The Importance of Digital Health Literacy in an Evolving Parkinson’s Disease Care System
Abstract
Digital health technologies are growing at a rapid pace and changing the healthcare landscape. Our current understanding of digital health literacy in Parkinson’s disease (PD) is limited. In this review, we discuss the potential challenges of low digital health literacy in PD with particular attention to telehealth, deep brain stimulation, wearable sensors, and smartphone applications. We also highlight inequities in access to digital health technologies. Future research is needed to better understand digital health literacy among individuals with PD and to develop effective solutions. We must invest resources to evaluate, understand, and enhance digital health literacy for individuals with PD.
INTRODUCTION
Health literacy refers to “the degree to which individuals have the ability to find, understand, and use information and services to inform health-related decisions and actions for themselves and others” [1]. Among older adults, low health literacy is associated with worse health outcomes, including increased risk of hospitalization and mortality [2]. With the advent of new technology emerged the concept of eHealth literacy, which was originally defined as the ability to obtain, critically appraise, and apply health information from electronic sources [3]. However, this definition reflects a time when the primary health application of technology for the individual was the use of the internet to access health information. It does not adequately capture the current state of technology or the growth of the digital health space. In today’s increasingly digital world, individuals have access to a wide array of digital health technologies; these include online platforms that support synchronous telehealth visits and asynchronous communication with health teams, and smartphone applications and sensors that support remote monitoring, facilitate treatment interventions, and assist in self-management. In this changing world, digital literacy plays an increasingly important role in accessing health services and is recognized as a social determinant of health [4]. A new definition of digital health literacy that encompasses the ability to generate health information (e.g., by completing patient-reported outcome measures digitally) [5], capture health data (e.g., using activity trackers to monitor physical activity), exchange health information (e.g., through telehealth visits) [6], and administer and adjust treatment (e.g., by changing settings for portable medication pumps) is needed to reflect our changing environment.
Digital health technologies are increasingly used in Parkinson’s disease (PD) and have the potential to improve care and expand access to care. Yet, little is known about digital health literacy in PD, where motor and cognitive impairment might be expected to present additional challenges. In this review, we discuss what is known about digital health literacy in PD, review the potential challenge of low digital health literacy in PD with particular attention to telehealth, deep brain stimulation, wearable sensors, and smartphone applications, highlight inequities in access to digital health technologies, and explore future directions.
DIGITAL HEALTH TECHNOLOGY USE AND LITERACY AMONG OLDER ADULTS
In the United States, 93% of adults use the internet and 77% of adults have home broadband internet service [7]. However, access to home broadband varies widely by demographics—only 71% of Black adults, 65% of Hispanic adults, 64% of older adults, 57% of adults from low-income households, and 46% of adults with less than a high school education have home broadband. Technology adoption is lower among low income versus high income households; 23% of adults from low income households report having broadband service, a smartphone, a computer and a tablet, compared to 63% of adults from high income households [8]. Differences in access to technology influence the accessibility of healthcare. For example, 63% of Black adults compared to 49% of white adults agree that not having high-speed internet produces a disadvantage in accessing healthcare providers [9].
Among older adults, surveys suggest that low digital health literacy is commonplace. In a recent survey of 200 predominantly low-income, community-dwelling older adults, 73% had low digital health literacy [10]. In a survey of nearly 300 hospitalized adults (median age 58 years old), 34% had low digital health literacy [11]. Both studies used the eHealth Literacy Scale (eHEALS), which only assesses an individual’s perception of their ability to obtain, critically appraise, and apply health information from the internet [12], to assess digital health literacy and therefore likely underestimate the prevalence of low digital health literacy. Adequate digital health literacy is associated with higher levels of education, younger age, digital device ownership, and home internet access [11, 13].
No studies have directly assessed digital health literacy in PD. Results from studies that assessed health literacy, use of technology, and comfort with technology provide some insights. Among a sample of individuals with PD without dementia, 30% had low health literacy.[14] In a small survey-based study, 21% of individuals with PD rated their technology skills as absent or poor.[15] In a qualitative study, individuals with PD and their care partners identified lack of adequate digital skills as a barrier to use of digital health resources [16]. Individuals with PD from underrepresented groups view digital literacy as a barrier to using digital health for their disease management [16]. In another survey-based study of individuals with PD, usage of a smartphone decreased with advancing age and was influenced by motor impairment [17].
DIGITAL HEALTH APPLICATIONS IN PARKINSON’S DISEASE
Telehealth
Telehealth, which encompasses the use of real-time audio-visual technology to examine, monitor, and provide medical care, is well-suited for PD given the predominantly visual nature of the exam as well as the progressive disability experienced by individuals with PD that may impede travel to in-person appointments. Research has demonstrated the short- and long-term feasibility [18–21], patient/provider satisfaction [22–24], cost savings [18, 25, 26], and validity [27] of telehealth visits in PD. Successful management of PD requires multidisciplinary care, and prior research has demonstrated the ability to deliver rehabilitative therapy services and other treatments (Fig. 1). For example, virtual delivery of the Lee Silverman Voice Treatment (LSVT) has proven effective [28], being noninferior to in-person therapy [29]. Successful remote physical therapy assessments and exercise programs have been reported [30, 31], including a 7-week virtual therapy program that resulted in a significant improvement in physical performance [31]. A randomized controlled trial of telephone-based cognitive behavior therapy for depression in PD resulted in a significant reduction in depressive symptoms compared to community-based in-person treatment [32]. With an expansion in services available through telehealth, it is increasingly important that individuals with PD possess the necessary digital health literacy to access these essential services.
Fig. 1
Uses of digital health technologies in Parkinson’s disease care.
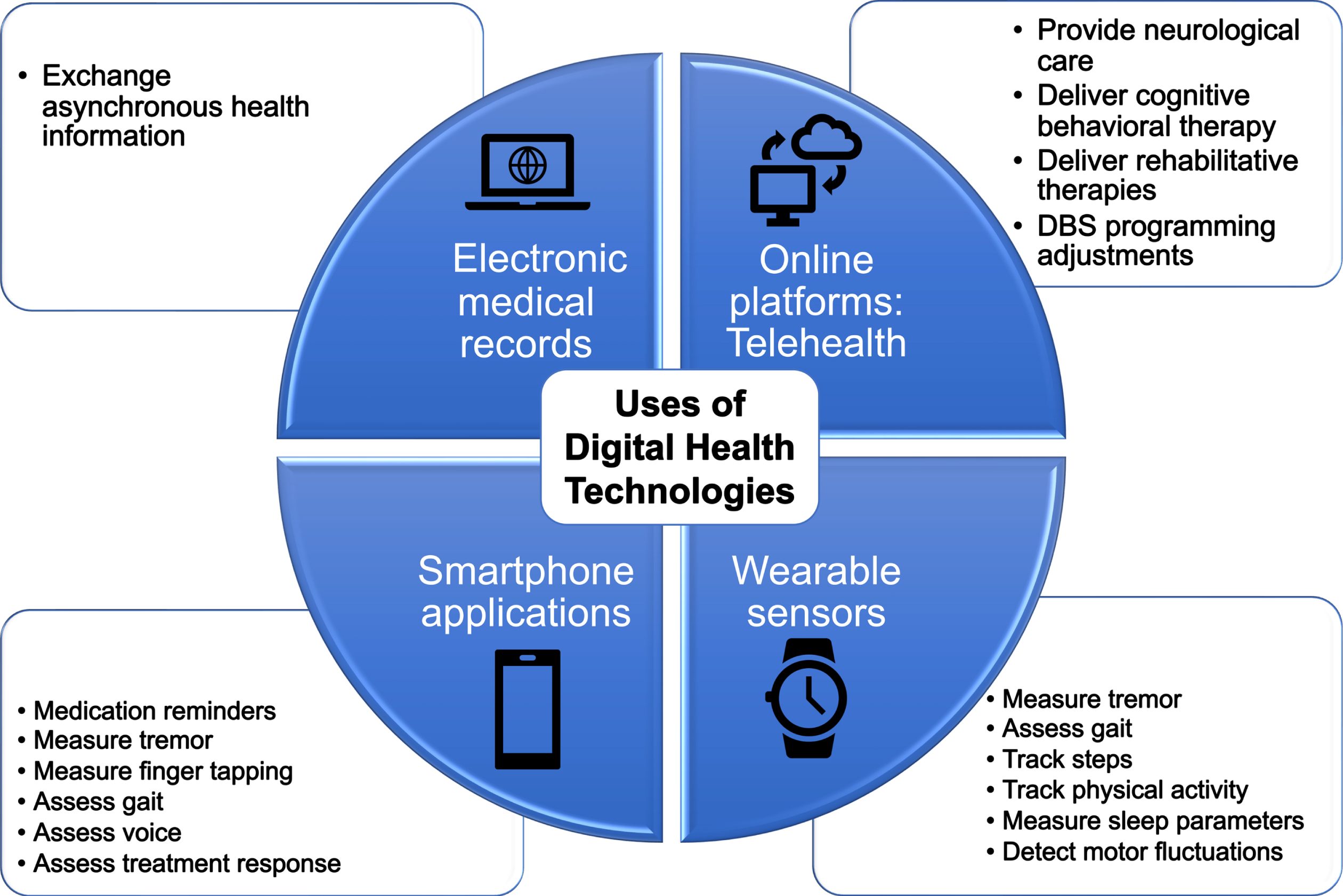
The COVID-19 pandemic led to a significant surge in telehealth visits for PD in early 2020. The proportion of individuals with PD participating in telehealth rose from 10% before the pandemic to 64% during its initial stages [33]. A global survey sponsored by the International Parkinson and Movement Disorder Society Telemedicine Group in 2020 confirmed the widespread increase in telehealth utilization [34]. However, despite its rapid expansion, a survey revealed that telehealth usage was highest among individuals with PD who had higher incomes, higher levels of education, and who were white [33]. An academic movement center observed that Black individuals were less likely to engage in telehealth compared to white individuals [22].
Despite the substantial growth and potential advantages of telehealth in PD care, there are still notable challenges that must be addressed. A cohort study involving 258 individuals with PD who completed follow-up telehealth visits found that only approximately 30% of participants agreed that video visits were “better overall,” and even fewer felt they received “better care” [20]. Although telehealth visits became necessary during the pandemic, not all patients expressed a desire to continue with such visits post-pandemic; one study reported that only 26–48% of individuals with PD planned to continue with telehealth visits [35]. In a single-center study of over 60,000 telehealth visits conducted across various disciplines during the COVID-19 pandemic, visits with poor connectivity, extended wait times, or difficulties being seen, examined, or understood by the provider were associated with lower patient satisfaction [36]. Low digital health literacy may be one important driver of dissatisfaction with telehealth visits.
While telehealth visits have ebbed as the pandemic has receded, they are here to stay. We must consider the challenges posed by telehealth visits and ensure that individuals with PD have the necessary level of digital health literacy to make proper use telehealth visits when appropriate. Individuals must possess several different technological skills to successfully complete a telehealth visit and exchange health information. For example, individuals with PD must successfully navigate one of a myriad of videoconferencing platforms [37], troubleshoot any technological problems that arise, successfully adjust their camera to facilitate examination, and navigate an online patient portal system in order to obtain a visit summary. And the process is likely to differ from healthcare system-to-healthcare system and even provider-to-provider, placing an even higher burden on individuals with PD. Concerns persist regarding limited digital literacy among individuals with PD [34, 38]. Robust educational outreach efforts and technical resources are needed to improve digital health illiteracy, foster engagement and participation in the exciting growing field of telehealth, and improve clinical outcomes in PD care.
Remote programming in deep brain stimulation
Deep brain stimulation (DBS) therapy is a well-established surgical treatment for select individuals with PD. The consultation process for DBS typically involves multi-disciplinary screening evaluations to identify optimal candidates for improved outcomes, along with extensive patient education. Patient education materials for DBS are often accessed through internet-based resources, which may be challenging for those with lower digital health literacy. Additionally, available patient education materials for DBS are often written above the recommended sixth-grade educational level set by the National Institutes of Health and American Medical Association [39–41]. This is compounded by patients often lacking understanding of basic brain structure and function, which further hinders their ability to make informed decisions about their health. In a recent randomized controlled study, the use of a 3D printed brain model during patient education for DBS surgery consultation was shown to improve patient confidence and understanding of the therapy compared to patient education alone, thus enhancing health literacy [39].
The efficacy of DBS treatment is highly dependent not only on the selection of ideal candidates but on proper programming [42] and telehealth can play a role in both. Remote evaluation for deep brain stimulation candidacy has been shown to be both feasible and economical [43]. Asynchronous and synchronous remote programming are available for DBS management. With asynchronous programming, a schedule of programming adjustments may be prescribed and communicated through electronic medical record (EMR) messages. In addition, patient programmer settings can be set to allow for pre-determined adjustments, which can be implemented by the individual or an experienced caregiver under the guidance of a DBS specialist during a video-based telehealth session.
Synchronous remote programming enables real-time adjustments during telehealth visits. Remote DBS programming has been available in China since 2017 with two manufacturers, PINS Medical and SceneRay Corporation, Ltd., and more recently in the US with the introduction of Abbott’s Neurospheretrademark Virtual Clinic in 2021. Research has demonstrated that remote DBS programming is safe and effective [44], and saves significant travel time and reduces costs [45–47]. A recent survey reported patient satisfaction with the Abbott DBS programming interface, and the majority of respondents felt that the visits were comparable to in-person visits [48]. Remote DBS programming may be particularly helpful in certain cases, such as when frequent adjustments are required or to address stimulation side effects that were not seen during the programming session [49]. Despite these exciting advances, not all patients are comfortable with this technology or possess the digital health literacy to properly adjust treatment, and may have cognitive, motor, or psychiatric impairment which can pose further challenges for remote DBS care. To provide equitable access to such advanced technology, we must ensure that individuals with PD are able to appropriately evaluate the pros and cons of such technology and navigate the programmers.
In June 2020, Medtronic introduced the first brain signal sensing DBS system, Percept PCtrademark. The Percepttrademark system utilizes Brainsensetrademark technology, which enables sensing and recording of local field potentials through the contacts adjacent to the active stimulation contact [50]. While this device is not capable of real-time remote programming as previously described, it can be configured so patients can track different “events.” This information can be reviewed by the clinician at a follow-up visit and potentially provide guidance for a more optimal programming configuration. BrainSensetrademark technology is a preliminary step towards developing a commercialized system that can deliver adaptive (closed-loop) stimulation, which may eventually enable the DBS system to modify settings without the need for a clinical intervention [49]. Ultimately, this may be a more viable option for individuals with PD with limited digital health literacy, providing even more opportunities for this subgroup.
A disparity in access to DBS already exists and as DBS technology advances, we must prioritize inclusivity. According to one study, white individuals with PD are five times more likely than Black individuals to undergo DBS [51]. Asian individuals, Black individuals, women and individuals from a socioeconomically marginalized group are less likely to undergo DBS [52]. In a recent study of remote DBS programming, participants were required to be fluent in English and 91% of those assigned to remote DBS programming were white [53]. To achieve health equity, future studies should show a commitment to be inclusive of individuals from underserved groups, non-English speakers, and those with less access to technology.
Wearables and smartphone applications
Sensor-based digital health technologies, such as smartphones and wearables, are increasingly being recognized as means to provide comprehensive, objective, real-world assessment of individuals with PD and improve care. Such technology is already readily available. Approximately 85% of U.S. adults own a smartphone [54], and 20% wear a smart watch or fitness tracker [55]. Research studies suggest that digital health technologies can be used to measure specific PD motor constructs [56], assess gait [57–59], track physical activity [60], measure sleep parameters [61], monitor response to medications [62, 63], detect motor fluctuations [62, 63], and predict meaningful events [64]. While for the most part this research has not yet translated to clinical practice, there are many commercially available fitness trackers and PD-specific digital products [65, 66], and the time is quickly approaching. Individuals with PD are beginning to expect a certain amount of familiarity with these approaches on the part of their healthcare team. In turn, healthcare teams are beginning to consider use of these digital health technologies to inform clinical management.
The use of sensor-based digital health technologies to capture personal health data requires an understanding of how to select the appropriate device for the intended use, the ability to perform a range of different digital skills (e.g., proper wear, charging, completion of health assessments, uploading and transmission of health data), and an understanding of how to interpret the data. While no studies have specifically examined digital health literacy in PD, much less with specific emphasis on sensor-based digital health technologies, usability studies that examine the ease of use of digital health technologies may inform our understanding. One study examined the usability of a digital health system (smartwatch, smartphone and wearable inertial monitoring unit) among individuals with PD over a 7-day monitoring period and found a high median usability score that inversely correlated with age [67]. In a separate qualitative study, individuals with PD identified wearable device design (e.g., need for battery replacement) as a potential barrier to use [68]. In another study, usability of a camera system was influenced by age and motor impairment while the usability of a tablet-based application was influenced by cognitive status and digital health literacy [69]. These results need to be interpreted with caution as individuals who participate in such studies are a self-selected group likely to have more familiarity with digital health technologies than the general PD population. However, such work highlights that digital solutions need to be tailored to the specific population and that successful use of digital health technologies may be dependent on age and/or disease stage.
FUTURE DIRECTIONS
The growth in digital health technologies has changed the face of healthcare. It is easy to imagine a world, in the not-too-distant future, where healthcare services are more easily accessible online than in-person, wearable and ambient sensors are routinely used to monitor health and adjust treatment remotely, autonomous optimization of DBS settings is the standard, and digital health technologies are routinely used to deliver treatments for a range of motor and non-motor symptoms. To prepare for such a world, we must invest in digital health literacy.
Future research should determine the prevalence of digital health literacy among individuals with PD. It is important to note that digital health literacy is not a fixed trait and can be improved over time. Accurately identifying individuals with low digital health literacy is a critical step in determining how to allocate targeted resources and implementing effective interventions. Routine screening for digital health literacy during healthcare visits may be instrumental in identifying those in need of support. Integrating digital health literacy into social determinants of health documentation within EMR systems can facilitate this process.
Evidence-based strategies for enhancing digital health literacy need to be developed. One potential intervention to improve digital health literacy is the implementation of educational programs and technology assistance initiatives (Table 1), which should be made available to patients and their caregivers at little to no cost [4]. We must devote appropriate healthcare system resources towards the improved education of individuals with PD and their caregivers on the use of EMR systems, telehealth, remote DBS programming, wearable devices, and other sensors to optimize their health [53]. Digital health technology companies also have a role to play; they must ensure the usability of their products and appropriateness for individuals with PD.
Table 1
Digital health literacy existing challenges and potential solutions
| Existing challenges | Potential solutions |
| Lack of access to high-speed broadband internet connection | •Advocate for expansion of affordable high-speed broadband/wireless services |
| Difficulty with independent Patient Portal login, access, and use | •Programs and aids to educate and assist patients with navigation of electronic medical record technologies |
| Obstacles to initiate and conduct a telehealth visit | •Accessible training materials in a variety of formats (e.g., written web-based instructions, video demonstrations) |
| •Clinic-based telehealth coordinator assistance to prepare patients prior to visit and assist during visit | |
| Barriers to independently set-up and navigate remote DBS programmer for adjustments and/or event tracking | •Real-time technical support |
| •Accessible training materials in a variety of formats (e.g., written web-based instructions, video demonstrations) | |
| Challenges with wearable sensor selection, use, and data interpretation | •Advocate for affordable access to digital health technologies |
| •Clinic-based guidance on the appropriate selection of sensors | |
| •Provide guidance on how to evaluate the adequacy of data privacy and protections | |
| Difficulty with smartphone navigation and application use | •Partner with community organizations and schools on digital health literacy programs |
| •Conduct in-person classes with student volunteers | |
| Lack of access to materials and support in languages other than English | •Ensure availability of educational resources in languages other than English |
While technology may improve the care that many individuals with PD receive, it threatens to amplify existing health disparities. The digital divide is the gap between individuals who have access to digital technologies and the necessary digital literacy to use them and those who do not [70]. As noted above, the digital divide primarily affects individuals with PD from underserved groups and interventions should be appropriately targeted. To dismantle these health disparities, we need to provide equitable access to technology and commit additional resources towards improving the digital health literacy of underserved individuals and their families. One important initiative is to provide patients with health services in the language they prefer. For example, Spanish language clinics could improve Hispanic patient understanding and adherence to digital health technology. Strong partnerships with community organizations will also be needed to improve digital health literacy. Policy changes are also necessary to help dismantle disparities in care. For example, we should advocate for affordable, home broadband access for all and for policies that ensure equal and fair reimbursement of telehealth services [71]. Lastly, research studies that evaluate digital health solutions and strategies for improving digital health literacy, must prioritize the inclusion of underserved populations.
Telehealth, wearable sensors, smartphone applications, and other digital health technologies offer transformative potential in healthcare. We must invest not only in the development of new technologies but the assessment, understanding, and improvement of digital health literacy. Without this investment, we will not be able to capitalize on the potential of digital health technologies and stand to worsen existing healthcare disparities.
ACKNOWLEDGMENTS
The authors have no acknowledgments to report.
FUNDING
The authors have no funding to report.
CONFLICT OF INTEREST
Dr. Schneider receives research support from the National Institutes of Health, the Michael J. Fox Foundation for Parkinson’s Research, Biohaven Pharmaceuticals, Bial, and Acadia Pharmaceuticals. She has served on an advisory board for Sun Pharma Advanced Research Company and receives financial support from Clintrex Research Corporation for serving on a Data and Safety Monitoring Board.
Dr. Valdovinos has no conflicts of interest.
Dr. Esper receives research support from the Centers for Disease Control and Prevention and royalties from UpToDate. She has received consultancy fees from NeuroOne Medical Technologies Corporation.
REFERENCES
[1] | Centers for Disease Control and Prevention, What is health literacy? Take action. Find out. https://www.cdc.gov/healthliteracy/learn/index.html, Accessed on March 31, 2023. |
[2] | Berkman ND , Sheridan SL , Donahue KE , Halpern DJ , Crotty K ((2011) ) Low health literacy and health outcomes: An updated systematic review, Ann Intern Med 155: , 97–107. |
[3] | Norman CD , Skinner HA ((2006) ) eHealth literacy: Essential skills for consumer health in a networked world, J Med Internet Res 8: , e9. |
[4] | Campanozzi LL , Gibelli F , Bailo P , Nittari G , Sirignano A , Ricci G ((2023) ) The role of digital literacy in achieving health equity in the third millennium society: A literature review. Front Public Health 11: , 1109323. |
[5] | Griebel L , Enwald H , Gilstad H , Pohl A-L , Moreland J , Sedlmayr M ((2018) ) eHealth literacy research—Quo vadis? Inform Health Soc Care 43: , 427–442. |
[6] | Paige SR , Stellefson M , Krieger JL , Anderson-Lewis C , Cheong J , Stopka C ((2018) ) Proposing a transactional model of eHealth literacy: Concept analysis, J Med Internet Res 20: , e10175. |
[7] | Pew Research Center, Internet/Broadband Fact Sheet. https://www.pewresearch.org/internet/fact-sheet/internetbroadband/, Accessed on July 9, 2023. |
[8] | Vogels E (2021) Digital divide persists even as Americans with lower incomes make gains in tech adoption. Pew Research Center, https://www.pewresearch.org/shortreads/2021/06/22/digital-divide-persists-even-as-americans-with-lower-incomes-make-gains-in-tech-adoption/, Accessed on July 9, 2023. |
[9] | Atske S , Perrin A (2021) Home broadband adoption, computer ownership vary by race, ethnicity in the U.S. Pew Research Center, https://www.pewresearch.org/shortreads/2021/07/16/home-broadband-adoption-computerownership-vary-by-race-ethnicity-in-the-u-s/, Accessed on July 9, 2023. |
[10] | Arcury TA , Sandberg JC , Melius KP , Quandt SA , Leng X , Latulipe C , Miller DP , Smith DA , Bertoni AG ((2020) ) Older adult internet use and eHealth literacy. J Appl Gerontol 39: , 141–150. |
[11] | Cheng J , Arora VM , Kappel N , Vollbrecht H , Meltzer DO , Press V ((2023) ) Assessing disparities in video-telehealth use and eHealth literacy among hospitalized patients: Cross-sectional observational study, JMIR Form Res 7: , e44501. |
[12] | Norman CD , Skinner HA ((2006) ) eHEALS: The eHealth Literacy Scale, J Med Internet Res 8: , e27. |
[13] | Liu S , Zhao H , Fu J , Kong D , Zhong Z , Hong Y , Tan J , Luo Y ((2022) ) Current status and influencing factors of digital health literacy among community-dwelling older adults in Southwest China: A cross-sectional study. BMC Public Health 22: , 996. |
[14] | Fleisher JE , Shah K , Fitts W , Dahodwala NA ((2016) ) Associations and implications of low health literacy in Parkinson’s disease. Mov Disord Clin Pract 3: , 250–256. |
[15] | Kenny L , Moore K , O’Riordan C , Fox S , Barton J , Tedesco S , Sica M , Crowe C , Alamäki A , Condell J , Nordström A , Timmons S ((2022) ) The views and needs of people with Parkinson disease regarding wearable devices for disease monitoring: Mixed methods exploration, JMIR Form Res 6: , e27418. |
[16] | Nimmons D , Armstrong M , Pigott J , Walters K , Schrag A , Ogunleye D , Dowridge W , Read J , Davies N ((2022) ) Exploring the experiences of people and family carers from under-represented groups in self-managing Parkinson’s disease and their use of digital health to do this. Digit Health 8: , 20552076221102261. |
[17] | Marxreiter F , Buttler U , Gassner H , Gandor F , Gladow T , Eskofier B , Winkler J , Ebersbach G , Klucken J ((2020) ) The use of digital technology and media in German Parkinson’s disease patients. J Parkinsons Dis 10: , 717–727. |
[18] | Beck CA , Beran DB , Biglan KM , Boyd CM , Dorsey ER , Schmidt PN , Simone R , Willis AW , Galifianakis NB , Katz M , Tanner CM , Dodenhoff K , Aldred J , Carter J , Fraser A , Jimenez-Shahed J , Hunter C , Spindler M , Reichwein S , Mari Z , Dunlop B , Morgan JC , McLane D , Hickey P , Gauger L , Richard IH , Mejia NI , Bwala G , Nance M , Shih LC , Singer C , Vargas-Parra S , Zadikoff C , Okon N , Feigin A , Ayan J , Vaughan C , Pahwa R , Dhall R , Hassan A , DeMello S , Riggare SS , Wicks P , Achey MA , Elson MJ , Goldenthal S , Keenan HT , Korn R , Schwarz H , Sharma S , Stevenson EA , Zhu W Connect.Parkinson Investigators ((2017) ) National randomized controlled trial of virtual house calls for Parkinson disease. Neurology 89: , 1152–1161. |
[19] | Dorsey ER , Venkataraman V , Grana MJ , Bull MT , George BP , Boyd CM , Beck CA , Rajan B , Seidmann A , Biglan KM ((2013) ) Randomized controlled clinical trial of “virtual house calls” for Parkinson disease. JAMA Neurol 70: , 565–570. |
[20] | Korn RE , Wagle Shukla A , Katz M , Keenan HT , Goldenthal S , Auinger P , Zhu W , Dodge M , Rizer K , Achey MA , Byrd E , Barbano R , Richard I , Andrzejewski KL , Schwarz HB , Dorsey ER , Biglan KM , Kang G , Kanchana S , Rodriguez R , Tanner CM , Galifianakis NB ((2017) ) Virtual visits for Parkinson disease: A multicenter noncontrolled cohort. Neurol Clin Pract 7: , 283–295. |
[21] | Samii A , Ryan-Dykes P , Tsukuda RA , Zink C , Franks R , Nichol WP ((2006) ) Telemedicine for delivery of health care in Parkinson’s disease. J Telemed Telecare 12: , 16–18. |
[22] | Esper CD , Scorr L , Papazian S , Bartholomew D , Esper GJ , Factor SA ((2021) ) Telemedicine in an academic movement disorders center during COVID-19. J Mov Disord 14: , 119–125. |
[23] | Qiang JK , Marras C ((2015) ) Telemedicine in Parkinson’s disease: A patient perspective at a tertiary care centre. Parkinsonism Relat Disord 21: , 525–528. |
[24] | Stillerova T , Liddle J , Gustafsson L , Lamont R , Silburn P ((2016) ) Remotely assessing symptoms of Parkinson’s disease using videoconferencing: A feasibility study. Neurol Res Int 2016: , 4802570. |
[25] | Xu Y , Feeney MP , Surface M , Novak D , Troche MS , Beck JC , Alcalay RN ((2022) ) Attitudes toward telehealth services among people living with Parkinson’s disease: A survey study. Mov Disord 37: , 1289–1294. |
[26] | Podlewska AM , van Wamelen DJ ((2022) ) Parkinson’s disease and Covid-19: The effect and use of telemedicine. Int Rev Neurobiol 165: , 263–281. |
[27] | Schneider RB , Myers TL , Tarolli CG , Amodeo K , Adams JL , Jensen-Roberts S , Dorsey ER ((2020) ) Remote administration of the MDS-UPDRS in the time of COVID-19 and beyond. J Parkinsons Dis 10: , 1379–1382. |
[28] | Griffin M , Bentley J , Shanks J , Wood C ((2018) ) The effectiveness of Lee Silverman Voice Treatment therapy issued interactively through an iPad device: A non-inferiority study. J Telemed Telecare 24: , 209–215. |
[29] | Constantinescu G , Theodoros D , Russell T , Ward E , Wilson S , Wootton R ((2011) ) Treating disordered speech and voice in Parkinson’s disease online: A randomized controlled non-inferiority trial. Int J Lang Commun Disord 46: , 1–16. |
[30] | Quinn L , Macpherson C , Long K , Shah H ((2020) ) Promoting physical activity via telehealth in people with Parkinson disease: The path forward after the COVID-19 pandemic? Phys Ther 100: , 1730–1736. |
[31] | Gandolfi M , Geroin C , Dimitrova E , Boldrini P , Waldner A , Bonadiman S , Picelli A , Regazzo S , Stirbu E , Primon D , Bosello C , Gravina AR , Peron L , Trevisan M , Garcia AC , Menel A , Bloccari L , Valè N , Saltuari L , Tinazzi M , Smania N ((2017) ) Virtual reality telerehabilitation for postural instability in Parkinson’s disease: A multicenter, single-blind, randomized, controlled trial. BioMed Res Int 2017: , 796282. |
[32] | Dobkin RD , Mann SL , Gara MA , Interian A , Rodriguez KM , Menza M ((2020) ) Telephone-based cognitive behavioral therapy for depression in Parkinson disease: A randomized controlled trial, Neurology 94: , e1764–e1773. |
[33] | Feeney MP , Xu Y , Surface M , Shah H , Vanegas-Arroyave N , Chan AK , Delaney E , Przedborski S , Beck JC , Alcalay RN ((2021) ) The impact of COVID-19 and social distancing on people with Parkinson’s disease: A survey study. NPJ Parkinsons Dis 7: , 10. |
[34] | Hassan A , Mari Z , Gatto EM , Cardozo A , Youn J , Okubadejo N , Bajwa JA , Shalash A , Fujioka S , Aldaajani Z , Cubo E the International Telemedicine Study Group Adams J , Afshari M , Aldaajani Z , Aldred J , Bajwa JA , Ben-Pazi H , Bloem B , Browne P , Buesing K , Cardozo A , Chan B , Chouinard S , Cubo E , Doumbe J , Dekker M , Fujioka S , Galifianakis N , Gatto E , Goetz C , Guttman M , Hassan A , Hatcher-Martin J , Katz M , Mari Z , Moukheiber E , Ojo O , Okubadejo N , Pantelyat A , Prakash N , Shalash A , Spindler M , Tanner C , Youn J ((2020) ) Global survey on telemedicine utilization for movement disorders during the COVID -19 pandemic. Mov Disord 35: , 1701–1711. |
[35] | de Rus Jacquet A , Bogard S , Normandeau CP , Degroot C , Postuma RB , Dupre N , Miyasaki JM , Monchi O , Martino D , Fon EA , Cicchetti F ((2021) ) Clinical perception and management of Parkinson’s disease during the COVID-19 pandemic: A Canadian experience. Parkinsonism Relat Disord 91: , 66–76. |
[36] | Rodrigues A , Yu JS , Bhambhvani H , Uppstrom T , Ricci WM , Dines JS , Hayden-Gephart M ((2022) ) Patient experience and satisfaction with telemedicine during coronavirus disease 2019: A multi-institution experience. Telemed E-Health 28: , 150–157. |
[37] | Cubo E , Arnaiz-Rodriguez A , Arnaiz-Gonzalez Á , Díez-Pastor JF , Spindler M , Cardozo A , Garcia-Bustillo A , Mari Z , Bloem BR ((2021) ) Videoconferencing software options for telemedicine: A review for movement disorder neurologists. Front Neurol 12: , 745917. |
[38] | Shalash A , Spindler M , Cubo E ((2021) ) Global perspective on telemedicine for Parkinson’s disease, J Parkinsons Dis 11: , S11–S18. |
[39] | Hirt L , Kern DS , Ojemann S , Grassia F , Kramer D , Thompson JA ((2022) ) Use of three-dimensional printed brain models during deep brain stimulation surgery consultation for patient health literacy: A randomized controlled investigation, World Neurosurg 162: , e526–e533. |
[40] | Shlobin NA , Rosenow JM ((2022) ) Ethical considerations in the implantation of neuromodulatory devices. Neuromodulation 25: , 222–231. |
[41] | Walsh TM , Volsko TA ((2008) ) Readability assessment of internet-based consumer health information. Respir Care 53: , 1310–1315. |
[42] | Sharma VD , Patel M , Miocinovic S ((2020) ) Surgical treatment of Parkinson’s disease: Devices and lesion approaches. Neurotherapeutics 17: , 1525–1538. |
[43] | Witek N , Heath SL , Ouyang B , Tanner CM , Galifianakis NB ((2020) ) Remote telemedicine evaluation of deep brain stimulation candidacy: Retrospective cohort analysis. Neurol Clin Pract 10: , 199–205. |
[44] | Li D , Zhang C , Gault J , Wang W , Liu J , Shao M , Zhao Y , Zeljic K , Gao G , Sun B ((2017) ) Remotely programmed deep brain stimulation of the bilateral subthalamic nucleus for the treatment of primary Parkinson disease: A randomized controlled trial investigating the safety and efficacy of a novel deep brain stimulation system. Stereotact Funct Neurosurg 95: , 174–182. |
[45] | Xu J , Wang J , Keith S , Zhang M , Yang C , Yuan Q , Qiu Y , Hu X , Wu X ((2021) ) Management of Parkinson’s disease patients after DBS by remote programming: Preliminary application of single center during quarantine of 2019-nCoV. J Neurol 268: , 1295–1303. |
[46] | Ma Y , Miao S , Zhou R , Zhang Q , Chen H , Liang Y ((2021) ) Application of remote deep brain stimulation programming for Parkinson’s disease patients, World Neurosurg 147: , e255–e261. |
[47] | Pinter D , Jardahazi E , Janszky J , Kovacs N ((2022) ) Potential clinical and economic benefits of remote deep brain stimulation programming. Sci Rep 12: , 17420. |
[48] | Esper CD , Merola A , Himes L , Patel N , Bezchlibnyk YB , Falconer D , Weiss D , Luca C , Cheeran B , Mari Z ((2022) ) Necessity and feasibility of remote tele-programming of deep brain stimulation systems in Parkinson’s disease. Parkinsonism Relat Disord 96: , 38–42. |
[49] | Sharma VD , Safarpour D , Mehta SH , Vanegas-Arroyave N , Weiss D , Cooney JW , Mari Z , Fasano A ((2021) ) Telemedicine and deep brain stimulation - Current practices and recommendations. Parkinsonism Relat Disord 89: , 199–205. |
[50] | Jimenez-Shahed J ((2021) ) Device profile of the percept PC deep brain stimulation system for the treatment of Parkinson’s disease and related disorders. Expert Rev Med Devices 18: , 319–332. |
[51] | Cramer SW , Do TH , Palzer EF , Naik A , Rice AL , Novy SG , Hanson JT , Piazza AN , Howard MA , Huling JD , Chen CC , McGovern RA ((2022) ) Persistent racial disparities in deep brain stimulation for Parkinson’s disease. Ann Neurol 92: , 246–254. |
[52] | Willis AW , Schootman M , Kung N , Wang X-Y , Perlmutter JS , Racette BA ((2014) ) Disparities in deep brain stimulation surgery among insured elders with Parkinson disease. Neurology 82: , 163–171. |
[53] | Duffley G , Lutz BJ , Szabo A , Wright A , Hess CW , Ramirez-Zamora A , Zeilman P , Chiu S , Foote KD , Okun MS , Butson CR ((2021) ) Home health management of Parkinson disease deep brain stimulation: A randomized clinical trial. JAMA Neurol 78: , 972–981. |
[54] | Pew Research Center, Mobile Fact Sheet. https://www.pewresearch.org/internet/fact-sheet/mobile/, Accessed on June 12, 2023. |
[55] | Vogels E (2020) About one-in-five Americans use a smart watch or fitness tracker. Pew Research Center, https://www.pewresearch.org/short-reads/2020/01/09/about-one-in-five-americans-use-a-smart-watch-or-fitness-tracker/, Accessed May 15, 2023. |
[56] | Adams JL , Kangarloo T , Tracey B , O’Donnell P , Volfson D , Latzman RD , Zach N , Alexander R , Bergethon P , Cosman J , Anderson D , Best A , Severson J , Kostrzebski MA , Auinger P , Wilmot P , Pohlson Y , Waddell E , Jensen-Roberts S , Gong Y , Kilambi KP , Herrero TR , Ray Dorsey E Parkinson Study Group Watch-PD Study Investigators and Collaborators ((2023) ) Using a smartwatch and smartphone to assess early Parkinson’s disease in the WATCH-PD study. NPJ Parkinsons Dis 9: , 64. |
[57] | Ellis RJ , Ng YS , Zhu S , Tan DM , Anderson B , Schlaug G , Wang Y ((2015) ) A validated smartphone-based assessment of gait and gait variability in Parkinson’s disease, PloS One 10: , e0141694. |
[58] | Hobert MA , Nussbaum S , Heger T , Berg D , Maetzler W , Heinzel S ((2019) ) Progressive gait deficits in Parkinson’s disease: A wearable-based biannual 5-year prospective study. Front Aging Neurosci 11: , 22. |
[59] | Liu Y , Zhang G , Tarolli CG , Hristov R , Jensen-Roberts S , Waddell EM , Myers TL , Pawlik ME , Soto JM , Wilson RM , Yang Y , Nordahl T , Lizarraga KJ , Adams JL , Schneider RB , Kieburtz K , Ellis T , Dorsey ER , Katabi D ((2022) ) Monitoring gait at home with radio waves in Parkinson’s disease: A marker of severity, progression, and medication response, Sci Transl Med 14: , eadc9669. |
[60] | Pradhan S , Kelly VE ((2019) ) Quantifying physical activity in early Parkinson disease using a commercial activity monitor. Parkinsonism Relat Disord 66: , 171–175. |
[61] | Mirelman A , Hillel I , Rochester L , Del Din S , Bloem BR , Avanzino L , Nieuwboer A , Maidan I , Herman T , Thaler A , Gurevich T , Kestenbaum M , Orr-Urtreger A , Brys M , Cedarbaum JM , Giladi N , Hausdorff JM ((2020) ) Tossing and turning in bed: Nocturnal movements in Parkinson’sdDisease. Mov Disord 35: , 959–968. |
[62] | Zhan A , Mohan S , Tarolli C , Schneider RB , Adams JL , Sharma S , Elson MJ , Spear KL , Glidden AM , Little MA , Terzis A , Dorsey ER , Saria S ((2018) ) Using smartphones and machine learning to quantify Parkinson disease severity: The Mobile Parkinson Disease Score. JAMA Neurol 75: , 876. |
[63] | Omberg L , Chaibub Neto E , Perumal TM , Pratap A , Tediarjo A , Adams J , Bloem BR , Bot BM , Elson M , Goldman SM , Kellen MR , Kieburtz K , Klein A , Little MA , Schneider R , Suver C , Tarolli C , Tanner CM , Trister AD , Wilbanks J , Dorsey ER , Mangravite LM ((2022) ) Remote smartphone monitoring of Parkinson’s disease and individual response to therapy. Nat Biotechnol 40: , 480–487. |
[64] | Lo C , Arora S , Baig F , Lawton MA , El Mouden C , Barber TR , Ruffmann C , Klein JC , Brown P , Ben-Shlomo Y , Vos M , Hu MT ((2019) ) Predicting motor, cognitive & functional impairment in Parkinson’s. Ann Clin Transl Neurol 6: , 1498–1509. |
[65] | Great Lakes NeuroTechnologies, https://www.glneurotech.com/, Accessed July 27, 2023. |
[66] | Global Kinetics, Meet PKG. https://pkgcare.com/patients/meet-pkg/, Accessed July 27, 2023. |
[67] | Debelle H , Packer E , Beales E , Bailey HGB , Mc Ardle R , Brown P , Hunter H , Ciravegna F , Ireson N , Evers J , Niessen M , Shi JQ , Yarnall AJ , Rochester L , Alcock L , Del Din S ((2023) ) Feasibility and usability of a digital health technology system to monitor mobility and assess medication adherence in mild-to-moderate Parkinson’s disease. Front Neurol 14: , 1111260. |
[68] | Ozanne A , Johansson D , Hällgren Graneheim U , Malmgren K , Bergquist F , Alt Murphy M ((2018) ) Wearables in epilepsy and Parkinson’s disease-A focus group study. Acta Neurol Scand 137: , 188–194. |
[69] | Bendig J , Spanz A , Leidig J , Frank A , Stahr M , Reichmann H , Loewenbrück KF , Falkenburger BH ((2022) ) Measuring the usability of eHealth solutions for patients with Parkinson disease: Observational study, JMIR Form Res 6: , e39954. |
[70] | Norris P (2001) Digital Divide: Civic Engagement, Information Poverty, and the Internet Worldwide, Cambridge University Press, Cambridge. |
[71] | Dorsey ER , Vlaanderen FP , Engelen LJ , Kieburtz K , Zhu W , Biglan KM , Faber MJ , Bloem BR ((2016) ) Moving Parkinson care to the home. Mov Disord 31: , 1258–1262. |




