Views on the Desirability of Diagnosing Sporadic Cerebral Amyloid Angiopathy with Biological Evidence
Abstract
Background:
Sporadic cerebral amyloid angiopathy (sCAA) research of the past decade has increasingly focused on developing biomarkers that allow for an earlier and more accurate sCAA-diagnosis. Considering that sCAA does not have treatment options available (yet), more fundamental questions concerning the desirability of using such early-sCAA biomarkers in clinical practice need to be addressed.
Objective:
In this qualitative interview study, we aim to explore the views of vascular neurologists on the purpose and possible consequences of an earlier and more accurate sCAA-diagnosis, using new biomarkers.
Methods:
Vascular neurologists from around the world were approached via email and interviewed via video call. Topics included views on current sCAA diagnostic practice, considerations on the use of new biomarkers, and expectations and hopes for the future. All interviews were transcribed ad verbatim using a transcription program (Otter.ai). Transcripts were analyzed using inductive content analysis.
Results:
We interviewed 14 vascular neurologists. Views regarding the desirability of new sCAA-biomarkers differed substantially between interviewees as to when and in whom these biomarkers could be of benefit in clinical practice. These differences were mainly reported with regards to prognosis, risk stratification, and biological precision, between general stroke neurologists and neurologists with specific sCAA-expertise.
Conclusion:
Views on the use of sCAA-biomarkers in clinical practice differ substantially between vascular neurologists. There is particularly no consensus regarding when, and in whom sCAA biomarkers could be useful in clinical practice.
INTRODUCTION
Sporadic cerebral amyloid angiopathy (sCAA) is common in older adults and is characterized by the accumulation of amyloid-β proteins in cerebral arteries and arterioles [1]. Amyloid-β accumulation can lead to weakening of the cerebral vasculature, increasing the risk for intracerebral hemorrhages (ICH) and cognitive impairment [2].
In the past decade, sCAA research has increasingly focused on developing biomarkers, e.g., in cerebrospinal fluid and (even more so) MRI, that allow for an earlier and more accurate sCAA-diagnosis. The current gold standard for a sCAA-diagnosis is based on biopsy, or autopsy findings [3]. For a diagnosis during life, the (modified) Boston-criteria can be used [4, 5]. These criteria comprise clinical, radiological, and pathological features, but are primarily tailored to detect microbleeds and superficial siderosis on brain magnetic resonance imaging (MRI), which may be manifestations of relatively late stage sCAA [6, 7]. Other limitations of the Boston-criteria are the relatively low sensitivity (44–70%) for diagnosing sCAA compared to biopsy and autopsy findings, and the fact that not every person can or wants to undergo an MRI-scan [8]. Consequently, estimations on the incidence of sCAA in population-based studies vary substantially [1].
The assumption is that early detection of sCAA might have clinical benefits, including informing patients about their risk to develop ICH or cognitive impairment, or providing early intervention possibilities once these become available [9–12].
Despite the research focus on finding new sCAA biomarkers, more fundamental questions concerning the desirability of early CAA diagnostics in clinical practice, e.g., if an early CAA diagnosis is desirable, for which patients this would apply and at what moment, were never scientifically addressed. The aim of this study is therefore to explore the views of vascular neurologists around the world on the purpose and possible consequences of an earlier and more accurate sCAA-diagnosis in clinical practice, using biomarkers, and to provide a link between biomarker research developments and the potential benefits of these biomarkers for (future) patients in clinicalpractice.
METHODS
Study design
This interview-study was part of the Biomarkers for Cognitive Impairment due to Cerebral Amyloid Angiopathy (BIONIC) project, which investigates the potential of biomarkers for a better diagnosis in clinical practice [13]. The local ethical committee approved the BIONIC project, as is stated in the Acknowledgments section. Consolidated criteria for Reporting Qualitative (COREQ) criteria were used to ensure the quality of reporting [14].
Participants
Between December 2020 and June 2021, practicing vascular neurologists from various parts of the world were approached via e-mail, and were informed about the content and purpose of the BIONIC-interview project. We invited vascular neurologists only, because they are more likely to have contemplated about the use of sCAA biomarkers in clinical practice given that they might diagnose sCAA in clinical practice—the sCAA diagnosis is generally used in specialist setting. Of these vascular neurologists, some had expertise sCAA while others had expertise in general stroke. To be able to collect a broad range of views and opinions, we used purposive sampling for the selection of the interviewees, taking field of expertise (sCAA or general stroke), years of work experience, and location (international, preferably one interviewee per research institute/clinical department) into account. Some interviewees were involved in sCAA research. We sampled from our own direct and indirect (inter)national network in combination with a non-exhaustive search in Medline. Enrolment ended when no new answers came up in the interviews in response to the main research questions, i.e. when thematic saturation was reached [15]. One interviewee requested for an overview of the interview questions prior to the interview. All interviews were audio-recorded with informed consent given verbally at the start of the interview. All audio recordings were collected in a secured digital workspace.
Data collection
In preparation of the interviews, we performed one pilot-interview with a general stroke neurologist. Two researchers conducted the semi-structured interviews (MS and HA) via videocall. One had a background in medical ethics and had experience in conducting interviews, the other had a background in clinical neurology and was trained by the former researcher to conduct the interviews.
The interview guide (Supplementary Material) was composed by four researchers (MS, ER, MP, and HA). This guide comprised three parts: 1) Views on current sCAA practice; 2) Considerations on the use of new biomarkers; 3) Expectations and hopes for the future. Part 1 included the interviewees’ current diagnostic workup for diagnosing sCAA; interviewees were asked if they would use this diagnosis in clinical practice and whether they experience obstacles in sCAA-diagnostics. In part 2, we discussed considerations both in favor and against the usage of new sCAA biomarkers in clinical practice. Part 3 comprised of the future perspective; interviewees were asked what role biomarkers could play in sCAA-diagnostics, and what they hoped to see for the future. We made small adjustments to the interview questions after each interview, if deemed necessary.
Coding and data analysis
The interviews were transcribed ad verbatim using a transcription program (Otter.ai). Data analysis started directly after the first interview, using inductive content analysis [16]. In the first phase of coding and analysis, two researchers (MS and HA) independently coded the interviews to limit the risk of bias and subjectivity in the analyses. Codes were then compared and discussed until consensus was reached about a preliminary coding tree, which was then used by the researcher (HA) to code remaining transcripts. After each interview the coding tree was updated. If new codes emerged, all interviews were reviewed according to the new codes. We combined open codes into axial codes, categories, and themes. The final coding tree was discussed with other researchers (MP, ER) and adjusted accordingly. Disagreements were settled by consensus.
In the second phase, the remaining interviews were coded by HA, and subsequently reviewed by MS. This coding was based on the initial coding tree that was adapted and elaborated based on new concepts emerging from the data. In the third phase, axial coding of the data was performed to identify clusters of considerations in the interviews. HA performed this coding, which was then reviewed by MS and later discussed with MP and ER. Disagreements were settled by consensus. Data collection and analysis continued until no new codes, concepts or patterns could be identified from the data. Analyses were supported using a qualitative data analysis software program (Atlas.ti 8.4.20).
RESULTS
Study population
We approached 30 potential interviewees between November 2020 and June 2021. 14 neurologists did not respond to our repeated request, one declined because of change in work responsibilities, another declined because of time constraints. Eventually, we interviewed 14 vascular neurologists with expertise in general stroke and/or sCAA. Due to risk of identification of interviewees in the relatively small field of stroke/sCAA, we do not include a table illustrating characteristic per participant. Interviewees were from the United States, Canada, United Kingdom, France, Denmark, and the Netherlands. Ten were men and four were women. Most worked at university hospitals as practicing neurologists and researchers, memory centers included. Work experience as a medical specialist in the field of neurology ranged from one to over 30 years. Of 14 neurologists, six had specific expertise in sCAA. All neurologists were familiar with CAA-diagnostics in clinical practice, but variation existed in the depth and frequency of their own involvement in diagnosing CAA. Interview duration ranged from 20–45 minutes. The inclusion of experts from a variety of countries and fields of expertise (sCAA and general stroke), renders our purposive sample effective.
Considerations for the general use of sCAA-biomarkers
Views on the desirability of implementing new sCAA-biomarkers in current practice varied substantially between the interviewees. Distinguishing subtypes of sCAA, monitoring disease progression, preventing complications (e.g., hemorrhagic stroke), and providing individuals with a prognosis were reported as generally useful properties of scAA biomarkers, but differences existed as to when, and in whom these purposes would be useful in clinical practice. Table 1 depicts an overview of illustrative quotes regarding neurologists’ views on the development and implementation of new biomarkers. The considerations mentioned seemed to be partly driven by participants’ fields of expertise, namely sCAA or general stroke. Those with specific expertise in sCAA generally seemed to argue more towards the potential benefits of the use of new sCAA-biomarkers. These considerations were expressed from a more groundbreaking point of view, because they regarded sCAA as a problem that applies to a greater population (e.g., asymptomatic older individuals), than solely those who have already been diagnosed with the illness after symptoms became apparent. Arguments in favor of a broader use of new sCAA-biomarkers in clinical practice were sometimes also used for the general population. Whereas those with expertise in general stroke generally seemed to use more terms and conditions as to when, and in what populations new biomarkers would be of added value in clinical practice, taking the clinical presentation and patient/person subpopulation into account in their argumentation.
Table 1
Statements made by interviewees concerning current sCAA-diagnostics and considerations in favor/against the implementation of novel biomarkers
| Current sCAA workup: Boston-criteria |
| “The Boston criteria are the best that we have so far.” |
| “So far, the Boston criteria work pretty well in terms of diagnosing sCAA.” |
| “Current Boston criteria cause a barrier between research and clinical practice.” |
| “The problem with the Boston criteria is that it’s too specific and not sensitive enough.” |
| “Boston criteria often misapplied by clinicians with no expertise of sCAA.” |
| “Current biomarkers do not particularly map well to clinical function.” |
| Considerations in favor/against implementing new biomarker |
| Prognosis |
| “ A biomarker should not fulfil prognostic information, because it is too nonspecific and heterogeneous between patients, therefore being unreliable.” |
| “ Every patient wants to know what’s going on, and what their prognosis is.” |
| “ Prognosis of cognitive decline as part of sCAA diagnosis is very much part of the diagnostic and therapeutic work-up plan.” |
| “ Patients would rather not know details of prognosis if no treatment is available.” |
| “Biomarker useful for prognosis when it induces change in person’s lifestyle.” |
| Informing |
| “As a clinician, I do recognize microbleeds and cognitive decline and take it into account, but I don’t discuss it much further with patients because of the lack of knowledge how to deal with that information.” |
| “It can be helpful for patients to know more about their illness, even if there are no treatments available.” |
| “Knowledge on illness can damage one’s life.” |
| “Research shows that there is no societal benefit to knowing early about incurable conditions.’ |
| “Telling all the possible details when it comes to hypothetical prognosis and neurological complications is burdensome for patients and their family.” |
| “Explanation might cause relieve in patients.” |
| “In some cases, when there are no treatment options available, you must protect the patient and not explain the diagnosis (in detail).” |
| Risk stratification |
| “Being able to stratify the individual risk of ischemic stroke and recurrent hemorrhage would help clinicians make better therapeutic decisions.” |
| “Knowing the severity of sCAA, modifies the therapeutic approach.” |
| “Being able to stratify the individual risk of ischemic stroke and recurrent hemorrhage would help clinicians make better therapeutic decisions.” |
| “sCAA biomarkers not useful for prognosis of (recurrent) hemorrhage, because risk of hemorrhage will stay the same nonetheless.” |
| “Being able to make multiple diagnoses and separate illness from another, might be helpful for risk stratification.” |
| Prevention |
| “Diagnostic biomarker can be helpful in guiding secondary prevention strategies.” |
| “Being able to prevent a second hemorrhage, and even better, first hemorrhage.” |
| “Biomarker to distinguish illness in earlier stage, might be beneficial for therapeutic consequences.” |
| “In sCAA patients who present with stroke, the focus is to prevent recurrent stroke.” |
| “There might be an economic value as to predict and prevent future disability.” |
| Biological precision |
| “Biomarkers are usually extremely unspecific and many times not useful in clinical practice.” |
| “Diagnosing a disease with some biological precision would also lead to better information on the course of the disease, which could be helpful for the future.” |
| “Diagnosing sCAA with more biological precision would help cover the knowledge gap in sCAA.” |
| “Diagnosing a disease with some biological precision would also lead to better information on the course of the disease, which could be helpful for the future.” |
| “Biological precision has only to do with a clinical diagnosis, and not necessarily with the course of the disease.” |
| Treatment and tracking treatment effects |
| “Few sCAA researchers or clinicians would recommend early screening until we understand more about disease modifying treatment.” |
| “Given that there are no treatment options available yet for sCAA, there is no need for distinguishing sCAA patients from one another.” |
| “If we would have disease modifying treatments, you would want to search for sCAA markers in asymptomatic people.” |
| “It would be useful to be able to provide patients with more personalized treatment.” |
| “Purpose biomarkers might become more useful when treatments become available.” |
Some interviewees were very specific about the purpose and setting in which a new biomarker would be useful for current practice;
“You get a person who shows up at age 70, with a lobar hemorrhage. Being able to say: yes, this is amyloid angiopathy, and you’re likely to have another bleed. That’s not very helpful. Because even if the testing was negative, I would say you’re likely to have another bleed.”. . . “Where it may be helpful is in a rare case of a hemorrhage. If you’re 30, or 40, and you have a hemorrhage, perhaps these biomarkers would be a bit more helpful. Just because that’s a very unusual scenario. And you don’t really know what their prognosis is. But but that’s very rare. It would be like one patient a year.”
Besides field of expertise, global differences in the organization of health care also seemed to influence the reasoning for the usage of new biomarkers, particularly differences in how the financial costs of biomarker testing was covered. Depending on the country of origin, some interviewees further substantiated their reluctance towards expanding the use of biomarkers in clinical practice, due to the high medical costs that are linked with these investigations (e.g., MRI and positron emission tomography, PET scans). For these clinicians, additional investigations were only performed, if findings were expected to have clinical consequences for the patient. Others mentioned that there can be a financial incentive for doctors, depending on the country, to offer biomarker testing.
“In some countries, hospitals make money by doing tests, and they make money when a person has a disease, right? The hospital receives funding from a third party to do all of these things, and they make a profit out of it. Whereas our system is the exact opposite. We lose money every time we do a test. We have an envelope of funding for all of our tests in our health care. But the more you use, the less there is for everybody. So in our context, we have a mantra: if it doesn’t change management, why would you do it?”
Figure 1 schematically illustrates the interviewees’ considerations in favor of and against new biomarkers, and possible consequences of implementing such biomarkers in clinical practice.
Fig. 1
Consequences and considerations for the implementation of new sCAA biomarkers in (future) clinical practice.
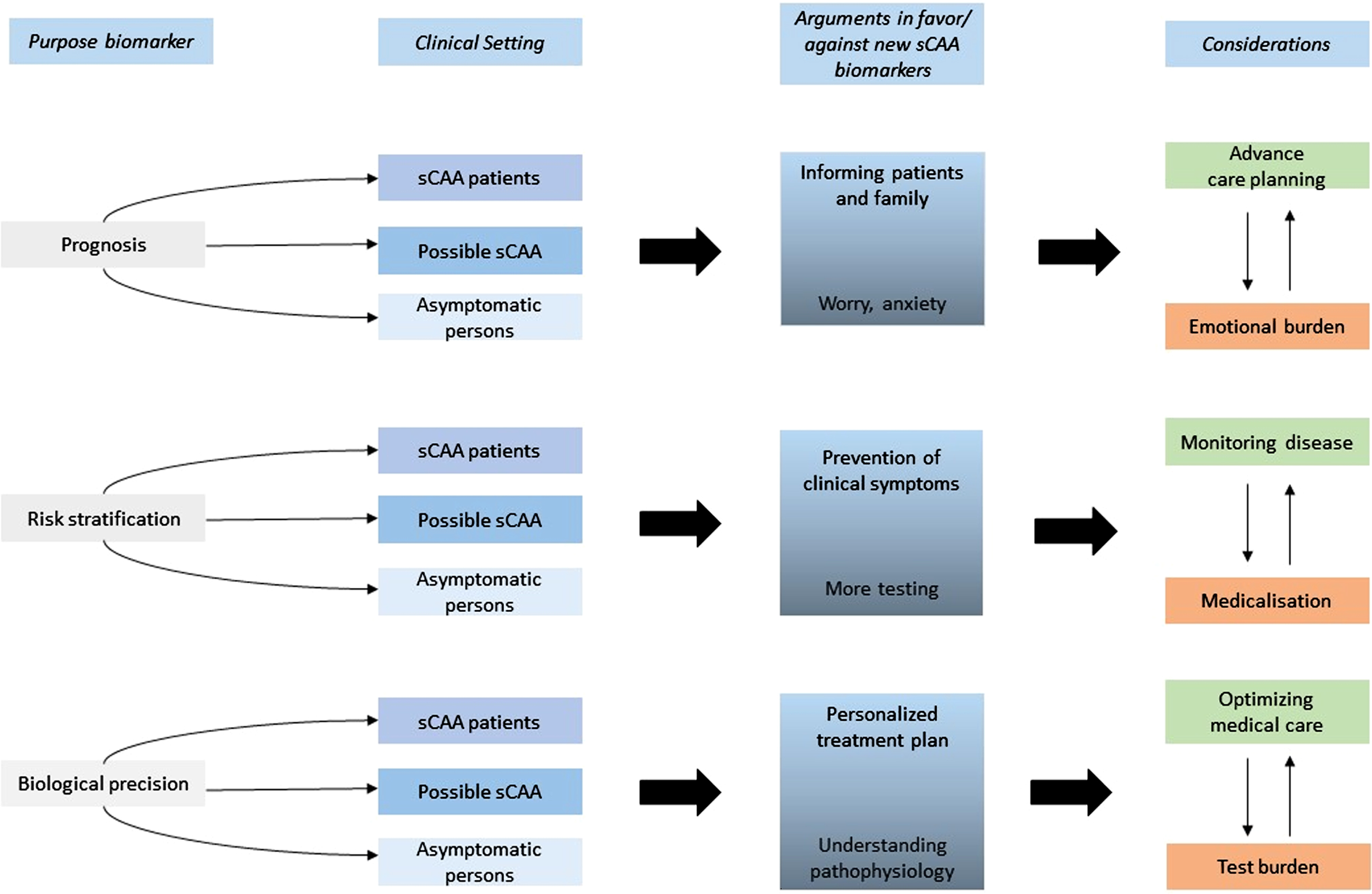
Analysis of the interviews showed that considerations regarding the use of new biomarkers varied most within three themes: Prognosis; Risk stratification; and Biological precision. We will discuss these theme-based considerations in more detail.
Prognosis
The theme of providing a prognosis as a potential benefit of sCAA biomarkers was limited to the patient group of individuals with (suspected) sCAA. Being able to provide a (more accurate) prognosis due to sCAA biomarkers was considered to be an important part of the diagnostic process by many, arguing that this information can be helpful when informing patients and family members on the expected disease progression. Many of these interviewees found the idea of not informing patients on the prognosis, if this information would be or could become available, because of lack of treatment options) as paternalistic, and sometimes unethical.
“But, you know, from an ethical standpoint, we’ve definitely moved to the tell everybody, everything. You don’t have to hit them on the head with the diagnosis, if they don’t want to hear it. But I think everybody wants to know what’s going on and wants to know their prognosis.”
Others were more reluctant towards prognosis, partly because of the lack of current treatment options. Some elaborated that the complexity of sCAA as a disease, including the heterogeneity in clinical manifestations, makes a CAA prognosis by definition unreliable.
“A biomarker should not fulfil prognostic information, because it is too nonpecific and hetero-geneous between patients, therefore being unreliable.”] Most of these interviewees believed that a prognosis would primarily be useful if there would be treatment options available or practical lifestyle consequences (e.g., in the context of advance care planning).
Risk stratification
The theme of risk stratification was discussed in a broader context, both with respect to asymptomatic individuals, those with suspected sCAA, and sCAA patients. Gaining information on the risk of developing clinical symptoms due to sCAA (such as hemorrhages and cognitive impairment) was thought to be particularly useful for sCAA patients to be able to prevent neurological complications because of medical management of stroke risk.
“.. I mean, that there’s this very careful weighing of risks versus benefits that has to be done. From a research standpoint, having those biomarkers would allow us to say: Okay, now, let me see what the risk of bleeding is with the biomarker and without the biomarker, and then model in some way whether you’re more likely to prevent a stroke or more likely to cause a stroke [when administering anti-coagulants]. There’s a danger of misuse of information, I think, for sure. But I think that’s a danger that can be handled, I think that it just would take thoughtful analysis and really clear directions on how it should be used and how it shouldn’t be used.”
When it concerned those with ‘possible sCAA’ according to the Boston criteria, and asymptomatic persons, views on the implementation of a biomarker test for sCAA risk stratification varied. Some argued that this could lead to more testing in clinical practice, possibly leading to over-medicalization, with little beneficial value for the patient/individual, which was thought to be unfavorable for both patients as well as doctors.
The implementation of sCAA biomarker testing in the asymptomatic, older population was not considered appropriate by many, even when these biomarkers could diagnose CAA more accurately. Concerns arose about the possibility of medicalization and inducing worry and anxiety.
“[in response to bringing CAA biomarker tests from research to clinical practice:] I think that all people fall in love with their own ideas. I do too. I think that we should be very much aware of the difference between what we actually know and what we think and what we hope. We as doctors should realize that a lot of what we do is certainly not evidence based. And just because we do this as doctors, it doesn’t mean it’s science, it can be experienced, it can be good, you know, good artisan. But before we implement something new, we should do the trials.”
Others focused more on the notion that a new biomarker could help predict the risk of developing sCAA at an early stage, which was thought to be helpful for advance care planning and treatment management, especially when treatment options would become available in the future.
“So it’d be nice to know that they don’t have either a sCAA or AD. And my hope is that [knowing] this ratio [sCAA vs. AD pathology] may help[..], those with a higher ratio of AD to sCAA, I would say probably have a worse prognosis. They might be better responders to cholinesterase inhibitors . . . “This would all be useful information to me, even if I can’t cure either disease, right now, it’s more prognostic information, as I’m describing, but to be able to know that, so it’s probably not yes or no, it’s probably like a score, you know, your amyloid-beta ratio.”] Views on what kind of patient population could benefit from knowing once’s risk to develop symptoms due to sCAA, differed.
Biological precision
Most interviewees found the Boston-criteria, currently used to diagnose sCAA, not accurate enough (particularly regarding its relatively low sensitivity) and its interpretation complex at times. Of these interviewees, some argued that novel biomarkers could help provide a more accurate diagnosis at an earlier stage of the disease. A smaller number of interviewees argued that they found the current Boston-criteria sufficiently accurate for standard sCAA-diagnostics in current practice. They did not express a sense of urgency for the development for new sCAA-biomarkers for being able to diagnose CAA with more biological precision but found other functionalities more relevant;
“Yes, there is a need for new biomarkers, but I don’t think this is specific to sCAA... Currently, our knowledge of CAA is based on a very heterogeneous group of patients. I would say that we would need better biomarkers to be able to disentangle the different subtypes of sCAA.”
Others reported that despite sCAA-diagnostics not being ideal, the implementation of novel biomarkers would only be useful in a research setting, and not on a larger scale in clinical practice, because of the lack of treatment options available.
The main incentive for diagnosing sCAA with more biological precision was to be able to provide patients with a more personalized treatment plan. This was particularly mentioned for patients with less typical sCAA-symptoms, in whom neurologists face greater dilemmas regarding treatment (e.g., cessation of the use of anticoagulants in patients with atrial fibrillation to avoid increasing the risk of stroke in those with sCAA). According to most neurologists, the ideal of diagnosing sCAA with more biological precision was deemed not applicable for asymptomatic persons, because they believed reaching consensus on whether there is an added value of new sCAA-biomarkers (and if so, in which subpopulations) is crucial before starting general population screening. Furthermore, some of these interviewees added that testing an asymptomatic population at a larger scale, for instance with a blood test by the general practitioner, would result in higher test burden, increase stress and anxiety, with little to no added value for this subpopulation. Others advocated for the implementation of new biomarkers in this group regardless, given the beneficial value of better understanding the pathophysiology of sCAA, next to being able to provide a future risk-stratification to those who undergo testing. The latter was thought to be particularly useful for future research and for the development of more effective treatment options.
DISCUSSION
This qualitative study on the desirability of the implementation of new sCAA-biomarkers revealed that views regarding the added value of these biomarkers differed as to when, and in whom these biomarkers should be used in clinical practice. Considerations for the desirability of new biomarkers varied most within three themes: prognosis, risk stratification, and biological precision. The direction of these considerations seemed partly entangled with participants’ field of expertise (sCAA or general stroke), but also by global differences in the ability to cover financial costs of additional medical examinations.
Many interviewees considered providing a prognosis as part of the diagnostic process in individuals with suspected sCAA who visit their practice, regardless of whether treatment options are available. Others argued that the lack of treatment options and the complexity of sCAA as a disease entity, make a prognosis less accurate, and therefore not very useful.
Risk stratification through biomarker testing was considered useful for sCAA patients by most interviewees. Estimating ICH risk in high-risk group patients (e.g., those who use anticoagulant therapy) was found particularly useful because it could help to optimize a treatment plan to avoid ICH. Others worried about over-medicalization and considered the beneficial value of knowing this kind of information limited, because they considered risk of ICH inherently associated with sCAA.
Lastly, the property of biomarkers to offer biological precision when diagnosing sCAA was often compared with the current use of the Boston-criteria. There was great variability of opinions regarding the usage and reliability of the Boston-criteria. Some did not rely too much on these criteria when making a sCAA-diagnosis and felt no need for a new diagnostic framework based on biomarkers, whereas others considered them as not ideal, and advocated for the need of a new biomarker with more accurate properties to help diagnose sCAA more accurately.
The differential findings in our study could take root in nuanced differences regarding the concept of personalized medicine. In the last two decades, research purposes have increasingly focused on developing biomarker tests for prediction, prevention, and precision-based medicine [17–20]. The development of biomarkers has taken flight within the medical field more generally, based on the underlying idea that by measuring bodily parameters, one would be able to characterize specific processes in the body that may be markers for health and disease [21], thereby enabling a more accurate diagnosis or prognosis, potentially leading to new intervention possibilities. With this goal in mind, the use of sCAA biomarkers may almost seem self-evident.
An important notion in this discussion is however, that molecular parameters only become relevant markers of disease when they can be aligned with relevant clinical events.
The widespread implementation of biomarkers under the guise of personalized medicine may in some diseases provoke complex dilemmas, for example related to finding a balance between providing accurate diagnostic information that is clinically relevant and trying to avoid over-medicalization. In multi-factorial diseases, in which the underlying pathological mechanisms are still largely unknown, the implementation of new biomarkers is less straightforward because it remains uncertain to what extent they reflect a ‘pathological mechanism’ that is causing the symptoms. Recent discussions on the use of biomarkers in the Alzheimer’s disease (AD) field illustrate this complex dilemma well. The weak correlation between AD-related neuropathological changes and clinical symptoms complicates the distinction between being ‘at risk’ for disease and having disease. Those who believe that AD biomarkers reflect the pathological cause of symptoms, tend to be more favorable towards biomarker testing. Furthermore, the prognostic abilities of AD-biomarkers are disputed, given that a considerable number of people with mild cognitive impairment and with positive AD biomarkers may never develop AD [24].
Parallels can be drawn with sCAA. Studies have shown that 23–48% of persons of 65 years and older have sCAA-related neuropathological changes at autopsy [1]. As in AD, this raises the question whether having sCAA neuropathological changes at autopsy reflect general ageing. Again, the framework of the implementation of new biomarkers is possibly more complicated because biological observations may not automatically be linked to clinically (relevant) observations. This is further supported by a recent review, that reported the great variety of definitions used for diagnosing sCAA [1]. These standards may depend on the clinical setting, e.g., primary care versus university hospital, in which different doctors have different experiences regarding the usefulness of implementing more biomarkers, but also on the variety of definitions that are used for one disease [1]. Biomarker tests may fit better with one definition of scAA than with another. Determining a cut-off value for biomarker tests in clinical practice, distinguishing ‘healthy’ from ‘disease’, would therefore be complicated when it comes to screening asymptomatic older persons for sCAA-related pathological findings.
Appreciation of the intrinsic and intangible value of having more diagnostic certainty (“knowing for the sake of knowing”) also differs between patients, physicians, and those who manage care (i.e., policy makers or care managers). These parties involved may also have misperceptions about how the other group appreciates the value of diagnostic certainty [25]. It is worth mentioning that despite good intentions of wanting to inform patients (and perhaps asymptomatic individuals) on current/future disease risk, this may have detrimental rather than beneficial effects [11, 26, 27]. According to patients, knowing one is ‘at risk’ for disease, with no options for prevention or treatment, can cause stress, anxiety, and depression, because the acquired knowledge is not actionable [28]. In a recent focus group study, (future) sCAA patients and caregivers confirmed [29] that information about disease can be ‘premature’ and even ‘harmful’ when recipients feel unable to act upon the information for their (self-) care. This was specifically the case when symptoms were related to (incipient) cognitive impairment. New biomarkers may potentially improve the prediction of ICH, but the question if and when an ICH could occur will most likely remain unanswered. Patients and relatives may benefit from information on the cause of ICH, but undoubtedly would derive more benefit if specific preventive treatments were available, which is currently not the case.
One could therefore argue that doctors who assume that patients would want to know everything, and therefore perform extensive testing without enabling the patient the right not to know, can in a sense, also be viewed as paternalistic.
Lastly, several studies have investigated the financial appreciation (i.e., ‘willingness to pay’) of patients and the general public for diagnostic information that may not influence care strategies [26, 27]. Knowing the views of those who pursue diagnostic tests for a disease for which there is no treatment, such as sCAA, i.e., researchers, or physicians, who decide in clinical practice on the use of the test, received little attention so far. The added value of ‘better’ diagnostic testing for sCAA however remains unclear—or, at the minimum, not yet explicitly defined—even though all the research efforts invested in finding sCAA biomarkers seems to assume that diagnostic testing always leads to scientific and/or clinical improvement. This may feed into the often-described phenomenon of physicians involving excessive diagnostic testing in the hunt for diagnostic certainty, even against the advice of clinical guidelines [30, 31]. Identifying who may benefit from improved diagnostic testing, and who may not, in parallel of the development of these tests, could avoid excessive use of diagnostic tests and channel its development and use in the most appropriate way.
Strengths of this study include the wide variety of participant characteristics, particularly considering a balanced number of participants with expertise in sCAA or general stroke. Furthermore, the international aspect of this study presents a diverse spectrum, taking the cultural norms, patient populations, and financial abilities to cover for diagnostic testing that may differ between healthcare centers worldwide into account. Other strengths of this study include data-triangulation and the combination of experiences from clinical practice with analysis of moral reasoning. Also, the involvement of a multidisciplinary research team contributed to addressing the new sCAA-biomarkers from diverse relevant angles. A limitation of this study is the fact that 16/30 invited neurologists did not respond to our invitation or declined because of busy work schedules. These refusals seemed at random and could not clearly be explained by specificcharacteristics.
A potential future indication for using biomarkers to confirm sCAA would be to predict Amyloid Related Imaging Abnormalities in patients who would be prescribed anti-amyloid therapy for AD. Currently, in Europe there is not approved anti-amyloid therapy, and in the US, there is a highly controversial conditional approval of aducanumab, which so far is unlikely to be prescribed at a large scale.
Conclusion
Our study findings show that there is a great variety in views concerning the use of new sCAA-biomarkers between vascular neurologists from around the world. There is particularly no consensus regarding when and in whom sCAA biomarkers could be useful in clinical practice. The substantially different views between vascular neurologist may evoke the debate on who would benefit from these biomarkers in clinical practice, and what arguments are important to consider in the usage of these novel biomarkers in the near future.
ACKNOWLEDGMENTS
This study has been approved by the Medical Ethics Committee Arnhem-Nijmegen (file no. 2017-3810). All authors critically revised the manuscript.
We thank all clinicians who were willing to participate in the interview study.
This study was part of the BIONIC project (no. 733050822), made possible by ZonMW.
BIONIC is part of ‘Memorabel’, the research and innovation program for dementia, as part of the Dutch national ‘Deltaplan for Dementia’: zonmw.nl/dementiaresearch.
This project is also funded by The Netherlands Organization for Health Research and Development (ZonMw) VIDI grant 91718303 to E. Richard. The funders did not play a role in any part (such as initiation, execution, or interpretation of the results) of this manuscript.
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/22-0052r1).
REFERENCES
[1] | Jäkel L , De Kort AM , Klijn CJM , Schreuder F , Verbeek MM ((2022) ) Prevalence of cerebral amyloid angiopathy: A systematic review and meta-analysis. Alzheimers Dement 18: , 10–28. |
[2] | van Etten ES , Gurol ME , van der Grond J , Haan J , Viswanathan A , Schwab KM , Ayres AM , Algra A , Rosand J , van Buchem MA , Terwindt GM , Greenberg SM , Wermer MJ ((2016) ) Recurrent hemorrhage risk and mortality in hereditary and sporadic cerebral amyloid angiopathy. Neurology 87: , 1482–1487. |
[3] | Vinters HV ((1987) ) Cerebral amyloid angiopathy. A critical review. Stroke 18: , 311–324. |
[4] | Greenberg SM , Charidimou A ((2018) ) Diagnosis of cerebral amyloid angiopathy: Evolution of the Boston Criteria. Stroke 49: , 491–497. |
[5] | Greenberg SM , Rebeck GW , Vonsattel JP , Gomez-Isla T , Hyman BT ((1995) ) Apolipoprotein E epsilon 4 and cerebral hemorrhage associated with amyloid angiopathy. Ann Neurol 38: , 254–259. |
[6] | Vonsattel JP , Myers RH , Hedley-Whyte ET , Ropper AH , Bird ED , Richardson EP Jr. ((1991) ) Cerebral amyloid angiopathy without and with cerebral hemorrhages: A comparative histological study. Ann Neurol 30: , 637–649. |
[7] | Mandybur TI ((1986) ) Cerebral amyloid angiopathy: The vascular pathology and complications. J Neuropathol Exp Neurol 45: , 79–90. |
[8] | Knudsen KA , Rosand J , Karluk D , Greenberg SM ((2001) ) Clinical diagnosis of cerebral amyloid angiopathy: Validation of the Boston criteria. Neurology 56: , 537–539. |
[9] | Shoamanesh A , Preis SR , Beiser AS , Vasan RS , Benjamin EJ , Kase CS , Wolf PA , DeCarli C , Romero JR , Seshadri S ((2015) ) Inflammatory biomarkers, cerebral microbleeds, and small vessel disease: Framingham Heart Study. Neurology 84: , 825–832. |
[10] | Charidimou A , Friedrich JO , Greenberg SM , Viswanathan A ((2018) ) Core cerebrospinal fluid biomarker profile in cerebral amyloid angiopathy: A meta-analysis. Neurology 90: , e754–e762. |
[11] | Schermer MHN , Richard E ((2019) ) On the reconceptualization of Alzheimer’s disease. Bioethics 33: , 138–145. |
[12] | Whitehouse PJ ((2019) ) Ethical issues in early diagnosis and prevention of Alzheimer disease. Dialogues Clin Neurosci 21: , 101–108. |
[13] | |
[14] | Tong A , Sainsbury P , Craig J ((2007) ) Consolidated criteria for reporting qualitative research (COREQ): A 32-item checklist for interviews and focus groups. Int J Qual Health Care 19: , 349–357. |
[15] | Saunders B , Sim J , Kingstone T , Baker S , Waterfield J , Bartlam B , Burroughs H , Jinks C ((2018) ) Saturation in qualitative research: Exploring its conceptualization and operationalization. Qual Quant 52: , 1893–1907. |
[16] | Hsieh HF , Shannon SE ((2005) ) Three approaches to qualitative content analysis. Qual Health Res 15: , 1277–1288. |
[17] | Welcome Trust and European Commission (2005) From Biobanks to Biomarkers. Translating the potential of human population genetics research to improve the quality of health of the EU citizen. |
[18] | Hamburg MA , Collins FS ((2010) ) The path to personalized medicine. N Engl J Med 363: , 301–304. |
[19] | National Research Council Committee on A Aging Frame work for Developing a New Taxonomy of Disease (2011) The National Academies Collection: Reports funded by National Institutes of Health. In TowardPrecision Medicine: Building a Knowledge Network for Biomedical Research and a New Taxonomy of Disease National Academies Press (US). National Academy of Sciences., Washington (DC). |
[20] | Hood L , Flores M ((2012) ) A personal view on systems medicine and the emergence of proactive P4 medicine: Predictive, preventive, personalized and participatory. N Biotechnol 29: , 613–624. |
[21] | Schleidgen S , Klingler C , Bertram T , Rogowski WH , Marckmann G ((2013) ) What is personalized medicine: Sharpening a vague term based on a systematic literature review. BMC Med Ethics 14: , 55. |
[22] | Wilson JMG, Jungner G,World Health Organization (1968) Principles and practice of screening for disease. World Health Organization.https://apps.who.int/iris/handle/10665/37650 |
[23] | Brooks M ((2009) ) Breast cancer screening and biomarkers. Methods Mol Biol 472: , 307–321. |
[24] | Mitchell AJ , Shiri-Feshki M ((2009) ) Rate of progression of mild cognitive impairment to dementia–meta-analysis of 41 robust inception cohort studies. Acta Psychiatr Scand 119: , 252–265. |
[25] | Hirth RA , Bloom BS , Chernew ME , Fendrick AM ((2000) ) Patient, physician, and payer perceptions and misperceptions of willingness to pay for diagnostic certainty. Int J Technol Assess Health Care 16: , 35–49. |
[26] | Kopits IM , Chen C , Roberts JS , Uhlmann W , Green RC ((2011) ) Willingness to pay for genetic testing for Alzheimer’s disease: A measure of personal utility. Genet Test Mol Biomarkers 15: , 871–875. |
[27] | Mühlbacher AC , Kaczynski A ((2016) ) Making good decisions in healthcare with multi-criteria decision analysis: The use, current research and future development of MCDA. Appl Health Econ Health Policy 14: , 29–40. |
[28] | Milne R , Bunnik E , Diaz A , Richard E , Badger S , Gove D , Georges J , Fauria K , Molinuevo JL , Wells K , Ritchie C , Brayne C ((2018) ) Perspectives on communicating biomarker-based assessments of Alzheimer’s disease to cognitively healthy individuals. J Alzheimers Dis 62: , 487–498. |
[29] | van der Burg S , Schreuder F , Klijn CJM , Verbeek MM ((2019) ) Valuing biomarker diagnostics for dementia care: Enhancing the reflection of patients, their care-givers and members of the wider public. Med Health Care Philos 22: , 439–451. |
[30] | Kassirer JP ((1989) ) Diagnostic reasoning. Ann Intern Med 110: , 893–900. |
[31] | Epstein RM , Franks P , Shields CG , Meldrum SC , Miller KN , Campbell TL , Fiscella K ((2005) ) Patient-centered communication and diagnostic testing. Ann Fam Med 3: , 415–421. |




