Preliminary assessment of a standardized vision screening guideline in a pediatric inpatient rehabilitation unit
Abstract
OBJECTIVE:
This study aimed to evaluate the impact of a standardized vision screen guideline on occupational therapy vision screens in a pediatric inpatient rehabilitation unit.
METHODS:
Charts of patients admitted to a pediatric inpatient rehabilitation before guideline implementation (n = 47) versus charts after implementation (n = 47) were randomly, retrospectively reviewed to explore differences in visual skills screened and use of standardized assessments.
RESULTS:
Significant improvements (p < = 0.05) were found in the number of visual skills screened (p = 0.034), use of standardized assessments (p = 0.005), and screening of the specific visual skills of accommodative amplitude (p = 0.05), suppression (p = 0.015), and double vision (p < 0.001).
CONCLUSION:
Implementation of a standardized vision screen guideline improved the frequency of vision screens during occupational therapy evaluations in a pediatric inpatient rehabilitation unit. The use of standardized assessments may also improve the quality of vision screens by encouraging staff to complete more comprehensive vision screens, including screening more visual skills, and by prompting use of standardized assessments, which can improve accuracy of screening procedures.
1Introduction
Occupational therapists (OTs) are concerned with an individual’s ability to participate in daily life activities, including completion of desired, necessary, and expected tasks. Vision problems can negatively impact performance in many daily occupations such as reading, writing, academic tasks, driving, participation in sports/leisure tasks, instrumental activities of daily living, and more [1, 2]. Common symptoms of visual impairments include headaches, eyestrain, blurred vision, double vision, reading problems, poor athletic performance, difficulty with rhythmic activities, lack of coordination and balance, difficulty handwriting, and difficulty learning left and right [1, 2]. Fourteen percent of typically developing children have a non-strabismic binocular vision or accommodative disorder, and 12% will have a strabismic binocular vision disorder [3]. Children with developmental disabilities such as cerebral palsy, Down syndrome, autism, and learning disabilities have a much higher prevalence of visual impairments than their peers [2, 4–7]. Untreated, undocumented vision problems could limit a child’s independence with daily tasks and interfere with the child’s progress toward goals with occupational therapy intervention.
The American Academy of Pediatrics (AAP) and the American Academy of Ophthalmology (AAO) recommend that all children receive vision screens at birth, at 6–12 months, at 12–36 months, between 3–5 years, and intermittently after the age of five [9]. It is recommended that vision checks include screening for strabismus, amblyopia, refractive errors, other “focusing” problems, and eye movements [9]. Wall [10] and Kemper [11] found that many pediatricians do not follow AAP guidelines for vision screening, especially in children under age five [10, 11]. Many pediatricians recommended vision tests before the child was five years old but were not performing all the recommended tests at each health visit [10]. Even when pediatricians report they are assessing visual skills, they are not consistently using objective measures as recommended by the AAP [11]. The primary tests completed at preschool and infant health visits include red reflex testing, visual acuity testing, and cover or stereopsis testing [10, 11].
Based on the populations commonly served, it is therefore likely that pediatric OTs are evaluating and treating children with visual impairments [12]. They might not have had a recent visual screening, and if it was completed, the child might not have had a comprehensive enough screen to detect all the potential visual impairments that could impact their ability to engage in daily occupations. Thus, the OT needs to be able to also provide comprehensive visual screening to identify and address all client factors and performance skill deficits that could be impacting occupational performance.
In addition to working with children with increased risk for developmental visual impairments, those working on an inpatient rehabilitation unit are likely to serve clients with increased risk of visual impairments due to acute injuries or illnesses. Children with brain injuries present with increased incidence of visual impairments such as convergence insufficiency, accommodative dysfunction, and saccadic eye movement dysfunction [8]. Pollack [13] surveyed OTs working in inpatient adult stroke settings and found that they were not consistently or comprehensively screening vision despite working with a population with a well-documented incidence of visual deficits [13]. Sixty-two percent of respondents reported their unit did not have a vision-related protocol or management plans. Only 69% of respondents reported they carried out a visual assessment during their initial occupational therapy evaluation [13]. Most respondents (89% ) indicated the primary visual skills assessed were visual attention and scanning [13]. Less than half of respondents endorsed assessment of binocular vision or ocular movements when completing vision screens [13]. Similarly, Reiser [14] found a wide variety of visual screening practices used by OTs in patients with concussion, another population with well-established increased prevalence of vision impairments [14]. Only 29% of therapists surveyed completed vision-specific screens as part of their evaluation for individuals with concussion [14]. The most common visual skill assessed was acuity, followed by accommodation. Less than 50% of therapists completing vision screens were evaluating skills such as binocular vision or eye movements [14]. Additionally, therapists assessing visual skills primarily used functional tasks to evaluate skills versus objective measures for all skills except convergence insufficiency [14]. It is important to ensure all visual impairments that could be impacting occupational performance are considered with occupational therapy treatment planning and intervention; thus, OTs should be given clear guidance related to suitable and appropriate assessments of vision to ensure comprehensive care is provided to clients.
Given the lack of standardization of care of vision screens completed by OTs, a “consensus panel was convened and charged with evaluating existing vision tests and proposing the composition of a clinically feasible and psychometrically optimal occupational therapy vision screen for service members with traumatic brain injuries” (TBIs) [15]. Using a modified nominal group technique, the panel identified a “better practice” occupational therapy TBI vision screen which includes nine subtests to assist OTs in consistently administering a comprehensive vision screen. Radomski [15] recommends all OTs working with adults who have mild traumatic brain injury (mTBI) administer this “best practice” screen to ensure they are making appropriate referrals for full vision examination by specialists and that clients receive necessary therapy and education to manage vision problems during functional activities.
Information about vision should be considered when treatment planning for children admitted to an inpatient rehabilitation unit. Current examinations of pediatric vision may be limited, with minimal attention to screening for binocular vision, accommodation, or eye movements. Although not a replacement for a comprehensive vision examination from an eye doctor, OTs can be trained to screen a child’s vision, identify the potential need for additional examination, and educate caregivers on the impact visual impairments could be having on a child’s occupational performance. Although Radomski [15] suggested the “best practice” screen be used with adults with mTBI, all of their recommended measures include administration guidelines or normative values for children. It therefore can be used appropriately in pediatric populations. Research shows that children are often not receiving a visual screen at all or, when the vision screen is completed, it does not include adequate screening of the visual skills needed to effectively participate in their daily activities. This study sought to identify if using the Radomski [15] proposed vision screen with modifications for the pediatric population could afford a pediatric occupational therapy department a standardized process to complete visual screens. It aimed to identify if a standardized process for visual screens would be more accurate and comprehensive in identifying visual needs than subjective observations and/or other non-standardized methods. It was hypothesized that implementation of the guideline would result in therapists completing more vision screens, which would result in screening more visual skills and would also increase the use of standardized assessments when screening vision.
2Methods
2.1Context
A retrospective study was designed to evaluate the impact of screening and documentation of pediatric occupational therapy vision screening before and after the implementation of a formal vision screening guideline for OTs on the inpatient rehabilitation team at the Children’s Hospital of Philadelphia (CHOP).
2.1.1Developing the vision screen guideline
CHOP’s occupational therapy documentation is completed using the electronic medical record software EPIC® (Epic Electronic Health Record, Madison, Wisconsin, USA).
This evaluation data is input into flowsheets before being pulled into a note. Prior to implementation of the updated vision screen guideline, there were two flowsheets specifically designated for vision: vision basic and vision complex. The preexisting flowsheets included drop-down options and free text ability for the following visual skills: functional acuity, refraction, eye disease, patient-reported visual symptoms, ocular range of motion, amblyopia, head position, ptosis, fixation, pursuits, saccades, convergence, peripheral visual fields, and diplopia assessment.
The updated vision screening guideline consisted of a two-part vision screen which included a section titled Standard Vision Screen (SVS) and Standard Vision Screen Plus (SVS+). Per the guideline, OTs should complete the SVS in every evaluation. Based on results of the SVS or child’s diagnosis, the therapist may complete the comprehensive vision screen, SVS+ (see Figure 1).
Fig. 1
Vision screen guideline decision tree.
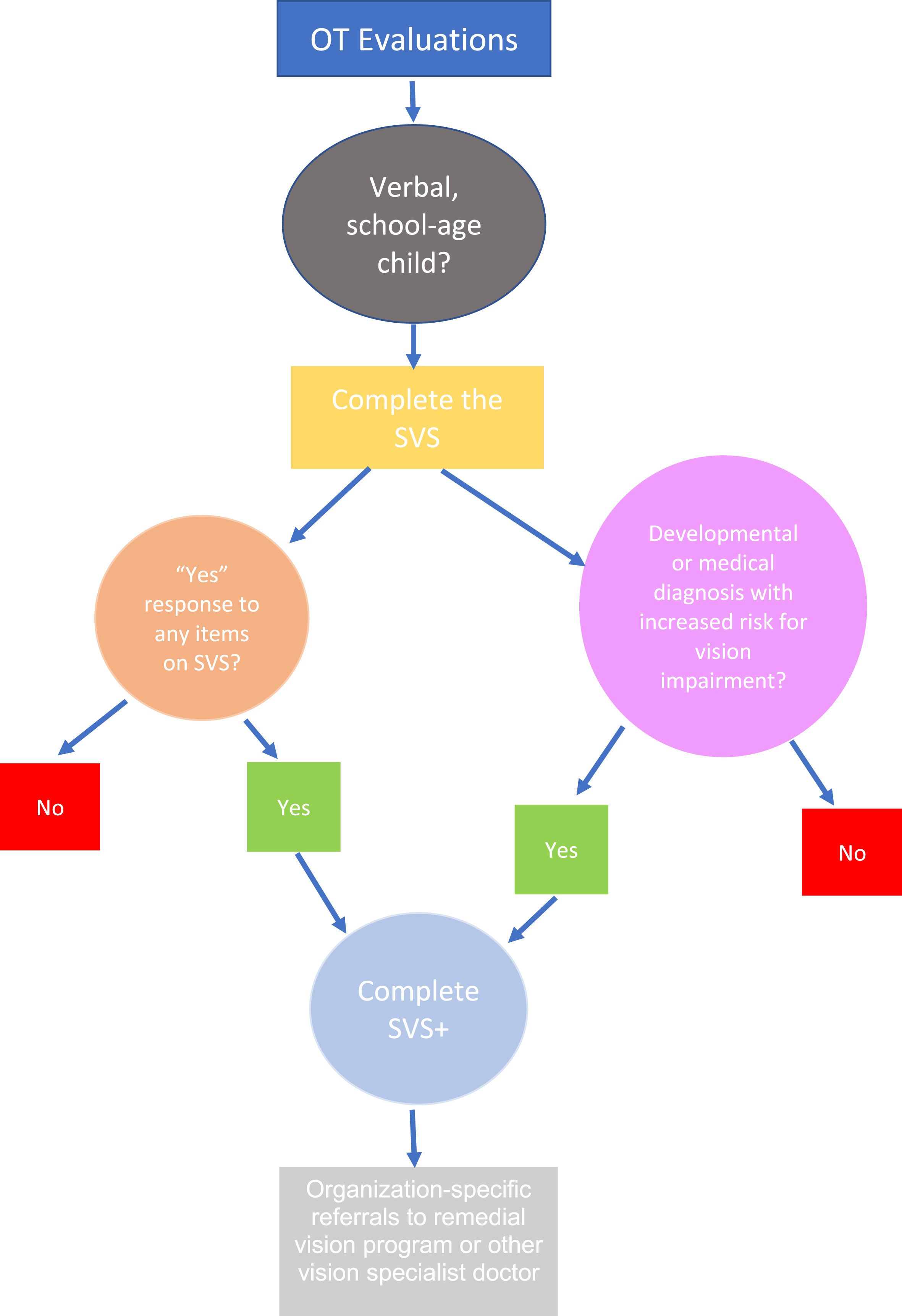
The updated vision screen guideline was introduced to the occupational therapy department at CHOP on January 1, 2021, via a virtual in-service. An in-person hands-on lab was held in February 2021 with the inpatient rehabilitation team to provide additional opportunity to practice the SVS+ and all assessments included in the screening as part of the new guideline. Charts from January 11, 2019, through November 2, 2021, were pulled for analysis.
2.1.2SVS
The SVS was intended to be a quick two-part screen that includes a clinician’s screening of the child’s face for obvious signs of visual impairment as well as a brief screen for common symptoms of visual deficits. The SVS includes an examination of the child’s face for signs of ptosis, visible misalignment of the eyes, and nystagmus. The therapist should confirm with parents if there is a history of eye misalignment or nystagmus and ask the child if they have ever experienced any of the following: headaches with reading or close work, blurry vision, or double vision. If there is an affirmative answer to any of the items on the SVS, therapists are to complete the full SVS+. Additionally, if the child has a known or suspected vision problem, a known or suspected learning disability, and/or a history of an acquired brain injury, the OT will complete the SVS+.
2.1.3SVS+
The SVS+ was adapted from the Radomski “best practice” vision screen [14]. It includes all visual skills recommended to be evaluated and all standardized assessments recommended for screening these visual skills except for the visual symptom questionnaire. The vision symptom questionnaire was adapted from a visual symptom screen created and published by Scheiman et al. [7] and was adapted to screen visual symptoms more appropriately in children. All other standardized vision assessments included in the SVS+ are intended for use in pediatric populations and include pediatric age-based norms.
The visual skills screened include visual symptoms, far acuity, accommodative amplitude, near point of convergence, eye alignment and binocular vision, saccadic eye movement, visual pursuits, and visual fields. Far acuity is assessed using a Snellen eye test chart for 10 feet. This chart was chosen due to environment and space restrictions within the hospital. Accommodative amplitude and near point of convergence are both assessed using a convergence ruler. Eye alignment and binocular vision are screened using the binocular vision assessment (BVA) screening software using the Worth 4 Dot and Phoria tests. The guideline also includes an option to use the Modified Thorington Procedure to screen for phoria. This was included particularly for use on the inpatient acute medical floors as children are often unable to transition to the therapy gym with the BVA software. Inclusion of the Modified Thorington Procedure improved the generalizability of the visual screen to other settings beyond an inpatient rehabilitation unit. Saccades are screened using the Developmental Eye Movement (DEM) test [16]. The Northeastern State University College of Optometry (NSUCO) Oculomotor Test saccades screen is also included as an option for children unable to complete the DEM. Pursuits are screened using the NSUCO. Visual fields are screened using confrontation field testing. Except for the DEM, all screens are utilized in intended pass or fail manner. The DEM is scored using age-based normative values. If a child scores lower than one standard deviations below the mean compared to same age peers, the child fails the DEM screen. Referral to a specialist eye doctor for a comprehensive vision screen is recommended for all children who fail any screens within the SVS+.
2.1.4Changes to documentation
Changes were made to the electronic medical record to reflect the clinical practice changes implemented.
2.2Samples
2.2.1Patients
Sample size for each cohort was originally set to 50, as this seemed achievable given the working time frame of the study. Given the 18-bed inpatient rehabilitation unit at CHOP, the sample size of 50 seemed appropriate to get results representative of the target population. The pre-implementation group (Cohort A) included children admitted to the CHOP inpatient rehabilitation unit from January 11, 2019, to January 31, 2021. The post-implementation group (Cohort B) included children admitted to the CHOP inpatient rehabilitation unit from February 1, 2021, to November 2, 2021, which represented approximately nine months of data immediately following implementation. Children six years or older were included during inpatient rehabilitation unit admission for both groups. Those admitted to the inpatient rehabilitation unit with a primary diagnosis of functional neurologic disorder or to participate in the Amplified Musculoskeletal Pain Syndrome program were excluded. Both groups received evaluations and vision screening from the same six primary OTs on the inpatient rehabilitation unit.
A research analyst queried CHOP’S ARCUS Rehabilitation database to identify initial possible participants who met previously identified inclusion and exclusion criteria [18]. Four hundred ninety seven medical records for Cohort A and 171 medical records for Cohort B were identified. One hundred seven medical records from Cohort A and 49 medical records from Cohort B were then excluded due to a diagnosis of functional neurologic disorder or multiple admissions for a single individual, which occurred due to medical procedures. Fifty potential participants were then selected using a random number generator. Research ID numbers were assigned once the final data set was identified. A manual review of medical records was then completed by the lead author, who identified an additional three patients from Cohort A and three from Cohort B for exclusion due to a diagnosis of functional neurologic disorder that had not been coded into the medical records. Forty-seven medical records for Cohort A and 47 for Cohort B were then used for analysis.
Arousal level was classified by the primary author by review of charts and occupational therapy inpatient rehabilitation admission initial evaluations. For purposes of comparing groups, arousal was categorized as minimal, impaired, or unimpaired consciousness.
Cognition and language status were classified by the primary author by review of charts and occupational therapy inpatient rehabilitation admission initial evaluations. For purposes of comparing groups, cognition and language were categorized as moderate to severe, mild, or no impairment. There was not consistent assessment of language and cognition across charts reviewed; some patients received cognition WeeFIM scores; however, others included primary evaluating OT subjective assessment of cognition and language [18]. If cognition and language skills were noted to be normal by subjective assessment or WeeFIM (score of six [modified independent] or seven [independent]) on the five cognition items, the patient was classified as no impairment. If there were higher level cognition or language impairments noted by the primary evaluating therapist, such as WeeFIM scores of four (minimal assistance) or five (stand by assistance), or therapist documentation of higher-level executive function or memory concerns, the patient was classified as mild impairment. If the cognition WeeFIM scores or subjective assessment documented multiple concerns related to language and cognition, for example, WeeFIM scores of three out of seven or less, or documentation of difficulty following verbal directions, the patient was classified as moderate to severe impairment. Average WeeFIM scores of four (minimal assistance) were included in the mild cognitive impairment category for classifying cognition since a patient scoring a four would be able to follow two unrelated verbal commands, communicate clearly 75% of the time, and recall 75% of daily information [18]. It was determined that these cognitive skills would enable a patient to complete the standardized test items included on the SVS+.
Due to the variety of diagnoses for children admitted to the inpatient rehabilitation unit at the CHOP, they were categorized into one of the following general categories: neurologic (other than functional neurologic disorder), orthopedic, or other. Children considered neurologic included those admitted after TBI, pediatric stroke, other acquired brain injuries such as encephalopathy, or with a seizure disorder. Those considered orthopedic included children admitted after orthopedic surgery. Children considered other had a primary diagnosis of deconditioning due to cardiac, pulmonary, or non-neurologically-based oncology diagnosis. Admitting diagnosis was determined by the admitting rehabilitation physician and identified via chart review.
2.3Measures
For both cohorts, it was noted whether the OT screened for the following skills during the child’s admission to the inpatient rehabilitation unit at CHOP: visual symptoms, far acuity, accommodative amplitude, near point of convergence, suppression, double vision, phoria, saccades, pursuits, and visual fields. These items were selected as they are the visual skills included on the SVS+. Binary criteria (yes/no) were used, indicating whether the visual skill was screened. Additionally, the number of visual skills assessed and how many standardized assessments were used to screen vision were counted. When counting the number of visual skills screened, fixation was included in addition to the visual skills listed above because it was included as a visual skill on occupational therapy evaluation templates prior to implementation of the updated visual screening guideline. It continues to be available on occupational therapy evaluation templates. If a therapist noted presence or lack of blurry vision or that the child denied visual changes, credit was given for screening for visual symptoms. When counting the number of visual skills assessed, repetitive skills were not included. For example, if a therapist noted intact tracking but also commented on saccades and pursuits, this was counted as two skills instead of three, as tracking is technically a combination of saccadic and pursuit eye movements. If the therapist reported that a child did not have amblyopia, this was counted as screening for it even if there was no additional supporting documentation. According to the Academy of Ophthalmology, amblyopia is diagnosed by measuring visual acuity on an eye chart or using an instrument-based devices [9]. Per chart review and understanding of visual screening procedures on the inpatient rehabilitation unit prior to implementation of the visual screening guideline, OTs were not using either of these methods during vision screens; it remains unclear how, if at all, OTs were screening for amblyopia prior to implementation of the guideline. If the therapist noted that the child could read signs, clocks, or other environmental items, far acuity was counted as screened. Many therapists noted if a child did or did not wear corrective lenses at baseline, but this information was not sufficient to be counted as a screen for far acuity. Some OTs included findings from a recent ophthalmology or neuro-ophthalmology evaluation; however, this information was not included as satisfying the OT screening for visual skill.
2.4Analysis
Gender, diagnosis, and primary OT were compared using Pearson X2. Arousal and cognitive/language levels were compared using a Mann Whitney U test for ordinal data. Mean age was compared using an independent t-test. The number of visual skills assessed and the number of standardized assessments used were compared using independent t-tests to explore group differences. Categorical variables (for example, was a vision screen completed: yes or no) were compared using a Pearson X2 test. Significance for all tests was determined to be p < /=.05.
3Results
Both cohorts consisted of 47 children who met inclusion criteria following randomization. Other than language/cognitive status, there were no significant differences in baseline characteristics between the cohorts. Cohorts A and B had patients with a similar mean age (12 years, 14 years), proportion of females (48.9% , 40.4% ), primary admitting diagnosis (neurologic diagnosis for 63.8% , 57.4% ), and arousal level at admission (no impairment of consciousness 95.8% , 91.5% ). Both cohorts also had a similar distribution of which rehabilitation OT completed the initial evaluation. Cohort B had more children with mild and moderate cognitive and language impairments: 41% of Cohort A were patients without cognitive or language impairments, but only 17% of Cohort B.
3.1Comparison of vision screening between Cohort A and Cohort B
After implementation of the vision screen guideline, there was a significant increase in the number of visual skills that were assessed and a significant increase in the number of standardized assessments used to screen vision. See Table 2.
Table 1
Patient population comparison between cohorts
| Characteristics | Cohort A | Cohort B | Group comparability |
| Mean Age, years | 12.68 (SD = 3.77) | 14.28 (SD = 4.26) | Independent t-test; p = .058 |
| Gender, % female | 48.9 (n = 23) | 40.4 (n = 19) | Pearson Chi-Squared; p = .407 |
| Primary admitting diagnosis (PAD) neurologic, % | 63.8 (n = 30) | 57.4 (n = 27) | Pearson Chi-Squared; p = .796 |
| PAD orthopedic, % | 21.3 (n = 10) | 23.5 (n = 11) | |
| PAD other, % | 14.9 (n = 7) | 19.1 (n = 9) | |
| No impairment of consciousness at admission, % | 95.8 (n = 45) | 91.5 (n = 42) | Mann Whitney U; p = .245 |
| Impaired consciousness at admission, % | 2.1 (n = 1) | 6.3 (n = 3) | |
| Minimally conscious at admission, % | 2.1 (n = 1) | 4.2 (n = 2) | |
| No cognitive or language impairment at admission, % | 40.5 (n = 19) | 17 (n = 8) | Mann Whitney U; p = .035* |
| Mild cognitive or language impairment at admission, % | 36.1 (n = 18) | 53.2 (n = 25) | |
| Moderate to severe cognitive or language impairment at admission, % | 23.4 (n = 10) | 29.8 (n = 14) | |
| Primary occupational therapist A, % | 21.4 (n = 10) | 19.1 (n = 9) | Pearson Chi-squared; p = .582 |
| Primary occupational therapist B, % | 17 (n = 8) | 17 (n = 8) | |
| Primary occupational therapist C, % | 21.4 (n = 10) | 21.4 (n = 10) | |
| Primary occupational therapist D, % | 21.4 (n = 10) | 17 (n = 8) | |
| Primary occupational therapist E, % | 12.8 (n = 6) | 23.4 (n = 11) | |
| Primary occupational therapist F, % | 6 (n = 3) | 2.1 (n = 1) |
Table 2
Mean visual skills assessed and mean number of standardized assessments used
| Cohort A Mean | Cohort B Mean | Independent t-tests | |
| Number of visual skills screened | 3.47 (SD 1.86) | 4.7 (SD 3.39) | p=.031* |
| Mean difference: –1.234 | |||
| 95% CI: –2.354, –.114 | |||
| Number of standardized assessments used in vision screen | .02 (SD.146) | 1.36 (SD 3.12) | p=.004* |
| Mean difference: –1.34 | |||
| 95% CI: –2.246, –.434 |
The total number of patients who received a vision screen at initial evaluation was significantly less in Cohort B than in Cohort A, although Cohort B underwent significantly more screening for accommodative amplitude, suppression, and double vision. There was no significant change in the screening of visual symptoms, far acuity, near point of convergence, phoria, saccades, pursuits, or visual fields. See Table 3.
Table 3
Visual skills assessed
| % yes in Cohort A | % yes in Cohort B | Pearson X2 | |
| Vision screen completed | 97 (n = 48) | 79 (n = 37) | p=.004* |
| Visual symptoms | 46 (n = 22) | 53 (n = 25) | p=.54 |
| Double vision | 19 (n = 9) | 53 (n = 25) | p<.001* |
| Pursuits | 57 (n = 27) | 49 (n = 23) | p=.414 |
| Near point of convergence | 36 (n = 17) | 34 (n = 16) | p=.831 |
| Saccades | 28 (n = 13) | 30 (n = 14) | p=1 |
| Phoria | 11 (n = 5) | 19 (n = 9) | p=.251 |
| Visual fields | 21 (n = 10) | 19 (n = 9) | p=1 |
| Suppression | 2 (n = 1) | 17 (n = 8) | p=.015* |
| Far acuity | 12 (n = 6) | 15 (n = 7) | p=.0768 |
| Accommodative amplitude | 2 (n = 1) | 13 (n = 6) | p=.05* |
Table 3 caption: Percentage of visual screens completed as well as the percentage of visual skills screened as part of the OT evaluation in each Cohort.
3.2Evaluation of two-part process in Cohort B
Thirty seven of the 47 patients reviewed in Cohort B received a vision screen. Of those 37, 28 should have received additional screening with the SVS+ due to a positive answer to items on the SVS or due to admitting diagnosis. Eight of the 28 patients (28.5% ) did receive additional vision screening via completion of the SVS+, whereas 20 did not receive the SVS+ despite indications per the guideline. For six of those 20 patients (30% ), the evaluating OT indicated that the SVS+ was unable to be completed due to patient deficits in language, cognition, or arousal. Seven of them (35% ) did not answer “yes” to any questions on the SVS; however, they should have received the SVS+ per the vision screen guideline because a primary admitting diagnosis put the patient at increased risk of visual impairments.
4Discussion
As members of the interdisciplinary team who address functional vision problems, occupational therapy practitioners should be screening patients’ visual functions routinely [15]. However, Pollack [13] and Reiser [14] found that OTs working in populations with increased likelihood of visual deficits were not consistently screening vision as part of their standard evaluation. Pollack [13] also found that 62% of respondents worked on a unit or in an institution that did not have a specific vision-related protocol or management plan. This study aimed to evaluate if implementation of a vision screening protocol could improve vision screening procedures.
Two of the three intended outcomes of the study were achieved. OTs screened significantly more visual skills than they were prior to implementation of the guideline. Also, they were using significantly more standardized assessments in lieu of subjective therapist observations when screening vision than prior to implementation of the guideline. After implementation of the guideline, there was a significant improvement in the number of visual skills assessed, which reflects a more comprehensive evaluation of the child’s vision. Specifically, therapists were significantly more likely to screen for accommodative amplitude, suppression, and double vision following implementation of the guideline than they were prior.
This is important due to increased prevalence of accommodative disorders, suppression, and binocular vision disorders in children with cerebral palsy, Down syndrome, and learning disabilities [2–7]. Additionally, deficits in these skill areas can often have a functional impact. Accommodative disorders can result in difficulties with reading and writing [19]. Binocular vision deficits can result in double or blurred vision [19]. Pediatric patients admitted to an inpatient rehabilitation unit are asked to participate in many reading and writing tasks during their daily participation in rehabilitative therapies. Additionally, deficits such as double and blurred vision can impact a child throughout the day. For example, these deficits can limit a child’s ability to safely or comfortably complete basic functional transfers and mobility or reach and grasp items during self-care completion. Suppression can limit depth perception and balance, further impacting a child’s ability to participate in a variety of activities expected of them [19]. It is important for pediatric OTs to consider the presence and impact of visual deficits when treatment planning, adapting daily tasks and environments, and educating caregivers.
Despite the lack of significant improvement in the screening of some visual skills, such as convergence insufficiency, phoria, saccades, pursuits, and visual fields, there was a significant improvement in the use of standardized assessments when screening vision overall. Based on chart reviews of OTs’ evaluations prior to the updated guideline, most were using subjective means to screen visual impairments. A primary goal of the study was to improve the quality and accuracy of vision screens implemented by OTs by shifting to the use of standardized, objective measures.
Skills such as phoria are unable to be assessed without specific technology or assessments. Documentation utilized prior to implementation of the updated vision screen guideline included a flowsheet for therapists to note whether a child presented with strabismus or phoria. Therapists were documenting if a visible eye misalignment was noted. However, phorias are not visible to the naked eye and therefore cannot be assessed without an appropriate tool [1]. Similarly, chart reviews showed that therapists were screening far acuity at baseline by asking the child to read signs, clocks, or other environmental items. This brings into question the accuracy of the vision screens being completed prior to the implementation of the vision screen guideline. This study was not specific enough to capture specific changes made to screening of each visual skill, such as a shift from a quick, objective screen of far acuity to standardized screening using the Snellen chart. Similarly, there was no significant improvement in the assessment of visual symptoms; however, prior to the guideline, chart reviews revealed that therapists were assessing visual symptoms by asking the child or caregivers if the child had blurry vision or was experiencing visual changes. If the therapist used the SVS+, screening of visual symptoms was completed using a standardized questionnaire of 11 questions reflecting a more comprehensive screen of a wider variety of common visual symptoms. An overall improvement in the use of standardized assessments following implementation of the guideline should reflect improved quality and accuracy of vision screens completed.
Another potential reason why there was no significant improvement in the assessment of some specific visual skills after implementation of the vision screen guideline could be related to the two-part screening process. Per the guideline, therapists were encouraged to complete the SVS during every occupational therapy evaluation. If there were no affirmative answers to items on the SVS and there was not an acute diagnosis that warranted additional screening, the guideline did not recommend it. Of the 37 in Cohort B who received a vision screen, nine did not require additional vision screening per the guideline; there was no answer of “yes” to any items on the SVS and their primary admitting diagnosis did not warrant additional vision screening. Prior to implementation of the guideline, these patients could have received additional, unnecessary vision screening. This could represent an unintended positive outcome of implementation of the vision screen guideline, i.e., a reduction in time spent on unnecessary, non-standardized screening.
Despite improvements in two of the three intended outcomes of the study, there was not a significant increase in visual screens completed overall. One possible reason for this is that there was a significant difference in cognitive and language deficits between the cohorts. Cohort B had significantly more participants with cognitive and language deficits, which could have impacted their ability to participate and complete many of the standardized assessments included in the SVS+. Of the 37 who received a vision screen in Cohort B, six should have received additional screening via the SVS+; however, the primary OT who completed the initial evaluation noted they were unable to complete it due to language, cognition, or arousal deficits. The updated vision screen guideline was intended for pediatric clients who were verbal and at least three years old. Most of the assessments included on the SVS+ required patients to follow at least one-step motor commands and verbalize responses. The updated vision screen guidelines do not support visual screening of pediatric clients with cognitive, language, or arousal impairments. Thus, Cohort B might have seen a reduction in the number of vision screens completed overall due to these limitations.
The two-part vision screen process could have further impacted results in Cohort B due to lack of education or understanding by the primary therapist on the need to complete the SVS+ based on primary admitting diagnosis. Seven patients in Cohort B should have received SVS+ screening based on primary admitting diagnosis but did not have a “yes” on any items on the SVS. Without this, therapists could have determined additional vision screening was not necessary. This could reflect an educational need to emphasize the importance of both criteria (performance on SVS and primary admitting diagnosis) when determining need to complete the SVS+.
5Limitations
Limitations of this study included an inability to evaluate improvements in quality and accuracy of specific visual skill assessment (for example, how accurate a therapist was in screening for phoria). There was a lack of appropriate equipment and training to accurately assess many visual skills prior to implementation of the vision screen guideline. The availability of correct screening equipment (such as the BVA, convergence and accommodation ruler, and DEM assessment), as well as the vision screen training that occurred with implementation of the vision screen guideline, should have resulted in more accurate assessment of many visual skills. However, the scope of this study and the retrospective chart review process limited the ability to definitively evaluate specific changes in quality and accuracy. The study was limited to demonstrating only a significant improvement in quantity of visual skills assessed and overall use of standardized assessments.
Future studies could investigate if use of the brief SVS during an occupational therapy evaluation allowed an OT to screen efficiently and effectively for visual impairment while simultaneously increasing the amount of time available for other assessments in the evaluation.
The timing of this study might have influenced results and evaluation of changes made following implementation of the guideline. Charts were pulled beginning five days after implementation of the guideline and for only nine months following implementation; the time frame was dictated by requirements to complete this research in line with the primary author’s doctoral capstone schedule. Any change, especially change at a large institution such as CHOP, takes time. The time frame used for chart reviews for this study might not have been long enough to accurately capture the changes to vision screening processes. Over time, therapists are able to update their workflow and practice. Capturing data further from implementation of the vision screen guideline could better reflect changes that occurred.
The generalizability of this study is limited as it evaluated the impact of a vision screen guideline only on a pediatric inpatient rehabilitation unit. It would be beneficial to repeat this study on other adult and pediatric occupational therapy locations to identify if this vision screen guideline could be useful to improve vision screens in other areas of practice.
The vision screening guideline itself was limited in that it was appropriate for use only in school-age, verbal children. This study revealed that the current vision screen guideline may not be suitable to screen vision in pediatric clients with impaired arousal level, cognition, or language skills. Future studies might consider excluding pediatric patients with these impairments in this type of comparison. Additionally, to improve the ability of an OT to complete a comprehensive vision screen with all patients served in the pediatric hospital environment, it would be beneficial to identify an appropriate vision screen to be completed in infants as well as in pediatric patients with impaired arousal, cognition and/or language.
6Conclusions
Implementation of a standardized vision screen guideline may improve the inclusion of vision screens in an occupational therapy evaluation on a pediatric inpatient rehabilitation unit. It may also improve the quality of vision screens completed by OTs by increasing the use of standardized assessments to screen for visual impairments and by increasing the number of visual skills screened by OTs.
Acknowledgments
We would like to thank our colleagues for their ongoing support and contributions to this improvement project. We would specifically like to thank Sarah Jamieson for her contribution to creating the vision screen guideline and her role in educating the inpatient rehabilitation team at the Children’s Hospital of Philadelphia on updated processes and procedures related to the updated vision screen guideline.
Conflict of interest
There was no funding obtained or used to enable this research. The authors have no conflict of interest to report.
Ethical considerations
In accordance with institutional review board (IRB) standards, this project was submitted to the IRB and met criteria to be exempt from review, as the study used retrospective chart reviews and study participants were de-identified.
References
[1] | Scheiman M . Understanding and managing vision deficits: A guide for occupational therapists. 3 rd ed. Thorofare, NJ SLACK Incorporated; (2011) . |
[2] | Salt A , Sargent J . Common visual problems in children with disability. Arch Dis Child. (2014) ;99: (12):1163–8. doi: 10.1136/archdischild-2013-305267. |
[3] | Scheiman M , Gallaway M , Coulter R , et al. Prevalence of vision and ocular disease conditions in a clinical pediatric population. J Am Optom Assoc (1996) ;67: (4):193–202. |
[4] | Scheiman MM . Optometric findings in children with cerebral palsy. Am J Optom Physiol Opt. (1984) ;61: (5):321–3. doi: 10.1097/00006324-198405000-00005. |
[5] | Jaeger EA . Ocular findings in Down’s syndrome. Trans Am Ophthalmol Soc. (1980) ;78: , 808–45. |
[6] | Scharre JE , Creedon MP . Assessment of visual function in autistic children. Optom Vis Sci. (1992) ;69: (6):433–9. doi: 10.1097/00006324-199206000-00004. |
[7] | Scheiman M , Rouse MW . Optometric management of learning-related vision problems. 2 nd ed. Mosby Inc; St. Louis, MO.2006. |
[8] | Master CL , Scheiman M , Gallaway M , et al. Vision diagnoses are common after concussion in adolescents. Clin Pediatr (Phila). (2016) ;55: (3):260–7. doi: 10.1177/0009922815594367. |
[9] | Gudgel D . Eye Screening for Children. American Academy of Ophthalmology; 2021 [updated 23 March 2021; cited 20 November 2022]. Available from: https://www.aao.org/eye-health/tips-prevention/children-eye-screening. |
[10] | Wall TC , Marsh-Tootle W , Evans HH , Fargason CA Jr , Ashworth CS , Hardin JM . Compliance with vision-screening guidelines among a national sample of pediatricians. Ambul Pediatr. (2002) ;2: (6):449–55. doi: 10.1367/1539-4409(2002)002<0449:cwvsga>2.0.co;2. |
[11] | Kemper AR , Clark SJ . Preschool vision screening in pediatric practices. Clin Pediatr (Phila). (2006) ;45: (3):263-6. doi: 10.1177/000992280604500309. |
[12] | More DH . Fact Sheet: Occupational therapy’s role with children and youth. Bethesda, MD: American Occupational Therapy Association; 2015 [cited 7 April 2023]. Available from: https://irp.cdn-website.com/de1c86c3/files/uploaded/Children%20and%20Youth%20fact%20sheet%20AOTA.pdf |
[13] | Pollock A , Hazelton C , Brady M . Visual problems after stroke: a survey of current practice by occupational therapists working in UK stroke inpatient settings. Top Stroke Rehabil. (2011) ;18: (sup1):643-51. doi: 10.1310/tsr18s01-643. |
[14] | Reiser A , Bunin G , Scheiman M . Concussion-Related Vision Disorder Practice Patterns in Occupational Therapy: A Survey. The Open Journal of Occupational Therapy. (2020) ;8: (4):1-20. doi: 10.15453/2168-6408.1737. |
[15] | Radomski MV , Finkelstein M , Llanos I , Scheiman M , Wagener SG . Composition of a vision screen for servicemembers with traumatic brain injury: Consensus using a modified nominal group technique. Am J Occup Ther. (2014) ;68: (4):422-9. doi: 10.5014/ajot.2014.011445. |
[16] | Garzia RP , Richman JE , Nicholson SB , Gaines CS . A new visual-verbal saccade test: The Developmental Eye Movement test (DEM). Journal of the American Optometric Association. (1990) ;61: (2):124–135. |
[17] | Camacho P . CHOP Center for Rehabilitation Research Registry [database]. Version 1.4. Arcus at Children’s Hospital of Philadelphia. [cited 28 January 2022]. |
[18] | Slomine BS , Grasmick PH , Suskauer SJ , Saloria CF . Psychometric properties of the Cognitive and Linguistic Scale: A follow-up study. Rehabil Psychol. (2016) ;61: (3):328-35. doi: 10.1037/rep0000096. |
[19] | Harris PF . Diagnosis and treatment of oculomotor dysfunction. In: Taub MB , Bartuccio M , Maino DM , editors. Visual Diagnosis and Care of the Patient with Special Needs. Philadelphia: Lippincott Williams & Wilkins; (2012) . |
[20] | Roberts PS , Rizzo J-R , Hreha K , et al. A conceptual model for vision rehabilitation. J Rehabil Res Dev. (2016) ;53: (6):693–704. doi: 10.1682/JRRD.2015.06.0113. |




