Clinical Outcomes by Nephrectomy Status In METEOR, A Randomized Phase 3 Trial of Cabozantinib Versus Everolimus in Patients with Advanced Renal Cell Carcinoma
Abstract
Background:
We investigated outcomes with cabozantinib versus everolimus in patients with advanced renal cell carcinoma (RCC) with or without prior nephrectomy in the phase 3 METEOR trial (NCT01865747).
Methods:
Patients (N = 658) with advanced clear cell RCC and prior treatment with≥1 VEGFR tyrosine kinase inhibitor (TKI) were randomized to cabozantinib 60 mg/day or everolimus 10 mg/day. Pre-specified subgroup analyses of progression-free survival (PFS), overall survival (OS), and objective response rate (ORR) were conducted by prior nephrectomy status. Response was assessed by independent radiology committee.
Results:
Most enrolled patients (85%) had prior nephrectomy. Baseline prognostic factors (e.g. MSKCC risk group) were less favorable for patients without prior nephrectomy. Cabozantinib improved outcomes versus everolimus in the subgroups with and without nephrectomy—hazard ratios (95% CIs) of 0.51 (0.41–0.64) and 0.51 (0.30–0.86), respectively, for PFS, and 0.66 (0.52–0.84) and 0.75 (0.44–1.27), respectively, for OS. Median OS was numerically longer in patients with versus those without prior nephrectomy in both treatment arms. ORR for cabozantinib versus everolimus was 17% versus 4% for the prior nephrectomy subgroup and 21% versus 2% for the subgroup without prior nephrectomy. Among evaluable patients without prior nephrectomy, reductions of renal target lesions occurred in 94% (16/17) of patients in the cabozantinib arm versus 44% (8/18) in the everolimus arm. The safety profiles of both subgroups were generally consistent with that of the overall study population.
Conclusion:
Cabozantinib improved PFS, ORR, and OS compared with everolimus in patients with advanced RCC irrespective of nephrectomy status.
INTRODUCTION
The clinical benefit of nephrectomy in patients with metastatic renal cell carcinoma (RCC) was initially reported in 2001 [1–3]. In phase 3 trials, patients who underwent nephrectomy followed by interferon-α had improved overall survival (OS) compared with patients treated with interferon-α therapy alone. The underlying biology of this benefit was not fully understood; but in addition to tumor debulking, data indicated that nephrectomy reduced immunosuppressive cytokines and tumor-promoting growth factors [4–6]. Nephrectomy with cytokine therapy became a standard of care. However, since the introduction of targeted therapies in 2005 (e.g., vascular endothelial growth factor receptor [VEGFR] tyrosine kinase inhibitors [TKIs]), the use of nephrectomy for metastatic RCC has steadily declined. VEGFR TKIs became the preferred first-line therapy, and phase 3 trials were not conducted to evaluate nephrectomy with targeted therapies [5, 7–9]. Despite this, approximately 35% of patients with advanced RCC still receive nephrectomy as part of their initial treatment [7–9]; and clinical trials evaluating TKIs have predominantly enrolled patients who received prior nephrectomy for localized or metastatic disease [10–14].
The continued use of nephrectomy has been supported by data from retrospective studies that show OS advantages for patients with advanced RCC who were treated with targeted therapy and had received a prior nephrectomy for early- or late-stage disease compared with patients who received targeted therapy without prior nephrectomy [7, 9]. Interestingly, the phase 3 CARMENA trial recently reported that initial treatment with the VEGFR TKI sunitinib alone was non-inferior to treatment with nephrectomy followed by adjuvant sunitinib in patients with metastatic RCC and intermediate- or poor-risk disease [15]. More data are needed across the spectrum of patients with RCC to better define the role of nephrectomy and understand treatment outcomes with systemic therapies by prior nephrectomy status.
Cabozantinib is an orally bioavailable inhibitor of pro-oncogenic tyrosine kinases including VEGFRs, MET, and AXL [16]. The United States Food and Drug Administration initially approved cabozantinib for patients with advanced RCC who had received prior anti-angiogenic therapy, based on the pivotal phase 3 METEOR trial, which compared cabozantinib with everolimus (NCT01865747) [17, 18]. The indication was expanded to all patients with advanced RCC based on outcomes from the randomized phase 2 CABOSUN trial, which compared first-line cabozantinib with sunitinib in patients with intermediate- or poor-risk disease [19, 20].
In METEOR, cabozantinib improved progression-free survival (PFS), OS, and objective response rate (ORR) compared with everolimus in patients with advanced RCC who had received prior anti-angiogenic therapy [17, 18]. Prior nephrectomy was not part of the eligibility criteria of METEOR, but nephrectomy status was a predefined subgroup in the statistical analysis plan given its prognostic significance in historical studies. Here we present the analysis of clinical outcomes for cabozantinib versus everolimus by prior nephrectomy status.
PATIENTS AND METHODS
Patients and treatment
METEOR was an international, randomized, open-label, phase 3 clinical trial (NCT01865747). The study design and methods have been previously reported [17, 18]. Key eligibility criteria included a diagnosis of RCC with a clear-cell component, measurable disease per Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST v1.1) [21], and prior treatment with≥1 VEGFR TKI. Radiographic progression during or within 6 months of the most recent VEGFR TKI regimen was required. The last dose of a VEGFR TKI must have been received from 6 months to 2 weeks before randomization. Karnofsky Performance Status score of≥70 and adequate organ function were required. Prior therapy with cabozantinib or a mammalian target of rapamycin inhibitor was not permitted.
Patients were randomized 1 : 1 to receive cabozantinib (60 mg once daily) or everolimus (10 mg once daily). Stratification was by Memorial Sloan Kettering Cancer Center (MSKCC) risk group (favorable, intermediate, or poor) [22, 23] and number of prior VEGFR TKIs (1 or≥2). Dose reductions (to 40 mg and 20 mg for cabozantinib or to 5 mg and 2.5 mg for everolimus) were allowed to manage adverse events (AEs). The study was conducted in accordance with the Good Clinical Practice guidelines and the Declaration of Helsinki. The protocol was approved by the ethics committee or institutional review board at each center, and all patients provided written informed consent.
Assessments
Tumor assessment by computed tomography or magnetic resonance imaging was performed at screening, every 8 weeks for the first 12 months, and every 12 weeks thereafter. Tumor response was assessed by RECIST v1.1 [21], per independent radiology committee (IRC). Safety was evaluated every 2 weeks for the first 8 weeks and every 4 weeks thereafter until treatment discontinuation. A follow-up visit was planned 30 days after treatment discontinuation. AEs were reported according to National Cancer Institute Common Terminology Criteria for Adverse Events, v4.0.
Data analysis
The primary endpoint of PFS, and the secondary endpoints of OS, ORR and safety for METEOR have been reported previously [17, 18]. Pre-specified subgroup analyses included outcomes by nephrectomy status. PFS and OS were estimated with the Kaplan–Meier method. No adjustments for multiplicity were made for subgroup analyses. Confidence intervals (CIs) are considered descriptive, and hazard ratios (HRs) are unstratified. Safety was assessed in all patients who received at least one dose of study treatment. PFS and ORR had a data cut-off of May 22, 2015; and OS and safety had a data cut-off of December 31, 2015.
Results
Patients
From August 8, 2013, to November 24, 2014, 658 patients were randomized 1 : 1 to receive cabozantinib (N = 330) or everolimus (N = 328). A total of 283 (86%) patients in the cabozantinib arm and 279 (85%) patients in the everolimus arm had undergone prior nephrectomy; 47 (14%) and 49 (15%) patients, respectively, had not had prior nephrectomy. Baseline prognostic factors, including Eastern Cooperative Oncology Group performance status, MSKCC and International Metastatic Renal Cell Carcinoma Database Consortium risk group, time from diagnosis to randomization, and median sum of diameters for tumor target lesions, were less favorable for the subgroup without prior nephrectomy relative to the prior nephrectomy subgroup. Baseline characteristics were generally balanced between treatment arms in each subgroup (Table 1). Approximately 70% of patients with prior nephrectomy and 76% of patients without prior nephrectomy had received only 1 prior VEGFR TKI, and sunitinib was received by the majority of patients in both subgroups (Supplementary Materials Table S1).
Table 1
Baseline characteristics
| Prior nephrectomy | No prior nephrectomy | |||
| Cabozantinib (N = 283) | Everolimus (N = 279) | Cabozantinib (N = 47) | Everolimus (N = 49) | |
| Median age, yr (range) | 62 (32–86) | 61 (31–84) | 63 (36–82) | 63 (34–81) |
| Male, n (%) | 217 (77) | 201 (72) | 36 (77) | 40 (82) |
| Enrollment region, n (%) | ||||
| Europe | 140 (49) | 131 (47) | 27 (57) | 22 (45) |
| North America | 104 (37) | 105 (38) | 14 (30) | 17 (35) |
| Asia Pacific + Latin America | 39 (14) | 43 (15) | 6 (13) | 10 (20) |
| Median time since diagnosis to randomization, yr (range) | 3.0 (0–30) | 2.8 (0–33) | 1.1 (0–17) | 1.0 (0–11) |
| ECOG performance statusa, n (%) | ||||
| 0 | 197 (70) | 186 (67) | 29 (62) | 30 (61) |
| 1 | 86 (30) | 93 (33) | 18 (38) | 19 (39) |
| MSKCC risk group, n (%) | ||||
| Favorable | 135 (48) | 134 (48) | 15 (32) | 16 (33) |
| Intermediate | 117 (41) | 115 (41) | 22 (47) | 20 (41) |
| Poor | 31 (11) | 30 (11) | 10 (21) | 13 (27) |
| IMDC risk group, n (%) | ||||
| Favorable | 60 (21) | 59 (21) | 6 (13) | 3 (6) |
| Intermediate | 184 (65) | 182 (65) | 26 (55) | 32 (65) |
| Poor | 39 (14) | 38 (14) | 15 (32) | 14 (29) |
| Median target lesion SOD per IRC, mm (range) | 61.3 (0–291) | 62.6 (0–258) | 93.7 (0–277) | 104.3 (0–217) |
| Metastatic sites per IRC, n (%) | ||||
| Lung | 173 (61) | 180 (65) | 31 (66) | 32 (65) |
| Liver | 71 (25) | 92 (33) | 17 (36) | 11 (22) |
| Bone | 67 (24) | 47 (17) | 10 (21) | 18 (37) |
aKarnofsky performance status was converted to ECOG status using ECOG 0 for Karnofsky score of 100 and 90, and ECOG 1 for score of 80 and 70. ECOG, Eastern Cooperative Oncology Group; IRC, independent radiology committee; IMDC, International Metastatic Renal Cell Carcinoma Database Consortium; MSKCC, Memorial Sloan Kettering Cancer Center; SOD, sum of diameters.
Efficacy outcomes
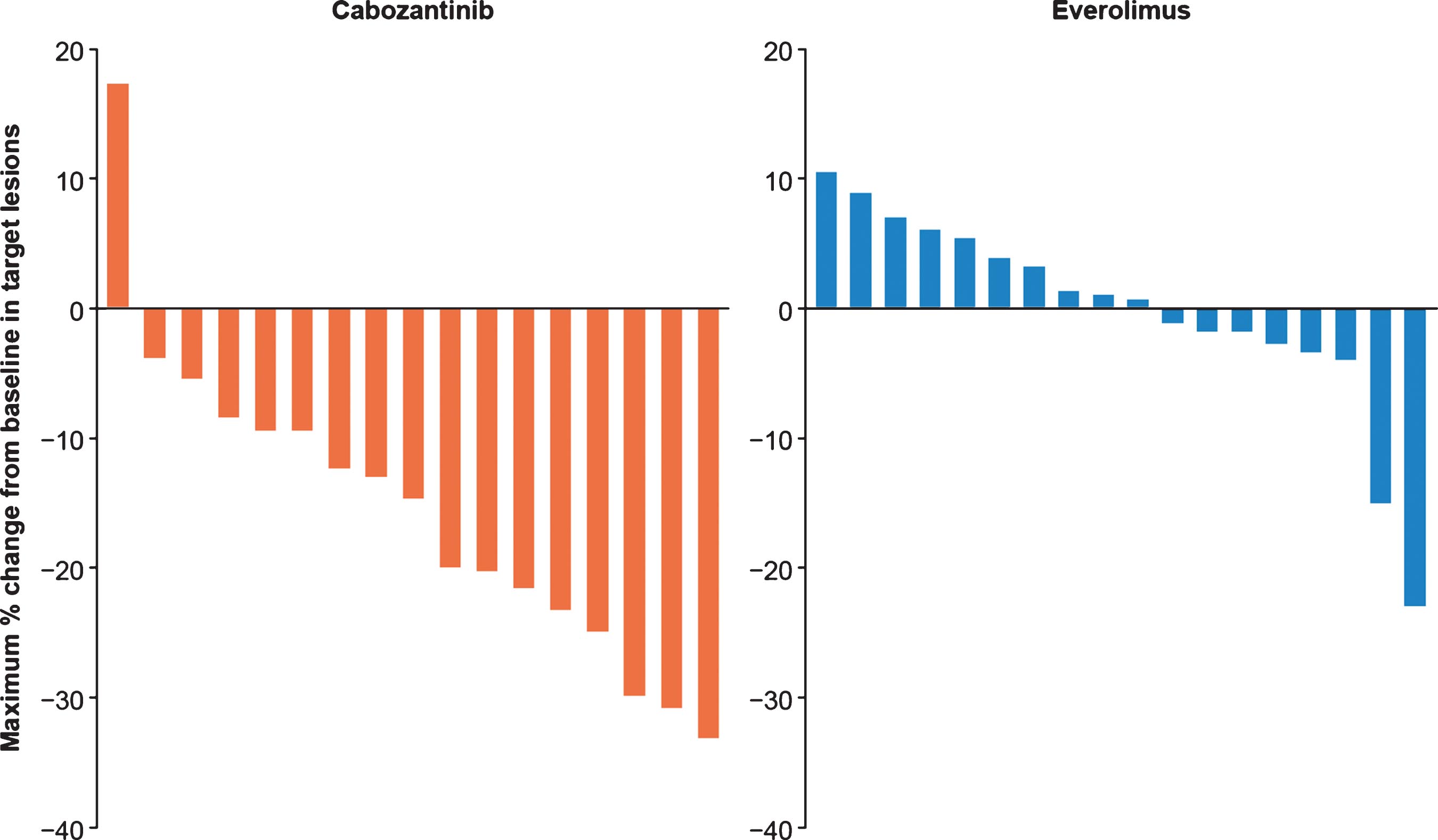
Cabozantinib improved PFS (Fig. 1) and OS (Fig. 2) compared with everolimus irrespective of nephrectomy status. In the prior nephrectomy subgroup, median PFS was 7.4 months for cabozantinib versus 3.9 months for everolimus (HR 0.51, 95% CI 0.41–0.64), and median OS was 22 months versus 17.2 months (HR 0.66, 95% CI 0.52–0.84). For patients without prior nephrectomy, median PFS was 6.6 months for cabozantinib versus 4.4 months for everolimus (HR 0.51, 95% CI 0.30–0.86), and median OS was 16.3 months versus 12.5 months (HR 0.75, 95% CI 0.44–1.27). The ORR per IRC was 17% in the cabozantinib arm versus 4% in the everolimus arm for patients with prior nephrectomy and 21% versus 2% for patients without prior nephrectomy (Table 2). All responses were partial. Among patients without prior nephrectomy who had a target lesion in the kidney and at least one post-baseline assessment, 94% (16/17) in the cabozantinib arm versus 44% (8/18) in the everolimus arm had a decrease in their sum of diameters for target lesions (Fig. 3).
Fig.1
Kaplan-Meier analyses of progression-free survival. Disease progression was assessed by an independent radiology committee. Data are through May 22, 2015. CI, confidence interval; PFS, progression-free survival; NE, not estimable.
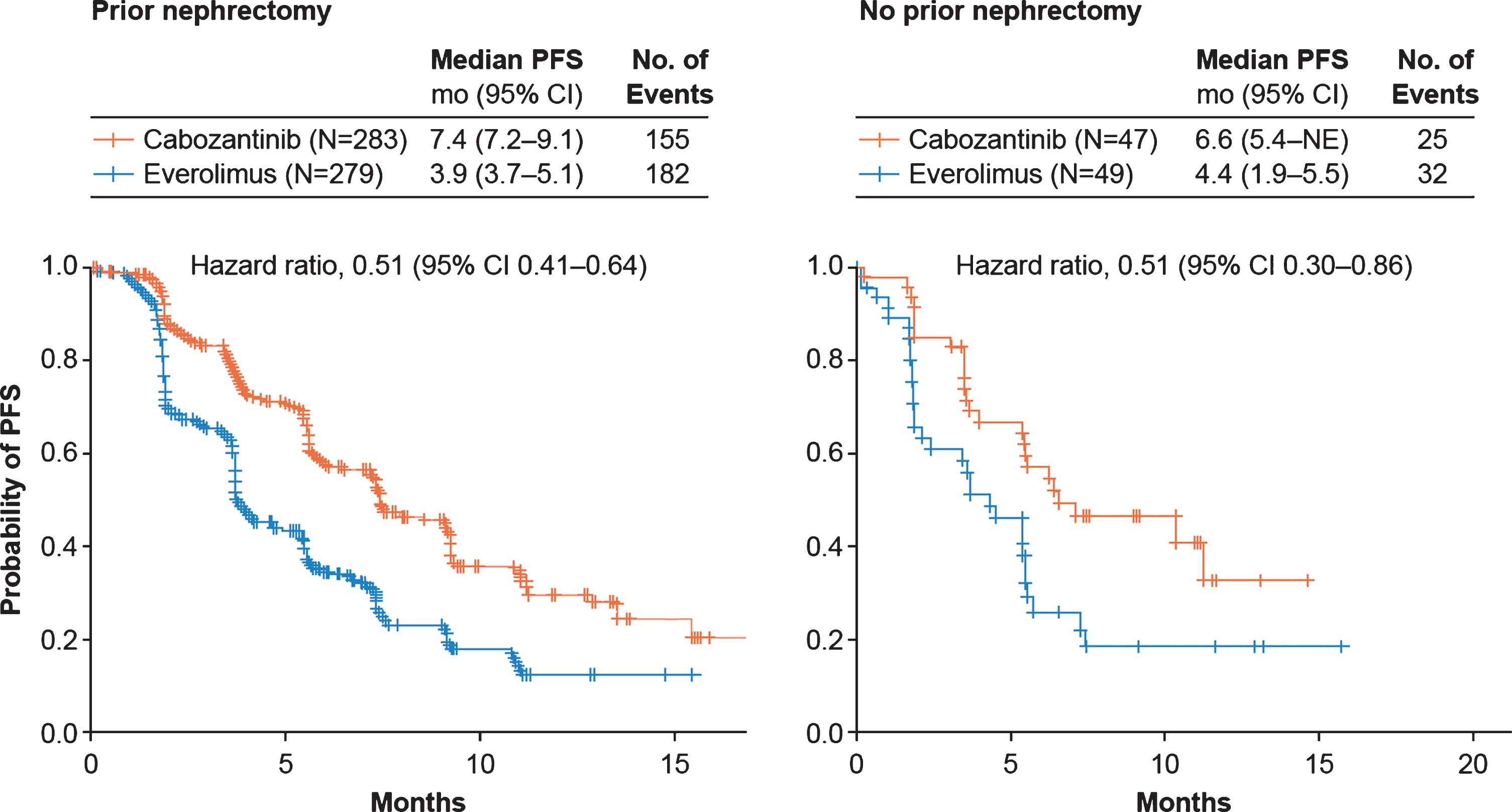
Fig.2
Kaplan-Meier analyses of overall survival. Data are through December 31, 2015. CI, confidence interval; OS, overall survival; NE, not estimable.
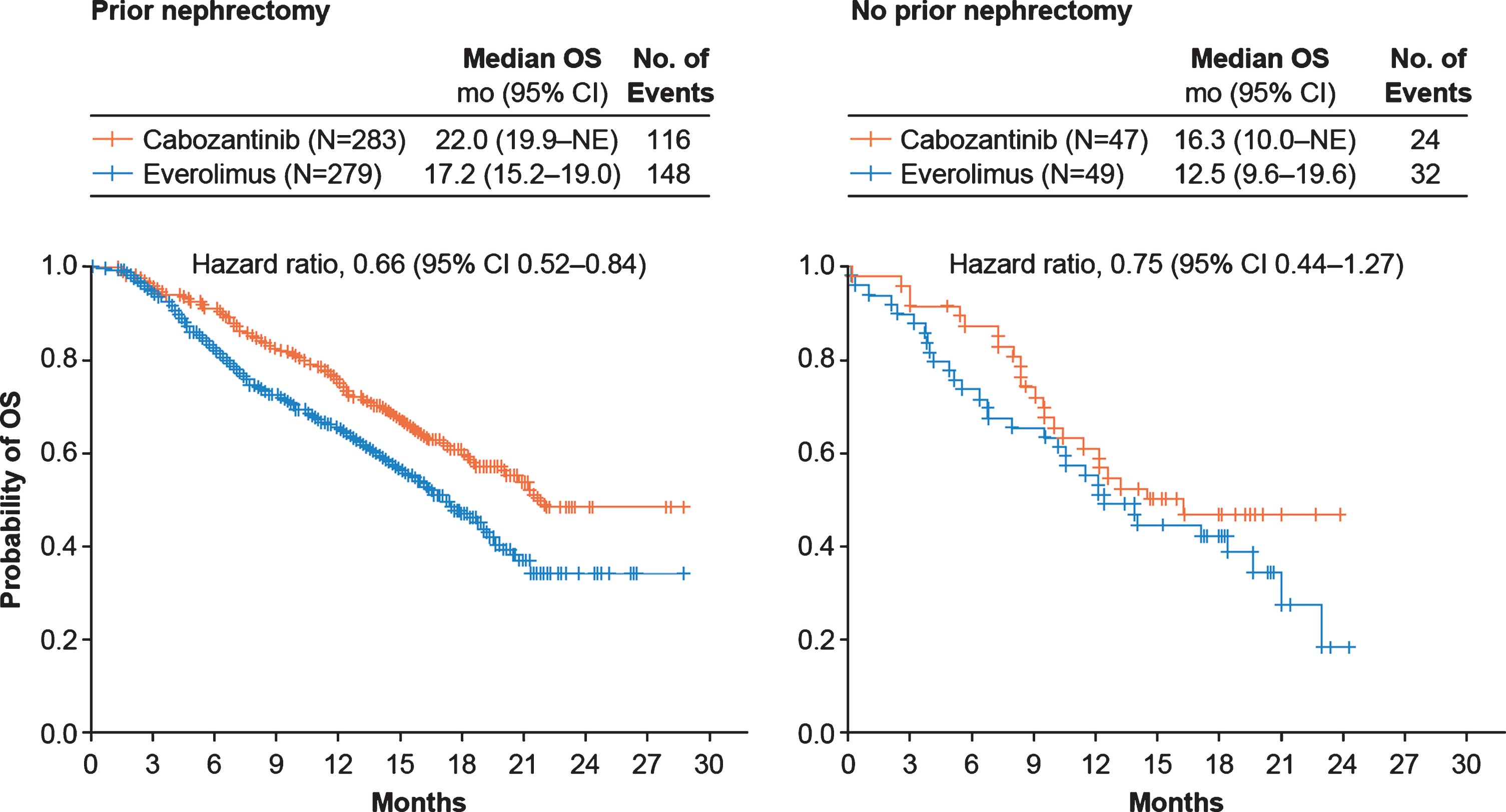
Table 2
Best Overall Tumor Response per RECIST v1.1
| Prior nephrectomy | No prior nephrectomy | |||
| Cabozantinib (N = 283) | Everolimus (N = 279) | Cabozantinib (N = 47) | Everolimus (N = 49) | |
| IRC | ||||
| ORR,a % (95% CI) | 17 (12–21) | 4 (2–6) | 21 (11–36) | 2 (0–11) |
| Best overall response, n (%) | ||||
| Confirmed partial response | 47 (17) | 10 (4) | 10 (21) | 1 (2) |
| Stable disease | 185 (65) | 175 (63) | 31 (66) | 28 (57) |
| Progressive disease | 36 (13) | 76 (27) | 5 (11) | 12 (24) |
| Not evaluable or missing | 15 (5) | 18 (6) | 1 (2) | 8 (16) |
| Investigator assessed, % | ||||
| ORR,a % (95% CI) | 24 (19–30) | 5 (3–8) | 19 (9–33) | 2 (0–11) |
| Best overall response, n (%) | ||||
| Confirmed partial response | 69 (24) | 13 (5) | 9 (19) | 1 (2) |
| Stable disease | 174 (61) | 176 (63) | 35 (74) | 29 (59) |
| Progressive disease | 27 (10) | 73 (26) | 2 (4) | 14 (29) |
| Not evaluable or missing | 13 (5) | 15 (5) | 1 (2) | 5 (10) |
aAll responses were partial responses. CI, confidence interval; IRC, independent radiology committee; ORR, objective response rate; RECIST, Response Evaluation Criteria in Solid Tumors; v, version.
Fig.3
Best change in target lesion size for primary kidney tumor in patients without prior nephrectomy Results are shown for patients with a target lesion in the kidney and≥1 post-baseline assessment.
Subsequent anti-cancer therapy
Overall, 50% of patients received subsequent anticancer therapy (Supplementary Materials Table S2). The proportion of patients who received a VEGFR TKI as subsequent therapy in the cabozantinib and everolimus arms were 22% and 47% in the prior nephrectomy subgroup, and 30% and 53% in the subgroup without prior nephrectomy. Axitinib was the most common subsequent VEGFR TKI administered. No patients underwent nephrectomy subsequent to enrollment in METEOR.
Safety
Exposure and dose reductions
The median duration of exposure with cabozantinib was similar for patients with and without prior nephrectomy, as was the duration of everolimus exposure (Table 3). Patients receiving cabozantinib underwent more frequent dose reductions than patients receiving everolimus. In the prior nephrectomy subgroup, 12% of patients in the cabozantinib arm and 11% of patients in the everolimus arm discontinued treatment due to AEs, with corresponding values of 13% and 6% in the subgroup without prior nephrectomy.
Table 3
Study treatment and dose reductions
| Prior nephrectomy | No prior nephrectomy | |||
| Cabozantinib (N = 284) | Everolimus (N = 274) | Cabozantinib (N = 47) | Everolimus (N = 48) | |
| Median (range) duration of exposure, weeks | 37 (3–121) | 19 (1–108) | 36 (1–96) | 19 (1–98) |
| Patients receiving dose reductions, n (%) | 180 (63) | 65 (24) | 26 (55) | 11 (23) |
| Median (range) average daily dose, mg | 43 (13–69) | 9 (3–12) | 43 (14–65) | 9 (3–11) |
| Median time (range) to first dose reduction, weeks | 8.1 (2.1–77.6) | 9.0 (1.7–68.4) | 6.5 (1.4–36.7) | 8.9 (3.1–28.1) |
Adverse events
The overall safety profiles of cabozantinib and everolimus were consistent across both subgroups (Table 4). In the prior nephrectomy subgroup, 70% of patients in the cabozantinib arm versus 60% of patients in the everolimus arm experienced grade 3 or 4 adverse events, with corresponding values of 79% versus 60% in patients without prior nephrectomy. The most common grade 3 or 4 adverse events in patients who received cabozantinib included hypertension, diarrhea, fatigue and palmar-plantar erythrodysesthesia. In the cabozantinib arm, 3% of patients with prior nephrectomy experienced grade 3 or 4 proteinuria, compared with 1% in the everolimus arm. There were no grade 3 or 4 proteinuria events in patients without prior nephrectomy.
Table 4
All-Causality Grade 3 or 4 Adverse Events
| Prior nephrectomy | No prior nephrectomy | |||
| Cabozantinib (N = 284) | Everolimus (N = 274) | Cabozantinib (N = 47) | Everolimus (N = 48) | |
| Any adverse event,a n (%) | 198 (70) | 164 (60) | 37 (79) | 29 (60) |
| Hypertension | 42 (15) | 10 (4) | 7 (15) | 2 (4) |
| Diarrhea | 31 (11) | 4 (1) | 12 (26) | 3 (6) |
| Fatigue | 29 (10) | 21 (8) | 7 (15) | 3 (6) |
| Palmar-plantar erythrodysesthesia | 22 (8) | 3 (1) | 5 (11) | 0 |
| Anemia | 14 (5) | 43 (16) | 5 (11) | 10 (21) |
| Hyperglycemia | 3 (1) | 14 (5) | 0 | 2 (4) |
| Hypomagnesemia | 15 (5) | 0 | 1 (2) | 0 |
| Hypokalemia | 13 (5) | 6 (2) | 3 (6) | 0 |
| Hyponatremia | 11 (4) | 7 (3) | 4 (9) | 1 (2) |
| Dyspnea | 3 (1) | 14 (5) | 7 (15) | 0 |
| Nausea | 11 (4) | 1 (<1) | 4 (9) | 0 |
| Abdominal pain | 9 (3) | 4 (1) | 3 (6) | 1 (2) |
| Gamma-glutamyltransferase increased | 2 (1) | 8 (3) | 2 (4) | 4 (8) |
| Mucosal inflammation | 4 (1) | 7 (3) | 1 (2) | 4 (8) |
| Asthenia | 10 (4) | 5 (2) | 5 (11) | 3 (6) |
| Adverse events of special interest,b n (%) | ||||
| Proteinuria | 8 (3) | 2 (1) | 0 | 0 |
| Renal failure | 1 (<1) | 1 (<1) | 0 | 0 |
| Renal impairment | 0 | 1 (<1) | 0 | 0 |
| Creatinine renal clearance decreased | 0 | 0 | 0 | 0 |
aGrade 3 or 4 events that occurred at≥5.0% frequency in either treatment arm; bevents potentially related to renal impairment.
DISCUSSION
Despite phase 3 studies establishing the benefit of nephrectomy followed by cytokine therapy for patients with metastatic RCC, there has been a lack of prospective studies to define its role with targeted therapies [1–3]. Observational studies have indicated a survival advantage when nephrectomy was eventually followed with targeted therapies, but the benefit did not appear to extend across all patient populations, particularly for patients with poor-risk disease, and may have been the result of selection bias [7, 24–26]. The recent phase 3 CARMENA trial demonstrated definitive evidence that first-line treatment with sunitinib alone was non-inferior to treatment with nephrectomy plus adjuvant sunitinib for patients with intermediate or poor risk metastatic RCC [15]. However, it remains unclear if CARMENA results will extend to other targeted therapies, to checkpoint inhibitors, or to patients with favorable-risk disease who are more likely to be candidates for nephrectomy. Further, there may be a potential neoadjuvant role as targeted therapies may improve outcomes and help to inform a post-nephrectomy treatment plan. In the recent SURTIME trial, patients with synchronous metastatic RCC were randomized to neoadjuvant sunitinib followed by nephrectomy (in the absence of progression) and then adjuvant sunitinib versus nephrectomy followed by adjuvant sunitinib. Because of poor patient accrual, this study was closed early. The progression-free rate at 28 weeks did not differ between patients who received neoadjuvant/adjuvant sunitinib versus patients who received adjuvant sunitinib (43% vs. 42%), while OS analysis suggested improvement with neoadjuvant/adjuvant sunitinib (median 32.4 vs. 15.0 months; P = 0.03) [27].
There have also been limited data on outcomes in patients receiving targeted therapies in the second-line setting with respect to prior nephrectomy status, particularly patients without prior nephrectomy [10, 11, 28]. In the METEOR study, cabozantinib improved PFS, OS, and ORR compared with everolimus in patients with advanced RCC after prior antiangiogenic therapy [17, 18]. The median PFS was 7.4 months for the cabozantinib arm versus 3.9 months for the everolimus arm (HR 0.51; 95% CI 0.41–0.62; P < 0.0001), median OS was 21.4 months versus 16.5 months (HR = 0.66; 95% CI 0.53–0.83; P = 0.00026), and the ORR was 17% versus 3% (P < 0.0001) [18]. More than 85% of the patients enrolled in METEOR had received prior nephrectomy.
In the prespecified subgroup analysis of METEOR reported here, cabozantinib was associated with improved PFS, OS, and ORR compared with everolimus irrespective of prior nephrectomy status. Results in the prior nephrectomy subgroup were comparable to those of the overall study population and likely reflect the large proportion of randomized patients who had received prior nephrectomy [18]. PFS and ORR results for patients without prior nephrectomy were comparable to those of the overall study population, while median OS was shorter in both treatment arms compared with corresponding values from the overall study population. The METEOR trial was conducted in an era when nephrectomy was considered a standard of care in RCC patients with synchronous metastases and favorable risk; the shorter OS in the non-nephrectomy subgroup may be related to less favorable prognostic characteristics that led to the decision to not perform nephrectomy. Patients without prior nephrectomy tended to have greater disease burden at baseline and other factors associated with poor prognosis than patients who had prior nephrectomy. In an exploratory analysis, we found no evidence of an interaction between treatment and nephrectomy status with respect to survival outcomes (data not shown). Regardless, the benefit associated with cabozantinib was maintained as the HRs for PFS and OS by prior nephrectomy status were comparable with those of the overall study population.
The types and incidence of AEs in both subgroups were consistent with those in the overall study population and the safety profiles of cabozantinib and everolimus [18, 19, 29]. The most common grade 3 or 4 AEs reported in patients receiving cabozantinib included hypertension, palmar-plantar erythrodysesthesia, diarrhea, and fatigue. The rates of grade 3 or 4 AEs related to renal dysfunction were low in both treatment arms, with all events occurring in patients with prior nephrectomy. There was no notable difference in the duration of exposure, the incidence of dose reductions, or the average daily dose based on prior nephrectomy status. Dose reductions were common for cabozantinib, but treatment discontinuations due to AEs were infrequent, indicating AEs were manageable with dose modifications and supportive care.
Because the RCC treatment landscape is evolving rapidly, the clinical benefit of nephrectomy will require additional studies in the context of emerging treatment strategies. Recently the CABOSUN study assessed cabozantinib as a first-line therapy and demonstrated superior PFS compared with sunitinib in patients with intermediate or poor risk advanced RCC [19]; this benefit was maintained in the subgroups of patients with or without prior nephrectomy [30]. Taken together, data from METEOR and CABOSUN support the use of single-agent cabozantinib in advanced RCC in patients with or without prior nephrectomy and consideration of neoadjuvant strategies to be tested in clinical trials. Cabozantinib is also being assessed in combination with checkpoint inhibitors [31, 32]; early phase studies have demonstrated clinical antitumor activity to support further development [33], including a phase 3 study of cabozantinib in combination with nivolumab (NCT03141177). There are limited data on outcomes with second-line nivolumab by prior nephrectomy status [34, 35]; subgroup analysis of the phase 3 CheckMate 214 study demonstrated that the OS benefit with nivolumab-ipilimumab compared with sunitinib as a first-line therapy was maintained in patients with or without prior nephrectomy [36].
CONCLUSION
Cabozantinib improved PFS, ORR, and OS compared with everolimus in patients with advanced RCC and prior anti-angiogenic therapy irrespective of nephrectomy status. Nephrectomy status had no notable impact on the safety and tolerability of cabozantinib.
FUNDING
The study was funded by Exelixis, Inc. Patients treated at Memorial Sloan Kettering Cancer Center were supported in part by Memorial Sloan Kettering Cancer Center Support Grant/Core Grant (P30 CA008748). Medical writing and editorial support was provided by Suvajit Sen, PhD (Exelixis, Inc.), and Fishawack Communications Inc. (Conshohocken, PA, USA), which was funded by Exelixis.
AUTHOR CONTRIBUTIONS
All authors have contributed toward the preparation of the manuscript.
CONFLICTS OF INTEREST
NMT reports honoraria, consulting/advisory roles, research funding, and travel/accommodation/ expenses from Bristol-Myers Squibb, Exelixis, Novartis, honoraria, consulting/advisory role, travel/accommodations/expenses from Calithera Biosciences, Eisai Medical Research, Nektar, Pfizer, honoraria, consulting/advisory roles from Ono Pharmaceutical, consulting/advisory role and travel/accommodation/expenses from Oncorena, research funding from Epizyme and Mirati Therapeutics, outside the submitted work.
TP reports honoraria, consulting/advisory roles,research funding, and travel/accommodation/expenses from AstraZeneca and Roche/Genentech, honoraria and consulting/advisory roles from Merck, honoraria, consulting/advisory roles, and travel/accommodation/expenses from Bristol-Myers Squibb, honoraria and consulting/advisory roles from Novartis, honoraria, consulting/advisory roles, and travel/accommodation expenses from Pfizer, consulting/advisory roles from Incyte and Seattle Genetics, research funding from AstraZeneca/MedImmune, travel/accommodation/expenses from MSD, outside the submitted work.
BE reports honoraria, consulting/advisory roles, travel/accommodations/expenses from Bristol-Myers Squibb, Ipsen, Roche/Genentech, Pfizer honoraria and consulting/advisory role from EUSA Pharma, Novartis, consulting/advisory role from AVEO, outside the submitted work.
FD reports grants from Pfizer and Ipsen outside of the submitted work.
VG reports grants and personal fees from Pfizer, grants, personal fees and non-financial support from BMS, Ipsen, personal fees and non-financial support from Roche, Novartis, EISAI, grants and personal fees from EUSA Pharma, personal fees from MSD, during the conduct of the study; grants, personal fees, non-financial support and other from MSD, grants from Novartis, grants, personal fees and other from AstraZeneca, grants, personal fees, non-financial support and other from BMS, personal fees and non-financial support from MerckSerono, PharmaMar, personal fees from Roche, Pfizer, Lilly, Janssen, Nanobiotix, outside the submitted work.
CNS reports consultancy fees and institutional funding from Eisai, Pfizer, and consultancy fees from Novartis, IPSEN, BMS, Roche, Bayer, and Merck, and institutional funding from Roche-Genentech and Exelixis, outside the submitted work.
MS reports grants and personal fees from Pfizer, Roche, personal fees from Ipsen, BMS, EUSA, EISAI, during the conduct of the study, outside the submitted work.
PS reports consulting/advisory fees from Plexxikon, Loxo, Ellipses Pharma, Deciphera, Servier, Merck, Genmab, Adaptimmune, Intellisphere, Transgene, consulting/advisory fees and institution research funding from Eisai, Lilly, Blueprint Medicines, and institution research funding from Boehringer Ingelheim, CoBioRes NV, Exelixis, G1 Therapeutics, Novartis, and PharmaMar, outside the submitted work.
KP reports personal fees from Ipsen, during the conduct of the study; personal fees from MSD, BMS, Pfizer, Orion Pharma, Roche, outside the submitted work, and Faron Pharmaceuticals, Stock holder, outside the submitted work.
DN reports personal fees from BMS, Hoffmann la Roche, Eisai, outside the submitted work.
BM reports personal fees and other from BMS, Roche, MSD, Merck Serono, Novartis, personal fees from Pfizer, Ipsen, Sanofi, Astellas, Janssen, Eisai, Bayer, Eli Lilly, AstraZeneca, Amgen, Servier, Pierre Fabre, outside the submitted work.
DC reports employment and stock options from Exelixis, Inc.
C Scheffold reports other from Exelixis, Inc, during the conduct of the study; in addition, C Scheffold has a patent Exelixis, Inc issued.
RJM reports consultancy fees and clinical trial support to his institution from Pfizer, Eisai, and Genentech/Roche, consultancy fees from Novartis, Exelixis, Merck, Incyte, and Lilly, and clinical trial support to his institution from Bristol-Myers Squibb, outside the submitted work.
TKC reports employment and leadership roles at Dana Farber Cancer Hospital, leadership roles at NCCN, honoraria and consulting/advisory roles from Alexion Pharmaceuticals, alligent, ASCO, Clinical Care Options, EMD Serono, Harborside Press, HERON, Kidney Cancer Journal, Lancet Oncology, Lilly, Lpath, Michael J. Hennessy Associates, Navinata Healthcare, NCCN, NEJM, PlatformQ Health, Sanofi/Aventis, UpToDate, honoraria from Genentech/Roche, consulting/advisory roles and institutional research funding from Roche/Genentech, honoraria, consulting/advisory roles, and institutional research funding from Analysis Group, AstraZeneca, Bayer, Bristol-Myers Squibb, Cerulean Pharma, Corvus Pharmaceuticals, Eisai, Exelixis, Foundation Medicine, GlaxoSmith-Kline, Ipsen, Merck, Novartis, Peloton Therapeutics, Pfizer, Prometheus, institutional research funding from Agensys, Calithera Biosciences, Celldex, Roche, Seattle Genetics/Astellas, Takeda, TRACON Pharma, Medical writing and editorial assistance support may have been funded by Communications companies funded by pharmaceutical companies such as ClinicalThinking, Health Interactions, Envision Pharma Group, Fishawack Group of Companies, Parexel, outside the submitted work.
C Szczylik has no conflict of interest to disclose.
ACKNOWLEDGMENTS
We thank the patients, their families, the investigators and site staff, and the study teams participating in this trial.
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/KCA-190080.
REFERENCES
[1] | Flanigan RC , Salmon SE , Blumenstein BA , Bearman SI , Roy V , McGrath PC , Caton JR Jr , MunshiN, CrawfordED. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med. (2001) ;345: (23):1655–9. |
[2] | Flanigan RC , Mickisch G , Sylvester R , Tangen C , Van Poppel H , Crawford ED . Cytoreductive nephrectomy in patients with metastatic renal cancer: A combined analysis. J Urol. (2004) ;171: (3):1071–6. |
[3] | Mickisch GH , Garin A , van H , de Prijck L , Sylvester R , European Organisation for R, Treatment of Cancer Genitourinary G. Radical nephrectomy plus interferon-alfa-based immunotherapy compared with interferon alfa alone in metastatic renal-cell carcinoma: A randomised trial. Lancet. (2001) ;358: (9286)966–70. |
[4] | Sato K , Tsuchiya N , Sasaki R , Shimoda N , Satoh S , Ogawa O , Kato T . Increased serum levels of vascular endothelial growth factor in patients with renal cell carcinoma. Jpn J Cancer Res. (1999) ;90: (8):874–9. |
[5] | Rini BI , Campbell SC . The evolving role of surgery for advanced renal cell carcinoma in the era of molecular targeted therapy. J Urol. (2007) ;177: (6):1978–84. |
[6] | Rini BI , Campbell SC , Escudier B . Renal cell carcinoma. Lancet. (2009) ;373: (9669):1119–32. |
[7] | Hanna N , Sun M , Meyer CP , Nguyen PL , Pal SK , Chang SL , de Velasco G , Trinh QD , Choueiri TK . Survival analyses of patients with metastatic renal cancer treated with targeted therapy with or without cytoreductive nephrectomy: A National Cancer Data Base Study. J Clin Oncol. (2016) ;34: (27):3267–75. |
[8] | Culp SH . Cytoreductive nephrectomy and its role in the present-day period of targeted therapy. Ther Adv Urol. (2015) ;7: (5):275–85. |
[9] | Conti SL , Thomas IC , Hagedorn JC , Chung BI , Chertow GM , Wagner TH , Brooks JD , Srinivas S , Leppert JT . Utilization of cytoreductive nephrectomy and patient survival in the targeted therapy era. Int J Cancer. (2014) ;134: (9):2245–52. |
[10] | Vaishampayan UN . The Role of Nephrectomy for Kidney Cancer in the Era of Targeted and Immune Therapies. Am Soc Clin Oncol Educ Book. (2016) ;35: :e16–20. |
[11] | Motzer RJ , Escudier B , Tomczak P , Hutson TE , Michaelson MD , Negrier S , Oudard S , Gore ME , Tarazi J , Hariharan S , Chen C , Rosbrook B , Kim S , Rini BI . Axitinib versus sorafenib as second-line treatment for advanced renal cell carcinoma: Overall survival analysis and updated results from a randomised phase 3 trial. Lancet Oncol. (2013) ;14: (6):552–62. |
[12] | Motzer RJ , Hutson TE , Tomczak P , Michaelson MD , Bukowski RM , Rixe O , Oudard S , Negrier S , Szczylik C , Kim ST , Chen I , Bycott PW , Baum CM , Figlin RA . Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. (2007) ;356: (2):115–24. |
[13] | Motzer RJ , Hutson TE , Cella D , Reeves J , Hawkins R , Guo J , Nathan P , Staehler M , de Souza P , Merchan JR , Boleti E , Fife K , Jin J , Jones R , Uemura H , De Giorgi U , Harmenberg U , Wang J , Sternberg CN , Deen K , McCann L , Hackshaw MD , Crescenzo R , Pandite LN , Choueiri TK . Pazopanib versus sunitinib in metastatic renal-cell carcinoma. N Engl J Med. (2013) ;369: (8):722–31. |
[14] | Motzer RJ , Hutson TE , Glen H , Michaelson MD , Molina A , Eisen T , Jassem J , Zolnierek J , Maroto JP , Mellado B , Melichar B , Tomasek J , Kremer A , Kim HJ , Wood K , Dutcus C , Larkin J . Lenvatinib, everolimus, and the combination in patients with metastatic renal cell carcinoma: A randomised, phase 2, open-label, multicentre trial. Lancet Oncol. (2015) ;16: (15):1473–82. |
[15] | Mejean A , Ravaud A , Thezenas S , Colas S , Beauval JB , Bensalah K , Geoffrois L , Thiery-Vuillemin A , Cormier L , Lang H , Guy L , Gravis G , Rolland F , Linassier C , Lechevallier E , Beisland C , Aitchison M , Oudard S , Patard JJ , Theodore C , Chevreau C , Laguerre B , Hubert J , Gross-Goupil M , Bernhard JC , Albiges L , Timsit MO , Lebret T , Escudier B . Sunitinib alone or after nephrectomy in metastatic renal-cell carcinoma. N Engl J Med. (2018) ;379: (5):417–27. |
[16] | Yakes FM , Chen J , Tan J , Yamaguchi K , Shi Y , Yu P , Qian F , Chu F , Bentzien F , Cancilla B , Orf J , You A , Laird AD , Engst S , Lee L , Lesch J , Chou YC , Joly AH . Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther. (2011) ;10: (12):2298–308. |
[17] | Choueiri TK , Escudier B , Powles T , Mainwaring PN , Rini BI , Donskov F , Hammers H , Hutson TE , Lee JL , Peltola K , Roth BJ , Bjarnason GA , Geczi L , Keam B , Maroto P , Heng DY , Schmidinger M , Kantoff PW , Borgman-Hagey A , Hessel C , Scheffold C , Schwab GM , Tannir NM , Motzer RJ , Investigators M. Cabozantinib versus everolimus in advanced renal-cell carcinoma. N Engl J Med. (2015) ;373: (19):1814–23. |
[18] | Choueiri TK , Escudier B , Powles T , Tannir NM , Mainwaring PN , Rini BI , Hammers HJ , Donskov F , Roth BJ , Peltola K , Lee JL , Heng DYC , Schmidinger M , Agarwal N , Sternberg CN , McDermott DF , Aftab DT , Hessel C , Scheffold C , Schwab G , Hutson TE , Pal S , Motzer RJ , investigators M. Cabozantinib versus everolimus in advanced renal cell carcinoma (METEOR): Final results from a randomised, open-label, phase 3 trial. Lancet Oncol. (2016) ;17: (7):917–27. |
[19] | Choueiri TK , Halabi S , Sanford BL , Hahn O , Michaelson MD , Walsh MK , Feldman DR , Olencki T , Picus J , Small EJ , Dakhil S , George DJ , Morris MJ . Cabozantinib versus sunitinib as initial targeted therapy for patients with metastatic renal cell carcinoma of poor or intermediate risk: The Alliance A03 CABOSUN Trial. J Clin Oncol. (2017) ;35: (6):591–7. |
[20] | Choueiri TK , Hessel C , Halabi S , Sanford B , Michaelson MD , Hahn O , Walsh M , Olencki T , Picus J , Small EJ , Dakhil S , Feldman DR , Mangeshkar M , Scheffold C , George D , Morris MJ . Cabozantinib versus sunitinib as initial therapy for metastatic renal cell carcinoma of intermediate or poor risk (AllianceA031203 CABOSUN randomised trial): Progression-free survival by independent review and overall survival update. Eur J Cancer. (2018) ;94: (:115–25. |
[21] | Eisenhauer EA , Therasse P , Bogaerts J , Schwartz LH , Sargent D , Ford R , Dancey J , Arbuck S , Gwyther S , Mooney M , Rubinstein L , Shankar L , Dodd L , Kaplan R , Lacombe D , Verweij J . New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur J Cancer. (2009) ;45: (2):228–47. |
[22] | Motzer RJ , Bacik J , Schwartz LH , Reuter V , Russo P , Marion S , Mazumdar M . Prognostic factors for survival in previously treated patients with metastatic renal cell carcinoma. J Clin Oncol. (2004) ;22: (3):454–63. |
[23] | Motzer RJ , Bacik J , Murphy BA , Russo P , Mazumdar M . Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol. (2002) ;20: (1):289–96. |
[24] | Choueiri TK , Xie W , Kollmannsberger C , North S , Knox JJ , Lampard JG , McDermott DF , Rini BI , Heng DY . The impact of cytoreductive nephrectomy on survival of patients with metastatic renal cell carcinoma receiving vascular endothelial growth factor targeted therapy. J Urol. (2011) ;185: (1):60–6. |
[25] | Heng DY , Wells JC , Rini BI , Beuselinck B , Lee JL , Knox JJ , Bjarnason GA , Pal SK , Kollmannsberger CK , Yuasa T , Srinivas S , Donskov F , Bamias A , Wood LA , Ernst DS , Agarwal N , Vaishampayan UN , Rha SY , Kim JJ , Choueiri TK . Cytoreductive nephrectomy in patients with synchronous metastases from renal cell carcinoma: Results from the International Metastatic Renal Cell Carcinoma Database Consortium. Eur Urol. (2014) ;66: (4):704–10. |
[26] | Bhindi B , Habermann EB , Mason RJ , Costello BA , Pagliaro LC , Thompson RH , Leibovich BC , Boorjian SA . Comparative survival following initial cytoreductive nephrectomy versus initial targeted therapy for metastatic renal cell carcinoma. J Urol. (2018) . |
[27] | Bex A , Mulders P , Jewett M , Wagstaff J , van Thienen JV , Blank CU , van Velthoven R , Del Pilar Laguna M , Wood L , van Melick HHE , Aarts MJ , Lattouf JB , Powles T , de Jong IJ , Rottey S , Tombal B , Marreaud S , Collette S , Collette L , Haanen J . Comparison of immediate vs deferred cytoreductive nephrectomy in patients with synchronous metastatic renal cell carcinoma receiving sunitinib: The surtime randomized clinical trial. JAMA Oncol. (2018) . |
[28] | Ueda K , Suekane S , Hirano T , Ogasawara N , Chikui K , Uemura K , Nakiri M , Nishihara K , Matsuo M , Igawa T . Efficacy of axitinib as second-line treatment in locally advanced and metastatic renal cell carcinoma. Anticancer Res. (2018) ;38: (9):5387–92. |
[29] | Motzer RJ , Escudier B , Oudard S , Hutson TE , Porta C , Bracarda S , Grunwald V , Thompson JA , Figlin RA , Hollaender N , Urbanowitz G , Berg WJ , Kay A , Lebwohl D , Ravaud A . Efficacy of everolimus in advanced renal cell carcinoma: A double-blind, randomised, placebo-controlled phase III trial. Lancet. (2008) ;372: (9637):449–56. |
[30] | George DJ , Hessel C , Halabi S , Sanford BL , Michaelson MD , Hahn OM , Walsh MK , Olencki T , Picus J , Small EJ , Dakhil SR , Feldman DR , Mangeshkar M , Scheffold C , Morris MJ , Choueiri TK . Cabozantinib versus sunitinib for previously untreated patients with advanced renal cell carcinoma (RCC) of intermediate or poor risk: Subgroup analysis of progression-free survival (PFS) and objective response rate (ORR) in the Alliance A031203 CABOSUN trial. Abstract (582) and poster (E19) presented at Genitourinary Cancers Symposium of the American Society of Clinical Oncology; San Francisco, CA; February 8-10, 2018. 2018: |
[31] | Nadal R , Mortazavi A , Stein M , et al. Clinical efficacy of cabozantinib plus nivolumab (CaboNivo) and CaboNivo plus ipilimumab (CaboNivoIpi) in patients (pts) with chemotherapy-refractory metastatic urothelial carcinoma (mUC) either naïve (n) or refractory (r) to checkpoint inhibitor (CPI). J Clin Oncol. (2018) ;36: (suppl):abstr4528. |
[32] | Agarwal N , Vaishampayan U , Green M , di Nucci F , Chang P , Scheffold C , Pal S , for the XL184-021 study group. Phase 1b study (COSMIC-021) of cabozantinib in combination with atezolizumab: Results of the dose escalation stage in patients (pts) with treatment-naïve advanced renal cell carcinoma (RCC). Ann Oncol. (2018) ;29: (suppl 8):Abstract 872P. |
[33] | Nadal RM , Mortazavi A , Stein M , Pal SK , Davarpanah NN , Parnes HL , Ning Y-M , Cordes LM , Bagheri MH , Lindenberg L , Thompson R , Steinberg SM , Moore T , Lancaster T , Velez M , Mena E , Costello R , Bottaro D , Dahut WL , Apolo AB . Results of phase I plus expansion cohorts of cabozantinib (Cabo) plus nivolumab (Nivo) and CaboNivo plus ipilimumab (Ipi) in patients (pts) with with metastatic urothelial carcinoma (mUC) and other genitourinary (GU) malignancies. J Clin Oncol. (2018) ;36: (6 suppl):Abstract 515. |
[34] | Motzer RJ , Escudier B , McDermott DF , George S , Hammers HJ , Srinivas S , Tykodi SS , Sosman JA , Procopio G , Plimack ER , Castellano D , Choueiri TK , Gurney H , Donskov F , Bono P , Wagstaff J , Gauler TC , Ueda T , Tomita Y , Schutz FA , Kollmannsberger C , Larkin J , Ravaud A , Simon JS , Xu LA , Waxman IM , Sharma P . Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med. (2015) ;373: (19):1803–13. |
[35] | Escudier B , Sharma P , McDermott DF , George S , Hammers HJ , Srinivas S , Tykodi SS , Sosman JA , Procopio G , Plimack ER , Castellano D , Gurney H , Donskov F , Peltola K , Wagstaff J , Gauler TC , Ueda T , Zhao H , Waxman IM , Motzer RJ . CheckMate 025 randomized phase 3 study: Outcomes by key baseline factors and prior therapy for nivolumab versus everolimus in advanced renal cell carcinoma. Eur Urol. (2017) ;72: (6):962–71. |
[36] | Motzer RJ , Tannir NM , McDermott DF , Aren Frontera O , Melichar B , Choueiri TK , Plimack ER , Barthelemy P , Porta C , George S , Powles T , Donskov F , Neiman V , Kollmannsberger CK , Salman P , Gurney H , Hawkins R , Ravaud A , Grimm MO , Bracarda S , Barrios CH , Tomita Y , Castellano D , Rini BI , Chen AC , Mekan S , McHenry MB , Wind-Rotolo M , Doan J , Sharma P , Hammers HJ , Escudier B . Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med. (2018) ;378: (14):1277–90. |




