Multi-slice spiral computed tomography diagnosis of juxta-papillary duodenal diverticulum and its relationship with biliopancreatic diseases
Abstract
BACKGROUND:
Juxta-papillary duodenal diverticula (JPDD) are common but are usually asymptomatic, and they are often diagnosed by coincidence.
OBJECTIVE:
To analyse the anatomy and classification of JPDD and its relationship with biliary and pancreatic disorders, and to explore the diagnostic value of multi-slice spiral computed tomography (MSCT) in patients with JPDD.
METHODS:
The imaging data of patients with JPDD, which was obtained via abdominal computed tomography examination and confirmed via gastroscopy and/or upper gastrointestinal barium enema, in our hospital from 1 January 2019 to 31 December 2020 were retrospectively analysed. All patients were scanned using MSCT, and the imaging findings, classification and grading were analysed.
RESULTS:
A total of 119 duodenal diverticula were detected in 96 patients, including 73 single diverticula and 23 multiple diverticula. The imaging findings were mainly cystic lesions of the inner wall of the duodenum protruding to the outside of the cavity. The thin layer showed a narrow neck connected with the duodenal cavity, and the shape and size of the diverticula were different: 67 central-type cases and 29 peripheral-type cases. There were 50 cases of type I, 33 cases of type II, 19 cases of type III and six cases of type IV. Furthermore, there were seven small, 87 medium and 14 large diverticula. The differences in the location and size of the JPDD in MSCT grading were statistically significant (
CONCLUSION:
The MSCT method has an important diagnostic value for the classification of JPDD, and MSCT images are helpful in the clinical evaluation of patients with JPDD and the selection of treatment options.
1.Introduction
A duodenal diverticulum is a cystic hernia that protrudes through the weak mucosa of the duodenal wall and is mostly asymptomatic [1]. A juxta-papillary duodenal diverticulum (JPDD) refers to a diverticulum that occurs 2–3 cm away from the duodenal papilla [2]. Due to its close relationship with the anatomy of the duodenal papilla, it is often combined with various biliary and pancreatic diseases in clinical practice, which is termed duodenal papillary diverticulum syndrome or Lemmel’s syndrome [3]. The patients are mainly middle-aged and elderly people who experience recurrent upper abdominal pain [4]. Computed tomography (CT) is a common examination method, but it is easy to ignore the simultaneous existence of JPDD in CT diagnosis [5].
At present, 64-slice and above multi-slice spiral CT (MSCT) can perform sub-millimetre thin-layer scanning and multi-directional observation, which improves the imaging level of the duodenum and hepatobiliary regions [6]. The MSCT method can identify four different types of JPDD, and the use of this classification can help describe the disease more accurately [7]. To further analyse the imaging findings of JPDD syndrome, this study retrospectively analysed the MSCT examination and imaging data of patients to provide a valuable reference for the clinical diagnosis and treatment of this disease.
2.Materials and methods
2.1Research participants
A total of 96 patients with JPDD who were examined using abdominal CT scanning and gastroscopy and/or upper gastrointestinal barium enema from 1 January 2019 to 31 December 2020 in our hospital in Yantai City were selected as the research participants. The inclusion criteria were as follows: (1) complete medical records; (2) the disease is confirmed by relevant examinations and meets the clinical diagnostic criteria of JPDD in the eighth edition of Surgery [8]; and (3) the participants understand the research content and agree to participate in this study. The exclusion criteria were as follows: (1) incomplete image data; (2) mental illness or cognitive dysfunction; and (3) lack of cooperation with the researchers.
The patients consisted of 52 men and 44 women, and their mean age was 67
2.2Examination methods
The instrument used for the examination was a Somatom Definition AS
2.3Observation indicators
The results of the MSCT examination were analysed, including in terms of imaging findings, classification and grading.
Based on the relationship between nipple and diverticulum, the classification of diverticula is divided into a central type (the nipple opening is located at the edge or inside of the diverticulum opening) and a peripheral type (the nipple opening is within 3 cm of the diverticulum) [9].
Based on the classification method devised by Wiesner et al., the classification of the diverticula was divided into four types: (1) type I: ventral type of ampullary sphincter complex; (2) type II: dorsal type of ampullary sphincter complex; (3) type III: two-leaf type; (4) type IV: small nipple ventral type [10]. These videos were read by two experienced abdominal radiologists to document the location and size of JPDD and the incidence of biliary and pancreatic diseases.
The diverticula were divided into three types according to diameter: small diverticulum (
The MSCT grading of the JPDD was as follows: grade 0
2.4Statistical methods
The date were analysed using SPSS 19.0 statistical software. The measurement data were expressed as (
3.Results
3.1Location and size of the diverticula
A total of 113 JPDD were detected in the 96 patients, including 73 cases of single diverticula and 23 cases of multiple diverticula. The diverticula were located above the nipple in 69 cases, below the nipple in 21 cases and at the edge of the nipple in six cases. The diameter of the diverticula ranged from 0.5 to 4.3 cm, with an average of 2.54
Figure 1.
A–B: male, 49 years old, enhanced scan portal venous phase, type I, medium diverticulum, pancreatic swelling, a small amount of exudative changes around the pancreas, thickening of the left prerenal fascia, considering JPDD with edematous pancreatitis, MSCT image grade 1. C–D: male, 72 years old, abdominal plain scan MPR coronal and axial, type I, large diverticulum, annular high-density lesions in the gallbladder, considering JPDD combined with gallstone MSCT image grade 2; E–F: male, 63 years old, MPR axial and coronal plain abdominal scan, type III, large diverticulum, multiple nodular high-density shadows in the common bile duct, thickening and blurring of the bile duct wall, bile duct dilatation above the obstruction level, considering JPDD with multiple common bile duct stones and cholangitis, MSCT imaging grade 2.
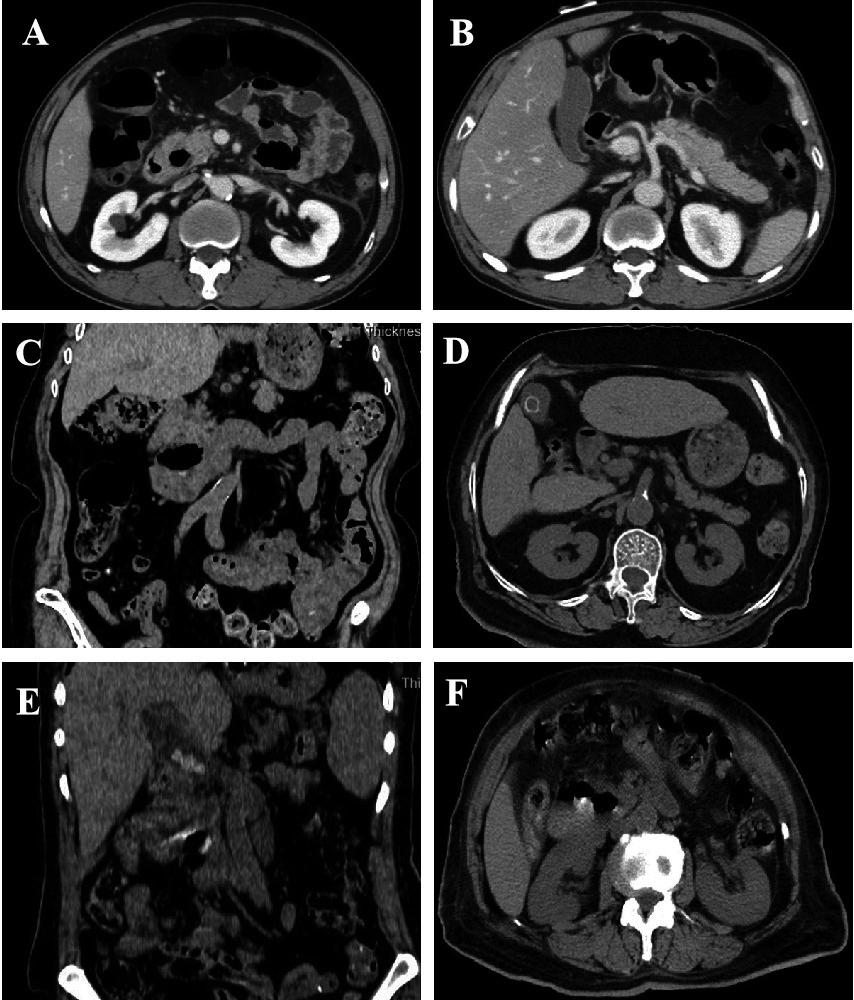
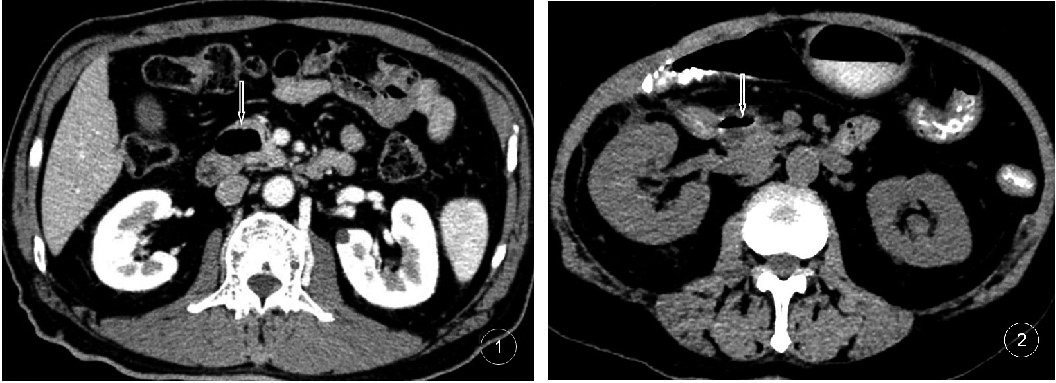
Figure 2.
1 Male, 68 years old, JPDD containing gas; 2 Female, 71 years old, JPDD containing a gas-liquid level and a small amount of high-density contrast medium, being compressed adjacent to the lower end of the common bile duct.
3.2Imaging findings of the diverticula
On the MSCT images, the JPDD mainly manifested as a pocket-like lesion protruding from the inner wall of the duodenum to the outside of the cavity.The thin layer showed the neck of the lesion and the duodenal cavity, and the neck was narrow. Nineteen (19.80%) cysts contained gas (Fig. 2), 18 (18.75%) cysts contained liquid and 31 (32.29%) cysts contained a gas-liquid plane (Fig. 2). Six cases (6.25%) had mixed-density shadows of gas-liquid and food in the cystic cavity. In the enhanced scan, 22 cases (22.92%) of the diverticulum wall presented mild to moderate enhancement consistent with the enhancement of the duodenal wall, and the contents of the diverticula were not enhanced. The shapes and sizes of the diverticula (mostly round or oval, with eight lobulated cases) were different. Fourteen cases were complicated with diverticulitis and mainly manifested as rough and thickened diverticulum walls with significant enhancement. The multiplanar reconstruction showed a clear diverticula narrow neck.
3.3Classification of the diverticula
There were 67 central-type cases and 29 peripheral-type cases, while there were 68 bile duct dilatation cases, 36 bile duct stones cases, 24 cholecystitis cases and 11 gallbladder stones cases (with multiple lesions) (Table 1).
Table 1
Classification of JDD and combined biliary and pancreatic diseases
| Diverticulum classification |
| Bile duct dilatation | Bile duct stones | Cholecystitis | Gallbladder stones |
|---|---|---|---|---|---|
| Central type | 67 | 56 | 22 | 11 | 6 |
| Peripheral | 29 | 12 | 14 | 13 | 5 |
| Total | 96 | 68 | 36 | 24 | 11 |
Table 2
MSCT grading in JDD types
| Classification/ grading | I | II | III | IV | Small diverticulum | Medium diverticulum | Large diverticulum |
|---|---|---|---|---|---|---|---|
| 0 | 14 | 15 | 8 | 5 | 5 | 30 | 7 |
| 1 | 22 | 2 | 8 | 0 | 2 | 36 | 4 |
| 2 | 13 | 6 | 3 | 1 | 0 | 20 | 3 |
| 3 | 1 | 0 | 0 | 0 | 0 | 1 | 0 |
| Total | 50 | 33 | 19 | 6 | 7 | 87 | 14 |
Of the 113 diverticulum types, 50 were type I, 33 were type II, 19 were type III and six were type IV. The incidence of grade 0 was 70.6% (36/51), the incidence of grade 1 was 51.4% (18/35), the incidence of grade 2 was 52.4% (11/21) and the incidence of grade 3 was 16.7% (1/6). The total incidence was 58.4% (66/113). There was a significant difference in the incidence of JPDD (grade 0–3) among different types (
4.Discussion
In recent years, with the continuous development of imaging diagnostic techniques, the early diagnosis rate of JPDD has improved. The MSCT method has been widely used in the clinical diagnosis of this disease and has become the preferred examination method [2], presenting a fast-scanning tool with a wide range. The method can be developed using multi-planar reconstruction, surface covering and other technologies, and it can obtain clearer image maps, which have the advantages of high sensitivity and high specificity [6]. In this study, the MSCT and imaging data of a sample of patients were retrospectively analysed, as well as the imaging manifestations of JPDD, which provided a valuable reference for the clinical diagnosis and treatment of this disease.
A typical JPDD is a capsular bag-like lesion protruding from the inner wall of the duodenum to the outer cavity. The thin layer shows the neck connecting the lesion and the duodenal cavity. The diverticulum cavity often contains a gas-liquid plane. The enhanced scan has mild to moderate enhancement, and the shape and size of the diverticula are different [12, 13]. In terms of clinical diagnosis, the patient’s medical history, symptoms and CT imaging findings generally allow for a clear diagnosis [14]. When the CT imaging reveals a simple liquid cystic shadow, attention should be paid to differential diagnosis with pancreatic cysts and cystic tumours to avoid misdiagnosis [15].
However, in the diagnosis of upper abdominal CT, the duodenal diverticulum is often overlooked and missed. The diagnosis of the 96 cases of JPDD in this group indicated that the previous diagnostic doctors had insufficient understanding and low vigilance. The CT manifestations of the duodenal diverticulum in this group were capsular bag-like protrusions beside the nipple, which were more common in the medial wall of the descending segment. The CT manifestations may vary due to the different contents of the diverticulum. Enhanced scan diverticulum wall visible enhancement, but also because the wall is too thin no obvious enhancement, content no enhancement. Through the observation using thin-layer CT, combined with MPR imaging, the display of the duodenal diverticulum is more reliable, clearly showing the shape, location, size and surrounding tissues and organs of the lesion. Duodenal CPR imaging can straighten the curved duodenal circle and better display the relationship between the duodenum and the diverticulum [16, 17]. It is recommended to find the opening between the diverticulum and the duodenal cavity or the intestinal mucosa extending from the duodenum to distinguish it from other lesions containing gas and liquid around the duodenum, such as gastrointestinal perforation, pancreatic pseudocyst and pancreatic cystadenoma.
In this study, the classification criteria for JPDD were mainly based on the type of biliary and pancreatic diseases and follow-up treatments. Patients with simple JPDD without biliary and pancreatic complications were clinically classified as grade 0 to prevent JPDD syndrome. Grade 1 mainly involved biliary and pancreatic inflammation, which can be alleviated and controlled by active medical treatment. Grade 2 was associated with various types of biliary calculi or obstructive jaundice. At present, endoscopic retrograde cholangiopancreatography or surgical treatment is advocated in clinical practice [18] to control any inflammation and reduce any hepatopancreatic damage. Grade 3 referred to all types of acute and severe diseases caused by JPDD, such as severe pancreatitis or cholestatic cirrhosis. These patients are critical and face complications and often require multidisciplinary comprehensive treatment. Treating doctors should be highly vigilant in clinical practice [19]. Through the analysis of this group, it was found that grades 0 and 1 accounted for most patients with JPDD, followed by grade 2 and grade 3. Most patients with JPDD only need follow-up or conservative medical treatment, while a small number of patients with grades 2 and 3 require surgery. Preoperative MSCT examinations are helpful in evaluating the condition of such patients.
This study involved a number of limitations. First, this was a retrospective study, and there may have been inevitable deviations. Second, this was a single-centre study, and future research needs to include multi-centre studies. In addition, this study did not compare the diagnostic performance of CT, MRI or ERCP, since the number of relevant examinations in our hospital is small. Clinical samples will be increased in future studies and these will include MRI or ERCP result reports for comparison, while the images will be re-evaluated.
5.Conclusion
MSCT not only allows for a classification diagnosis of JPDD but also an accurate image classification, which is helpful for clinical evaluation and the appropriate selection of treatment options.
Ethics statement
This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of 970 Hospital of the PLA JLSF. Written informed consent was obtained from all patients.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Funding
Not applicable.
Author contributions
LW and WD conceived the study, HJ and ZG participated in its design and coordination, and SJ helped draft the manuscript. All authors read and approved the final manuscript.
Conflict of interest
None of the authors have any personal, financial, commercial, or academic conflicts of interest.
References
[1] | Thakran J, Meyer A, Adamek HE. Duodenal diverticulum as the cause of unexplained duodenal stenosis. Dtsch Arztebl Int. (2020) Oct 20; 117: (48): 830. doi: 10.3238/arztebl.2020.0830b. |
[2] | Martín M, Gómez P, Amarelo M, Sáenz M. Lemmel’s syndrome: An uncommon complication of periampullary duodenal diverticulum. Rev Esp Enferm Dig. (2022) Jan; 114: (1): 57-58. doi: 10.17235/reed.2021.8258/2021. |
[3] | Gao AR, Matta A, Seth R, Bande D. Lemmel’s syndrome secondary to common bile duct compression by an inflamed duodenal diverticulum. Cureus. (2021) Aug 6; 13: (8): e16959. doi: 10.7759/cureus.16959. |
[4] | Houssni JE, Cherraqi A, Chehrastane R, Jellal S, Hsain IH, Oukacem S, Adjou N, Saouab R, En Nouali H, Fenni JE. Lemmel syndrome: An unusual cause of biliary obstruction secondary to a duodenal juxta-ampullary diverticulum: A report of two cases. Radiol Case Rep. (2023) Jan 10; 18: (3): 1147-1151. doi: 10.1016/j.radcr.2022.11.013. |
[5] | Rojas RA, Reyes MC, Peñaherrera MV, Gualacata EV, Morillo G, Villacres OP. Lemmel’s syndrome: Presentation of an uncommon cholangitis cause and a risk factor for failed endoscopic retrograde cholangiopancreatography. Case report. Int J Surg Case Rep. (2022) Jan; 90: : 106698. doi: 10.1016/j.ijscr.2021.106698. |
[6] | Tang L, Zhong J, Feng H, Huang Y. The clinical application value of MSCT in the classification and grading of duodenal peripapillary diverticulum. Chinese Journal of Integrated Traditional and Western Medicine Imaging. (2018) ; 16: (06): 598-600+603. |
[7] | Liang P. MSCT findings and diagnostic value of Lemmel’s syndrome. Journal of Medical Imaging. (2020) ; 30: (01): 83-86. |
[8] | Chen X, Wang J. Surgery. 8th edition [M]. Beijing: People’s Health Publishing House (2013) ; 368: : 453-454. |
[9] | Li J, Wu T, Yang K, Yang Y. Surgical diagnosis and treatment of biliary obstruction caused by duodenal papillary diverticulum. Chinese Journal of General Surgery. (2018) ; 27: (02): 231-235. |
[10] | Azzam AZ, Alsinan TA, Alrebeh GA, Alhaider T, Alnaqaeb LJ, Amin TM. Lemmel syndrome as a rare cause of prolonged right hypochondrial pain: A case report. Cureus. (2021) Dec 1; 13: (12): e20093. doi: 10.7759/cureus.20093. |
[11] | Tang L, Zhong J, Feng H, Huang Y. The clinical application value of MSCT in the classification and grading of duodenal peripapillary diverticulum. Chinese Journal of Integrated Traditional and Western Medicine Imaging. (2018) ; 16: (06): 598-600+603. |
[12] | Yang J, Xu H, Zhang M, Zhang J. Discussion on the value of ultrasound diagnosis of Lemmel syndrome. Chinese Journal of Medical Ultrasound (Electronic Edition). (2018) ; 15: (02): 144-147. |
[13] | Jin T, Wang L. One case of Lemmel syndrome with CA19-9 elevation in ultrasound. Zhejiang Medical Journal. (2019) ; 41: (19): 2119-2120. |
[14] | Aourarh B, Tamzaourte M, Benhamdane A, Berrag S, Adioui T, Aourarh A, Belkouchi L, Outznit M, Saouab R. An unusual cause of biliary tract obstruction: Lemmel syndrome. Clin Med Insights Case Rep. (2021) Dec 21; 14: : 11795476211063321. doi: 10.1177/11795476211063321. |
[15] | Zorzetti N, Lauro A, Cervellera M, Panarese A, Khouzam S, Marino IR, Sorrenti S, D’Andrea V, Tonini V. Debris-filled duodenal diverticulum and lemmel’s syndrome. Dig Dis Sci. (2022) Jul; 67: (7): 2805-2808. doi: 10.1007/s10620-022-07571-z. |
[16] | Zhu F, Zhang Q, Zhang J, Chang Y, Zhang J, Lu X, Yang C. Analysis of changes in the diagnosis and treatment of Lemmel syndrome in China in the past 20 years. Journal of Clinical Hepatobiliary Diseases. (2018) ; 34: (03): 583-587. |
[17] | Dávila Arias C, García Pérez PV, Moya Sánchez E. Acute cholangitis in the context of Lemmel syndrome with signs of diverticulitis. Rev Esp Enferm Dig. (2021) Apr; 113: (4): 298-299. doi: 10.17235/reed.2020.7095/2020. |
[18] | Alzerwi NAN. Recurrent ascending cholangitis with acute pancreatitis and pancreatic atrophy caused by a juxtapapillary duodenal diverticulum: A case report and literature review. Medicine (Baltimore). (2020) Jul 2; 99: (27): e21111. doi: 10.1097/MD.0000000000021111. |
[19] | Volpe A, Risi C, Erra M, Cioffi A, Casella V, Fenza G. Lemmel’s syndrome due to giant periampullary diverticulum: Report of a case. Radiol Case Rep. (2021) Oct 4; 16: (12): 3783-3786. doi: 10.1016/j.radcr.2021.08.068. |




