Polymorphism of the Forkhead box-O3 (FOXO3) Longevity Gene rs2802292 and senescence-associated secretory phenotype (SASP) in Indonesian Elderly Population
Abstract
BACKGROUND:
Forkhead box O3 (FOXO3) is a transcription factor that regulates stress resistance, metabolism, cell cycle, and apoptosis. Several studies exhibited the association of the FOXO3 polymorphism rs2802292 with human longevity and protects individuals from degenerative diseases. The emergence of degenerative diseases in the elderly is associated with the accumulation of senescent cells that secrete a secretome known as a senescence-associated secretory phenotype (SASP) consists of several cytokines, chemokines, growth factors, proteases.
OBJECTIVE:
The aim of this research was to analysis polymorphism of FOXO3 gene rs2802292 G-allele and its impact to the SASP by measuring IL-1α, IL-6, IL-8, and IL-10 in Indonesian elderly populations.
METHODS:
This study was conducted on 92 elderly subjects living at Jakarta. DNA was isolated from the whole blood then continued with PCR and sequencing for FOXO3 rs2802292 analysis. SASP was analyzed from the plasma using Luminex. Statistical analysis was performed using ANOVA and Kruskal Wallis.
RESULTS:
The results showed that FOXO3 rs2802292 was detected in Indonesian elderly population as follows GG, GT, TT genotype frequencies were 17.4 %, 42.4 % and 40.2 % respectively. Meanwhile, G and T allele frequencies based on the Hardy-Weinberg equilibrium test were 0.385 and 0.615 respectively. GG genotype was significantly found in subjects with longevity. Nevertheless, SASP analysis was found no significant differences both among GG, GT, TT genotypes.
CONCLUSION:
The frequency of FOXO3 rs2802292 G-allele in Indonesian elderly population was 0.385 and significantly detected in subjects with longevity. However, the FOXO3 rs2802292 polymorphism did not affect cytokines level as the component of SASP.
1Introduction
Elderly is a person who has reached the age of 60 years and over [1]. At present, the population aged 60 years and over has increased from 1 billion in 2020 to 1.4 billion. It is estimated that by 2050, the world population aged 60 and over will double to 2.1 billion [1]. Evolutionary theorists have shown that longevity is determined by various environmental and genetic factors [2]. Research proves that there is a protective gene, namely Forkhead box 3 (FOXO3) as a transcription factor that plays a role in individuals with longevity [3]. The FOXO3 gene, located specifically at chromosome 6q21, holds a pivotal role in orchestrating diverse biological processes by modulating activity and shaping the expression of effector genes engaged in processes such as proliferation, apoptosis, survival, cell-cycle progression, and response to DNA damage [4]. The subcellular localization of FOXO3a plays a crucial role in determining the functions and overall behavior of this protein. Notably, phosphorylation of FOXO3a facilitates its translocation from the nucleus to the cytoplasm, where it forms associations with 14-3-3 proteins, effectively impeding reentry into the nucleus [4]. FOXO3 serves as the primary controller, maintaining longevity by responding to diverse stimuli such as growth factors, hormones and nutrition. These factors play a role in regulating metabolism, stress resistance, the cell cycle and apoptosis [5]. One notable discovery highlighted in the literature is the apparent role of FOXO3 as a “resilience” gene. In the context of the brain, maintaining the pool of stem cells aligns with the goal of enhancing the brain’s resilience to age-related pathology, thereby providing a safeguard against cognitive decline. In various organs and systems, FOXO3 may contribute to increased resilience, potentially decelerating the progression of age-related conditions like coronary heart disease (CHD) and cardiometabolic diseases and it was suggested can slow down the disease process, resulting in a significant reduction in mortality risk [6]. The close association of FOXO3 with longevity underscores its pivotal role in maintaining balance. A thorough understanding of FOXOs, as exemplifying how biological systems tackle the intricacies of sustaining equilibrium for life, has the potential to advance understanding the molecular foundations of age-related diseases, lifespan and effective strategies for its management [7].
FOXO proteins, recognized as transcription factors, operate in the cell nucleus to regulate genes, with their activity primarily governed by their movement between the cytoplasm and the nucleus. The regulation of FOXO proteins primarily relies on posttranslational modifications, and because these alterations can be reversed, it is possible to achieve the activation and inactivation of FOXO through pharmacological interventions [8]. Therapeutic targeting can focus on four categories: regulating FOXO expression, post-translational modifications, interactions between proteins and FOXO, and interactions between DNA and FOXO. Potential applications for therapy and prevention encompass a range of conditions such as aging, cancer, diabetes, as well as cardiovascular and neurodegenerative diseases [9]. Various small-molecule compounds, such as nuclear export inhibitors, PI3K, mechanistic target of rapamycin (mTOR), AKT, phosphoinositide-dependent kinase 1 (PDK1), calmodulin inhibitors, and certain chemotherapeutic drugs, have demonstrated the capability to influence the subcellular localization of FOXO proteins [10, 11].
In addition to advancing targeted therapies for FOXO3 expression, it has been demonstrated that the existence of polymorphisms in FOXO3 correlates with heightened protein expression and is linked to increased longevity. Several case-control studies have investigated the association of the FOXO3 polymorphism with human longevity in Japanese, Italian, German populations and China [12–14]. A meta-analysis of eight case-control studies indicated that the rs2802292 G-allele correlated with longevity in subgroups of individuals in various countries [14]. There is evidence that polymorphic FOXO3 alleles may protect individuals against degenerative diseases [15]. In the elderly male Japan and American population, rs2802292 G-allele is a protective factor for reducing mortality of coronary heart disease. The frequency of the G allele varies in each population, that is 0.83 in African, 0.39 in America, 0.43 in Europe, 0.31 in East Asia and 0.56 in South Asia populations [16]. Until now, There has been no report on the frequency of the G allele in the Indonesian elderly population.
The emergence of degenerative diseases in the elderly is associated with the accumulation of aging cells that secrete a secretome known as a senescence-associated secretory phenotype (SASP) [17]. SASP consists of cytokines (such as interleukins/IL-6, IL-1, IL-8), chemokines (such as CXCL-5, CXCL-6, CCL-7), growth factors (such as Insuline –like Growth Factor/IGF, IGF-binding proteins/IGFBP), proteases (Matrix metalloproteinases/MMP-1, MMP-3, MMP-10), lipids, and other molecules that have autocrine and paracrine effects [18]. The effect of autocrine can cause cell growth to stop due to these cells increase the secretion of various secretomes. By paracrine effect, the secretome can change the phenotype of a young cell to an old one. The resulting SASP can damage tissue structures thereby the tissues cannot function normally [19]. In aging pulmonary arteries, the smooth muscle releases components of the SASP, including IL-6 and IL-8, along with other extracellular matrix proteins. These substances contribute to the thickening of the tunica intima in blood vessels, leading to the development of pulmonary hypertension [20]. The presence of SASP was linked to the thickening of the pulmonary artery wall in individuals diagnosed with chronic obstructive pulmonary disease. Additionally, the secretion of matrix metalloproteinases (MMPs) by aging skin cells also plays a role in reducing collagen levels and thinning both the dermal and epidermal layers of the skin [21].
The regulation of SASP occurs at both the transcriptional and post-transcriptional levels. The primary controller of the expression of SASP components, including various interleukins and chemokines, is nuclear factor-κB (NF-κB) [22]. NF-κB activation leads to the production of interleukins, cytokines, and other molecules associated with inflammation [23]. Research proved that FOXO3 regulates NF-κB activation in immune cells [24]. Moreover, the interaction of FOXO3 with NF-κB prevents translocation and activation of NF-κB in myeloid cell signaling pathways [25]. In mammals FOXO3 signaling is linked to homeostatic regulation of the immune system. Study form Lin et al about FOXO3 knockout mice having lymphoproliferative disease associated with inflammation in several tissues, showing increased cytokine secretion, multisystemic inflammation was shown to be caused by a lack of FOXO3 to inhibit NF-κB [26]. Moreover, increased FOXO3 expression can inhibit NF-κB-induced TNF-α induction [26]. However, until now it is not known how FOXO3 plays a role in aging cells and their SASP secretions. FOXO3 polymorphism will increase FOXO3 expression and is associated with longevity and reduced mortality due to degenerative diseases, but the mechanism underlying this process is still unclear. Hardiany et al [27] found that data on FOXO3 mRNA expression in 60 samples of elderly women in Jakarta showed varied results. FOXO3 expression in the age group 60–70 years was 0.001–10.48 with a median of 0.62, while FOXO3 expression in the age group > 70 years was 0.003–4.593 with a median value of 0.175. The variation in expression value is thought to be caused by polymorphisms in the FOXO3 gene. Until now, there has been no research in Indonesia that analyzed the FOXO3 polymorphism. In addition, no studies have explored the impact of these polymorphisms on SASP. Therefore, the aim of this research was to analysis polymorphism of FOXO3 gene rs2802292 G-allele and its impact to the SASP by measuring several cytokines (IL-1α, IL-6, IL-8, and IL-10) in Indonesian elderly populations.
2Materials and methods
It was a cross-sectional study conducted in 92 elderly subjects from the Integrated Development Post at three different sub-districts (Cilandak, Kebayoran Baru, and Pesanggarahan) in Jakarta. The inclusion criteria were male or female aged 60 years and over who signed informed consent. The exclusion criteria were a smoking or drinking alcohol history in the last year and suffer from acute infection. Ethical clearance was issued by Ethic Committee Faculty of Medicine Universitas Indonesia with ethical number KET-100/UN2.F1/ETIK/PPM.00.02/2023.
2.1Isolation of DNA
DNA was isolated from whole blood using a genomic DNA purification wizard kit (Promega) based on Manual Instruction kit. Cell lysis solution was added to 300μL of whole blood and incubated for 10 minutes. After centrifugation, supernatant was discarded and nuclei lysis solution was added into pellet white blood cells. RNase solution was added, followed by addition of protein precipitation solution. After centrifugation, supernatant was poured into 1.5 mL of microtube containing isopropanol to precipitate DNA. DNA pellet was washed by 70% of ethanol and rehydrated in 100μL of DNA rehydration solution. Rehydrated DNA was stored at 4°C for further analysis.
2.2Polymerase Chain Reaction (PCR) and Sequencing of FOXO3 rs2802292
Three microliters of DNA template were amplified using 15μL GoTaq Green Master Mix (Promega), 1.5μL primer and 9μL nuclease free water by PCR machine (Applied Biosystem). FOXO3 primer sequences were CAGCTTCTGAGTGACAGAGTG (Forward) and TTCTTCCCTAGAGAGCAGCAG (Reverse) produced 157 bp amplicon [28]. DNA amplification was run for 29 cycles with an initial denaturation of 95°C for 15 minutes, then entered into a cycle consisting of denaturation of 94°C for 1 minute, annealing at 60°C for 1 minute, and extension at 72°C for 1 minute. At the end of the cycle, the extension time was extended at 72°C for 10 minutes. After the DNA amplification process was complete, the amplicon was stored at 4°C. The amplification results of PCR products were visualized using 1.5% agarose gel electrophoresis in 1x TAE buffer and stained by GelRedTM (Biotium 41003). Sanger sequencing was performed to PCR product by Genetika Science Ltd., and analyzed using BioEdit software.
2.3SASP (IL-1α, IL-6, IL-8 and IL-10) analysis
Plasma was separated from the whole blood by centrifugation at 3,500 G for 15 minutes. SASP was analyzed from the plasma using a Magnetic Luminex Assay (R&D system brand biotech, Minneapolis, USA). In this method, several analytes were tested at once based on magnetic beads. This method is very suitable for analysis with a limited number of samples. Fifty microliters of plasma, standard and control samples were mixed with 50μL of microparticle cocktail and incubated for 2 hours at room temperature in a shaker with a speed of 800 rpm. Then the liquid in the well was discarded and filled with 100μL wash buffer (repeated 3 times). In each well, 50μL of Biotin-antibody cocktail was introduced and incubated for 1 hour at room temperature on a shaker set at 800 rpm. Following that, the wash buffer was used for the washing step. Subsequently, 50μL of streptavidin-PE was added to each well and incubated for 30 minutes at room temperature on a shaker at 800 rpm. Then the washing steps were repeated using the wash buffer 3 times by adding 100μL of wash buffer to each well. After all procedures were completed, each well was read using the Luminex analyzer 100/200 TM.
2.4Formula for calculating mean of age and SASP in G and T allele
Mean of age/SASP in G allele = [(mean of age/SASP in GG genotype x number of GG genotype)+(mean of age/SASP in GT genotype x number of GT genotype)]/ total number of GG and GT genotype.
Mean of age/SASP in T allele = [(mean of age/SASP in TT genotype x number of TT genotype)+(mean of age/SASP in GT genotype x number of GT genotype)]/ total number of TT and GT genotype.
2.5Statistical analysis
Data analysis was performed using SPSS software version 26. The normality test used the Kolmogorov Smirnov test. While the homogeneity test used the Levene test. Analysis of differences between groups was carried out by parametric test ANOVA for normally distributed and homogeneous data. For non-normally distributed data and non-homogeneous variances, the Kruskal Wallis non-parametric test was used. For normally distributed data, the presentation of results involved a graph displaying the mean±standard deviation (SD). In the case of non-normally distributed data, the presentation comprised a median (minimum-maximum) representation. P < 0.05 was set as the significant result.
3Results
3.1Subject characteristics
The number of subjects in this study were 92 elderlies with an average age of 70.21±6.28 years were recruited from Cilandak, Kebayoran Baru and Pesanggrahan sub-district, South Jakarta. The sub-districts were selected using the Probability Proportional to Size method based on population data for the elderly in South Jakarta. Based on gender, the number of male elderlies were 22 people (23.9%) and 70 people (76.1%) for the female. Systolic blood pressure was 130 (100–170) mmHg and diastolic blood pressure was 80 (50–110) mmHg. About 53.3% of the subjects exhibited normal blood pressure, while 46.7% showed signs of hypertension. Moreover, random blood glucose was 112 (78–333) mg/dL. A majority of subjects (91.3%) displayed normal blood glucose, while 8.7% exhibited elevated levels. Notably, there was a higher representation of female elderly individuals compared to males, and the majority of elderly participants demonstrated normal blood pressure and glucose levels.
3.2Distribution of FOXO3 rs2802292 genotypes & alleles
FOXO3 rs2802292 polymorphism was analyzed by sequencing showed that GG genotype frequency was 17.4 %, GT genotype frequency was 42.4 % and TT genotype frequency was 40.2 % . Whereas, the G and T allele frequencies based on the Hardy-Weinberg equilibrium test were 0.385 and 0.615 respectively. A single peak allele G for homozygous GG, a double peak allele G and T for heterozygous GT, and a single peak allele T for homozygous TT were shown by arrow in Fig. 1. The GG genotype was found significantly in older age as shown in Table 1. The mean age for G allele was 71.18 years old and 69.51 years old for T allele. The findings affirm the presence of the FOXO3 rs2802292 polymorphism in elderly individuals residing in South Jakarta, establishing a significant correlation with increasing age. However, there was no significant difference in blood pressure and random blood glucose based on FOXO3 rs2802292 genotype (Table 1).
Fig. 1
FOXO3 rs2802292 sequencing results. (A) GG homozygous genotype. (B) GT heterozygous genotype. (C) TT homozygous genotype.
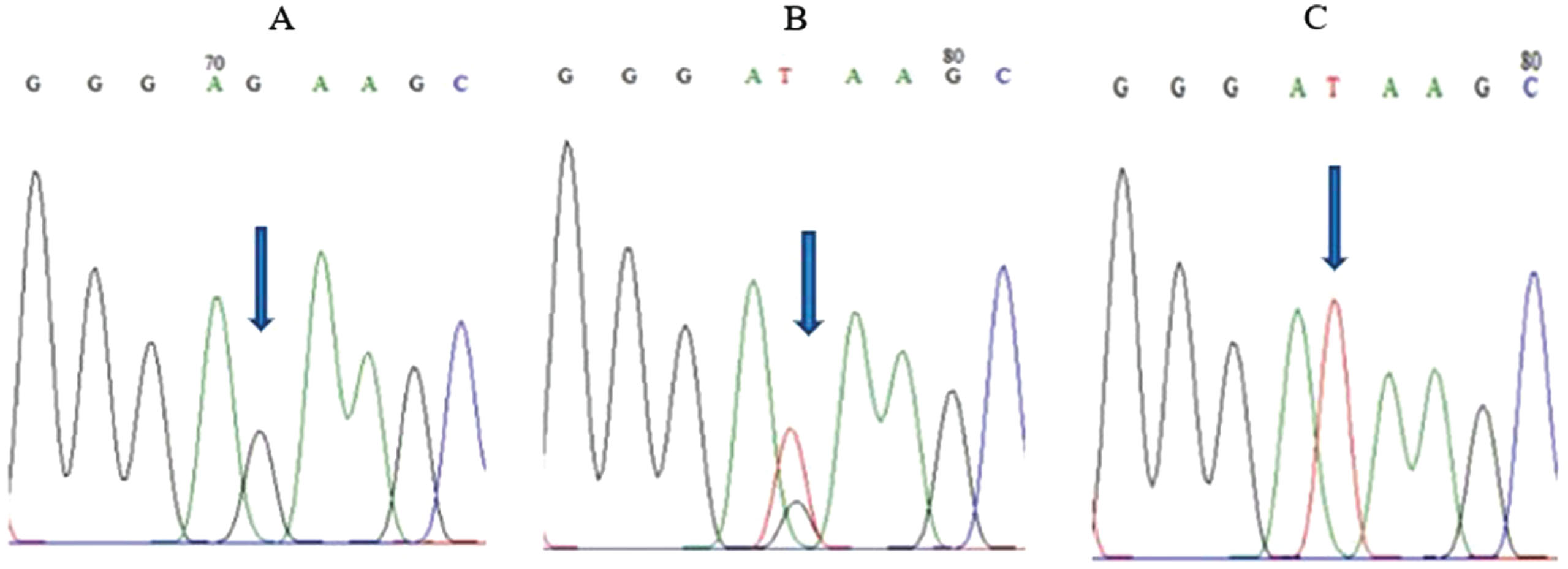
Table 1
The Frequency of FOXO3 rs2802292 genotype
| FOXO 3 rs2802292 | n (%) | Age (year old) | Systolic blood pressure (mmHg) | Diastolic blood pressure (mmHg) | Random blood glucose (mg/dL) |
| Mean±SD | Median (min-max) | Median (min-max) | Median (min-max) | ||
| Genotype | |||||
| GG | 16 (17.4) | 73.5±5.93 | 130 (100-160) | 80 (70-90) | 114.5 (82-298) |
| GT | 39 (42.4) | 70.23±6.67 | 130 (100-160) | 70 (50-90) | 117 (78-333) |
| TT | 37 (40.2) | 68.76±5.56 | 130 (100-170) | 80 (50-110) | 108 (85-306) |
| p = 0.04* | p = 0.349 | p = 0.210 | p = 0.375 |
Note: * significant.
3.3Analysis of SASP parameters based on SNP FOXO3 rs2802292
The SASP parameters were assessed through the measurement of IL-1α, IL-6, IL-8, and IL-10 utilizing the Luminex assay. The level of each cytokine was 2.12 (1.61–9.96) pg/mL for IL-1α, 2.05 (1.43–6.24) pg/mL for IL-6, 10.32 (0.38–24.09) pg/mL for IL-8 and 2.86 (1.70–148.34) pg/mL for IL-10. There was no significant difference in all SASP level based on the genotype as shown in Fig. 2 and Table 2. However, mean of inflammatory cytokine IL-1α and IL-6 were lower in the G allele than T allele. Moreover, mean of anti-inflammatory cytokine IL-10 level was higher in the G allele compare to the T allele (Table 2). This result indicates that the polymorphism of FOXO3 rs2802292 does not appear to influence cytokines level as the component of SASP in these elderly subjects.
Fig. 2
Distribution of SASP level (pg/mL) based on genotype FOXO rs2802292. (A). IL-1α level, (B). IL-6 level, (C). IL-8 level (D) IL-10 level.
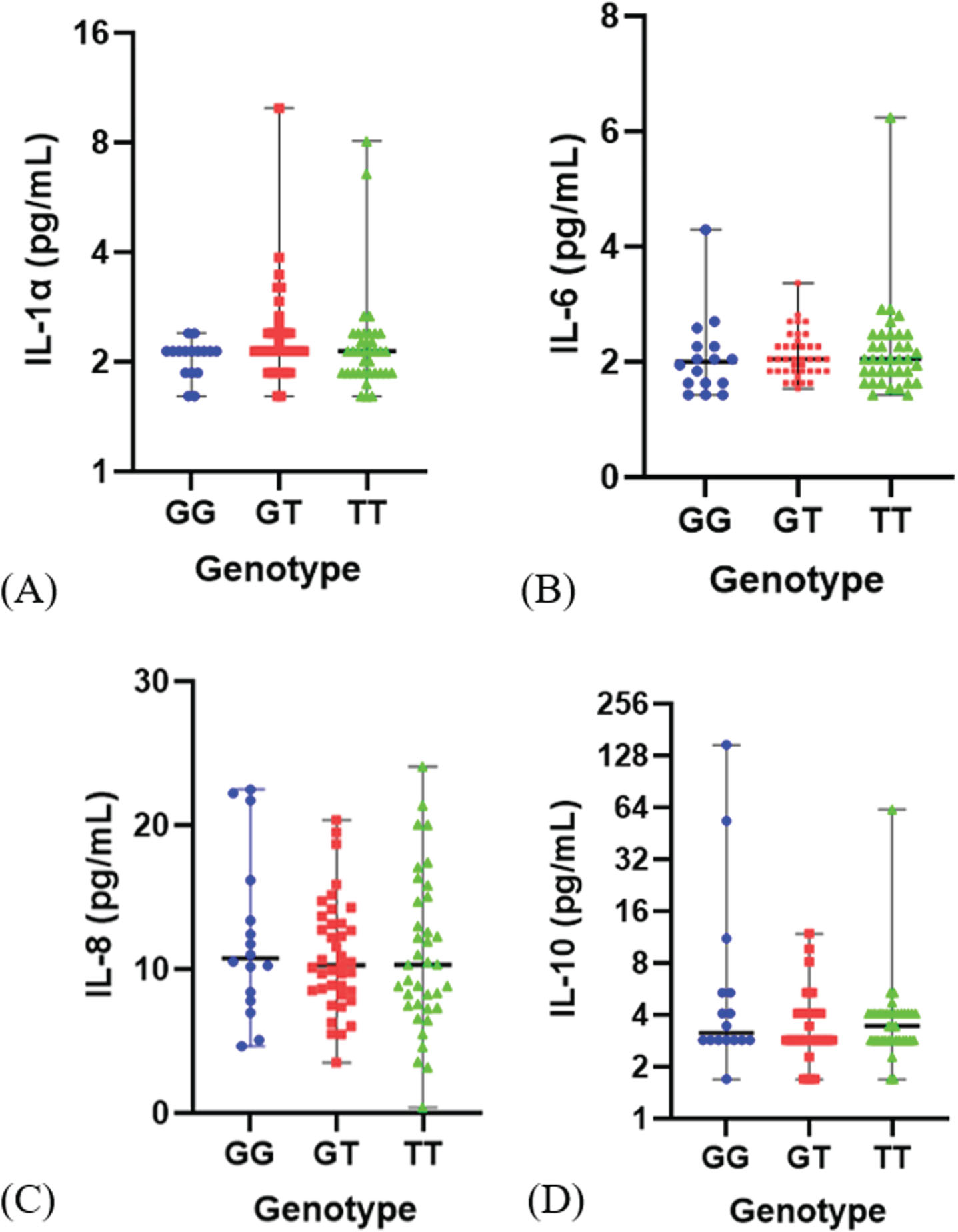
Table 2
SASP level based on FOXO3 rs2802292
| FOXO 3 | SASP Level (pg/mL) | |||
| rs2802292 | IL-1α | IL-6 | IL-8 | IL-10 |
| Genotype | ||||
| GG (n = 16) | 2.13 (1.61-2.40) | 2.0 (1.43-4.30) | 12.19±5.74 | 3.16 (1.70-148.34) |
| GT (n = 39) | 2.13 (1.61-9.96) | 2.03 (1.54-3.37) | 10.95±3.84 | 2.86 (1.70-11.90) |
| TT (n = 37) | 2.13 (1.61-8.06) | 2.05 (1.43-6.24) | 10.99±5.38 | 3.46 (1.70-62.38) |
| p = 0.140 | p = 0.593 | p = 0.658 | p = 0.449 | |
| Allele | ||||
| G | 2.36 | 2.09 | 11.31 | 7.31 |
| T | 2.42 | 2.13 | 10.96 | 4.38 |
4Discussion
Being a part of the FOXO subfamily, the FOXO3 gene is situated at a distinct location on chromosome 6q21, which is linked to human longevity. This association is supported by the identification of genetic variants in FOXO3 that are correlated with remarkable longevity observed in various organisms, including worms, flies, and mammals [29]. Over the last decade, many studies have successfully found that FOXOs play important role in many cellular processes. FOXOs transcriptionally will either inhibit or activate downstream target genes. In humans, FOXO3 contains approximately 40 non-coding single-nucleotide polymorphisms (SNPs) that common to be found for example rs2802292, that consistently reported to be correlated with longevity in both Caucasian and Asian [14, 30]. In this study, FOXO3 rs2802292 was also detected in Indonesian elderlies’ population living at Jakarta, the GG genotype subjects had a significant higher average age (73.5 years) than the GT and TT genotypes. Subjects carrying the G allele had a longevity (71.18 years) than subjects carrying the T allele (69.51 years). This is in line with several previous studies, where the G allele at rs2802292 has an association with the longevity of humans from various populations [14]. Significantly, research has demonstrated that FOXO3 functions as a mediator in key biological processes, playing a crucial role in responding to oxidative stress, DNA damage, starvation, and caloric restriction. The ultimate outcome is an extension of lifespan and the prevention of diseases associated with aging [16]. Nevertheless, the blood pressure and blood glucose levels of the subjects in our study were not affected by the FOXO3 rs2802292, likely attributable to the fact that over half of the subjects have normal blood pressure, and the majority of them have normal blood sugar levels. Nutritional status, lifestyle, comorbid and environmental factors probably contribute more toward the change of blood pressure and blood glucose which was not analyzed in this study [31].
In general, SNPs can impact gene regulation through various mechanisms. They may function as splice-site variants, act as enhancer elements in transcription, cause changes in amino acids, or influence the structure of chromatin [32]. Numerous non-coding genetic variations, such as SNPs, found in loci associated with risk, selectively modify gene expression in a manner specific to both the disease and the tissue. This occurs by regulating the activity of cis-regulatory elements, particularly enhancers, through mechanisms involving the binding of transcription factors [33]. A recent discovery revealed that the 90 base pair sequence surrounding the intronic SNP rs2802292 in FOXO3 serves as an enhancer. Moreover, various cellular stress conditions were found to trigger the recruitment of Heat Shock Factor-1 (HSF1), leading to the promotion of FOXO3 expression [16]. HSF1 is a stress-induced transcription factor involved in a variety of physiological processes, including metabolism, development, senescence and controls multiple loci that participate in the stress response [34]. In the presence of nutrient, genotoxic, and oxidative stress, the homozygosity for the G-allele at rs2802292 enhances the expression of FOXO3 via HSF1. This is because the G-allele binds to HSF-1, activating downstream gene targets involved in antioxidant expression, metabolic regulation, and transcriptional programs for DNA repair. These effects occur in both the nucleus and mitochondria, resulting in heightened stress tolerance [35]. Therefore, the subjects with the intronic rs2802292 G-allele have the chance to reach healthy aging.
In old age, the immune system deteriorates, thus elderly were more susceptible to infection. Nevertheless, contribution of FOXO3 in regulating immune system reduce the risk of infection in elderly. For instance, FOXO3 protein plays an important role in age-dependent as it decreases in expression relates to anti-inflammatory behavior loss in old mice macrophages [29]. The inflammatory response was less severe in the monocytes of elderlies with the rs2802292 G-allele in FOXO3 gene due to the reduction in pro-inflammatory cytokines. Also, there was an increase in the production of anti-inflammatory cytokines, including IL-10 [36]. Dagdeviren et al [37] proved that overexpression IL-10 in old mice increase insulin sensitivity and decrease interferon-γ and interleukin-1β as the inflammatory cytokines in skeletal muscle. Although SASP components both pro-inflammatory (IL-1α, IL-6, IL-8) and anti-inflammatory (IL-10) cytokine level were not significant difference among genotypes group, however G-allele had a lower mean of inflammatory cytokine (IL-1α and IL-6) as well as had a higher anti-inflammatory cytokine (IL-10) compare to the T-allele. The IL-1α, IL-6 and IL-10 as the component of SASP appears to be influenced by polymorphism of FOXO3 rs2802292. However further research is needed using more subjects to obtain significancy impact. In addition, other SASP components such as chemokine, growth factors and proteases should be measured in the next research which are probably significantly affected by FOXO3 rs2802292 as well.
In conclusion, the frequency of FOXO3 rs2802292 G-allele in Indonesian elderly population was 0.385 and significantly detected in subjects with longevity. However, the FOXO3 rs2802292 polymorphism did not affect cytokines level as the component of SASP.
Acknowledgments
Authors would like to thank to Associate Professor Beben Benyamin, PhD (University of South Australia) for his support on this project. Moreover, thank to Directorate Research & Community Engagement Universitas Indonesia for funding this research (International Indexed Publication Q2/PUTI Q2 UI 2022, grant number: NKB-1242/UN2.RST/HKP.05.00/2022).
Funding
International Indexed Publication Q2/PUTI Q2 UI 2022, grant number: NKB-1242/UN2.RST/HKP.05.00/2022.
Authors contribution
Conception: Novi Silvia Hardiany, Febriana Catur Iswanti; Performance of work: Wahyu Nurfiyana; Interpretation of data: Novi Silvia Hardiany, Wahyu Nurfiyana, Febriana Catur Iswanti; Writing the Article: Novi Silvia Hardiany, Wahyu Nurfiyana.
Conflicts of interest
The authors have no conflict of interest to report.
References
[1] | World Population Ageing 2015: Highlights. World Population Ageing 2015: Highlights. 2016. |
[2] | McDonald RB , Ruhe RC . Aging and longevity: Why knowing the difference is important to nutrition research. Nutrients. (2011) ;3: (3):274–282. |
[3] | Martins R , Lithgow GJ , Link W . Long live FOXO: Unraveling the role of FOXO proteins in aging and longevity. Aging Cell. (2016) ;15: (2):196–207. |
[4] | Huang H , Tindall DJ . Dynamic FoxO transcription factors. J Cell Sci. (2007) ;120: :2479–2487. |
[5] | Webb AE , Brunet A . FOXO transcription factor key regulator ceullar quality control. Trends Biochem Sci. (2015) ;39: (4):159–169. |
[6] | Donlon TA , Morris BJ , Masaki KH , Chen R , Davy PMC , Kallianpur KJ et al. FOXO3, a resilience gene: Impact on lifespan, healthspan, and deathspan. J Gerontol. A Biol. Sci. Med. Sci. (2022) ;77: (8):1479–1484. |
[7] | Gui T , Burgering BMT . FOXOs: masters of the equilibrium. FEBS J. (2022) ;289: (24):7918–7939. |
[8] | Jimenez L , Silva A , Calissi G , Grenho I , Monteiro R , Mayoral-Varo V et al. Screening health-promoting compounds for their capacity to induce the activity of FOXO3. J Gerontol A Biol Sci Med Sci. (2022) ;77: (8):1485–1493. |
[9] | Orea-Soufi A , Paik J , Braganca J , Donlon TA , Willcox BJ , Link W . FOXO transcription factors as therapeutic targets in human diseases. Trends Pharmacol Sci. (2022) ;43: (12):1070–1084. |
[10] | Calissi G , Lam EW , Link W . Therapeutic strategies targeting FOXO tran-scription factors. Nat Rev Drug Discov. (2021) ;20: (1):21–38. |
[11] | McIntyre RL , Liu YJ , Hu M , Morris BJ , Willcox BJ , Donlon TA et al. Pharmaceutical and nutraceutical activation of FOXO3 for healthy longevity. Ageing Res Rev. (2022) ;78: :101621. |
[12] | Li Y , Wang WJ , Cao H , Lu J , Wu C , Hu FY et al. Genetic association of FOXO1A and FOXO3A with longevity trait in Han Chinese populations. Hum Mol Genet. (2008) ;18: (24):4897–4904. |
[13] | Sun L , Hu C , Zheng C , Qian Y , Liang Q , Lv Z et al. FOXO3 variants are beneficial for longevity in Southern Chinese living in the Red River Basin: A case-control study and meta-analysis. Sci Rep. (2015) ;5: :1–7. |
[14] | Bao JM , Song XL , Hong YQ , Zhu HL , Li C , Zhang T et al. Association between FOXO3A gene polymorphisms and human longevity: A meta-analysis. Asian J Androl. (2014) ;16: (3):446–452. |
[15] | Mao YQ , Liu JF , Han B , Wang LS . Longevity-associated forkhead box O3 (FOXO3) single nucleotide polymorphisms are associated with type 2 diabetes mellitus in chinese elderly women. Med Sci Monit. (2019) ;25: :2966–2975. |
[16] | Sanese P , Forte G , Disciglio V , Grossi V , Simone C . FOXO3 on the Road to Longevity: Lessons From SNPs and Chromatin Hubs. Comput Struct Biotechnol J. (2019) ;17: :737–745. |
[17] | Byun HO , Lee KY , Kim JM , Yoon G . From cell senescence to age related disease: differential mechanisms of action of senescence-associated secretory phenotype. BMB Rep. (2015) ;48: (10):549–558. |
[18] | Borodkina AV , Deryabin PI , Giukova AA , Nikolsky NN . Social life ofsenescence cells: What is SASP and why study it? Acta Naturae. (2018) ;10: (36):4–14. |
[19] | Cuollo L , Antonangeli F , Santoni A , Soriani A . The Senescence-Associated Secretory Phenotype and Age-Related Diseases. Biology (Basel). (2020) ;9: (485):1–16. |
[20] | Noureddine H , Gary-Bobo G , Alifano M , Marcos E , Saker M , Vienney N et al. Pulmonary artery smooth muscle cell senescence is a pathogenic mechanism for pulmonary hypertension in chronic lung disease. Circ Res. (2011) ;109: (5):543–553. |
[21] | Campisi J , Sndersen J , Kapahi P , Melov S . Cellular senescence: a link between cancer and age-related degenerative disease? Semin Cancer Bio. (2011) ;21: (6):354–359. |
[22] | Lopes-Paciencia S , Saint-Germain E , Rowell MC , Ruiz AF , Kalegari P , Ferbeyre G . The senescence-associated secretory phenotype and its regulation. Cytokine. (2019) ;117: :15–22. |
[23] | Salminen A , Kauppinen A , Kaarniranta K . Emerging role of NF-κB signaling in the induction of senescence-associated secretory phenotype (SASP). Cell Signal. (2012) ;24: (4):835–845. |
[24] | Su H , Bidere N , Lenardo M . Another Fork in the Road: that diseasedevelopment is associated with NF-κB. Immunity. (2004) ;21: :133–134. |
[25] | Thompson MG , Larson M , Vidrine A , Barrios A , Navarro F , Meyers K et al. FOXO3–NF-κB RelA protein complexes reduce proinflammatory cell signaling and function. J Immunol. (2015) ;195: (12):5637–5647. |
[26] | Lin L , Hron JD , Peng SL . Regulation of NF-κB, Th activation, and autoinflammation by the forkhead transcription factor Foxo3a. Immunity. (2004) ;21: (2):203–213. |
[27] | Hardiany NS , Sucitra , Paramita R . The mRNA expression of Forkhead box O3a (FOXO3a) as a longevity-Associated gene in leucocytes of elderly women. J Pak Med Assoc. (2021) ;71: (2):S74–77. |
[28] | Forte G , Grossi V , Celestini V , Lucisano G , Scardapane M , Varvara D et al. Characterization of the rs2802292 SNP identifies FOXO3A as a modifier locus predicting cancer risk in patients with PJS and PHTS hamartomatous polyposis syndromes. BMC Cancer. (2014) ;14: (1):1–6. |
[29] | Stefanetti RJ , Voisin S , Russell A , Lamon S . Recent advances in understanding the role of FOXO3 [version 1; referees: 4 approved]. FResearch. (2018) ;7: :1–8. |
[30] | Willcox BJ , Donlon TA , He Q , Chen R , Grove JS , Yano K et al. FOXO3A genotype is strongly associated with human longevity. Proc Natl Acad Sci U S A. (2008) ;105: (37):13987–13992. |
[31] | Tsimihodimos V , Gonzalez-Villalpando C , Meigs JB , Ferrannini E . Hypertension and diabetes mellitus coprediction and time trajectories. Hypertension. (2018) ;71: (3):422–428. |
[32] | Long HK , Prescott SL , Wysocka J . Ever-changing landscape: Transcriptional enhancers in development and evolution. Cell. (2016) ;167: (5):1170–1187. |
[33] | Cowper-Sallari R , Zhang X , Wright JB , Bailey SD , Cole MD , Eeckhoute J et al. Breast cancer risk-associated SNPs modulate the affinity of chromatin for FOXA1 and alter gene expression. Nat Genet. (2012) ;44: (11):1191–1198. |
[34] | Anckar J , Sistonen L . Regulation of HSF1 function in the heat stress response: Implications in aging and disease. Annu Rev Biochem. (2011) ;80: :1089–1115. |
[35] | Grossi V , Forte J , Sanese P , Peserico A , Tezil T , Signorile ML et al. The longevity SNP rs2802292 uncovered: HSF1 activates stress-dependent expression of FOXO3 through an intronic enhancer. Nucleic Acid Res. (2018) ;46: (11):5587–5600. |
[36] | Morris BJ , Willcox DC , Donlon TA , Willcox BJ . FOXO A Major Gene for Human Longevity –A Mini-Review. Gerontology. (2015) ;61: (6):515–525. |
[37] | Dagdeviren S , Jung DY , Friedline RH , Noh HL , Kim JH , Patel PR et al. IL-10 prevents aging-associated inflammation and insulin resistance in skeletal muscle. FASEB J. (2016) ;31: (2):701–710. |




