40-Year Incidence of Early-Onset Parkinson’s Disease in Southeast Minnesota
Abstract
Background:
Parkinson’s disease (PD) most commonly surfaces at middle age. An earlier onset is named early-onset Parkinson’s disease (EOPD), but the exact definition is a matter of ongoing scientific debate.
Objective:
To investigate 40-year EOPD incidence trends in a population-based cohort of parkinsonism in Olmsted County, Minnesota.
Methods:
We used the Rochester Epidemiology Project (REP) to identify all incident EOPD cases in Olmsted County, 1976–2015. A movement-disorder specialist reviewed all cases to confirm the EOPD diagnosis. For EOPD definition, we used two age cut-offs: motor-symptom onset at or before 50 and 55 years.
Results:
EOPD incidence was 1.43/100,000 person-years for ≤55 and 0.55/100,000 for ≤50 years. Men had a higher incidence in both groups [1.84 vs. 1.03 (p = 0.04); and 0.70 vs. 0.40 (p = 0.24), respectively]. EOPD incidence of patients with motor-symptom onset before age 55 increased from 1.02/100.000 person-year 1976–1985, to 1.32/100.000 person-year 2006–2015. A similar trend was observed when ≤50 years cut-off was used (0.28/100,000 person-years 1976–1985, to 0.59/100,000 person-year 2006–2015). However, negative binomial regression found no significant change in incidence per 10 years (RR = 1.04 and 1.24 in the two groups). Incidence was consistently higher in men than women. Median time from EOPD-symptom onset to death was shorter in the EOPD ≤55 group (21.9 years) compared to the EOPD ≤50 group (25.6 years).
Conclusion:
We observed an increased trend in the incidence of EOPD with both cut-off ages. Overall, incidence of EOPD was 1.43 (≤55) and 0.55 (≤50) cases per 100,000 person-years, higher in men.
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disease [1]; the incidence of PD has been increasing in the population [2, 3], estimated to triple in the next 50 years [4]. The youngest cases of PD are defined as early-onset PD (EOPD) and carry an additional set of societal, familiar, social, and job-related consequences. Although the exact age cut-off for the definition of EOPD is still under scrutiny [5], we previously reported an incidence of 0.81/100,000 person-years when motor-symptom onset was ≤50 years of age, and 2.05/100,000 person-years when it was ≤55 years of age [5]. Compared to overall PD (incidence of 14.2/100,000 person-years), the incidence of EOPD is 17.5-fold lower and 6.9-fold lower when using ≤50 and ≤55 as cut-off ages.
In previous studies, we reported incidence trends of the of PD in Olmsted County (MN) between 1976 and 2005 [2], showing a progressive, increased incidence of PD in the population in 30 years. We used the same methodology to explore the incidence trends of EOPD, but we expanded our original cohort (1976–2005) to include 10 more years of observation (from 2006 to 2015) in Olmsted County, obtaining a study period of 40 years.
We also added 2 additional years (2016–2017) of follow-up after the incidence period to ensure catchment of those patients who had motor-symptom onset during the 40-year incidence period but received a PD diagnosis between 2016 and 2017. This methodology had already been used in a previous study regarding the incidence of EOPD in 7 counties in Minnesota [5].
MATERIALS AND METHODS
Ascertainment of cases
We used the Rochester Epidemiology Project (REP) medical records-linkage system to establish our cohort of EOPD between 1976 and 2015. The full cohort comprises 3 cohorts established at different times. The first cohort (1976–1990) was created in 1999 [6], the second (1991–2005) in 2013 [7], and the third has been defined for this current study, spanning from 2006 to 2015.
The methodology for case detection, as well as parkinsonism and EOPD definitions were the same as previously published [5]. We defined a case of EOPD (≤55) if a patient had motor-symptom onset (regardless of diagnosis date) before the 56th birthday, whereas he or she was included as a case of EOPD (≤50) if the motor-symptom onset occurred before the 51st birthday.
In brief, we used the Rep database to screen for potential cases of PD, setting as geographical frame Olmsted County (MN) and as temporal timeframe 1976–2015. The exact list of ICD codes and rationale is presented elsewhere [5]. Our codes are designed to maximize catchment of PD patients; hence they are thought to maximize sensitivity at the cost of specificity. Following the initial computerized phase, a second manual phase is performed by the authors, which includes reading the clinical charts of all patients selected by the software to establish which cases are true positive vs false positive. Once a cohort of PD patients is identified, clinical chart review is performed, and data such as motor symptoms onset, diagnosis date, motor phenotype, date and cause of death are recorded.
Statistical analysis
The incidence of EOPD in Olmsted County over the 40-year study period was evaluated by comparing incident cases of EOPD against the at-risk Olmsted County population captured by the REP each year. Incidence rates are calculated relative to 100,000 person-years and age-adjusted based on the age threshold of EOPD being evaluated. The change in EOPD incidence over time was assessed using negative binomial regression accounting for calendar year and sex. Differences in EOPD incidence by decade and between men and women are reported as relative risks (RRs) with 95% confidence intervals.
Secondary analysis compared the duration of time between EOPD symptoms onset and clinical diagnosis across the 40-year study period using linear regression adjusted for patient age and sex. Results are reported as the expected change per 10 years with 95% CI.
Changes in EOPD incidence were also evaluated across individuals born during different decades. Patients were grouped into 10-year birth cohorts centered around the beginning of each decade and cohort-specific incidences of EOPD were calculated.
Standard protocol approvals, registrations, and patient consents
This study was approved by the Mayo Clinic and Olmsted Medical Center Institutional Review Boards. Participating patients (or their legally authorized representatives) provided informed written consent for use of their medical information for research.
Data sharing
Data from the Rochester Epidemiology Project (REP) medical records-linkage system are available upon request from qualified investigators.
RESULTS
≤55 years cohort
We identified 55 cases of EOPD (≤55) in Olmsted County (MN) between 1976 and 2015. These included 35 (64%) men and 20 (36%) women, and their demographic characteristics are shown in Table 1. Rest tremor, bradykinesia, and rigidity were reported in approximately 90% of the patients, whereas impaired postural reflexes were present only in 50%.
Table 1
Birth cohorts and phenotype of our cohort of EOPD patients, both ages cut-off
| ≤55 years at motor symptoms onset | |||
| Birth cohort | Female (N = 20) | Male (N = 35) | Total (N = 55) |
| 1920 | 0 (0.0%) | 1 (2.9%) | 1 (1.8%) |
| 1930 | 2 (10.0%) | 6 (17.1%) | 8 (14.5%) |
| 1940 | 5 (25.0%) | 6 (17.1%) | 11 (20.0%) |
| 1950 | 6 (30.0%) | 12 (34.3%) | 18 (32.7%) |
| 1960 | 6 (30.0%) | 7 (20.0%) | 13 (23.6%) |
| 1970 | 1 (5.0%) | 3 (8.6%) | 4 (7.3%) |
| Rest tremor | 17 (85.0%) | 32 (91.4%) | 49 (89.1%) |
| Bradykinesia | 17 (85.0%) | 32 (91.4%) | 49 (89.1%) |
| Impaired postural reflexes | 10 (50.0%) | 18 (51.4%) | 28 (50.9%) |
| Rigidity | 19 (95.0%) | 31 (88.6%) | 50 (90.9%) |
| ≤50 years at motor symptoms onset | |||
| Birth cohort | Female (N = 7) | Male (N = 12) | Total (N = 19) |
| 1930 | 1 (14.3%) | 1 (8.3%) | 2 (10.5%) |
| 1940 | 0 (0.0%) | 1 (8.3%) | 1 (5.3%) |
| 1950 | 0 (0.0%) | 4 (33.3%) | 4 (21.1%) |
| 1960 | 5 (71.4%) | 3 (25.0%) | 8 (42.1%) |
| 1970 | 1 (14.3%) | 3 (25.0%) | 4 (21.1%) |
| Rest tremor | 5 (71.4%) | 12 (100.0%) | 17 (89.5%) |
| Bradykinesia | 7 (100.0%) | 12 (100.0%) | 19 (100.0%) |
| Impaired postural reflexes | 5 (71.4%) | 3 (25.0%) | 8 (42.1%) |
| Rigidity | 7 (100.0%) | 12 (100.0%) | 19 (100.0%) |
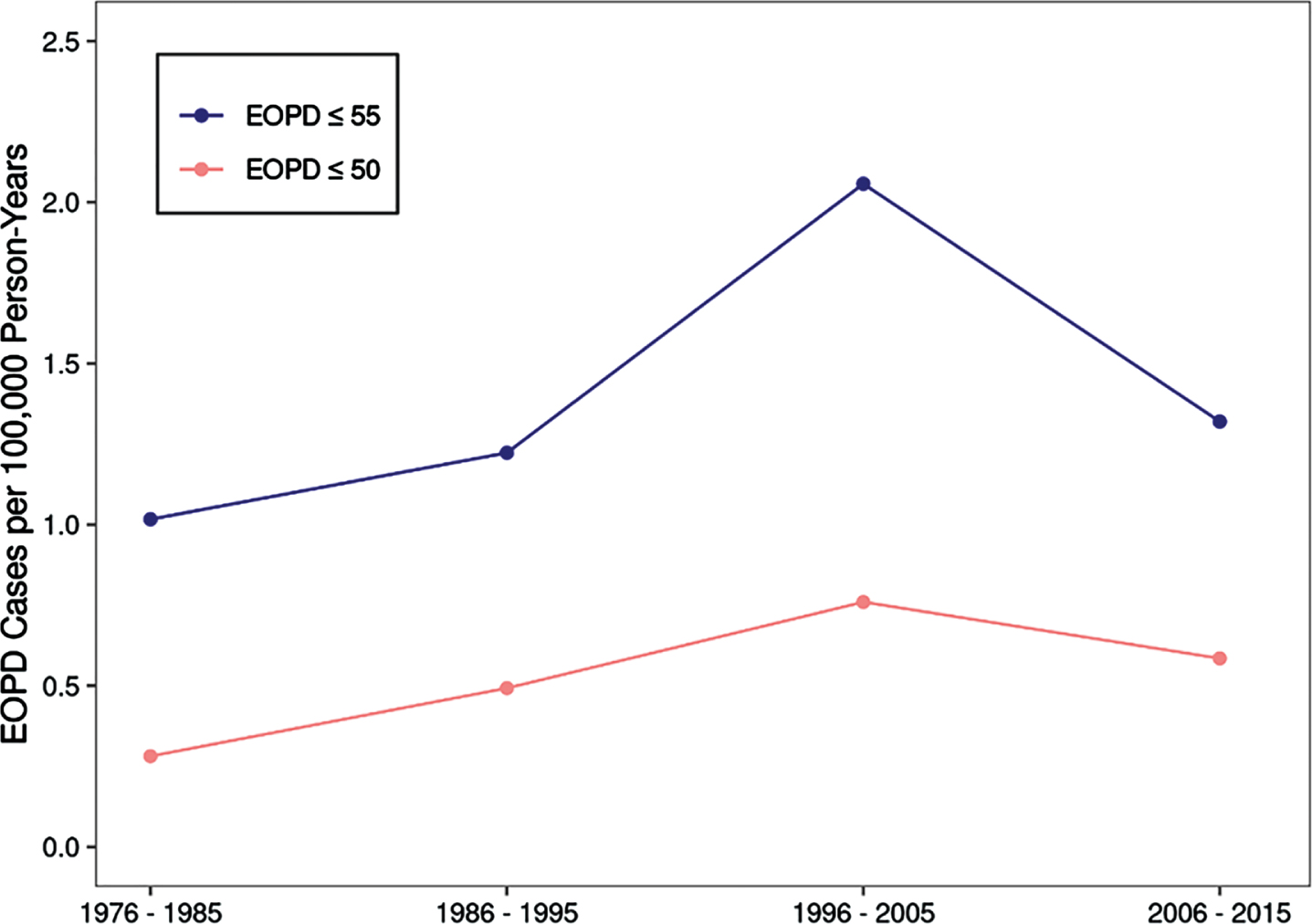
The overall incidence of EOPD (≤55) was 1.43 cases per 100,000 person-years in Olmsted County. Men had a significantly greater risk of EOPD with an incidence of 1.84 cases per 100,000 compared to women, with an incidence of 1.03 cases per 100,000 person-years (RR = 1.79, 95% CI: 1.03 – 3.09, p = 0.04). Exploring the different decades, incidence was between 1976 and 1985 was 1.02 (1.56 in men and 0.50 in women); between 1986 and 1995, it was 1.22 (1.79 in men and 0.67 in women); between 1996 and 2005, it was 2.06 (2.35 in men and 1.77 in women); and in the last decade, 2006–2015, it was 1.32 (1.61 in men and 1.04 in women, Table 2).
Table 2
Incidence of EOPD over time, both ages cut-off
| Incidence of EOPD ≤55 years at motor symptoms onset | |||
| Motor symptoms onset | Female (N = 20) | Male (N = 35) | Total (N = 55) |
| 1976–85 | 0.50 | 1.56 | 1.02 |
| 1986–95 | 0.66 | 1.79 | 1.22 |
| 1996–2005 | 1.77 | 2.35 | 2.06 |
| 2006–2015 | 1.04 | 1.61 | 1.32 |
| Incidence of EOPD ≤50 years at motor symptoms onset | |||
| Motor symptoms onset | Female (N = 7) | Male (N = 12) | Total (N = 19) |
| 1976–85 | 0.28 | 0.29 | 0.28 |
| 1986–95 | 0.00 | 0.99 | 0.49 |
| 1996–2005 | 0.44 | 1.08 | 0.76 |
| 2006–2015 | 0.77 | 0.40 | 0.59 |
Figure 1 shows the yearly incidence of EOPD (≤55 and ≤50) for the full population of Olmsted County. Negative binomial regression found no significant change in overall incidence per 10 years (RR = 1.06 per 10 years, 95% CI 0.84 – 1.34, p = 0.64). Moreover, there was no significant change observed for women (RR = 1.25 per 10 years. 95% CI 0.84–1.87, p = 0.26) or men (RR = 0.96 per 10 years, 95% CI 0.72 – 1.29, p = 0.79).
Fig. 1
Incidence of EOPD in Olmsted County (1976–2015) broken down by age cut-off.
The median time from EOPD symptom onset to clinical diagnosis was 1.35 (IQR 0.51 – 2.00) years for the full cohort, 1.40 (0.41 – 2.19) years in men, and 1.26 (IQR 0.66 – 1.98) years in women. There was no change in time from symptom onset to diagnosis across the study period (difference = 0.08 per 10 years, 95% CI: – 0.27 to 0.42, p = 0.66).
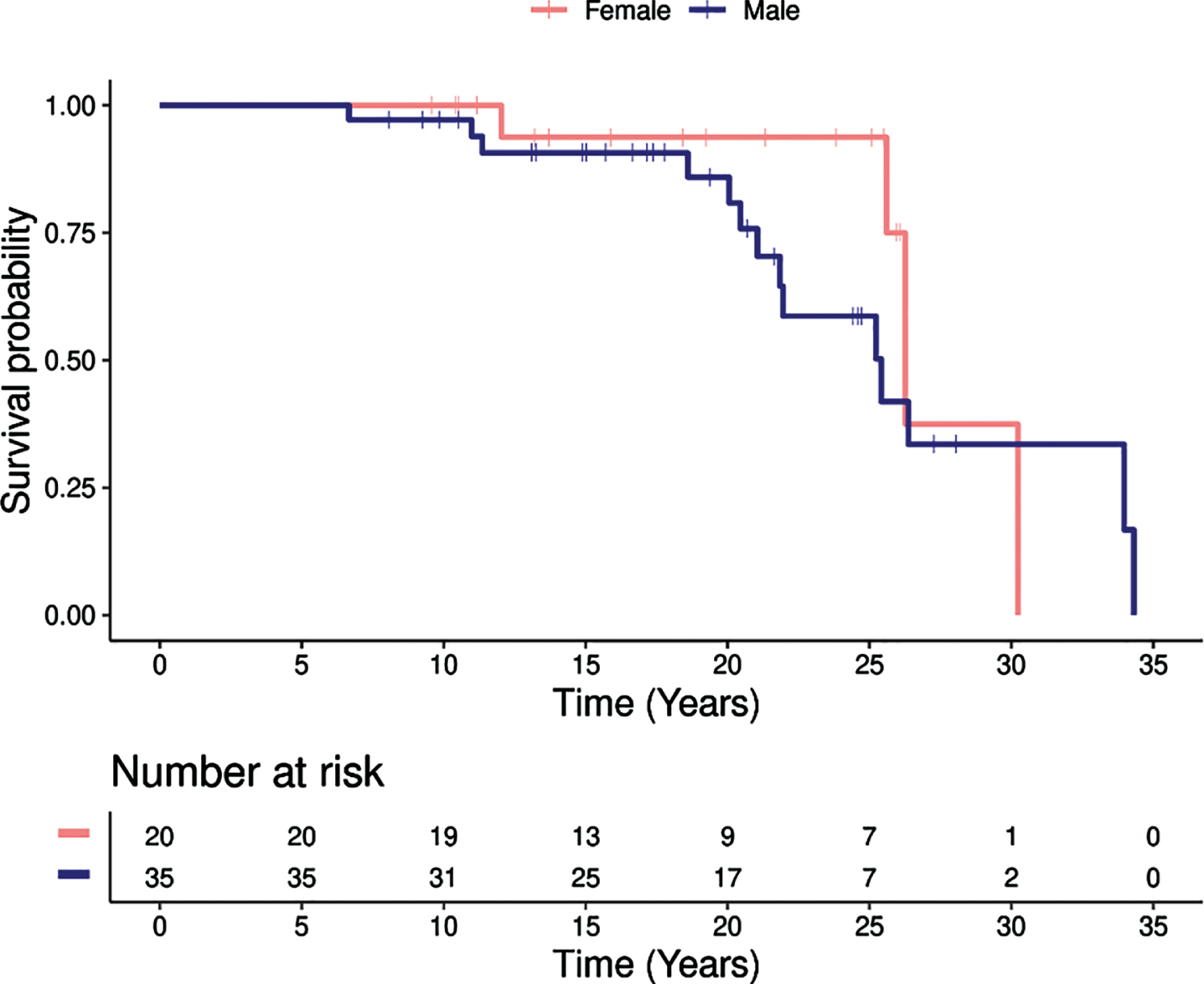
At time of data collection, 18/55 (32.7%) patients in the EOPD (≤55) cohort had died including 14 men (77.8%) and 4 women (22.2%). The median age of death was 75 (IQR: 66 – 79) years; it was 74 (IQR: 65 – 79) years in men and 77 (IQR: 73 – 78) years in women. Time from EOPD motor symptom onset to death was 21.9 (IQR: 19.0 – 26.1) years, 21.5 (IQR: 19.0 – 25.4) years in men and 25.9 (IQR: 22.2 – 27.3) years in women. Figure 2 displays the survival of the EOPD cohort following the EOPD motor-symptom onset divided by patient sex: no significant difference in the risk of death between males and females (p = 0.31). In exploring the birth cohorts, we observed an increased clustering of EOPD in the more recent birth cohort explored (1940, 50, and 60, Table 1).
Fig. 2
Survival following the EOPD motor symptoms onset, broken down by sex.
≤50 years cohort
We identified 19 cases of EOPD (≤50) in Olmsted County (MN) between 1976 and 2015. Importantly, these patients are also part of the ≤50-years cohort. These were 12 (63%) men and 7 (37%) women, and their characteristics are reported in Table 1. In terms of motor phenotype, rest tremor, bradykinesia, and rigidity were reported in 90 to 100% of them; impaired postural reflexes were present in approximately 42% (Table 1).
The overall incidence of EOPD (≤50) was 0.55 cases per 100,000 person-years in Olmsted County. In males it was 0.70 cases per 100,000 person-years, and in females it was 0.40 cases per 100,000 person-years. This difference was not statistically significant (RR = 1.74, 95% CI: 0.68 – 4.41, p = 0.24). Breaking down by decade, incidence between 1976 and 1985 was 0.28 (0.29 in men and 0.28 in women); between 1986 and 1995, it was 0.49 (0.99 in men and 0.00 in women); between 1996 and 2005, it was 0.76 (1.08 in men and 0.44 in women); and in the last decade, 2006 to 2015, it was 0.59 (0.40 in men and 0.77 in women, Table 2).
Negative binomial regression found no significant change in overall incidence per 10 years (RR = 1.23 per 10 years, 95% CI 0.80 – 1.87, p = 0.34). Moreover, there was no significant change observed for females (RR = 1.81 per 10 years, 95% CI 0.81–4.05, p = 0.15) or males (RR = 1.01 per 10 years, 95% CI 0.60 – 1.69, p = 0.97).
At time of data collection, only 4/19 (21.1%) patients in the EOPD (≤50) cohort had died. Time from EOPD motor symptoms onset to death was 25.6 (IQR: 20.4 – 31.2) years including 21.1 (IQR: 19.8 – 27.5) in men and 30.2 in the only woman. There was more frequent clustering in the 1950, 1960, and 1970 birth cohorts (84.2% overall) as compared to the cohorts born in the 1930s and 1940s (Table 1).
DISCUSSION
We report that incidence of EOPD (using both age cut-off) increased in Olmsted County across four decades. However, in the last decade (2006–2015), the incidence of EOPD was lower than in the previous one (1996–2005). We hypothesize that this decrement in incidence is an artifact related to our methodology and to the shorter follow-up period compared with the previous decades. Indeed, in PD, there is a known delay between motor-symptom onset and PD diagnosis [8]. In EOPD this latency is expected to be higher [9], as it has been shown to require a longer time to diagnosis, more neurologists to perform the diagnosis, and more visits/investigation compared to LOPD [9]. This is probably because neurologists are less likely to consider PD in a younger person.
In our previous studies, we considered a 5-year period at the end of every timeframe analyzed to be sure of catching those cases that had onset within the timeframe but a later diagnosis. (For example, in Savica et al. [7], the timeframe was 1991 to 2005, but the analysis was extended until 2010 to capture cases with a diagnosis after 2005.) In this study, however, we were able to expand our case identification only until 2017 (2-year follow-up after the 1976–2015timeframe) [5]. Thus, missing some cases in the later years of our timeframe may have underestimated the incidence in the last decade, especially given our relatively small cohort (55 patients overall and 15 patients in the last decade). This could be especially true in women, who apparently have a longer time from motor-symptom onset to first physician and movement-disorder specialist encounter, in non-age-restricted PD [10].
The recorded lifetime presence of motor features in EOPD was similar to those previously reported [5]. Indeed, rest tremor, rigidity, and bradykinesia had a lifetime prevalence (we treated them as ever/never variables) of ≥90% in this study; however, the percentage seems to be higher than was observed in other studies [11–14]. We speculate that the difference between our study and others is secondary to differences in clinical criteria and case identification.
Interestingly, we did not observe a long delay between reported symptoms of onset and diagnosis. Indeed, given the potential unusual manifestation of symptoms of onset in EOPD, we were expecting a longer delay; on the other hand, we can explain these unexpected results because of the unique access to medical care available to the population of Southeast Minnesota. Therefore, it is possible that we were able to recognize cases earlier than what is noted in a different area with a less medicalized population and less access to care.
Median survival after EOPD motor-symptom onset in our ≤55 years cohort was 21.9 years and 25.6 years when onset was ≤50 years; this difference presumably reflected the influence of age. Not surprisingly, median survival in our EOPD cases was longer than reported for non-age-restricted overall PD (7–14 years after diagnosis, [15]). We note that survival in our EOPD cohort differed from another study [16], where it was 32 years in the <50 group and 18.5 years in the 50-to-69 group. This discrepancy should be interpreted cautiously because of the small sample size of our cohort (only had 19 cases of EOPD [≤50] versus 60 cases in Mehanna et al. [16]).
We observed PD clustering in the more recent birth cohorts (1940s, 1950s, and 1960s) compared to earlier birth cohorts. These findings are particularly interesting because they support the theory that the frequency of PD, and in particular EOPD, has increased more recently in the population, not necessarily due to earlier recognition by the neurologists/physicians; indeed, the delay in reaching a diagnosis [10] is expected to be more prolonged in EOPD than PD.
The increased incidence we observed may have been influenced by multiple factors, including environmental risk factors. The association between some common risk factors and PD (e.g., pollutants, pesticides, comorbidities) is well established, but specific studies accounting risk factors for EOPD only are lacking [17]. EOPD may have different pathologic pathways than LOPD and different interactions with external and internal agents.
In addition, clinical suspicion of EOPD might have increased as decades passed, with increasing awareness of this subtype of PD. However, this effect would mostly be impactful towards the end of the 40-year time frame.
Our study had several strengths. First, using the REP provided the opportunity to access the lifelong medical history of all Olmsted County residents; hence, we caught all incident EOPD cases in the geographical and temporal settings chosen. Second, to the best of our knowledge, this is one of the few studies in the literature to report the population-based trends of EOPD.
We also must acknowledge the limitations of our methodology. First, medical records are not standardized for research purposes; hence, some clinical information may not be systematically available. Second, we had a relatively small cohort; however, EOPD is a rare condition, therefore a larger sample size is only possible with multicenter population-based studies. Third, most of these patients did not have a pathological confirmation of the diagnosis because they are still living; however, we previously reported a clinical-pathological correlation of 86.7% [18] in our cohort of PD cases, using the same methodology for case ascertainment.
In conclusion, we observed a non-significant increase in incidence of EOPD (both ≤55 and ≤50 years of age) in Olmsted County between 1976 and 2015. Overall, incidence of EOPD was 1.43 (≤55) and 0.55 (≤50) cases per 100,000 person-years, significantly higher in men than in women only in the≤55 cohort.
ACKNOWLEDGMENTS
The authors thank Ms. Lea Dacy for editing and formatting assistance.
FUNDING
This publication was made possible by the Mayo Clinic CTSA through grant number UL1 TR002377 from the National Center for Advancing Translational Sciences (NCATS), a component of the National Institutes of Health (NIH) and R01 AG034676 from the National Institute on Aging of the National Institutes of Health and by the Mayo Foundation for Medical Education and Research. The funder had no role in the conception or preparation of this study.
CONFLICT OF INTEREST
Rodolfo Savica is an Editorial Board member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer review. The other authors have no conflicts of interest to report.
References
[1] | de Lau LML , Breteler MMB ((2006) ) Epidemiology of Parkinson’s disease. Lancet Neurol 5: , 525–535. |
[2] | Savica R , Grossardt BR , Bower JH , Ahlskog JE , Rocca WA ((2016) ) Time trends in the incidence of Parkinson disease. JAMA Neurol 73: , 981–989. |
[3] | Marras C , Beck JC , Bower JH , Roberts E , Ritz B , Ross GW , Abbott RD , Savica R , Van Den Eeden SK , Willis AW , Tanner CM , Parkinson’s Foundation P4 Group ((2018) ) Prevalence of Parkinson’s disease across North America. NPJ Parkinsons Dis 4: , 21. |
[4] | Dorsey ER , Sherer T , Okun MS , Bloem BR ((2018) ) The emerging evidence of the Parkinson pandemic. J Parkinsons Dis 8: , S3–S8. |
[5] | Camerucci E , Stang CD , Hajeb M , Turcano P , Mullan AF , Martin P , Ross OA , Bower JH , Mielke MM , Savica R ((2021) ) Early-onset Parkinsonism and early-onset Parkinson’s disease: A population-based study (2010-2015). J Parkinsons Dis 11: , 1197–1207. |
[6] | Bower JH , Maraganore DM , McDonnell SK , Rocca WA ((1999) ) Incidence and distribution of parkinsonism in Olmsted County, Minnesota, 1976–1990. Neurology 52: , 1214–1214. |
[7] | Savica R , Grossardt BR , Bower JH , Ahlskog JE , Rocca WA ((2013) ) Incidence and pathology of synucleinopathies and tauopathies related to parkinsonism. JAMA Neurol 70: , 859–866. |
[8] | Cervantes Arriaga A , Rodriguez Violante M , Camacho Ordonez A , Gonzalez Latapi P , Lopez Ruiz M , Estrada Bellmann I , Zuniga Ramirez C , Otero Cerdeira E ((2014) ) [Time from motor symptoms onset to diagnosis of Parkinson’s disease in Mexico]. Gac Med Mex 150: Suppl 2, 242–247. |
[9] | Rana AQ , Siddiqui I , Yousuf MS ((2012) ) Challenges in diagnosis of young onset Parkinson’s disease. J Neurol Sci 323: , 113–116. |
[10] | Saunders-Pullman R , Wang C , Stanley K , Bressman SB ((2011) ) Diagnosis and referral delay in women with Parkinson’s disease. Gend Med 8: , 209–217. |
[11] | Friedman A ((1994) ) Old-onset Parkinson’s disease compared with young-onset disease: Clinical differences and similarities. Acta Neurol Scand 89: , 258–261. |
[12] | Giovannini P , Piccolo I , Genitrini S , Soliveri P , Girotti F , Geminiani G , Scigliano G , Caraceni T ((1991) ) Early-onset Parkinson’s disease. Mov Disord 6: , 36–42. |
[13] | Mehanna R , Jankovic J ((2019) ) Young-onset Parkinson’s disease: Its unique features and their impact on quality of life. Parkinsonism Relat Disord 65: , 39–48. |
[14] | Wickremaratchi MM , Knipe MD , Sastry BS , Morgan E , Jones A , Salmon R , Weiser R , Moran M , Davies D , Ebenezer L , Raha S , Robertson NP , Butler CC , Ben-Shlomo Y , Morris HR ((2011) ) The motor phenotype of Parkinson’s disease in relation to age at onset. Mov Disord 26: , 457–463. |
[15] | Macleod AD , Taylor KS , Counsell CE ((2014) ) Mortality in Parkinson’s disease: A systematic review and meta-analysis. Mov Disord 29: , 1615–1622. |
[16] | Mehanna R , Moore S , Hou JG , Sarwar AI , Lai EC ((2014) ) Comparing clinical features of young onset, middle onset and late onset Parkinson’s disease. Parkinsonism Relat Disord 20: , 530–534. |
[17] | Bellou V , Belbasis L , Tzoulaki I , Evangelou E , Ioannidis JP ((2016) ) Environmental risk factors and Parkinson’s disease: An umbrella review of meta-analyses. Parkinsonism Relat Disord 23: , 1–9. |
[18] | Turcano P , Mielke MM , Josephs KA , Bower JH , Parisi JE , Boeve BF , Savica R ((2017) ) Clinicopathologic discrepancies in a population-based incidence study of parkinsonism in olmsted county: 1991-2010. Mov Disord 32: , 1439–1446. |




