (0.81) |
| Dynamic right (°) | –3.73 (1.02) |
| Dynamic centre (°) | –0.15 (0.82) |
| Dynamic left (°) | 3.94 (0.92) |
2.2Procedures
The neuro-otology evaluation included ear examination, eye movement recordings (including saccades, smooth pursuit and optokinetic nistagmus), sinusoidal rotation at 0.16 Hz and at 1.28 Hz (60°/s peak velocity), static visual vertical (average of 10 trials) and dynamic visual vertical during unilateral centrifugation (300°/s; 3.5 cm) (I-Portal-NOCT-Professional, Neuro-Kinetics, Pittsburgh).
2.3Exposure to the magnetic field
To control for the right/left side of the excitatory/inhibitory MVS, each subject was exposed to the magnet twice (one week in-between), with safety screening before each exposure, once entering the bore “head-first” and once entering the bore ”feet-first” (always with the head within the bore) in random order. In order to avoid experiment-induced bias or suggestibility, no mention of any mental or physical perceptions related to the exposure was made prior to participation in the study.
At morning (8:00–10:00AM), after a night of sleep, the following sequence was applied:
1. Nearby to the scanner room, participants replied for the first time to questionnaires of DD symptoms (Depersonalization/derealization by Cox & Swinson) [22], and state anxiety (STAI short version) [23].
2. Entering the scanner room, they were placed on the MRI’s table (Magneton Skyra, Siemens, Muenchen) in a conventional supine position, with the closed eyes and covered with a soft eye-mask, and the head supported by a head rest with cushions but no straps,
3. To enable mainly horizontal and anterior semicircular canal stimulation [24, 25], the Reid’s line (from the lateral canthus of the eye to the tragus) was positioned at circa 70° by small adjustments on neck extension,
4. The table was displaced towards the bore of the scanner, at circa 10cm/s, up to a position where the tragus were 20cm within the bore, at the level of the scanner magnet [26].
5. Participants were exposed to the magnetic field during 5 minutes, with the instruction to stay awake and keep the head still, while the examiner waited just outside the bore.
6. Immediately after coming-out from the bore the eye mask was removed, and motion-perception in and just coming out of the bore was assessed (checklist; Appendix 1A).
7. Within the first 3 minutes after coming-out from the scanner, participants replied again to the DD and STAI questionnaires.
8. During the following 7 days, using a structured diary (Appendix 1B) and a daily mobile-message reminder, participants were instructed to self-report on the presence of any dizziness and disorientation as well as whether their daily routine performance differed from usual.
9. A week later, the second exposure was performed, and the full sequence was repeated.
To assess reproducibility of the results, participants were invited to replicate the study protocol one month later; 13 subjects came back, but 2 of them did not complete the procedures due to work commitments. Then, 11 participants with similar characteristics to the whole group (4 women/7 men, 19 to 30 years old) completed the protocol for a second time.
2.4Inventories
The 28-item depersonalization/derealization inventory by Cox & Swinson [22] is an instrument designed to assess this phenomenology in clinically anxiety states, rather than in the context of dissociative disorders, with the purpose of enabling the correlation of symptoms with concurrent neurophysiological variables. The severity of each inventory item is coded on a 5 point scale where 0=does not occur, 1=mild, 2=moderate, 3=severe and 4=very severe. A total score is obtained by the sum of the individual scores (range 0 to 112).
The 6-item state trait anxiety inventory short version is the smallest subset of both anxiety-present and anxiety-absent items from the full-form of the STAI [23]. The severity of each inventory item is coded using a 4 points Likert scale according to the subjective experienced intensity of each described feeling: for anxiety present items 0=not at all, 3=very much so; for anxiety-absent items 0=very much so, 3=not at all. A total score is obtained by the sum of the individual scores (range 0 to 18).
2.5Statistical analysis
Statistical analysis was performed with a significant level of 0.05 (CSS 5.5, Statsoft Inc,Tulsa). In order to assess the psychological responses to the magnetic vestibular stimulation, it was essential to verify in which subjects the intended vestibular stimulation was effective in the 3T MRI scanner; then, subjects were classified according to the report of motion or no motion perception related to the exposure to the magnetic field. Then, to evaluate if the differences among the subgroups were related to differences on the vestibulo-ocular gain, analysis of variance (ANOVA) with Duncan’s test was performed.
Finally, a multivariate analysis of covariance (MANCOVA) was performed to evaluate the differences on the repeated pre- and post- exposure reports of DD symptoms and state anxiety, taking into account both motion perception (vestibular stimulation) and individual predisposition (psychological questionnaires). Additionally, the Pearson’s correlation coefficient was used to detect interaction between the report of DD symptoms and state anxiety.
3Results
3.1Perception of motion in/out of the bore of the MRI scanner
During the two exposures, circa 40% of the subjects perceived self-motion in/out of the bore (20 subjects for the first exposure and 19 subjects for the second exposure). Among them, 31% (n = 15) and 29% (n = 14) respectively, reported motion-perception just while in the bore, 2% (n = 1) and 6% (n = 3) reported motion-perception just coming out of the bore, and 8% (n = 4) and 4% (n = 2) reported motion-perception both in and out of the bore. The reported movement was mainly rotation in the yaw (axial) plane of the head (75% of the times,n = 15), but some subjects reported linear motion along the longitudinal axis of the body (20%, n = 4), and one subject reported a complex movement including linear motion backwards, like falling (5%). The direction of the rotation was consistent with right/left MVS in the bore and it reversed in those who also reported motion just coming-out of the bore. The duration of the perception was variable from 3–300 seconds (typical time circa 5s),
Dizziness was reported by 12% (n = 6) of the subjects during the first exposure and 14% (N = 7) during the second exposure. However, in the first exposure, two subjects who entered the bore “feet first”, reported severe dizziness and nausea, and one of them required medication (dimenhidrinate 50mg). During the second exposure nobody reported nausea. The participant who required medication after the first exposure also reported motion-perception in and out of the bore (circa 1 minute), whereas during the second exposure, she reported mild dizziness with a brief motion-perception of circa 5 seconds.
3.2Vestibulo-ocular reflex assessment
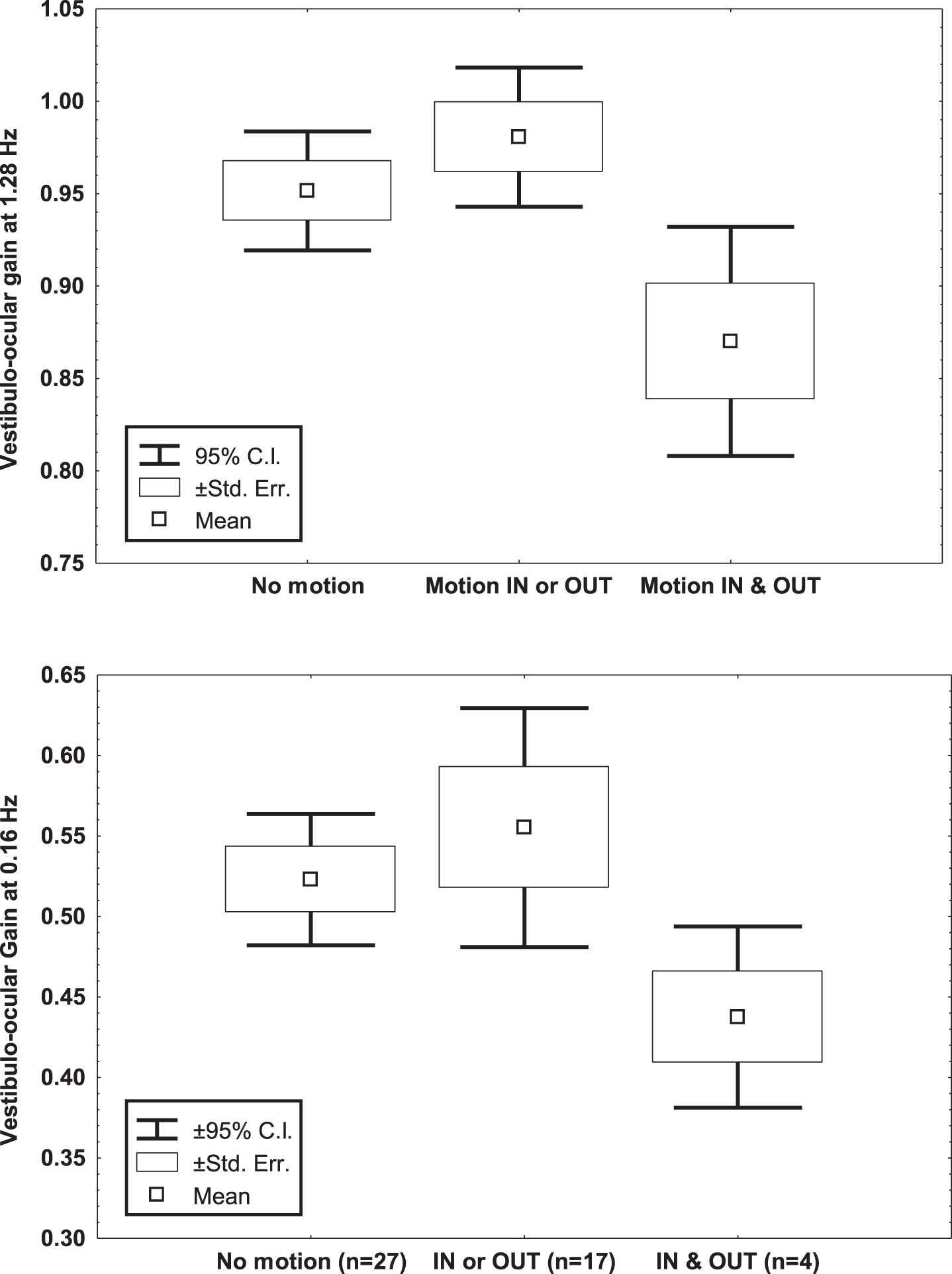
The main vestibular results are shown in Table 1. Analysis on the vestibulo-ocular responses to sinusoidal rotation showed that the subjects who reported motion-perception in and out of the bore had lower vestibulo-ocular gain in darkness than those reporting no motion-perception or motion-perception in or out of the bore. This was significant just for rotation at 1.28 Hz (ANOVA & Duncan test, M = 0.94, p = 0.04 & M = 0.97, p = 0.01), due to variability at 0.16 Hz (M = 0.52, p = 0.16 and M = 0.55, p = 0.06) (Fig. 1). Of note, no differences were observed among subgroups on the righ/left response asymmetry or the response phase.
Fig. 1
Mean, standard error of the mean and 95% Confidence Interval of the mean of the vestibulo-ocular gain to sinusoidal rotation at 0.16 and 1.28 Hz (60°/s peak velocity), of 48 young healthy volunteers, according to the report of motion perception in/ out the bore of the 3T MRI scanner.
3.3Depersonalization/derealization symptoms
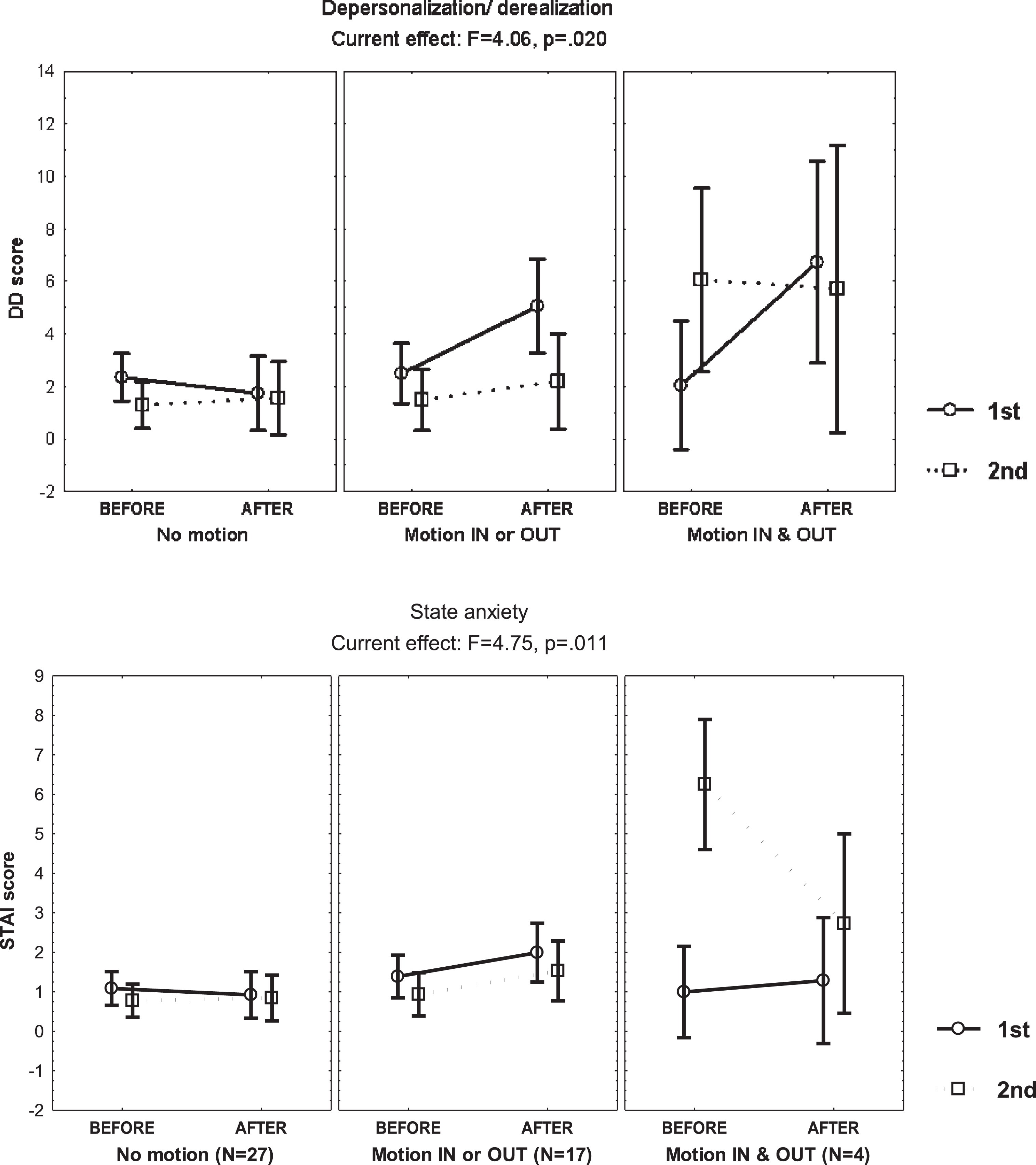
Table 2 shows the DD score observed before and after the exposure, during the first and second exposures, with an increase in the subjects who reported motion-perception in/out of the bore just during the first exposure, and no change in those who denied motion-perception (MANCOVA, F = 4.06, p = 0.02) (Fig. 2). However, in subjects who reported motion perception just in or out of the bore, the DD score increase was smaller than anticipated by caloric stimulation [12].
Table 2
Mean and standard error of the mean of the depersonalization/derealization total score and the State-Trait Anxiety Inventory score of 48 young adults before and after exposure to the magnetic field of the 3T MRI, during the first and second exposures, according to motion perception
| Exposure 1 Mean (Standard error) | Exposure 2 Mean (Standard error) | |||||
| Motion perception | No motion | In or out | In &out | No motion | In or out | In &out |
| (n = 27) | (n = 17) | (n = 4) | (n = 27) | (n = 17) | (n = 4) | |
| Depersonalization/derealization | ||||||
| Before | 2.2 (0.3) | 2.3 (0.6) | 3.0 (2.1) | 1.3 (0.3) | 1.4 (0.6) | 3.5 (3.1) |
| After | 1.6 (0.3) | 4.8 (1.5) | 8.2 (4.4) | 1.2 (0.3) | 2.5 (0.8) | 3.7 (2.5) |
| State-Trait Anxiety Inventory | ||||||
| Before | 1.0 (0.2) | 1.4 (0.3) | 0.7 (0.4) | 0.6 (0.1) | 1.1 (0.3) | 3.2 (1.4) |
| After | 0.9 (0.2) | 2.0 (0.4) | 1 (1) | 0.6 (0.2) | 2.0 (0.4) | 1.0 (1.0) |
Fig. 2
Mean and standard error of the mean of the depersonalization/derealization (DD) score and the state anxiety score of 48 young healthy volunteers, before and after static magnetic field exposure, according to the report of motion-perception in/out the bore of the 3T MRI scanner, computed at mean sleep hours per week and scores on the Hospital Anxiety and Depression Scale, the Burnout Measure, the Dissociative Experiences Scale and the Claustrophobia Questionnaire.
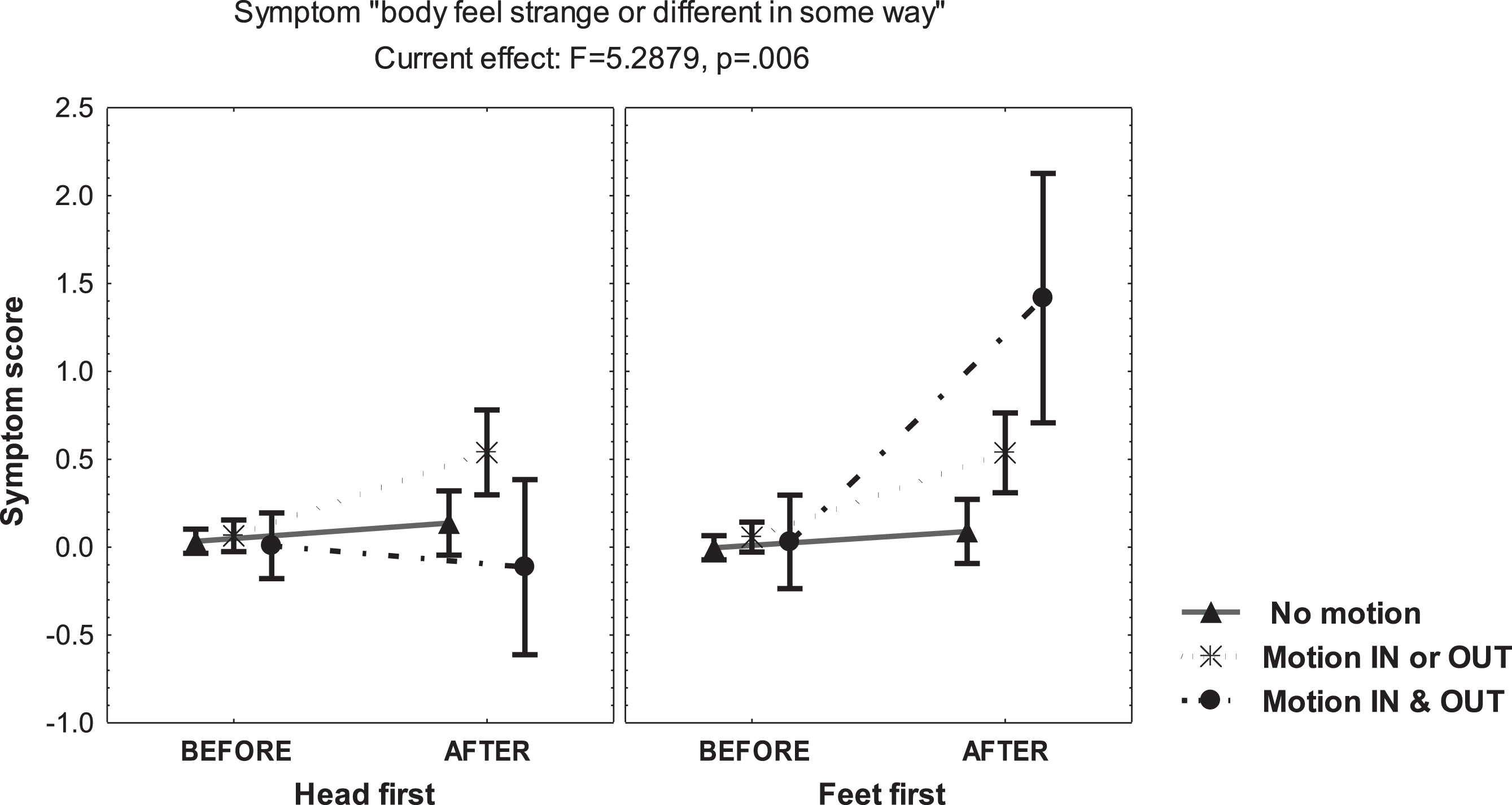
The main increase of DD symptoms was reported just by the subgroup of subjects reporting motion-perception in and out of the bore, mainly when entering the scanner “feet first” (MANCOVA, F =7.79, p = 0.0007) (Fig. 3). Interestingly, during this condition, the subgroup perceiving self-motion scored increase of the questionnaire item “body feels strange or different in some way” (MANCOVA, F = 5.28, p = 0.006) (Fig. 4). However, during the second exposure, the DD score displayed no change in both, those reporting motion-perception in/out of the bore and those denying motion-perception, which supports that a factor contributing to derealization was the novelty of the sensory conflict in susceptible individuals.
Fig. 3
Mean and standard error of the mean score of the depersonalization/derealization (DD) score of 48 young healthy volunteers, before and after static magnetic field exposure, either “head/feet first”, according to the report of motion-perception in/out the bore of the 3T MRI scanner, computed at mean sleep hours per week.
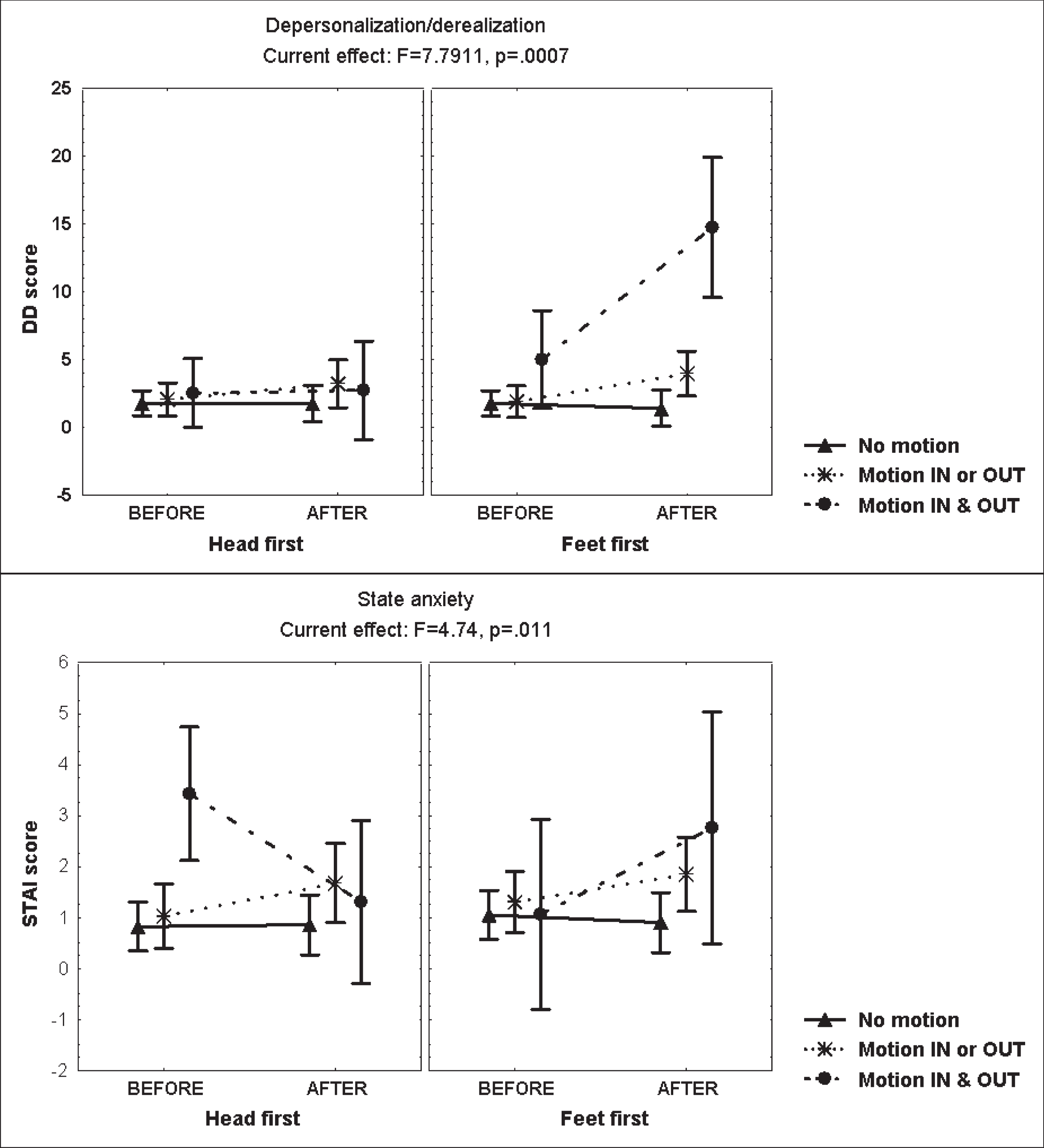
Fig. 4
Mean and standard error of the mean of the score on the symptom “body feels strange or different in some way” of 48 young healthy volunteers, before and after static magnetic field exposure, according to the report of motion-perception in/out the bore of the 3T MRI scanner, computed at mean sleep hours per week.
3.4State anxiety
There was no clear effect of scanner exposure on state anxiety (Table 2), but a decrease during the second exposure was observed in the subgroup reporting motion-perception in and out of the bore. In turn, this was related to an increase of the anxiety score before entering the bore (Fig. 2); particularly in the subject with nausea requiring medication during the first exposure, who returned anxious for the second exposure (STAI score of 7) but reported no anxiety afterwards (STAI score of 0).
3.5Delayed dizziness and disorientation after scanner exposure
After the first exposure, 25% (n = 12) of the sub-jects reported dizziness during the first day (Table 3A). Although at day 7, the frequency of dizziness decreased to 8% (n = 4), it was persistent in one subject, who complained of difficulty reading (dizziness when focusing), and headaches for three days post-exposure, interfering with daily activities. The frequency of dizziness during day 1 after the second exposure was 14% (n = 7), decreasing to 2% (n = 1) at day 7; the same subject who experienced difficulty reading after the first exposure also reported interference with his activities due to headache after re-exposure. Two other subjects reported difficulty reading during the following week post-exposure, but they were able to perform their daily activities as usual.
Table 3
Percentage (number) of subjects reporting usual performance of their daily-life activities, good orientation and dizziness during the first week after exposure to the magnetic field. A. data from 48 subjects after the 1st and 2nd exposures, and B. data from 11 subjects, after the 3rd and 4th exposures (replication)
| A | Day 1 | Day 2 | Day 3 | Day 4 | Day 5 | Day 6 | Day 7 |
| Exposure 1 | |||||||
| Usual performance | 97% (47) | 97% (47) | 97% (47) | 97% (47) | 97% (47) | 97% (47) | 97% (47) |
| Usual orientation | 97% (47) | 97% (47) | 97% (47) | 97% (47) | 97% (47) | 95% (46) | 97% (47) |
| Dizziness | 25% (12) | 8% (4) | 14% (7) | 12% (6) | 14% (7) | 8% (4) | 8% (4) |
| Exposure 2 | |||||||
| Usual performance | 100% (48) | 97% (47) | 100% (48) | 100% (48) | 100% (48) | 100% (48) | 100% (48) |
| Usual orientation | 100% (48) | 100% (48) | 100% (48) | 100% (48) | 100% (48) | 100% (48) | 100% (48) |
| Dizziness | 14% (7) | 8% (4) | 6% (3) | 2% (1) | 2% (1) | 4% (2) | 2% (1) |
| B | |||||||
| Exposure 1 | |||||||
| Usual performance | 90% (10) | 90% (10) | 90% (10) | 90% (10) | 90% (10) | 90% (10) | 90% (10) |
| Usual orientation | 90% (10) | 90% (10) | 90% (10) | 90% (10) | 90% (10) | 90% (10) | 90% (10) |
| Dizziness | 45% (5) | 9% (1) | 18% (2) | 18% (2) | 27% (3) | 9% (1) | 9% (1) |
| Exposure 2 | |||||||
| Usual performance | 81% (9) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) |
| Usual orientation | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) |
| Dizziness | 27% (3) | 0% (0) | 0% (0) | 0% (0) | 0% (0) | 0% (0) | 0% (0) |
| Exposure 3 | |||||||
| Usual performance | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) |
| Usual orientation | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) |
| Dizziness | 27% (3) | 9% (1) | 0% (0) | 0% (0) | 0% (0) | 0% (0) | 0% (0) |
| Exposure 4 | |||||||
| Usual performance | 100% (11) | 100% (11) | 90% (10) | 100% (11) | 100% (11) | 100% (11) | 100% (11) |
| Usual orientation | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) | 100% (11) |
| Dizziness | 27% (3) | 0% (0) | 9% (1) | 0% (0) | 0% (0) | 0% (0) | 0% (0) |
3.6Interaction among the study variables
A simple correlation between DD an STAI scores was observed only during the first exposure, when the DD score pre-exposure was related to the STAI score pre-exposure (r = 0.29, p = 0.049) and the DD score post-exposure was related to the STAI score post-exposure (r = 0.39, p = 0.006). No correlation was present during the second exposure.
A repeated measures multivariate analysis showed that DD scores after any exposure were influenced by entering the bore “feet first” (beta =0 .50, 95% C.I. 0.22–0.76), motion-perception (beta =0 .45, 95% C.I. 0.25–0.64), and the mean sleep hours/week (beta =0 .25, 95% C.I. 0.07–0.43) with a whole model multiple R of 0.58 (MANCOVA, F = 7.66, p = 0.00001). There was no influence from the scale scores of burnout, dissociative experiences, anxiety and depression and motion sickness, or from the report of alcohol consumption (mild in all cases), or tobacco consumption (infrequent).
A similar repeated measures multivariate analysis on the STAI score after any exposure showed influence from the anxiety and depression scale (beta = 0.33, 95% C.I. 0.09–0.57) and absence of motion-perception (beta = 0.26, 95% C.I. 0.03–0.48), with no influence from other variables nor full significance of the whole model multiple R (MANCOVA, p = 0.056).
3.7Replication of the study protocol
The subjects who performed the study protocol twice reported the perception of motion in/out of the bore with a similar frequency than the entire group: 45% (n = 5) for the first exposure and 54% (n = 6) for the second exposure; while during replication it was 63% (n = 7) for the third exposure and 45% (n = 5) for the fourth exposure. However, in this subgroup, only one subject reported motion-perception both in and out of the bore during the four exposures. Then, analysis was performed considering just two groups: no motion-perception and motion-perception,
After one month, baseline DD and STAI scores were similar to those observed before the first exposure to the magnetic field. After the increase of the DD and STAI scores observed during the first exposure in those who experienced motion-perception, there was no change during the following three exposures. During the follow-up the majority of subjects performed their daily life activities as usual (Table 3B).
4Discussion
The summary of findings in normal subjects exposed to a static magnetic field of a 3T MRI scanner is:
- DD symptoms were associated with scanner-induced motion-perception and mean sleep hours/week, mainly during the first exposure and when entering the scanner “feet first”.
- State anxiety was related to trait anxiety during the first scanner exposure, when a weak correlation was observed between DD scores and state anxiety scores.
- DD symptoms, state anxiety and motion-percep-tion were independent from general motion sickness susceptibility.
- There was poor reproducibility of the DD symptoms during repeated exposures.
- Motion-perception was reported by circa half of the subjects and dizziness by less than 15%.
- Due to considerable individual susceptibility, a few subjects reported severe dizziness, capable of interfering with daily life during a week, and of influencing state anxiety during a subsequent scanner exposure.
The association of the perception of motion with increase in DD symptoms suggests that derealization symptoms were related to MVS. This is consistent with previous reports of DD symptoms emerging in healthy subjects during caloric vestibular stimulation [12], centrifugation [27], and in vestibular disease [12, 28, 29]. However, the results show that perceptual reactions evoked by 3T MRI exposure may be variant, and they are smaller than those observed during caloric stimulation [12].
Our findings indicate that the development of scanner related derealization is a multifactorial phenomenon. These include, the sensory conflict induced by MVS, the number of sleep hours/week, the “first exposure” effect and, for some hitherto unexplained reason, the “feet first” condition. Facing a novel experience and the sensory conflict induced by MVS (both somatosensory-vestibular and intra-vestibular conflicts) [25] are easy to understand as contributory to the emergence of DD features in the scanner. Unexpectedly, however, the first exposure of the body itself to a strong magnetic field, during “feet first” entrance (i.e. prior to the head entering the bore) also seemed to have played a part, which requires further study.
In the study design, we considered that the selec-tion of participants among healthcare students/pro-fessionals acquainted with hospital environment and the use of MRI scanning, could introduce effects from burnout and sleeping hours/week. However, the multivariate analysis showed no influence from burnout, since all subjects had low burnout scores, while there was a positive relationship between sleep hours and the DD score. This finding is consistent with the evidence that sleeping habits are part of the psychological make up of young adults [30] and, further, that self-reported sleep duration is related to morbidity/mortality [31] and cognitive function [32]. However, our study design does not allow us to determine whether self-reported sleep hours describes sleep duration accurately or time in bed or sleep disturbances.
In this study, state anxiety was related to trait anxiety, denoting that the novelty effect may have been more stressful for some of the participants. The finding of the contribution of trait anxiety to state anxiety is consistent with the multidimensional concept of anxiety, since the level of state anxiety may be dependent upon both the person trait anxiety and the stressful situation [33]. Reciprocally, the low reproducibility of the DD symptoms during subsequent magnetic exposures likely reflects the lack of novelty of the procedure, considering that the history of experienced stimuli during a sequential sensory estimation task can bias perception [34, 35].
The short duration of the motion-perception related to the exposure, when the oculomotor (nystagmic) response is known to continue as long as the stimulus [10, 25], is an example of the dissociation between perceptual and reflex vestibular responses. To start with, vestibulo-perceptual thresholds are higher than vestibulo-ocular reflex thresholds but, more importantly, vestibulo-perceptual adaption can be quicker and more profound than vestibulo-ocular reflex adaptation in healthy subjects [36], in trained subjects such as dancers [37], and in vestibular patients [38]. The finding that subjects who reported motion perception in and out of the bore showed the lowest vestibular gain to rotation implies that central vestibular processing and adaptation modulated subjects’ individual susceptibility for motion perception and derealization symptoms.
As expected, motion-perception in a 3T MRI scanner was less frequent than that reported in 7T MRI scanners [10, 25], likely related to the strength of the MVS [7]. However, our study shows that even in a conventional 3T MRI scanner, without any additional recording devices and excluding claustrophobic subjects, a brief exposure in a standard head position can provoke negative experiences in susceptible individuals. A main contribution of our study is to have established that such negative experiences, well known to radiologists and clinicians in general, are due not only to psychological factors. One can only presume that additional worries in patients, whose scans results might lead to life changing scenarios, may make the situation even worse but further work is needed to clarify this point.
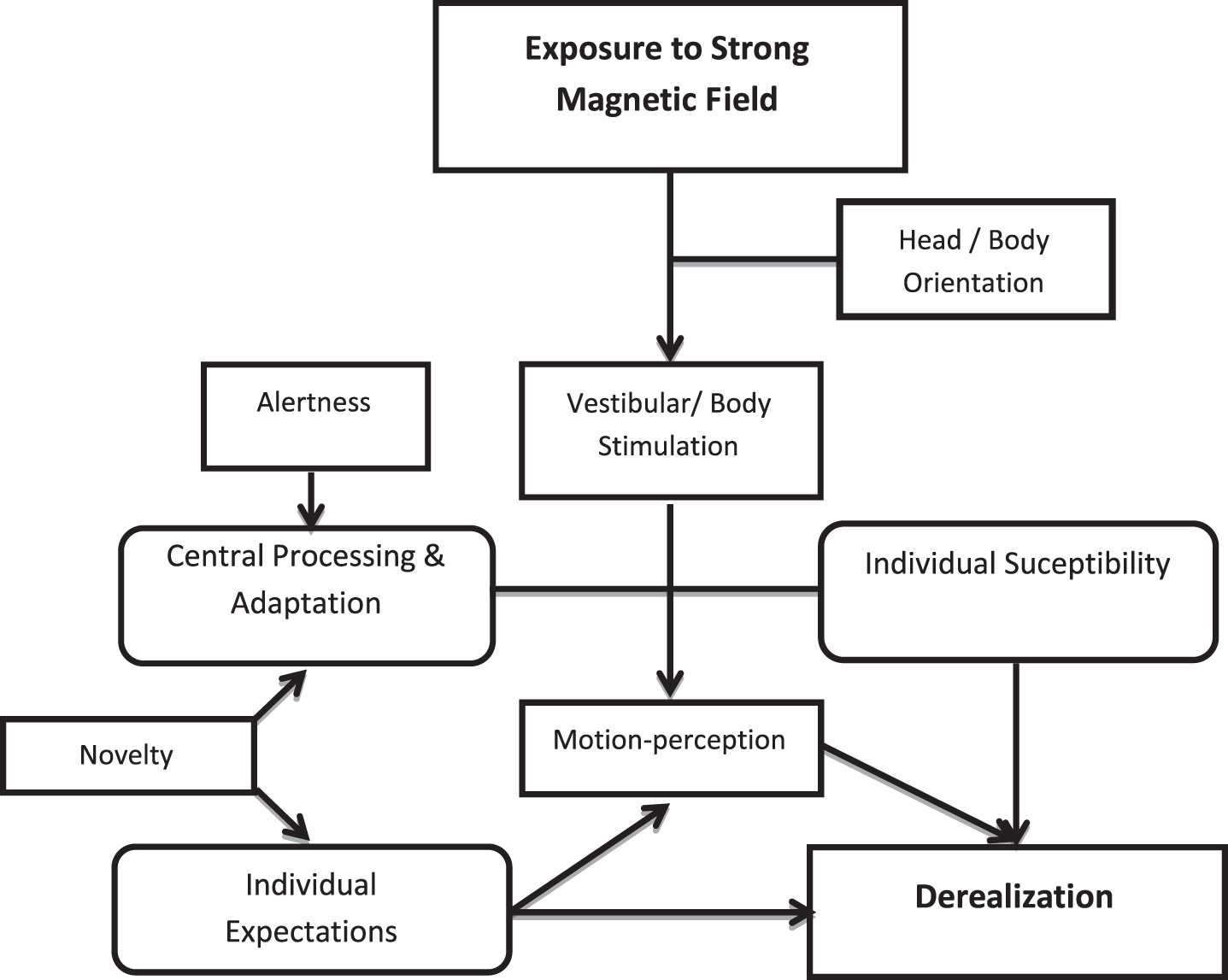
Our knowledge of how vestibular adaptation and compensation play a critical part in clinical recovery after a vestibular insult has mostly focused on reflex function, while perceptual processes are less understood [37]. This study supports that the appearance of derealization features in the MRI scanner depends on a combination of MVS, novelty and individual factors. A schematic diagram of their interaction is represented in Fig. 5. The results suggests that MVS could be used as a model to assess responses to sensory conflict, including the report of chronic/persistent dizziness, like those encountered in the functional syndrome of Persistent Postural-Perceptual Dizziness [14]. As much as such patients develop chronic dizziness as a result of a combination of acute vestibular disease, psychological features and idiosyncratic perceptual styles [40].
Fig. 5
Theoretical model of the interaction between individual factors and strong magnetic stimulation to provoke derealization.
One limitation of the study is the lack of recording of the vestibulo-ocular responses in the scanner. However, in order to establish the natural occurrence of vestibular and DD feelings in our subjects, it was essential that no added apparatus was introduced. Another limitation is that the cognitive expectation of no experiencing movement may have influenced the report of motion-perception [41]. However, despite the strict selection criteria, not including subjects with migraine, claustrophobia or other disorders that could influence the results, some participants exhibited dizziness and some reported after-effects.
In conclusion, derealization symptoms experienced by young healthy adults during the first exposure to the static magnetic field of a 3T MRI scanner are influenced by the induced motion-perception, the novelty of the experience, the mean sleep hours/week and orientation of the head and body on entering the bore. However, individual susceptibility may occur, and this can influence anxiety levels before a subsequent scan. Forewarning subjects of these predisposing factors may increase tolerance to MRI scanning.
Conflict of interest
None declared.
Appendix 1
A
Immediately after coming out from the bore, to assess motion-perception the following questions (check list) were asked by the examiner:
1- At any moment within the bore, did you feel dizzy?
If you did: For how long did you feel dizzy?
2- At any moment within the bore, did you have a rotational sensation?
If you did: In which direction was the rotation? How long did it last?
3- Just after coming-out from the bore, did you have any rotational sensation?
If you did: In which direction was the rotation? How long did it last?
4- If you experienced a rotational sensation within the bore as well as on coming out from the bore: Was it similar the two times? or Was there any difference between them? If there was a difference, describe the difference.
B
To assess feelings of dizziness/disorientation after the MRI scanner exposure, every day for 7 days, subjects replied to the following Yes/No questions with an option for comment (structured diary):
1. Today, did you perform your regular activities as usual?
2. Today, when going to customary places, did you feel disoriented?
3. Today, at any moment, did you feel dizzy or disoriented?
Please write any comment.
Acknowledgments
We thank Ricardo Soriano Rodríguez, Carlos Gar-cía Ramírez and Lourdes Rodríguez López for their technical assistance at the MRI Department, and Silvia Hidalgo Tobón for her advice on MRI safety issues.
References
[1] | Meléndez J.C. and McCrank E. , Anxiety-Related Reactions Associated With Magnetic Resonance Imaging Examinations, JAMA 270: ((1993) ), 745–747. |
[2] | Napp A.E. , Enders J. , Roehle R. , Diederichs G. , Rief M. , Zimmermann E. , Martus P. and Dewey M. , Analysis and Prediction of Claustrophobia during MR Imaging with the Claustrophobia Questionnaire: An Observational Prospective 18-month Single-Center Study of 6500 Patients, Radiology 283: ((2017) ), 148–157. |
[3] | Katz R.C. , Wilson L. and Frazer N. , Anxiety and its determinants in patients undergoing magnetic resonance imaging, J Behav Ther Exp Psychiatry 25: ((1994) ), 131–134. |
[4] | Spielberger C.D. , (Ed.), Anxiety and behaviour, Academic Press, New York ((1966) ), pp. 361–398 |
[5] | Bloxham J. and Gubbins D. , The secular variation of Earth’s magnetic field, Nature 317: ((1985) ), 777–781. |
[6] | de Vocht F. , Batistatou E. , Mölter A. , Kromhout H. , Schaap K. , van Tongeren M. , Crozier S. , Gowl P. and Keevil S. , Transient health symptoms of MRI staff working with 1.5 and 3.0 Tesla scanners in the UK, Eur Radiol 25: ((2015) ), 2718–2726. |
[7] | Schaap K. , de Vries Y. C. , Mason C.K. , de Vocht F. , Portengen L. and Kromhout H. , Occupational exposure of healthcare and research staff to static magnetic stray fields from 1.5–7 Tesla MRI scanners is associated with reporting of transient symptoms, Occup Environ Med 71: ((2014) ), 423–429. |
[8] | Roberts D.C. , Marcelli V. , Gillen J.S. , Carey J.P. , Della Santina C.C. and Zee D.S. , MRI magnetic field stimulates rotational sensors of the brain, Curr Biol 21: ((2011) ), 1635–1640. |
[9] | Theysohn J.M. , Kraff O. , Eilers K. , Andrade D. , Gerwig M. , Timmann D. , Schmitt F. , Ladd M.E. , Ladd S.C. and Bitz A.K. , Vestibular effects of a 7 Tesla MRI examination compared to 1.5 T and 0 T in healthy volunteers, PLoS One 9: ((2014) ), e92104. |
[10] | Ward B.K. , Otero-Millan J. , Jareonsettasin P. , Schubert M.C. , Roberts D.C. and Zee D.S. , Magnetic Vestibular Stimulation (MVS) As a Technique for Understanding the Normal and Diseased Labyrinth, Front Neurol 8: ((2017) ), 122. |
[11] | American Psychiatric Association, Diagnostic and Statistical Manual of Mental Disorders. Fifth Edition, American Psychiatric Association, Washington D.C. 2013. |
[12] | Sang Y.P.F. , Jáuregui-Renaud K. , Green D.A. , Bronstein A.M. and Gresty M. , Depersonalization/ derealization symptoms in vestibular disease, J Neurol Neurosurg Psychiatry 77: ((2006) ), 760–766. |
[13] | Cappon D. and Banks R. , Orientational perception. A review and preliminary study of distortion in orientational perception, Arch Gen Psychiat 5: ((1961) ), 380–392. |
[14] | Staab J.P. , Eckhardt-Henn A. , Horii A. , Jacob R. , Strupp M. , Brandt T. and Bronstein A. , Diagnostic criteria for persistent postural-perceptual dizziness (PPPD): Consensus document of the committee for the Classification of Vestibular Disorders of the Bárány Society,, J Vestib Res 27: ((2017) ), 191–208. |
[15] | Kanal E. , Barkovich A.J. and Bell C. , for the Expert Panel on MR Safety. ACR Guidance Document on MR Safe Practices:, J Magn Resonance Imag 37: ((2013) ), 501–530. |
[16] | Radomsky A.S. , Rachman S. , Thordarson D.S. , McIsaac H.K. and Teachman B.A. , The Claustrophobia Questionnaire, J Anxiety Disord 15: ((2001) ), 287–297. |
[17] | Sheehan D.V. , Lecrubier Y. , Sheehan K.H. , Amorim P. , Janavs J. , Weiller E. , Hergueta T. , Baker R. and Dunbar G.C. , The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10, J Clin Psychiatry 59: (Suppl 20) ((1998) ), 22–33. |
[18] | Malach-Pines A. , The Burnout Measure, Short Version, Int J Stress Manag 12: ((2005) ), 78–88. |
[19] | Bernstein E.M. and Putman F.W. , Development, reliability, and validity of a dissociation scale, J Ner Ment Dis 174: ((1986) ), 727–735. |
[20] | Zigmond A.S. and Snaith R.P. , The Hospital Anxiety And Depression Scale, Acta Psychiatr Scand 67: ((1983) ), 361–370. |
[21] | Golding J. , Predicting individual differences in motion sickness susceptibility by questionnaire, Pers Individ Dif 41: ((2006) ), 237–248. |
[22] | Cox B.J. and Swinson R.P. , Instrument to assess deperson-alization-derealization in panic disorder, Depress Anxiety 15: ((2002) ), 172–175. |
[23] | Marteau T.M. and Bekker H. , The development of a six-item short-form of the state scale of the Spielberger State-Trait Anxiety Inventory (STAI), Br J Clin Psycol 31: (Pt 3) ((1992) ), 301–306. |
[24] | Mian O.S. , Li Y. , Antunes A. , Glover P.M. and Day B.L. , On the Vertigo Due to Static Magnetic Fields, PLoS One 8: ((2013) ), e78748. |
[25] | Mian O.S. , Li Y. , Antunes A. , Glover P.M. and Day B.L. , Effect of head pitch and roll orientations on magnetically induced vertigo, J Physiol 594: ((2016) ), 1051–1067. |
[26] | Siemens AG , Healthcare Sector. System Owner's Manual: Magnetom Skyra. Siemens AG 2010-2012; Muenche. 2012. |
[27] | Aranda Moreno C. and Jáuregui-Renaud K. , Derealization during utricular stimulation,, J Vestib Res 26: ((2016) ), 425–431. |
[28] | Gómez-Alvarez F.B. and Jáuregui-Renaud K. , Psychological Symptoms and Spatial Orientation During the First 3 Months After Acute Unilateral Vestibular Lesion, Arch Med Res 42: ((2011) ), 97–103. |
[29] | Jáuregui-Renaud K. , Ramos-Toledo V. , Aguilar-Bolaños M. , Montaño-Velázquez B. and Pliego-Maldonado A. , Symptoms of detachment from the self or from the environment in patients with an acquired deficiency of the special senses, J Vestib Res 18: ((2008) ), 129–137. |
[30] | Johns M.W. , Bruce D.W. and Masterton B.T. , Psychological correlates of sleep habits reported by healthy young adults, Br J Med Psychol 47: ((1974) ), 181–187. |
[31] | Grandnera M.A. and Drummond S. , Who are the long sleepers? Towards an understanding of the mortality relationship, Sleep Med Rev 11: ((2007) ), 341–360. |
[32] | Kronholm E. , Sallinen M. , Suutama T. , Sulkava R. , Era P. and Partonen T. , Self-reported sleep duration and cognitive functioning in the general population, J Sleep Res 18: ((2009) ), 436–446. |
[33] | Endler N.S. and Kocovski N.L. , State and trait anxiety revisited, Anxiety Disord 15: ((2001) ), 231–245. |
[34] | Rader A.A. , Oman C.M. and Merfeld D.M. , Motion perception during variable-radius swing motion in darkness, J Neurophysiol 102: ((2009) ), 2232–2244. |
[35] | Prsa M. , Jimenez-Rezende D. and Blanke O. , Inference of perceptual priors from path dynamics of passive self-motion, J Neurophysiol 113: ((2015) ), 1400–1413. |
[36] | Pettorossi V.E. , Panichi R. , Botti F.M. , Kyriakareli A. , Ferraresi A. , Faralli M. , Schieppati M. and Bronstein A.M. , Prolonged asymmetric vestibular stimulation induces opposite, long-term effects on self-motion perception and ocular responses, J Physiol 591: (7) ((2013) ), 1907–1920. |
[37] | Nigmatullina Y. , Hellyer P.J. , Nachev P. , Sharp D.J. and Seemungal B.M. , The neuroanatomical correlates of training-related perceptuo-reflex uncoupling in dancers, Cereb Cortex 25: ((2015) ), 554–562. |
[38] | Panichi R. , Faralli M. , Bruni R. , Kiriakarely A. , Occhigrossi C. , Ferraresi A. and Bronstein A.M. , Asymmetric vestibular stimulation reveals persistent disruption of motion perception in unilateral vestibular lesions, J Neurophysiol 118: ((2017) ), 2819–2832. |
[39] | Bronstein A.M. and Dieterich M. , Long-term clinical outcome in vestibular neuritis, Curr Opin Neurol 32: ((2019) ), 174–180. |
[40] | Cousins S. , Kaski D. , Cutfield N. , Arshad Q. , Ahmad H. , Gresty M.A. , Seemungal B.M. , Golding J. and Bronstein A.M. , Predictors of clinical recovery from vestibular neuritis: a prospective study, Ann Clin Transl Neurol 224: ((2017) ), 340–346. |
[41] | Wertheim A.H. , Mesland B.S. and Bles W. , Cognitive suppression of tilt sensations during linear horizontal self-motion in the dark, Perception 30: ((2001) ), 733–741. |




