Evaluation of Nocturnal Symptoms in Chinese Parkinson’s Disease Patients Based on the PDSS-2 Scale: A Multicenter Cross-Sectional Study
Abstract
Background:
Nocturnal symptoms have a significant effect on the quality of life in Parkinson’s disease (PD) patients.
Objective:
This study aimed to investigate the prevalence and associated factors of nocturnal symptoms in Chinese PD patients.
Methods:
This multicenter cross-sectional study included 1,500 patients with primary PD from 18 centers in China was carried out between February 2019 and February 2020. Questionnaires including Parkinson’s disease sleep scale 2 (PDSS-2), Parkinson’s disease questionnaire 8 (PDQ-8), Beck depression inventory (BDI), and generalized anxiety disorder scale 7 (GAD-7) were used to assess nocturnal symptoms, quality of life, depression, and anxiety.
Results:
Among 1,500 Chinese PD patients, 576 (38.4%) reported nocturnal symptoms. Of them, 59.2% were older than 65 years. The PDQ-8 total score was higher in patients with nocturnal symptoms (p < 0.01). Moderate and severe depression was reported more often in patients with nocturnal symptoms (p < 0.01), and the occurrence and severity of anxiety were higher as well (p < 0.01). Longer disease duration and higher Hoehn-Yahr (HY) stage were independently associated with nocturnal symptoms (p < 0.01). Education level, depression, disease course, HY stage, and nocturnal symptoms were related to the quality of life in Chinese PD patients (p < 0.01).
Conclusion:
Our study found that 38.4% of Chinese PD patients have nocturnal symptoms, even in early and mid-stage PD. Nocturnal symptoms were associated with worse quality of life and higher incidences of depression and anxiety. Nocturnal symptoms should be included in the assessment and care plan, especially in patients with longer disease courses and higher HY stages.
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disease worldwide, which is caused by the progressive degeneration of the central nervous system, affecting motor and non-motor functions and resulting in disability [1]. In 2016, about 6.1 million people were diagnosed with PD, which was 2.4 times higher compared to 1990 [2]. In China, the prevalence rate of PD is 1.7% for people older than 65 years old, similar to that in European and American countries [3]. With the aging of the Chinese population, the number of Chinese PD patients will increase to 5 million in 2030 and might constitute almost half of all PD patients globally [4]. It is suggested that the characteristics of Chinese PD patients are the most representative in the world [4]. Besides, the burden of PD patients is not only associated with rising costs for medication or hospitalization and nursing home care but also a serious decrease in patients’ quality of life [4, 5]. Therefore, it is necessary to strengthen the understanding and management of PD patients in China.
In addition to common symptoms such as resting tremor, rigidity, bradykinesia, posture, and gait disorders, PD patients also suffer from sleep-related symptoms, such as insomnia, rapid eye movement sleep behavior disorder (RBD), excessive daytime sleepiness (EDS), restless legs syndrome (RLS), nighttime akinesia, and difficulties in getting up [6, 7]. The causes of nocturnal symptoms of PD included both the disease itself and the side effects of medications [8]. Nocturnal symptoms significantly affect the quality of life in PD patients [9], but they are often ignored or underestimated in clinical practice [10].
A large questionnaire-based survey in Germany showed that 84.4% of PD patients had a poor quality of life and symptoms, including early morning akinesia (55.4%) and sleep disorders (43.5%), which are common forms of nocturnal symptoms [11]. Studies in the UK [12] and Slovakia [13] demonstrated that 50% and 63% of PD patients showed nocturnal symptoms, respectively. Previous studies in Chinese PD patients [14–17] evaluated the nocturnal symptoms and quality of life and found that most of them had sleep problems. Similar results were also observed in the Japanese [10] population.
Nocturnal symptoms present a challenge for physicians to diagnose as they mainly appear at night and are frequently compensated by fatigue and EDS [9]. Hence, numerous scales have been used to evaluate the sleep-related symptoms of PD, such as the Epworth Sleepiness Scale (ESS) and Parkinson’s disease sleep scale (PDSS) [18–21]. PDSS-2 has been developed recently, which could better cover nocturnal symptoms not included previously (e.g., restless leg syndrome, nocturnal pain, akinesia, and sleep apnea) [22–24]. PDSS-2 score has already been utilized in studies to identify changes in sleep quality in PD patients and proved to be less methodology- and sample-dependent compared to other scores [25, 26].
This study aimed to explore the prevalence and associated factors of nocturnal symptoms in a large group of Chinese PD patients, as well as the relationship between nocturnal symptoms and quality of life. The study might help strengthen our understanding of the prevalence and impact of sleep-related symptoms in PD and perhaps help manage these symptoms.
METHODS
Ethics approval and consent to participate
This study was approved by the ethics committee of each participating center. The subjects were informed in detail of the study and informed consents were signed before participating. Only individuals who gave written consent to participate were included in the study.
Study design and participants
This cross-sectional study included patients diagnosed with primary PD based on the Movement Disorder Society (MDS) clinical diagnostic criteria for PD in 18 healthcare centers in China between February 2019 and February 2020. Patients in the study were from the outpatient clinics, mostly in the early and mid-term stages of PD. The participants also had to be able to describe their symptoms and were willing to cooperate with the investigation. Those who could not complete the questionnaire as required were excluded from the study.
Data collection
All the data was gathered with a questionnaire. The questionnaire collected information on baseline data, nocturnal symptoms, quality of life, depression, and anxiety.
Baseline data included gender, age, education, marital status, employment, comorbid diseases (hypertension, diabetes, dyslipidemia, cerebral infarction, coronary heart disease, chronic obstructive pulmonary disease, sleep apnea syndrome), PD clinical characteristics (disease duration, the first symptom of bradykinesia, tremor, rigidity or mixed type, Hoehn-Yahr stage), PD surgical treatment, and pharmaceutical treatment (current PD treatment, sleep disorders treatment, and psychosomatic treatment, if applicable).
Assessments
The nocturnal symptoms were assessed using PDSS-2 including three main domains: sleep disturbance, nocturnal motor symptoms, and nocturnal PD symptoms. The total score of PDSS-2 ≥15 was considered having nocturnal symptoms [26].
PD patients’ quality of life was evaluated using the eight-item Parkinson’s disease questionnaire (PDQ-8), including mobility, daily life behavior, emotional satisfaction, stigma, social support, cognition, communication, and physical discomfort. Each item is scored 0-4 and the total score of PDQ-8 is 0-32, with higher scores indicating a worse quality of life.
Depression was evaluated using the total score of the Beck depression inventory (BDI, 0–63 points). The degree of depression was classified as no depression (0–13 points), mild depression (14–19 points), moderate depression (20–28 points), and severe depression (29–63 points).
Anxiety was evaluated using the generalized anxiety disorder scale (GAD-7): no symptoms (0–4), mild (5–9), moderate (10–14), and severe (15–21).
Sample size calculation
The sample size of this study was estimated by the PDSS-2 score. The reference data were the results of a nocturnal symptom survey of PD patients in Taiwan [27]. Among the 15 nocturnal symptoms of PD patients in the literature, item 15 (woke up or dyspnea at night caused by snoring) had the lowest prevalence, which was 12.4%; the highest prevalence was item 11 (muscle cramps of upper or lower limbs during night sleep), which was 93.3%. According to the calculation of α= 0.05 and allowable error = 0.15*P, 1,206 and 12 patients were required for surveys on the two nocturnal symptoms, respectively. Simultaneously, the total PDSS-2 score of the patients in the literature is 13.94±6.58. The total scores of nocturnal motor symptoms, PD-related nocturnal symptoms, and sleep disorders were 2.92±2.73, 2.84±2.90, and 8.16±3.56 points, respectively. According to the calculation of α= 0.05 and allowable error = 0.1*mean, the sample sizes required for the above four items were 86, 336, 401, and 73 cases, respectively. Therefore, the number of patients required in this study was 1,206. Considering an estimated effective response rate of approximately 80%, 1,500 participants were planned to be enrolled.
Statistical analysis
The quantitative indicators conforming to the normal distribution were described by mean±SD, and the p-value was calculated by the student t-test. The quantitative indicators that did not conform to the normal distribution were described by median (IQR), and the p-value was calculated by the Mann-Whitney U test. The qualitative index was described by n (%), and the p-value was calculated by the Chi-square test or Fisher’s exact test. Correlation was analyzed using the Pearson or Spearman test (if both variables were in a normal distribution, Pearson was used, otherwise – Spearman). Multivariate logistic regression analysis was used to analyze the related factors of nocturnal symptoms, presenting the odds ratio (OR) and corresponding 95% confidence interval (CI). The following influencing factors were included in the model, including demographics, sociological factors (including age group, gender, education level, employment, and marital status), comorbid diseases (including hypertension, diabetes, dyslipidemia, cerebral infarction, coronary heart disease, chronic obstructive pulmonary disease, sleep apnea syndrome, depression, anxiety, RBD, and RLS), disease duration, first symptom (bradykinesia, tremor, rigidity, and mixed), PD treatment (deep brain stimulation, surgery, pharmaceutical treatment, and no treatment), and Hoehn-Yahr stage.
The linear regression method was used to analyze the influencing factors of patients’ quality of life. The following factors were included in the model, including demographics, sociological factors, comorbid diseases, disease duration, first symptom, PD treatment, Hoehn-Yahr stage, prescription of sleep medicine or psychosomatic treatment, and nocturnal symptoms (PDSS-2 score of ≥15 vs. <15).
In cases when scores of the scales were missing, the mean value was used to fill in. Only the missing data of scores of the scales were filled in; for other variables (e.g., demographic characteristics in Table 1), the numbers of cases with valid data were indicated.
Table 1
Demographic characteristics of Chinese PD patients
| Variable | Total (n = 1,500) | Without nocturnal symptoms (n = 924) | With nocturnal symptoms (n = 576) | p |
| Age (y) (n = 1,494) | 64.69 (9.41) | 63.83 (9.58) | 66.05 (8.97) | <0.01 |
| Age group (n = 1,494) | 0.01 | |||
| 0–44 y | 35 (2.3%) | 28 (3.0%) | 7 (1.2%) | |
| 45–54 y | 180 (12.0%) | 122 (13.3%) | 58 (10.1%) | |
| 55–64 y | 464 (31.1%) | 295 (32.1%) | 169 (29.4%) | |
| ≥65 y | 815 (54.6%) | 475 (51.6%) | 340 (59.2%) | |
| Gender (n = 1,485) | 0.82 | |||
| Male | 783 (52.7%) | 484 (53.0%) | 299 (52.4%) | |
| Education level (n = 1,481) | <0.01 | |||
| ≤5 y | 253 (17.1%) | 133 (14.6%) | 120 (21.1%) | |
| 5–9 y | 552 (37.3%) | 337 (36.9%) | 215 (37.9%) | |
| 10–14 y | 490 (33.1%) | 310 (34.0%) | 180 (31.7%) | |
| ≥15 y | 186 (12.6%) | 133 (14.6%) | 53 (9.3%) | |
| Employment (n = 1,479) | 296 (20.0%) | 200 (21.9%) | 96 (17.0%) | 0.02 |
| Marital status (n = 1,488) | 0.07 | |||
| Unmarried | 12 (0.8%) | 5 (0.5%) | 7 (1.2%) | |
| Married | 1402 (94.2%) | 875 (95.3%) | 527 (92.5%) | |
| Divorced | 9 (0.6%) | 3 (0.3%) | 6 (1.1%) | |
| Widowed | 65 (4.4%) | 35 (3.8%) | 30 (5.3%) | |
| Hypertension (n = 1,462) | 359 (24.6%) | 216 (24.0%) | 143 (25.4%) | 0.55 |
| Diabetes (n = 1,462) | 149 (10.2%) | 88 (9.8%) | 61 (10.8%) | 0.52 |
| Dyslipidemia (n = 1,462) | 120 (8.2%) | 68 (7.6%) | 52 (9.2%) | 0.26 |
| Cerebral infarction (n = 1,462) | 91 (6.2%) | 53 (5.9%) | 38 (6.7%) | 0.51 |
| Coronary heart disease (n = 1,462) | 107 (7.3%) | 58 (6.5%) | 49 (8.7%) | 0.11 |
| Chronic obstructive pulmonary disease (n = 1,462) | 14 (1.0%) | 6 (0.7%) | 8 (1.4%) | 0.15 |
| Sleep apnea syndrome (n = 1,462) | 13 (0.9%) | 6 (0.7%) | 7 (1.2%) | 0.25 |
| Depression (n = 1,462) | 119 (8.1%) | 53 (5.9%) | 66 (11.7%) | <0.01 |
| Anxiety (n = 1,462) | 144 (9.8%) | 76 (8.5%) | 68 (12.1%) | 0.02 |
| RBD (n = 1,462) | 142 (9.7%) | 63 (7.0%) | 79 (14.0%) | <0.01 |
| RLS (n = 14,62) | 56 (3.8%) | 26 (2.9%) | 30 (5.3%) | 0.02 |
PD, Parkinson’s disease; RBD, rapid eye movement sleep behavior disorder; RLS, restless legs syndrome.
Statistical analysis was conducted using SAS V9.4 software. p < 0.05 was considered statistically significant.
RESULTS
Demographic and general characteristics of Chinese PD patients
A total of 1,500 patients were included in the study. As shown in Table 1, 576 (38.4%) PD patients were found to have nocturnal symptoms, of which 59.2% were older than 65 years. There was no statistical difference in sex, marital status, or comorbid diseases/conditions between the two groups (all p > 0.05). However, compared with the patients without nocturnal symptoms, those with nocturnal symptoms had a lower education level (p < 0.01), lower employment rate (p = 0.02), higher depression rate (p < 0.01), higher anxiety rate (p = 0.02), higher RBD rate (p < 0.01), and higher restless leg syndrome rate (p = 0.02).
As shown in Table 2, the patients with nocturnal symptoms had a longer disease duration (6.41±4.86 vs. 4.27±3.79 years, p < 0.01). First symptoms were similar between the two groups, except that a higher proportion of patients with nocturnal symptoms had rigidity (11.5% vs. 7.8%, p = 0.02). The proportion of Hoehn-Yahr stage 1 was lower (15.8% vs. 35.6%), and those of stage 3 (38.0% vs. 23.8%) and stage 4 (10.8% vs. 1.5%) were higher in patients with nocturnal symptoms (p < 0.01). Only 1.6% and 0.4% of patients with and without nocturnal symptoms were on stage 5. Patients having nocturnal symptoms also reported a higher proportion of “wearing off” phenomenon (45.5% vs. 21.6%, p < 0.01).
Table 2
Clinical characteristics of PD patients with/without nocturnal symptoms
| Variable | Total (n = 1,500) | Without nocturnal symptoms (n = 924) | With nocturnal symptoms (n = 576) | p |
| Disease duration (y) (n = 1,496) | 5.09 (4.36) | 4.27 (3.79) | 6.41 (4.86) | <0.01 |
| 0–1 | 188 (12.6%) | 136 (14.8%) | 52 (9.0%) | |
| 1–2 | 259 (17.3%) | 187 (20.3%) | 72 (12.5%) | |
| 2–5 | 440 (29.4%) | 306 (33.2%) | 134 (23.3%) | |
| 5–10 | 400 (26.7%) | 208 (22.6%) | 192 (33.4%) | |
| ≥10 | 209 (14.0%) | 84 (9.1%) | 125 (21.7%) | |
| First symptoms | ||||
| Bradykinesia (n = 1494) | 497 (33.3%) | 297 (32.3%) | 200 (34.8%) | 0.31 |
| Tremor (n = 1494) | 795 (53.2%) | 502 (54.6%) | 293 (51.0%) | 0.18 |
| Rigidity (n = 1494) | 138 (9.2%) | 72 (7.8%) | 66 (11.5%) | 0.02 |
| Mixed (n = 1494) | 153 (10.2%) | 93 (10.1%) | 60 (10.5%) | 0.83 |
| Hoehn-Yahr scale (n = 1,500) | <0.01 | |||
| Stage 1 | 420 (28.0%) | 329 (35.6%) | 91 (15.8%) | |
| Stage 2 | 552 (36.8%) | 357 (38.6%) | 195 (33.9%) | |
| Stage 3 | 439 (29.3%) | 220 (23.8%) | 219 (38.0%) | |
| Stage 4 | 76 (5.1%) | 14 (1.5%) | 62 (10.8%) | |
| Stage 5 | 13 (0.9%) | 4 (0.4%) | 9 (1.6%) | |
| Wearing-off phenomenon (n = 1,498) | 461 (30.8%) | 199 (21.6%) | 262 (45.5%) | <0.01 |
| PD treatment (n = 1,494) | ||||
| Deep brain stimulation | 61 (4.1%) | 32 (3.5%) | 29 (5.1%) | 0.13 |
| Nerve destruction | 7 (0.5%) | 3 (0.3%) | 4 (0.7%) | 0.31 |
| Pharmaceutical treatment | 1,392 (93.2%) | 838 (91.2%) | 554 (96.3%) | 0.02 |
| Anticholinergics | 120 (8.4%) | 82 (9.2%) | 38 (7.0%) | 0.13 |
| Amantadine | 305 (21.4%) | 167 (19.0%) | 138 (25.3%) | <0.01 |
| Levodopa | 1,253 (83.9%) | 725 (78.9%) | 528 (91.8%) | <0.01 |
| Dopamine receptor agonists | 930 (62.5%) | 563 (61.3%) | 367 (64.5%) | 0.22 |
| MAO-B inhibitor | 326 (21.8%) | 209 (22.7%) | 117 (20.3%) | 0.28 |
| COMT inhibitor | 189 (12.6%) | 81 (8.8%) | 108 (18.8%) | <0.01 |
| Triple formulation | 22 (1.5%) | 11 (1.2%) | 11 (1.9%) | 0.26 |
| Other PD drugs | 22 (1.5%) | 11 (1.2%) | 11 (1.9%) | 0.43 |
| No treatment | 86 (5.8%) | 70 (7.6%) | 16 (2.8%) | <0.01 |
| Sleep disorder treatment (n = 1,438) | 101 (7.0%) | 34 (3.8%) | 67 (12.2%) | <0.01 |
| Psychotropic treatment (n = 1,439) | 146 (10.1%) | 71 (8.0%) | 75 (13.5%) | <0.01 |
PD, Parkinson’s disease; MAO-B, monoamine oxidase-B; COMT, catechol-O-methyltransferase.
A significantly higher proportions of patients in the nocturnal symptoms group was receiving pharmaceutical treatment for PD (96.3% vs. 91.2%, p = 0.02), including amantadine (25.3% vs. 19.0%, p < 0.01), levodopa (91.8% vs. 78.9%, p < 0.01), and catechol-o-methyltransferase (COMT) inhibitors (18.8% vs. 8.8%, p < 0.01). The proportions of patients receiving sleep disorder treatment (12.2% vs. 3.8%, p < 0.01) and psychosomatic treatment (13.5% vs. 8.0%, p < 0.01) were significantly higher in the nocturnal symptoms group as well, and that of patients receiving no treatment was lower (2.8% vs. 7.6%, p < 0.01).
The scale score of nocturnal symptoms in Chinese PD patients
As shown in Fig. 1, the average PDSS-2 total score of all patients was 13.68, with a sleep disturbance score of 7.45, a night motor symptoms score of 3.56, and a nocturnal PD symptoms score of 2.67. The most complained item was “get up at night to pass urine” (average score: 2.46), followed by “difficulties in staying asleep” (average score: 1.56) and “bad sleep quality” (average score: 1.43).
Fig. 1
Evaluation of nocturnal symptoms in Chinese PD patients with or without nocturnal symptoms. A) The Parkinson’s disease sleep scale 2 (PDSS-2) score, sleep disturbance score, night motor symptoms score, and nocturnal PD symptoms score; B) the Parkinson’s disease questionnaire 8 (PDQ-8) score; C) the presence and severity of depression; D) the presence and severity of anxiety.
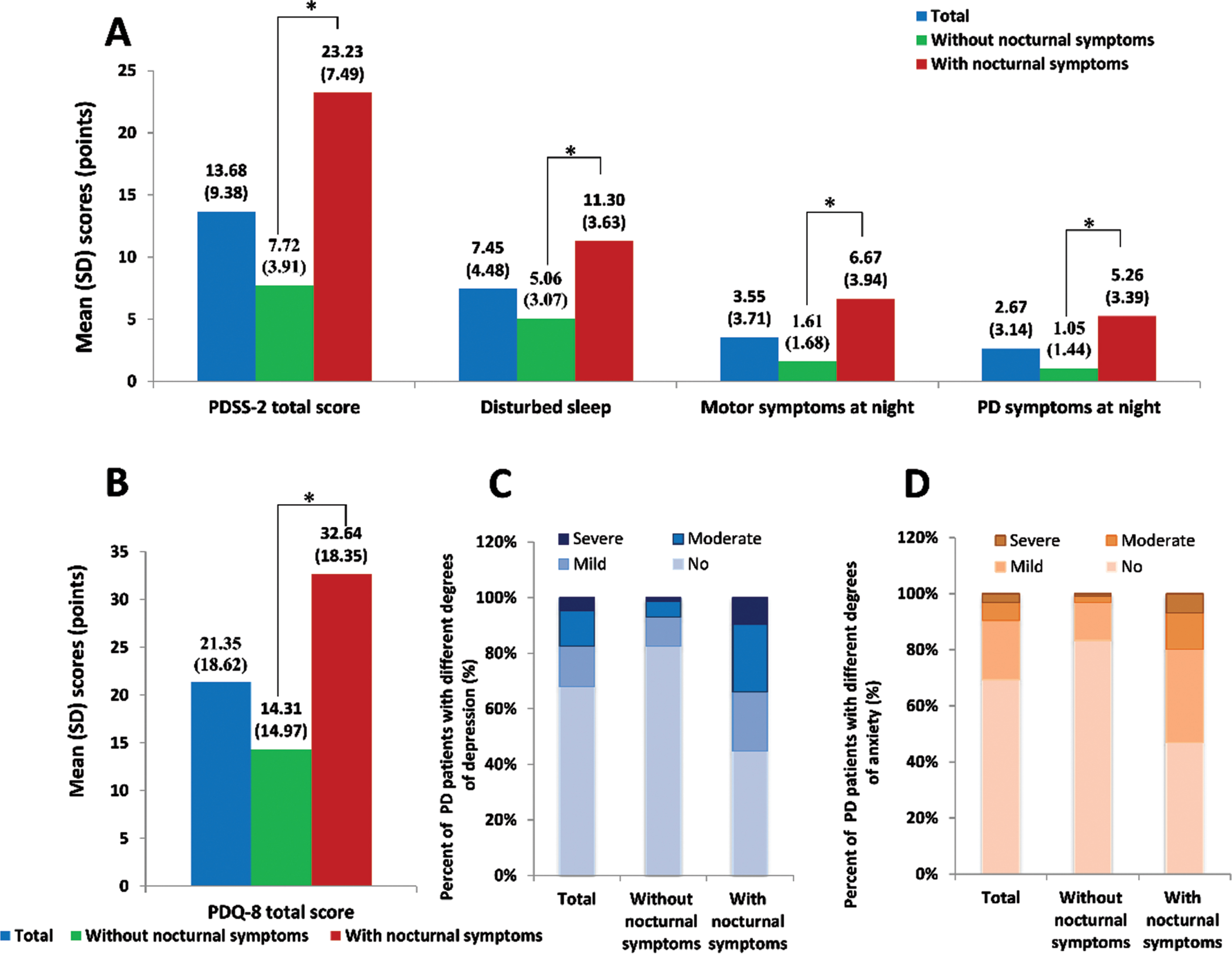
In the nocturnal symptoms group, the PDSS-2 total score was significantly higher than the non-nocturnal symptoms group (23.23±7.49 vs. 7.72±3.91). The scores of sleep disturbance, night movement symptom, and nocturnal PD symptom in both groups were 11.30±3.63 vs. 5.06±3.07, 6.67±3.94 vs. 1.61±1.68, and 5.26±3.39 vs. 1.05±1.44, respectively, indicating that the frequencies of other motor symptoms at night and other PD symptoms at night were approximately five times higher in patients with nocturnal symptoms.
In addition, the PDQ-8 total score was 32.64±18.35 points in patients with nocturnal symptoms compared with 14.31±14.97 in those without nocturnal symptoms (p < 0.01). Moderate and severe depression was reported more often (p < 0.01), and the occurrence and severity of anxiety were higher as well in the former compared to the latter group (p < 0.01).
Correlations between different scores in Chinese PD patients
As demonstrated in Table 3, among Chinese PD patients, the PDSS-2 total score, PDQ-8 total score, BDI total score, and GAD-7 total score were all strongly correlated (p < 0.01). The strongest positive link was found between the PDSS-2 total score and PDQ-8 total score (r = 0.5804, p < 0.01).
Table 3
Spearman correlation between PDSS-2 total score, PDQ-8 total score, BDI total score, and GAD-7 total score
| PDSS-2 total score | PDQ-8 total score | BDI total score | GAD-7 total score | |
| PDSS-2 total score | – | 0.5804 | 0.5241 | 0.4615 |
| PDQ-8 total score | 0.5804 | – | 0.6006 | 0.5837 |
| BDI total score | 0.5241 | 0.6006 | – | 0.6389 |
| GAD-7 total score | 0.4615 | 0.5837 | 0.6389 | – |
The correlation coefficients in the table assume that the p values obtained from the test are less than 0.001. PDSS-2, Parkinson’s disease sleep scale 2; PDQ-8, Parkinson’s disease questionnaire 8; BDI, Beck depression inventory; GAD-7, generalized anxiety disorder scale 7.
Factors related to the development of nocturnal symptoms in Chinese PD patients
As shown in Table 4, education level, depression, RBD, longer disease duration, and higher Hoehn-Yahr stage were related to the development of nocturnal symptoms in patients with PD. Most notably, longer disease duration (≥10 vs. 0–1 years, OR = 2.29; 95% CI: 1.44–3.64; 5–10 vs. 0–1 years, OR = 1.96; 95% CI: 1.30–2.93) and Hoehn-Yahr stage (stage 5 vs. stage 1, OR = 5.63; 95% CI: 1.62–19.56; stage 4 vs. stage 1, OR = 9.89; 95% CI: 5.05–19.38; stage 3 vs. stage 1, OR = 2.58; 95% CI: 1.87–3.55; stage 2 vs. stage 1, OR = 1.63; 95% CI: 1.20–2.21) were independently associated with nocturnal symptoms in PD patients (p < 0.01).
Table 4
Factors associated with nocturnal symptoms in Chinese PD patients
| Variables | OR | 95% CI | |
| Lower confidence bounds | Upper confidence bounds | ||
| Education level | |||
| ≤5 y | 2.34 | 1.50 | 3.65 |
| 5–9 y | 1.58 | 1.06 | 2.36 |
| 10–14 y | 1.54 | 1.02 | 2.31 |
| ≥15 y | Reference | ||
| Depression | 1.72 | 1.15 | 2.59 |
| RBD | 1.99 | 1.37 | 2.91 |
| Disease duration | |||
| ≥10 y | 2.29 | 1.44 | 3.64 |
| 5–10 y | 1.96 | 1.30 | 2.93 |
| 2–5 y | 1.09 | 0.73 | 1.62 |
| 1–2 y | 0.98 | 0.63 | 1.53 |
| 0–1 y | Reference | ||
| Hoehn-Yahr scale | |||
| Stage 5 | 5.63 | 1.62 | 19.56 |
| Stage 4 | 9.89 | 5.05 | 19.38 |
| Stage 3 | 2.58 | 1.87 | 3.55 |
| Stage 2 | 1.63 | 1.20 | 2.21 |
| Stage 1 | Reference | ||
PD, Parkinson’s disease; OR, odds ratio; CI, confidence interval; RBD, rapid eye movement sleep behavior disorder.
Factors related to the quality of life in Chinese PD patients
As shown in Table 5, factors related to the quality of life in Chinese PD patients included nocturnal symptoms, education level, depression, disease duration, and Hoehn-Yahr stage (all p < 0.01).
Table 5
Factors related to the quality of life in Chinese PD patients
| Source of variation | Degrees of freedom | Type III sum of squares | Mean square | F value | p |
| Education level | 3 | 5193.23 | 1731.08 | 7.51 | <0.0001 |
| Depression | 1 | 10380.31 | 10380.31 | 45.01 | <0.0001 |
| Disease course | 4 | 7278.14 | 1819.54 | 7.89 | <0.0001 |
| Hoehn-Yahr staging | 4 | 19711.12 | 4927.78 | 21.37 | <0.0001 |
| Nocturnal symptoms | 1 | 58167.39 | 58167.39 | 252.22 | <0.0001 |
PD, Parkinson’s disease.
DISCUSSION
Nocturnal symptoms are common in PD patients and might seriously affect the quality of their life [9, 12]. Due to the difficulties in health monitoring during nighttime, patients and doctors often ignore those symptoms, leading to persistence or worsening [28]. This study explored the occurrence of nocturnal symptoms and associated factors in a large group of Chinese patients with PD and the relationship between nocturnal symptoms and quality of life, aiming to strengthen doctors’ and patients’ understanding and draw attention to nocturnal symptoms of PD.
To our knowledge, this is the first national study to investigate the prevalence of nocturnal symptoms in outpatients diagnosed with PD in China. With a large sample size of 1500 cases, we reported that in the Chinese population, 38.4% of patients with early- and mid-stage PD were found to have nocturnal symptoms, of which 59.2% were older than 65 years. The most common symptoms were sleep disorders and factors related to the development of nocturnal symptoms included a lower education level, presence of advanced depression, longer disease duration, and a higher Hoehn-Yahr stage.
Bhidayasiri et al. [29] reported a high prevalence rate of nocturnal symptoms the Thai population (96.6% of all PD patients). In the present study, the prevalence of nocturnal symptoms in Chinese PD patients was as low as 38.4%. Several reasons may explain the inconsistent results, despite that both studies are among the Asian population. Firstly, the sample sizes of both studies were different, and the vast majority of the patients included in our study were at early and middle stages, which may lead to a lower prevalence of nocturnal symptoms. Secondly, the evaluation tools differed (the PDSS-2 scale in this study and the MPDSS scale in Bhidayasiri’s study). Thirdly, the present study set stricter diagnostic criteria for nocturnal symptoms. Suzuki et al. [10] showed that a total score of PDSS-2 ≥15 could help to detect lack of sleep in PD patients. Nevertheless, an MPDSS score ≤6 was considered abnormal in Bhidayasiri’s study. Studies in the Caucasian population evaluated nocturnal symptoms using Pittsburgh Sleep Quality Index and Epworth Sleepiness Scale, and the prevalence rates were slightly higher than our results (50% in the UK [12] and 63% in Slovakia [13]).
Pandey et al. [12] assessed the quality of sleep by Pittsburgh Sleep Quality Index and EDS by Epworth Sleepiness Scale and reported nocturnal symptoms in 50% of patients (50/100). Poor sleep quality was not related to low quality of life in the abovementioned study, with authors hypothesizing it being the secondary outcome to reported depression and anxiety. In our study, multiple factors were found to be related to the quality of life among Chinese PD patients, including the presence of nocturnal symptoms (p < 0.001). Most likely, the difference could be explained by the different sample size, as previous studies also supported the association between sleep quality and quality of life [9].
On the other hand, Havlikova [13], using the same scales and almost the same number of patients (all of whom were Caucasians), reported various sleep-related problems during nighttime in 73.1%, but only 24.7% of patients had PSQI score >5, with no significant association between depression and lower quality of life. At the same time, the authors noted a question of appropriate evaluation for depression in the sample, as only patients who were able and motivated to come in for examination and interview were usually included in the studies.
Therefore, choosing the evaluation scale is essential for accurate assessment of nocturnal symptoms. PDSS-2 is a second revision of the visual analog scale PDSS addressing 15 commonly reported symptoms associated with sleep disturbance. The original PDSS scale was reviewed to match the diagnostic needs for PD-specific sleep disorders, such as sleep apnea, RBD, and RLS [30]. For that purpose, all night symptoms were divided into three main domains: motor symptoms at night, PD symptoms at night, and disturbed sleep. PDSS-2 score has already been used in studies to identify the changes in sleep quality of PD patients and proved to be less methodology- and sample-dependent compared to other scores [25, 26]. In the present study, we found that PDSS-2 is more understandable and recognized by physicians, and a total score of PDSS-2 in its three domains helps successfully indicate which nocturnal symptoms seriously affect the sleep and life of PD patients. It was also found that the sleep disturbance domain had the largest proportion in the group; this result suggested that attention should be paid to this particular domain in clinical practice, and appropriate intervention methods should be selected. According to the 2019 MDS Parkinson’s Non-motor Symptom Management Consensus, those methods might include PD treatment adjustment, sleep hygiene consultation or prescription of sleeping medication, and the option of continuous positive airway pressure for insomnia patients with obstructive sleep apnea [31].
In our study, PD patients with advanced age, long duration of disease, late Hoehn-Yahr stage, rigidity as the first symptom, RBD, mental illness, and comorbidity were more likely to have nocturnal symptoms. Still, it has to be noted RLS and RBD are part of PDSS-2, and such associations are not surprising. Nevertheless, it is suggested that attention should be paid to the nocturnal symptoms and sleep conditions of PD patients [32, 33] with the factors mentioned above in clinical practice. Simultaneously, this study found that the pharmaceutical treatment amount and medication frequency in the group with nocturnal symptoms were higher compared to the group without nocturnal symptoms.
We noted that education, depression, RBD, PD duration, and Hoehn-Yahr stage were associated with nocturnal symptoms in Chinese PD patients. It is highly advised to focus on the factors with clinical significance and remind doctors and patients of what actions should be taken, such as strengthening doctor-patient communication [34] to improve the understanding of and attention to PD nocturnal symptoms and provide education to patients. The presence of nocturnal symptoms is therefore related to the increase in disease duration and aggravation of other symptoms [10, 17, 28, 35]. It suggests that clinicians should strengthen the monitoring of people at high risk of nocturnal symptoms and pay attention to their treatment, as suggested by similar studies [32, 33]. Depression and RBD are also associated with nocturnal symptoms in PD patients, further indicating the need to evaluate the patient’s mental state and quality of life.
Previous studies showed that anti-PD drugs, in particular L-dopa, adversely affect nocturnal sleep in patients with PD, especially in terms of sleep quality and nocturia [36, 37]. In this study, the patients with nocturnal symptoms were taking more anti-PD drugs than those without nocturnal symptoms, although the multivariable analysis did not identify anti-PD drugs as being associated with the development of nocturnal symptoms. Nevertheless, the top two highest subscores of PDSS-2 were nocturnal motor and PD symptoms. Hence, it is possible that dosing up with anti-PD drugs might cause some resistance to rescuing PD and nocturnal symptoms in the nocturnal symptom group. Furthermore, previous trials revealed that the long-acting dopamine agonist rotigotine rescued some items of the nocturnal motor dysfunction in PDSS-2 [38–40]. Additional studies are necessary to examine the role of anti-PD drugs in developing nocturnal symptoms.
Our study points out that it is necessary to carry out early screening, control, and improve nocturnal symptoms so that patients can obtain a better quality of life. In recent years, with the advancement of medical technology, clinical specialists have access to effective disease monitoring and treatment methods (symptomatic and personalized medicine) through newly developed scales (PDSS-2) and electronic medical equipment (e.g., PSG, wearable devices) [4, 41, 42]. It may help monitor and effectively relieve nocturnal symptoms of PD patients [43].
The fact that there is still insufficient attention to PD nocturnal symptoms and a serious lack of relevant evaluation tools and standards [44, 45] is concerning. Long disease duration means that the same PD patients sometimes are used to evaluate the nocturnal symptoms under different scoring scales and evaluation standards. The evaluation criteria that meet the actual situation and clinical practice in China are needed. It will help identify the nocturnal symptoms of PD in a timely and effective manner and improve the prognosis of nocturnal symptoms in patients with PD.
This study has some limitations. First, it was a cross-sectional study, and it can only analyze the associations, not causality. Second, it was based on the PDSS-2 scale to observe the symptoms of PD patients at night and the PDSS-2 scale to assess night sleep. Although items 13 and 14 include morning tremors, fatigue, and lethargy, they cannot cover the entire night and early morning. Scales evaluated by physicians should be established. Third, the study was only on patients recruited from outpatient departments, and 94% were in the early-to-middle stages. Further studies are needed to explore nocturnal symptoms in patients with severe PD. Finally, in clinical practice, information on nocturnal symptoms of PD is mainly obtained by self-report and scale evaluation, which not only is subjective but also makes it difficult to obtain accurate and comprehensive data. Based on the current research results and the limitations, it should be noted that the PDSS-2 scale is insufficient to obtain full information on nocturnal symptoms in PD patients, while ESS and PSQI demonstrate previous inconsistencies in evaluation. Therefore, new revisions of the existing scales and the development of new scales/questionnaires should be prioritized. Portable wearable sensor equipment might be another promising way to deal with this problem in the future.
Conclusion
This study found that 38.4% of Chinese PD patients have nocturnal symptoms, even in early- and mid-stage PD, of which 59.2% were older than 65. PD stage and duration were independent factors associated with the development of nocturnal symptoms. Nocturnal symptoms were significantly associated with the quality of life in PD patients, and thus should be included in the assessment and care plan for patients with PD.
ACKNOWLEDGMENTS
The authors appreciate Dr. Kun Nie, Dr. Chao Han, Dr. Jifeng Guo, and Dr. Pei Huang for collecting materials for this manuscript.
FUNDING
This study was funded by UCB Pharma. The sponsor was the Chinese Society of Gerontology and Geriatrics, and the co-sponsor was the Parkinson’s Disease and Movement Disorders Group of the Neurology Society of the Chinese Medical Association.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
DATA AVAILABILITY
The data supporting the findings of this study are available on request from the corresponding author.
References
[1] | Tysnes OB , Storstein A ((2017) ) Epidemiology of Parkinson’s disease. J Neural Transm (Vienna) 124: , 901–905. |
[2] | ((2018) ) Global, regional, and national burden of Parkinson’s disease, 1990-2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol 17: , 939–953. |
[3] | Zhang ZX , Roman GC , Hong Z , Wu CB , Qu QM , Huang JB , Zhou B , Geng ZP , Wu JX , Wen HB , Zhao H , Zahner GE ((2005) ) Parkinson’s disease in China: Prevalence in Beijing, Xian, and Shanghai. Lancet 365: , 595–597. |
[4] | Lopiano L , Modugno N , Marano P , Sensi M , Meco G , Solla P , Gusmaroli G , Tamma F , Mancini F , Quatrale R , Zangaglia R , Bentivoglio A , Eleopra R , Gualberti G , Melzi G , Antonini A ((2019) ) Motor and non-motor outcomes in patients with advanced Parkinson’s disease treated with levodopa/carbidopa intestinal gel: Final results of the GREENFIELD observational study. J Neurol 266: , 2164–2176. |
[5] | Sveinbjornsdottir S ((2016) ) The clinical symptoms of Parkinson’s disease. J Neurochem 139: Suppl 1, 318–324. |
[6] | Arnulf I , Konofal E , Merino-Andreu M , Houeto JL , Mesnage V , Welter ML , Lacomblez L , Golmard JL , Derenne JP , Agid Y ((2002) ) Parkinson’s disease and sleepiness: An integral part of PD. Neurology 58: , 1019–1024. |
[7] | Lang AE ((2011) ) A critical appraisal of the premotor symptoms of Parkinson’s disease: Potential usefulness in early diagnosis and design of neuroprotective trials. Mov Disord 26: , 775–783. |
[8] | Hirata K , Högl B , Tan EK , Videnovic A ((2015) ) Sleep problems in Parkinson’s disease. Parkinsons Dis 2015: , 507948. |
[9] | Amara AW , Chahine LM , Videnovic A ((2017) ) Treatment of sleep dysfunction in Parkinson’s disease. Curr Treat Options Neurol 19: , 26. |
[10] | Suzuki K , Miyamoto M , Miyamoto T , Iwanami M , Hirata K ((2011) ) Sleep disturbances associated with Parkinson’s disease. Parkinsons Dis 2011: , 219056. |
[11] | Wüllner U , Fuchs G , Reketat N , Randerath O , Kassubek J ((2012) ) Requirements for Parkinson’s disease pharmacotherapy from the patients’ perspective: A questionnaire-based survey. Curr Med Res Opin 28: , 1239–1246. |
[12] | Pandey S , Bajaj BK , Wadhwa A , Anand KS ((2016) ) Impact of sleep quality on the quality of life of patients with Parkinson’s disease: A questionnaire based study. Clin Neurol Neurosurg 148: , 29–34. |
[13] | Havlikova E , van Dijk JP , Nagyova I , Rosenberger J , Middel B , Dubayova T , Gdovinova Z , Groothoff JW ((2011) ) The impact of sleep and mood disorders on quality of life in Parkinson’s disease patients. J Neuroly 258: , 2222–2229. |
[14] | Yu RL , Tan CH , Wu RM ((2015) ) The impact of nocturnal disturbances on daily quality of life in patients with Parkinson’s disease. Neuropsychiatr Dis Treat 11: , 2005–2012. |
[15] | Lin YY , Chen RS , Lu CS , Huang YZ , Weng YH , Yeh TH , Lin WY , Hung J ((2017) ) Sleep disturbances in Taiwanese patients with Parkinson’s disease. Brain Behav 7: , e00806. |
[16] | Xue F , Wang FY , Mao CJ , Guo SP , Chen J , Li J , Wang QJ , Bei HZ , Yu Q , Liu CF ((2018) ) Analysis of nocturnal hypokinesia and sleep quality in Parkinson’s disease. J Clin Neurosci 54: , 96–101. |
[17] | Xiang W , Sun YQ , Teoh HC ((2018) ) Comparison of nocturnal symptoms in advanced Parkinson’s disease patients with sleep disturbances: Pramipexole sustained release versus immediate release formulations. Drug Des Devel Ther 12: , 2017–2024. |
[18] | Shulman LM , Taback RL , Rabinstein AA , Weiner WJ ((2002) ) Non-recognition of depression and other non-motor symptoms in Parkinson’s disease. Parkinsonism Relat Disord 8: , 193–197. |
[19] | Barone P , Amboni M , Vitale C , Bonavita V ((2004) ) Treatment ofnocturnal disturbances and excessive daytime sleepiness inParkinson’s disease. Neurology 63: , S35–38. |
[20] | Martinez-Martin P , Rodriguez-Blazquez C , Kurtis MM , Chaudhuri KR ((2011) ) The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov Disord 26: , 399–406. |
[21] | Viwattanakulvanid P , Kaewwilai L , Jitkritsadakul O , Brenden NR , Setthawatcharawanich S , Boonrod N , Mekawichai P , Bhidayasiri R ((2014) ) The impact of the nocturnal disabilities of Parkinson’s disease on caregivers’ burden: Implications for interventions. J Neural Transm (Vienna) 121: Suppl 1, S15–24. |
[22] | Trenkwalder C , Kohnen R , Högl B , Metta V , Sixel-Döring F , Frauscher B , Hülsmann J , Martinez-Martin P , Chaudhuri KR ((2011) ) Parkinson’s disease sleep scale–validation of the revised version PDSS-2. Mov Disord 26: , 644–652. |
[23] | Yang HJ , Kim HJ , Koh SB , Kim JS , Ahn TB , Cheon SM , Cho JW , Kim YJ , Ma HI , Park MY , Baik JS , Lee PH , Chung SJ , Kim JM , Song IU , Kim JY , Sung YH , Kwon DY , Lee JH , Lee JY , Kim JS , Yun JY , Kim HJ , Hong JY , Kim MJ , Youn J , Kim JS , Oh ES , Yoon WT , You S , Kwon KY , Park HE , Lee SY , Kim Y , Kim HT , Kim SJ ((2020) ) Subtypes of sleep disturbance in Parkinson’s disease based on the cross-culturally validated Korean version of Parkinson’s Disease Sleep Scale-2. J Clin Neurol 16: , 66–74. |
[24] | Suzuki K , Miyamoto M , Miyamoto T , Tatsumoto M , Watanabe Y , Suzuki S , Iwanami M , Sada T , Kadowaki T , Numao A , Trenkwalder C , Hirata K ((2012) ) Nocturnal disturbances and restlessness in Parkinson’s disease: Using the Japanese version of the Parkinson’s disease sleep scale-2. J Neurol Sci 318: , 76–81. |
[25] | Wang G , Cheng Q , Zeng J , Bai L , Liu GD , Zhang Y , Tan YY , Pan J , Hong Z , Wang Y , Chen SD ((2008) ) Sleep disorders in Chinese patients with Parkinson’s disease: Validation study of a Chinese version of Parkinson’s disease sleep scale. J Neurol Sci 271: , 153–157. |
[26] | Suzuki K , Miyamoto T , Miyamoto M , Suzuki S , Numao A , Watanabe Y , Tatsumoto M , Sakuta H , Watanabe Y , Fujita H , Iwanami M , Sada T , Kadowaki T , Hashimoto K , Trenkwalder C , Hirata K ((2015) ) Evaluation of cutoff scores for the Parkinson’s disease sleep scale-2. Acta Neurol Scand 131: , 426–430. |
[27] | Bhidayasiri R , Truong DD ((2012) ) Therapeutic strategies for nonmotor symptoms in early Parkinson’s disease: The case for a higher priority and stronger evidence. Parkinsonism Relat Disord 18: Suppl 1, S110–113. |
[28] | Todorova A , Jenner P , Ray Chaudhuri K ((2014) ) Non-motor Parkinson’s: Integral to motor Parkinson’s, yet often neglected. Pract Neurol 14: , 310–322. |
[29] | Bhidayasiri R , Mekawichai P , Jitkritsadakul O , Panyakaew P , Kaewwilai L , Boonrod N , Petchrutchatachart S , Jagota P , Boonpeng K , Singmaneesakulchai S , Setthawatcharawanich S ((2014) ) Nocturnal journey of body and mind in Parkinson’s disease: The manifestations, risk factors and their relationship to daytime symptoms. Evidence from the NIGHT-PD study. J Neural Transm (Vienna) 121: Suppl 1, S59–68. |
[30] | Arnaldi D , Cordano C , De Carli F , Accardo J , Ferrara M , Picco A , Tamburini T , Brugnolo A , Abbruzzese G , Nobili F ((2016) ) Parkinson’s Disease Sleep Scale 2: Application in an Italian population. Neurol Sci 37: , 283–288. |
[31] | Roychowdhury S , Forsyth DR ((2012) ) Sleep disturbance in Parkinson disease. J Clin Gerontol Geriatr 3: , 53–61. |
[32] | Dhawan V , Healy DG , Pal S , Chaudhuri KR ((2006) ) Sleep-related problems of Parkinson’s disease. Age Ageing 35: , 220–228. |
[33] | Stefani A , Högl B ((2020) ) Sleep in Parkinson’s disease. Neuropsychopharmacology 45: , 121–128. |
[34] | Ha JF , Longnecker N ((2010) ) Doctor-patient communication: A review. Ochsner J 10: , 38–43. |
[35] | Rektorova I , Aarsland D , Chaudhuri KR , Strafella AP ((2011) ) Nonmotor symptoms of Parkinson’s disease. Parkinsons Dis 2011: , 351461. |
[36] | Taguchi S , Koide H , Oiwa H , Hayashi M , Ogawa K , Ito C , Nakashima K , Yuasa T , Yasumoto A , Ando H , Fujikake A , Fukuoka T , Tokui K , Izumi M , Tsunoda Y , Kawagashira Y , Okada Y , Niwa JI , Doyu M ((2021) ) Antiparkinsonian drugs as potent contributors to nocturnal sleep in patients with Parkinson’s disease. PLoS One 16: , e0255274. |
[37] | Bollu PC , Sahota P ((2017) ) Sleep and Parkinson disease. Mo Med 114: , 381–386. |
[38] | Trenkwalder C , Kies B , Rudzinska M , Fine J , Nikl J , Honczarenko K , Dioszeghy P , Hill D , Anderson T , Myllyla V , Kassubek J , Steiger M , Zucconi M , Tolosa E , Poewe W , Surmann E , Whitesides J , Boroojerdi B , Chaudhuri KR , Recover Study Group ((2011) ) Rotigotine effects on early morning motor function and sleep in Parkinson’s disease: A double-blind, randomized, placebo-controlled study (RECOVER). Mov Disord 26: , 90–99. |
[39] | Vallderiola F , Compta Y , Aparicio J , Tarradellas J , Salazar G , Oliver JM , Callen A , Delgado T , Nobbe F ((2015) ) Effects of night-time use of rotigotine on nocturnal symptoms in Parkinson’s Disease. Parkinsons Dis 2015: , 475630. |
[40] | Suzuki K ((2021) ) Current update on clinically relevant sleep issues in Parkinson’s disease: A narrative review. J Parkinsons Dis 11: , 971–992. |
[41] | Madrid-Navarro CJ , Escamilla-Sevilla F , Mínguez-Castellanos A , Campos M , Ruiz-Abellán F , Madrid JA , Rol MA ((2018) ) Multidimensional circadian monitoring by wearable biosensors in Parkinson’s disease. Front Neurol 9: , 157. |
[42] | Vasconcelos L ((2019) ) Parkinson’s disease rehabilitation:Effectiveness approaches and new perspectives. In Physical Therapy Effectiveness Bernardo-Filho M, Cunha de Sá-Caputo D, Taiar R, eds. IntechOpen, Rijeka. https://doi.org/10.5772/intechopen.89360. |
[43] | Pirtošek Z , Bajenaru O , Kovács N , Milanov I , Relja M , Skorvanek M ((2020) ) Update on the management of Parkinson’s diseasefor general neurologists. Parkinsons Dis 2020: , 9131474. |
[44] | Loddo G , Calandra-Buonaura G , Sambati L , Giannini G , Cecere A , Cortelli P , Provini F ((2017) ) The Treatment of Sleep disorders in Parkinson’s disease: From research to clinical practice. Front Neurol 8: , 42. |
[45] | Schrempf W , Brandt MD , Storch A , Reichmann H ((2014) ) Sleep disorders in Parkinson’s disease. J Parkinsons Dis 4: , 211–221. |




