Early rehabilitation treatment in hospitalized patients with severe COVID-19: Effects on autonomy and quality of life
Abstract
PURPOSE:
The aim of our study is to evaluate effects of early rehabilitation treatment in hospitalized patients with severe COVID-19, in order to improve patients’ autonomy and quality of life.
METHODS:
This observational study has been performed in hospitalized patients with severe COVID-19. All patients were evaluated at T0 using specific scales: Modified Barthel Index (MBI) for autonomy in ADL, Mini Mental State Examination (MMSE) for cognitive status, Borg scale for dyspnoea, EQ5D scale for quality of life. In absence of contraindications for the rehabilitation treatment, patients start early a rehabilitation protocol consisting of one session (30 minutes) per day, for 2 to 3 weeks; these scales have been repeated at patient’s demission (T1).
RESULTS:
70 patients (37 women and 33 men, with average age of 71 years) with severe COVID-19 were included in the study. After rehabilitation treatment, MBI increases statistically significantly from T0 to T1 (39.8±35.0 with 95% CI 31.6–48, vs 69.8±38.1 with 95% CI 60.8–78.7, p < 0.001); besides MBI at T0 correlates inversely and statistically significantly with all EQ-5D variables at T0, similarly at T1 (p < 0.001), indicating the improvement of autonomy and therefore of the quality of life. The MMSE correlates statistically significantly with MBI at T0 and T1 (r = 0.569, r = 0.747 respectively, p < 0.001), indicating that an adequate cognitive status is connected with a greater increase in autonomy in ADL after rehabilitation treatment. MBI correlates directly and significantly with the PaO2/FiO2 value both at T0 and T1 (r = 0.263 with p = 0.039, r = 0.389 with p = 0.023 respectively), indicating that improving the oxygen exchanges also improves the patient’s autonomy.
CONCLUSIONS:
An early rehabilitation treatment should promote autonomy and a better quality of life in patients with COVID-19.
1Introduction
Coronavirus disease (COVID-19) is an infectious and contagious disease, of viral origin, caused by a Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) [1, 2]. COVID-19 can show a range of clinical manifestations, including cough, fever, myalgias, gastrointestinal symptoms, neurological manifestations [1–3]. Diagnosis of COVID-19 is commonly made through detection of SARS-CoV-2 RNA by PCR testing of a nasopharyngeal swab or other specimens, including saliva [4]. Most patients are asymptomatic or develop mild symptoms without requiring special treatment; however, frail patients with other comorbidities (such as cardiovascular disease, diabetes, chronic respiratory disease, obesity, cancer, immunosuppressive conditions) [5] may develop severe illness (including severe respiratory failure with acute respiratory distress syndrome (ARDS) and multi-organ failure), associated to secondary damages due to prolonged bed rest, muscle weakness, motor deconditioning, postural impairment, joint stiffness, pain, fatigue, immobilization syndrome [5]. In these patients a multidisciplinary assessment, with early respiratory and neuromuscular rehabilitation treatment, is absolutely necessary in order to guarantee a rapid functional recovery and limit the damage caused by prolonged bed rest [6–10].
COVID-19 can be classified, as reported by Ghandi et al. [11] in:
• Asymptomatic,
• Mild (mild symptoms including fever, cough or change in taste or smell, no dyspnea; requiring clinical monitoring and supportive care),
• Moderate (clinical or radiographic evidence of lower respiratory tract disease; oxygen saturation ≥94, requiring clinical monitoring and sometimes hospitalization),
• Severe (Oxygen saturation < 94%; respiratory rate ≥30 breaths/min; lung infiltrates > 50%, requiring hospitalization, oxygen and specific therapies),
• Critical (Respiratory failure, shock and multiorgan dysfunction or failure, requiring critical care and specific therapy).
As reported in literature, in severe and critically ill patients, rehabilitation treatment should begin early, encouraging, as soon as possible, physical activity along with posture and breathing management [12]. Many studies reported that rehabilitation in the acute phase would be helpful to mitigate sequelae of COVID, suggesting that early mobilization improves respiratory function and maintains oxygen saturation levels [12–18].
The aim of our study is to show our experience of early rehabilitation treatment in hospitalized patients with severe COVID-19, and its effects in order to improve the autonomy of patients and their quality of life (QoL).
2Methods
131 patients with COVID-19 diagnosis, confirmed by PCR testing (nasopharyngeal swab), were hospitalized in non-intensive care unit in the period between January and May 2021. All patients at the entrance (TO) received clinical-instrumental examinations such as Chest CT, electrocardiogram (ECG) and echocardiogram, hemogasanalysis, laboratory tests including routine blood chemistry, d-dimer, CRP (C Reactive Protein), vitamin D assay and other specific exams considering patient’s pathologies. All patients were treated with low molecular weight heparin, cortisone therapies, oxygen therapy (using nasal cannulae, Venturi mask or, if necessary, CPAP/BiPAP mode ventilation), antiviral therapy (remdesevir), and with other personalized therapies depending on individual clinical conditions including antibiotic therapy.
In all patients the Physiatrist, assisted by the ward Internist, performed:
• MMSE (Mini Mental State Examination) for a basic cognitive assessment,
• MBI (Modified Barthel Index) for autonomy in Activities Daily Living (ADL),
• Borg scale for dyspnoea,
• EQ-5D scale (European Quality of Life Five Dimension) for quality of life.
These scales were repeated at patient’s demission (T1).
MBI [19] is divided into 5 categories: 1) 0 –24 points (total dependence), 2) 25 –49 points (severe dependence), 3) 50 –74 points (moderate dependence), 4) 79 –90 points (mild dependence) and 5) 91 –99 points (minimum dependence). Borg scale for dyspnoea [20] ranged from 0 to 10, 0 indicating no symptoms and 10 severe dyspnoea symptoms. EQ-5D scale includes 5 parameters (mobility, self-care, usual activities, pain/discomfort, anxiety/depression), each evaluated in three levels (nothing, moderate and severe problems for each parameter) [21].
Rehabilitation treatment includes mobilization exercises, muscle strengthening, reconditioning to sitting position, postural passages, walking training, breathing exercises. The early rehabilitation protocol consists of one session (30 minutes) per day, with increasing intensity exercises based on the characteristics of the patient, for 2 to 3 weeks, adapted for each patient (according to the ventilatory support, general clinical condition, patient’s days of hospitalization and recovery of his autonomy). The rehabilitation treatment is interrupted if the clinical picture worsens (instability of internal parameters); generally, it begins within 2–3 days after hospitalization.
2.1Inclusion criteria to the rehabilitation treatment
• patients with severe COVID (Oxygen saturation < 94%; respiratory rate ≥30 breaths/min; lung infiltrates > 50%, requiring hospitalization, oxygen and specific therapies);
• cardio-respiratory stability condition (heart rate < 100 bpm and > 40 bpm; blood pressure < 180 mm/Hg and > 90 mm/Hg; P/F (Pa02/Fi02)* > 200);
• Glasgow Coma Scale > 9;
• body temperature < 37°C;
• adequate level of collaboration.
2.2Exclusion criteria to rehabilitation treatment
• Pneumonia in the acute phase with respiratory failure (spo2 < 90%, P/F < 200),
• Acute / subacute ischemic heart disease (heart attack within the last three months),
• uncontrolled arrhythmias (such as high-response ventricular fibrillation),
• uncontrolled hypertension,
• Acute / subacute heart failure,
• Acute progressive liver disease or renal failure,
• Acute / subacute cerebral ischemia with a state of restlessness and psychomotor agitation.
Vitamin D. In patients who had a vitamin D value < 20 ng/ml, an attack protocol with cholecalciferol 50,000 IU/day was performed for 7 days, at the end of which it was re-evaluated; if it was still deficient, the therapy was continued for 7 more days.
This observational study was performed in accordance with the Strengthening The Reporting Of Observational Studies In Epidemiology (Strobe) Guidelines. All patients gave their written informed consent to the rehabilitation treatment, and all authors were instructed to protect the privacy and follow the study procedures according to Helsinki Declaration; the study was approved by the Internal Review Board. This observational study was not registered on Clinical Trials Registry, because it is not an intervention trial, and reflects normal clinical practice.
SPSS program was used to analyze statistical data; t-Student test was used to evaluate data pre and post rehabilitation treatment; Pearson’s correlation was used for data correlation; p value < 0.05 was considered as statistically significant.
3Results
According to inclusion criteria, 82/131 hospitalized patients were included in our study. Of these 82, 10 patients got worse and died and 2 were intubated and transferred to another department; therefore, 70 patients were included in the study (see flow-chart).
Flow chart
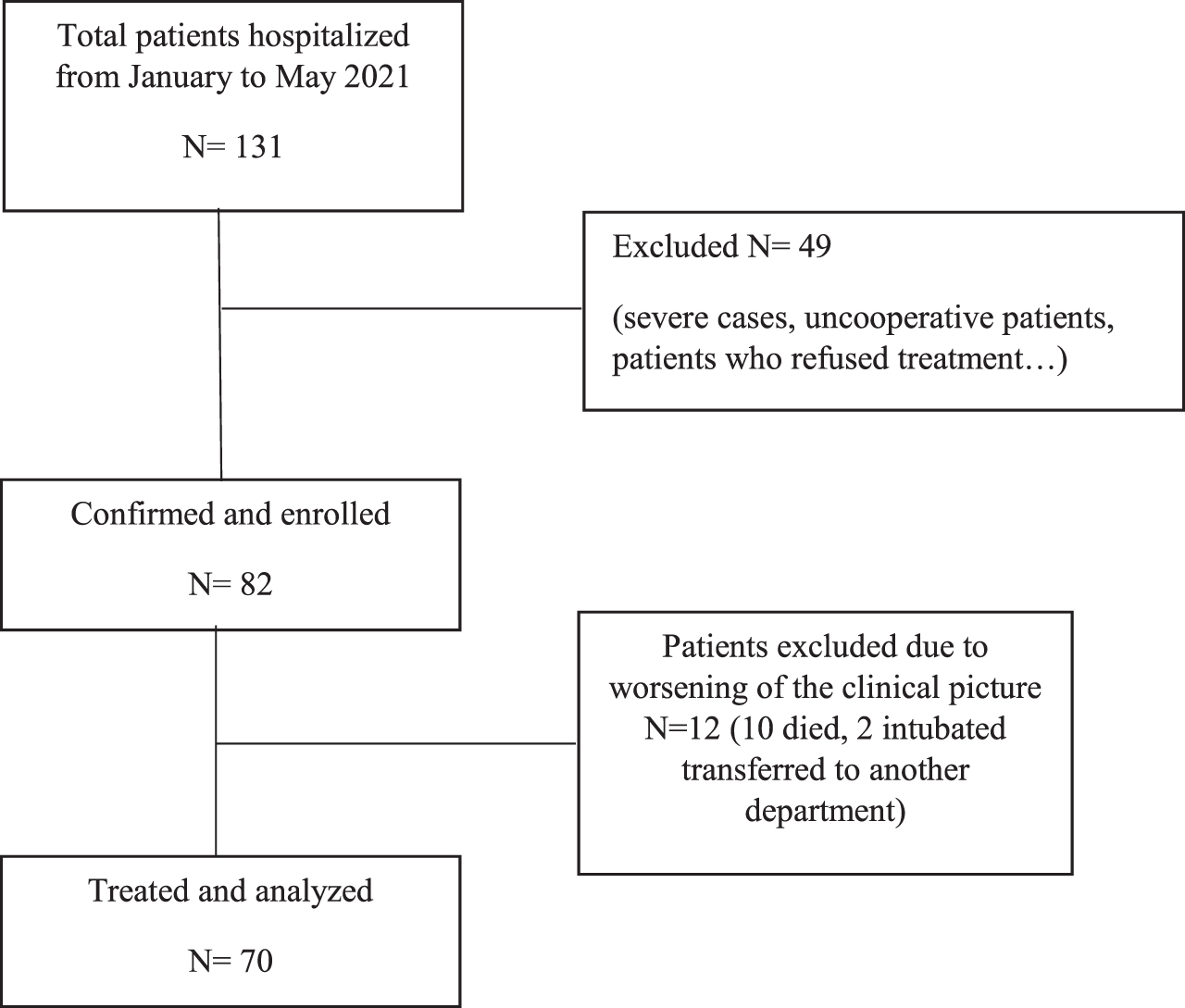
Table 1 summarizes demographic characteristics, blood exams and average values of the evaluation scales used at T0 and T1. The sample was composed by 37 women and 33 men, with an average age of 71 years (SD 71.4±12.6). The average hospitalization was 24 days (SD 23.8±11.6); the average rehabilitation treatments were 13.4 sessions (SD 13.2±7.8). All patients required oxygen therapy, 27 with nasal cannulas and 43 in Venturi masks. Main comorbidities reported were: Hypertension (65 patients), Diabetes (48 patients), COPD Chronic Obstructive Pulmonary Disease (21 patients), Obesity (BMI > 30 in 12 patients). 8 patients were hospitalized with neurological symptoms as the first COVID19 manifestation: Ischemic Stroke (6 patients), Haemorrhagic Stroke (1 patient) and Transient Ischemic Attack (1).
Table 1
Characteristics of the sample, blood chemistry tests and evaluation scales at T0 and T1
| Sex | Females (n = 37) 53 % |
| Males (n = 33) 47% | |
| Age | 71.6 (SD 71.4±12.6) |
| Oxygen support | Nasal cannulas (n = 27) 38.5% |
| Venturi Mask (n = 43) 61.5% | |
| Comorbidities | Hypertension 65 (92%), Diabetes 48 (68.5%), |
| COPD 21 (30%), Obesity (BMI > 30) 12 (17%) | |
| Proximate medical history: | |
| Ischemic Stroke 6 (8.5%), Transient Ischemic Attack 1 (1.4%), Hemorragyc Stroke 1 (1.4%) | |
| Days of hospitalization | 24.14 days±11.6 |
| MMSE | 24,1±8.9 |
| D-dimer | T0: 1238,8±2291.78μg/mL |
| T1: 748.9±661.47μg/mL | |
| CRP | T0: 4,72±4.82 mg/dL |
| T1: 3,8±3.86 mg/dL | |
| Vitamine D | T0: 14.5±9.8 ng/mL |
| MBI | T0: 39.8±35.01 |
| T1: 69.82±38.12 | |
| BORG dyspnoea scale | T0: 1.86±0.21 |
| T1: 0.21±0.48 |
MMSE (Mini Mental State Examination), MBI (Modified Barthel Index), CRP (C-Reactive Protein).
Statistical analysis shows that MBI increases statistically significantly from T0 to T1 (39.8±35.0 with 95% CI 31.6–48, vs 69.8±38.1 with 95% CI 60.8–78.7, p < 0.001), indicating an improvement in autonomy thanks to the early rehabilitation treatment and general clinical picture. About QoL improvement, in Fig. 1a and 1b are shown EQ-5D percentage parameters distribution at T0 and T1: there is a statistically significant increase from T0 to T1 (p < 0.001) in the percentage of patients who respond “no problem” for each parameter (mobility, self-care, usual activities, pain/discomfort, anxiety/depression), indicating a global improvement in patients’ quality of life.
Fig. 1a
EQ-5D percentage parameters distribution at T0 (mobility, self-care, usual activities, pain/discomfort, anxiety/depression).
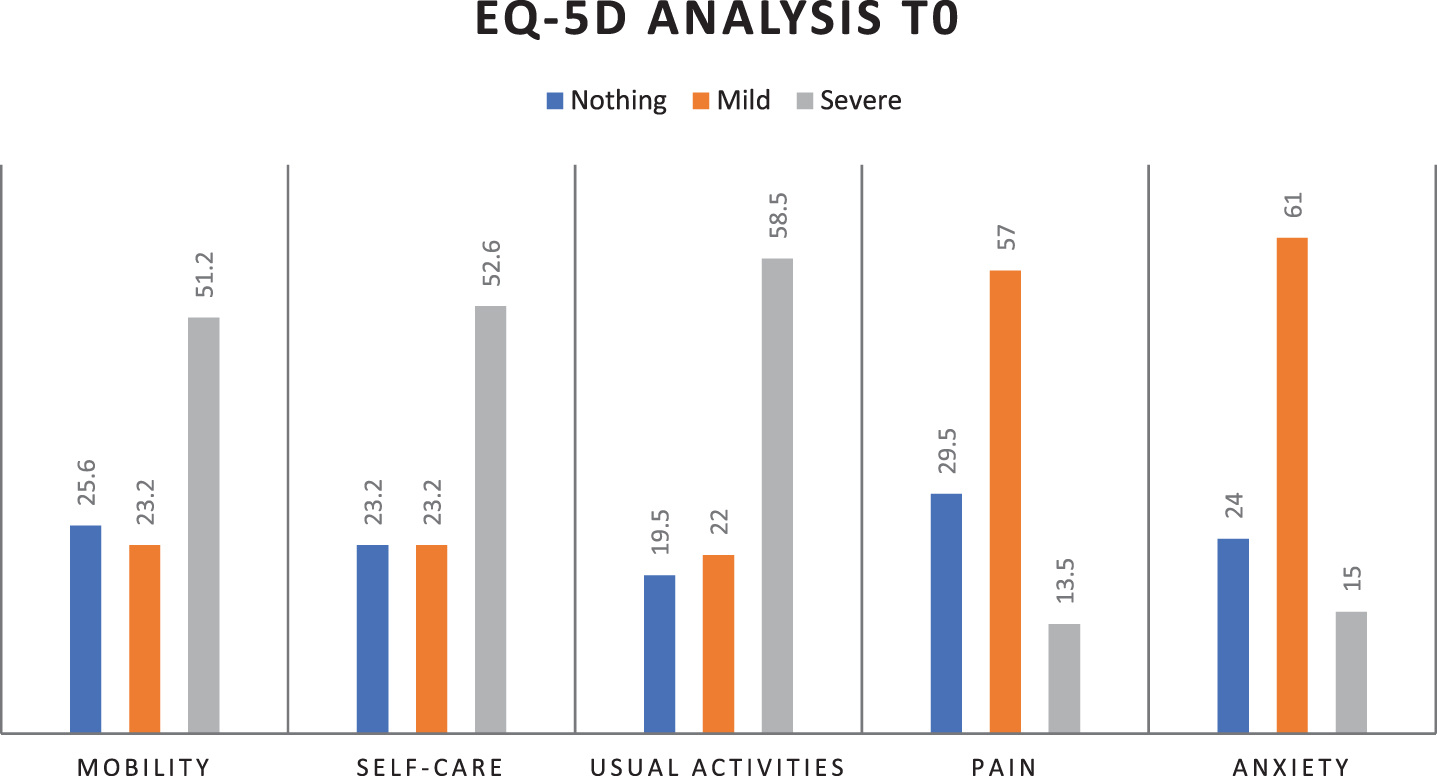
Fig. 1b
EQ-5D percentage parameters distribution at T1 (mobility, self-care, usual activities, pain/discomfort, anxiety/depression).
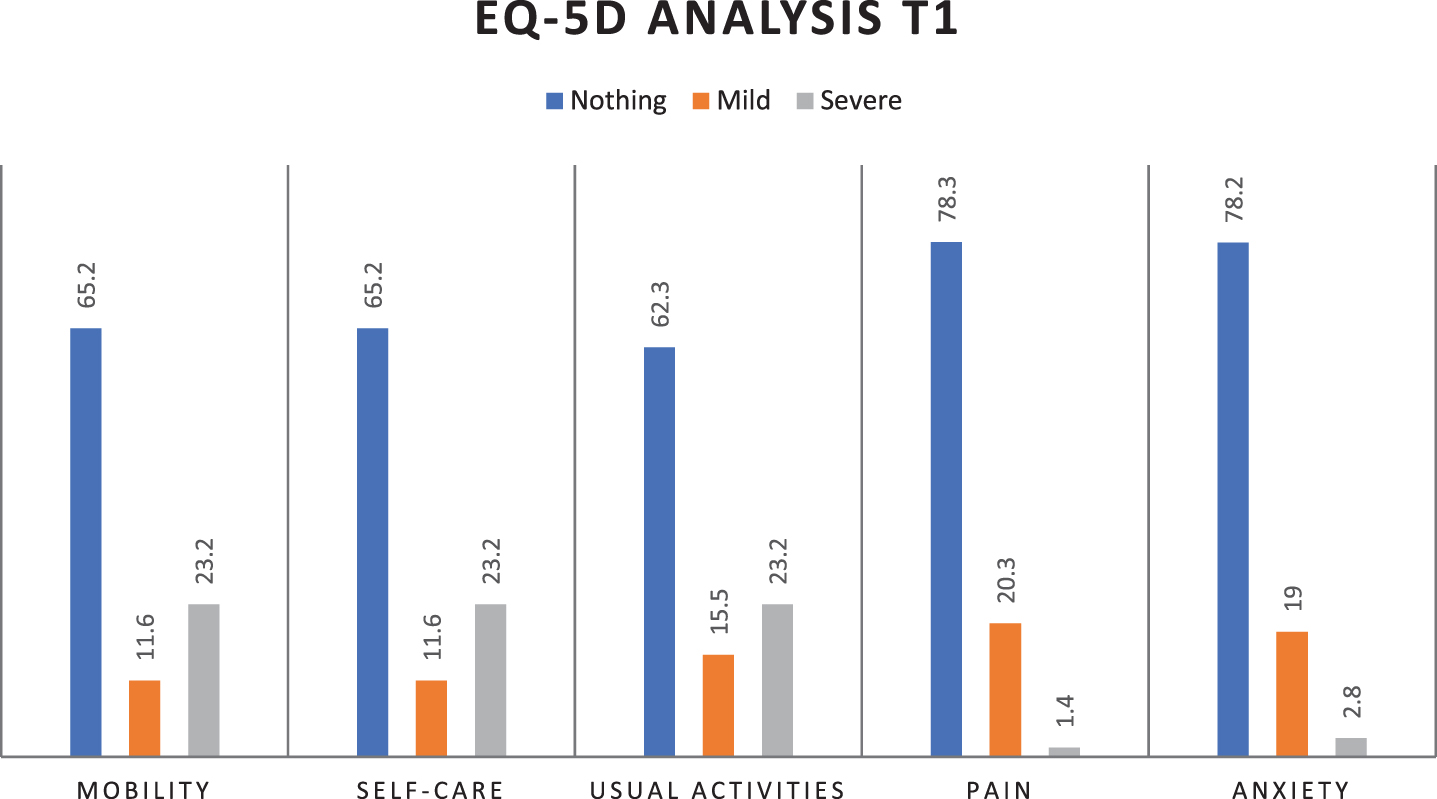
Table 2 shows results obtained using Pearson’s correlation test: interestingly, MBI at T0 correlates inversely and statistically significantly with all EQ-5D T0 variables and similarly the same results are repeated at T1 and correlating the delta MBI (MBI T1 –MBI T0) with the delta EQ-5D (EQ5D T1 –EQ5D T0) (p < 0.001); these results indicate that improving the autonomy has a positive effect on the patients’ quality of life.
Table 2
Correlations between MBI and EQ5D, MMSE, P/f, oxygen, d-dimer, CRP, Vitamin D at T0, T1 and delta T1-T0 obtained using Pearson’s correlation
| MBI T0 | MBI T1 | Δ MBI (MBI T1-MBI T0) | |
| EQ5D | Mobility | Mobility | Mobility |
| P < 0.001 | P < 0.001 | P < 0.001 | |
| R = –0.939 | R = –0.664 | R = –0.873 | |
| Self-care | Self-care | Self-care | |
| P < 0.001 | P < 0.001 | P < 0.001 | |
| R = –0.915 | R = –0.664 | R = –0.839 | |
| Usual activities | Usual activities | Usual activities | |
| P < 0.001 | P < 0.001 | P < 0.001 | |
| R = –0.875 | R = –0.677 | R = –0.806 | |
| Pain/discomfort | Pain/discomfort | Pain/discomfort | |
| P = 0.001 | P < 0.001 | P = 0.035 | |
| R = –0.400 | R = –0.493 | R = –0.255 | |
| Anxiety/depression | Anxiety/depression | Anxiety/depression | |
| P = 0.002 | P < 0.002 | P = 0.009 | |
| R = –0.361 | R = –0.791 | R = –0.315 | |
| MMSE | P < 0.001 | P < 0.001 | P = 0.049 |
| R = 0.569 | R = 0.747 | R = 0.244 | |
| P/F | P = 0.039 | P = 0.023 | |
| R = 0.263 | R = 0.389 | ||
| Liters Oxigen | P = 0.053 | P = 0.151 | |
| R = –0.232 | R = –0.173 | ||
| D-dimer | P = 0.591 | P = 0.776 | |
| R = –0.66 | R = –0.038 | ||
| CRP | P = 0.069 | P = 0.051 | |
| R = –0.220 | R = –0.251 | ||
| Vitamin D | P = 0.637 | ||
| R = 0.59 |
MBI (Modified Barthel Index), MMSE (Mini Mental State Examination), CRP (C-Reactive Protein), P/F (Pa02 / Fi02).
The MMSE correlates statistically significantly with MBI T0, MBI T1 and with delta MBI (r = 0.569, r = 0.747 and r = 0.244 respectively, p < 0.001), indicating that an adequate cognitive status correlates with a better increase in autonomy in ADL (Activities of Daily Living) after the rehabilitation treatment. Moreover, MBI values correlate directly and significantly with the P/F value calculated on the hemogasanalysis, both at T0 and T1 (r = 0.263 with p = 0.039, r = 0.389 with p = 0.023 respectively), indicating that improving the oxygen exchanges provides a better autonomy. On the other hand, analysing some phlogosis indices (C Reactive Protein and D-dimer), there was no statistically significant correlation with MBI (r = –0.22 with p = 0.06 at T0, r = –0.66 with p = 0.59 at T0 respectively); similar results were obtained correlating MBI with Vitamin D value (r = 0.59 with p = 0.637 at T0), and MBI with oxygen liter therapy (r = 0.232 with p = 0.053 at T0).
4Discussion
Early rehabilitation treatment in hospitalized COVID patients is an extremely important and very topical issue [6, 12–18]: the aim of rehabilitation in the acute phase is to prevent neuro-musculoskeletal damage, aiding the autonomy recovery in ADL and improving Quality of Life.
As reported in literature by international guidelines on COVID rehabilitation [13, 23–30], neuromuscular and respiratory rehabilitation is suggested for hospitalized patients with moderate and severe COVID-19, in association to a psychological support. Respiratory therapy includes mostly respiratory muscle training through various exercises including cough exercise, diaphragmatic training and stretching; though, airway clearance techniques (such as stimulated cough maneuvers and airway suctioning) are not recommended for patients with significant bronchial obstruction [13]; similarly, respiratory muscle training are not recommended during the acute phase when the patient is sedated or in a more critical condition [13]. Musculoskeletal therapies include mostly passive and active-assisted range of motion, stretching and pumping exercise for limbs, sit-to-stand training, walking, balance and aerobic training; finally, a psychosocial therapy is promoted through psychological counselling and sleep-promotion activities.
Rehabilitation prescription should be adapted to each individual patient according to their comorbidities, stage of recovery, severity of the symptoms. As reported by Wasilewski et al., duration, frequency and intensity of rehabilitation protocols are different in literature, but generally authors suggest a rehabilitative treatment once-twice a day for 15–45 minutes. Chinese guidelines [24] also focus on the application of specific evaluation scales for Basic Activities of Daily Living (BADL) and Instrumental Activities of Daily Living (IADL), with the aim of also setting up rehabilitative programmes for these aspects.
As reported in literature [31–33], COVID greatly affects the psychological sphere, causing a depressive state that affects daily life: our study highlights how the improvement of autonomy is associated with a better quality of life.
The presence of comorbidities in our sample (hypertension, COPD, diabetes type 2, dyslipidaemia, obesity) is similar to that reported in the literature [5]. Moreover, about initial symptoms shown by patients, we had 8 patients with neurological manifestations: this data is very interesting, although it is difficult to say if COVID is the real cause of these symptoms, considering the presence of various cardiovascular risk factors in these patients. Interestingly, in this group of patients we reported a lower need for oxygen support. Many neurological symptoms in COVID patients have been reported in literature [34–37], but surely further investigations will be necessary to better know this aspect.
Our study shows certainly some limitations: the sample size, the lack of patients’ follow-up after hospital discharge, the absence of control group, the non-homogeneity of the sample due to comorbidities shown by patients, the absence of a psychologist; in addition, although some authors [6], recommend a MMSE > 24 as an inclusion criterion for rehabilitation treatment, in our study we also included patients with lower MMSE, obtaining improvements in terms of autonomy and QoL worse than patients with a better cognitive status.
In conclusion, an early rehabilitation treatment should promote autonomy and a better QoL for patients. A multidisciplinary evaluation, an Individual Rehabilitation Project, and the use of appropriate evaluation scales are fundamental for COVID patients’ management; finally, it is certainly important to perform a careful follow-up, in order to identify and treat possible neuro-muscular disorders that can be associated with long COVID syndrome [38, 39].
Acknowledgments
We thank all the medical and health personnel of the COVID Medicine Department for their availability, work and dedication in these difficult months of the COVID pandemic.
Conflict of interest
The authors have no conflict of interest to report.
References
[1] | Wang C , Horby PW , Hayden FG , Gao GF , A novel coronavirus outbreak of global health concern, Lancet. (2020) ;395: :470–3. |
[2] | Ren LL , Wang YM , Wu ZQ , Xiang ZC , Guo L , Xu T , et al., Identification of a novel coronavirus causing severe pneumonia in human: A descriptive study, Chin Med J (2020) ;133: :1015–24. |
[3] | Guo YR , Cao QD , Hong ZS , Tan YY , Chen AD , Jin HJ , et al., The origin, transmission and clinical therapies on coronavirus disease (COVID-19) outbreak –an update on the status, Mil Med Res (2020) ;7: :11. |
[4] | Lai KCC , Lam W , Laboratory testing for the diagnosis of COVID-19, Biochem Biophys Res Commun. (2021) ;538: :226–30. |
[5] | Ejaz H , Alsrhani A , Zafar A , Javed H , Junaid K , Abdalla AE . et al., COVID-19 and comorbidities: Deleterious impact on infected patients, J Infect Public Health (2020) ;13: :1833–9. |
[6] | Curci C , Pisano F , Bonacci E , Camozzi DM , Ceravolo C , Bergonzi R , et al., Early rehabilitation in post-acute COVID-19 patients: Data from an Italian COVID-19 Rehabilitation Unit and proposal of a treatment protocol, Eur J Phys Rehabil Med (2020) ;56: :633–41. |
[7] | Lazzeri M , Lanza A , Bellini R , Belfiore A , Cecchetto S , Gaudiello G , et al., Respiratory physiotherapy in patients with Covid-19 infection in acute setting: A position paper of the italian association of respiratory physiotherapists (ARIR), Monaldi Arch Chest Dis (2020) ;26: :90. |
[8] | Kiekens C , Boldrini P , Andreoli A , Avesani R , Gamna F , Grandi M , et al., Rehabilitation and respiratory management in the acute and early post-acute phase, “Instant paper from the field” on rehabilitation answers to the coVid-19 emergency. Eur J Phys Rehabil Med (2020) ;56: :323–6. |
[9] | Simpson R , Robinson L , Rehabilitation following critical illness in people with COVID-19 infection, Am J Phys Med Rehabil (2020) ;99: :470–4. |
[10] | Li J , Effect and enlightenment of rehabilitation medicine in COVID-19 management, Eur J Phys Rehabil Med (2020) ;56: :335–8. |
[11] | Gandhi RT , Lynch JB , Del Rio C , Mild or moderate Covid-19, New Eng J Med (2020) ;383: :1757–66. |
[12] | Agostini F , Mangone M , Ruiu P , Paolucci T , Santilli V , Bernetti A , Rehabilitation setting during and after Covid- An overview on recommendations, J Rehabil Med (2021) ;19: :53. |
[13] | Wasilewski MB , Cimino SR , Kokorelias KM , Simpson R , Hitzig SL , Robinson L , Providing rehabilitation to patients recovering from COVID- A scoping review, PM&R. (2021) ;19: .8. doi 10.1002/pmrj.12669. |
[14] | Simpson R , Robinson L , Rehabilitation after critical illness in people with COVID-19 infection, Am J Phys Med Rehabil (2020) ;99: :470–4. |
[15] | Brugliera L , Spina A , Castellazzi P , Cimino P , Tettamanti A , Houdayer E , et al., Rehabilitation of COVID-19 patients, J Rehabil Med (2020) ;52: :1–3. |
[16] | Candan SA , Elibol N , Abdullahi A , Consideration of prevention and management of long-term consequences of post-acute respiratory distress syndrome in patients with COVID-19, Physiother Theory Pract (2020) ;36: :663–8. |
[17] | Kalirathinam D , Guruchandran R , Subramani P , Comprehensive physiotherapy management in COVID-19 –A narrative review, Sci Med (2020) ;30: :e38030. |
[18] | Zen B , Chen D , Qiu Z , Zhang M , Wang G . Expert consensus on protocol of rehabilitation for COVID-19 patients using framework and approaches of WHO international Family Classifications, Aging (2020) ;3: :82–94. |
[19] | Ambrosino P , Papa A , Maniscalco M , Di Minno MND , COVID-19 and functional disability: Current insights and rehabilitation strategies. Postgraduate Med J. 2020; 469-70. |
[20] | Ohura T , Hase K , Nakajima Y , Nakayama T , Validity and reliability of a performance evaluation tool based on the modified Barthel Index for stroke patients, BMC Med Res Methodol (2017) ;17: :131. |
[21] | EuroQol-a new facility for the measurement of health-related quality of life. Health Policy (1990) ;16: :199–208. |
[22] | Crisafulli E , Clini EM . Measures of dyspnea in pulmonary rehabilitation, Multidiscip Respir Med (2010) ;5: :202–10. |
[23] | Barker-Davies R , O’Sullivan O , Prathima Senaratne KPP , Baker P , Cranley M , Dharm-Datta S , et al., The Stanford Hall consensus statement for post-COVID-19 rehabilitation, Br J Sports Med (2020) ;54: :949–59. |
[24] | Zhao HM , Xie YX , Wang C , Recommendations for respiratory rehabilitation in adults with coVid-19, Chin Med J (2020) ;133: :1595–602. |
[25] | Aytur YK , Koseoglu BF , Ozyemisci Taskiran O , Ordu-Gokkaya NK , Unsal DS , Sonel TB , et al., Pulmonary rehabilitation principles in SARS-COV2 Infection (COVID-19): A guideline for the acute and subacute rehabilitation, Turk J Phys Med Rehabil (2020) ;66: :104–20. |
[26] | Ministry of Health and Allied Health Aotearoa NewZeland. Guidance for the Rehabilitation of People with Recovering from COVID-19 in Aotearoa NewZeland.Wellington, New Zeland; Ministry of Health and Allied Health Aotearoa New Zeland; 2020. |
[27] | Collaborative N.P.I.C.R. Responding to COVID-19 and beyond: A framework for assessing early rehabilitation needs following treatment in intensive care. 2020. Society BSoRMaIC, editor, 36. |
[28] | Chartered Society of Physiotherapy. Rehabilitation and COVID-19 ’ CSP Policy Statement. London, UK; Chartered Society of Physiotherapy; 2020. |
[29] | Ross D , Allport F , Carter R , COVID-19 Patient Rehabilitation Booklet – Information for Patients. Leeds,UK;National Health Service (NHS); 2020. |
[30] | World Health Organization Regional Office for Europe. Support for Rehabilitation Self-Management after COVID- 9 Related Illnes. Copenhagen, Denmark: World Health Organization; 2020. |
[31] | Bajwah S , Wilcock A , Towers R , Costantini M , Bausewein C , Simon ST , et al., Managing the supportive care needs of those affected by COVID-19, Eur Respir J. (2020) ;55: :2000815. |
[32] | Van Der Sar-van Der Brugge S , Talman S , Boonman-De Winter L , de Mol M , Hoefman E , van Etten RW , De Backer IC , Pulmonary function and health-related quality of life after COVID-19 pneumonia, Respir Med (2021) ;176: :106272. |
[33] | Nguyen HC , Nguyen MH , Do BN , Tran CQ , Nguyen TTP , Pharm KM , et al., People with suspected COVID-19 symptoms were more likely depressed and had lower health-related quality of life: The potential benefit of health literacy, J Clin Med (2020) ;9: :965. |
[34] | Mao L , Jin H , Wang H , Hu Y , Chen S , He Q , et al., Neurologic manifestations of hospitalized patients with coronavirus disease in Wuhan, China, JAMA Neurol. (2020) ;77: :683–90. |
[35] | Zhang Y , Xiao M , Zhang S , Xia P , Cao W , Jiang W , et al., Coagulopathy and antiphospholipid antibodies in patients with Covid-19, N Engl J Med (2020) ;382: :e38. |
[36] | Thachil J , Tang N , Gando S , Falanga A , Cattaneo M , Levi M , et al., ISTH interim guidance on recognition and management of coagulopathy in COVID-19, J Thromb Haemost (2020) ;18: :1023–26. |
[37] | Perry RJ , Smith CJ , Roffe C , Simister R , Narayanamoorthi S , Marigold R , et al., Characteristics and outcomes of COVID-19 associated stroke: A UK multicentre case-control study, J Neurol Neurosurg Psychiatry. (2021) ;92: :242–8. |
[38] | Baig AM , Chronic COVID syndrome: Need for an appropriate medical terminology for long-COVID and COVID long-haulers, J Med Virol (2021) ;93: :2555–6. |
[39] | Halpin S , O’Connor R , Sivan M , Long COVID and chronic COVID syndromes, J Med Virol (2021) ;93: :1242–3. |



