Effectiveness of a multidisciplinary rehabilitation program in real-world patients with chronic back pain: A pilot cohort data analysis
Abstract
BACKGROUND
: Randomized clinical trials (RCT) suggest a multidisciplinary approach to pain rehabilitation is superior to other active treatments in improving pain intensity, function, disability, and pain interference for patients with chronic pain, with small effect size (ds
OBJECTIVE:
The current study examined the effectiveness of a multidisciplinary program to a cognitive and behavioral therapy (pain-CBT) in real-world patients with chronic back pain.
METHODS:
Twenty-eight patients (
RESULTS:
We found significant improvement in mobility and pain behavior only after a multidisciplinary program (
CONCLUSIONS:
The effect of a multidisciplinary rehabilitation program observed in RCT would be generalizable to real-world practice.
1.Introduction
Chronic back pain is the leading cause of disability worldwide [1] and the one-year prevalence of activity-limiting back pain is about 38% [2]. Studies have shown that chronic back pain impacts physical [3, 4] and psychosocial function [5, 6]. This multi-factorial nature of chronic back pain makes it imperative that treatment approaches should be multimodal; and typically include medical, physical, and psychological intervention approaches. Cumulative evidence suggests a multidisciplinary approach to pain rehabilitation is superior to other active treatments in improving pain intensity, function, disability, and pain interference for patients with chronic pain, with small effect size (ds
The most popular psychological interventions for chronic pain are Cognitive Behavioral Therapy (CBT) [12, 13], Acceptance and Commitment Therapy (ACT) [14], and Mindfulness-based program [15]. CBT aims to reduce disability through learning adaptive cognitive and behavioral strategies, and it shows small to medium effects on pain outcomes [13]. ACT focuses on patient engagement in valued life activities while accepting unwanted pain, thoughts, and feelings, with the overall goal of enhancing ‘psychological flexibility’ [16], and ACT includes mindfulness strategy [17]. ACT is similarly efficacious in improving physical function [17, 18]. Our new multidisciplinary rehabilitative program, labeled Backs in Action (BIA), has been offered to patients as an adjunctive treatment to their usual medical care. The BIA is comprised of an individualized physical therapy (PT) and psychological programs (CBT and ACT).
Using the CHOIR-CAT survey, the current study aimed to compare the effectiveness of a newly developed multidisciplinary rehabilitation program to the pain-CBT in a pilot cohort of real-world patients with chronic back pain. We hypothesized that treatment outcomes would be comparable between pain-CBT and BIA programs because we matched patients with their need for CBT only or intensive rehabilitation program.
Table 1
Comparison of the content of the two programs
| Pain-CBT | BIA | |
|---|---|---|
| Pain psychology | ||
| Psychoeducation about pain | X | X |
| Relaxation techniques | X | X |
| SMART goal settings | X | |
| Cognitive restructuring | X | X |
| Activity pacing | X | X |
| Sleep hygiene | X | X |
| Scheduled pleasant activity | X | |
| Planning for discharge and pain flare-up | X | X |
| Values | X | |
| Mindfulness | X | |
| Committed action | X | |
| Defusion | X | |
| Self as context | X | |
| PT program | ||
| Mindful Movement – Tai Chi for Rehabilitation Sequence* | X | |
| Mindful Movement-Gentle Yoga | X | |
| Gym Exercise Program – Graded Aerobic Exercise | X | |
| Gym Exercise Program-Individual Exercises | X | |
| Pain Neuroscience Education | X |
*:Full video can be found at https://www.youtube.com/watch?v=0DiwrHRWS4A.
2.Method
2.1Enrollment process
This was a retrospective cohort study of patients who attend the BIA or pain-CBT at a tertiary pain clinic between January 2017 and November 2019. Inclusion criteria for both programs were patients with any chronic back pain (i.e., neck, upper, and lower back pain). The BIA was designed for those who significantly reduced daily function and needed an intensive outpatient program whereas the pain-CBT was for patients who had relatively better daily function and needed to learn self-management skills. All patients in both groups were referred by pain physicians, but the enrollment process was different between the two programs. All patients in the BIA were evaluated by pain psychologists to determine their appropriateness for the group-based pain psychology program and by physical therapists to determine whether the patient could safely participate in the exercise-based interventions. Tests and measures during the physical therapy evaluation included lumbar ROM, core testing, balance testing, a six-minute walk test (6MWT) with BP/HR measurements pre-and post 6MWT [19]. Then, patients who had an insurance approval attended the BIA program. In contrast, not all patients in the pain-CBT underwent psychology evaluation because pain physicians could make a referral to the pain-CBT. The pain-CBT was periodically offered free of charge and insurance approval was not needed.
2.2Intervention content
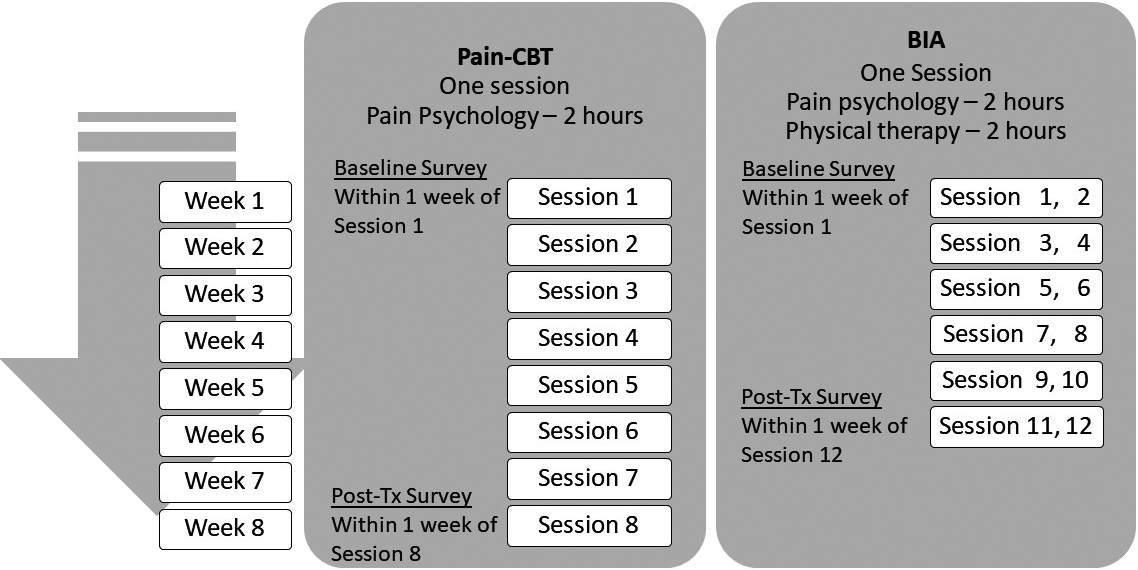
Our programs covered the published CBT [20] and ACT contents [21]. Specifically, the pain-CBT included pain psychoeducation and pain self-management techniques, listed in Table 1. The pain-CBT was run by a pain psychologist or 4 supervised postdoctoral psychology fellows. The 2-hour CBT was offered over 8 weeks (total 16 hours). In addition to the pain-CBT, the BIA covered the ACT content: values, mindfulness, committed action, defusion, and self as context [21]. The BIA was run by a pain psychologist and a physical therapist. The BIA consisted of 12 sessions over a 6-week period (total 24 hours of pain psychology and 24 hours of PT sessions). In each BIA session were 2-hour pain psychology and 2-hour PT (Fig. 1).
Figure 1.
Comparison of survey administration and the treatment course for Pain-CBT and BIA programs. Post-Tx: post-treatment.
Our PT program included the evidenced-based educational contents and exercise interventions: pain neuroscience education as well as graded aerobic exercise and supervised/individualized resistance exercise [22, 23](Table 1). Tai Chi and Yoga were also included as they had shown to reduce pain and improve disability for patients with low back pain [24, 25]. The Tai Chi program was led by a Physical Therapist that was certified in Tai Chi for Rehabilitation Sequence [24]. The Tai Chi was taught in seated and standing positions. The yoga was based off the published protocol [25], including supine poses, seated poses and standing poses for 20 minutes each. Patients had a graded aerobic exercise program that increased in duration by 10–20% each week throughout the 6 weeks. The individual exercise program, which was created by the physical therapist at the initial evaluation, focused each individual’s impairments and self-selected goals. The individual program was also upgraded each week throughout the program.
2.3Measurements
Retrospective chart reviews were conducted to assess medical diagnoses. The current study extracted data of the following measures completed within one week of the first and last sessions.
Our primary outcome was the Patient-Reported Outcomes Measurement Information System (PROMIS)
Secondary outcomes were the PROMIS Pain Intensity measure assessing the intensity of pain at worst and average for the past 7 days on a 0–10 scale [28] and the Pain Catastrophizing Scale (PCS) assessing levels of catastrophic thinking about pain [29]. The total PCS scores range from 0 to 52, with higher scores indicating greater pain catastrophizing. Finally, the body map was used to assess the number of painful sites, ranging from 0 to 45 [30].
2.4Statistical analysis
Appropriate (
Table 2
Comparison of the baseline characteristics of the patients in the pain-CBT and BIA programs
| Pain-CBT ( | BIA ( | |||||
|
| (%) |
| (%) |
|
| |
| Sex (Female) | 14 | (77.8) | 23 | (82.1) | 0.13 | 0.716 |
| Race (White/Caucasian) | 12 | (66.7) | 15 | (53.6) | 0.78 | 0.379 |
| Marital status (Married) | 9 | (50.0) | 17 | (60.7) | 0.51 | 0.474 |
| Education (Bachelor’s or higher)* | 12 | (66.7) | 20 | (74.1) | 0.29 | 0.591 |
| Currently not working | 11 | (61.1) | 19 | (67.9) | 0.22 | 0.639 |
| Currently being on disability | 5 | (17.9) | 5 | (27.8) | 0.63 | 0.426 |
| Pain caused by an injury/accident | 7 | (38.9) | 8 | (28.6) | 0.53 | 0.466 |
| Legal claim for pain* | 4 | (23.5) | 1 | (3.6) | – | 0.060 |
| Median | (IQR) | Median | (IQR) |
|
| |
| Number of painful sites | 10.5 | 6.0–15.3 | 11.5 | 7.5–24.5 | 205.50 | 0.295 |
| Pain duration (years) | 4.6 | 2.9–10.3 | 9.8 | 4.3–17.3 | 171.00 | 0.068 |
| Comorbid Conditions^ | 0.5 | 0.0–4.0 | 2.0 | 1.0–5.0 | 179.50 | 0.095 |
|
| SD |
| SD |
|
| |
| Age | 58.9 | 14.3 | 57.6 | 14.8 | 0.773 | |
| PCS | 17.7 | 7.8 | 16.8 | 10.0 | 0.747 | |
| Worst pain | 7.4 | 1.6 | 6.7 | 2.1 | 0.245 | |
| Average pain | 5.0 | 1.7 | 4.6 | 2.1 | 0.510 | |
| PROMIS T scores | ||||||
| Mobility | 58.2 | 7.4 | 61.0 | 5.1 | 1.49 | 0.143 |
| Pain Interference | 64.2 | 5.3 | 64.6 | 5.4 | 0.27 | 0.788 |
| Pain Behaviors | 58.8 | 2.7 | 59.5 | 3.2 | 0.72 | 0.474 |
| Fatigue | 61.2 | 10.1 | 60.6 | 9.3 | 0.843 | |
| Sleep Impairment | 56.8 | 11.2 | 59.1 | 8.4 | 0.79 | 0.432 |
| Depression | 54.7 | 9.3 | 57.1 | 6.9 | 1.04 | 0.307 |
| Anxiety | 58.1 | 10.1 | 57.5 | 7.7 | 0.823 | |
| Emotional Support | 50.9 | 8.2 | 52.0 | 9.0 | 0.44 | 0.664 |
| Social Isolation | 51.7 | 9.9 | 52.8 | 6.5 | 0.47 | 0.644 |
| Social Role Satisfaction | 59.6 | 6.8 | 59.9 | 5.6 | 0.18 | 0.856 |
*: missing
Table 3
The mean differences of T scores between the pre- and post-treatments
| Total (Pain-CBT and BIA) ( | Pain-CBT ( | BIA ( | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Mean difference | SD |
| Mean difference | SD |
| Mean difference | SD |
| |
| PCS* |
| 8.73 | 0.57 | 7.48 | 9.59 | ||||
| PROMIS T scores | |||||||||
| [Physical Health] | |||||||||
| Mobility | 3.66 | 0.31 | 0.67 | 3.41 | 0.20 |
| 3.38 |
| |
| Pain Interference |
| 4.54 | 0.73 | 4.20 | 4.71 | ||||
| Pain Behaviors | 3.82 | 0.34 | 0.22 | 2.92 | 0.08 |
| 4.06 |
| |
| Fatigue |
| 8.31 | 0.47 | 7.49 | 8.93 | ||||
| Sleep Impairment | 6.89 | 0.20 | 6.34 | 7.28 | |||||
| [Psychosocial Health] | |||||||||
| Depression |
| 7.19 | 0.44 | 5.31 | 7.96 | ||||
| Anxiety |
| 8.63 | 0.29 | 7.86 | 9.04 | ||||
| Emotional Support | 5.43 | 0.26 | 0.00 | 5.08 | 5.55 | ||||
| Social Isolation | 6.13 | 0.21 | 5.37 | 6.41 | |||||
| Social Role Satisfaction |
| 5.79 | 0.54 | 4.30 | 6.56 | ||||
Note: *Changes in PCS total scores. The others are changes in T scores. Bolded values in the total (Pain-CBT and BIA group) column indicate a significant time effect in a post-hoc test and bolded values in the Pain-CBT and BIA column indicate a significant time by group interaction effect (
We collected data until our sample size was a total of 40 patients for the current pilot study and intended to have 20 for each group. However, our final sample included 18 patients for pain-CBT and 28 patients for BIA and the current study computed the effect sizes, which can be used for a future study’s power analysis. No significant violation of multicollinearity and homogeneity were observed as all correlation coefficients were
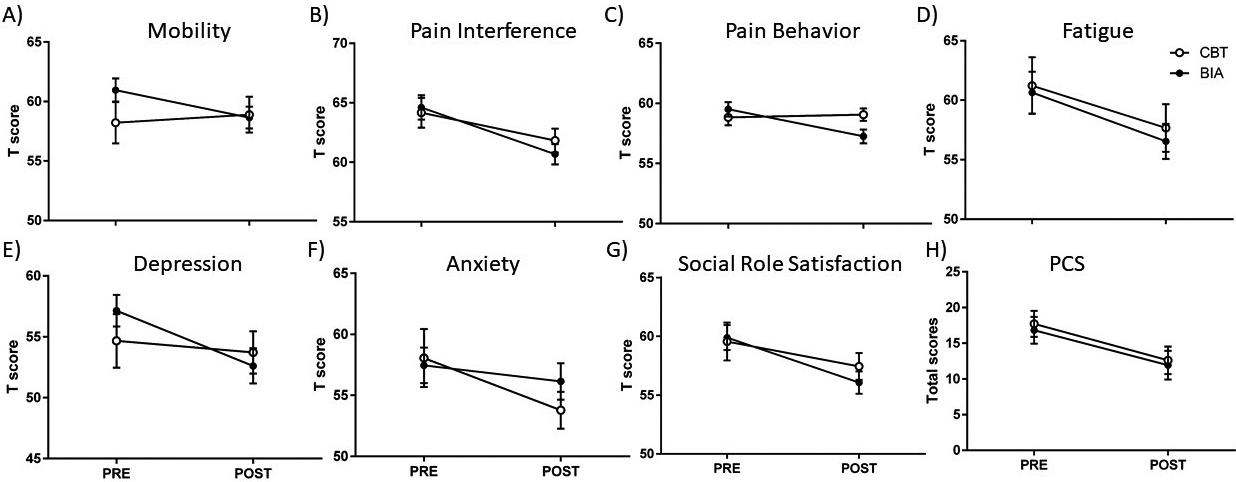
Figure 2.
Comparisons of pre- and post-treatment outcomes. A)–G) are PROMIS T scores of Mobility, Pain Interference, Pain Behavior, Fatigue, Depression, Anxiety, and Social Role Satisfaction, respectively, with higher T scores indicating worse health status in each domain. H) is PCS total score. Errors
3.Results
3.1Baseline patient characteristics
Sociodemographic states were not significantly different between the two groups (
The two groups were not significantly different in the number of painful sites (
3.2Health outcomes of the pain-CBT and BIA programs
A 2(time)
We conducted sensitivity analysis for the significant interaction effect. When entering legal claims as a covariate, the previously significant interaction effect in mobility,
Another 2(time)
Finally, a 2(time)
4.Discussion
The current study compared the effectiveness of a multidisciplinary rehabilitation program to pain-CBT in real-world patients with chronic back pain. As hypothesized, we found that the BIA and pain-CBT groups showed statistically significant improvement in pain catastrophizing, pain interference, fatigue, depression, anxiety, and social role satisfaction. These treatment effects were small to medium effect sizes. Different from our hypothesis, only the BIA group showed a significant improvement in mobility and pain behaviors.
A meta-analysis reveals that multidisciplinary programs with functional restoration approach produce greater improvement in function for patients with disabling chronic pain than usual care [8]. Based on our results, pain interference improved in both groups, yet physical function/mobility improved only after the BIA, highlighting the additional benefit of improving physical function by the PT inclusion program. Our results are consistent with a meta-analysis showing that multidisciplinary programs with functional restoration approach produce greater improvement in function for patients with disabling chronic pain than usual care [8, 10], but inconsistent with a RCT showing that both CBT alone and CBT and PT programs produce greater improvement in physical function at 3, 6, and 12-month follow-ups, compared to exercise only or usual care [32]. Potentially, the current study may be underpower to detect the effect of CBT on physical function or need to follow-up a longer period as there may be a delayed effect, referred to as “sleeper effect” [33]. ACT components in the BIA may also contribute to the improvement of physical function. The CBT is based on control strategies and it focuses on learning effective pain management skills to reduce pain and the impact of pain on life. In contrast, the ACT teaches that controlling or avoiding pain is ineffective and it focuses on reducing “avoidance” of unwanted thoughts, feeling, and sensations, allowing patients to engage in values driven behaviors that increase life satisfaction and meaning. In supporting this view, an experiment has demonstrated that physical impairment is decreased by acceptance strategies but increased by control strategies [34].
We also found that pain behaviors were reduced only after the BIA. Pain behaviors are verbal and non-verbal expressions of pain experience and suffering [35]. Greater pain behaviors are associated with worse pain and general health status [35]. Studies have noted that ACT [36, 37] and PT [38] reduce pain behaviors such as less sick leave and medical visits whereas CBT prevents from taking more sick leave and medical visits in the future [39]. Notably, levels of acceptance and value-driven action do not predict medical visits after ACT [37]. We speculate that reduction of pain behaviors may be related to a) learning the relationships between behaviors moving away from their valued-life and the associated internal struggles, and b) engagement in strategies to reduce the internal struggles, by incorporating defusion and mindfulness skills.
Our small to moderate effects of pain-CBT or BIA on pain interference, fatigue, depression, anxiety, social role satisfaction, and pain catastrophizing were consistent with RCTs. More importantly, despite heterogeneity of our sample with multiple comorbidities and no randomization, our findings are similar to RCT of CBT [13], ACT [17], and multidisciplinary programs [7, 8, 9, 10], suggesting RCT findings are generalizable to typical patients attending a rehabilitation program at an outpatient setting. Furthermore, different from patients in RCT, which usually offers compensation for participants’ class attendance and time for surveys, our patients attended the programs as recommended by their treating clinicians and voluntarily completed the survey for no compensation. Therefore, our patients had less external motivation.
The current study has several limitations. Additional study with a large cohort of patients is warranted to determine the effectiveness of a multidisciplinary rehabilitation program in real-world practice. The current study assessed the outcomes of patients who attended the first and last session, and voluntarily completed the CHOIR survey before and after the program. Future study should conduct intent-to-treat analysis and examine the sleeper effect. Finally, our treatment effects related to PT or ACT components should be examined in a RCT. Despite these limitations, to the best of our knowledge, this was the first study to show that RCT results of multidisciplinary rehabilitation program for chronic back pain would be generalizable to real-world practice.
The current study showed that pain catastrophizing, pain interference, fatigue, depression, anxiety, and social role satisfaction were improved after pain-CBT and a multidisciplinary rehabilitation program. Yet, improvement of physical function and pain behaviors were observed only after the multidisciplinary rehabilitation program, which included additional ACT and PT components.
Conflict of interest
None to report.
Funding
Dr. Dokyoung You (K23DA048972), Dr. Maisa Ziadni (K23DA047473) and Dr. Sean Mackey (K24DA0 29262) received funding from the NIH National Institute on Drug Abuse. Dr. Sean Mackey also received support from Redlich Pain Research Endowment.
References
[1] | Vos T, Allen C, Arora M, Barber RM, Bhutta ZA, Brown A, Carter A, Casey DC, Charlson FJ, Chen AZ. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet. (2016) ; 388: (10053): 1545-1602. |
[2] | Hoy D, Bain C, Williams G, March L, Brooks P, Blyth F, Woolf A, Vos T, Buchbinder R. A systematic review of the global prevalence of low back pain. Arthritis Rheum. (2012) ; 64: (6): 2028-2037. |
[3] | Hammill RR, Beazell JR, Hart JM. Neuromuscular consequences of low back pain and core dysfunction. Clin Sports Med. (2008) ; 27: (3): 449-462. |
[4] | Picavet H, Schouten J. Musculoskeletal pain in the Netherlands: prevalences, consequences and risk groups, the DMC3-study. Pain. (2003) ; 102: (1-2): 167-178. |
[5] | Meyer T, Cooper J, Raspe H. Disabling low back pain and depressive symptoms in the community-dwelling elderly: a prospective study. Spine. (2007) ; 32: (21): 2380-2386. |
[6] | Strunin L, Boden LI. Family consequences of chronic back pain. Soc Sci Med. (2004) ; 58: (7): 1385-1393. |
[7] | Scascighini L, Toma V, Dober-Spielmann S, Sprott H. Multidisciplinary treatment for chronic pain: a systematic review of interventions and outcomes. Rheumatology. (2008) ; 47: (5): 670-678. |
[8] | Guzmán J, Esmail R, Karjalainen K, Malmivaara A, Irvin E, Bombardier C. Multidisciplinary rehabilitation for chronic low back pain: systematic review. BMJ. (2001) ; 322: (7301): 1511-1516. |
[9] | Hoffman BM, Papas RK, Chatkoff DK, Kerns RD. Meta-analysis of psychological interventions for chronic low back pain. Health Psychol. (2007) ; 26: (1): 1-9. |
[10] | Kamper SJ, Apeldoorn A, Chiarotto A, Smeets R, Ostelo R, Guzman J, Van Tulder M. Multidisciplinary biopsychosocial rehabilitation for chronic low back pain: Cochrane systematic review and meta-analysis. BMJ. (2015) ; 350. doi: 10.1136/bmj.h444. |
[11] | Chambers DA, Feero WG, Khoury MJ. Convergence of implementation science, precision medicine, and the learning health care system: a new model for biomedical research. JAMA. (2016) ; 315: (18): 1941-1942. |
[12] | de Williams AC, Eccleston C, Morley S. Psychological therapies for the management of chronic pain (excluding headache) in adults. Cochrane database of systematic reviews (2012) (11). doi: 10.1002/14651858.CD007407.pub3. |
[13] | Ehde DM, Dillworth TM, Turner JA. Cognitive-behavioral therapy for individuals with chronic pain: efficacy, innovations, and directions for research. Am Psychol. (2014) ; 69: (2): 153-166. |
[14] | McCracken LM, Vowles KE. Acceptance and commitment therapy and mindfulness for chronic pain: Model, process, and progress. Am Psychol. (2014) ; 69: (2): 178-187. |
[15] | Castelnuovo G, Giusti EM, Manzoni GM, Saviola D, Gatti A, Gabrielli S, Lacerenza M, Pietrabissa G, Cattivelli R, Spatola CA. Psychological treatments and psychotherapies in the neurorehabilitation of pain: evidences and recommendations from the Italian Consensus Conference on Pain in Neurorehabilitation. Front Psychol. (2016) ; 7: : 115. doi: 10.3389/fpsyg.2016.00115. |
[16] | Hayes SC, Villatte M, Levin M, Hildebrandt M. Open, aware, and active: Contextual approaches as an emerging trend in the behavioral and cognitive therapies. Annu Rev Clin Psychol. (2011) ; 7: : 141-168. |
[17] | Gaudiano BA. A review of acceptance and commitment therapy (ACT) and recommendations for continued scientific advancement. SRMHP. (2011) ; 8: (2): 5-22. |
[18] | Hann KE, McCracken LM. A systematic review of randomized controlled trials of Acceptance and Commitment Therapy for adults with chronic pain: Outcome domains, design quality, and efficacy. J Contextual Behav Sci. (2014) ; 3: (4): 217-227. |
[19] | Gibbons WJ, Fruchter N, Sloan S, Levy RD. Reference values for a multiple repetition 6-minute walk test in healthy adults older than 20 years. J Cardiopulm Rehabil Prev. (2001) ; 21: (2): 87-93. |
[20] | Darnall BD, Mackey SC, Lorig K, Kao M-C, Mardian A, Stieg R, Porter J, DeBruyne K, Murphy J, Perez L. Comparative Effectiveness of Cognitive Behavioral Therapy for Chronic Pain and Chronic Pain Self-Management within the Context of Voluntary Patient-Centered Prescription Opioid Tapering: The EMPOWER Study Protocol. Pain Med. (2019) ; 21: (8): 1523-1531. doi: 10.1093/pm/pnz285. |
[21] | Wetherell JL, Afari N, Rutledge T, Sorrell JT, Stoddard JA, Petkus AJ, Solomon BC, Lehman DH, Liu L, Lang AJ. A randomized, controlled trial of acceptance and commitment therapy and cognitive-behavioral therapy for chronic pain. Pain. (2011) ; 152: (9): 2098-2107. |
[22] | Booth J, Moseley GL, Schiltenwolf M, Cashin A, Davies M, Hübscher M. Exercise for chronic musculoskeletal pain: a biopsychosocial approach. Musculoskeletal Care. (2017) ; 15: (4): 413-421. |
[23] | Meeus M, Nijs J, Van Wilgen P, Noten S, Goubert D, Huijnen I. Moving on to movement in patients with chronic joint pain. Pain. (2016) ; 1: (10): 23-35. |
[24] | Hall AM, Maher CG, Lam P, Ferreira M, Latimer J. Tai chi exercise for treatment of pain and disability in people with persistent low back pain: a randomized controlled trial. Arthritis Care Res. (2011) ; 63: (11): 1576-1583. |
[25] | Saper RB, Lemaster C, Delitto A, Sherman KJ, Herman PM, Sadikova E, Stevans J, Keosaian JE, Cerrada CJ, Femia AL. Yoga, physical therapy, or education for chronic low back pain: a randomized noninferiority trial. Ann Intern Med. (2017) ; 167: (2): 85-94. |
[26] | Cella D, Riley W, Stone A, Rothrock N, Reeve B, Yount S, Amtmann D, Bode R, Buysse D, Choi S. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005–2008. J Clin Epidemiol. (2010) ; 63: (11): 1179-1194. |
[27] | Cella D, Gershon R, Lai J-S, Choi S. The future of outcomes measurement: item banking, tailored short-forms, and computerized adaptive assessment. Qual Life Res. (2007) ; 16: (1): 133-141. |
[28] | Cook KF, Dunn W, Griffith JW, Morrison MT, Tanquary J, Sabata D, Victorson D, Carey LM, MacDermid JC, Dudgeon BJ. Pain assessment using the NIH Toolbox. Neurology. (2013) ; 80: (11 Supplement 3): S49-S53. |
[29] | Sullivan MJ, Bishop SR, Pivik J. The pain catastrophizing scale: development and validation. Psychol Assess. (1995) ; 7: (4): 524-532. |
[30] | Kutch JJ, Ichesco E, Hampson JP, Labus JS, Farmer MA, Martucci KT, Ness TJ, Deutsch G, Apkarian AV, Mackey SC. Brain signature and functional impact of centralized pain: a multidisciplinary approach to the study of chronic pelvic pain (MAPP) network study. Pain. (2017) ; 158: (10): 1979-1991. |
[31] | Cohen J. Statistical power for the social sciences. Hillsdale, NJ: Laurence Erlbaum and Associates, (1988) ; 98-101. |
[32] | Donta ST, Clauw DJ, Engel CC, Jr., Guarino P, Peduzzi P, Williams DA, Skinner JS, Barkhuizen A, Taylor T, Kazis LE. Cognitive behavioral therapy and aerobic exercise for Gulf War veterans’ illnesses: a randomized controlled trial. JAMA. (2003) ; 289: (11): 1396-1404. |
[33] | Rawson RA, McCann MJ, Flammino F, Shoptaw S, Miotto K, Reiber C, Ling W. A comparison of contingency management and cognitive-behavioral approaches for stimulant-dependent individuals. Addiction. (2006) ; 101: (2): 267-274. |
[34] | Vowles KE, McCracken LM, Eccleston C. Processes of change in treatment for chronic pain: the contributions of pain, acceptance, and catastrophizing. Eur J Pain. (2007) ; 11: (7): 779-787. |
[35] | Revicki DA, Chen W-H, Harnam N, Cook KF, Amtmann D, Callahan LF, Jensen MP, Keefe FJ. Development and psychometric analysis of the PROMIS pain behavior item bank. Pain. (2009) ; 146: (1-2): 158-169. |
[36] | Dahl J, Wilson KG, Nilsson A. Acceptance and commitment therapy and the treatment of persons at risk for long-term disability resulting from stress and pain symptoms: A preliminary randomized trial. Behav Ther. (2004) ; 35: (4): 785-801. |
[37] | McCracken LM, Gutiérrez-Martínez O. Processes of change in psychological flexibility in an interdisciplinary group-based treatment for chronic pain based on Acceptance and Commitment Therapy. Behav Res Ther. (2011) ; 49: (4): 267-274. |
[38] | Ritz JM, Childs JD, Wainner RS, Flynn TW. Primary care referral of patients with low back pain to physical therapy: impact on future health care utilization and costs. Spine. (2012) ; 37: (25): 2114-2121. |
[39] | Linton SJ, Boersma K, Jansson M, Svärd L, Botvalde M. The effects of cognitive-behavioral and physical therapy preventive interventions on pain-related sick leave: a randomized controlled trial. Clin J Pain. (2005) ; 21: (2): 109-119. |




