The Effectiveness of Inpatient Rehabilitation in Parkinson’s Disease: A Systematic Review of Recent Studies
Abstract
Background:
Parkinson’s disease (PD) is a progressive disease, which is associated with the loss of activities of daily living independency. Several rehabilitation options have been studied during the last years, to improve mobility and independency.
Objective:
This systematic review will focus on inpatient multidisciplinary rehabilitation (MR) in people with Parkinson’s disease (PwPD), based on recent studies from 2020 onwards.
Methods:
Search strategy in three databases included: multidisciplinary rehabilitation, Parkinson’s Disease, inpatient rehabilitation, motor-, functional- and cognitive performance, cost-effectiveness, Quality of Life, and medication changes/Levodopa equivalent daily doses.
Results:
Twenty-two studies were included, consisting of 13 studies dealing with inpatient MR and 9 studies on inpatient non-MR interventions. Inpatient PD multidisciplinary rehabilitation proved to be effective, as well as non-MR rehabilitation.
Conclusions:
This review confirms the efficacy of inpatient MR and non-MR in PD, but is skeptical about the past and current study designs. New study designs, including new physical training methods, more attention to medication and costs, new biomarkers, artificial intelligence, and the use of wearables, will hopefully change rehabilitation trials in PwPD in the future.
INTRODUCTION
Background
Parkinson’s disease (PD) is a complex neurodegenerative disorder, which leads to many motor- and non-motor problems, including neuropsychiatric-, sleep-, and autonomic signs and symptoms [1–3]. Severe symptoms in one domain, or combinations of symptoms in several domains impair activities of daily living (ADL), as is the case in people with PD (PwPD), showing increasing ADL deficits over time, which results in an increase of the burden on caregivers. Finally, this may result in hospital admission or admission to one of the Parkinson rehabilitation programs, in order to improve ADL independency and extending the period of being able to live at home. This review will focus on the effectiveness of inpatient PD rehabilitation programs and will summarize the set-up and outcomes of recent studies in this field, including long-term- and cost-effectiveness data, if available. It is quite important to get a clear understanding of these issues, because the overall numbers of PwPD are increasing, globally from 6.2 million at this moment, to 12.9 million PwPD in 2040 [4, 5]. As a result, health care costs will rise exponentially. Institutional care is one of the most important drivers of overall costs related to PD, representing 67% of the direct costs [6–9]. A nationwide retrospective cohort study initiated from University of Pennsylvania identified 469,055 PwPD who received Medicare benefits in 2002. Nearly 25% (more than 100,000 in total) resided in a long-term care facility. Women with PD had greater odds of nursing facility residence (adjusted odds ratio (AOR) 1.34, 95% confidence interval (CI) 1.30–1.38) compared to men. A multicenter study in Sydney described that 48% of the PwPD were finally admitted in a nursing home [10–12]. Therefore, efficient PD rehabilitation is a very important instrument to improve ADL, hopefully leading to postponement of definite nursing home admission. Without proper cost-effectiveness evidence, it will be difficult for policymakers, insurance companies, clinicians, and PwPD and their caregivers to identify the value of these rehabilitation interventions [13].
The aim of this review is to provide an overview of all studies on inpatient rehabilitation of PwPD, performed between 2020 and 2023, focusing on outcomes related to motor-, functional-, and cognitive performance, cost-effectiveness, quality of life (QoL), and medication changes. We will grade the available evidence, identifying existing gaps in knowledge and making recommendations for the current inpatient rehabilitation programs of PwPD, finally suggesting which future studies should be performed to guide this field.
Overview of PD inpatient rehabilitation data before 2020
Different rehabilitation program models already exist for PwPD, including inpatient multidisciplinary rehabilitation, integrated interdisciplinary models in an outpatient setting, community-based interventional programs or a combination of these concepts. This review will focus on inpatient rehabilitation programs. Various inpatient multidisciplinary models were developed during the last decade, including the German PD-Multimodel Complex Treatment (MCT) model [14–17], the Italian Multidisciplinary Intensive Rehabilitation Treatment concept [18–22], the Dutch inpatient rehabilitation program at Parkinson Expertise Centers (Point for Parkinson), and the Hamburg Parkinson Day-Clinic, indicated for complex PwPD patients, with in- and outpatient care and the use of sophisticated treatment strategies [23]. Most of the models use serial evaluations, followed by an overall care plan, generated by the expertise team on each necessary discipline. Not all the models pay attention to medication (personalized medicine) but are mainly focusing on physiotherapy and physical exercise for PwPD with mild-to-moderate disability Hoehn&Yahr (H&Y) stage 1-3. Also little attention is paid to people with advanced PD (including cognitive impairment), to overall costs and long-term effects. In addition to these existing models, several physical exercise programs (e.g., dancing, climbing, music therapy), or adjunct tools (e.g., virtual reality and robotics), are offered as add-on treatments [24–32]. A Cochrane report on the effect of physical exercise for PwPD, including 156 randomized controlled trials (RCTs) with a total of 7,939 participants, concluded that most types of physical exercise improved the movement patterns and quality of life, whereas the efficacy of these types of physical exercise did not show significant differences. This large Cochrane meta-analysis also concluded that larger, well-conducted studies are needed to increase confidence in the evidence, also including people with advanced disease and cognitive impairments, to be able to generalize the findings to a broader range of PwPD [33]. In addition, there is also a lack of robust evidence for interventions to reduce hospitalization [34] and nursing home admission [35–38] The same applies to the adjunct tools and techniques with small sample sizes, high risk of bias and no long-term evidence [39–45].
METHODS
Search methods
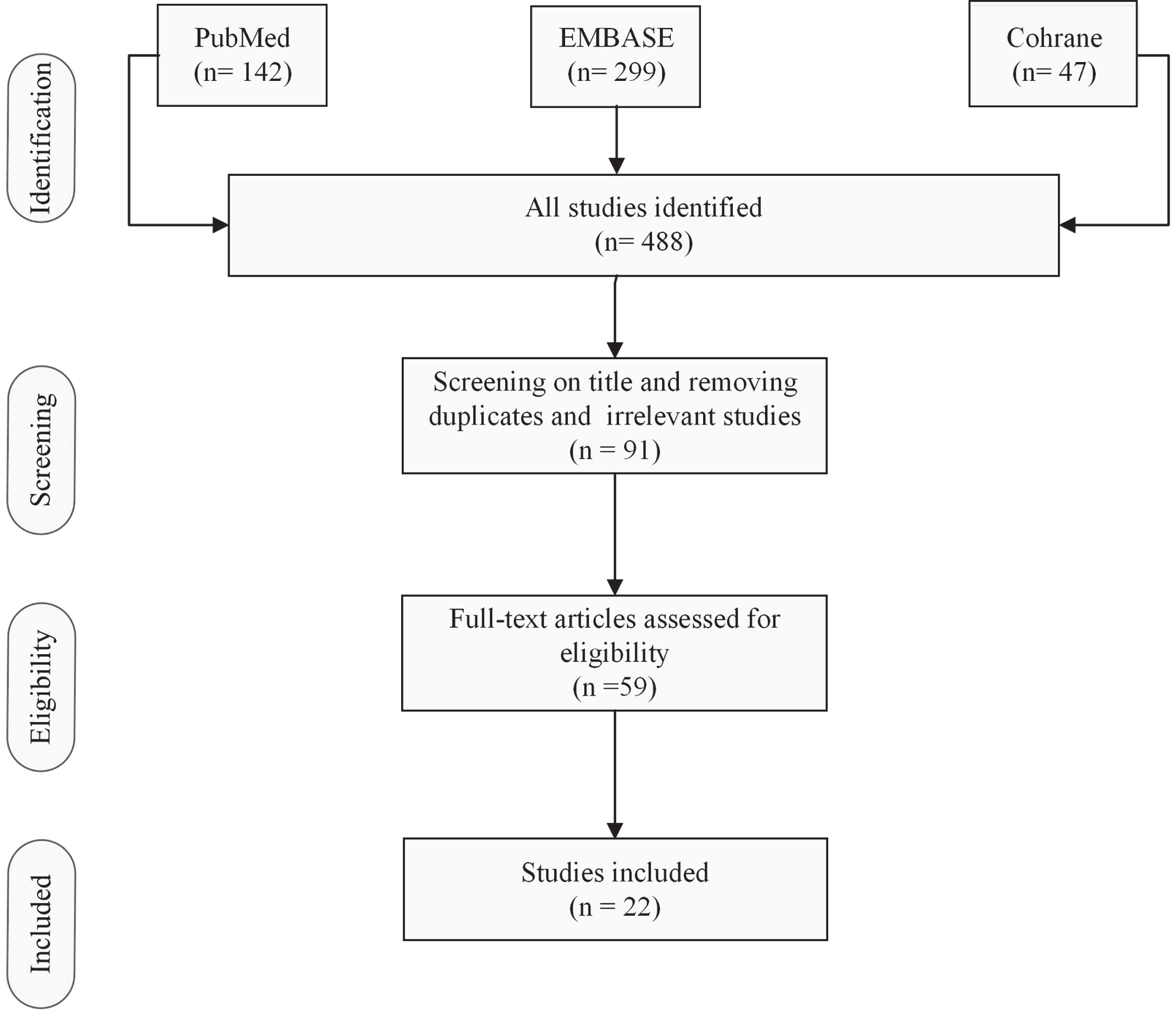
A search was carried out in the following scientific database: PubMed, Embase, Cochrane library (Fig. 1). Review was carried out following the PRISMA guidelines. Search strategy included: multidisciplinary or interdisciplinary rehabilitation, Parkinson’s Disease, inpatient- outpatient: In PubMed, EMBASE and Cochrane library: Search MeSH terms: ‘rehabilitation AND Parkinson’, ‘inpatient AND rehabilitation AND Parkinson’, ‘inpatient AND rehabilitation AND Parkinson’s disease’ and ‘inpatient AND multidisciplinary rehabilitation AND Parkinson’.
Fig. 1
Flowchart of the literature and search strategy (Search MeSH terms: ‘inpatient AND rehabilitation’ ‘inpatient AND rehabilitation AND Parkinson/Parkinson’s disease’).
Selection criteria
Inclusion criteria. The inclusion criteria are summarized in Table 1. Only full text publications in scientific journals (excluding abstracts and posters), written in English were selected for thisreview.
Table 1
Inclusion criteria of selected publications published between Jan 2020-June 2023
| Item | Inclusion of: |
| Design | Randomized controlled trials and non-randomized controlled trials as crossover trials, pre-post test with no control, prospective and retrospective trials |
| Participants | People with Parkinson’s disease n > 10, healthy controls |
| Intervention | Inpatient rehabilitation |
| Comparisons | Usual care, T0 and T1, short-term, long-term |
| Outcome measures | Outcomes related to motor-, functional- and cognitive performance, cost-effectiveness, Quality of Life (QoL) and medication changes |
A classical meta-analysis could not be performed, because of the clinical heterogeneity of the study groups, and the variability in endpoints. All study characteristics are summarized in Table 1. Assessment of each study was done by the first author (in- and exclusion) and was verified by the last author. The quality of evidence of all included studies was rated independently by both authors, and disagreements between the authors were discussed, and solved by consensus, according to the GRADE system [46]. Reasons for downgrading the overall rating were based on the risk of bias, study design, industry sponsorship, inconsistency of data, indirectness, imprecision, substantial loss to follow-up of participants, and unblinded outcome assessment. We were also aware of the fact that studies with statistically non-significant results may not have been reported or submitted for publication (resp. selective non-reporting bias and publication bias). Reasons for upgrading non-randomized studies consisted of a large magnitude of effect or a low influence of confounding factors.
In order to compare the studies quantitatively, we expressed the change vs. baseline of 6 parameters as a percentage, including motor symptoms, ADL, QoL, LEDD change, and cost-effectiveness. The results of these comparisons are summarized in Table 2, which also shows the study design, adding extra information on the strength of the data. Last but not least, the overall value of data is interpreted in the context of decision makers. Will outcomes likely change the landscape of PD rehabilitation?
Table 2
Study characteristics of I. Multidisciplinary rehabilitation (MR) interventions and II. Non-MR interventions
| Author [ref] | Intervention and duration | Objective | Disease severity-H&Y-DD (y) | Subjects (n) | Study design | Duration follow-up (from baseline) | Primary- and Secondary endpoints | Funding | GRADE (Quality of evidence) |
| I. Multidisciplinary rehabilitation (MR) interventions | |||||||||
| Controlled studies (non-randomized) | |||||||||
| Steendam et al. (2023) [47] | 6 weeks MR including optimization of pharmacotherapy+outpatient follow-up | ADL improvement and delay of nursing home admission | H&Y IG mean 4.38 CG mean 4.32 DD (y) IG median 8.0 CG median 9.0 Cognition (scopa-cog score) IG 22.0 CG 15.0 | Total n = 43 IG n = 24 CG n = 19 | Prospective, controlled study | 2 years | Primary outcomes: - ADL (ALDS) - % living independently at home Secondary outcomes - medication (LEDD) - motor performance (SCOPA-SPES) - cognition (SCOPA-COG) - hallucinations (NPI) - depression (BDI). | UMCG innovation fund and prof. Van der Valk Stichting | ⊕⊕ ⊕ ◯ |
| Li et al. (2023) [48] | 2 weeks MR | Change in neurovascular coupling on MRI | H&Y IG mean 2.0 (0.1) CG not mentioned DD (y) IG mean 8.7 (1.1) CG not mentioned Cognition (MMSE) IG 27.1 (0.8) (MoCA 24.5 (1.0) CG not measured | Total n = 61 IG n = 31 CG n = 30 | Prospective controlled study | 2 weeks (discharge) | Outcomes: - motor performance (UPDRS-III) - MRI a. resting-state ASL b. resting-state BOLD scan c. The global and regional CBF–fALFF correlation | National Natural Science Foundation of China Grants, National Key research and Development Program of China and the Science and Technology Development Fund of Beijing Rehabilitation Hospital, Capital Medical University Grants. | ⊕⊕ ⊕ ◯ |
| Wagner et al. (2022) [49] | 1. 3 weeks MR 2. 9 months physiotherapy (App-based) 3 times/ week),+outpatient follow-up | Effectiveness (added value) of tablet-based physiotherapy | H&Y IG mean 2.57 (0.7) CG mean 2.54 (0.7) DD (y) IG mean 7.75 (6.2) CG mean 8.23 (5.1) Cognition? | Total n = 230 IG n = 93 CG n = 137 | Prospective controlled study | 9 months | Primary outcome - QoL (PDQ-8)Secondary outcomes-participation restrictions (IMET) - Fear of falling (FES-I) - Sleep disorder (PDSS) – Anxiety/Depression (PHQ-4) - Comorbidity (SCQ-D) - Pain single item - Performance capability - Physical activity (Federal Health survey) | Innovations Fond Funding Programme of the federal Joint Committee, Project DEAL | ⊕⊕ ◯◯ |
| Prospective open label studies | |||||||||
| Chen et al. (2021) [22] | 2 weeks MR | Effect of PD subtypes on efficacy MIR | H&Y PIGD group 3.0 (1.0) TD group 2.0 (1.0) Indeterminate group 2.5 (1.0) DD (y) PIGD group 6.5 (5.0) TD group 5.0 (4.0) Indeterminate group 4.5 (4.0) Cognition (MMSE) PIGD group 28 (2.0) TD group 27 (6.0) | Total n = 69 PIGD n = 36 TD n = 19 Indeterminate group n = 14 | Prospective open label study | 2 weeks (discharge) | Primary outcome - motor performance (UPDRS-III) Secondary outcomes - Balance (BBS) - mobility (TUG, 10MT, 6MWD, M-PAS) - Strength (5xSTS) | National Key research and Development Program Sub-project and the start-up fund for scientific research Talents of Beijing Rehabilitation Hospital, Capital Medical University of China | ⊕⊕ ◯◯ |
| Lo Buono et al. (2021) [50] | 8 weeks MR | Changes in anxiety and depression and quality of life | H&Y Not mentioned DD Mean 7.57 (±3.46) Cognition (MMSE) 27 (25-28) | n =100 | Prospective open label study | 60 days | Outcome - motor performance (UPDRS-III) - ADL functioning (BI) -neuropsychological function/cognition (ACE-R) - depression (BDI-II) -Anxiety (HAMA-A) -QoL (PDQ-39 - speech language (Clinical Bedside Swallowing examinations, Robertson dysarthria profile) | Italian department of Health | ⊕◯◯◯ |
| Scherbaum et al. (2020) [14] | 2 weeks MR including optimization of pharmacotherapy | Effectiveness of PD-MCT midterm outcome and QoL | H&Y Median 3 (2.5-3) DD (y) Mean 8.5 (5.3) Cognition (MoCA) mean 22.5 (4.6) | n =47 | Prospective open label study | 6 weeks | Primary outcome - QoL (PDQ-39 and EuroQol) Secondary outcomes - Motor function (MDS-UPDRS-III, TUG and PPT) - Depression (BDI-II) - Global change (PGIC) | Deutsche Parkinson Vereinigung Bundesverband | ⊕⊕ ◯◯ |
| Hartelt et al. (2020) [15] | 2 weeks MIRincluding optimization of pharmacotherapy | Effect of PD-MCT on motor symptoms and motor complications | H&Y Median 3 (2.5-3) DD (y) Mean 8.5 (5.3) Cognition (MoCA) mean 22.5 (4.6) | n =47 | Prospective, open label study | 8 weeks | Outcomes - motor assessment (MDS-UPDRS-II, III, IV, TUG, BBS, PPT) - non-motor assessment(MDS-UPDRS-I, BDI-II, AES-D, HAMD-17) | Deutsche Parkinson Vereinigung Bundesverband (Grant no. 33.17-92907). | ⊕⊕ ◯◯ |
| Nielsen et al. (2020) [51] | 2 weeks MIR | Effect on mobility, physical function, and health related quality of life (HRQoL) | H&Y Total 2.1 (SD1.1) Center1/ CST 2.1(SD 0.7) Center 2/VRC 2.2 (SD 0.7) DD Total 7.5 (SD 4.2) CST 7.5 (SD3.5) VCR 7.5 (7.1) Cognition ? | Total n = 214 Two centers involved CST n = 108 VRC n = 106 | Prospective open label study | 4 months,10 months (only PDQ-39) | Primary outcome - QoL (PDQ39)Secondary outcomes - Handgrip strength, - motor (TUG), - Anxiety/ Depression (HADS), - Falls (FES-I) | SANO: Center for Health and Rehabilitation, Danish Association for Rheumatism, Copenhagen Denmark Danish Parkinson’s Association | ⊕⊕ ◯◯ |
| Retrospective studies | |||||||||
| Krause et al. (2022) [52] | 3 weeks MIR Two groups: 1. 3-days/week group (9 treatments) 2. 2-days/ a week (6 treatments) (partly inpatient) | Effect on motor and non-motor symptoms, and QoL | H&Y Total H&Y Mean 2.55 (0.7) DD (y) Mean 10.8 (7.9) Cognition (Mini-mental state-MMST) mean 28.66 (2.11) | Total n = 143 3 G n = 70 2 G n = 73 | Retrospective study | 3 weeks (discharge) | Outcomes - motor performance(UPDRS-III) - psychosocial function (scopa-ps) - Depression (BDI) - QoL (PDQ39, SF36) - Sleep (PDSS, ESS) - Impulsiveness (Quip) - Apathy (SAS) - change in medication - Balance (BBS) - mobility (TUG, MSST)) - Strength (StSt) | Projekt DEAL, German research foundation (DFG) | ⊕⊕ ◯◯ |
| Michels et al. (2022) [53] | 3 weeks MR | Effect of PKB on motor abilities, cognitive profiles and reported depressive symptoms and psychosocial functioning. | H&Y mean 2.78 (0.67) DD (y) mean 108.67 months (74.76) ≈ 9.06 y Cognition MMSE 27.53 (2.35) MoCA 24.0 (4.06) | n =40 | Retrospective study | 3 weeks (discharge) | Outcomes - motor performance (UPDRS-III) - Cognition (MMSE, MoCA) -Attention (TAP, TMT-A) -Memory (WMS-R, VLMT, MCGCF) -visuospatial function (MCGCF, CORSI) - language (CERAD+, Boston naming test) - executive function (WMS-R, RWT, TMT-B - neuropsychological tests (BDI-II, scopa-ps) | Not applicable | ⊕⊕ ◯◯ |
| Ziegler et al. (2022) [54] | 2 weeks MR | Effect of PD-MCT and identify predictors concerning ADL disability. | H&Y Not mentioned (disease severity ICD classification) DD (y) Not mentioned Cognition (MoCA) mean 19.9 (4.7) | n =591 | Retrospective study, | 6 weeks | Primary outcome -ADL (UPDRS-II) Secondary predictors: Basic parameters -age - gender - baseline ADL (UPDRS-II) - baseline motor score (UPDRS-III) -comorbidity | Deutsche Parkinson Vereinigung (DPV) and the Deutsche Stiftung Neurologie (DSN). | ⊕⊕ ◯◯ |
| Heimrich et al. (2021) [17] | 7-21 days MR | PD-MCT to identify predictors of motor improvement and long-term dynamics of health-related quality of life (HR-QoL) | H&Y Median 3.0 (2.5-4.0) DD (y) Mean 9.4 (6.3) Cognition (MoCA) mean 19.9 (4.7) | Total n = 159 PD n = 134 APS n = 25 | Retrospective study | 1 months (Telephone interview) 12 months (only SF-12) | Outcomes - motor performance (UPDRS-III, Tinetti test) - changes medication (LEDD) - HR QoL (SF-12)* Baseline - non-motor symptoms (NMSQ)Depression (HADS-D, BDI-II) - Cognition (MoCA) | Deutsche Forschunsgemeinschaft (DFG, German Research Foundation), Interdisciplinary Center of Clinical Research of the Medical Faculty of Jena and a grant from Bundesministerium fur Bildung und Forschung (BMBF) | ⊕⊕ ◯◯ |
| Meloni et al. (2021) [55] | 3-4 weeks MR | Effect on functional, cognitive, and geriatric domains | H&Y Total mean 3.88 (SD 0.91) Mild-moderate group 3.4 (SD 1.10) Severe stage group 4.21 (SD 0.58) DD (y) Mean 10.43 (SD 6.15) Cognition (MMSE) Baseline 24.79 (SD 5.82) | n =24 Mild-moderate group n = 10 Severe stage group n = 14 | Retrospective, study | 3-4 weeks (discharge) | Outcomes - Functional performance (BI) - cognition (MMSE, Token test, Phonemic and Semantic fluency, Copy and Recall Rey’s Figure, Ravens Colored Progressive Matrices) - geriatric domains (Numeric Rating scale, Norton scale, Conley scale) | Italian Ministry of Health Ricerca Corrente: RIN network | ⊕⊕ ◯◯ |
| II. Non-MR interventions | |||||||||
| Controlled studies (randomized) | |||||||||
| Jäggi et al. (2023) [56] | 2-4 weeks Exergame (Dividat Senso) rehabilitation, integrated into rehabilitation program | To determine the feasibility of exergaming and the effect on motor- and cognitive performance | H&Y IG median 3 CG median 3 DD (y) IG mean 7 CG mean 12.8 Cognition (MMSE) IG mean 27.79 CG mean 27.57 | Total n = 40; IG n = 19 CG n = 21 | Randomized, placebo controlled study | 2-4 weeks (discharge) | Primary outcomes Feasibility of the training -adherence rate - attrition rate AE - experience questions (SUS) - enjoyment (NASA-TLX) Secondary outcomes Cognitive - Go/No-Go test - color word interference test (D-KEFS) Motor - Reaction Time (RTT) - preferred-, maximum- and dual task- gait speed - physical performance (SPPB) - walking (TUG, 5xStS, TMT) | Swiss Federal Institute of Technology Zurich | ⊕⊕ ⊕ ◯ |
| Raciti et al. (2022) [57] | 8 weeks robotic therapy with an exoskeleton (ArmeoSpring/ Hocoma Inc, Zurich) | Evaluate the effect on hand dexterity and overall motor functions | H&Y IG median 2 (2-3) CG median 2 (2-3) DD (y) IG mean 5.3 (3.$) (6.2) CG mean 6.2 (4.6) Cognition Not mentioned | Total n = 24 IG n = 15 CG n = 9 | Single-blinded, randomized placebo-controlled study | 8 weeks(discharge) | Primary outcome –Functional performance (9HPT) Secondary outcomes - Motor performance (UPDRS-III, MI-UE, FMA-UE) - Functional performance (FIM) -Pain (P-NRS) | No external funding | ⊕⊕ ⊕ ◯ |
| Goosses et al. (2020) [58] | 8 days Music-Assisted Treadmill Training (MATT) | Feasibility and effect of the MATT program on motor- and cognitive functions, mood, fatigue and QoL | H&Y IG median 2.5 (R = 1.5) CG median 2.5 (R = 1.5) DD Mean 8.63 (6.60) IG 8.64 (5.49) CG 6.94 (4.25) Cognition (MoCA) 25 for both groups | Total n = 32 IG n = 15 CG n = 17 | Randomized controlled study | 8 days 6-weeks (telephone interview) | Outcomes - Pat subjective training perception - acceptance/feasibility of the therapy (Likert scale) - Cognition (MoCA, TAP, WMSR, LPS50+) - Motor function (UPDRS-III, -Functional integrity of lower extremity, walking, balance, gross and fine hand function, - FOG - Mood (GDS) - QoL (PDQ-39) - Fatigue (PSF-16) - Functional (FIM) | Budget resource of the participating study sites. The Gaittrainer 3 was sponsored by Biodex Medical systems Inc | ⊕⊕ ⊕ ◯ |
| Zhu et al. (2020) [59] | 12 week Tai Chi (+outpatient) | Effect of Tai Chi on motor- and non-motor symptoms | H&Y IG mean 2 (2.2) CG mean 2 (1.2) DD IG mean 4.68 (0.43) CG mean 4.00 (0.39) Cognition (MoCA) IG mean 21.37 (2.52) CG mean 22.05 (2.78) | Total n = 41 IG n = 19 CG n = 22 | Randomized placebo-controlled study | 12 weeks | Primary outcome - Motor performance (UPDRS-III, BBS) Secondary outcomes Non-motor - sleep quality (PDSS) - depression (HAMD) - anxiety state (HAMA) - cognitive function (MoCA) - quality of life (PDQ-39), | Project of Science Technology Department of Zhejiang Province | ⊕⊕ ⊕ ◯ |
| Controlled studies (non-randomized) | |||||||||
| De Luca et al. (2020) [60] | 8-week music assisted therapy (inpatient) | Effect on non-motor symptoms | H&Y Total mean 1.62 (0.57) IG mean 1.5 (0.53) CG mean 1.7 (0.59) DD Mean ? Cognition MMSE > 23 | Total n = 40 IG n = 20 CG n = 20 | Cross-sectional controlled study | 8 weeks (discharge) | Outcomes - psychological (PGWBI) - QoL (HRQoL) - Coping (Brief-COPE) - motor performance (FIM, TUG, 10mWT) | No external funding | ⊕⊕ ◯◯ |
| Brognara et al. (2020) [61] | Single mechanical stimulation by wearing insoles during 5 min | Effect of foot plantar stimulation on gait parameters | H&Y Not mentioned DD Mean not mentioned (residential profile) Cognition ? | Total n = 24 IG n = 12 CG n = 12 | Cross-sectional controlled study | 30 minutes | Outcomes Gait parameters - stride length - stride asymmetry - stride variability - pitch contact UPDRS total score Correlation between Gait and UPDRS total. | No external funding | ⊕◯◯◯ |
| Prospective open label studies | |||||||||
| Gassner et al. (2023) [62] | 4-week inpatient therapeutic climbing | Feasibility of therapeutic climbing integrated in rehabilitation and effect on gait | H&Y tot mean 1.92 H&Y1 16% (n = 4) H&Y2 68% (n = 17) H&Y3 (n = 4) DD (y) mean 7 (1-23) Cognition Not mentioned | n =26 | Prospective feasibility study | 4 weeks (discharge) | Primary outcome - Self-perceived differences in health and well-being (survey; no validation of survey) Secondary outcomes - 10MWT - Functional Gait Assessment - 2MWT - TAT - 9-HPT | Hilde-Ulrichs Foundation for Parkinson research | ⊕⊕ ◯ |
| Brandín-De la Cruz et al. (2020) [63] | 4-week virtual reality and antigravity treadmill training | Feasibility and efficacy of mechanical gait assistance combined with virtual reality | H&Y Mean 2.63 DD (y) Not mentioned Cognition Not mentioned | n =12 | Prospective feasibility study | 4 weeks (discharge) | Primary outcome -Motor performance (6MWT) Secondary outcomes - Gait speed (10MWT), - Balance (Tinetti scale), - QoL (SF-36) | Not mentioned | ⊕◯◯◯ |
| Retrospective studies | |||||||||
| Koyanagi et al. (2021) [64] | 4-week body weight-supported overground gait training (BWSOGT) | Effect of BWSOGT on functional and motor performance | H&Y IG median 3 (2-4) CG median 3 (2-4) DD (y) IG median 7 (5-13) CG median 7.5 (4.25- 10.5) Cognition (MMSE) IG median 26 (20-28) CG median 24.5 (20-27.5) | Total n = 37 IG n = 19 CG n = 18 | Retrospective case-controlled study | 4 weeks (discharge) | Outcomes - Functional performance (UPDRS-II) - Motor performance (UPDRS-III, 10-MWT, Velocity, stride length, 6-MWT, TUG, BBS, and FOG) | No specific funding | ⊕⊕ ◯◯ |
ADL, activities of daily living; APS, Atypical Parkinsonism; CBF, Cerebral blood flow; CG, Control group; DD, disease duration; fALFF, fractional amplitude of low-frequency; FOG, freezing of Gait; HC, healthy controls; IG, Intervention group; MR, multidisciplinary rehabilitation; PIGD, postural instability and gait difficulty-predominant disease; QoL, Quality of Life; Resting state BOLD, blood oxygen level dependent; Resting-state ASL, arterial spin labelling; Resting-state BOLD scan, blood oxygen level-dependent; TD, tremor dominant; UMCG, University Medical Center Groningen. Scales/tests: ACE-R, Addenbrooke’s Cognitive Examination-Revised; AES-D, Apathy evaluation scale, ALDS, AMC Linear Disability scale; BI, Barthel Index; BBS, Berg-Balance Scale; BDI, Beck depression inventory; Brief-COPE, Brief coping orientation in problems experiences; CERAD+, Consortium to Establish a Registry for Alzheimer’s Disease-Plus; Conley scale, assess the fall risk; CORSI, block tapping; D-KEFS, Delis–Kaplan Executive Function System; FES-I, Falls Efficacy Scale-international; FIM, Functional Independence Measure; FMA-UE, Fugl-Meyer Assessment for the upper extremity; FTSTS, 5-times sit to stand test; GDS, Geriatric Depression Scale; HADS, Hospital Anxiety and Depression Scale; HAMA-A, Hamilton Anxiety Rating Scale; HAMD, Hamilton rating scale for depression; 9-HPT, 9 hole Peg test; HRQoL, Health related Quality of life; IMET, Index zur Messung von Einschränkungen der Teilhabe (Measurement of Restrictions on Participation); LPS50+, Leistungsprüfsystem (cognition); MCGCF, Medical College of Georgia Complex Figures; MI-UE, Motricity Index for Upper Extremity; MoCA, Montreal Cognitive Assessment; MMSE, Mini-Mental State Examination; MMST, Mini Mental State; M-PAS, Modified Parkinson activity scale; MSST, Minute-Sit-to-Stand-Test; 6MT and 10MT test, 6 and 10 meter walk test; 6MWD test, 6 min walk distance test; NASA-TLX, Task-load Index; NMSQ, non-motor symptoms Questionnaire; Norton scale, risk of contracting pressure ulcers; NPI, NeuroPsychiatric Inventory; PDSS, Parkinson’s disease sleeping scale; PDQ-8 and -39, Parkinson’s disease questionnaire; PGIC, patient’s Global Impression of change; PGWBI, Psychological general well-being Index; PHQ-4, Patient Health Questionnaire for Depression and Anxiety; P-NRS, numerical rating scale of pain; PPT, Purdue Pegboard test; PSF, Parkinson Fatigue Scale; Quip, Questionnaire for Impulsive-compulsive Disorders in PD; RRT, Reaction Time Test; RWT, Regensburger Wortflüssigkeitstest; SAS, Starkstein Apathy scale; SCOPA, SCales for Outcome in PArkinson’s Disease; SCOPA-SPES, short Parkinson’s evaluation scale; SCOPA-COG, COGnition, SCOPA-PS, PsychoSocial; SCQ-D, Self-Administered Comorbidity Questionnaire; SF 36 and SF-12, Sort form health survey; SPPB, Short physical performance battery; 5xStS, 5 times Sit-to-Stand; SUS, system usability scale; TAP, Test of Attentional Performance; TAT, Tinetti Assessment Tool; TMT-A, Trail making test; TUG, Timed up-and-Go test; UPDRS I-IV, Unified Parkinson’s disease rating scale; VLMT, Verbal Learning and memory test; WMS-R, Wechsler Memory Scale revised; Assessment by GRADE (imprecision, inconsistency, indirectness, and publication bias), may affect the confidence in the results: ⊕⊕⊕ ⊕ High: We are very confident that the true effect lies close to that of the estimate of the effect. ⊕⊕ ⊕ ◯ Moderate: We are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.⊕⊕ ◯◯ Low: Our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. ⊕◯◯◯ Very low: We have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect.
Multidisciplinary treatment was defined as involving at least two different disciplines, including mostly a neurologist, physiotherapist, occupational therapist, Parkinson’s disease nurse, psychiatrist or (neuro)psychologist, social worker, dietician, and speech therapist. The H&Y rating and disease duration were used to characterize the severity of the disease.
RESULTS
Study characteristics and the quality of evidence
Overall, 22 studies were selected (Table 2), consisting of 13 studies covering inpatient multidisciplinary rehabilitation (MR) and 9 studies dealing with non-MR interventions, but sometimes added to an existing rehabilitation program, which was not the objective of the study. The MR studies consisted of only 3 controlled trials, 5 open label prospective trials, and 5 retrospective studies. The non-MR studies consisted of 6 controlled trials, including 4 randomized trials, 2 prospective open label trials, and 1 retrospective trial. This means that the overall quality of study designs of the non-MR interventions group was superior to the MR interventions group. The majority of the studies described the effect of the intervention on motor-, non-motor symptoms and quality of life scores as an endpoint. One study also measured the ability to prevent nursing home admission [47] and one study analyzed the effect of the treatment on neurovascular coupling, a MRI-based endpoint [48].
The multidisciplinary studies included PwPD with mild-to-moderate disability (H&Y 1-3, mean 2.9) with a disease duration in between 4.7-10.8 years (mean 8.5 years). Only 3 MR studies were controlled trials [47–49]. All of them were non-randomized trials, so including a risk of selection bias. However, 2 controlled MR studies showed a large magnitude of effect on different outcome measurements [47, 48] and have been upgraded from low to moderate quality of evidence for that reason. The other 10 MR studies were either prospective open label (n = 5) or retrospective studies (n = 5). The size of the studies differed significantly, varying between 24 and 591 PwPD, with a smaller number of participants in most prospective studies, and larger populations in the retrospective studies. Most trials reported loss of data and subjects dropping out of the studies. Selection- and recall biases also have affected the results negatively. The duration of the interventions varied from 1 to 8 weeks, with a mean duration of 6.5 weeks in the multidisciplinary studies. Long-term effects were measured in only 5 studies, 8 weeks up to 2 years. Funding was mentioned, if applicable, in all studies and most of the studies were funded by innovation funds, grants or financially supported by governments (12 studies). No studies had been supported by commercial parties.
The 9 non-MR studies consisted of virtual reality and antigravity treadmill training for gait rehabilitation [63], Music-Assisted Treadmill Training [58], body weight-supported overground gait training [64], gravity-supporting exoskeleton training [57], therapeutic climbing [62], exergames integrated in regular rehabilitation [56], and Tai Chi training [59]. The non-MR interventions only studied short-term outcomes. The H&Y scores varied from 1-3 (mean 2.3) with a disease duration between 4.7-10.8 years (mean 6.5 years). The non-MR group included 4 RCTs [56–59], 2 non-randomized controlled studies [60, 61], 2 prospective open label studies [62, 63], and 1 retrospective study [64]. Blinding issues, short duration of the intervention, confounders, inconsistency in measurements and high drop out rate/lost-to follow up, no intention to treat analysis and small sample size were reasons to downgrade the 4 RCTs from high to moderate quality of evidence. The number of subjects in this group varied between 12 and 41. The duration of the interventions varied from 30 min to 12 weeks (mean 5.4 weeks). All interventions only described short-term effects. Funding was described in all studies, except one who did not mention the funding [63]. One study was funded by a company providing the treatment assessed [58].
Quantitative comparisons of the main domains
Motor improvement
The most common outcome measure in the included studies was the motor score, most frequently assessed by the MDS-UPDRS III (8 studies in the MR group and 4 in the non-MR group, see Tables 2 and 3). Non-MR studies especially focused on gait speed, and on step- and stride length.
The short-term motor improvement in the controlled MR group varied between 17–19% [47–49]. Short-term motor improvement in the open label MR group varied between 13–30% [14, 15, 22, 50, 51]. Only 1 controlled MR study reported long-term change of motor scores, which worsened 2% after 2 years vs. baseline [47]. Long-term motor effects in the open label MR group varied from 16–17% [14, 15, 51].
The short-term motor scores of the controlled non-MR interventions varied from 8–26% [56, 57, 59, 60], whereas the open label studies varied from 15-65% [62, 63] in their overall motor scores. None of the non-MR interventions described long-term effects.
Functional improvement
Functional improvement was only measured in 10 out of 22 studies (5 studies in the MR group [15, 47, 50, 54, 55] and 5 in the non-MR group [56, 57, 62–64], see Tables 2 and 3) and was measured by a great variety of tests including the UPDRS-II, Barthel index, SF-36, 9HPT, and 10MWT.
Short-term outcome in the MR group improved in one controlled study with 18% [47], in two open label studies with 14% [15] and 31% [50]. Long-term effects in the MR group were measured in two studies. One controlled study reported an improvement of 6% after 2 years [47] and one open label study showed an improvement of 2.6% after 8 weeks [15].
Short-term effects in the controlled non-MR group were reported in two studies, both showing a score of 19% [56, 57]. In the prospective open label non-MR group functional outcome varied from 13–25% [62, 63]. None of the non-MR interventions described long-term effects on functional scales.
Improvement in cognition
Only 7 studies; 5 in the MR group [47, 50, 52, 53, 55] and 2 in the non-MR [56, 59] (Table 2) measured cognitive function, using different tests including MMSE, SCOPA-COG, MMST, MoCA, Go/no go, and D-KEFS.
Short-term cognitive improvement was seen in 4 studies within the MR group (Table 3); 27% in a controlled study [47], 4% in a non-controlled study [50] and only 1% in a retrospective study [52]. One retrospective study showed 1.5% worsening of cognition [53]. One controlled study in the MR group showed 9% worsening of cognition after 2 years, which was not significantly different vs. baseline [47].
Table 3
Percentage improvement in motor, functional, cognition and quality of life and medication changes/optimalization (LEDD in mg) in multidisciplinary interventions and non-multidisciplinary interventions. Short-term effects are expressed as % improvement vs. baseline. Most studies did not report long term effects, whereas the duration of follow-up showed significant variation
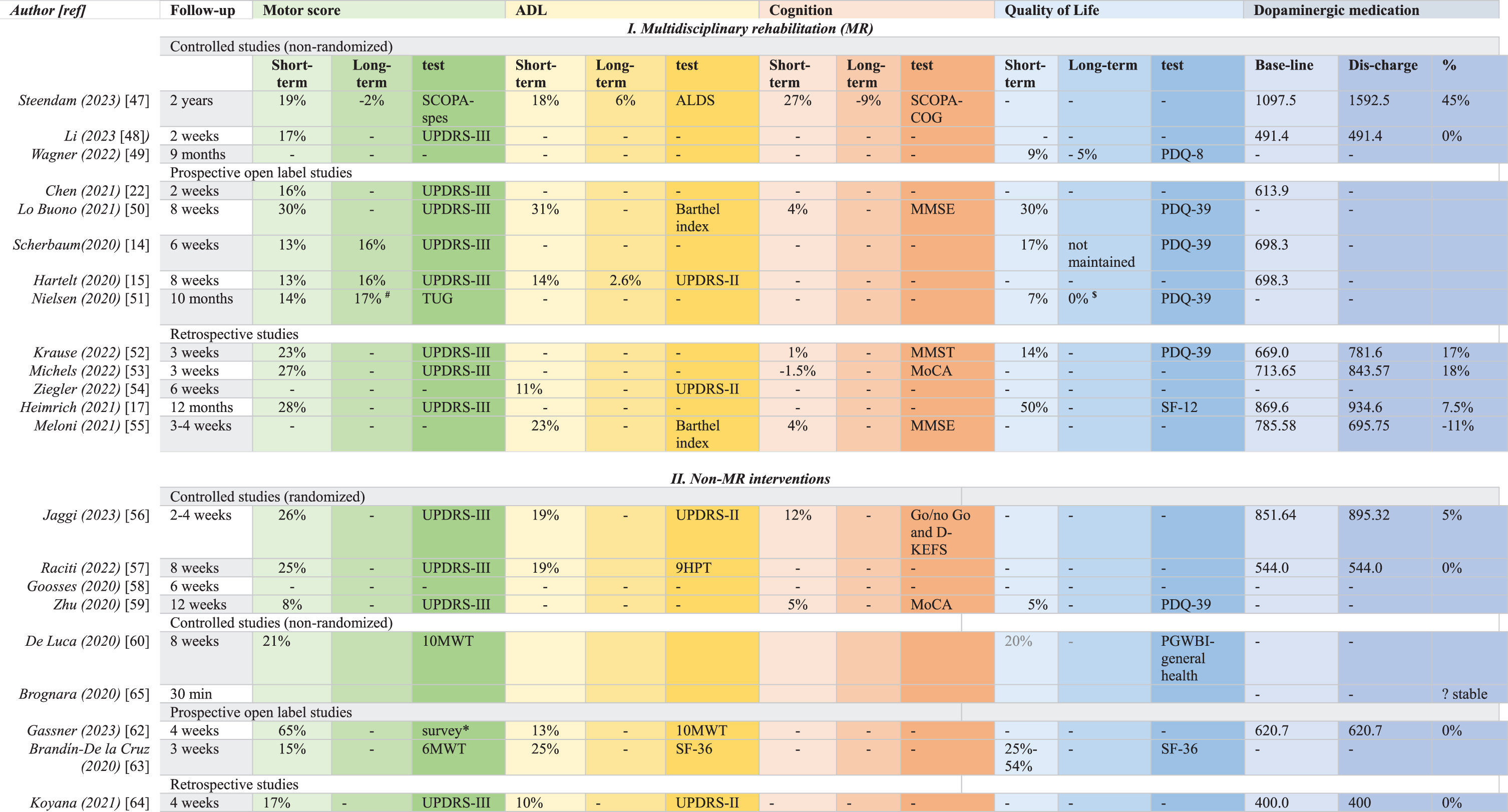 |
# measurement at 4 months $ measurement at 10 months *measured by a non validated survey.
Two RCTs in the non-MR group showed short-term cognitive improvement of 12% [56] and 5% [59]. None of the non-MR interventions described long-term effects on cognition.
Quality of life
Quality of life was assessed in 6 studies of the MR group [14, 17, 49–52] and in 2 studies of the non-MR group [59, 63], measured by PDQ-8, PDQ-39, SF-12, and SF-36 (Table 2).
Short-term improvement of QoL in the controlled MR group was 9% [49] and varied between 7–30% in the open label MR group [14, 50, 51]. Long-term data on QoL in 1 controlled study within the MR group [49], whereas 1 open label study showed stable QoL measures over 10 months [51].
The non-MR data showed significant short-term QoL improvement in 1 RCT of 5% [59], vs. 20% improvement in a controlled trial [60]. The open label non-MR studies showed 25–54% improvement of QoL [63]. No long-term data on QoL were reported in any of the non-MR interventions.
Medication changes
Although an optimal medication regimen is very important to support PwPD, not all interventions included medication optimization as an endpoint. LEDD changes were only registered in 6 studies [17, 47, 48, 52, 53, 55] of the MR group (Table 2) Two controlled studies [47, 48] showed a LEDD increase of 45% [47] and an unchanged LEDD [48]. Three non-controlled studies [17, 52, 53] showed an increase in between 7.5- and 18%. One retrospective study [55] showed a decrease in LEDD of 11%.
The non-MR group contained 4 studies [56, 57, 62, 64] which reported pre-and post test doses of dopaminergic drugs (LEDD). However, in 3 studies the LEDD was kept stable during the intervention, in order to differentiate between medication effects and the effects of the intervention [57, 62, 64].
Cost-effectiveness
Only 1 study [47] assessed the costs of the MR intervention and the outpatient follow-up. The inpatient costs were 12.500 € for about 6 weeks in the clinic, whereas the costs of the follow-up, including extra paramedical support were 4.000 € /3 months, resulting in total costs of 44.500 € /patient over 2-years in the intervention group, compared to 180.000 € /patient over 2-years in the control group, which was attributed to institutional costs related to nursing home admission (90.000 euro per year).
DISCUSSION
This review focused on 22 inpatient rehabilitation studies published in the last 3 years, 13 studies based on a multidisciplinary approach in rehabilitation centers or hospitals. This many studies in only 3 years shows the interest in the field. However, only 9 studies were performed with control groups, from which only 4 studies were randomized, only in the non-multidisciplinary field. This resulted in an overall rather poor GRADE rating, with only 6 studies reaching a moderate grade, no studies with a high grade and most studies having a low or even very low grade.
Previous recommendations of 20 expert centers in multidisciplinary PD care [66] indicated that an outpatient setting is the preferred way to organize care for most PwPD. However, an inpatient setting is the preferred way to perform PD rehabilitation, especially if multidisciplinary care is needed [21], people are about to lose their independence [47], or need to be treated with advanced therapies [66]. The final success of inpatient rehabilitation is highly dependent from proper communication with the involved outpatient (allied) healthcare professionals [47, 49]. For instance, a higher frequency of visits of the general physician was significantly associated with longer survival time, fewer inpatient days, and lower health care costs [67].
The issue of suboptimal medication regimens is very important. Suboptimal medication leads to impaired ADL and reduced QOL, and to unnecessary neuropsychiatric symptoms. Collaboration between pharmacists and physicians during all stages of the rehabilitation process therefore is required and beneficial [68, 69].
Strengths and limitations
Most of the included studies are no RCT studies, which is a risk for bias. PwPD with cognitive decline and/or co-morbidity were mostly excluded from the trials, whereas most trials included only PwPD in early H&Y stages (H&Y 1-3), with a relative short disease duration. So a significant proportion of PwPD were not represented by these studies [33, 70].
Some trials reported significant numbers lost to follow-up, even up to 20% or more, which implicates an great risk on selection bias [71]. However, it is good to realize that the review period overlapped with the COVID-19 pandemic, which might have contributed to less favorable continuation rates [72].
The lack of sustained benefit and long-term data is a major criticism of most multidisciplinary interventions included in this overview. The major limitation to compare all included studies properly was the huge diversity in endpoints. We have tried to overcome this issue, using percentages of change. However, the same percentage change of different scales may not represent the same magnitude of improvement, due to the metrics of the scale. Also the rather short follow-up periods, and the limited information on medication changes over time, have strongly limited the value and long-term impact of our conclusions.
Last but not least, this dataset showed a serious lack of functional data, which is quite astonishing talking about rehabilitation programs. Only 10 out of 22 studies reported a functional outcome, and just 9 studies included a QoL measure. Despite the suggestions made in previous reviews and meta-analyses, almost nothing has changed in the methodology and design of rehabilitation trials over the last years. Researchers have continued in the same vein, resulting in the same weak recommendations.
Conclusions
The 6 moderate grade studies permit some conclusions, which might benefit the field. The only long-term controlled multidisciplinary study [47] clearly showed that a focused inpatient program including medication optimization [69], followed by an intensive outpatient program, was able to keep PwPD stable for 2 years, whereas a matched control group all stayed in the nursing home. This means that focused PD rehabilitation, including medication optimization with advanced therapies like L-dopa- and apomorphine infusions if indicated, is able to postpone definite nursing home admission with years, resulting in a huge improvement of ADL and with a significant cost reduction at the same time. The short-term controlled MR study with intensive PD rehabilitation [48] confirmed these positive effects of focused PD rehabilitation. Finally, the controlled non-MR studies indicate that exergaming, treadmill training, Taichi and exoskeleton-supported PD rehabilitation seem to be effective interventions or add-ons to existing PD rehabilitation programs, at least on the short-term. However, no long-term data on any of these new interventions were reported.
Future perspectives
This review also makes clear that RCTs are perhaps not the best way to evaluate the structure and efficacy of rehabilitation models, because of the highly variable modes of intervention. RCTs are also difficult to execute if PwPD have cognitive pathology, which was the reason they had been excluded from almost all trials described above. As a result, it is hard to say if the current results can be applied in more vulnerable population of PwPD, having cognitive and behavioral problems [33, 70].
So, it is really time to harmonize the design of rehabilitation trials with PwPD, including bigger and broader populations, with a longer duration of follow-up and standardized endpoints, including functional measures, which are less dependent from the cognitive abilities of the participants [70, 73–75].
Finally, new objectives and new endpoints should be introduced in future PD rehabilitation trials, using recent insights in the pathophysiology and subtyping of PD. Thus, modulating the gut’s microbiome by diets, pre- and probiotics, (potentially) disease modifying drugs, anaerobic exercise and/or lifestyle changes constitute interesting new objectives to integrate in new trial designs. Biomarkers might be integrated in new study designs to tailor a personalized approach to rehabilitation, termed “rehabilomics” [76, 77]. Aerobic exercise, including high intensity training, in early PD has shown to increase the aerobic capacity, endurance, and seems to slow down the progression of motor symptoms [78, 79]. Exercise paradigms, incorporating both goal-based practice and aerobic training might work synergistically to promote neuroplasticity. Another approach is to look at dual task practices, without aerobic exercise, which provides insight into the role of cognitive motor training, without the exercise component [80, 81]. Preliminary data indicate an association between effective rehabilitation and brain reorganization, with restoration of the cortico–subcortical pathway and activation of compensatory networks, e.g., the frontoparietal networks [82, 83]. These approaches indicate a possible role of targeted rehabilitation in disease modification [48]. Finally, future trials should implement telehealth and wearables. Providing telehealth to participants exercising at home has proven to be substantially more time efficient for the physiotherapist, compared to center-based classes [84]. Participants identified a variety of benefits, including the reduction of financial- and travel-related burdens [85]. Digitalization and increased connectivity and the expanding capabilities of sensors will allow that more care can be given at home [86]. Artificial intelligence can be helpful to remotely assess the motor performance of PwPD. Finger-tapping is commonly used in neurological exams to evaluate bradykinesia. A recent study [87] developed computer algorithms to obtain objective measurements obtained from PwPD sitting in front of a webcam, that aligned with the MDS-UPDRS guideline, which proved to be strongly correlated with the neurologists’ ratings.
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
ESO has no conflict of interest to report. TVL reports consultancy: AbbVie, Britannia Pharm., Clexio; Grants: MJFF, Dutch Brain Foundation, UMCG, Parkinson NL and honoraria: AbbVie, Centrapharm, Britannia Pharm., Eurocept, Genilec, all outside the submitted work.
DATA AVAILABILITY
Data is available on request from the corresponding author.
REFERENCES
[1] | Pfeiffer RF ((2016) ) Non-motor symptoms in Parkinson’s disease. Parkinsonism Relat Disord 22 Suppl 1: , S119–S122. |
[2] | Post B , van der Eijk M , Munneke M , Bloem BR ((2011) ) Multidisciplinary care for Parkinson’s disease: Not if, but how!. Postgrad Med J 87: , 575–578. |
[3] | Chaudhuri KR , Healy DG , Schapira AH V , National Institute for Clinical Excellence ((2006) ) Non-motor symptoms of Parkinson’s disease: Diagnosis and management. Lancet Neurol 5: , 235–245. |
[4] | Dorsey ER , Sherer T , Okun MS , Bloem BR ((2018) ) The emerging evidence of the Parkinson pandemic. J Parkinsons Dis 8: , S3–S8. |
[5] | De Miranda BR , Goldman SM , Miller GW , Greenamyre JT , Dorsey ER ((2022) ) Preventing Parkinson’s disease: An environmental agenda. J Parkinsons Dis 12: , 45–68. |
[6] | Vossius C , Nilsen OB , Larsen JP ((2009) ) Parkinson’s disease and nursing home placement: The economic impact of the need for care. Eur J Neurol 16: , 194–200. |
[7] | Findley LJ , Wood E , Lowin J , Roeder C , Bergman A , Schifflers M ((2011) ) The economic burden of advanced Parkinson’s disease: An analysis of a UK patient dataset. J Med Econ 14: , 130–139. |
[8] | Huse DM , Schulman K , Orsini L , Castelli-Haley J , Kennedy S , Lenhart G ((2005) ) Burden of illness in Parkinson’s disease. Mov Disord 20: , 1449–1454. |
[9] | Martinez-Martin P , Macaulay D , Jalundhwala YJ , Mu F , Ohashi E , Marshall T , Sail K ((2019) ) The long-term direct and indirect economic burden among Parkinson’s disease caregivers in the United States. Mov Disord 34: , 236–245. |
[10] | Hely MA , Reid WGJ , Adena MA , Halliday GM , Morris JGL ((2008) ) The Sydney multicenter study of Parkinson’s disease: The inevitability of dementia at 20 years. Mov Disord 23: , 837–844. |
[11] | Safarpour D , Thibault DP , DeSanto CL , Boyd CM , Dorsey ER , Racette BA , Willis AW ((2015) ) Nursing home and end-of-life care in Parkinson disease. Neurology 85: , 413–419. |
[12] | Li Y , McLernon DJ , Counsell CE , Macleod AD ((2024) ) Incidence and risk factors of institutionalisation in Parkinson’s disease and atypical parkinsonism. Parkinsonism Relat Disord 118: , 105928. |
[13] | Jordan N , Deutsch A ((2022) ) Why and how to demonstrate the value of rehabilitation services. Arch Phys Med Rehabil 103: , S172–S177. |
[14] | Scherbaum R , Hartelt E , Kinkel M , Gold R , Muhlack S , Tönges L ((2020) ) Parkinson’s disease multimodal complex treatment improves motor symptoms, depression and quality of life. J Neurol 267: , 954–965. |
[15] | Hartelt E , Scherbaum R , Kinkel M , Gold R , Muhlack S , Tönges L ((2020) ) Parkinson’s Disease Multimodal Complex Treatment (PD-MCT): Analysis of therapeutic effects and predictors for improvement. J Clin Med 9: , 1874. |
[16] | Richter D , Bartig D , Muhlack S , Hartelt E , Scherbaum R , Katsanos AH , Müller T , Jost W , Ebersbach G , Gold R , Krogias C , Tönges L ((2019) ) Dynamics of Parkinson’s disease multimodal complex treatment in Germany from 2010-2016: Patient characteristics, access to treatment, and formation of regional centers. Cells 8: , 151. |
[17] | Heimrich KG , Prell T ((2021) ) Short- and long-term effect of Parkinson’s disease multimodal complex treatment. Brain Sci 11: , 1460. |
[18] | Frazzitta G , Bertotti G , Riboldazzi G , Turla M , Uccellini D , Boveri N , Guaglio G , Perini M , Comi C , Balbi P , Maestri R ((2012) ) Effectiveness of intensive inpatient rehabilitation treatment on disease progression in parkinsonian patients: A randomized controlled trial with 1-year follow-up. Neurorehabil Neural Repair 26: , 144–150. |
[19] | Frazzitta G , Maestri R , Bertotti G , Riboldazzi G , Boveri N , Perini M , Uccellini D , Turla M , Comi C , Pezzoli G , Ghilardi MF ((2015) ) Intensive rehabilitation treatment in early Parkinson’s disease: A randomized pilot study with a 2-year follow-up. Neurorehabil Neural Repair 29: , 123–131. |
[20] | Ferrazzoli D , Ortelli P , Zivi I , Cian V , Urso E , Ghilardi MF , Maestri R , Frazzitta G ((2018) ) Efficacy of intensive multidisciplinary rehabilitation in Parkinson’s disease: A randomised controlled study. J Neurol Neurosurg Psychiatry 89: , 828–835. |
[21] | Monticone M , Ambrosini E , Laurini A , Rocca B , Foti C ((2015) ) In-patient multidisciplinary rehabilitation for Parkinson’s disease: A randomized controlled trial. Mov Disord 30: , 1050–1058. |
[22] | Chen K-K , Jin Z-H , Gao L , Qi L , Zhen Q-X , Liu C , Wang P , Liu Y-H , Wang R-D , Liu Y-J , Fang J-P , Su Y , Yan X-Y , Liu A-X , Fang B-Y ((2021) ) Efficacy of short-term multidisciplinary intensive rehabilitation in patients with different Parkinson’s disease motor subtypes: A prospective pilot study with 3-month follow-up. Neural Regen Res 16: , 1336–1343. |
[23] | Fründt O , Mainka T , Schönwald B , Müller B , Dicusar P , Gerloff C , Buhmann C ((2018) ) The Hamburg Parkinson day-clinic: A new treatment concept at the border of in- and outpatient care. J Neural Transm (Vienna) 125: , 1461–1472. |
[24] | Johansson H , Folkerts A-K , Hammarström I , Kalbe E , Leavy B ((2023) ) Effects of motor-cognitive training on dual-task performance in people with Parkinson’s disease: A systematic review and meta-analysis. J Neurol 270: , 2890–2907. |
[25] | Pereira APS , Marinho V , Gupta D , Magalhães F , Ayres C , Teixeira S ((2019) ) Music therapy and dance as gait rehabilitation in patients with Parkinson disease: A review of evidence. J Geriatr Psychiatry Neurol 32: , 49–56. |
[26] | Machado Sotomayor MJ , Arufe-Giráldez V , Ruíz-Rico G , Navarro-Patón R ((2021) ) Music therapy and Parkinson’s disease: A systematic review from 2015-2020. Int J Environ Res Public Health 18: , 11618. |
[27] | Song R , Grabowska W , Park M , Osypiuk K , Vergara-Diaz GP , Bonato P , Hausdorff JM , Fox M , Sudarsky LR , Macklin E , Wayne PM ((2017) ) The impact of Tai Chi and Qigong mind-body exercises on motor and non-motor function and quality of life in Parkinson’s disease: A systematic review and meta-analysis. Parkinsonism Relat Disord 41: , 3–13. |
[28] | Lee H , Ko B ((2023) ) Effects of music-based interventions on motor and non-motor symptoms in patients with Parkinson’s disease: A systematic review and meta-analysis. Int J Environ Res Public Health 20: , 1046. |
[29] | Kola S , Subramanian I ((2023) ) Updates in Parkinson’s disease integrative therapies: An evidence-based review. Curr Neurol Neurosci Rep 23: , 717–726. |
[30] | Gilat M , Ginis P , Zoetewei D , De Vleeschhauwer J , Hulzinga F , D’Cruz N , Nieuwboer A ((2021) ) A systematic review on exercise and training-based interventions for freezing of gait in Parkinson’s disease. NPJ Parkinsons Dis 7: , 81. |
[31] | Seid AA , Demirdel E , Aychiluhm SB , Mohammed AA ((2022) ) Multidisciplinary rehabilitation for people with Parkinson’s disease: A systematic review and meta-analysis. Parkinsons Dis 2022: , 2355781. |
[32] | Yoon SY ((2022) ) Update on Parkinson’s disease rehabilitation. Brain Neurorehabilitation 15: , e15. |
[33] | Ernst M , Folkerts A-K , Gollan R , Lieker E , Caro-Valenzuela J , Adams A , Cryns N , Monsef I , Dresen A , Roheger M , Eggers C , Skoetz N , Kalbe E ((2023) ) Physical exercise for people with Parkinson’s disease: A systematic review and network meta-analysis. Cochrane Database Syst Rev 1: , CD013856. |
[34] | Muzerengi S , Herd C , Rick C , Clarke CE ((2016) ) A systematic review of interventions to reduce hospitalisation in Parkinson’s disease. Parkinsonism Relat Disord 24: , 3–7. |
[35] | Holden SK , Jones WE , Baker KA , Boersma IM , Kluger BM ((2016) ) Outcome measures for Parkinson’s disease dementia: A systematic review. Mov Disord Clin Pract 3: , 9–18. |
[36] | Darweesh SKL , Raphael KG , Brundin P , Matthews H , Wyse RK , Chen H , Bloem BR ((2018) ) Parkinson matters. J Parkinsons Dis 8: , 495–498. |
[37] | Pereira F , Verloo H , von Gunten A , Del Río Carral M , Meyer-Massetti C , Martins MM , Wernli B ((2022) ) Unplanned nursing home admission among discharged polymedicated older inpatients: A single-centre, registry-based study in Switzerland. BMJ Open 12: , e057444. |
[38] | Shih TM , Sail KR , Jalundhwala YJ , Sullivan J , van Eijndhoven E , Zadikoff C , Marshall TS , Lakdawalla DN ((2020) ) The effect of functional status impairment on nursing home admission risk among patients with advanced Parkinson’s disease. J Med Econ 23: , 297–307. |
[39] | Dockx K , Bekkers EM , Van den Bergh V , Ginis P , Rochester L , Hausdorff JM , Mirelman A , Nieuwboer A ((2016) ) Virtual reality for rehabilitation in Parkinson’s disease. Cochrane Database Syst Rev 12: , CD010760. |
[40] | Sarasso E , Gardoni A , Tettamanti A , Agosta F , Filippi M , Corbetta D ((2022) ) Virtual reality balance training to improve balance and mobility in Parkinson’s disease: A systematic review and meta-analysis. J Neurol 269: , 1873–1888. |
[41] | Zanatta F , Farhane-Medina NZ , Adorni R , Steca P , Giardini A , D’Addario M , Pierobon A ((2023) ) Combining robot-assisted therapy with virtual reality or using it alone? A systematic review on health-related quality of life in neurological patients. Health Qual Life Outcomes 21: , 18. |
[42] | Tao Y , Luo J , Tian J , Peng S , Wang H , Cao J , Wen Z , Zhang X ((2023) ) The role of robot-assisted training on rehabilitation outcomes in Parkinson’s disease: A systematic review and meta-analysis. Disabil Rehabil, doi: 10.1080/09638288.2023.2266178. |
[43] | Xue X , Yang X , Deng Z ((2023) ) Efficacy of rehabilitation robot-assisted gait training on lower extremity dyskinesia in patients with Parkinson’s disease: A systematic review and meta-analysis. Ageing Res Rev 85: , 101837. |
[44] | Özden F ((2023) ) The effect of mobile application-based rehabilitation in patients with Parkinson’s disease: A systematic review and meta-analysis. Clin Neurol Neurosurg 225: , 107579. |
[45] | Proud EL , Miller KJ , Morris ME , McGinley JL , Blennerhassett JM ((2023) ) Effects of upper limb exercise or training on hand dexterity and function in people with Parkinson disease: A systematic review and meta-analysis. Arch Phys Med Rehabil, doi: 10.1016/j.apmr.2023.11.009. |
[46] | Ryan R , Hill S ((2016) ) How to GRADE the quality of the evidence. Cochrane Consumers and Communication Group. |
[47] | Steendam-Oldekamp E , Weerkamp N , Vonk JM , Bloem BR , van Laar T ((2023) ) Combined multidisciplinary in/outpatient rehabilitation delays definite nursing home admission in advanced Parkinson’s disease patients. Front Neurol 14: , 1128891. |
[48] | Li T , Wang L , Piao Z , Chen K , Yu X , Wen Q , Suo D , Zhang C , Funahashi S , Pei G , Fang B , Yan T ((2023) ) Altered neurovascular coupling for multidisciplinary intensive rehabilitation in Parkinson’s disease. J Neurosci 43: , 1256–1266. |
[49] | Wagner L , Hauptmann B , Hoffmann A-K , Jochems N , Schmeier B , Schrader A , Kohlmann T , Deck R ((2022) ) Evaluation of an individualized, tablet-based physiotherapy training programme for patients with Parkinson’s disease: The ParkProTrain study, a quasi-randomised controlled trial. BMC Neurol 22: , 176. |
[50] | Lo Buono V , Palmeri R , De Salvo S , Berenati M , Greco A , Ciurleo R , Sorbera C , Cimino V , Corallo F , Bramanti P , Marino S , Di Lorenzo G , Bonanno L ((2021) ) Anxiety, depression, and quality of life in Parkinson’s disease: The implications of multidisciplinary treatment. Neural Regen Res 16: , 587–590. |
[51] | Nielsen C , Siersma V , Ghaziani E , Beyer N , Magnusson SP , Couppé C ((2020) ) Health-related quality of life and physical function in individuals with Parkinson’s disease after a multidisciplinary rehabilitation regimen-a prospective cohort feasibility study. Int J Environ Res Public Health 17: , 7668. |
[52] | Krause P , Berking S , Astalosch M , Grünheid R , Kühn AA ((2022) ) Motor and non-motor improvements following short-term multidisciplinary day-clinic care in Parkinson's disease. J Neural Transm (Vienna) 129: , 1419–1426. |
[53] | Michels J , van der Wurp H , Kalbe E , Rehberg S , Storch A , Linse K , Schneider C , Gräber S , Berg D , Dams J , Balzer-Geldsetzer M , Hilker-Roggendorf R , Oberschmidt C , Baudrexel S , Witt K , Schmidt N , Deuschl G , Mollenhauer B , Trenkwalder C , Liepelt-Scarfone I , Spottke A , Roeske S , Wüllner U , Wittchen H-U , Riedel O , Kassubek J , Dodel R , Schulz JB , Costa AS , Reetz K ((2022) ) Long-term cognitive decline related to the motor phenotype in Parkinson’s disease. J Parkinsons Dis 12: , 905–916. |
[54] | Ziegler K , Messner M , Paulig M , Starrost K , Reuschenbach B , Fietzek UM , Ceballos-Baumann AO ((2023) ) Activities of daily living are improved by inpatient multimodal complex treatment for PD-a real-world cohort study. Mov Disord Clin Pract 10: , 42–54. |
[55] | Meloni M , Saibene FL , Di Tella S , Di Cesare M , Borgnis F , Nemni R , Baglio F ((2021) ) Functional and cognitive improvement after an intensive inpatient multidisciplinary rehabilitation program in mild to severe Parkinson’s disease: A retrospective and observational study. Front Neurol 12: , 626041. |
[56] | Jäggi S , Wachter A , Adcock M , de Bruin ED , Möller JC , Marks D , Schweinfurther R , Giannouli E ((2023) ) Feasibility and effects of cognitive-motor exergames on fall risk factors in typical and atypical Parkinson’s inpatients: A randomized controlled pilot study. Eur J Med Res 28: , 30. |
[57] | Raciti L , Pignolo L , Perini V , Pullia M , Porcari B , Latella D , Isgrò M , Naro A , Calabrò RS ((2022) ) Improving upper extremity bradykinesia in Parkinson’s disease: A randomized clinical trial on the use of gravity-supporting exoskeletons. J Clin Med 11: , 2543. |
[58] | Gooßes M , Saliger J , Folkerts A-K , Nielsen J , Zierer J , Schmoll P , Niepold A , Colbach L , Leemhuis J , Engels L , van Krüchten M , Ophey A , Allert N , Karbe H , Kalbe E ((2020) ) Feasibility of music-assisted treadmill training in Parkinson’s disease patients with and without deep brain stimulation: Insights from an ongoing pilot randomized controlled trial. Front Neurol 11: , 790. |
[59] | Zhu M , Zhang Y , Pan J , Fu C , Wang Y ((2020) ) Effect of simplified Tai Chi exercise on relieving symptoms of patients with mild to moderate Parkinson’s disease. J Sports Med Phys Fitness 60: , 282–288. |
[60] | De Luca R , Latella D , Maggio MG , Leonardi S , Sorbera C , Di Lorenzo G , Balletta T , Cannavò A , Naro A , Impellizzeri F , Calabrò RS ((2020) ) Do patients with PD benefit from music assisted therapy plus treadmill-based gait training? An exploratory study focused on behavioral outcomes. Int J Neurosci 130: , 933–940. |
[61] | Brognara L , Cauli O ((2020) ) Mechanical plantar foot stimulation in Parkinson’s disease: A scoping review. Diseases 8: , 12. |
[62] | Gassner L , Dabnichki P , Pokan R , Schmoeger M , Willinger U , Maetzler W , Moser H , Zach H ((2023) ) Therapeutic climbing in Parkinson’s disease: Differences in self-reported health and well-being, feasibility and clinical changes. Physiother Theory Pract 39: , 1163–1177. |
[63] | Brandín-De la Cruz N , Secorro N , Calvo S , Benyoucef Y , Herrero P , Bellosta-López P ((2020) ) Immersive virtual reality and antigravity treadmill training for gait rehabilitation in Parkinson’s disease: A pilot and feasibility study. Rev Neurol 71: , 447–454. |
[64] | Koyanagi Y , Fukushi I , Nakamura M , Suzuki K , Oda N , Aita T , Seki H ((2021) ) The effect of body weight-supported overground gait training for patients with Parkinson’s disease: A retrospective case-control observational study. PLoS One 16: , e0254415. |
[65] | Brognara L , Mafla-España MA , Gil-Molina I , Castillo-Verdejo Y , Cauli O ((2022) ) The effects of 3D custom foot orthotics with mechanical plantar stimulation in older individuals with cognitive impairment: A pilot study. Brain Sci 12: , 1669. |
[66] | Radder DLM , Nonnekes J , van Nimwegen M , Eggers C , Abbruzzese G , Alves G , Browner N , Chaudhuri KR , Ebersbach G , Ferreira JJ , Fleisher JE , Fletcher P , Frazzitta G , Giladi N , Guttman M , Iansek R , Khandhar S , Klucken J , Lafontaine A-L , Marras C , Nutt J , Okun MS , Parashos SA , Munneke M , Bloem BR ((2020) ) Recommendations for the organization of multidisciplinary clinical care teams in Parkinson’s disease. J Parkinsons Dis 10: , 1087–1098. |
[67] | Fujita T , Babazono A , Kim S-A , Jamal A , Li Y ((2021) ) Effects of physician visit frequency for Parkinson’s disease treatment on mortality, hospitalization, and costs: A retrospective cohort study. BMC Geriatr 21: , 707. |
[68] | Ensing HT , Koster ES , Stuijt CCM , van Dooren AA , Bouvy ML ((2015) ) Bridging the gap between hospital and primary care: The pharmacist home visit. Int J Clin Pharm 37: , 430–434. |
[69] | Stuijt CCM , van den Bemt BJF , Boerlage VE , Janssen MJA , Taxis K , Karapinar-Çarkit F ((2022) ) Differences in medication reconciliation interventions between six hospitals: A mixed method study. BMC Health Serv Res 22: , 722. |
[70] | Bloem BR , Ypinga JHL , Willis A , Canning CG , Barker RA , Munneke M , De Vries NM ((2018) ) Using Medical claims analyses to understand interventions for Parkinson patients. J Parkinsons Dis 8: , 45–58. |
[71] | Dumville JC , Torgerson DJ , Hewitt CE ((2006) ) Reporting attrition in randomised controlled trials. BMJ 332: , 969–971. |
[72] | Braun T , Weidmann R , Möller JC , Ammann A , Marks D ((2021) ) The impact of a coronavirus disease 2019 pandemic-related interruption of regular physical rehabilitation on functional abilities in a patient with two chronic neurological diseases: A case report. J Med Case Rep 15: , 503. |
[73] | Lidstone SC , Bayley M , Lang AE ((2020) ) The evidence for multidisciplinary care in Parkinson’s disease. Expert Rev Neurother 20: , 539–549. |
[74] | Ernst M , Folkerts A-K , Gollan R , Lieker E , Caro-Valenzuela J , Adams A , Cryns N , Monsef I , Dresen A , Roheger M , Eggers C , Skoetz N , Kalbe E ((2023) ) Physical exercise for people with Parkinson’s disease: A systematic review and network meta-analysis. Cochrane Database Syst Rev 1: , CD013856. |
[75] | Kalbe E , Aarsland D , Folkerts A-K ((2018) ) Cognitive interventions in Parkinson’s disease: Where we want to go within 20 years. J Parkinsons Dis 8: , S107–S113. |
[76] | Wagner AK ((2017) ) TBI rehabilomics research: An exemplar of a biomarker-based approach to precision care for populations with disability. Curr Neurol Neurosci Rep 17: , 84. |
[77] | Cao W , Zhang X , Qiu H ((2023) ) Rehabilomics: A state-of-the-art review of framework, application, and future considerations. Front Neurol 14: , 1103349. |
[78] | Schootemeijer S , van der Kolk NM , Bloem BR , de Vries NM ((2020) ) Current perspectives on aerobic exercise in people with Parkinson’s disease. Neurotherapeutics 17: , 1418–1433. |
[79] | van der Kolk NM , de Vries NM , Kessels RPC , Joosten H , Zwinderman AH , Post B , Bloem BR ((2019) ) Effectiveness of home-based and remotely supervised aerobic exercise in Parkinson’s disease: A double-blind, randomised controlled trial. Lancet Neurol 18: , 998–1008. |
[80] | Petzinger GM , Fisher BE , McEwen S , Beeler JA , Walsh JP , Jakowec MW ((2013) ) Exercise-enhanced neuroplasticity targeting motor and cognitive circuitry in Parkinson’s disease. Lancet Neurol 12: , 716–26. |
[81] | Petzinger GM , Holschneider DP , Fisher BE , McEwen S , Kintz N , Halliday M , Toy W , Walsh JW , Beeler J , Jakowec MW ((2015) ) The effects of exercise on dopamine neurotransmission in Parkinson’s disease: Targeting neuroplasticity to modulate basal ganglia circuitry. Brain Plast 1: , 29–39. |
[82] | Marinelli L , Trompetto C , Canneva S , Mori L , Nobili F , Fattapposta F , Currà A , Abbruzzese G , Ghilardi MF ((2017) ) Learning “how to learn”: Super declarative motor learning is impaired in Parkinson’s disease. Neural Plast 2017: , 3162087. |
[83] | Baglio F , Pirastru A , Bergsland N , Cazzoli M , Tavazzi E ((2022) ) Neuroplasticity mediated by motor rehabilitation in Parkinson’s disease: A systematic review on structural and functional MRI markers. Rev Neurosci 33: , 213–226. |
[84] | Flynn A , Preston E , Dennis S , Canning CG , Allen NE ((2021) ) Home-based exercise monitored with telehealth is feasible and acceptable compared to centre-based exercise in Parkinson’s disease: A randomised pilot study. Clin Rehabil 35: , 728–739. |
[85] | Goldman JG , Merkitch D , Brewington D , Peirce H , Rho M , Jayabalan P , Curran J , Brennan K ((2023) ) Patient experiences receiving rehabilitation care via telehealth: Identifying opportunities for remote care. Front Rehabil Sci 4: , 1049554. |
[86] | Luis-Martínez R , Monje MHG , Antonini A , Sánchez-Ferro Á , Mestre TA ((2020) ) Technology-enabled care: Integrating multidisciplinary care in Parkinson’s disease through digital technology. Front Neurol 11: , 575975. |
[87] | Islam MS , Rahman W , Abdelkader A , Lee S , Yang PT , Purks JL , Adams JL , Schneider RB , Dorsey ER , Hoque E ((2023) ) Using AI to measure Parkinson’s disease severity at home. NPJ Digit Med 6: , 156. |




