A Retrospective Study on Clinical Assessment of Cognitive Impairment in a Swedish Cohort: Is There Inequality Between Natives and Foreign-Born?
Abstract
Background:
People with a migration background are underrepresented in dementia research and disfavored in assessment and treatment, and many foreign-born individuals with dementia remain undiagnosed.
Objective:
The aim of this study was to examine whether there is inequality in the clinical assessment of dementia between native and foreign-born individuals in Sweden.
Methods:
Information was gathered retrospectively from a cohort of 91 native and 36 foreign-born patients attending four memory clinics in Skåne, Sweden. Data included information on cognitive test results, cerebrospinal fluid biomarkers, scores at structural imaging scales of global cortical atrophy (GCA), medial temporal lobe atrophy (MTA) and the Fazekas scale, laboratory measures of thyroid-stimulating hormone, calcium, albumin, homocysteine, hemoglobin, cobalamin (vitamin B12), and folate (vitamin B9), contact with health care, and treatment.
Results:
Foreign-born patients had lower educational level and scored lower on Mini-Mental State Examination and Clock Drawing Test (p < 0.001–0.011). Relatives initiated contact with health care to a higher extent in the foreign-born group (p = 0.031). Foreign-born patients had less white matter lesions (p = 0.018). Additionally, Alzheimer’s disease (AD) biomarkers were significantly less used in foreign-born patients to support an AD diagnosis (p = 0.001). No significant differences were found for scores on GCA and MTA, laboratory measures, or initiated treatment.
Conclusion:
Although native and foreign-born patients were predominantly homogenous regarding examined variables, differences in the diagnostic process and underlying biological correlates of dementia exist and need to be further investigated in a larger sample.
INTRODUCTION
Dementia is a common global problem: more than 55 million people are living with dementia worldwide, and it is predicted that by the year 2030, that number will be around 75 million individuals [1, 2]. The extent of epidemiological studies concerning dementia is higher in developed countries compared to developing countries [3]. Less than 10% of all population-based dementia studies are carried out in low- and middle-income countries, despite the fact that nearly 70% of people with dementia live here [4]. There is a general lack of ethnic and geographic diversity in dementia research, where a recent systematic review found that 89% of the included studies originated from North America or Europe [5]. Moreover, only 22% of the analyzed studies reported on ethnicity, and even in these studies 89% of participants were white [5].
Growing evidence shows that people in minority ethnic (ME) groups are disfavored in the process of assessment and treatment of dementia [6–8]. This group of people gets access to health care later in the course of the disease, and when they eventually get a dementia diagnosis they are less likely than the native population to get appropriate medication [6]. Barriers for help seeking in ME groups are key factors in explaining why these groups show up later to caregivers [7]. The barriers include linguistic difficulties, lack of understanding of what dementia is, lack of understanding of how and where to seek help for dementia, and experienced discrimination and dismissal of symptoms [7]. Furthermore, one study found that 64% of European dementia centers thought it was more complicated to assess people of ME groups than natives, and that it was thought to be significantly more challenging in countries with a more recent history of immigration [8]. Reported difficulties in the clinical evaluation of people of ME groups included limited linguistic skills, educational level, religious issues, and a lack of cognitive tests and rating scales suitable for the patients [8]. There also appears to be a difference in the use of blood tests, structural imaging, and lumbar puncture between ME groups and natives; for example it was reported that only 8% of ethnic minority patients always got a lumbar puncture, partially due to problems in gaining consent for these tests [8]. Furthermore, former studies have suggested a potential difference in cerebrospinal fluid (CSF) biomarkers and structural imaging based on ethnicity [9, 10].
The inequality in research, assessment, and treatment of dementia between ethnic groups poses a contemporary problem. There are among 272 million (as of 2019) international migrants worldwide, where 12% are age 65 and over [11]. It is estimated that there are around 476,500 dementia cases in the foreign-born population in Europe, which constitutes around 6.5% of the total number of dementia cases in Europe [12]. Findings suggest that many migrants with dementia remain undiagnosed: in Italy, which is one of the most immigrant-dense countries in Europe, only 3.1% of outpatients with cognitive difficulties who attended a memory clinic or neurology clinic were migrants [13].
The aim of this paper is to examine whether there is inequality in the process of dementia diagnosis between native and foreign-born individuals. This includes analysis of the health care process, clinical assessment, and treatment options, and will cover time and quality of investigation, clinical presentation and laboratory/imaging findings, level of laboratory abnormalities linked to risk of dementia [14–22], and time to appropriate treatment.
MATERIALS AND METHODS
Study population
This was a retrospective cohort study, where the cohort was originally used to validate cognitive tests prospectively [23]. In the original study, information about the project was sent to all patients, both native and foreign-born, about to undergo cognitive investigation at four memory clinics in Skåne, Sweden: Ängelholm, Hässleholm, Malmö, and Ystad, between October 2018 and October 2019. The ones who agreed to participate were included in the study, resulting in a cohort consisting of 127 patients [23]. A total of 91 (72%) were native Swedish patients, and 36 (28%) were foreign-born patients. As a rule, foreign-born people were offered an interpreter so that they could be examined in their first language, but in a total of 12 cases an interpreter was considered unnecessary because of adequate Swedish fluency or a present relative that could help interpret if needed. The foreign-born group was also categorized into high (n = 12) versus low/middle (n = 21) income of their country of origin, according to World Bank Group’s country classifications by income level [24]. For some foreign-born patients (n = 3), information on income level could not be gathered as their country of origin no longer exists as the same country and is therefore not included on the World Bank Group’s lists.
Data collection
The data collection in this study was done by using a code key to get access to the deidentified participants’ medical records in Melior, a Swedish journal software, through a specific researcher user account. Already available information included the participants’ age, gender, years of education, and first language/origin (native Swedish or foreign-born), and scores on the cognitive tests Mini-Mental State Examination (MMSE), Clock drawing test (CDT), scores on the questionnaires Geriatric Depression Scale (GDS) and Functional Activities Questionnaire (FAQ). In the context of this study, the new variables collected included date of their first appointment/diagnosis at the memory clinics, question on the referral, and whether it was the patients themselves, their relatives or health care personnel who noticed symptoms of cognitive impairment and therefore initiated contact with health care. Information on CSF biomarkers, structural imaging, laboratory measures, and treatment were also gathered, see below.
Methods
CSF biomarkers
When performed, lumbar puncture was done at the memory clinic followed by measurement of Alzheimer’s disease (AD) CSF biomarkers amyloid-β 42 (Aβ42), the ratio Aβ42/40, total tau protein (T-tau), and phosphorylated tau protein 181 (P-tau 181). CSF biomarkers were measured at the Neurochemistry laboratory at Mölndal Hospital, University of Gothenburg, according to established protocols [25]. Aβ42/40 was chosen over Aβ42 to define pathological Aβ status. Based on biomarker levels, patients were further classified according to the A/T/N classification, where A + represents pathologic Aβ42/40 ratio, T + pathological P-tau 181, and N + pathological T-tau [26]. A + patients having T + or N + or both were considered as fulfilling the IWG-2 criteria for pathological AD biomarker status [27].
Structural imaging
For those patients who underwent computed tomography (CT) or magnetic resonance imaging (MRI) of the brain, information on global cortical atrophy (GCA), medial temporal lobe atrophy (MTA), and Fazekas scale (quantification of white matter lesions) scores were retrieved from the specialist’s report. These scales are routinely used in structural imaging assessment of dementia [28]. MTA ranges from 0–4 (0 = normal, 1 = normal, 2 = increased width of the choroid fissure and temporal horn, mild loss of hippocampal height, 3 = severely increased width of the choroid fissure, moderate widening of the temporal horn and moderate loss of hippocampal height, 4 = severely increased width of the choroid fissure and temporal horn, marked atrophy of the hippocampus), GCA ranges from 0–3 (0 = normal, 1 = slight widening of sulci, 2 = gyral volume loss, 3 = marked widening of sulci with severe gyral volume loss), and Fazekas scale ranges from 0–3 (0 = normal, 1 = multiple punctate white matter lesions, 2 = incipient confluence of punctate white matter lesions, 3 = confluent white matter lesions) [28].
Laboratory measures
Information on pathological findings in laboratory measures linked to potential reversible causes of cognitive symptoms or to higher risk of dementia was retrieved, namely thyroid-stimulating hormone (TSH), calcium, albumin, homocysteine, hemoglobin, cobalamin (vitamin B12), and folate (vitamin B9) [14–22]. A level of laboratory abnormalities was calculated by dichotomizing each variable based on whether the laboratory values were normal or pathological. Hereafter, the number of pathological values were added for each patient, resulting in a “level of laboratory abnormalities”.
Treatment and former health care contact
Information was collected about whether the patients had ever been in contact with a psychiatric clinic, and what medicines they were currently taking at the first appointment, whether they got any new medicines from the memory clinic and if so which ones. Only neurological and psychiatric medicines were included: different antidepressants and mood stabilizers, benzodiazepines, antipsychotic medicines, antiepileptic medicines, other sedatives and anxiolytics, treatment for Parkinson’s disease, central nervous system stimulants, acetylcholinesterase inhibitors (AChEIs) and memantine.
Statistical analysis
Statistical analysis was done in IBM SPSS Statistics version 26. Gauss distribution was assessed by examining the Skewness-Kurtosis normality test. T-test or Mann-Whitney U test were used alternatively based on the skewness of the variables. T-test was also sometimes used on ordinal variables, for example MMSE score and scores of imaging findings, to increase the statistical power of the comparison [29], along with non-parametric tests. Pearson Chi-square was used to determine the significance of differences on categorical variables.
To examine correlation between two categorical variables, Spearman’s r (rs) was calculated. Binary logistic regression was used to calculate associations between ethnic background and dichotomous variables.
Ethics
The study was approved by the regional Ethics Review Board in Lund, Sweden, and by Samrådsgrupp KVB (consultation group for quality registers and health care databases).
RESULTS
Characteristics of the study population
Characteristics of the 127 study participants are presented in Table 1. There were no statistically significant differences in age and gender between the native and foreign-born group (Table 1). The native individuals had a significantly higher number of years of education (mean 11.2) than foreign-born individuals (mean 9.2, p = 0.013, 95% CI 0.446 –3.580) (Table 1). On average, foreign-born scored lower on MMSE and CDT (p < 0.001 and p = 0.011, respectively). The difference at MMSE was still significant when adjusting for years of education (p < 0.001), but not the difference at CDT (p > 0.05). Both MMSE and CDT scores correlated positively with years of education (rs = 0.461 and 0.455, p < 0.001).
Table 1
Cohort characteristics
| Characteristic | Native | Foreign-born | p |
| Number of patients | 91 | 36 | |
| Age, mean y (SD) | 70.5 (12.0) | 70.8 (12.9) | 0.907 |
| Gender, male/female (% male) | 45/46 (49%) | 13/23 (36%) | 0.174 |
| Education, mean y (SD) | 11.2 (3.6) | 9.2 (4.1) | 0.013 |
| MMSE, mean (SD) | 24.6 (5.2) | 18.6 (5.7) | <0.001 |
| CDT, mean (SD) | 3.1 (1.2) | 2.5 (1.3) | 0.011 |
Demographics of the 127 participants. T-test was used for continuous variables and Pearson Chi-square test for categorical variables. p-value < 0.05 was considered statistically significant. MMSE, Mini-Mental State Examination; CDT, Clock Drawing Test.
The foreign-born patients’ country of origin and years spent in Sweden before the first appointment at the memory clinic is presented in Supplementary Table 1. The 36 foreign-born patients originated from a total of 20 different countries, where 5 patients originated from neighboring Scandinavian countries, 15 from other European countries, 7 from Middle Eastern countries, 3 from Asian countries, 4 from South American countries, and 2 from African countries (Supplementary Table 1 and Supplementary Figure 1). The spread of years spent in Sweden were 1–55 years, with a median of 27 years (Supplementary Table 1).
Performed investigations and clinical presentation
The number of patients who got a dementia diagnosis in each group and the average time to diagnoses are also presented. No statistically significant disparities were found. The most common reason a patient did not undergo a lumbar puncture (Table 2) was a low suspicion of dementia based on anamnesis, cognitive testing and structural imaging (n = 21), and the next most common reason was that the patients themselves refused to undergo it (n = 11). Other reasons were a high suspicion of dementia based on anamnesis, cognitive testing, and structural imaging (n = 5), so that a lumbar puncture was not necessary to diagnose the patient, structural abnormalities of the spine such as scoliosis or arthrodesis of the spine (n = 2), and a risk of bleeding due to medication with anticoagulants (n = 1). A pie chart of diagnosis for the patients not undergoing lumbar puncture is presented in the Supplementary Figure 2. Analysis on income status of the country of origin, and number of educational years for the patients undergoing or not undergoing lumbar puncture showed no statistically significant differences. The reason two native patients did not get any structural imaging (Table 2) was repeated non-attendance, and a former diagnosis of Parkinson’s disease where structural imaging was done about 4 years prior and a new CT scan was thought to be unnecessary, respectively.
Table 2
Content and outcome of investigation
| Native n = 91 | Foreign-born n = 36 | p | |
| Lumbar puncture, n (%) | 54 (59%) | 26 (72%) | 0.175 |
| Structural imaging, n (%) | 89 (98%) | 36 (100%) | 0.370 |
| Dementia diagnosis, n (%) | 42 (46%) | 21 (58%) | 0.259 |
| Mean time to diagnosis, days (SD) | 159 (92) | 176 (53) | 0.578 |
T-test was used for continuous variables and Pearson Chi-square test for categorical variables. p-value < 0.05 was considered statistically significant. In the dementia diagnosis category, n = 89 for native individuals. Time to diagnosis refers to the number of days from the first appointment at a memory clinic to the eventual diagnosis, and in this category n = 68 for native individuals and n = 10 for foreign-born individuals.
There were no statistical differences in content and outcome of investigation when adjusted for the subgroups of the foreign-born patients’ origin (p > 0.05, Supplementary Table 2).
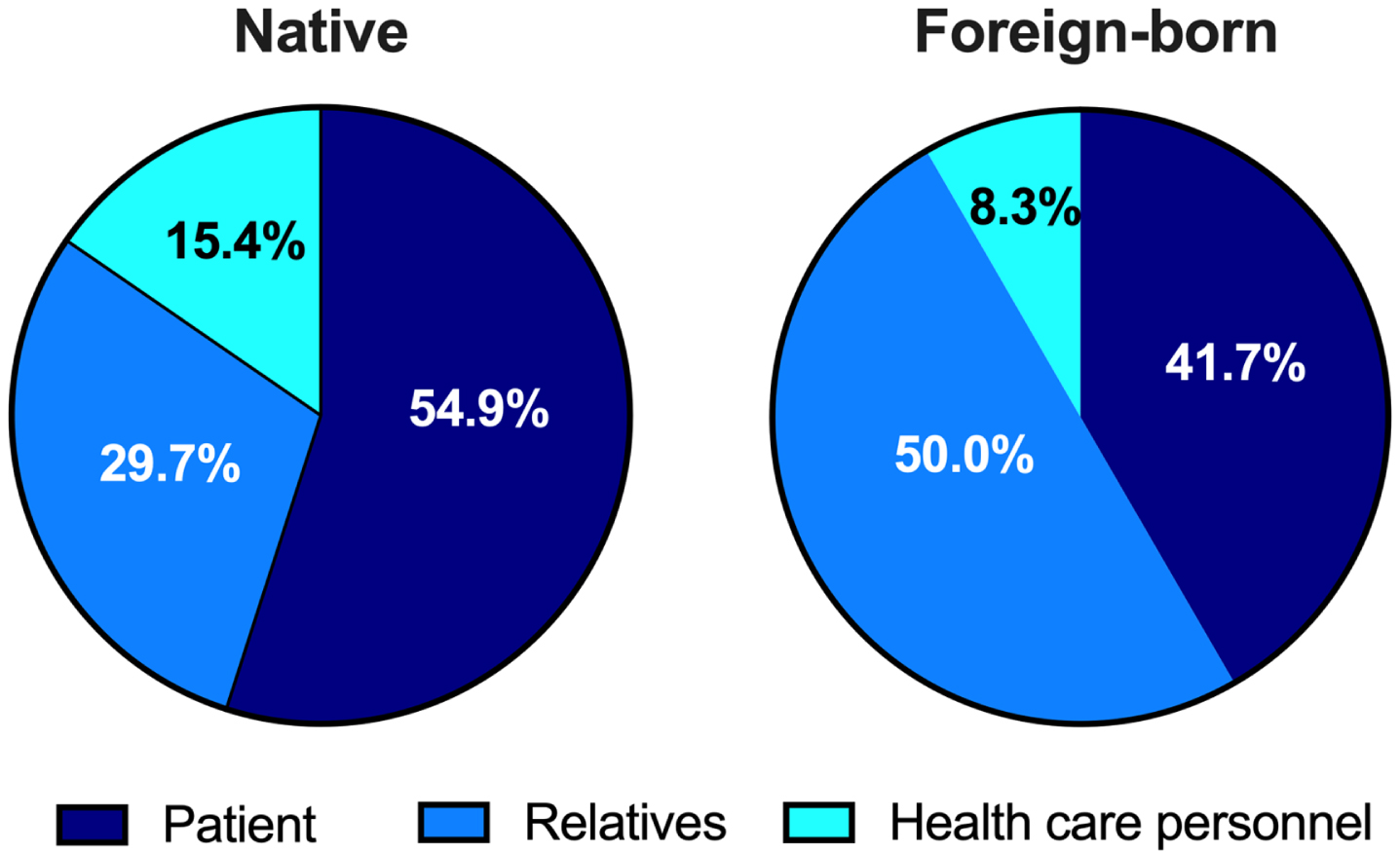
Contact with health care was initiated by relatives for foreign-born individuals to a greater extent compared to native individuals (p = 0.031, Table 3, Fig. 1). Furthermore, a higher share of foreign-born patients was taking antipsychotic and antiepileptic medicines at their first appointment at a memory clinic than native patients (p = 0.005 and p = 0.003, respectively) (Table 3). No significant differences were found regarding former contact with a psychiatric clinic, or whether the patients were on antidepressants and mood stabilizers, benzodiazepines, other sedatives and anxiolytic medicine, treatment for Parkinson’s disease, central nervous system stimulants, or treatment for dementia at baseline (p > 0.05, Table 3).
Table 3
Type of established contact with healthcare and current medication at baseline
| Native n = 91 | Foreign-born n = 36 | p | |
| Patient initiated contact, n(%) | 50 (55%) | 15 (42%) | 0.177 |
| Relatives initiated contact, n(%) | 27 (30%) | 18 (50%) | 0.031 |
| Health care personnel initiated contact, n(%) | 14 (15%) | 3 (8%) | 0.293 |
| Former contact with psychiatric clinic, n(%) | 19 (21%) | 7 (19%) | 0.857 |
| Already on neurological or psychiatric medicines at first appointment, n(%) | 38 (42%) | 10 (28%) | 0.453 |
| Antidepressants and mood stabilizers | 25 (27%) | 4 (11%) | 0.236 |
| Antipsychotics | 1 (1%) | 3 (8%) | 0.005 |
| Benzodiazepines | 7 (8%) | 3 (8%) | 0.326 |
| Antiepileptics | 1 (1%) | 3 (8%) | 0.003 |
| Other sedatives and anxiolytics | 17 (19%) | 4 (11%) | 0.987 |
| Treatment for Parkinson’s disease | 4 (4%) | 0 | 0.309 |
| Central nervous system stimulants | 1 (1%) | 0 | 0.623 |
| Treatment for dementia | 2 (2%) | 0 | 0.482 |
Pearson Chi-square test was used, p-value<0.05 was considered statistically significant. Regarding treatment, n = 88 in the native group and n = 19 in the foreign-born group.
Fig. 1
Pie charts showing the distribution concerning who, of three subgroups, initiated contact with health care concerning cognitive problems in natives and foreign-born respectively.
There were no statistical differences in type of established contact with health care and current medication at baseline when adjusted for the subgroups of the foreign-born patients’ origin, (p > 0.05, Supplementary Table 3).
Investigation findings and comorbidity
CSF biomarkers
There was no significant difference in the proportion of patients receiving an AD diagnosis between the native and foreign-born group (Table 4). Thirty-one out of 36 patients diagnosed with AD in the native group had a pathological AD biomarker status (defined with A/T/N classification) according to the diagnostic IWG-2 criteria (A + /T+/N+or A + in combination with T + or N+) [27]. In the foreign-born group, the majority of patients receiving an AD diagnosis had either not undergone a lumbar puncture (n = 4) or did not fulfill the IWG-2 criteria for pathological AD biomarker status (n = 4) (Table 4) [27]. Foreign-born received their AD diagnosis based on clinical findings rather than pathological biomarker status to a significantly higher extent (p = 0.001). All of the patients clinically diagnosed with AD in the respective groups subsequently received dementia treatment (Table 4). Three native patients and three foreign-born patients who had a pathological biomarker did not receive an AD diagnosis (Table 4). Instead, the native patients received the diagnoses mild cognitive impairment (n = 2) and localized brain atrophy (n = 1), and the foreign-born the diagnoses mild cognitive impairment (n = 1), vascular dementia (n = 1) and unspecified dementia (n = 1). A total of two native patients and one foreign-born with pathological AD biomarker status did not receive dementia treatment (Table 4).
Table 4
Biomarker findings and assigned diagnosis and treatment
| Native n = 91 | Foreign-born n = 36 | p | |
| AD diagnosis, n (%) | 36 (40%) | 10 (28%) | 0.213 |
| A + /T+/N+ | 12 | 0 | |
| A + and either T + or N+ | 19 | 2 | |
| A + /T-/N- | 3 | 3 | |
| A-/T+/N+ | 0 | 1 | |
| No lumbar puncture | 2 | 4 | |
| Symptomatic treatment after AD diagnosis, n (%) | 36 (100%) | 10 (100%) | – |
| Pathological biomarker profile without AD diagnosis, n (%) | 3 (3%) | 3 (8%) | 0.828 |
| A + /T+/N+ | 1 | 0 | |
| A + and either T + or N+ | 2 | 3 | |
| Symptomatic treatment after pathological biomarker profile without AD diagnosis, n (%) | 1 (33%) | 2 (67%) | 0.414 |
Pearson Chi-square test was used, p-value < 0.05 was considered statistically significant. Symptomatic treatment includes both Acetylcholinesterase inhibitors (AChEIs) and Memantine. AD = Alzheimer’s disease.
Structural imaging
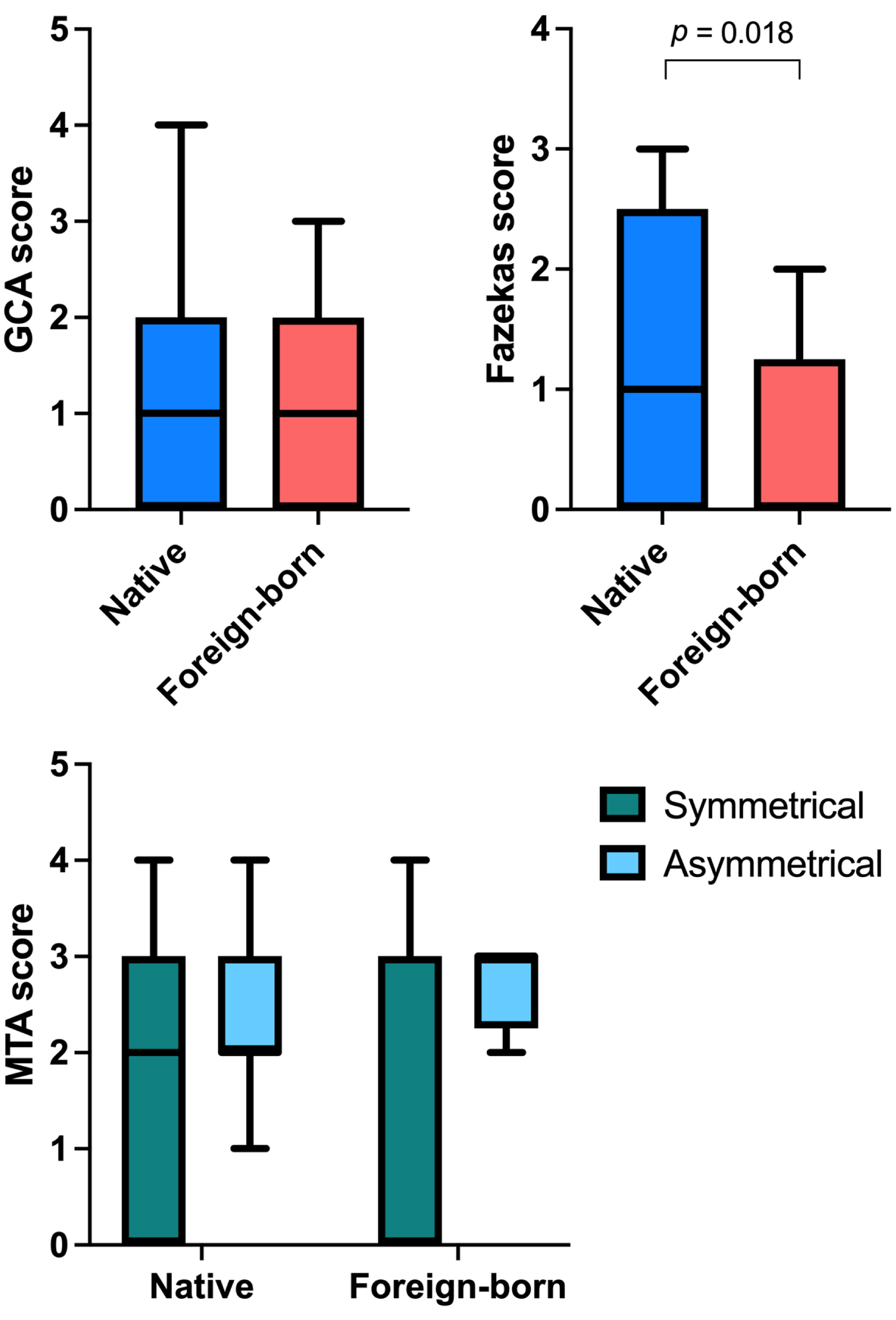
Foreign-born patients had a significantly lower Fazekas score compared to native patients (p = 0.018) (Fig. 2). No significant differences were observed for GCA or MTA. Mann-Whitney U test showed similar p-values as the ones from T-test (p = 0.022 for Fazekas score, other comparisons not significant). Furthermore, there were significant correlations between MMSE score and GCA score (rs=–0.298, p = 0.021) and MMSE score and MTA score (rs=–0.396, p < 0.001) in the native group, and in the foreign-born group there was a significant correlation between MMSE score and MTA score (rs=–0.458, p = 0.011). No significant correlation between MMSE score and Fazekas score was found for either of the groups (p > 0.05).
Fig. 2
Box plots showing global central atrophy (GCA) scores, Fazekas scores, and symmetrical and asymmetrical medial temporal lobe atrophy (MTA) scores on structural imaging for native and foreign-born patients. T-test was used, p-value<0.05 was considered statistically significant.
Laboratory measures
No significant differences were observed between the groups regarding level of laboratory abnormalities (p > 0.05, SupplementaryTable 4), or its correlation with findings on cognitive tests and structural imaging (p > 0.05, Supplementary Table 5). Abnormal levels of TSH, calcium, homocysteine, or hemoglobin were not associated to country of origin (p > 0.05, Supplementary Table 6). Albumin could not be examined as no foreign-born patient had pathological albumin, which resulted in a perfect separation on the logistic regression. As no patient had pathological concentrations of cobalamin or folate, these variables were not included.
Treatment
No statistically significant differences between the groups were found concerning AChEIs and memantine (p > 0.05, Table 5), antidepressants and mood stabilizers, antipsychotics, benzodiazepines, antiepileptics, other sedatives and anxiolytics, or treatment for Parkinson’s disease (p > 0.05, Supplementary Table 7). There were no significant differences in time from first appointment to treatment with AChEIs or Memantine (p > 0.05, Table 5). The most common treatment combination were AChEIs+Memantin for both native and foreign-born people, and there were no statistically significant differences in treatment combinations (p > 0.05, Supplementary Table 8).
Table 5
Initiated treatment for dementia
| Native n = 91 | Foreign-born n = 36 | p | |
| Treatment for dementia, n(%) | 45 (49%) | 15 (42%) | 0.428 |
| Treatment with AChEIs | 39 (43%) | 10 (28%) | 0.083 |
| Treatment with Memantine | 33 (36%) | 13 (36%) | 0.290 |
| Time from first appointment to treatment with AChEIs, days Mean (min-max) | 167 (0–670) | 273 (0–769) | 0.120 |
| Time from first appointment to treatment with Memantine, days Mean (min-max) | 293 (0–912) | 376 (126–796) | 0.349 |
Pearson Chi-square test was used for categorical variables, T-test was used for continuous variables. p-value < 0.05 was considered statistically significant. AChEIs, acetylcholinesterase inhibitors.
A significantly longer time from first appointment to treatment with AChEIs was found for patients originating from European countries outside Scandinavia (p = 0.003, Supplementary Table 9). Other than this, there were no statistically significant differences in initiated treatment for dementia when adjusted for the subgroups of the foreign-born patients’ origin (p > 0.05, Supplementary Table 9).
DISCUSSION
This retrospective cohort study aimed to examine whether there are inequalities in the investigation of cognitive impairment based on ethnic origin in a Swedish cohort of 127 patients, of which 91 were native and 36 foreign-born. The main focus of comparison were investigation findings of cognitive tests, lumbar puncture and structural imaging, contact with health care, comorbidity, and treatment.
Overall, we observed no substantial differences between native and foreign-born patients regarding access to health care, examinations provided and initiated therapy. This deviates from former studies indicating multiple differences in the dementia care process based on origin [6–8, 13, 30]. Although reassuring in terms of equality in the clinical assessment, the results should be taken with caution since they only reflect assessment of dementia in southern Sweden.
Although the groups were mostly homogeneous, some differences between groups were present and are worth being investigated. We found that foreign-born patients scored lower at MMSE and CDT, but, surprisingly, adjusting for years of education affected only the difference in CDT scores and not in MMSE scores. Education is one of the multiple confounders, together with language, culture and others, that have been shown to affect the MMSE and CDT scores [31–38]. For these reasons, researchers agree that more targeted cognitive tests are needed, and these tests should only be used as a first screening method and not for diagnosis, especially in ethnically diverse populations [32, 35, 36, 38]. We chose therefore not to focus on cognitive tests as primary outcomes and instead rely on objective laboratory and imaging biomarkers. Future studies should focus on determining the quality of examining these patients with a multicultural cognitive test battery instead of MMSE and CDT [39].
When looking at biomarkers, we found that, although the proportion of AD subjects in both groups was similar, most foreign-born patients were diagnosed with AD based on clinical symptoms, compared to natives where the biomarker profile also matched the clinical diagnosis (Table 4). This is not entirely surprising since AD is still nowadays a clinical diagnosis, despite increasing evidence linking pathological fluid biomarkers to the disease or future development of it, which has also led to the establishment of “biological” AD research criteria (NIA-AA), only based on biomarkers [40]. For these reasons, we chose to apply the IWG-2 research criteria to further classify the AD patients, since IWG-2 criteria require both a specific AD clinical phenotype and pathological biomarkers [27]. It is possible that foreign-born patients arrive later to the clinic and are diagnosed when the disease is more advanced and symptomatic, thus not requiring a lumbar puncture to establish the diagnosis. This is suggested also based on the differences in type of established contact with the clinic, with foreign-born relying more on family members (Table 3). This could be explained by elderly foreign-born people being less integrated in society and experiencing language barriers that prevent them from seeking contact themselves or make them wait longer before they seek help, as previously shown [7].
It is also important to note that some patients with a pathological biomarker profile did not receive an AD diagnosis or symptomatic treatment, possibly because the clinical phenotype did not match the AD one, or because they still had only minor symptoms (Table 4). This tendency was larger in the foreign-born group, albeit non significantly. In research settings, these subjects would be classified as “preclinical AD” according to the NIA-AA criteria and “at risk for progression to AD” according to the IWG-2 criteria [27, 40]. In light of the future development of disease modifying drugs for AD, it is important that these subjects are correctly diagnosed and recruited for clinical trials, and that foreign-born subjects and people from ethnic minorities are not underrepresented in these studies, as previously documented [5].
Regarding medication, other differences consisted in a larger number of foreign-born patients prescribed antipsychotic and antiepileptic medicine (3 patients versus 1 in the native group), and longer time to treatment in the non-European subgroup (n = 15) compared to the other ethnic subgroups (Supplementary Table 9). This could also reinforce the hypothesis that foreign-born have atypical presentations and later access to health care. However, due to the small groups examined and lack of difference when examining the foreign-born groups as a whole, we should take these findings with caution.
Regarding structural imaging scores, foreign-born patients had a tendency to lower atrophy and significantly less white matter lesions. In a former study, it was found that a minimal-atrophy AD subtype was associated with a lower educational level, which corresponds well to the results of this study [41]. This is also believed to be in line with the cognitive reserve hypothesis, which states that education results in a greater neural reserve and neural compensation, and the finding that underlying pathology in patients with AD is seemingly more advanced in patients with a higher educational level [42]. Certainly, it should be taken into account that, despite the many confounders mentioned earlier, clinical diagnoses still tend to rely on cognitive test scores, which are lower in people with less education, possibly resulting in subjects not yet severely atrophic receiving a dementia diagnosis [37, 38]. It would also have been interesting to examine the correlation over time between cognition and atrophy in the native and foreign-born group respectively, but it could not be done in our study as we had no longitudinal data.
This study is one of the first of its kind in Sweden. Similar studies have been conducted in Denmark and Norway, and the first study on inequality in dementia care between native and foreign-born people in Sweden was published earlier this year [43–45]. Compared to the previous Swedish study, ours has the strength that the foreign-born group accounted for 28.3% of the cohort, compared to 12% in the other study [45]. We also examined contact with healthcare, CSF biomarkers, and structural imaging scales, which the previous study did not [45]. Nevertheless, there are a few limitations. The total sample size is, as mentioned earlier, relatively small, consisting of 127 patients. In addition, the groups were unevenly distributed (natives n = 91, foreign-born n = 36). Also, some foreign-born patients (n = 12) did not have the need for an interpreter in the contact with the memory clinic. This means that the language barrier was eliminated for these patients, which may have influenced the results. However, the foreign-born group appeared balanced, with a total of 20 different countries represented. Only 5 patients originated from neighboring Scandinavian countries, while the most part originated from other European (n = 15) or non-European (n = 16) countries. This means that our results can not only be compared with previous studies, but even show a greater spread of ethnicities.
Conclusions
The findings in this study show that clinical assessment of dementia in a Swedish cohort of native and foreign-born patients was mostly homogenous regarding biomarkers and parameters examined. However, differences in the diagnostic process still exist. Choice of cognitive tests and weight of biomarkers in dementia diagnosis in culturally diverse population are still matter of discussion in the scientific community. It is of great importance that more research on dementia diagnostics and education of clinical practitioners and the general public is carried out on the issue of multiculturality, as well as similar studies on larger cohorts of patients with different ethnicities.
ACKNOWLEDGMENTS
The study was supported by the Swedish federal government under the ALF agreement, as well as by the Kockska foundation and Vinnova (grant number 2021-02680).
The authors have no conflict of interest to report.
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/22-0177r2).
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/JAD-220177.
REFERENCES
[1] | Dementia, World Health Organization, https://www.who.int/news-room/fact-sheets/detail/dementia, September 2, 2021, Accessed September 20, 2021. |
[2] | Stephan BCM , Birdi R , Tang EYH , Cosco TD , Donini LM , Licher S , Ikram MA , Siervo M , Robinson L ((2018) ) Secular trends in dementia prevalence and incidence worldwide: A systematic review. J Alzheimers Dis 66: , 653–680. |
[3] | Ferri CP , Prince M , Brayne C , Brodaty H , Fratiglioni L , Ganguli M , Hall K , Hasegawa K , Hendrie H , Huang Y , Jorm A , Mathers C , Menezes PR , Rimmer E , Scazufca M ((2005) ) Global prevalence of dementia: A Delphi consensus study. Lancet 366: , 2112–2117. |
[4] | Prince M ((2000) ) Dementia in developing countries. A consensus statement from the 10/66 Dementia Research Group. Int J Geriatr Psychiatry 15: , 14–20. |
[5] | Mooldijk SS , Licher S , Wolters FJ ((2021) ) Characterizing demographic, racial, and geographic diversity in dementia research: A systematic review. JAMA Neurol 78: , 1255–1261. |
[6] | Cooper C , Tandy AR , Balamurali TB , Livingston G ((2010) ) A systematic review and meta-analysis of ethnic differences in use of dementia treatment, care, and research. Am J Geriatr Psychiatry 18: , 193–203. |
[7] | Mukadam N , Cooper C , Livingston G ((2011) ) A systematic review of ethnicity and pathways to care in dementia. Int J Geriatr Psychiatry 26: , 12–20. |
[8] | Nielsen TR , Vogel A , Riepe MW , de Mendonça A , Rodriguez G , Nobili F , Gade A , Waldemar G ((2010) ) Assessment of dementia in ethnic minority patients in Europe: A European Alzheimer’s Disease Consortium survey. Int Psychogeriatr 23: , 86–95. |
[9] | Morris JC , Schindler SE , McCue LM , Moulder KL , Benzinger TLS , Cruchaga C , Fagan AM , Grant E , Gordon BA , Holtzman DM , Xiong C ((2019) ) Assessment of racial disparities in biomarkers for Alzheimer disease. JAMA Neurol 76: , 264–273. |
[10] | Brickman AM , Schupf N , Manly JJ , Luchsinger JA , Andrews H , Tang MX , Reitz C , Small SA , Mayeux R , DeCarli C , Brown TR ((2008) ) Brain morphology in older African Americans, Caribbean Hispanics, and Whites from Northern Manhattan. Arch Neurol 65: , 1053–1061. |
[11] | (2019) International Migration 2019: Report. United Nations, Department of Economic and Social Affairs, Population Division. ST/ESA/SER.A/438. |
[12] | Canevelli M , Lacorte E , Cova I , Zaccaria V , Valletta M , Raganato R , Bruno G , Bargagli AM , Pomati S , Pantoni L , Vanacore N ((2019) ) Estimating dementia cases amongst migrants living in Europe. Eur J Neurol 26: , 1191–1199. |
[13] | Cova I , Del Tedesco F , Maggiore L , Pantoni L , Pomati S ((2020) ) Cognitive disorders in migrants: Retrospective analysis in a Center for Cognitive Disorders and Dementia in Milan. Aging Clin Exp Res 32: , 535–538. |
[14] | George KM , Lutsey PL , Selvin E , Palta P , Windham BG , Folsom AR ((2019) ) Association between thyroid dysfunction and incident dementia in the atherosclerosis risk in communities neurocognitive study. J Endocrinol Metab 9: , 82–89. |
[15] | Smith AD , Refsum H , Bottiglieri T , Fenech M , Hooshmand B , McCaddon A , Miller JW , Rosenberg IH , Obeid R ((2018) ) Homocysteine and dementia: An international consensus statement. J Alzheimers Dis 62: , 561–570. |
[16] | Kern J , Kern S , Blennow K , Zetterberg H , Waern M , Guo X , Börjesson-Hanson A , Skoog I , Östling S ((2016) ) Calcium supplementation and risk of dementia in women with cerebrovascular disease. Neurology 87: , 1674–1680. |
[17] | Calvo-Rodriguez M , Hou SS , Snyder AC , Kharitonova EK , Russ AN , Das S , Fan Z , Muzikansky A , Garcia-Alloza M , Serrano-Pozo A , Hudry E , Bacskai BJ ((2020) ) Increased mitochondrial calcium levels associated with neuronal death in a mouse model of Alzheimer’s disease. Nat Commun 11: , 2146. |
[18] | Llewellyn DJ , Langa KM , Friedland RP , Lang IA ((2010) ) Serum albumin concentration and cognitive impairment. Curr Alzheimer Res 7: , 91–96. |
[19] | Zhang X , Bao G , Liu D , Yang Y , Li X , Cai G , Liu Y , Wu Y ((2021) ) The association between folate and Alzheimer’s disease: A systematic review and meta-analysis. Front Neurosci 15: , 661198. |
[20] | Moore E , Mander A , Ames D , Carne R , Sanders K , Watters D ((2012) ) Cognitive impairment and vitamin B12: A review. Int Psychogeriatr 24: , 541–556. |
[21] | Peters R , Burch L , Warner J , Beckett N , Poulter R , Bulpitt C ((2008) ) Haemoglobin, anaemia, dementia and cognitive decline in the elderly, a systematic review. BMC Geriatr 8: , 18. |
[22] | Wolters FJ , Zonneveld HI , Licher S , Cremers LGM , Group obot HBCCR , Ikram MK , Koudstaal PJ , Vernooij MW , Ikram MA ((2019) ) Hemoglobin and anemia in relation to dementia risk and accompanying changes on brain MRI. Neurology 93: , e917–e926. |
[23] | (2020) Ett mångkulturellt kognitivt testbatteri. Kunskapscentrum Demenssjukdomar, Migrationsskolan Rapport 2020:2. |
[24] | The World Bank, https://data.worldbank.org/. |
[25] | Blennow K , Hampel H , Weiner M , Zetterberg H ((2010) ) Cerebrospinal fluid and plasma biomarkers in Alzheimer disease. Nat Rev Neurol 6: , 131–144. |
[26] | Jack CR Jr , Bennett DA , Blennow K , Carrillo MC , Feldman HH , Frisoni GB , Hampel H , Jagust WJ , Johnson KA , Knopman DS , Petersen RC , Scheltens P , Sperling RA , Dubois B ((2016) ) A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 87: , 539–547. |
[27] | Dubois B , Feldman HH , Jacova C , Hampel H , Molinuevo JL , Blennow K , DeKosky ST , Gauthier S , Selkoe D , Bateman R , Cappa S , Crutch S , Engelborghs S , Frisoni GB , Fox NC , Galasko D , Habert MO , Jicha GA , Nordberg A , Pasquier F , Rabinovici G , Robert P , Rowe C , Salloway S , Sarazin M , Epelbaum S , de Souza LC , Vellas B , Visser PJ , Schneider L , Stern Y , Scheltens P , Cummings JL ((2014) ) Advancing research diagnostic criteria for Alzheimer’s disease: The IWG-2 criteria. Lancet Neurol 13: , 614–629. |
[28] | Wahlund L-O , Westman E , van Westen D , Wallin A , Shams S , Cavallin L , Larsson EM ; From the Imaging Cognitive Impairment Network (ICINET) ((2017) ) Imaging biomarkers of dementia: Recommended visual rating scales with teaching cases. Insights Imaging 8: , 79–90. |
[29] | Prajapati B , Dunne MCM , Armstrong RA (2010) Sample size estimation and statistical power analyses. Optometry Today 16/07. |
[30] | Ismail Z , Rajji TK , Shulman KI ((2010) ) Brief cognitive screening instruments: An update. Int J Geriatr Psychiatry 25: , 111–120. |
[31] | Pinto E , Peters R ((2009) ) Literature review of the Clock Drawing Test as a tool for cognitive screening. Dement Geriatr Cogn Disord 27: , 201–213. |
[32] | Pellicer-Espinosa I , Díaz-Orueta U ((2022) ) Cognitive screening instruments for older adults with low educational and literacy levels: A systematic review. J Appl Gerontol 41: , 1222–1231. |
[33] | Storey JE , Rowland JT , Basic D , Conforti DA ((2002) ) Accuracy of the clock drawing test for detecting dementia in a multicultural sample of elderly Australian patients. Int Psychogeriatr 14: , 259–271. |
[34] | Borson S , Brush M , Gil E , Scanlan J , Vitaliano P , Chen J , Cashman J , Sta Maria MM , Barnhart R , Roques J ((1999) ) The Clock Drawing Test: Utility for dementia detection in multiethnic elders. J Gerontol A Biol Sci Med Sci 54: , M534–M540. |
[35] | Paddick SM , Gray WK , McGuire J , Richardson J , Dotchin C , Walker RW ((2017) ) Cognitive screening tools for identification of dementia in illiterate and low-educated older adults, a systematic review and meta-analysis. Int Psychogeriatr 29: , 897–929. |
[36] | Palsetia D , Rao GP , Tiwari SC , Lodha P , De Sousa A ((2018) ) The Clock Drawing Test versus Mini-mental Status Examination as a screening tool for dementia: A clinical comparison. Indian J Psychol Med 40: , 1–10. |
[37] | Schmand B , Lindeboom J , Hooijer C , Jonker C ((1995) ) Relation between education and dementia: The role of test bias revisited. J Neurol Neurosurg Psychiatry 59: , 170–174. |
[38] | Ranson JM , Kuźma E , Hamilton W , Muniz-Terrera G , Langa KM , Llewellyn DJ ((2019) ) Predictors of dementia misclassification when using brief cognitive assessments. Neurol Clin Pract 9: , 109–117. |
[39] | Nielsen TR , Segers K , Vanderaspoilden V , Beinhoff U , Minthon L , Pissiota A , Bekkhus-Wetterberg P , Bjørkløf GH , Tsolaki M , Gkioka M , Waldemar G ((2019) ) Validation of a brief Multicultural Cognitive Examination (MCE) for evaluation of dementia. Int J Geriatr Psychiatry 34: , 982–989. |
[40] | Jack CR Jr , Bennett DA , Blennow K , Carrillo MC , Dunn B , Haeberlein SB , Holtzman DM , Jagust W , Jessen F , Karlawish J , Liu E , Molinuevo JL , Montine T , Phelps C , Rankin KP , Rowe CC , Scheltens P , Siemers E , Snyder HM , Sperling R ; Contributors ((2018) ) NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement 14: , 535–562. |
[41] | Persson K , Eldholm RS , Barca ML , Cavallin L , Ferreira D , Knapskog AB , Selbæk G , Brækhus A , Saltvedt I , Westman E , Engedal K ((2017) ) MRI-assessed atrophy subtypes in Alzheimer’s disease and the cognitive reserve hypothesis. PLoS One 12: , e0186595. |
[42] | Stern Y ((2012) ) Cognitive reserve in ageing and Alzheimer’s disease. Lancet Neurol 11: , 1006–1012. |
[43] | Stevnsborg L , Jensen-Dahm C , Nielsen TR , Gasse C , Waldemar G ((2016) ) Inequalities in access to treatment and care for patients with dementia and immigrant background: A Danish nationwide study. J Alzheimers Dis 54: , 505–514. |
[44] | Sagbakken M , Spilker RS , Nielsen TR ((2018) ) Dementia and immigrant groups: A qualitative study of challenges related to identifying, assessing, and diagnosing dementia. BMC Health Serv Res 18: , 910. |
[45] | Lindgren E , Sörenson J , Wattmo C , Kåreholt I , Nägga K ((2021) ) Differences in dementia care between Swedish-born and foreign-born from countries with different country level socioeconomic position: A nationwide register-based study. J Alzheimers Dis 84: , 1363–1371. |




