Physicochemical, microbial, and sensory properties of yogurt supplemented with flaxseeds during fermentation and refrigerated storage
Abstract
BACKGROUND:
Flaxseeds are miraculous seeds because of their richness in omega-3, fibers and other healthy compounds, they can be used as a natural remedy for several diseases.
OBJECTIVE:
This study was carried out to determine the feasibility of supplementing flaxseed powder to yogurt at 3% to benefit from its functional effects such as its richness in proteins, dietary fiber, polysaccharides, polyphenolic compounds and essential fatty acids for health.
METHODS:
Two types of milk powder were used for the manufacture of yogurt: skimmed milk powder at 15% (w/v) and whole milk powder at 13.7% (w/v). Physicochemical (pH and titratable acidity), microbial (Streptococcus thermophilus and Lactobacillus bulgaricus) and sensory (texture, odour, taste and color) properties of this supplementation were evaluated during the fermentation time and storage at 4°C.
RESULTS:
The addition of ground linseed (GFS) decreased the pH values and increased the titratable acidity values during fermentation and refrigerated storage, especially in the case of yogurt manufactured using whole milk powder, where the average pH of samples decreased from 4.37 to 4.20 after 28 days storage while the average titratable acidity increased from 78 to 105°D. Sensory evaluation revealed that supplemented yogurt had better sensory texture, taste and odour scores than the control sample. Supplementation of GFS significantly increased the average values of lactic acid bacteria CFU counts but at values not exceeding 1.25×108 CFU/ml for S. thermophilus and 0.85×107 CFU/ml for Lactobacilllus bulgaricus indicating the possibility of prolonging the shelf life of yogurt.
CONCLUSION:
The study concludes that the supplementation of yogurt by GFS is appreciated by the tasters and allows to keep a desirable physicochemical properties of the dairy product.
1Introduction
Yogurt is the most popular fermented fresh dairy product and most consumed in the world [1, 2]. It has several beneficial health effects because of its functional properties of viable microorganisms and the allegation of health recently authorized the consumption of yogurt to improve lactose digestion for individuals with intolerance for lactose [3].
The current dynamic of the market for dairy products, including yogurt, forces industry to constantly formulate new recipes. These recipes must meet the demands of consumers who expect natural products with low sugar, low fat and without preservatives while respecting the mouth feel and aroma, which largely determine product acceptability [4].
In recent years, many different food ingredients, such as dietary fiber [5], evening primrose oil [6], β-glucan [7, 8], nanopowdered chitosan [9], green and black teas [10] and lentil flour [11] have been included in yogurt formulations to improve nutritional value.
Flax (Linum usitassimum) belonging to the Lineaceae family, is an herbaceous annual plant with blue flowers, which produces small flat seeds varying from golden yellow to reddish or brown. Flaxseed has a crunchy texture and nutty flavor [12, 13].
Flax is one of the oldest commercial crops, the first traces of its use dating back to 8000 BC in Turkey [14]. Its name Linum usitatissinum, translated from the Latin “ lin most useful”, refers to its multiple uses. Currently, linen is used in the fields of textile industry (fiber), food (seed and oil) and chemical (oil) [15].
Presently, flaxseed has new prospects as functional food because of consumer’s growing interest for food with superb health benefits. Owing to its excellent nutritional profile and potential health benefits, it has become an attractive ingredient in the diets particularly designed for specific health benefits [16]. Flaxseed contains protein, dietary fiber, polysaccharides, polyphenolic compounds and essential fatty acids for health that help to prevent somes diseases. Its content of alpha-linolenic acid (ALA), an essential omega-3 fatty acids, dietary fiber and lignans has a special interest to consumers, food manufacturers andresearchers [15].
Flax is unique among oilseeds because of its high content of omega-3 fatty acid ALA. Flaxseeds contain between 35% and 45% oil, ALA representing about 50% to 60% fatty acids [17], it is one of the essential polyunsaturated fatty acid and reported to exhibit anti-inflammatory, anti-thrombotic and anti-arrhythmic properties [18]. Flax also contains linoleic acid (LA), an omega-6 essential fatty acid. Flax oil contains three times more omega-3 fatty acids than omega-6 fatty acids. The triple unsaturation of the alpha-linolenic acid (C18:3, 50 to 60%) gives linseed oil high reactivity concerning oxygen and thus a high drying ability. This property is interesting for oleochemical applications [19].
Flax seeds contain about 28% dietary fiber, which consist of 60% to 80% insoluble fiber and 20% to 40% soluble fiber (mucilage) [20].
Of all seeds and cereals, flaxseed is the one with the highest lignan content [21]. It is rich in secoisolariciresinol diglycoside (SDG), a precursor of mammalian lignans that are enterodiol and enterolactone [22]. SDG when ingested by humans influences a wide range of biological systems that keep humans healthy [23].
The growing popularity of flaxseed is due to its health benefits including the reduction of cardiovascular disease and cancer risk, particularly the breasts and prostate cancer, anti-inflammatory activity, a laxative effect and reduction of menopausal symptoms and osteoporosis [24]. Flaxseed oil supplementation for about four weeks resulted in protecting the mice against Streptococcus pneumonia infection [25]. Flaxseed and its oil reduces the growth of tumors at the later stage of carcinogenesis whereas mammalian lignin precursor exert the greatest inhibitory effect on the growth of new tumors [26].
Flaxseed possesses antioxidant and hepatoprotective properties. Several studies advocated the cholesterol lowering benefits of flaxseed meal [27–29]. A study on hypercholesterolemic rats fed on flaxseed chutney supplemented diet (15%) revealed significant reduction in LDL cholesterol and total serum cholesterol and no change in HDL cholesterol. In CCl4 intoxicated rats, lipid peroxidation products were neutralized by flaxseed lignans [30].
There is no report in the literature on the production of yogurt with flaxseed. Therefore, the objectives of this study were to investigate the possibility of adding ground flaxseeds to yogurt and the effects of this addition on the physicochemical, microbiological and sensory properties of the product during fermentation and storage.
2Materials and methods
2.1Flaxseeds
Flaxseeds purchased from a local supermarket in Algiers (Algeria) were sieved (1 mm mesh size), cleaned manually to remove all debris and gravel then ground to powder with Universal Mill M20 IKA IMLAB. The particle size of powder was standardized to 450 and 355 μm using Rotachoc Chopin (50 Hrz).
2.2Preparation of yogurt
Three lots of milk bases were prepared by reconstituting milk powder: the skimmed milk (fat content <1%) at 15% (w/v) [31] and the whole milk (28% fat) at 13.7% (w/v) [32] in distilled water, 12.5% of sugar (w/w) and 3% (w/w) ground flaxseeds were added to these two batches. The amount of flaxseeds added is the result of a preliminary sensory analysis. The third one was prepared as a control using whole milk powder at 13.7% (w/v) added with 12.5% sugar (w/w) in distilled water.
The reconstituted milk with the sugar was homogenized and heated at 95°C for 5 min. Subsequently, it was cooled to the incubation temperature (42–43°C) and inoculated with 10% (w / w) of commercial natural yogurt (fat content <1%) containing lactic ferments Streptococcus thermophilus and Lactobacillus delbrückii subsp. bulgaricus.
The incubation was carried out until complete gelation of the yogurt (4 hours), then, the incubation was stopped by cooling to 10°C and the samples were stored at this temperature for 24 h [9].
After stabilization, each yogurt sample was stored for 0, 7, 14, 21 and 28 days at 4°C in a refrigerator to evaluate the physicochemical, microbiological and sensory properties. Each batch of yogurt was prepared in triplicate.
2.3Chemical analysis
The pH of each yogurt sample was measured using an electronic pH meter laboratory (PH 210 Hanna Instruments HI 2210).
The titratable acidity of the samples was determined according to the IDF/ISO/AOAC Standard [33] by a potentiometric method (real acidity) which is a measurement technique allowing to passively evaluate the potential of a solution between two electrodes while affecting the solution in a minimal way. It is expressed in grams of lactic acid per 100 g of product, and is defined by the following equation:
Where:
At: titratable acidity
V is the volume (ml) of 0.1 M sodium hydroxide;
m is the mass (g) of the sample;
0.9 is the conversion factor of the lactic acid.
All samples were measured in triplicate.
The results are expressed in degree Dornic (°D)
2.4Consumer acceptance test
Sensory analyses (color, texture, taste and odour) of yogurts supplemented with GFS and yogurt control were evaluated by 10 tasters (members of Laboratory Soft Technology, Valorization, Physical Chemistry of Biological Materials and Biodiversity), 3 males and 7 women, of 22 to 50 years old recruited according to their motivation and availability to participate to the study.
The analysis is performed at room temperature, yogurt samples (10 g) were served in randomized order blinded in a transparent plastic cup coded. Consumers were asked to note their preference for acceptability using paper score-sheets.
Three coded samples were provided to panels at the same time. The consumer acceptance was determined using a 5-point scale [32]:
1 point: nonstandard product unfit for consumption
2 points: unsatisfactory quality product but can be used
3 points: product of satisfactory quality
4 points: product of good quality
5 points: product of excellent quality
2.5Microbiological analysis
Yogurt microbiological analysis were performed to determine the influence of the addition of flaxseeds on the kinetic of growth of lactic ferments. For viable counts, M17 culture media was used to quantify the S. thermophilus and MRS agar (agar of Man, Rogosa, Sharpe) for L. bulgaricus. Plates were incubated under anaerobic conditions at 37°C for 48 h and 37°C for 72 h, respectively [34]. Colonies were counted only in Petri dishes containing between 30 and 300 colonies [35, 36]. Samples were analyzed in triplicate.The number of colonies is expressed in CFU/ml according to the following formula:
Where:
CFU: unit forming colony
n:colony counts
v: volume collected
d: dilution
Only the preferred supplemented yogurt with GFS and the yogurt control will be considered for microbiological analysis.
During fermentation and storage period (28 days) at 4°C, pH, titratable acidity and the growth of lactic acid bacteria were measured.
2.6Statistical analysis
All statistical analyses were performed using SPSS version 21.0, IBM STATISTICS (USA). An ANOVA was performed using the general linear models procedure to determine significant differences among the samples. For Chemical analysis, means were compared by using Duncan’s test. For microbiological analysis, as we have two samples, means were compared by using Student’s test and the significance was defined at the 5% level.
3Results and discussion
3.1Acidification evolution during yogurt fermentation
Acidification evolution in yogurt fermentation is presented in Fig. 1. The addition of 3% ground flaxseed increased the acidification level, as compared to the control and this effect was found to be statistically significant (p < 0.05) after 1 h of incubation, These results are similar to those found by Zare et al. [11] in the study on yogurt supplemented with lentil powder. In supplemented yogurts, those with whole milk powder have pH slightly lower and it is statistically significant (p < 0.05) compared to samples with skim milk powder after 2 h of incubation.
Fig.1
Change in pH as a function of incubation time during the acidification of yogurt with whole milk powder (Y1) and yoghurt with skimmed milk powder (Y2) supplemented with 3% ground flaxseeds (GFS) and yogurt with whole milk powder as control. Bars with different letters within the same fermentation time differ significantly (p < 0.05).
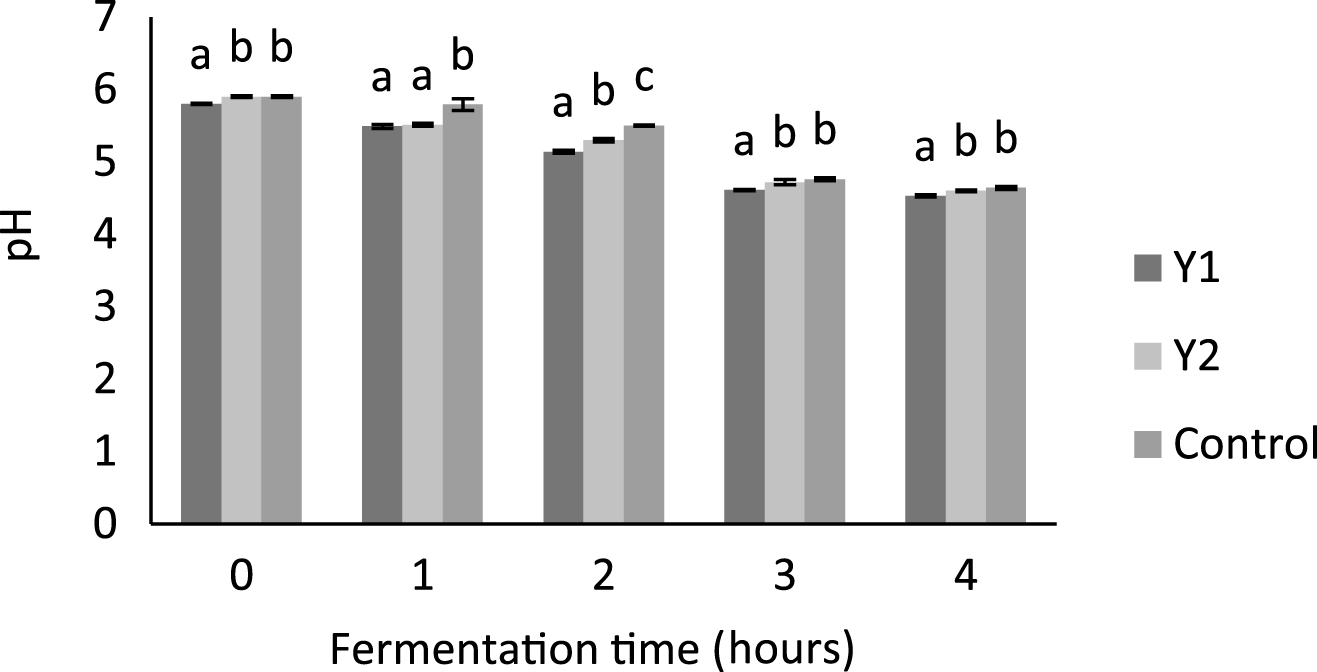
During fermentation, the casein particles aggregate as the pH approaches 4.6 resulting from the lactic acid production. This, together with the denaturation of the whey protein and its association with caseins induce an increase of gel viscosity and firmness [37–39].
Rate of acidification is one of the important detriments of yogurt making. So it is important to achieve a balance between strength of the gel as well as commercially feasible fermentation time. During fermentation time, lactic acid production increases with the growth of the starter culture S. thermophilus and L. bulgaricus.
The values of titratable acidity for all samples studied increased during fermentation (Table 1). The decrease in pH values and increase in titratable acidity values for yogurt samples during fermentation obtained in our study are in agreement with results obtained by Pelaes Vital et al. [40] for yogurt supplemented with Pleurotus ostreatus aqueous extract.
Table 1
Changes in titratable acidity (°D) during the fermentation time of yogurt with whole milk powder (Y1) and yogurt with skimmed milk powder (Y2) supplemented with ground flaxseeds. Control (yogurt with whole milk powder)
| Fermentation time (h) | Yogurt types | Titratable acidity (°D) |
| 0 | Y1 | 24.00±4.35 a |
| Y2 | 22.00±1.73 a | |
| Control | 20.00±2.64 a | |
| 1 | Y1 | 27.00±1.00 a |
| Y2 | 25.00±1.00 a | |
| Control | 25.00±1.73 a | |
| 2 | Y1 | 43.00±3,60 a |
| Y2 | 40.00±1.73 a | |
| Control | 27.00±1.73 b | |
| 3 | Y1 | 60.00±3.00 a |
| Y2 | 57.00±1.73 a | |
| Control | 52.00±1.00 b | |
| 4 | Y1 | 65.00±1.00 a |
| Y2 | 59.00±3.60 b | |
| Control | 57.00±2.64 b |
Values with different letters within the same column differ significantly (p < 0.05).
The time required to reach a pH lower or equal to 4.6 was shorter in the case of supplemented yogurts due to richness of flaxseeds of insoluble dietary fiber that promote the growth of lactic ferments S. thermophilus and L. bulgaricus (Fig. 2).
Fig.2
Sensory properties of yogurt with whole milk powder Y1 and yogurt with skimmed milk powder Y2 supplemented with 3% ground flaxseed and yogurt with whole milk powder as control after production (T1 to T10: tasters).
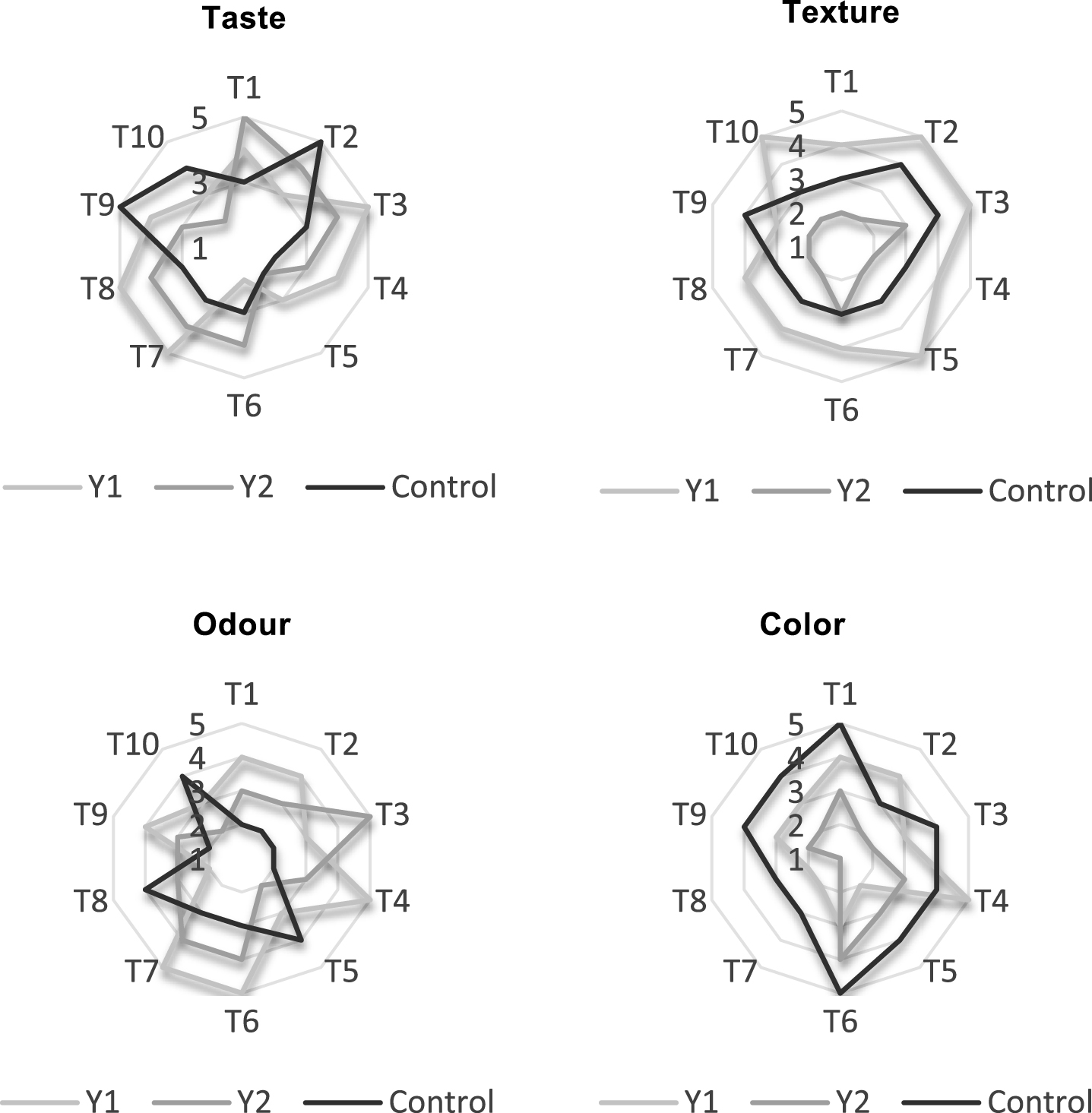
In our study, we found that supplemented whole yogurt gelation was faster; this can be explained by: first the presence of soluble fiber which is a viscous seed coat gum composed of neutral (75%) and acidic (25%) monosaccharides [41], flaxseed gum possesses good gelling, foaming and emulsifying capacity, and can potentially replace gum arabic in food emulsions [42–44]; and second the fat which plays an important role in controlling the firmness/viscosity and perceived creaminess of yogurt due to the formation of a larger number of smaller fat particles during homogenisation when they are stabilised by milk proteins and interact with the protein matrix [45].
3.2Acidification evolution during yogurt refrigerated storage
During the fermentation and storage at 4°C, pH, titratable acidity and lactic acid bacteria growth were measured at 7 days interval until 28 days.
The pH values decreased significantly during storage for all yogurts samples. Similar pH changes were observed during refrigerated storage at 4°C for 28 days for probiotic yogurt fortified with fiber-rich pineapple peel powder [46].
The normal pH of commercial yogurt products ranged from 4.0 to 4.4 [8, 47]. In the current study, increasing the storage period from 1 to 28 days considerably decreased the pH values for the control sample from 4.51 to 4.29. However it was observed that the pH values of supplemented yogurts were not considerably altered during storage for 28 days proving that the quality of yogurts enriched with GFS was not affected during storage. According to these results, it is meditated that adding flaxseeds into yogurt could extend the shelf life as found by Seo et al. [9] for yogurt supplemented with nanopowdered chitosan.
The addition of GFS into yogurt samples significantly increased the values of titratable acidity (Table 2). Furthermore, the values of titratable acidity for all samples studied were slightly increased when stored at 4°C for 28 days. These results are in accordance with those found by Ozturkoglu-Budak et al. [48] and Raho Ghalem et al. [49] who have respectively worked on yogurt supplemented with dried nut and yogurt supplemented with Rosmarinus officinalis oil.
Table 2
Effect of yogurt supplementation with ground flaxseeds on pH and titratable acidity (D°) during 28 days of storage at 4°C
| Yogurt types | Storage period (days) | ||||
| 1 | 7 | 14 | 21 | 28 | |
| pH | |||||
| Control | 4.51±0.03 (b) | 4.48±0.01 (b) | 4.33±0.06 (a) | 4.34±0.03 (b) | 4.29±0.01 (c) |
| Y1 | 4.37±0.01 (a) | 4.35±0.03 (a) | 4.27±0.02 (a) | 4.22±0.02 (a) | 4.20±0.01 (a) |
| Y2 | 4.41±0.01 (a) | 4.38±0.02 (a) | 4.29±0.04 (a) | 4.26±0.02 (a) | 4.24±0.02 (b) |
| Titratable acidity (D°) | |||||
| Control | 69.00±2.64 (b) | 73.00±1.00 (c) | 85.00±3.60 (b) | 87.00±1.73 (c) | 91.00±1.00 (c) |
| Y1 | 78.00±1.00 (a) | 83.00±2.64 (a) | 90.00±1.73 (a) | 100.00±1.73 (a) | 105.00±2.64 (a) |
| Y2 | 75.00±1.73 (a) | 79.00±1.73 (b) | 88.00±1.00 (a) | 92.00±2.64 (b) | 97.00±1.73 (b) |
Values with different letters within the same column differ significantly (p < 0.05).
3.3Sensory acceptance
Figure 3 shows the consumer acceptance of yogurts. Supplementation of yogurts by GFS largely influenced all the parameters analyzed. The control sample was acceptable. The substitution of the whole milk powder by the skimmed milk powder gave the lowest notes on texture and color. This can be explained by the rise in the surface of the ground flaxseed because of their richness in fat; on the visual point this effect is considered as undesirable. Moreover, the skimming of the milk gives a yogurt less firm, less creamy with an important syneresis. About taste and odour, tasters preferred Y1 to Y2. This preference can possibly be explained by the taste and smell of flaxseeds which are similar to those of hazelnuts. The same preference has been given to Y1 for texture due to the gelling power of linseed which gives firmer yogurts. Overall, in terms of ranking, tasters preferred Y1 followed by control and lastly Y2.
Fig.3
Streptococcus thermophilus (A) and Lactobacillus delbrueckii subsp. bulgaricus (B) counts in supplemented yogurts with 0% (control) and 3% of GFS during 28 days of storage at 4°C. Bars with different letters within the same fermentation time differ significantly (p < 0.05).
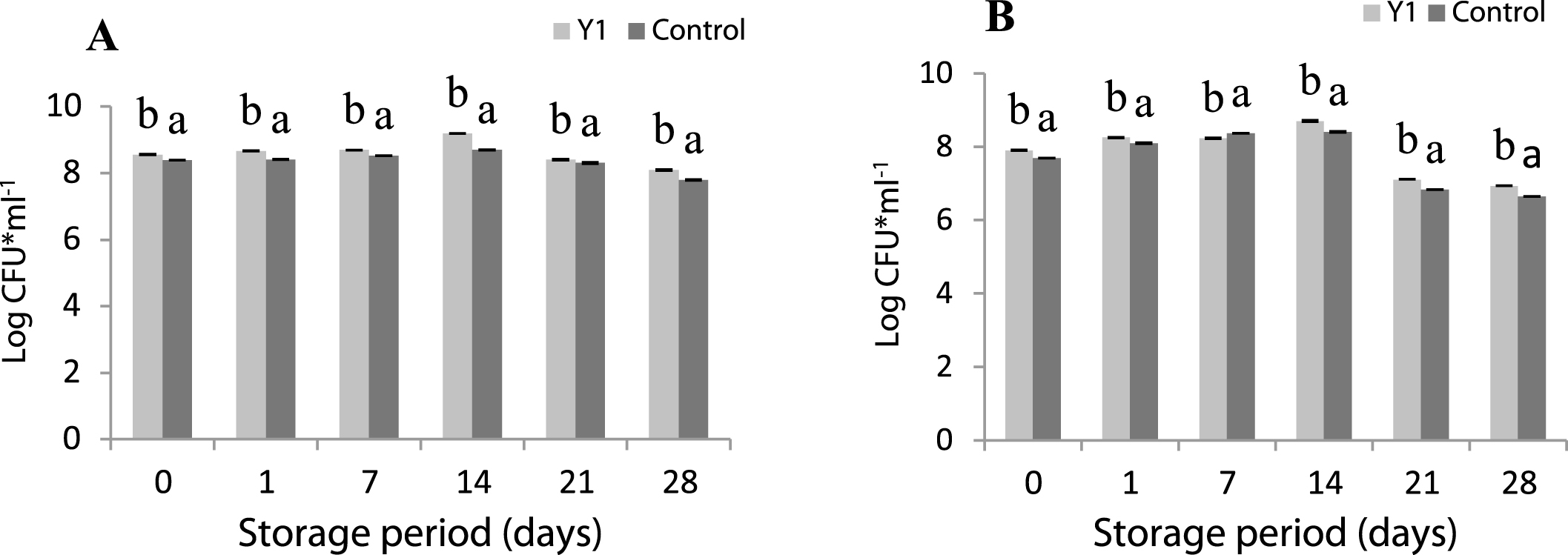
3.4Microbiological analysis
The yogurt simple Y2 was not considered for the microbiological analysis because it was not preferred by tasters in the sensorial analysis.
Lactobacillus delbrueckii ssp. bulgaricus produces essential amino acids because of its proteolytic nature, and the symbiotic relationship of L. delbrueckii ssp. bulgaricus and S. thermophilus is well established; the latter organism also produces growth factors for the former organism [50].
During the period of fermentation and the period of storage (Table 3 and Fig. 2), the growth of S. thermophilus is higher than that of L. bulgaricus. The same effect has been observed by Muniandy et al. [51] in the study on comparison of the effect of green, white and black tea on Streptococcus thermophilus and Lactobacillus spp. in yogurt during refrigerated storage.
Table 3
Effect of yogurt supplementation with ground flaxseeds (GFS) on viable counts (CFU/ml) during fermentation
| Yogurt types | Fermentation time (hours) | ||
| 1h30 | 2h30 | 3h30 | |
| Viability of S. thermophilus (CFU/ml) | |||
| Control | 9.02*107 ± 6.00*105 (a) | 9.98*107 ± 1.05*106 (a) | 2.45*108 ± 1.73*106 (a) |
| Y1 | 9.99*107 ± 1.01*106 (b) | 1.13*108 ± 1.73*106 (b) | 3.63*108 ± 2.64*106 (b) |
| Viability of L. bulgaricus (CFU/ml) | 1h30 | 2h30 | 3h30 |
| Control | 9.00*106 ± 2.00*104 (a) | 1.65*107 ± 5.56*106 (a) | 4.88*107 ± 6.08*105 (a) |
| Y1 | 9.30*106 ± 3.80*105 (a) | 5.21*107 ± 6.92*105 (b) | 7.94*107 ± 3.60*105 (b) |
Values with different letters within the same column differ significantly (p < 0.05).
The viable counts of S. thermophilus and L. bulgaricus increased during fermentation time in al samples. However, the addition of GFS causes greater significant growth of lactic acid bacteria compared to the control (Table 3). This, can be explained by the richness of flaxseeds in nutrients and dietary fibers which provided additional source of carbohydrate for lactic acid bacteria and had a strong positive effect on the activity of some lactic acid bacteria strains [52].
The growth kinetics of the lactic acid bacteria during 28 days of storage at 4°C is shown in Fig. 2. The growth rate obtained after one day storage for both samples was greater than that recommended by the Codex Alimentarius (107 CFU/g) [53]. The maximum concentration of both strains was obtained after two weeks of storage for all yogurts.
The addition of GFS significantly increased the number of viable lactic acid bacteria compared to control. Indeed, S. thermophilus reached a mean concentration of 9.20 log10 CFU*ml-1 at 14th day, which was higher than the control sample (8.70 log10 CFU*ml-1). Lactobacillus bulgaricus reached a mean concentration of 8.70 log10 CFU*ml-1 in fortified yogurt compared to 8.40 log10 CFU*ml-1 in the control at the same day.
The viability of strains decreased from the 21st day of refrigerated storage especially in yogurt control. At the end of the storage period, in supplemented yogurts, S. thermophilus achieved a mean concentration of 8.10 log10 CFU*ml-1, which was slightly higher than the control (7.80 log10 CFU*ml-1). The number of Lactobacillus bulgaricus found was 6.93 log10 CFU*ml-1 in fortified yogurt compared to 6.64 log10 CFU*ml-1 in the control.
The reduction of S. thermophilus and Lactobacillus bulgaricus during the last two weeks of storage could be attributed to L. delbrueckii ssp. bulgaricus which produces lactic acid during refrigerated storage. This process is known in the industry as post-acidification. Acid produced during refrigerated storage (post-acidification) causes loss of viability of probiotic bacteria [50, 54, 55].
4Conclusion
Our study was designed to develop a yogurt supplemented with ground flaxseeds and to evaluate the effects of this supplementation on the physicochemical, microbial, and sensory properties of the product during fermentation and storage at 4°C.
The data on the pH, titratable acidity, microbial, and sensory analysis obtained from the current study indicated that fortifying yogurt with GFS produced an acceptable product with potential beneficial health effects. The obtained pH and titratable acidity values allowed us to deduce that the addition of GFS to yogurt could extend the shelf life.
Concerning the sensory evaluation, the panelists gave the best notes for the odour, the texture and the taste to the whole milk powder yogurt supplemented; for the color, the control yogurt was the most preferred.
The multiplication of lactic acid bacteria was greater in supplemented yogurts which maintained a high level of S. thermophilus (1.25*108 CFU/ml), and Lactobacilllus spp. (0.85*107 CFU/ml) counts throughout the refrigerated storage period, and these values could be a desirable characteristic of the product to confer health benefits to consumers upon regular consumption.
Based on these results, we may deduce that the enrichment of yogurt with flaxseeds appears to be important, as it provides health benefits and a convenient food that satisfies consumer interest.
Conflict of interest
None to report.
References
[1] | Nakassaki K , Yanagisawa M , Kobayashi K . Microbiological quality of fermented milk produced by repeated-batch culture. Journal of Bioscience and Bioengineering. (2008) ;105: (1):73–6. doi: 10.1263/jbb.105.73 |
[2] | Ju Noh Hyo , Seo Min Hye , Lee Jun Ho , Chang Yoon Hyuk . Physicochemical and Sensory Properties of Yogurt Supplemented with Corni fructus during Storage. Preventive Nutrition and Food Science. (2013) ;18: (1):45–9. doi: 10.3746/pnf.2013.18.1.045 |
[3] | EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of health claims related to Yoghurt cultures and improving lactose digestion (ID 1143, 2976) pursuant to Article 13(1) of Regulation (EC) No 1924/2006: In EFSA Journal. (2010) ;8: (10):18. doi: 10.2903/j.efsa.2010.1763. Available online: http://www.efsa.europa.eu/efsajournal.htm. |
[4] | Paci Kora Ahmad . Physico-chemical and sensory interactions in stirred and flavored yoghurt: What are the respective impacts on the perception of texture and flavor? National Agricultural Institute. Paris-grignon; (2004) . |
[5] | Staffolo MD , Bertola N , Martino M , Bevilacqua A . Influence of dietary fiber addition on sensory and rheological properties of yogurt. Int Dairy J. (2004) ;14: :263–8. doi: 10.1016/j.idairyj.2003.08.004 |
[6] | Lee SJ , Hwang JH , Lee S , Ahn J , Kwak HS . Property changes and cholesterol-lowering effect in evening primrose oil-enriched and cholesterol-reduced yogurt. Int J Dairy Technol. (2006) ;60: (1):22–30. |
[7] | Gee VL , Vasanthan T , Temelli F . Viscosity of model yogurt systems enriched with barley β-glucan as influenced by starter cultures. Int Dairy J. (2007) ;17: :1083–8. doi: 10.1016/j.idairyj.2007.01.004 |
[8] | Sahan N , Yasar K , Hayaloglu AA . Physical, chemical and flavor quality of non-fat yogurt as affected by a β-glucan hydrocolloidal composite during storage. Food Hydrocoll. (2008) ;22: :1291–7. doi: 10.1016/j.foodhyd.2007.06.010 |
[9] | Seo MH , Lee SY , Chang YH , Kwak HS . Physicochemical, microbial, and sensory properties of yogurt supplemented with nanopowdered chitosan during storage. J Dairy Sci. (2009) ;92: (12):5907–16. doi: 10.3168/jds.2009-2520 |
[10] | Jaziri I , Ben Slama M , Mhadhbi H , Urdaci MC , Hamdi M . Effect ofgreen and black teas (Camellia sinensis L. ) on the characteristicmicroflora of yogurt during fermentation and refrigerated storage.Food Chem. (2009) ;112: :614–20. doi: 10.1016/j.foodchem.2008.06.017 |
[11] | Zare F , Boye JI , Orsat V , Champagne C , Simpson BK . Microbial, physical and sensory properties of yogurt supplemented with lentil flour. Food Research International. (2009) ;44: :2482–8. doi: 10.1016/j.foodres.2011.01.002 |
[12] | Morris HM . Flax: A health and nutrition primer. Flax Council of Canada. Winnipeg: Canada; (2007) . |
[13] | Rubilar M , Gutiérrez C , Vedugo M , Shene C , Sineiro J . Flaxseedas a source of functional ingredients. Journal of soil Science,Plant Nutrition. (2010) ;10: (3):373–7. doi.org.sci-hub.cc/ 10.4067/S0718-95162010000100010 |
[14] | Van Zeiste W . Palaeobotanical results of the 1970 season at Cayonu. Helinium: Turkey. (1972) . |
[15] | Vaisey-Genser MA , Morris D . Introduction History of the cultivation and uses of flaxseed. In: Muir AD , Wescott ND , editors. Flax, The genus Linum: CRC Press; (2003) . p. 1–22. |
[16] | Oomah D . Flaxseed as functional source. J Sci Food Agric. (2001) ;81: :889–94. doi: 10.1002/jsfa.898 |
[17] | Wiesenborn D , et al. Proceedings of the 50th Flax Institute of the United States, (2002) . |
[18] | Simopoulos AP . Essential fatty acids in health and chronic diseases. Am J Clin Nutr. (1999) ;70: :560–9. |
[19] | Knörr W , Daute P , Grützmacher R , Höfer R . Development of new fields of application for linseed oil. Lipid / Fett. (1995) ;97: :165–9. |
[20] | Bhatty RS . Nutrient composition of whole flaxseed and flaxseed meal. In: Cunnane SC , Thompson LU editors. Flaxseed in human nutrition. Champaign: AOCS Press; (1995) . p. 22–42. |
[21] | Meagher LP , Beecher GR . Assessment of data on the lignan content of foods. J Food Compos Anal. (2000) ;13: :935–47. doi: 10.1006/jfca.2000.0932 |
[22] | Bierenbaum ML , Reichstein R , Watkins TR . Reducing atherogenic risk in hyperlipidemic humans with flaxseed supplementation: A preliminary report. J Am Coll Nutr. (1993) ;12: :501–4. |
[23] | Muir AD . Flax lignans: New opportunities for functional foods. Food Sci Technol Bull Funct Foods. (2010) ;6: (6):61–79. |
[24] | Goyal A , Sharma V , Upadhyay N , Gill S , Sihag M . Flax and flaxseed oil: An ancient medicine & modern functional food. Journal of Food Science and Technology-Mysore. (2014) ;51: (9):1633–53. doi: 10.1007/s13197-013-1247-9 |
[25] | Saini A , Harjai K , Mohan H , Punia RPS , Chhibber S . Long-term flaxseed oil supplementation diet protects BALB/c mice against Streptococcus pneumonia infection. Med Microbiol Immunol. (2010) ;199: :27–34. doi: 10.1007/s00430-009-0132-7 |
[26] | Thompson LU , Rickard SE , Orcheson LJ , Seidl MM . Flaxseed and its lignin anditsoil components reduce mammary tumor growth at a late stage of carcinogenesis. Carcinogenesis. (1996) ;17: (6):1373–76. |
[27] | Cunnane SC , et al. High linolenic acid flaxseed (Linumusitatissimum): Some nutritional properties in humans. Br J Nutr. (1993) ;69: :443–53. |
[28] | Ridges L , Sunderland R , Moerman K , Meyer B , Astheimer L , Howe P . Cholesterol lowering benefits of soy and linseed enriched foods. Asia Pac J Clin Nutr. (2001) ;10: (3):204–11. |
[29] | Bhathena SJ , Ali AA , Haudenschild C , Latham P , Ranich T , Mohamed AI , et al. Dietary flaxseed meal is more protective than soyprotein concentrate against hypertriglycerdemia and steatosis ofthe liver in an animal model of obesity. J Am Coll Nutr. (2003) ;22: :157–64. |
[30] | Shakir KAF , Madhusudan B . Hypocholesterolemic and hepatoprotective effects of flaxseed chutney: Evidence from animal studies. Int J Clin Biochem. (2007) ;22: (1):117–21. doi: 10.1007/BF02912893 |
[31] | Sanz T , Salvador A , Jime'nez A , Fiszman SM . Yogurt enrichment with functional asparagus fibre. Effect of fibre extraction method on rheological properties, colour, and sensory acceptance. Eur Food Res Technol. (2008) ;227: :1515–21. doi: 10.1007/s00217-008-0874-2 |
[32] | Amellal H . Technological abilities of some common varieties of dates: Formulation of a yoghurt naturally sweetened and flavored. [Doctoral thesis]. Boumerdes (Algeria) University M’hamed Bougara, Faculty of Science; (2007) . |
[33] | IDF: Yogurt: Determination of titratable acidity. IDF/ISO/AOAC Standarda 150:1991a. International Dairy Federation, Brussels, Belgium. |
[34] | IDF: Yogurt: Enumeration of characteristic microorganisms, Colony Count Technique at 37°C. IDF/ISO Standard 117:2003b. International Dairy Federation, Brussels, Belgium. |
[35] | Lim Orathai , Suntornsuk Worapot , Suntornsuk Leena . Capillary zone electrophoresis for enumeration of Lactobacillus delbrueckii subsbulgaricus and Streptococcus thermophilus in yogurt. Journal of Chromatography B. (2009) ;877: (8-9):710–18. doi: 10.1016/j.jchromb.2009.02.014 |
[36] | Bonnefoy C , Guillet F , Leyral G , Verne-Bourdais E . Microbiology and quality of agro-food industries. France:Edition Doin; (2002) . |
[37] | Lucey JA , Singh H . Formation and physical properties of acid milk gels: A review. Food Res Int. (1997) ;30: :529–42. doi: 10.1016/S0963-9969(98)00015-5 |
[38] | Lucey JA . Formation and physical properties of milk protein gels. J Dairy Sci. (2002) ;85: :281–94. doi: 10.3168/jds.S0022-0302(02)74078-2 |
[39] | Van Vliet T , Lakemond CMM , Visschers RW . Rheology and structure of milk protein gels. Current Opinion in Colloid & Interface Science. (2004) ;9: (5):298–304. doi: 10.1016/j.cocis.2004.09.002 |
[40] | Pelaes Vital AC , Akie Goto P , Hanai LN , Gomes-da-Costa SM , De Abreu Filho BA , Nakamura CV , et al. Microbiological, functional and rheological properties of low fat yogurt supplemented with Pleurotus ostreatus aqueous extract. LWT - Food Science and Technology. (2015) ;64: (2):1028–35. doi: 10.1016/j.lwt.2015.07.003 |
[41] | Warrand J , Michaud P , Picton L , Muller G , Courtois B , Ralainirina R , et al. Structural investigations of the neutral polysaccharide of Linum usitatissimum L. seeds mucilage. International Journal of Biological Macromolecules. (2005) ;35: (3-4):121–25. doi: 10.1016/j.ijbiomac.2004.12.006 |
[42] | Chen HH , Xu SY , Wang Z . Gelation properties of flaxseed gum. Journal of Food Engineering. (2006) ;77: (2):295–303. doi: 10.1016/j.jfoodeng.2005.06.033 |
[43] | Mazza G , Biliaderis CG . Functional properties of flax seed mucilage. Journal of Food Science. (1989) ;54: :1302–5. doi: 10.1111/j.1365-2621.1989.tb05978.x |
[44] | Wang Y , Li D , Wang LJ , Li SJ , Adhikari B . Effects of drying methods on the functional properties of flaxseed gum powders. Carbohydrate Polymers. (2010) ;81: :128–33. doi: 10.1016/j.carbpol.2010.02.005 |
[45] | Folkenberg DM , Martens M . Sensory properties of low fat yoghurts. Part A: Effect of fat content, fermentation culture and addition of non-fat dry milk on the sensory properties of plain yoghurts. Milchwissenschaft-Milk Science International. (2003) ;58: (1-2):48–51. |
[46] | Sah BNP , Vasiljevic T , McKechnie S , Donkor ON . Physicochemical, textural and rheological properties of probiotic yogurt fortified with fibre-rich pineapple peel powder during refrigerated storage. LWT - Food Science and Technology. (2016) ;65: :978–86. doi: 10.1016/j.lwt.2015.09.027 |
[47] | Kroger M . Quality of yogurt. J Dairy Sci. (1976) ;59: :344–50. |
[48] | Ozturkoglu-Budak S , Akal C , Yetisemiyen A . Effect of dried nut fortification on functional, physicochemical, textural, and microbiological properties of yogurt. J Dairy Sci. (2016) ;99: (11):8511–23. doi: 10.3168/jds.2016-11217 |
[49] | Raho Ghalem Bachir , Benattouche Zouaoui . Microbiological, physico-chemical and sensory quality aspects of yoghurt enriched with Rosmarinus officinalis oil. African Journal of Biotechnology. (2013) ;12: (2):192–98. doi: 10.5897/AJB12.1257 |
[50] | Shah NP . Probiotic bacteria: Selective enumeration and survival in dairy products. J Dairy Sci. (2000) ;83: (4):894–907. doi: 10.3168/jds.S0022-0302(00)74953-8 |
[51] | Muniandy Premalatha , Bakr Shori Amal , Salihin Baba Ahmad . Comparison of the effect of green, white and black tea on Streptococcus thermophilus and Lactobacillus spp. in yogurt during refrigerated Storage. Journal of the Association of Arab Universities for Basic and Applied Sciences. (2015) ;22: :26–30. |
[52] | Garcia-Perez FJ , Lario Y , Fernandez-Lopez J , Sayas E , Perez-Alvarez JA , Sendra E . Effect of orange fiber addition on yogurt color during fermentation and cold storage. Color Res Appl. (2005) ;30: :457–63. doi: 10.1002/col.20158 |
[53] | Bertolino Marta , Belviso Simona , Dal Bello Barbara , Ghirardello Daniela , Giordano Manuela , Rolle Luca , et al. Influence of the addition of different hazelnut skins on the physicochemical, antioxidant, polyphenol and sensory properties of yogurt. LWT - Food Science and Technology. (2015) ;63: (2):1145–54. doi: 10.1016/j.lwt.2015.03.113 |
[54] | Mishra V , Prasad DN . Application of in vitro methods for selection of Lactobacillus casei strains as potential probiotics. Int J Food Microbiol. (2005) ;103: (1):109–15. doi: 10.1016/j.ijfoodmicro.2004.10.047 |
[55] | Madureira AR , Amorim M , Gomes AM , Pintado ME , Malcata FX . Protective effect of whey cheese matrix on probiotic strains exposed to simulated gastrointestinal conditions. Food Res Int. (2011) ;44: (1):465–70. doi: 10.1016/j.foodres.2010.09.010 |




