Comparison of the kinetics of monomeric anthocyanins loss and colour changes in thermally treated Blackcurrant, Maqui Berry and Blueberry pulps from Argentina
Abstract
BACKGROUND:
Fruits like blackcurrant, blueberry and maqui berry stand out for their high content of anthocyanins that give products an attractive red colour, in addition to their rich composition of other polyphenols and vitamins. Comprehensive understanding of thermal properties of foods and quantitative changes in quality attributes are necessary for the proper design of thermal processes.
OBJECTIVE:
The aim of the present study was to evaluate the kinetics of monomeric anthocyanin (MAcy) and colour degradation due to thermal processing of pulps of blackcurrant, blueberry and maqui berry produced in El Bolsón, Argentina.
METHODS:
Anthocyanins content was measured with pH-differential method. CIELab parameters were obtained with a Minolta Spectrophotometer, total phenolics were determined by Folin-Ciocalteu method. MAcy degradation and evolution of colour parameter a* were fitted to a first–order model.
RESULTS:
Activation energies for anthocyanins losses were between 81 and 94 kJ/mol, and for the deterioration of colour between 85 and 89 kJ/mol. In the case of deterioration of anthocyanins, maqui berry presented the highest values of half-life times especially at 90°C. In the case of colour, blackcurrant showed greater stability showing average colour degradation half-life times between 5 and 11 times higher than those of anthocyanins loss.
CONCLUSIONS:
Kinetic parameters calculated for different berry juices can be used to design a thermal treatment to obtain a high retention of colour and bioactive compounds.
1Introduction
The development of products based on berries is gaining importance due to their potential use as a food ingredient, natural colourant and nutraceutical applications. In Argentina (Patagonia Region), berries like elderberry (Sambucus nigra), blackcurrant (Ribes nigrum), maqui berry (Aristotelia chilensis) and blueberry (Vaccinium corymbosum), are considered products with high quality. In addition to its consumption as fresh fruit and in the elaboration of juices, jams and preserves, producers have a great interest in a potential use as functional foods because of its high content of bioactive compounds.
Berries stand out for their high content of anthocyanins that give products an attractive red colour, in addition to their rich composition of other polyphenols, essential amino acids, vitamin C and minerals. Moreover, these fruits present high phenolic content and antioxidant activity [1–3]. Numerous studies have reported the effects of berry anthocyanins as an antioxidant, anticarcinogenic and immune–stimulating agents among other health benefits [4–6]. The stability of anthocyanins is influenced by several factors, such as temperature, light, pH, metal ions, oxygen. Degradation of these compounds has an effect on product quality and therefore consumer acceptance [7].
Differences in anthocyanin profiles of fruits and overall composition can also determine the level of deterioration. The presence of acylated or polyglycosylated anthocyanins, phenolics acids or inorganic salts has an effect on the stability of anthocyanins and resulting juice colour after processing and storage [8, 9]. Besides, chemical composition of fruits is conditioned by genetic, environmental factors and maturation degree [10].
Different thermal treatments are performed in products based on berries during the processing steps, with the aim of produce safety and acceptable shelf-life products. These processes, which generally involve temperatures lower than 100°C in acidic products, could lead to the degradation of anthocyanins and product quality [11]. The effect will be dependent on the length of time and treatment applied, so that the knowledge of the kinetic parameters of these processes (reaction rates, activation energy, and half-life times) will be essential to predict changes that will occur in the products during processing.
Proper design of thermal processes requires the comprehensive understanding of thermal properties of foods and quantitative changes in quality attributes in order to predict and control the food quality changes [12].
The aim of the present study was to determine anthocyanin content and evaluate the kinetics of anthocyanin and colour degradation due to thermal processing in pulps of three different berries: blueberry, blackcurrant and maqui berry from El Bolsón, Rio Negro, Argentina. The stability of the total phenolics content was also evaluated.
2Materials and methods
2.1Samples
Blueberry, blackcurrant and maqui berry pulps were provided by a producer from El Bolsón, Río Negro (Argentina). Fruits used by the producer for preparing pulps were of similar maturation degree, and were collected in areas next to El Bolsón. The pulps were obtained from fruit blanched in water (80°C, 3 min) and then crushed and peeled with an industrial pulper, packed, pasteurised (85°C, 15 min) and frozen at –18°C.
2.2Heat treatment of berries pulps to obtain kinetic parameters
Pulps (processed as described in 2.1) was thawed and put in Pyrex tubes, well capped to avoid evaporation and placed in a water bath at the constant temperature of 70°C or 75°C, 80°C and 90°C. At regular time intervals, samples were removed from the bath, cooled by plunging into ice water and then stored at –18°C until they were analysed. In order to obtain kinetic parameters (degradation rates, half-life times, activation energies), the total heating times at each temperature were selected based on previous studies’ data [13–17]. These experimental times are generally longer than those used in industrial processes, but allow obtain enough data to make the kinetic modelling. It is also important that the reaction is carried out far enough to distinguish the order of reaction [12].
2.3Preparation of extracts from berries pulps
Pulps thermally treated were used to prepare alcoholic extracts using 5 g of pulp. Samples were extracted twice in 20 ml ethanol: HCl 0.1N (85 : 15) (solvents were acquired from Merck, Darmstadt, Germany), then were centrifuged (g-force 2550×g, 15 min) and supernatant was mixed and utilised for measurements of total phenolics, monomeric anthocyanins, colour, colour density and polymeric colour. All the results are based on initial pulp taking into account the corresponding pulp density.
2.4Methods
2.4.1Physicochemical properties
The total soluble solids content was evaluated in °Brix with a manual refractometer Atago N2 (Tokyo, Japan), pH measurements were taken at 25°C using Hanna HI 8424 equipment (Hanna Instruments Inc., Woonsocket, RI, USA).
2.4.2Total phenolics
Total phenolics (TP) were determined in the extracts using the Folin–Ciocalteu method according to Waterhouse [18], (Folin–Ciocalteu reagent was purchased from Merck KgaA Darmstadt, Germany). Sample absorbance at 765 nm (spectrophotometer PG Instruments T60U UV-Vis, Leicestershire, United Kingdom) was measured, and phenolic concentrations were expressed as Gallic acid equivalent (GAE) in g/L calculated by means of a standard curve of Gallic acid (obtained from Anedra, Buenos Aires,Argentina).
2.4.3Monomeric anthocyanin content
Monomeric anthocyanin content (MAcy) of extracts was determined by pH differential method described by Giusti and Wrolstad [19], using potassium chloride buffer (pH 1.0) and sodium acetate buffer (pH 4.5). A sample of pulp extract was mixed with the corresponding buffer, in a dilution to achieve an adequate reading of absorbance, that was read at 510 and 700 nm. Monomeric anthocyanin content was expressed as cyanidin-3-glucoside in mg/L according to the following equation:
(1)
2.4.4Colour measurements
The colour of pulps was analysed using a Minolta Spectrophotometer CM-600d (Konica Minolta Observer), with D65 illuminant, and an observer angle of 2°. The calibration was done with standard white and black tiles.The colour measurement was made by applying 1 ml of pulp extract in plastic white containers. CIELab parameters (CIE 1976 L* a* b*) were L* for lightness, a* for redness and b* for yellowness. Calculations for h0 (hue angle) as arctan b*/a* were made. Total colour difference was calculated as
2.4.5Colour density, polymeric colour and percent polymeric colour
The percent polymeric colour was determined using the method described by Giusti and Wrolstad [19]. Samples of pulp extract were treated with a potassium metabisulfite solution for bleaching or with water as a control. Absorbance was read at 420, 510 and 700 nm. Colour density (CD) was calculated using the control sample according to Equation (1), being DF the dilution factor for Eqs. (1) and (2).
(2)
Polymeric colour (PC) was determined using the bisulfite-bleached sample according to Equation (2).
(3)
Percent polymeric colour (% PC) was calculated using the Equation (4).
(4)
2.4.6Kinetics model
Monomeric anthocyanin (MAcy) degradation and the evolution of colour parameter a* has been found to follow a first –order reaction model [20–22]:
(5)
(6)
(7)
(8)
C0 = MAcy content and a* colour parameter at initial time.
C = MAcy content and a* colour parameter after t hours of heating at a given temperature.
kMA: reaction rate constant for MAcy degradation.
ka: reaction rate constant for a* degradation.
t 1/2: half- life time, time needed for 50% degradation of MAcy or a* colour parameter.
k1, k2: degradation rates at T1 and T2.
2.5Data analysis
Thermal treatment of juices was performed by duplicate. At selected heating and storage times samples were analysed. All the parameters studied were determined in duplicate for each pulp sample, in the case of monomeric anthocyanins determination by pH differential method and colour measurements determinations were performed in triplicate. Average of measurements was reported. Colour parameters at initial and final time of thermal treatments were analysed by one-way analysis of variance (ANOVA) test using Infostat v.2009 (Universidad Nacional de Córdoba, Argentina). Means comparisons were carried out by Student Newman-Keuls (SNK) test at P < 0.05. Pearson’s correlations between anthocyanins content and colour parameters were performed.
3Results and discussion
3.1Degradation Kinetics of monomeric anthocyanins (MAcy) and a* colour parameter in blueberry, blackcurrant and maqui berry pulps during heat treatment
Table 1 shows the characteristics of different pulps. Blackcurrant (BC) has the highest total phenolics content followed by maqui berry (MB) and blueberry (BB). Percentage of soluble solids follows the same order (BC>MB>BB) but the relative ratio between soluble solids and phenolics content between the fruits is different: relative soluble solids content 1.7: 1.4: 1 and TP content: 4: 2.1: 1 (BC:MB:BB). This indicates that blackcurrant has a greater proportion of total phenolics per gramme of soluble solid. In the case of monomeric anthocyanins content, BC and MB have higher levels than BB, MB showing the highest content with 1795 mg/L. Based on the results showed in Table 1, the percentage of monomeric anthocyanins relative to TP is about: 14.4% for BC, 29.3% for MB and 9.7% for BB. In a previous work, similar relationships were obtained for elderberry pulp, that showed a MAcy content of 994 mg/L, that represented 15.8% of total phenolics [23].
Table 1
Physico-chemical characteristics of the different berry juices
| Total soluble solids (°Brix) | pH | Total phenolics content (mg/L) | Monomeric anthocyanins (mg/L) | |
| Blackcurrant | 15.0±0.1 | 3.07±0.01 | 11737±457 | 1222±47 |
| Maqui berry | 13.0±0.1 | 4.17±0.01 | 6128±310 | 1795±19 |
| Blueberry | 9.0±0.1 | 3.46±0.01 | 2963±136 | 287±20 |
Values are mean±standard error.
Figure 1 shows the evolution of MAcy degradation following a first-order reaction model described inequation (5). Degradation rates and half-life times (t1/2) of monomeric anthocyanins estimated at the different temperatures are shown in Table 2. In all cases, the r2 values were higher than 0.97, indicating a good data fit to the first-order reaction model.
Fig.1
Comparison of kinetics loss of monomeric anthocyanins and colour in heated berries form Río Negro, Argentina.
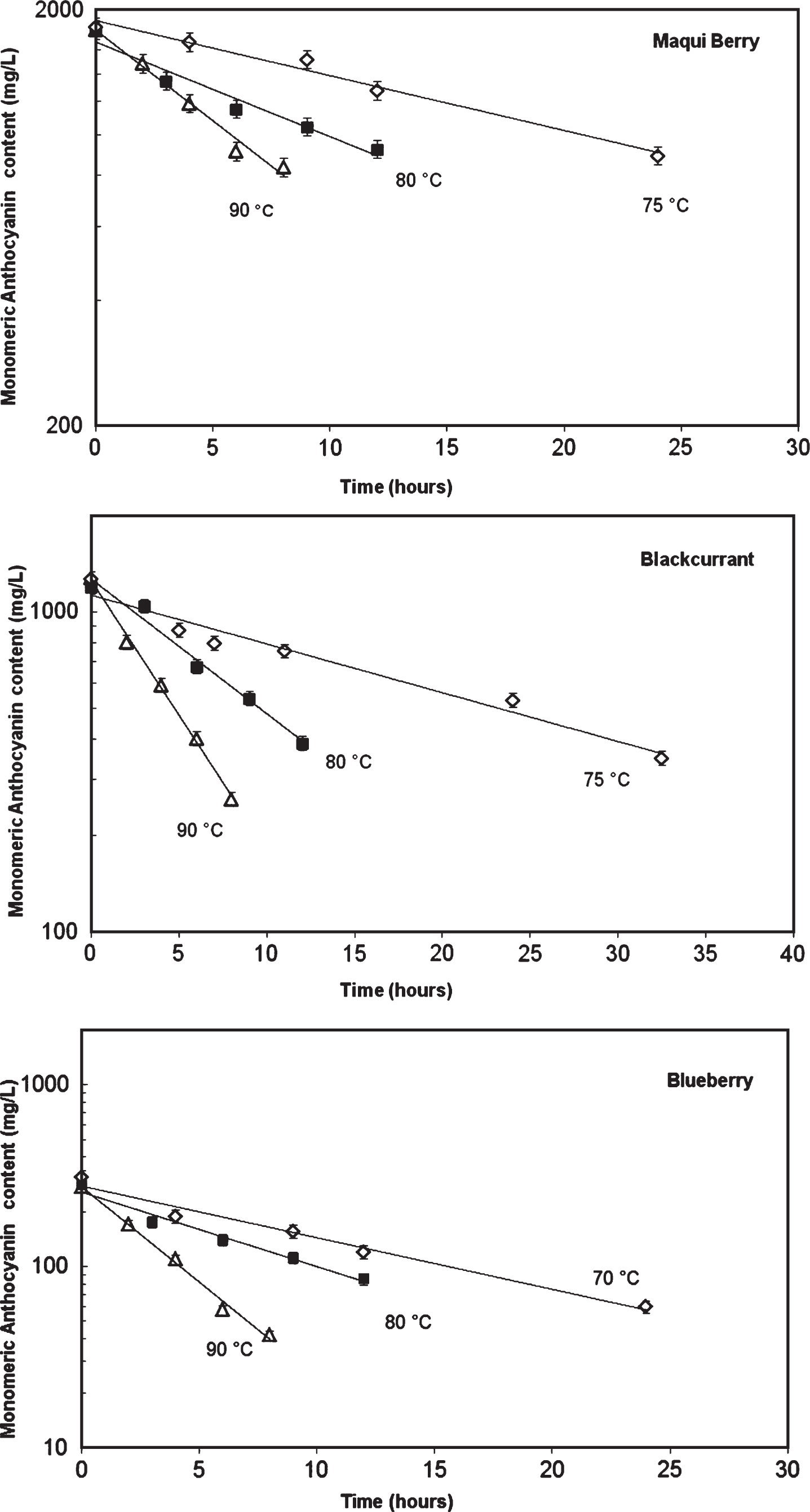
Table 2
Kinetic parameters describing the loss of Monomeric anthocyanins in heated berry juices
| Berry | Temperature (°C) | k (h–1) | t1/2 (h) | Ea (kJ/mol) | Q10 (80–90°C) |
| Blackcurrant | 75 | 0.0316±0.0016 | 21.9±1.3 | 94±4 | 2.0±0.1 |
| 80 | 0.0969±0.0058 | 7.2±0.4 | |||
| 90 | 0.1935±0.011 | 1.8±0.1 | |||
| Maqui berry | 75 | 0.0304±0.0018 | 22.8±1.6 | 81±4 | 1.9±0.1 |
| 80 | 0.0522±0.0003 | 13.3±0.8 | |||
| 90 | 0.1003±0.0050 | 6.9±0.3 | |||
| Blueberry | 70 | 0.0655±0.0033 | 12.7±0.8 | 92±5 | 2.6±0.1 |
| 80 | 0.0945±0.0042 | 7.3±0.4 | |||
| 90 | 0.2419±0.0145 | 1.4±0–1 |
Values are mean±standard error.
As shown in Table 2, half-life times decreased significantly with temperature showing the decrease in MAcy stability as temperature increases. Other authors have reported similar half-lives for blueberry juice 8.9° Brix, 8.6 h at 70°C and 5.1 h a 80°C [24], and for blackcurrant juice, 9 h at 80°C [25].
The effect of temperature on the degradation rate constants for MAcy was expressed by the linearized Arrhenius equation by plotting ln k against 1/T in which the temperature dependence of k was quantified by the activation energy Ea according to:
(9)
A lower activation energy value for MAcy degradation obtained for MB would indicate lower temperature dependence compared with blackcurrant and blueberry.
Q10 values in the range 80–90°C were between 1.9–2.6 for different fruits indicating that degradation rate duplicate when increasing treatment temperature in 10°C. These results show a significant effect of temperature on degradation anthocyanins for all fruits, although differences can be observed between them.
The stability of total phenolics content was evaluated during the heat treatment. In all cases, a high stability in the content of TP for all temperatures was observed: retention values between 82–89% for BB, 87–91% for BC and 75–90% for MB at the end of the thermal treatment. Figure 2 shows the evolution in TP for treatment at 90° C. These results agreed to Fischer et al. [16], who found that the total phenolic content was not markedly lowered upon heating pomegranate juices despite a significant decrease in anthocyanin content during heat treatment between 60 and 90°C.
Fig.2
Retention of Total Phenolics in various berries as a function of heating time at 90°C
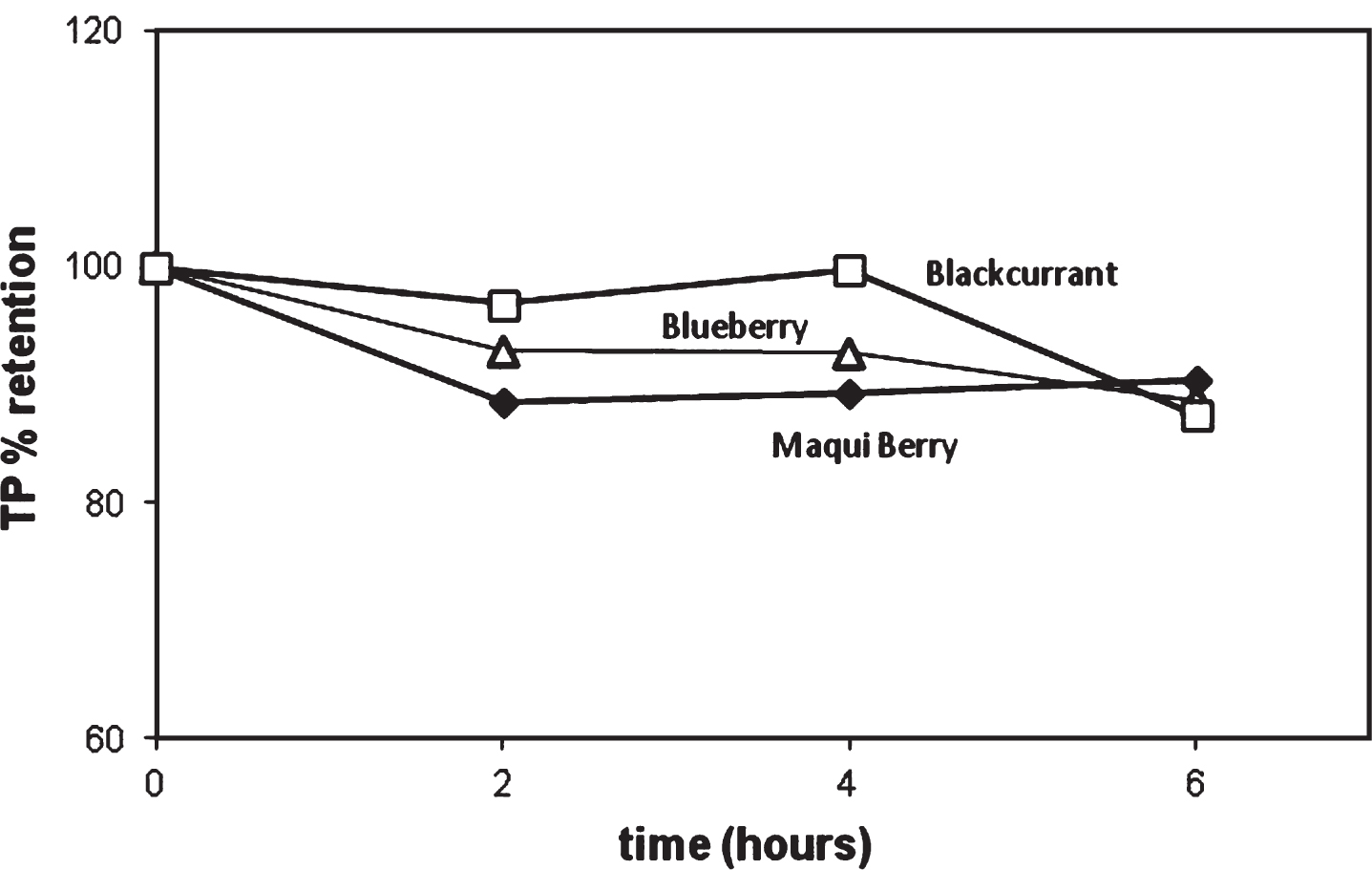
Table 3 shows kinetic parameters for degradation of colour parameter a*, following a first order model as for the degradation of monomeric anthocyanins.
Table 3
Kinetic parameters describing the loss of a* colour parameter in heated berry juices
| Berry | Temperature (°C) | k (h–1) | t1/2 (h) | Ea (kJ/mol) | Q10 (80–90°C) |
| Blackcurrant | 75 | 0.0065±0.0003 | 106.6±5.4 | 87.2±4 | 2.7±0.2 |
| 80 | 0.0131±0.0008 | 52.9±2.6 | |||
| 90 | 0.0352±0–0021 | 19.7±1.0 | |||
| Maqui berry | 75 | 0.0246±0.0015 | 20.2±1.2 | 84.9±5 | 2.0±0.1 |
| 80 | 0.0417±0.0021 | 16.6±1.0 | |||
| 90 | 0.0846±0.0005 | 8.2±0.4 | |||
| Blueberry | 70 | 0.0320±0.0016 | 21.7±1.1 | 89.5±4 | 2.2±0.1 |
| 80 | 0.0536±0.0037 | 12.9±0.8 | |||
| 90 | 0.1167±0.0058 | 5.9±0.4 |
Values are mean±standard error.
It can be seen that although the deterioration of MAcy and a* colour parameter have similar temperature dependence according to the obtained values of activation energies, colour degradation would occur slowly according to the values of k and t1/2 obtained. Table 4 shows a comparison between the kinetics of degradation of Macy and a* colour parameter. The ratio of activation energies for anthocyanins and colour deterioration was close to 1. In the case of half-life times, for maqui berry colour and anthocyanins kinetics of deterioration were similar (ratio 1.2), for blueberry t1/2 of colour deterioration was 4 times greater than anthocyanins losses at 90°C and for blackcurrant a much higher ratio was observed at all temperatures reaching values of colour deterioration 11 times lower than anthocyanins losses. This would indicate that for BC, while a deterioration of the monomeric anthocyanins is observed during the thermal process, the effect in colour is much lower and would take a long time to reach considerable deterioration values.
Table 4
Comparison of kinetic parameters between MA y and a* colour parameter
| Berry | Temperature (°C) | t1/2COLOUR/t1/2MAcy | EaCOLOUR/EaMAcy |
| Blackcurrant | 75 | 4.9 | 0.93 |
| 80 | 7.3 | ||
| 90 | 10.9 | ||
| Maqui berry | 75 | 1.2 | 1.04 |
| 80 | 1.2 | ||
| 90 | 1.2 | ||
| Blueberry | 70 | 1.7 | 0.98 |
| 80 | 1.8 | ||
| 90 | 4.2 |
This effect had already been observed in a previous work with commercial juices stored at 38°C, where black currant juice showed low colour deterioration after 180 days of storage [7].
As mentioned previously, in the case of colour parameter a*, the largest decrease was observed for BB and lower for BC. According to the values shown in Table 5, the percentage of red colour decrease was 60.7% for BB, 50.4% for MB and 24.3% for BC at 90°C.
Table 5
CIELab colour parameters of berries extracts before and after thermal treatment at 90°C (final time. 8 hours)
| Colour parameter | Fruit | |||
| Blueberry | Blackcurrant | Maqui Berry | ||
| L* | Initial | 31.56± 0.71b | 17.12±0.84b | 32.15±0.84b |
| Final | 44.10±0.82a | 25.52±1.31a | 38.10±0.09a | |
| a* | Initial | 38.24±0.51b | 48.23±0.83b | 29.33±0.45b |
| Final | 15.03±0.25a | 36.51±0.56a | 14.56±0.37a | |
| b* | Initial | –2.79±0.25b | 15.93±0.73b | 9.99±0.73b |
| Final | 5.18±0.33a | 6.03±0.39a | 16.28±0.78a | |
| h0 | Initial | –4.17±0.91b | 18.28±0.91b | 17.08±0.91b |
| Final | 19.01±0.03a | 9.48±0.30a | 48.25±0.29a | |
Values are means±SD (n = 3). For each colour parameter values in columns with the different superscript letters are significantly different (P < 0.05).
A good correlation was observed in the behaviour of the parameter MAcy and a* colour parameter in pulp extract during heating, showing an overall coefficient of 0.98 for blueberry and 0.97 for maqui berry. For black currant the overall coefficient was 0.87, this lower value of the correlation coefficient would also reflect the slow degradation of colour observed for this fruit even slower than observed for the other fruits in relation to the degradation of anthocyanins.
In general, it was observed an increase in L *, indicating an increase in brightness after heat treatment. Colour parameters b * and h0 showed an increase in the case of blueberry and maqui berry, indicating a shift to more browning tones. In the case of blackcurrant a decrease was observed.
Fischer et al. [17] observed similar colour changes after the thermal treatment of pomegranate juice. Extracts of juice showed a decrease of a* (52%) with an increase of b* (98%) and L* (4%) values, indicating a fading in colour which was ascribed to anthocyanin losses. In the case of blackcurrant, although a change in colour is perceived, the effect of browning would not be evident according to colour measurements.
Total colour difference (ΔE) values were 27.6±0.5, 17.5±0.4 and 17.1±0.3 for BB, BC and MB respectively, indicating more noticeable visual changes for blueberry juice.
The change in colour density (CD) and percentage of polymeric colour (% PC) during heating pulps were also studied. Figure 3 shows the evolution of colour indices CD and % PC at 90°C. Percent of PC, showed, in general, an increase for all fruits, at 90°C the increase was 46% to 94.4% for blueberry, 29.7% to 52.9% for blackcurrant and 35.9% to 60% for maqui berry, indicating that in part the observed colour changes are due to polymerization reactions between monomeric anthocyanins. The increase in % PC during thermal treatment has been reported in other anthocyanin-containing juices like grape or blueberry juice [26, 27].
Fig.3
Color density (CD: ■, ▴, ♦) and % polimeric colour (% PC: □, Δ, ♦) evolution for berry juices during heating at 90°C.
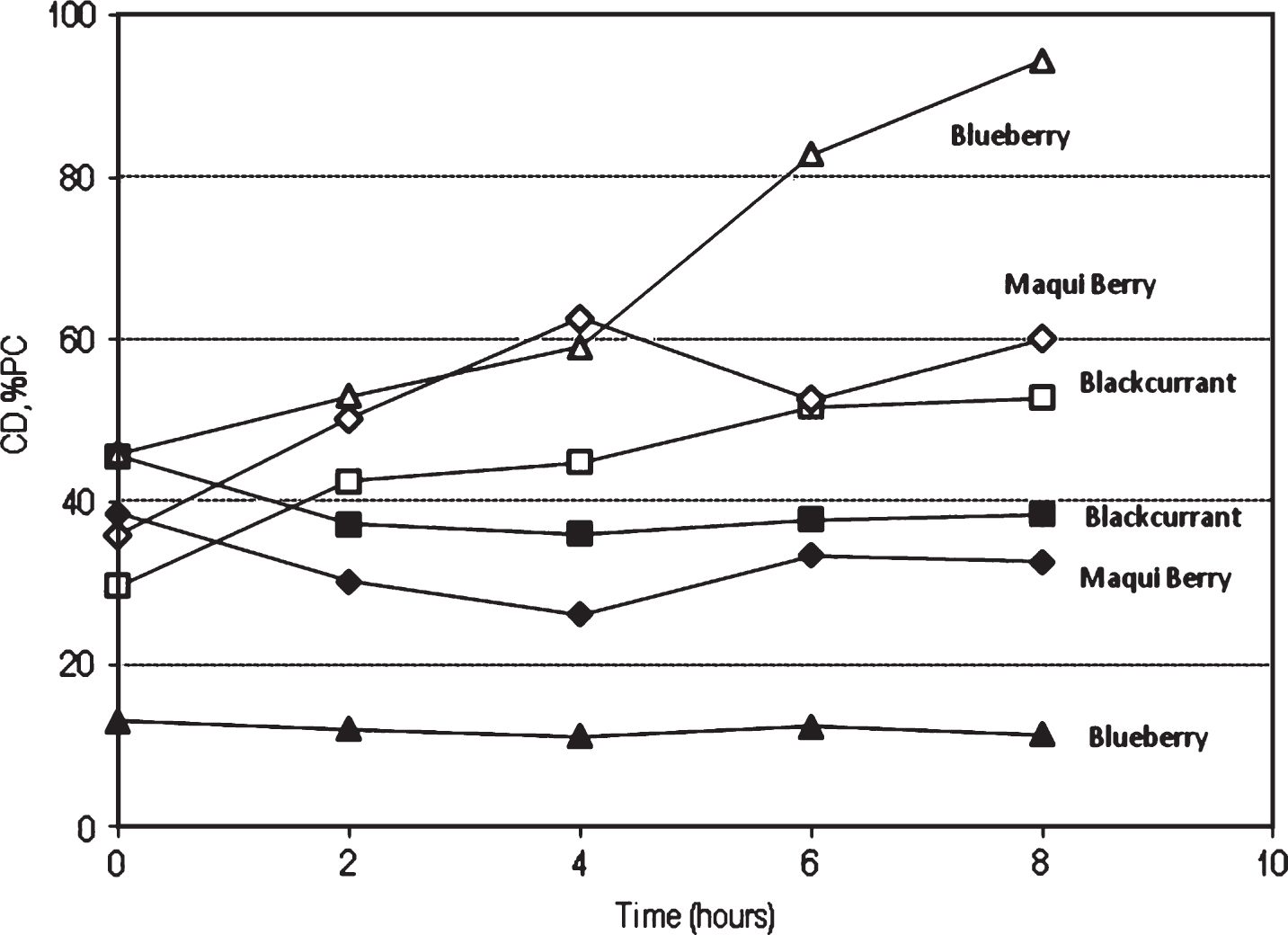
In general, results indicate that colour changes were much less evident than those observed for the monomeric anthocyanin content. Colour stabilisation phenomena may be produced by different reactions such as co-pigmentation of anthocyanins with other compounds (co-pigments), considered one of the main mechanisms of stabilisation of colour in plants [28–31]. In this phenomenon, the pigments and other colourless organic compounds or metallic ions form molecular or complex associations, generating a change in colour intensity. In the case of intramolecular copigmentation, the covalent acylation of the anthocyanin molecule stabilises the pigment, probably because the reactivity of the carbon C-2 and C-4 with nucleophilic reactants, e.g. water molecules, is hindered [8].
The increase in observation of the percentage of polymeric colour may indicate the formation of condensation reactions between anthocyanins and other polymeric compounds such as procyanidins, resulting in the formation of polymerised phenolic compounds that can be detected by the Folin method [13]. This phenomenon may explain the stability observed in the phenolic content.
The degree of colour stabilisation effects depends on the composition and anthocyanin profile of each fruit. Blackcurrant contains four major anthocyanins; the 3-glucosides and 3-rutinosides of cyanidin and delphinidin [8, 32–34]. Besides anthocyanins, BC contains hydroxycinnamic acids (p-coumaric, caffeic acid, ferulic acid) and hydroxybenzoic acids (gallic acid), flavonols (glycosides of myricetin, quercetin, kaempferol and isorhamnetin) and proanthocyanidins. The presence of these compounds has been associated with increased colour stability in blackcurrant, due to the formation of acylated anthocyanins [34–36].
Blueberry has a more complex anthocyanin profile, it was found the presence of 14 different anthocyanins being the most abundant delphinidin 3-glucoside, cyanidin 3-glucoside, petunidin 3-glucoside, malvidin 3-glucoside and cyanidin 3-arabinoside. It was also reported a low level of acylated anthocyanins or acylated pigments for this fruit [32, 35, 37]. Other compounds identified in BB were cinnamic acids and Flavonol –glycosides [37].
Maqui berry was characterised by the presence of glycosylated forms of delphinidin and cyanidin, with the important presence of polar poly glycosylated derivatives (3-glucosides, 3,5-diglucosides, 3-sambubiosides and 3- sambubioside-5-glucosides of delphinidin and cyanidin) [38]. It has been reported that anthocyanin molecules with complex patterns of glycosylation and acylation exhibit remarkable stability to heat treatments and light exposure [39]. This could explain the higher colour stability observed for blackcurrant and maqui berry, as reported in previous studies [8, 36]. Wrolstrad et al. [9] suggested that glycosidic substitution, and in particular, acylation of sugar residues with cinnamic acids, increase pigment stability. Furthermore, it has been observed that an increase in total anthocyanin concentration promotes higher colour stability [39]. This could be another reason for the increased colour stability observed for blackcurrant, followed by maqui berry and finally blueberry, since in the latter case is the fruit that has a more complex anthocyanin profile and lowest concentration of both anthocyanins and total phenolics. In the case of blackcurrant and maqui berry it has been reported a high concentration of major anthocyanins [32, 40].
The activation energies (Ea) values obtained for the thermal degradation of anthocyanins and colour indicate significant temperature dependence for these degradation processes. Other authors have reported Ea values in the same order for anthocyanin degradation in several fruit and vegetables: 71.9 kJ/mol for grapefruit [14], 74.8 kJ/mol for acerola pulp [41], 80.4 kJ/mol for blueberry [24], 92 kJ/mol for blackberry [42] and 94.4 kJ/mol for strawberry pure [43]. Q10 values have also been reported in the same range of temperatures, with similar values than those obtained in the present work: 2.3 between 70–80°C for black carrot [14], 2.95 between 60–70°C and 1,67 in the range 70–80°C for blueberry juice [24].
Selection of heating temperature has a significant effect on the final characteristics of the thermally treated product. Enzyme inactivation is one of the objectives of applying a heat treatment in such products based on fruits. It has been reported activity of peroxidases and phenolases in berries, such as phenol oxidases and polyphenol oxidases, which were found naturally in these fruits [8]. The processes of enzyme inactivation such as polyphenol oxidase (PPO) or peroxidase (PDO) have much higher temperature dependence with values of Ea between 214–295 kJ/mol [44–46]. Since activation energy for the degradation of anthocyanins and colour are much lower than those of enzymatic degradation, it should be selected a treatment at a higher temperature (e.g. 90°C), where further destruction of enzymes is achieved, and lower of quality factors as the content of bioactive compounds and colour.
4Conclusions
The results show that among the studied fruits, blackcurrant and maqui berry have the highest content of anthocyanins and total polyphenols. All fruits showed a degradation rate of colour much slower than the degradation of anthocyanins, especially blackcurrant showed the highest stability. This could be due to the relatively high amount of acylated anthocyanins and co pigments present in this fruit in comparison with the other fruits. This condition would be appreciated from the point of view of product acceptance by consumers.
The kinetic parameters calculated for the different fruits can be used to design a thermal treatment to obtain a high retention of attractive colour and bioactive compounds. While a significant dependence on temperature was observed according to values of Ea and Q10, it is less than other factors such as enzyme inactivation which is necessary to apply in fruit products.
The evolution of total phenolics content during heating indicate that the products obtained from these fruits and subjected to heat treatment between 70–90°C maintain the level of TP after treatment.
Acknowledgments
The authors would like to thank Frutas del Sur Andino S.A. (Río Negro, Argentina) for donating the commercial juices used in this study that was funded by CONICET (Project PIP -CONICET N 052).
References
[1] | Vulić JJ , Vračar LO , Šumić ZM . Chemical characteristics of cultivated elderberry fruit. Acta Periodica Technologica. (2008) ;39: :85–90. |
[2] | Pérez-Jiménez J , Neveu V , Vos F , Scalbert A . Identification of the 100 richest dietary sources of polyphenols: an application of the Phenol-Explorer database. European Journal of Clinical Nutrition. (2010) ;64: :S112–20. |
[3] | Granato D , Karnopp AR , van Ruth SM . Characterization and comparison of phenolic composition, antioxidant capacity and instrumental taste profile of juices from different botanical origins. Journal of the Science of Food and Agriculture. (2014) ;95: (10):1997–2006. |
[4] | Thorne Research. Sambucus nigra (Elderberry). Alternative Medicine Review. (2005) ;10: :51–5. |
[5] | Veberic R , Jakopic J , Stampar F , Schmitzer V . European elderberry (Sambucus nigra L.) rich in sugars, organic acids, anthocyanins and selected polyphenols. Food Chemistry. (2009) ;114: :511–5. |
[6] | Netzel M , Stintzing FC , Quaas D , Strass G , Carle R , Bitsch R , Bitsch I , Frank T . Renal excretion of antioxidative constituents from red beet in humans. Food Research International. (2005) ;38: :1051–8. |
[7] | Busso Casati C , Sánchez V , Baeza R , Magnani N , Evelson P , Zamora MC . Relationships between colour parameters, phenolic content and sensory changes of processed blueberry, elderberry and blackcurrant commercial juices. International Journal of Food Science and Technology. (2012) ;47: :1728–36. |
[8] | Rein MJ . Copigmentation reactions and color stability of berry anthocyanins (dissertation). EKT series 1331. University of Helsink. Department of Applied Chemistry and Microbiology. (2005) ;88 + 34 pp. |
[9] | Wrolstad R , Durst W , Lee J . Tracking color and pigment changes in anthocyanin products. Trends in Food Science & Technology. (2005) ;16: :423–8. |
[10] | Skupién K . Chemical composition of selected cultivars of highbush blueberry fruit (Vaccinium corymbosum L.). Folia Horticulturae. (2006) ;Ann. 18/2:47-56. |
[11] | Boranbayeva T , Karadeniz F , Yilmaz E . Effect of Storage on Anthocyanin Degradation in Black Mulberry Juice and Concentrates. Food and Bioprocess Technology. (2014) ;7: :1894–902. |
[12] | Ling B , Tang J , Kong F , Mitcham EJ , Wang S . Kinetics of food quality changes during thermal processing: A review. Food Bioprocess Technology. (2015) ;8: :343–58. |
[13] | Patras A , Brunton NP , O’Donnell C , Tiwari BK . Effect of thermal processing on anthocyanin stability in foods; mechanisms and kinetics of degradation. Review. Trends in Food Science & Technology. (2010) ;21: :3–11. |
[14] | Kirca A , Őzkan M , Cemeroğlu B . Stability of black carrot anthocyanins in various fruit juices and nectars. Food Chemistry. (2006) ;97: :598–605. |
[15] | Kirca A , Őzkan M , Cemeroğlu B . Effects of temperature solid content and pH on stability of black carrot anthocyanins. Food Chemistry. (2007) ;101: :212–8. |
[16] | Kara Ş , Erçelebi EA . Thermal degradation kinetics of anthocyanins and visual colour of Urmu mulberry (Morus nigra L.). Journal of Food Engineering. (2013) ;16: :541–7. |
[17] | Fischer UA , Carle R , Kammerer DR . Thermal stability of anthocyanins and colourless phenolics in pomegranate (Punica granatum L.) juices and model solutions. Food Chemistry. (2013) ;138: :1800–9. |
[18] | Waterhouse AL . Determination of total phenolics. In Wrolstad RE. editor. Current Protocols in Food Analytical Chemistry. New York: Wiley & Sons; (2001) . pp I1.1.1–I1.1.8. |
[19] | Giusti MM , Wrolstad RE . Characterization and measurement of anthocyanins by UV-visible spectroscopy. In: Wrolstad R , Acree T , An H , Decker E , Penner M , Reis D , Schawrtz S , Shoemaker C & Spoms P , editors. Current Protocols in Food Analytical Chemistry (1st edn). New York: John Wiley and Sons, Inc; (2001) . pp F1.2.1–F1.2.13. |
[20] | Maskan M . Production of pomegranate (Punica granatum L. ) juice concentrate by various heating methods: Colour degradation and kinetics. Journal of Food Engineering. (2006) ;72: :218–24. |
[21] | Yang Z , Han Y , Gu Z , Fan GZ . Thermal degradation kinetics of aqueous anthocyanins and visual colour of purple corn (Zea mays L.) cob. Innovative Food Science & Emerging Technologies. (2008) ;9: :341–7. |
[22] | Özşen D , Erge HS . Degradation kinetics of bioactive compounds and change in the antioxidant activity of wild strawberry (Fragaria vesca) pulp during heating. Food and Bioprocess Technology. (2013) ;9: :2261–7. |
[23] | Busso Casati C , Baeza R , Sanchez V , Catalano A , Lopez P , Zamora MC . Thermal degradation kinetics of monomeric anthocyanins, colour changes and storage effect in elderberry juices. Journal of Berry Research. (2015) ;5: (1):29–39. |
[24] | Pereira Kechinski C , Ramos Guimaraes V , Zapata Noreña C , Tessaro I , Ferreira Marczak L . Degradation kinetics of anthocyanin in blueberry juice during thermal treatment. Journal of Food Science. (2010) ;75: (2):173–6. |
[25] | Harbourne N , Jacquier JC , Morgan DJ , Lyng JG . Determination of the degradation kinetics of anthocyanins in a model juice system using isothermal and non-isothermal methods. Food Chemistry. (2008) ;111: :204–8. |
[26] | Danişman G , Arslan E , Toklucu AK . Kinetic Analysis of anthocyanin degradation and polymeric colour formation in grape juice during heating. Czech J Food Science. (2015) ;33: (2):103–8. |
[27] | Brownmiller C , Howard LR , Prior RL . Processing and storage effects on monomeric anthocyanins, percent polymeric colour and antioxidant capacity of processed blueberry products. Journal of Food Science. (2008) ;73: :H72–79. |
[28] | Davies AJ , Mazza G . Copigmentation of simple and acylated anthocyanins with colorless phenolic compounds. Journal of Agricultural and Food Chemistry. (1993) ;41: :716–20. |
[29] | Mazza G , Brouillard R . Recent developments in the stabilization of anthocyanins in food products. Food Chemistry. (1987) ;25: :207–25. |
[30] | Mazza G , Brouillard R . The mechanism of copigmentation of anthocyanins in aqueous solutions. Phytochemistry. (1990) ;29: :1097–102. |
[31] | Bakowska A , Kucharska AZ , Oszmianski J . The effects of heating, UV irradiation, and storage on stability of the anthocyanin-polyphenol copigment complex. Food Chemistry. (2003) ;81: :349–55. |
[32] | Jacobek L , Seruga M , Medvidović-Kosanović M , Novak I . Anthocyanin content and antioxidant activity of various red fruit juices. Deutsche Lebensmittel-Rundschau. (2007) ;103: :58–64. |
[33] | Laaksonen O , Sandell M , Nordlund E , Heiniö R-L , Malinen H-L , Jaakkola M , Kallio H . The effect of enzymatic treatment on blackcurrant (Ribes nigrum) juice flavour and its stability. Food Chemistry. (2012) ;130: :31–41. |
[34] | Mattila P , Hellström J , McDougall G , Dobson G , Pihlava J-M , Tiirikka T , Stewart D , Karjalainen R . Polyphenol and vitamin C contents in European commercial blackcurrant juice products. Food Chemistry. (2011) ;127: :1216–23. |
[35] | Obón JM , Díaz García MC , Castellar MR . Red fruit juice quality and authenticity control by HPLC. Journal of Food Composition and Analysis. (2011) ;24: :760–71. |
[36] | Wrolstad RE . Interaction of natural colors with other ingredients. Anthocyanin pigments- bioactivity and coloring properties. Journal of Food Science. (2004) ;69: (5):C419–25. |
[37] | Lee J , Durst RW , Wrolstad RE . Impact of juice processing on blueberry anthocyanins and polyphenolics: Comparison of two pretreatments. Journal of Food Science. (2002) ;67: (5):1660–7. |
[38] | Rojo L , Ribnicky D , Logendra S , Poulev A , Rojas-Silva P , Kuhn P , Dorn R , Grace M , Lila M , Raskin I . In vitro and in vivo anti-diabetic effects of anthocyanins from Maqui Berry (Aristotelia Chilensis). Food Chemistry. (2012) ;131: :387–96. |
[39] | Giusti M , Wrolstad R . Acylated anthocyanins from edible sources and their applications in food systems. Biochemical Engineering Journal. (2003) ;14: :217–25. |
[40] | Escribano-Bailón MT , Alcalde-Eon C , Muñoz O , Rivas-Gonzalo J , Santos-Buelga C . Anthocyanins in Berries of Maqui [Aristotelia chilensis (Mol.) Stuntz]. Phytochem Anal. (2006) ;17: (1):8–14. |
[41] | Mercali GD , Pez Jaeschke D , Tessaro IC . Degradation kinetics of anthocyanins in acerola pulp: Comparison between ohmic and conventional heat treatment. Food Chemistry. (2013) ;136: :853–7. |
[42] | Jimenez Oey I , van der Plancken I , Hendrickx M , van Loey A . Kinetic study on the thermal and pressure degradation of anthocyanins. Food Chemistry. (2010) ;123: :269–74. |
[43] | Verbeyst L , Oey I , van der Plancken I , Hendrickx M , van Loey A . Kinetic study on the thermal and pressure degradation of anthocyanins. Food Chemistry. (2010) ;123: :269–74. |
[44] | Fortea MI , Lopez-Miranda S , Serrano-Martinez A , Carreño J , Núñez-Delicado E . Kinetic characterization and thermal inactivation study of polyphenol oxidase from table grape (Crimson seedless). Food Chemistry. (2009) ;113: :1008–14. |
[45] | Terefe N , Yang YH , Knoerzer K , Buckow R , Versteeg C . High pressure and thermal inactivation kinetics of polyphenol oxidase and peroxidase in strawberry puree. Innovative Food Science and Emerging Technologies. (2010) ;11: :2–60. |
[46] | Cheng XF , Zhang M , Adhikari B . The inactivation kinetics of polyphenol oxidase in mushroom (Agaricus bisporus) during thermal and thermosonic treatments. Ultrasonics sonochemistry. (2013) ;20: :674–67. |




