Autism – A brief update
Abstract
Autism is a group of neurodevelopmental disorders that involve definite impairments in social interactions, disturbance in language, and a stereotyped pattern of behaviour. These clinical features are described as the core symptoms. The condition represents a very large number of diseases and syndromes that are individually rare. Therefore, most people will refer to autism in the plural – autisms.
The prevalence of autism has increased incredibly in the last three decades. However, although the number of people diagnosed with autism has increased, this is not the same as saying that there is an increase in the number of cases of autism. Most likely, many children and adults 40–50 years ago had autistic behaviour that went under other diagnoses.
The cause of the autistic features has been thoroughly discussed for many years and has been the subject of many research activities. The dominant view today is that genetic and environmental factors mainly cause autism. In this article we want to give a brief status quo of the clinic, epidemiology and causes of autism.
1Introduction
Autism has been one of the most debated medical conditions through the last decades, and our understanding of this neurodevelopmental disorder has completely changed. From being perceived as a specific and rare clinical entity, autism is nowadays considered to be relatively common, both in children and in the adult population. Our understanding of the clinical manifestations, the epidemiology and the etiology of autism has also completely changed since the condition was first described. It is no exaggeration to say that this field has undergone a total paradigm shift. One of the reasons for this is the significantly increased research activity that has taken place in recent decades. Figure 1 is a bar chart showing how the number of research publications dealing with autism have developed prior to 2021. As shown in the figure, there has been a tremendous increase in the number of published articles.
The name “autism” was already mentioned in the psychiatric literature as early as in the 1911 [1]. At that time, however, no one described and systematized information about this condition, and no one considered it likely that it had something to do with a medical condition. The vast majority was convinced that it was a psychiatric condition, and many believed it was a variant of schizophrenia [1].
Fig. 1
Literature presentations from 1944-2023.
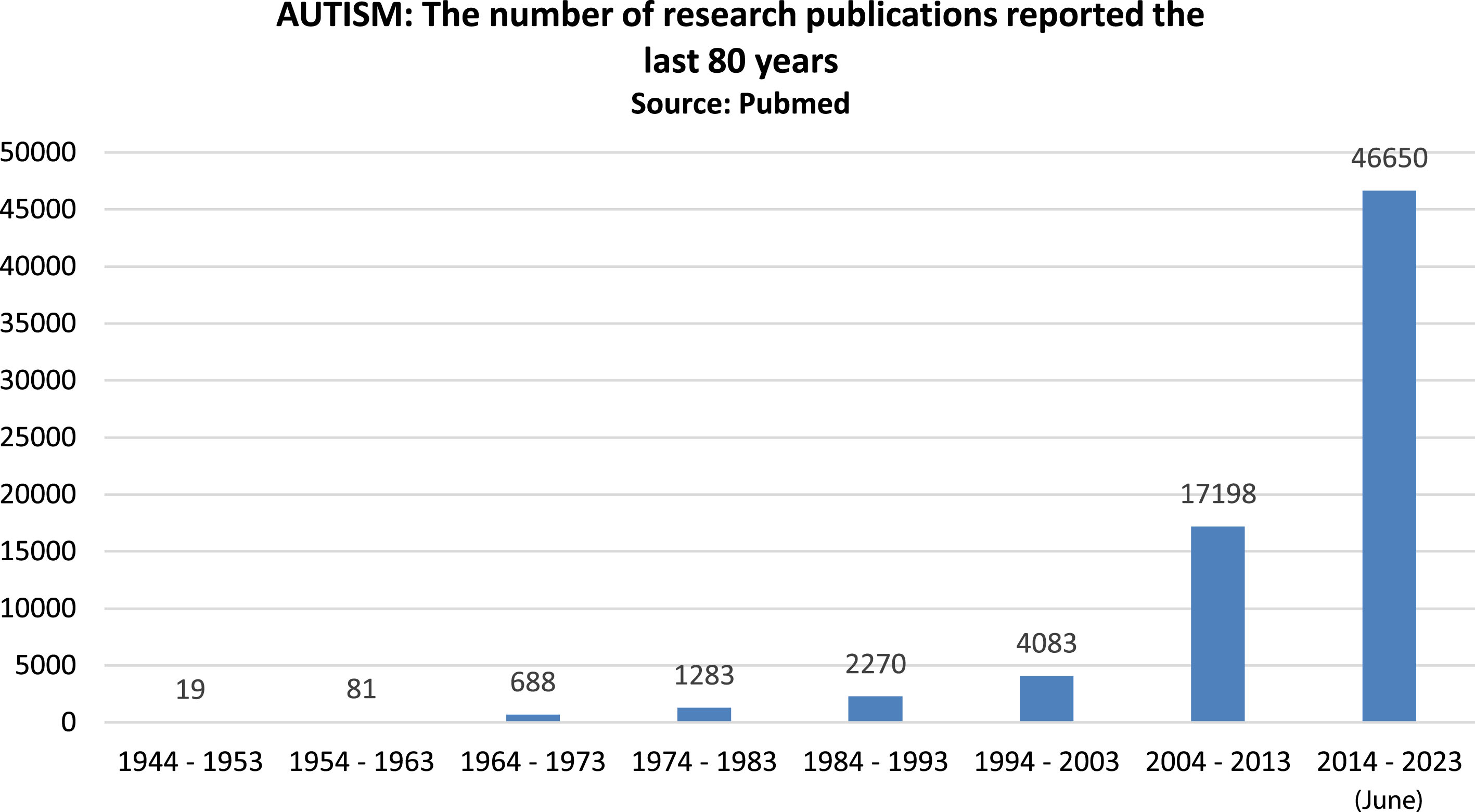
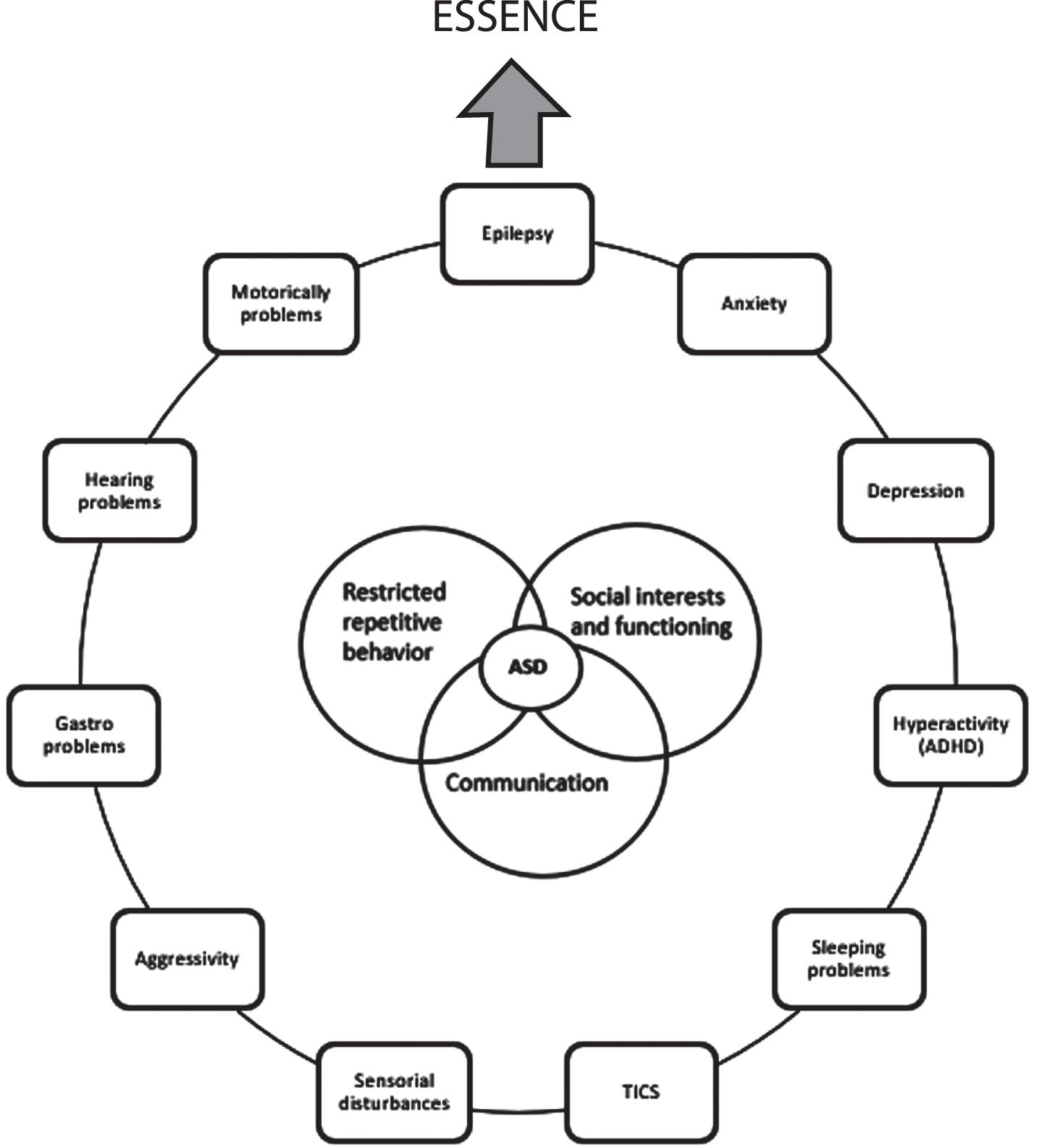
Fig. 2
The figure illustrates the core symptoms in autism and together with some of the comorbid clinical factors which can occur together with autism and its relation to the term ESSENCE.
In 1926, however, a Russian child psychiatrist, Grunya Efimovna Sukhareva, published an article describing autistic traits in six children [2]. She used the term “a schizoid psychopathy” to describe the clinical picture of autism. Interestingly, her description of the clinical picture is almost identical to the description of autism spectrum disorder in today‘s DSM-5 [3].
However, the one credited with first describing the clinical features of autism is Leo Kanner. In 1943, he published an article in a well-recognized child psychiatric journal, providing detailed symptoms and signs in eleven children with the same clinical features as Sukhareva [4]. In 1944, Kanner presented a larger patient population with the same clinical symptoms and signs [5]. This publication was intended for pediatricians.
Both articles aroused significant interest among both psychiatrists and paediatricians. All the patients were characterized as having impaired social interaction, impaired verbal and non-verbal communication, repetitive patterns of behaviour and so-called restricted interests. Kanner called the condition autism without mentioning Sukhareva‘s publication.
In 1944, an Austrian paediatrician named Hans Asperger, presented children with more or less the same clinical picture, except that they were more mildly clinically affected compared to Kanner‘s patients [6]. Among other characteristics, Asperger‘s cases had intelligence in the normal range and had good grammar and vocabulary. However, they had poor language skills and restricted interests. Many years later, the term Asperger syndrome was suggested for this condition [7]. Today, most people agree that Asperger syndrome is a variant of autism. Both these conditions have been earlier combined under the overall name of autism spectrum disorders (ASD). This article will use the name autism, meaning the whole spectrum of the autism phenotypes.
The concept of autism has evolved dramatically over the last four to five decades. Thus, both the clinical perception of autism and the research in the field have undergone major theoretical and practical changes. From being perceived as a specific clinical entity described by Kanner in 1943 [4], there has been a total paradigm shift in terms of clinical perception, epidemiological aspects, and, not least, the etiological background of autism.
In this present paper, we provide readers with a brief update on some of the clinical aspects of autism and epidemiological data and offer some thoughts about the causes that may underlie autism.
2Clinical aspects of autism
Autism is a neurobehavioral and neurodevelopmental disorder with a typical onset early in childhood. The diagnosis is classified in the International Classification and Diseases 11th Revision (ICD-11), a coding system maintained by the World Health Organization (WHO). In the ICD-11, autism is classified as a neurodevelopmental disorder and is listed under the category “Disorders of Social Interaction and Communication”.
According to ICD-11 the core symptoms of autism are characterized by persistent impairments in social communication as well as in social interaction and restricted repetitive patterns of behaviour, interests, or activities. It is still these symptoms that form the basic clinical manifestations of autism [8]. However, before these basic symptoms manifest, a substantial number of children later diagnosed with autism display mild or more serious degrees of developmental problems from very early childhood (i.e., intellectual disabilities, language impairments, motor problems) [9].
Furthermore, autism is a life-long disorder that, in youth and adulthood, often leads to impairments in different areas of life, for example in family, school, professional and the social environments [10].
In the nineteen forties, Kanner claimed that autism was one single disease, meaning that it was one single clinical entity. Clinical research, in the years following Kanner’s original publication, soon showed that this was not the case. It turned out that the core symptoms varied considerably, which gave a marked heterogeneity in the clinical picture. Furthermore, about 20 percent of the children developing autism undergo a so-called regressive development, starting very early and lasting in weeks or months. In this regressive phase, it seems as if development stops completely and that acquired functions sometimes are lost. This also occurs in some genetic syndromes associated with autism for example Rett syndrome and Angelman syndrome [9].
Secondly, the core symptoms rarely appear alone, but are usually associated with other neurological and psychiatric comorbidities, such as epilepsy, intellectual disabilities and ADHD [11–14]. Such comorbid factors are in fact the rule rather than the exception. The presence and the variation of comorbid factors also suggest a multifactorial etiology and that it involves many medical conditions. In other words, autism is not a single disease, but many.
Among those neuropsychiatric disorders associated with autism, Intellectual Developmental delay (ID), ADHD, anxiety, sleep disorders, eating problems, depression and obsessive-compulsive disorders are common. In several investigations especial ADHD has been shown to be very common in autism. In a meta-analysis done by Mutluer et al in 2022 [15] the mean prevalence of ADHD among autistic individuals was 26.2 percent, which was consistent with other older surveys. ID and anxiety disorders were reported with prevalences of 22.1 percent and 11.1 percent, respectively.
Wolff et al. [16] pointed out the heterogeneity of the intelligence quotient (IQ) reported through the years. For example, Fombone [17] reported, based on 20 epidemiological studies published between 1966 and 2001, that IQ below 70 varied from 40 to 100 percent with a mean 70 % . A more recent study [18] has reported that only 31 percent of children with autism were classified in the range of cognitive impairment (IQ < 70). Thus, epidemiological and clinical data on the IQ distribution in autism reveal that autistic individuals with intellectual disability seemed to have decreased from 70 percent down to 30 percent during the past 50 years [16].
Among the neurological comorbidities epilepsy is one of the most common conditions [19]. All types of seizures can occur, and in some children the first manifestation can be infantile spasms. Epilepsy occurs with higher rates with increasing age, and it is strongly correlated with the degree of ID. Other neurological disorders reported are cerebral palsy, motoric clumsiness and visual problems [20].
Furthermore, the concept of ESSENCE (Early Symptomatic Syndromes Eliciting Neurodevelopmental Clinical Examinations) was coined by Christopher Gillberg over ten years ago to highlight the high comorbidity and overlap between these behaviour phenotype syndromes presenting with major behavioural/cognitive/emotional problems at an early age [20, 21]
The ESSENCE concept refers to the whole group of neurodevelopmental/neuropsychiatric disorders that present with impairment symptoms in early childhood, including conditions such as intellectual disability, ADHD, autism and Tourette syndrome. Many of the ESSENCE disorders overlap with each other, creating a mosaic of symptoms that shift as the individual develops. Due to the great overlap, the presenting symptoms of autism may be outside the core autism symptom criteria. Clinical research in autism must therefore acknowledge this significant variation in phenotypic presentation and the overlap with other conditions. In a recent publication Fernell and Gillberg [20] write that “ESSENCE underscores that a first recognized symptom or identified disorder need to be followed over time in a holistic perspective, taking into account all possible developmental/behavioral problems, and not over-focusing on one specific disorder only”. Other authors share these views [22].
In summary, the present understanding of autism thus includes a spectrum of neurobiological disorders with overlapping phenotypic behaviours and with different etiological backgrounds. To acknowledge this great heterogeneity, many researchers are now talking about autisms in the plural – no longer autism in the singular.
Using the plural form of the autism emphasizes that the condition appears very differently in different people. Looking for one disorder with one cause has been fruitless, and the way forward now is to understand autism as a common pathway representing a plethora of underlying biological mechanisms. This understanding of autism and its biology will be of great importance in terms of the medical assessment and in the advice and prospects we give to parents or carers of children with autism. Furthermore, biological understanding paves the way for future targeted and precise treatment strategies [22].
3Epidemiology
Epidemiological research in autism has been in focus for many decades. A marked increase in prevalence has been seen throughout all these years, especially in the last 20–30 years. Interestingly, this increase is not limited to a specific geographical area but can be characterized as a global increase. The big question is, of course, why this increase has taken place.
Mapping and calculating the occurrence of autism is important. If we know how frequent a disease is, we can inform public policy, raise awareness, and develop research priorities [23]. It is, therefore, not surprising that epidemiological research has played such a large role. With the intention of mapping the prevalence of autism, a significant number of epidemiological studies have been published. These studies have been carried out in different populations and different age groups.
As a part of the epidemiological research, it has been a consistent feature in all investigations that have been carried out that the prevalence in males is about four to five times greater than in females [24]. However, critical voices have argued that this gender difference is over-estimated [25, 26]. A recently published article by Hervas claimed that autism is underdiagnosed in females [25]. Hervas wrote that females present with different clinical, cognitive, and biological characteristics which makes it more difficult to clarify the diagnosis. It has for many years been assumed that autism occurs more frequently in boys. Therefore, the publications from Hervas [25] and Lubinoff [26] are very interesting and should inspire further research.
If we look at the prevalence figures in more detail, it seems clear that from 1970 s until today, there has been a tremendous increase in patients diagnosed with autism. For example, in 1979, Wing and Gould found a prevalence as low as 0.04 % (4 per 10.000) [27], while Kim et al. in 2014, 35 years later, estimated the prevalence in the general population to be to around 1 % [28]. Thus, the diagnosis of autism has gone from very rare to quite common.
In the Scandinavian countries, for instance, in Norway, we have seen the same tendency. As far back as in 1998, Sponheim and Skjeldal [29] reported prevalence rates of 5.3 per 10.000, while Isaksen et al., 13 years later, found a prevalence 51 per 10.000 [30]. Particularly from 1990 onwards, the prevalence has increased, with some estimates of up to 120 per 10,000 for the whole spectrum and about 40 per 10,000 for classic childhood autism [31, 32].
Epidemiological figures from the USA have shown the most marked increase. Reports from the 2000s showed that one in every 250 children has a diagnosis of autism. Tehaconas and Adesman found the numbers even higher [33]. They claimed in 2013 that one in 88 children falls under the diagnostic criteria for autism. Furthermore, as late as in 2021, the US Center for Disease Control and Prevention (CDC) reported that one in 44 children aged 8 years was estimated to have autism [34]. However, this study pointed out large differences between the States in the USA. The highest figures were found in California. In the same study, they reported that autism was 4.2 times more prevalent among boys than among girls. The same tendency is found in several studies from European countries. In almost all European countries where these studies have been carried out, very high prevalence figures have been found compared to what was found 30 years ago.
In a recent published article by Zeidan et al. [23], the authors synthesized estimates of the prevalence of autism worldwide. They studied 71 articles published between 2012 and 2021. The median male-to-female ratio was 4.2. They found that the prevalence of autism ranged within and across regions, with a median prevalence of 100/10.000.
There has been an intense discussion on how to interpret the dramatic increase in the prevalence of autism. In this connection, it is important to point out that although the number of people diagnosed with autism has increased, this is not the same as saying that there is an increase in the number of cases of autism. The increase in prevalence figures could just as well mean that 40–50 years ago, there were many children who had autistic behaviours but who went undiagnosed or received other diagnoses. At different times, there have always been special diagnoses that have received special focus. Furthermore, part of the explanation for the increase in prevalence that has been seen in recent years may very well reflect different methodological approaches when diagnosing and selecting children with autism. This may explain the large variation seen in prevalence figures today compared to a few decades ago. In other words, the perception of which children and young people should come under the diagnostic umbrella of autism has changed. Many children with autistic features who were previously not included according to the criteria of the time, will today be diagnosed as having autism [35–37].
In this connection, it is important to emphasize that better developed welfare and health services make it easier to find children with autism. Furthermore, there are today, at least in European countries, better survey instruments which lead to more children being diagnosed.
Another important point is that it is difficult to compare studies on prevalence that took place 30–40 years ago with those taking place today. As mentioned, different methodological approaches can be one of the reasons for that. Another important thing is that one must expect that there is more knowledge and a higher awareness of the condition in the professional environment and in the general population, which also contributes to the discovery of more cases.
It also has to be mentioned that mild autistic symptoms are very frequent in the population, such symptoms often have no practical significance nor require special measures. Many people with such mild signs can be well integrated into society and the autistic traits can almost be characterized as a personality type. Some of these, however, will today be diagnosed within the autism spectrum, which probably also contributes to the increase in prevalence figures. The decisive factor, however, is whether the patients have a functional impairment or not. Functional level often varies during life, depending on whether contextual demands exceed the resources of a person with subclinical autism or not [20].
Overall, there is no doubt that there are significantly more children and young people who today fall under the diagnostic group of autism compared to investigations that were carried out before 1990. Of the various subgroups within autism, Asperger syndrome has especial shown an increase. The diagnosis of Asperger syndrome was relatively rare in the 1990 s and after that has shown to be relatively common. It is, however, important to know that both ICD-11 and DSM-5 now combine the various autism diagnoses (F84) into a single diagnosis of ‘autism spectrum disorder’ – ASD. Thus, neither Asperger syndrome, nor any of the other subgroups, will be used in the coming years. It will therefore be interesting to see how this affects the prevalence figures in the future.
Different studies have described different reasons for the increased prevalence. Lyal and Croen [38] believe that changes in the number of children who receive an autism diagnosis can always be seen in the context of the treatment available. In the same period that the increase in the number of cases of autism took place, the number of cases of children with mental retardation, without autism, decreased almost correspondingly. In any case, what we can conclude is that there are a high number of children and young people with autism who are diagnosed and thus may need extensive treatment.
4Etiology
Despite intense research efforts which have resulted in a large number of scientific publications, the biological background of autism is still poorly understood. Part of the challenge has been that autism has proven to be clinically very heterogeneous, and probably represents so many biological conditions [39. Furthermore, many of the studies are based on a very limited number of patients and poor phenotyping in larger samples 40].
Researchers have tried to define the unique or pathognomonic cause that could distinguish autism from other disorders. This concept has been completely abandoned. Although autism is undoubtedly partly a genetic condition, the etiology and pathogenesis of the autism phenotypes are to great extent, still unknown. However, our knowledge of this is growing strongly. We still have no biological marker for autism available, probably because the pathophysiological mechanisms behind autism are complex and diverse. Thus, the autism phenotypes must still be defined only by symptoms that are observed by the investigators and by asking questions of the caregivers [40, 41].
There is evidence that typical symptoms develop earlier than previously assumed. It is not rare to see autism features in the very first months of life. This contrasts with what was assumed 20–30 years ago and means that the pathophysiological mechanisms that cause autism become apparent very early in the child’s postnatal development. The pathophysiology seems to be dominated by both genetic and environmental factors involved.
4.1Neurochemical factors in autism
Many biochemical parameters have been investigated in patients with autism in the last 50 years. The reason for that is probably that the concentrations and dynamics of many neurotransmitters can affect neuron-related functions [42], and growing evidence suggesting that disturbances in the neurotransmitter system play a key role in autism [43]. Neurotransmitters have thus been the focus of research for many years.
Serotonin was one of the first neurotransmitters to be investigated in patients with autism. Serotonin is a monoamine neurotransmitter that is involved in regulating mood, anxiety and social behaviour. Studies have found that individuals with autism tend to have higher Serotonin levels compared to controls in children without autism. However, this seems to be age-depended. In small children the 5-HT synthesis is reduced during childhood but increases between the ages of 2 and 15, years reaching almost twice as much the normal adult value [44, 45]. Studies have examined platelet hyperosmolarity in autism patients. This increase appears to be unique to autism, as it has not been observed in intellectual disability or other neurodevelopmental disorders [43]. However, it is important to note that the results of these and other serotonin studies show no consistency.
Dopamine is another neurotransmitter that has been studied in autism [46]. This transmitter is involved in the regulation of movement, emotion, and motivation. Some studies have indicated altered dopamine signalling in mesocorticolimbic and nigrostriatal circuits in autism [34]. However, it is unclear what significance this has. Furthermore, studies have found that individuals with autism have alterations in the Dopamine receptors, which may contribute to the disorder‘s symptoms [47].
Gamma aminobutyric acid (GABA) is an important excitatory neurotransmitter in the developing brain. It is derived from Glutamate by the action of glutamic acid decarboxylase [43]. It has been shown that alterations in the gabaergic and glutaminergic systems can disturb the balance between the excitatory – inhibitory balance, and some believe that this could be a causative factor in autism. These changes in GABA transmitter concentrations have been associated with mutations in SHANK3 [48].
Nerve growth factors regulate the neuron survival and differentiation of the nerve cells. They play a main role as regulators for central nervous system (CNS) maturation, including that of synaptic synthesis, cellular proliferation and migration of the nerve cells. Among those, Brain-derived neurotrophic factor (BDNF) has been in focus regarding the understanding of autism. BDNF is also involved in the survival and differentiation of dopaminergic neurons in the developing brain and plays an important role in the formation and plasticity of synaptic connections. Furthermore, BDNF is also trophic for serotonergic neurons [49, 50].
The role of BDNF in autism is, however, still unclear. Many authors have found increased systemic levels of BDNF in children with autism [50], other studies report no differences compared to controls [51–53] and some report depressed levels [53].
The immune system has also been of increasing interest in autism research [54–56], and interestingly enough already Kanner implied in1957 that the immunological system could be a biological factor causing autism [57]. However, at that time, they had neither the knowledge nor the resources for follow-up.
There are several scientific reports that provide evidence that the immune system is activated in autism. However, the importance of immunological factors to the pathophysiology of autism is still uncertain. In this connection the previously mentioned possible dysfunction in the serotonergic system is interesting. Serotonin can be transformed from the essential amino acid trypthophan. However, trypthophan can also be transformed into neuroactive compounds including Kynurenic acid and Quinolinic acid. This is known as the kynurenine pathway. It has been postulated that there may be a link between inflammation or dysregulation in the immune system in autism, and enhanced kynurenine activation resulting in reduced Serotonin synthesis from Tryptophan and production of kynurenine metabolites capable of modulating glutaminergic activity [43, 56].
5Genetic factors
As early as the 1970 s, it was claimed that genetic causes play an important role in autism and in the following years the role of genetic influence in autism were increasingly debated. The current view emphasizes thus that genetic mechanisms play a particularly large role in the causal mechanisms of autism. This view is based on many twin concordance studies, the recurrence risk in families and the connection between chromosomal disorders and autism.
In 1977, Follstein and Rutter published the first twin study [58]. This study looked at the concordance in a sample of 11 monozygotic twins and 10 dizygotic twins. They found that 36 percent of the monozygotic twins were concordant while 0 percent of the dizygotic were not, which seriously suggested that autism was under a strong genetic influence. When they expanded the study to include those with a milder autism phenotype the results were even more convincing.
In the following years a substantial number of twin studies were undertaken with the same results as Follstein and Rutter [58]. Since the year 2000, thirty more twin studies have been completed. In 2016, Tick and co-workers conducted a systematic review of seven twin studies that had been published and identified a high twin heritability [59]. All in all, going through these studies, revealed a heritability range for autism from 64 to 91 percent. Furthermore, Ronald and Hoekstra went through other studies and found that the concordance was between 60 to 90 percent [60]. This is an older study, from 2011, but still it is important because it shows the same as Tick found later. Thus, the twin studies have given us strong evidence that autism is highly heritable. Although the results have varied somewhat, almost all the studies have shown a high concordance in monozygotic twins compared with dizygotic twins.
A substantial number of DNA studies have discovered genes that are involved in autism. The list of genes that can lead to the autistic phenotype is growing, and Styles et al. reported in 2020 that over 400 “Autism susceptibility genes” have been strongly linked, and 200 genes were weakly linked [61]. The contribution of each particular gene, however, within the autism population, was very minor [61]. It is, however, important to know that no gene is consistently mutated in every person with autism. Stated differently, there does not seem to be any gene that causes autism every time it is mutated [61].
Roughly speaking, we can say that two types of genetic conditions are important. The first is rare gene defects that can increase the risk of autism. This group includes a significant number of monogenic or chromosomal syndromes [62] for example Fragile X, Rett syndrome, Angelman syndrome, Tuberous sclerosis complex and Down syndrome. The group has been named by some as “syndromic autism”. The group also includes rare mutations in genes that encode synaptic proteins, such as NLGN3, NLGN4X, SHANK2 and SHANK3 [63]. Furthermore, whole-genome microarray studies have also found changes mentioned as “Copy Number Variants” (CNV). These are deletions and duplications of long stretches of DNA, and such changes can result in autism if particularly vulnerable autism genes are involved [61].
Secondly, in the group of individuals with autism and normal or high intelligence, the genetic predisposition for autism is usually due to the sum of the effects of several common gene variants, each of which has little effect. Sometimes the genetic predisposition can be explained by an interaction between rare and common genetic variants [63].
One important thing to note is that until now, no genes associated with autism result in only autism, but also involve intellectual developmental delay, epilepsy, and other comorbid factors. Put another way, so far, no specific gene or gene variant has been found that alone points to autism [63].
6Environmental risk factors
There is a growing body of research suggests that environmental factors, alongside genetic etiology, may play an important role in the development of autism. It is doubtful if one single factor is sufficient to yield autism, but probably it is so that a collection of them can be involved. In fact, there is evidence that the interactions between susceptible genes and environmental factors can be the major mechanism of autism etiology [64]. These environmental factors can include exposures that occur before, during or after birth (Table 1).
Table 1 gives an overview of those environmental factors which have, over the years, been discussed as associated with autism. It is important to note that not all the scientific works that have been done show a connection between risk factors and the development of autism. In many publications, it is not possible to demonstrate such a correlation. This is probably because there must be an interaction between environmental factors and genetic vulnerability to develop autism [64].
Here we will briefly mention the most important risk factors which have been discussed.
6.1Prenatal factors
Higher parental age has several times been reported as an important prenatal risk factor. This particularly applies to the age of the father [61, 65 –70]. Parental ages over 34 seems in many investigations to be critical. However, in some studies, this is not so conclusive [69–70].
It is worth noting that Sansafar et al. [70] claim that the risk of autism increases by 29 percent for every 10-years older the father is, and that those fathers older than 40 years have a more than two-fold greater risk of having an affected child compared to fathers aged 25–29 years [70]. When it comes to the mother’s age, we don’t have the same clear age effect, but in some publications in recent years most authors suggested that the mother’s age also has some negative effect on the development of autism in the child [61]
6.2Complications during the pregnancy
During the pregnancy infections and bleeding seems to present the greatest risk for autism in the child. Among the infections it is especially viral infections such as rubella, measles, mumps, chickenpox, influenza, herpes, varicella, and cytomegalovirus which are the biggest threats [61, 71–73]. It is, however, important to point out that many of the references that deal with a connection between viral infection in pregnancy and the later development of autism in the child are from an older date.
A substantial number of the manuscripts published about Covid and autism concerns the medical and psychosocial consequences of undergoing Covid 19 for people with autism. However, as far as we know, there is no evidence that Covid-19 infection is an etiological factor for autism. Such investigations have been called for [74].
Table 1
The able shows some environmental risk factors which have been claimed associated with autistic phenotype (modified after 57)
| SOME PRENATAL RISK FACTORS |
| Parental age |
| Birth order |
| Preeclampsia |
| Maternal diabetes |
| Maternal bleeding during pregnancy |
| Bacterial/viral infections during pregnancy |
| Prenatal medication use (antiepileptic drugs; Psychotropic drugs, Antidepressant drugs; Misoprostol; Thalidomide; Pesticides; air pollutants) |
| Hypertension in mother |
| Familial socio-economic status |
| Maternal iron, folate, and methionine deficiency |
| Weight gain during pregnancy |
| Threatened abortion |
| SOME NATAL RISK FACTORS |
| Preterm birth |
| Postterm birth |
| Cesaren delivery |
| Fetal complications |
| Asphyxia |
| Congenital anomalies |
| Fetal respiratory distress |
| Season of birth |
| SOME POSTNATAL RISK FACTORS |
| Low birth weight |
| Jaundice and hyperbilirubinemi |
| Birth defects |
| Early infant infections |
| Postnatal anemia |
| Encephalopathy |
| Postnatal vitamin D deficiency |
Metabolic syndromes such as diabetes, hypertension and obesity during the pregnancy have been reported to be risk factors for the child to develop autism [66, 75–78]. The reason that such maternal metabolic syndromes are a risk factor for autism is claimed to be due to cerebral hypoxia in utero with subsequent damage to the neurons [75].
Different kinds of prenatal medications have been put in context with increased risk of autism. This applies to prenatal use of psychiatric medications. Those drugs easily pass the placenta [66]. Other drugs that have been associated with autism are antiepileptic drugs, especially valproate, which can give a so-called valproate syndrome that leads to developmental delays, deficient motor activities and autistic behaviour [79, 80–81]. Different kinds of analgesics, for example paracetamol, have also been reported to cause autism [82]. The use of antidepressant medications in pregnancy has been claimed to be causing autism [83–85].
6.3Perinatal and postnatal factors
Abnormal gestational age, preterm (less than 35 weeks) and post-term pregnancy have been reported to increase the risk of autism [86–88]. Bleeding during pregnancy, umbilical-cord complications and caesarean delivery are associated with higher risk of autism possibly during cerebral hypoxia in the fetus [43, 61, 64, 77, 88].
Many postnatal risk factors have been associated with the development of autism, most of them are mentioned in table 1. Children who have had low birth weight and/or postnatal infections are presumably most vulnerable [64]. Postnatal jaundice has also been claimed to be a risk of autism and other mental disorders [43]. Further studies are needed to fully understand the associations between these environmental exposures and autism, as well as the mechanisms by which they might affect the development of the disorder. Furthermore, most environmental factors that increase the risk of autism do so in relatively small amounts and are found in combination with genetic factors that also play a role.
7Conclusions
Autism, meaning autism spectrum disorders (ASD), are a diverse group of disorders. They are characterized by difficulty in social interaction and communication and atypical patterns of activities and behaviours. Most often children and adults with autism have co-occurring neurological and psychiatric conditions, for example epilepsy, depression, and ADHD. The level of intellectual functioning varies. However, recent research claims that about 1/3 of the autistic individuals suffer from intellectual developmental delay.
The prevalence of autism has increased significantly through the last decades. The current data show mean prevalence figures of 1 to 1.5%, however, the this varies substantial across studies. Some studies have reported figures that are higher. In this article we discuss why the prevalence figures have increased so much.
The cause of autism seems to be multifactorial. Genetic mechanisms play a particularly large role as a cause of autism, and a substantial number of DNA studies have discovered a polygenic risk factor in autism. The last updates show that several hundreds of genes are linked to autism. A huge number of environmental factors, both prenatal, perinatal and postnatal factors are reported to be risk factors for autism. Interactions between susceptible genes and environmental factors can be the major mechanism of autism aetiology.
References
[1] | Bleuler E. The theory of schizophrenic negativism, J Nerv Ment Dis (WA White, Trans) 39: ((1912) ), 50–57. |
[2] | Manouilenko I. , Bejerot S. Sukhareva-Prior to Asperger and Kanner, Nord J Psychatry ((2015) ). DOI: 10.3109/08039.2015.1005022 |
[3] | American Psychiatric Association, Diagnostic and statistical manual of mental disorders, 5th edition, Arlington, VA:APA; 2013. |
[4] | Kanner L.J.N. Autistic disturbances of affective contact, Nervous Child 2: (3)((1943) ), 217–250. |
[5] | Kanner L. Early infantile Autism, The Journal of Pediatrics 25: ((1944) ), 211–217. |
[6] | Asperger H. Die Autistichen Psychopathien im Kindesalter, Archiv für Psychiatrie und Nervenkrankheiten 117: ((1944) ), 76–136. |
[7] | Wing L. Asperger‘s syndrome: a Clinical account, Psychological Medicine, Cambridge University Press 11: (1)((1981) ), 115–129. |
[8] | Lai M.C. , Lombardo M.V. and Baron-Cohen S. Autism, Lancet 383: (9920)((2014) ) 896–910. doi: 10.1016/S0140-6736(13)61539-1 |
[9] | Coleman M. and Gillberg C. The Autism, Oxford University Press ((2012) ). Doi: 10.1093/med/978199732128.00/0001. |
[10] | Roy M. and Strate P. Autism spectrum disorders in adulthood – symptoms, diagnosis and treatment, Dtsch Arztebl Int ((2023) ) 120: , 87–93 DOI: 10.3238/arztebl.m2022.0379 |
[11] | Canitano R. Epilepsy in autism spectrum disorders, Eur Child Adolesc Psychiatry 16: (1) ((2007) ), 61–66. doi: 10.1007/s00787-006-0563-2 |
[12] | Gillberg C. , Fernell E. Autism plus versus autism pure, J Autism Dev Disord 44: (12) ((2014) ), 3274–3276. doi: 10.1007/s10803-014-2163-1 |
[13] | Gjevik E. , Eldevik S. , Fjaeran-Granum T. and Sponheim E. Kiddie-SADS reveals high rates of DSM-IV disorders in children and adolescents with autism spectrum disorders, J Autism Dev Disord 41: (6) ((2011) ), 761–769. doi: 10.1007/s10803-010-1095-7 |
[14] | Sundelin H.E. , Larsson H. , Lichtenstein P. , Almqvist C. , Hultman C.M. , Tomson T. , Ludvigsson J.F. Autism and epilepsy: A population-based nationwide cohort study, Neurology 87: (2) ((2016) ), 192–197. doi: 10.1212/WNL.0000000000002836 |
[15] | Mutluer T. , Aslan Genc H. Özcan Morey,A. Yapici Eser,H. Ertinmaz B. and Can, M. . Population based psychiatric comorbidity in children and adolesecents with autism spectrum disorders: a meta-analysis, Front Psychiatry ((2022) ), 13.856208. DOI: 10.3389/fpsyt.2022.856208 |
[16] | Wolff N. , Stroth S. , Kamp-Becker I. , Roepke S. , Roessner V. Autism Spectrum Disorder and IQ – A complex interplay, Front Psychiatry ((2022) ). DOI: 10.3389/fpsyt.2022.856084 |
[17] | Fombonne E. Epidemiological surveys of autism and other pervasive developmental disorders: an update, J Autism Dev Disord 33: ((2003) ), 365–382. DOI: 10.1023/a:1025054610557 |
[18] | Baio J. , Wiggins L. , Christensen D.L. , Maenner M.J. , Daniels J. Prevalence of autism spectrum disorder among children aged 8 years – autism and developmental disabilities monitoring network, MMWR Surveillance Summary 67: ((2014) ), 1–23. |
[19] | Tuchman R. What is the relationship between autism spectrum disorders and epilepsy? Semin Pediatr Neurol 24: ((2017) ), 292–300. DOI: 10.1016/j.spen.2017.10.004 |
[20] | Fernell E. , Gillberg C. Autism under the umbrella of ESSENCE, Front in Psychiatry ((2023) ). DOI: 10.3389/fpsyt.2023.1002228 |
[21] | Gillberg C. The ESSENCE in child psychiatry: Early Symptomatic Syndromes Eliciting Neurodevelopmental Clinical Examinations, Res Dev Disabil 31: (6)((2010) ), 1543–1551. doi: 10.1016/j.ridd.2010.06 |
[22] | Eissa N. , Al-Houqani M. , Sadeq A. , Ojha S.K. , Sasse A. , Sadek B. Current Enlightenment About Etiology and Pharmacological Treatment of Autism Spectrum Disorder, Front Neurosci 12: ((2018) ), 304. doi:10.3389/fnins.2018.00304 |
[23] | Zeidan J. , Fombonne E. , Scorah J. , Ibrahim A. , Durkin M.S. , Saxena S. , Yusuf A. , Shih a. Global p 41: revalence of autism: A systematic review update, Aut Research 15: ((2022) ), 778–790. DOI: 10.1002/aur.2696 |
[24] | Baio J. , Wiggins L. , Christensen D.L. , Maenner M.J. , Daniels J. , Warren Z. Prevlance of autism spectrum disorder among children aged 8 years – autism and developmental disabilities monitoring network, 11 sites, United States, 2014 MMWR Surveill Summ 67: ((2018) ), 1. DOI: 10.15585/mmwr.ss6706a1 |
[25] | Hervàs A. Female gender and autism: under detection and my diagnoses, Medicina (B Aires) 82: (Suppl 1) ((2002) ), 37–42. |
[26] | Ochoa-Lubinoff C. , Bridget A. , Emily M. , Dillon F. Autism in Women, Neurol Clin 41: ((2023) ), 381–397. |
[27] | Wing L. , Gould J. Severe impairments of social interaction and associated abnormalities in children: epidemiology and classification, J Autism Dev Disord ((1979) ) 9: (1), 11–29. doi: 10.1007/BF01531288 |
[28] | Kim Y.S. , Fombonne E. , Koh Y.J. , Kim S.J. , Cheon K.A. , Leventhal B.L. A comparison of DSM-IV pervasive developmental disorder and DSM-5 autism spectrum disorder prevalence in an epidemiologic sample, J Am Acad Child Adolesc Psychiatry 53: (5) ((2014) ), 500–508. doi: 10.1016/j.jaac.2013.12.021 |
[29] | Sponheim E. , Skjeldal O. Autism and related disorders: epidemiological findings in a Norwegian study using ICD-10 diagnostic criteria, J Autism Dev Disord ((1998) ) 28: (3), 217–227. |
[30] | Isaksen J. , Diseth T.H. , Schjølberg S. and Skjeldal, O.H. Observed prevalence of autism spectrum disorders in two Norwegian counties, Eur J Paediatr Neurol ((2012) )1–7. doi: 10.1016/j.ejpn.2012.01.014 |
[31] | Baird G. , Simonoff E. , Pickles A. , Chandler S. , Loucas T. , Meldrum D. , Charman T. Prevalence of disorders of autism spectrum in a population cohort of children in South Thames: the Special Need and Autism Project (SNAP), Lancet 368: (9531)((2006) ), 210–215. doi: 10.1016/S0140-6736(06)69041-7 |
[32] | Gillberg C. , Wing L. Autism not an extremly rare disorder, Acta Psychiatrica Scandinavica ((1999) ) 99: (6), 399–406. |
[33] | Tchaconas A. , Adesman A. Autism spectrum disorders: a pediatric overview and update, Curr Opin Pediatr 25: (1)((2013) ), 130–144 |
[34] | Maenner M.J. et al., Prevalence and characteristics of Autism Spectrum Disorder among children aged 8 years – Autism and Developmental Disabilities Monitoring Network, 11 Sites, United states, 2018. MMWR Surveill Summ 70: (11)((2021) ), 1–16. DOI: 10.15585/mmwr.ss7011a1. |
[35] | Posserud M.B. , Lundervold A.J. , Gillberg C. Autistic features in a total population of 7– 9-year-old children assessed by the ASSQ (Autism Spectrum Screening Questionnaire), J Child Psychol Psychiatry 47: (2)((2006) ), 167–175 |
[36] | Posserud M.B. , Lundervold A.J. , Lie S.A. , Gillberg C. The prevalence of autism spectrum disorders: impact of diagnostic instrument and non-response bias, Soc Psychiatry Psychiatr Epidemiol 45: (3)((2010) ), 319–327. |
[37] | Isaksen J. , Diseth T.H. , Schølberg S. and Skjeldal, O.H. Autism spectrum disorders-are they really epidemic? Eur J Paediatr Neurol ((2013) ) DOI: 10.1016/j.ejpn.2013.03.003 |
[38] | Lyall K. , Croen L. , Daniels J. The changing Epidemiology of Autism Spectrum Disorders, Annu Rev Public Health 38: ((2017) ), 81–102. doi: 10.1146/annurev-publhealth-031816-044318. |
[39] | Eisa N. , Al-Houqani M. , Sadeq A. , Ojha S.K. , Sasse A. , Sadek B. Current Enlightenment about etiology and Pharmacological treatment of autism spectrum disorder, Front Neurosci 12: ((2018) ), 304 Doi: 10.3389/fnins.s018.00304 |
[40] | Cai N. , Revez J.A. , Adams M.J. , Andlauer T.F.M. , Breen G. , Byrne E. Minimal phenotyping yields genome-wide association signals of low specificity for major depression, Nature Genetics 52: (4)((2020) ), 437–447. doi: 10.1038/s41588-020-0594-5 |
[41] | Loth E. , Charman T. , Mason L. , Tillmann J. , Jones E.J.H. , Wooldridge C. , Buitelaar J.K. The EU-AIMS Longitudinal European Autism Project (LEAP): design and methodologies to identify and validate stratification biomarkers for autism spectrum disorders, Mol Autism 8: ((2017) ), 24. doi: 10.1186/s13229-017-0146-8 |
[42] | Fernandez M. , Mollinedo-Gajate I. , Penagarikano O. Neural Circuits for Social Cognition: Implications for autism, Neuroscience 370: ((2018) ), 148–162. |
[43] | L. Wang, B. Wang, C. Wu, J. Wang and M. Sun, Autism spectrum disorders: Neurodevelopmental Risk Factors, Biological Mechanism, and Precision Therapy, Int J Mol Sci ((2023) ) https://doi.org/103390/ijms24031819 |
[44] | Tan Z. , Wei H. , Song X. , Mai W. , Yan J. , Ye W. , Ling X. , Hou L. , Zhang S. , Yan S. Positron Emission Tomography in the Neuroimaging of Autism Spectrum Disorder: A review, Front Neurosci 16: ((2022) ), 806876. |
[45] | Liu Z. , Wang J. Xu,Q Hong,Q. Zhu J. and Chi,X. Research progress in Vitamin A and Autism spectrum diosorders, Behav Neurol ((2021) ) 2021: , 5417497 |
[46] | Mandic-Maravic V. , Grujicic R. , Milutinovic L. , Munjiza-Jovanov A.M. , Pejovic-Milovancevic M.P. Dopamine in Autism spectrum disorders – Focus on D2/D3 Partial agonists and their possible use in treatment, Front Psychiatry ((2022) ). doi. org/10.3389/fpsyt.2021.787097 |
[47] | Mandic-Maravic V. , Grujicic R. , Milutinovic L. , Munjiza-Jovanov A.M. , Pejovic-Milovancevic M.P. Dopamine in Autism spectrum disorders – Focus on D2/D3 Partial agonists and their possible use in treatment, Front Psychiatry ((2022) ). doi.org/10.3389/fpsyt.2021.787097. |
[48] | Horder J. , Petrinovic M.M. , Mendez M.A. , Bruns A. , Takumi T. , Spooren W. , Barker G.J. , Kunnecke B. , Murphy D.G. Glutamate and GABA in autism spectrum disorder- a translational magnetic resonance spectroscopy study in man and rodent models, Transl Psychiatry ((2018) ) 8: , 106. |
[49] | Binder D.K. , Scharfman H.E. Brain-derived neurotrophic factor, Grow Factors 22: (3)((2004) ), 123–131. |
[50] | Bryn V. , Halvorsen B. , Ueland T. , Isaksen J. , Kolkova K. , Ravn K. , Skjeldal O.H. Brain derived neurotrophic factor (BDNF) and autism spectrum disorders (ASD) in childhood, J Eur Paediatr Neurol 19: ((2015) ), 411–414. http://dx.org/10.1016/j.ejpn.2015.03.005 |
[51] | Mansour M. , Mohamed A. , Azam H. , Henedy M. Brain derived neurotrophic factor in autism, Curr Psychiatry 17: (1)((2010) ), 23–29. |
[52] | Nelson P.G. , Kudo T. , Song E.Y. Selected neurotrophins, neuropeptides and cytokines: developmental trajectory and concentrations in neonatal blood of children with autism or Down syndrome, Int J Dev Neurosci 24: (1)((2006) ), 73–80. |
[53] | Bryn V. , Dalsbotten Aass, H. Skjeldal, O.H. Isaksen,J. Saugstad O.D. and Ormstad,H. Cytokine Profile in Autism Spectrum Disorders in Children, J Mol Neurosci 61: ((2017) ), 1–7. doi: 10.1007/212031-016-0847-z |
[54] | Mead J. , Ashwood P. Evidence supporting an altered immune response in ASD, Immunol Lett 163: ((2015) ), 49–55. |
[55] | Gesundheit B. Immunological and autoimmune considerations of autism spectrum disorders, J Autoimmun 44: ((2013) ), 1–7. |
[56] | Bryn V. , Verkerk R. , Skjeldal O.H. , Saugstad O.D. , Ormstad H. Kynurenine pathway in Autism Spectrum Disorders in Children, Neuropsychobiology ((2017) ) doi: 10.1159/000488157 |
[57] | Kanner L. , Eisenberg G.L. Childhood schizophrenia symposium, VI Early infantile autism 1943–55, The American journal of Orthopsychatry 26: ((1956) ), 556–566. |
[58] | Follstein S. , Rutter M. Infantile autism: a genetic study of 21 twin pairs, J of Child Psyhology and Psychiatry 18: ((1977) ), 297–321. doi: 10.1111/j.1469-7610.1977.tb00443.x |
[59] | Tick B. , Bolton P. , Happé F. Rutter F, M. and Rijsdijk F. , Heritability of autism spectrum disorders: a meta-analysis of twin studies, Child Psychol and Psychiatr 57: (5)((2016) ), 585–595. |
[60] | Ronald A. , Hoekstra R.A. Autism spectrum disorders and autistic tratits: a decade of new twin studies, Am J Med Genet B Neuropsychiatr Genet 156: (3)((2011) ), 255–274. |
[61] | Styles M. , Alsharshani D. , Samara M. , Alsharshani M. , Khattab A. , Qoronfleh M.W. Risk factors, diagnosis, prognosis and treatment of autism, IMR Press FBL ((2020) )DOI: 10.2741/4873 |
[62] | Lord C. , Brugha T.S. , Charman T. Autism spectrum disorder, Nat Rev Dis Primers 6: ((2020) ), 5. |
[63] | Grove J. , Ripke S. , T.D. Identification of common genetic risk variants for autism spectrum disorder, Nat Genetic ((2019) ) 51: , 431–444. |
[64] | Karimi P. , Kamali E. , Mohammad S. , Karahmadi M. Enviromental factors influencing the risk of autism, J Res Med Sci 22: ((2017) ), 22–27. |
[65] | Durkin M.S. , Maenner M.J. , Newschaffer C.J. , Lee L.C. , Cunniff C.M. , Daniels J.L. Advanced parental age and the risk of autism spectrum disorder, Am J Epidemiol 168: ((2008) ), 1268–1276. |
[66] | Gardener H. , Speigelman D. , Buka S.L. Prenatal risk factors for autism: Comprehensive meta-analysis, Br J Psychiatry 195: ((2009) ), 7–14. |
[67] | Lee B.K. , McGrath J.J. Advancing parental age and autism:Multifactorial pathways, Trends Mol Med 21: ((2015) ), 118–125. |
[68] | Parner,E.T. Baron-Cohen,S. Lauritsen,M.B. Jørgensen,M. Schieve L.A. and Yeargin-Allsopp,M. Parental age and autism spectrum disorders, Ann Epidemiol 22: ((2010) ), 143–150. |
[69] | Karimi P. , Kamali E. , Mohammad S. , Karahmadi M. Enviromental factors influencing the risk of autism, J Res Med Sci 22: ((2017) ), 22–27. |
[70] | Sasanfar R. , Haddad S. , Tolouei A. , Santagelos S. Paternal age increases the risk for autism in an Iranian population sample, Mol Autism 112: ((2010) ). DOI: 10.1186/2040-2392-1-2 |
[71] | Libbey J. , Sweeten T. , McMahon W. , Fujinami R. Autistic disorder and viral infections, J Neurovirol ((2005) ). DOI: 1080/13550280590900553 |
[72] | London E. The enviroment as an etiologic factor in autism: a new directionfor research, Environ Health Perspect 108: (Suppl 3) ((2000) ), 401. DOI: 10.1289/ehp 00108s3401 |
[73] | Yamashita Y. , Fujimoto C. Possible association between congenital cytomegalovirus infection and autistic disorder, J Autism Dev Disord ((2003) ). DOI: 10.1023/A:1025023131029 |
[74] | Steinman,G. COVID-19 and autism, Med Hypotheses 142: ((2020) ). DOI: 10.1016/j.mehy.2020.109797 |
[75] | Eidelman A. , Samueloff A. The pathophysiology of the fetus of the diabetic mother, Semi Perinatol ((2002) ). DOI: 10.1053/sper.2002.34215 |
[76] | Krakowiak P. , Walker C. Bremer,A Baker,A. Ozonoff, S. Hansen R. and Hertz-Picciottol,L. Maternell Metabolic Conditions and Risk for Autism and Other Neurodevelopmental Disorders, Pediatrics ((2012) ). DOI: 10.1542/peds.2011-2583 |
[77] | Hultman C.M. , Sparén P. and Cnattingius, S. Perinatal risk factors for infantile autism, Epidemiology 13: ((2002) ), 417–423. |
[78] | Guinchat V. , Thorsen P.L. , Laurent C.C. , Bodeau N.C. , Cohen D. Pre-, peri- and neontala risk factor for autism, Acta Obstet Gynecol Scan ((2012) ). DOI: 10.1111/j.1600-0412.2011.01325.x |
[79] | Ornoy A. , Weinstein-Fudim L. , Ergaz Z.P. Prenatal factors associated with autsm spectrum disorders (ASD), Reprod Txicol ((2015) ). DOI: 10.1016/j.reprotox.2015.05.007 |
[80] | Narita M. , Oyabu A.I. , Mura Y.K. , Kamada N.Y. , Okoyama T.T. , Tano K.U. , Uchida A.N. , Arita N.N. Exploratory movement and behavioral alterations in a thalidomide or valproic acid-induced autism model rat, Neurosci Res ((2010) ) DOI: 10.1016/j.neures.2009.09.001 |
[81] | Ornoy A. Valproic acid in pregnancy: how much are we endangering the embryo and fetus? Reprod Toxicol ((2009) ) DOI: 10.1016/j.reprotox.2009.02.014 |
[82] | Bauer A.Z. , Kriebel D.P. Prenatal and perinatal analgesic exposure and autism: an ecological link, Environ Health ((2013) ). DOI: 10.1186/1476-069X-12-41 |
[83] | Gidaya N.B. , Lee B.K. , Burstyn I. , Yudell M. , Mortensen E.L. , Newschaffer C.J. In utero exposure to selective serotonin reuptake inhibitors and risk for autism spectrum disorder, J Autism Dev Disord ((2014) ). DOI: 10.1007/s10803-014-2128-4 |
[84] | Harrington R. , Lee L. , Crum R. , Zimmerman A. , Hertz-Picciotto I. Prenatal SSRI use and offspring with autism spectrum disorder or developmental delay, Pediatrics ((2014) ). DOI: 10.1542/peds.2013-3406 |
[85] | Rai D. Lee, B. Dalman,C. Golding, J. Lewis G. and Magnusson,C. Parental depression, maternal antidepressant use during pregnancy, and risk of autism spectrum disorders: population based case-control study, BMJ ((2013) ). DOI: 10.1136/bmj.f2059 . |
[86] | Larsson H.J. , Eaton W.W. Madsen,K.M. Vestergaard,M. Olesen A.V. and Agerbo, E. Risk factors forautism: Perinatal factors, parental psychiatrichistory, and socioeconomic status, Am J Epidemiol 161: ((2005) ), 916–925. |
[87] | Zhang X. , Lv C.C. , Tian J. , Miao R.J. , cXi W. , and Hertz-Picciotto I. Prenatal and perinatal risk factors for autismin China, J Autism Dev Disord 40: ((2010) ), 1311–1321. |
[88] | Gardener H. , Speigelman D. , Buka S.L. Perinatal and neonatal risk factors for autism: A comprehensive meta-analysis, Pediatrics 128: ((2011) ), 344–355. |




