Worsened Parkinson’s Disease Progression: Impact of the COVID-19 Pandemic
Abstract
Whilst some studies investigated the impact of viral infection or reduced access to medication during the COVID-19 pandemic in patients with Parkinson’s disease (PD), data on the effects of pandemic restrictions are still scarce. We retrospectively analyzed motor symptoms of longitudinally followed PD patients (n = 264) and compared motor disease progression before and during the COVID-19 pandemic. Additionally, we performed a trend analysis of the yearly evolution of motor symptoms in 755 patients from 2016 until 2021. We observed a worsening of motor symptoms and a significantly increased motor disease progression during pandemic-related restrictions as compared to before the COVID-19 outbreak.
INTRODUCTION
Parkinson’s disease (PD) represents a multisystem disorder with clinical key features pertaining to motor dysfunction. There is strong evidence that physiotherapy and physical activity with dedicated training improve not only levodopa-responsive but also axial motor symptoms [1]. For instance, a 2013 Cochrane review analyzed randomized controlled trials of physiotherapy versus no such intervention in patients with PD [2]. In this study comprising 39 trials with 1,827 patients, benefit for physiotherapy over up to 3 months was significant for clinician-rated Parkinson core motor symptoms, but also for gait velocity, freezing of gait, and postural stability. These are relevant symptoms related to ambulation of patients and quality of life. Additionally, data shows that COVID-19-dependent restrictions have negative effects on mental health [3] besides correlative evidence showing a worsening of non-motor features and potentially exacerbated motor symptoms in PD [4]. Further, a recent pilot study including mobile assessments to collect self-report data showed that it was physical activity that led to highest self-satisfaction in PD-patients during COVID-19 restrictions [5].
Therefore, it seems likely that the COVID-19 pandemic with all consecutive confinement strategies such as quarantine—leading to reduction of physical activity and physiotherapy, but also psychosocial challenges that may lead to psychiatric deterioration—negatively impacts progression of PD. In fact, during the first two waves of the COVID-19 pandemic in Switzerland, pharmacotherapy was always fully available, but many PD patients were partially or totally deprived of physiotherapy, and confinement further reduced their daily physical and social activities. In addition, fitness centers, dancing classes, and many other team sports that are popular among PD patients but also socio-cultural institutions (e.g., restaurants, art galleries) were closed over many months. This challenge has been discussed already early during the pandemic [6, 7] and has ever since been corroborated by the literature [3, 8–11]. For instance, a Dutch group found that the pandemic evokes psychological distress and lifestyle changes including reduced physical activity [12]. This is because most countries markedly reduced not only public life, but also canceled healthcare services including physiotherapy, with detrimental effects on chronically ill patients [8].
However, most of the present literature on COVID-19 and PD focuses on the impact of the infection itself and reduced access to PD medication on mortality, motor as well as non-motor symptoms [13–19]. For instance, one study found that patients with COVID-19 experienced new or worsening motor and non-motor symptoms [18]. It also found that PD patients without COVID-19 infection reported disrupted medical care, exercise, and social activities, and worsened motor and non-motor symptoms. Only few other studies focused on the impact of the pandemic restrictions on the course of PD. A questionnaire-based study from India found that home confinement worsened motor symptoms and quality of life in 38.5%of 832 PD patients [20]. In 100 PD patients from South Korea, another study found reduced exercise related to subjective worsening of PD symptoms [21]. Similarly, an Italian group observed that 60%of 74 PD patients reported a significant worsening of their general conditions during lockdown, the reduction of physical activity being the main risk factor for such change [22].
As the previous studies on the impact of pandemic restrictions on motor symptom progression were questionnaire-based and subjective, we aimed to retrospectively examine motor symptom severity and progression in PD patients during the pandemic and in the years before. We hypothesized that lack of physiotherapy and other physical training opportunities but also a social isolation-dependent increase of psychological stress during the pandemic crisis was mirrored by exacerbated motor disease severity and progression in PD patients.
MATERIALS AND METHODS
This is a retrospective analysis of data ascertained from the movement disorders outpatient unit in the Department of Neurology, University Hospital Zurich, Switzerland. All longitudinally followed patients with available scores of the third (motor) part of the Unified Parkinson Disease Rating Scale (UPDRS) of the International Parkinson and Movement Disorders Society (MDS) and with available entries regarding their levodopa equivalent dose (LED) during medication ON condition were included. The study was approved by the local ethics committee (“Kantonale Ethikkommission Zurich”). First, we aimed to conduct a descriptive analysis of the temporal trend of motor symptom severity: the data was resampled and represented as mean motor symptom scores per year from 2016 until 2021 (i.e., data was split into yearly time bins and the mean per time bin was calculated). The same procedure was applied to total LED. Next, motor disease progression was assessed by calculating the slope of a linearly fitted regression line through all data points per patient and timespan (i.e., slope of motor disease progression during 2018/2019 vs 2020/2021). To check whether a simple regression model was appropriate for the dataset, the residual values for each observation were plotted and inspected. For the analysis of disease progression, we included all PD patients who have had regular follow-up examinations in the years 2018/2019 and 2020/2021, and in whom a minimal observation time of at least two months in each timespan from September 2018 until December 2019 (16 months) and from January 2020 until April 2021 (16 months) was available.
Statistical testing was conducted using the built-in statistical libraries of Python (5.6.0, Python.org). To test whether there was a significant difference in motor symptom progression before as compared to during the pandemic crisis, the Wilcoxon signed-rank test was used. Significance was accepted at a p≤0.05-level.
RESULTS
Inspection of the yearly trend between 2016 and 2021 in 755 patients revealed exacerbated motor symptom severity during the pandemic crisis compared to before: whilst the mean MDS-UPDRS part III scores progressed only slowly from 2016 until the onset of the pandemic crisis (beginning of 2020), there is a steep, pharmacotherapy-independent increase from 2020 onwards (i.e., during the crisis) as the mean LED remained unchanged between 2016 and 2021 (Fig. 1A).
Fig. 1
A) Temporal trend analysis: Mean MDS-UPDRS part III scores of all PD patients (n = 755) resampled using a yearly time binning and comparing a time frame ranging from 2016–2021. B) Median (±SEM) motor disease progression as assessed by the slope (linearly fitted regression line through all data points per patient and timespan) of the MDS-UPDRS part-III score in each of 16 months before the crisis (until end of 2019) and 16 months during the crisis (until end of April 2021) of all PD patients who have had regular follow-up examinations during each timespan (n = 264).
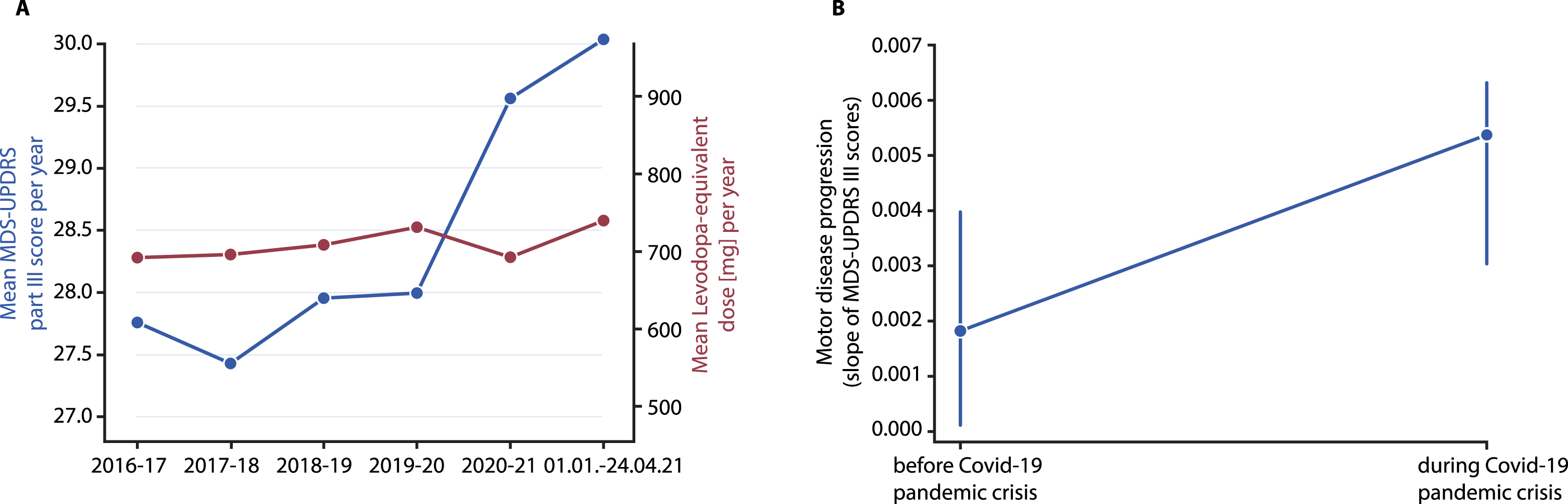
To investigate motor disease progression, we analyzed data from 264 PD-patients (Table 1). The sample consisted of 101 females and the mean disease duration was 14.5 years. Only 4 patients were diagnosed with a COVID-19 infection and in all of them, symptoms were benign, flu-like, and transient. Comparing motor disease progression before (September 2018 - December 2019, median (±SEM) observation time: 350±5.6 days, number of visits per period: 2–11) and during (January 2020 - April 2021, median (±SEM) observation time: 353±5.8 days, number of visits per period: 2–15) the pandemic crisis revealed a worsened progression during the crisis. This is reflected by the significantly increased slope of UPDRS part-III motor score progression during as compared to before the crisis (Z = –1.986, p = 0.047; Fig. 1B). This pertains to a median disease progression of 0.73 MDS-UPDRS part III points/year before the crisis as compared to a median increase of 1.82 points per year during the crisis. The axial-UPDRS-III score [23], indicating progression of axial motor symptoms, did not differ (p > 0.38).
Table 1
Characteristics of the study population
| PD patients (n = 264) | |
| Age (y; mean±SD) | 69.1±10.6 |
| Females (%) | 38.3 |
| Disease duration (y; mean±SD) | 14.5±7.5 |
| Axial UPDRS (mean±SD) | 6.1±4.3 |
| Number of patients with documented COVID-19 diagnosis | 4 |
DISCUSSION
This retrospective study reveals that motor symptom progression in PD patients in Switzerland as measured using the MDS-UPDRS III is worse during the months with pandemic restrictions comprising reduced physical activity and social isolation, compared to the same time period before the COVID-19 pandemic. This view of an increase in motor symptom progression is further reinforced when inspecting the temporal trend of yearly mean MDS-UPRDS III scores providing further evidence for a worsening of motor function during the time of confinement. Importantly, the increase in symptom progression is not due to a potential reduction in medication. These findings corroborate the assumption that discontinuity in exercise-based therapy and social contacts exerts detrimental effects on motor symptoms. Conversely, confinement-specific protective effects through mask-wearing and reduced social interactions led to substantially diminished influenza infections [24–26] with possibly overall improved health in the elderly. This is not reflected in the present data, which further suggests strong effects that seem to occur under conditions of containment leading to a worsened functional outcome. The fact that confinement and reduced medical services affect psychiatric health [3] but may also worsen progression of chronic diseases such as PD has consequences for the decision-making process of lawmakers and governmental bodies. Further, whilst treatments for improving functional outcomes through telemedicine offer opportunities to treat patients at home [8, 27–31], insurance bodies in many countries including Switzerland incomprehensibly require personal consultations for coverage of costs. More studies are needed to investigate whether telemedicine approaches can supplement traditional patient care [27, 32] and personal training.
This study has significant limitations, namely the retrospective setting and the assessment of MDS-UPDRS III in medication ON rather than OFF conditions. As physical daily activities and other contributing factors have not been measured in these patients, the study does not prove a causal link between confinement-dependent factors such as psychiatric status, activity level and worsened progression in PD. There is, however, evidence from qualitative studies suggesting an interrelation between physical activity and disease progression [14, 17, 19] and quantitative evidence that physical activity was significantly decreased during the stay-at-home mandate in PD-patients [5].
ACKNOWLEDGMENTS
This study was conducted in the absence of any financial support.
CONFLICT OF INTEREST
Christian R. Baumann received competitive grants from the Swiss National Science Foundation, the Hochschulmedizin Zurich (Flagship Grant), the Novartis Foundation, and Parkinson Schweiz, and unrestricted grants from AbbVie Pharma, and Roche; is founder and shareholder of Tossoo AG, which invests into non-pharmacological sleep modulation technologies; and received speaker honoraria from AbbVie Pharma. Heide Baumann-Vogel received a competitive grant from the Koetser Foundation Zurich. No other authors have any conflict of interest to report.
REFERENCES
[1] | Li F , Harmer P , Fitzgerald K , Eckstrom E , Stock R , Galver J , Maddalozzo G , Batya SS ((2012) ) Tai chi and postural stability in patients with Parkinson’s disease. N Engl J Med 366: , 511–519. |
[2] | Tomlinson CL , Patel S , Meek C , Herd CP , Clarke CE , Stowe R , Shah L , Sackley CM , Deane KH , Wheatley K , Ives N ((2013) ) Physiotherapy versus placebo or no intervention in Parkinson’s disease. Cochrane Database Syst Rev CD002817. |
[3] | Xiong J , Lipsitz O , Nasri F , Lui LMW , Gill H , Phan L , Chen-Li D , Iacobucci M , Ho R , Majeed A , McIntyre RS ((2020) ) Impact of COVID-19 pandemic on mental health in the general population: A systematic review. J Affect Disord 277: , 55–64. |
[4] | Antonini A , Barone P , Marconi R , Morgante L , Zappulla S , Pontieri FE , Ramat S , Ceravolo MG , Meco G , Cicarelli G , Pederzoli M , Manfredi M , Ceravolo R , Mucchiut M , Volpe G , Abbruzzese G , Bottacchi E , Bartolomei L , Ciacci G , Cannas A , Randisi MG , Petrone A , Baratti M , Toni V , Cossu G , Del Dotto P , Bentivoglio AR , Abrignani M , Scala R , Pennisi F , Quatrale R , Gaglio RM , Nicoletti A , Perini M , Avarello T , Pisani A , Scaglioni A , Martinelli PE , Iemolo F , Ferigo L , Simone P , Soliveri P , Troianiello B , Consoli D , Mauro A , Lopiano L , Nastasi G , Colosimo C ((2012) ) The progression of non-motor symptoms in Parkinson’s disease and their contribution to motor disability and quality of life. J Neurology 259: , 2621–2631. |
[5] | Templeton JM , Poellabauer C , Schneider S (2021) Negative effects of COVID-19 stay-at-home mandates on physical intervention outcomes: A preliminary study. J Parkinsons Dis, doi: 10.3233/JPD-212553 |
[6] | Papa SM , Brundin P , Fung VSC , Kang UJ , Burn DJ , Colosimo C , Chiang HL , Alcalay RN , Trenkwalder C ; MDS-Scientific Issues Committee ((2020) ) Impact of the COVID-19 pandemic on Parkinson’s disease and movement disorders. Mov Disord Clin Pract 7: , 357–360. |
[7] | Helmich RC , Bloem BR ((2020) ) The impact of the COVID-19 pandemic on Parkinson’s disease: Hidden sorrows and emerging opportunities. J Parkinsons Dis 10: , 351–354. |
[8] | Langer A , Gassner L , Flotz A , Hasenauer S , Gruber J , Wizany L , Pokan R , Maetzler W , Zach H ((2021) ) How COVID-19 will boost remote exercise-based treatment in Parkinson’s disease: A narrative review. NPJ Parkinsons Dis 7: , 25. |
[9] | Janiri D , Petracca M , Moccia L , Tricoli L , Piano C , Bove F , Imbimbo I , Simonetti A , Di Nicola M , Sani G , Calabresi P , Bentivoglio AR ((2020) ) COVID-19 pandemic and psychiatric symptoms: The impact on Parkinson’s disease in the elderly. Front Psychiatry 11: , 1306. |
[10] | Bek J , Groves M , Leventhal D , Poliakoff E ((2021) ) Dance at home for people with Parkinson’s during COVID-19 and beyond: Participation, perceptions, and prospects. Front Neurol 12: , 678124. |
[11] | Kelly MP , Leventhal D ((2021) ) Dance as lifeline: Transforming means for engagement and connection in times of social isolation. Health Promot Pract 22: (1 Suppl), 64S–69S. |
[12] | van der Heide A , Meinders MJ , Bloem BR , Helmich RC ((2020) ) The impact of the COVID-19 pandemic on psychological distress, physical activity, and symptom severity in Parkinson’s disease. J Parkinsons Dis 10: , 1355–1364. |
[13] | Cilia R , Bonvegna S , Straccia G , Andreasi NG , Elia AE , Romito LM , Devigili G , Cereda E , Eleopra R ((2020) ) Effects of COVID-19 on Parkinson’s disease clinical features: A community-based case-control study. Mov Disord 35: , 1287–1292. |
[14] | Fasano A , Cereda E , Barichella M , Cassani E , Ferri V , Zecchinelli AL , Pezzoli G ((2020) ) COVID-19 in Parkinson’s disease patients living in Lombardy, Italy. Mov Disord 35: , 1089–1093. |
[15] | Antonini A , Leta V , Teo J , Chaudhuri KR ((2020) ) Outcome of Parkinson’s disease patients affected by COVID-19. Mov Disord 35: , 905–908. |
[16] | Pavel A , Murray DK , Stoessl AJ ((2020) ) COVID-19 and selective vulnerability to Parkinson’s disease. Lancet Neurol 19: , 719. |
[17] | Brown EG , Chahine LM , Goldman SM , Korell M , Mann E , Kinel DR , Arnedo V , Kenneth ML , Tanner CM ((2020) ) The effect of the COVID-19 pandemic on people with Parkinson’s disease. J Parkinsons Dis 10: , 1365–1377. |
[18] | Cheong JLY , Goh ZHK , Marras C , Tanner CM , Kasten M , Noyce AJ , Movement Disorders Society Epidemiology Study Group ((2020) ) The impact of COVID-19 on access to Parkinson’s disease medication. Mov Disord 35: , 2129–2133. |
[19] | Vignatelli L , Zenesini C , Belotti L ((2020) ) Risk of hospitalization and death for COVID-19 in people with Parkinson’s disease or parkinsonism. Mov Disord 36: , 1–10. |
[20] | Kumar N , Gupta R , Kumar H , Mehta S , Rajan R , Kumar D , Kandadai RM , Desai S , Wadia P , Basu P , Mondal B , Sanchita , Rawat A , Meka SS , Mishal B , Prashanth LK , Srivastava AK , Goyal V ((2020) ) Impact of home confinement during COVID-19 pandemic on Parkinson’s disease. Parkinsonism Relat Disord 80: , 32–34. |
[21] | Song J , Ahn JH , Choi I , Mun JK , Cho JW , Youn J ((2020) ) The changes of exercise pattern and clinical symptoms in patients with Parkinson’s disease in the era of COVID-19 pandemic. Parkinsonism Relat Disord 80: , 148–151. |
[22] | Schirinzi T , Di Lazzaro G , Salimei C , Cerroni R , Liguori C , Scalise S , Alwardat M , Mercuri NB , Pierantozzi M , Stefani A , Pisani A ((2020) ) Physical activity changes and correlate effects in patients with Parkinson’s disease during COVID-19 lockdown. Mov Disord Clin Pract 7: , 797–802. |
[23] | Schreiner SJ , Imbach LL , Werth E , Poryazova R , Baumann-Vogel H , Valko PO , Murer T , Noain D , Baumann CR ((2019) ) Slow-wave sleep and motor progression in Parkinson disease. Ann Neurol 85: , 765–770. |
[24] | Hills T , Kearns N , Kearns C , Beasley R ((2020) ) Influenza control during the COVID-19 pandemic. Lancet 396: , 1633–1634. |
[25] | Soo RJJ , Chiew CJ , Ma S , Pung R , Lee V ((2020) ) Decreased influenza incidence under COVID-19 control measures, Singapore. Emerg Infect Dis 26: , 1933–1935. |
[26] | Olsen SJ , Azziz-Baumgartner E , Budd AP , Brammer L , Sullivan S , Pineda RF , Cohern C , Fry AM ((2020) ) Decreased influenza activity during the COVID-19 pandemic—United States, Australia, Chile, and South Africa, 2020. Am J Transplant 20: , 3681–3685. |
[27] | Hatcher-Martin JM , Adams JL , Anderson ER , Bove R , Burrus TM , Chehrenama M , Dolan O’Brien M , Eliashiv DS , Erten-Lyons D , Giesser BS , Moo LR , Narayanaswami P , Rossi MA , Soni M , Tariq N , Tsao JW , Vargas BB , Vota SA , Wessels SR , Planalp H , Govindarajan R ((2020) ) Telemedicine in neurology: Telemedicine Work Group of the American Academy of Neurology update. Neurology 94: , 30–38. |
[28] | De Marchi F , Contaldi E , Magistrelli L , Cantello R , Comi C , Mazzini L ((2021) ) Telehealth in neurodegenerative diseases: Opportunities and challenges for patients and physicians. Brain Sci 11: , 237. |
[29] | Ferreira D , Azevedo E , Araújo R ((2021) ) Teleneurology in Parkinson’s disease: A step-by-step video guide. Acta Neurol Scand 144: , 221–225. |
[30] | Quinn L , Macpherson C , Long K , Shah H ((2020) ) Promoting physical activity via telehealth in people with Parkinson disease: The path forward after the COVID-19 pandemic? Phys Ther 100: , 1730–1736. |
[31] | Ellis TD , Earhart GM ((2021) ) Digital therapeutics in Parkinson’s disease: Practical applications and future potential. J Parkinsons Dis 11: (s1), S95–S101. |
[32] | Shalash A , Spindler M , Cubo E ((2021) ) Global perspective on telemedicine for Parkinson’s disease. J Parkinsons Dis 11: (s1), S11–S18. |




