Physical Activity in Early Parkinson Disease
Abstract
Background:
Physical activity and exercise improve outcomes in Parkinson disease (PD), however little is known about activity levels in early PD patients.
Objective and Methods:
We examined self-reported activity scores and examined associations with clinical characteristics in 383 PD subjects and 175 healthy controls from the Parkinson Progression Markers Initiative (PPMI).
Results:
Activity scores were 8% lower for PD subjects than HC (162.6±86.2 vs 175.0±78.5, p = 0.10). Higher scores were associated with younger age and male sex. Only 47% of PD subjects and 44% of HC reported activity consistent with standard recommendations for adults.
Conclusions:
Our findings highlight the need to encourage exercise even in early PD.
INTRODUCTION
Physical activity is a modifiable lifestyle factor that may affect disease onset, severity, and progression in Parkinson disease (PD) [1]. Higher levels of midlife exercise, including strength, aerobic, and balance training, are associated with lower PD risk [2, 3], and PD patients who remain physically active have better health related quality of life [4] and lower rates of serious complications such as falls and fractures [5]. If activity improves outcomes in PD, then inactivity likely contributes to disability and represents an opportunity for intervention.
For these reasons, physical activity assessment is one component of the quality guidelines of the American Academy of Neurology for patients with PD [6]. Although guidelines for formal physical therapy programs in PD exist [7], there are currently no established targets for the prescription of general activity to PD patients. For all adults, the American Heart Association (AHA) recommends 150 minutes of moderate or 75 minutes of vigorous physical activity weekly, together with 2 days per week of strength training [8]. It is unclear whether an activity benchmark intended for the general population is achievable for PD patients with motor disability.
Despite increasing recognition of the importance of physical activity in disease management, little is known about prevailing activity habits in early PD. In moderate PD, activity is low, and near-sedentary behavior is associated with worsening disability [9, 10]. There is growing interest in activity levels in early disease, where interventions may be the most achievable and provide the longest potential benefit. Prior studies of activity in early PD [11, 12] are small and focus on walking rather than a range of physical activities. Here, we report the results of a cross-sectional analysis of baseline Physical Activity Scale in the Elderly (PASE) [13] scores and clinical correlates for deeply-phenotyped (with detailed clinical, neuropsychological, imaging and biomarker assessments) PD patients and healthy controls (HC) enrolled in the Parkinson Progression Markers Initiative (PPMI) [14], a large international multicenter cohort study of early PD patients.
METHODS
Subjects/assessments
Data were obtained from the PPMI database (http://www.ppmi-info.org/data), from centers located in the United States, Europe, Israel and Australia. Patients were enrolled within two years of a diagnosis of PD; HC were neurologically normal individuals recruited via local community and media outreach from the same movement disorders centers to provide a population norm. Individuals undergo regular assessments of cognitive, motor, and psychiatric function; biomarker data from serum, CSF, and imaging are also included in the database.
Data were downloaded on March 3, 2017. Physical activity levels were assessed using the PASE, which was added to PPMI in 2013 and queries frequency, intensity, and duration of time spent on leisure activity, household chores, and work/volunteer activity over the preceding week. Self-reported PASE scores are well-validated against objective measures of activity [15, 16] and can be compared to age- and gender-matched population norms. We selected PD and HC who had completed at least one PASE in the first year after introduction of the questionnaire. For those who had completed multiple PASE questionnaires, only the first score was used. We identified 387 PD subjects and 176 HC meeting these criteria. For these individuals, we acquired demographic characteristics, cognitive scores, motor and non-motor symptoms, and biomarker data, within one year of their initial PASE questionnaire.
Data analysis
Our primary outcome measure was the PASE score, computed using the PASE Administration and Scoring Manual [New England Research Institute, Watertown MA, USA]. The raw PASE data allows conversion of activity frequency, duration, and intensity into a METS-based total score, where higher scores indicate more exercise engagement; scores range from 0 to 500. Subscores for household chores and leisure activities were also calculated; the work-related item is not included in either subscore. We also examined the reported frequency and duration for moderate activities, vigorous activities, and strength training. Individuals who reported at least two days per week of strength training, together with either 150 minutes weekly moderate activity or 75 minutes of weekly vigorous activity, were considered to be adherent to AHA guidelines. All other individuals were coded as not adherent.
Because an initial inspection revealed that PASE scores for HC were higher than previously published population norms, we audited all data, blinded to diagnosis, and recoded when necessary, according to the PASE scoring manual. If either the PASE manual did not provide guidance or the participant did not report specific types of activities, the raw scores were not changed. Twenty percent of scores were recoded; the mean score difference between raw and recoded scores was 3.3 points for PD subjects and 4.5 points for HC; recoded scores were within 2.5% of raw scores. Of those who required recoding, 47.7% had over-reported activity and 52.3% had under-reported activity. Analysis used only the recoded scores. Auditing also revealed five PD subjects and one control who grossly over-reported activity (i.e. daily activity duration exceeded 24 hours). These individuals were excluded from further analysis.
Data were analyzed using SPSS version 20 [IBM, Cary NC, USA]. We performed t-tests comparing PD subjects with age- and gender-matched HC. We also conducted linear and logistic regression analyses on PD subjects to identify clinical characteristics associated with higher levels of physical activity and adherence to AHA guidelines, respectively.
RESULTS
The mean age [SD] of PD subjects was 61.6 (9.7) compared to 61.4 (11.0) for HC (p = 0.83). Of PD subjects, 65.5% were male, compared to 62.3% of HC (p = 0.46). Education levels were similar between the two groups (15.6 years for PD, 16.0 years for HC, p = 0.11). PD subjects had an average disease duration of 16.6 [SD 7.7] months at the time of first PASE, and most (n = 363, 95.5%) were unmedicated.
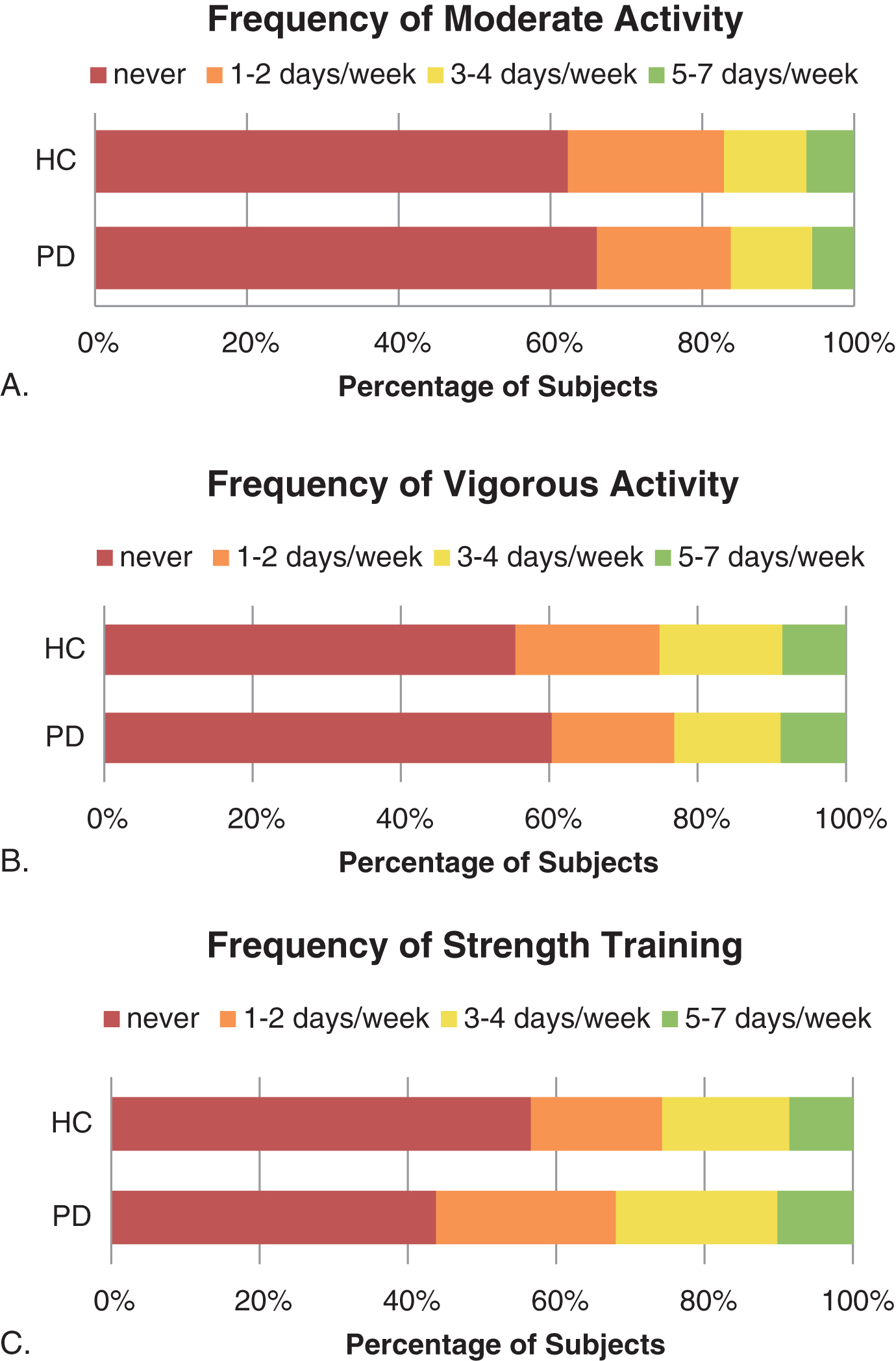
Mean PASE scores [SD] are shown in Table 1. Total PASE was slightly lower in PD subjects compared to HC. In subgroup analysis, this difference was driven largely by women over the age of 70, where mean PASE for PD subjects was 127.0 (69.0) compared to 207.1 (83.1) for controls (p = 0.0043). Excluding women over 70, the mean PASE was similar between PD subjects and HC (206.4 (94.6) vs. 208.8 (77.3), p = 0.784). There were no significant differences in household or leisure PASE subscores between PD and HC. Further analysis of moderate, vigorous, and strengthening activities (Fig. 1) showed that a higher proportion of PD subjects than HC participated in strength-building at least one day per week (58.5% vs 43.4%, p = 0.0004); there was no difference between groups for moderate activities (37.3% vs 37.7%, p = 0.928) or vigorous activities (42.8% vs 44.5%, p = 0.707).
Table 1
PASE scores (mean [standard deviation])
| PD (n = 383) | HC (n = 175) | p | ||
| Age/Gender | All | 162.6 (86.2) | 175.0 (78.5) | 0.10 |
| Men <65 | 227.2 (92.6) | 216.36 (86) | 0.397 | |
| Men 65–69 | 181.3 (94.9) | 206.98 (72.6) | 0.212 | |
| Men >70 | 155.8 (72.2) | 161.0 (57.0) | 0.747 | |
| Women <65 | 198.3 (90.9) | 211.2 (69.7) | 0.380 | |
| Women 65–69 | 166.3 (89.5) | 170.7 (56.4) | 0.859 | |
| Women >70 | 127.0 (69.0) | 207.1 (83.1) | 0.005 | |
| Activity Type | Household | 118.3 (76.0) | 126.6 (66.2) | 0.194 |
| Leisure | 43.3 (43.5) | 44.2 (41.8) | 0.812 |
Fig.1
Percentage of subjects reporting Moderate, Vigorous, and Strength-based activities, by frequency (A-C).
In linear regression analysis for PD subjects, higher PASE was associated with younger age (B = 0.412, p < 0.0001) and male gender (B = 0.124, p = 0.009). There were trends toward association of PASE with modified Schwab and England scores (B = 0.080, p = 0.088) and inverse association with depression (B = –0.083, p = 0.077). Other clinical characteristics that were not significantly associated with PASE included Montreal Cognitive Assessment, Epworth Sleepiness Scale, Hoehn & Yahr, UPDRS-III, Scopa-Autonomic, education level, or body mass index (data not shown).
The proportion of individuals meeting AHA activity guidelines was not significantly different between PD subjects (47%) and HC (44%) (p = 0.588). There were no gender differences in rates of adherence to activity guidelines (p = 0.113 for PD subjects, p = 0.826 for HC). In logistic regression analysis among PD subjects, AHA adherence increased with age (B = 0.954, p = 0.015), and there was a trend toward association with body mass index (B = 0.933, p = 0.089). Other clinical characteristics, including modified Schwab & England, Geriatric Depression Score, Montreal Cognitive Assessment, Epworth Sleepiness Score, Hoehn & Yahr, UPDRS-III, Scopa-Autonomic, and education level, were not significantly associated with AHA adherence (data not shown).
DISCUSSION
We found a trend toward slightly lower (8%, p = 0.1) activity levels in early PD subjects and similar adherence to AHA activity guidelines compared to matched controls. This suggests that early PD patients can maintain physical activity levels comparable to healthy peers. This finding is supported by small randomized trials demonstrating that PD patients are capable of increasing physical activity early in the disease [17–20]. However, the modest disparity in activity levels observed in this early cohort is likely to increase as the disease and motor disability progress [12, 21], highlighting the need for exercise counseling early in the disease course. Additionally, adequate symptomatic management at all stages likely carries the benefit of increased physical activity, which may further improve both motor and non-motor symptoms. In turn, the sometimes advocated delay or limitation of dopaminergic replacement therapy may further worsen disability by decreasing activity levels.
Apart from age and gender, other clinical characteristics were not associated with physical activity at baseline, suggesting that all patient groups would benefit from increased physical activity. Older patients, especially women, may be particularly vulnerable to inactivity and its attendant complications, including falls, fractures, and loss of independence. Interestingly, we identified a trend toward higher rates of AHA adherence with increasing body mass index, which could be related to additional exercise counseling for overweight individuals. Further studies are needed to identify the specific motivators and barriers to exercise among patients with PD; these can be used to design targeted activity interventions [10].
Our findings are consistent with prior data that physical activity levels do not meet recommended targets even for early PD [10]. However, most studies of physical activity in PD focus on walking behaviors, while the PASE queries multiple types of physical activity and provides a fuller picture of weekly fitness habits. The use of a validated instrument to measure physical activity in a large, deeply-phenotyped cohort of PD patients and matched controls are additional strengths of the current study. Nevertheless, some important limitations should be acknowledged. The PPMI cohort is highly motivated and may not be representative of the wider PD population. In particular they may be more likely to exercise, which could explain why PASE scores were higher than previously published population norms. The PASE questionnaire is self-administered and retrospective, and is therefore subject to bias. However, PASE scores are strongly correlated with objective measures of physical activity [15, 16]. We attempted to mitigate miscategorization of activity intensity by auditing subject-reported data against the official administration and scoring manual. Since not all subjects listed specific activities, and since some activities are not categorized in the scoring manual, it remains possible that some subject-reported scores still overestimated or underestimated true activity. Additionally, the self-reported nature of PASE prevents clarification of activity frequency or duration; we identified a few individuals who self-reported daily activities exceeding 24 hours, but we cannot verify that others did not also overreport or underreport activity frequency or duration. However, among scores that required auditing, the magnitude of change was small and subjects were equally likely to over or under-code their activity. Lastly, our cross-sectional study examined only initial PASE and clinical data, as the PASE was added as an assessment after the inception of the PPMI cohort and follow-up data are currently limited. The longitudinal design of PPMI will allow future studies to examine changes in physical activity as well as any potential association between baseline activity levels and clinical manifestations or biomarkers of disease progression.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The views expressed in this article are those of the authors and do not necessarily reflect the position or policy of the Department of Veterans Affairs or the United States government.
PPMI – a public-private partnership – is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners Abbvie, Avid Radiopharmaceuticals, Biogen Idec, BioLegend, Bristol-Myers Squibb, Eli Lilly & Co., F. Hoffman-La Roche, Ltd., GE Healthcare, Genentech, GlaxoSmithKline, Lundbeck, Merck, MesoScale Discovery, Piramal, Pfizer, Sanofi Genzyme, Servier, Takeda, Teva, and UCB.
REFERENCES
[1] | Ahlskog JE ((2011) ) Does vigorous exercise have a neuroprotective effect in Parkinson disease? Neurology 77: , 288–294. |
[2] | Thacker EL , Chen H , Patel AV , McCullough ML , Calle EE , Thun MJ , Schwarzschild MA , Ascherio A ((2008) ) Recreational physical activity and risk of Parkinson’s disease. Mov Disord 23: , 69–74. |
[3] | Xu Q , Park Y , Huang X , Hollenbeck A , Blair A , Schatzkin A , Chen H ((2010) ) Physical activities and future risk of Parkinson disease. Neurology 75: , 341–348. |
[4] | Rafferty MR , Schmidt PN , Luo ST , Li K , Marras C , Davis TL , Guttman M , Cubillos F , Simuni T ((2017) ) Regular exercise, quality of life, and mobility in Parkinson’s disease: A longitudinal analysis of national parkinson foundation quality improvement initiative data. J Parkinsons Dis 7: , 193–202. |
[5] | Canning CG , Sherrington C , Lord SR , Close JCT , Heritier S , Heller GZ , Howard K , Allen NE , Latt MD , Murray SM , O’Rourke SD , Paul SS , Song J , Fung VSC ((2015) ) Exercise for falls prevention in Parkinson disease. Neurology 84: , 304–312. |
[6] | Factor SA , Bennett A , Hohler AD , Wang D , Miyasaki JM ((2016) ) Quality improvement in neurology: Parkinson disease update quality measurement set Executive summary. Neurology 86: , 2278–2283. |
[7] | Keus SHJ , Bloem BR , Hendriks EJM , Bredero-Cohen AB , Munneke M , on behalf of the Practice Recommendations Development Group ((2007) ) Evidence-based analysis of physical therapy in Parkinson’s disease with recommendations for practice and research. Mov Disord 22: , 451–460. |
[8] | Nelson ME , Rejeski WJ , Blair SN , Duncan PW , Judge JO , King AC , Macera CA , Castaneda-Sceppa C , American College of Sports Medicine, American Heart Association ((2007) ) Physical activity and public health in older adults: Recommendation from the American College of Sports Medicine and the American Heart Association. Circulation 116: , 1094–1105. |
[9] | Ellis T , Motl RW ((2013) ) Physical activity behavior change in persons with neurologic disorders: Overview and examples from Parkinson disease and multiple sclerosis. J Neurol Phys Ther 37: , 85–90. |
[10] | Ellis T , Boudreau JK , DeAngelis TR , Brown LE , Cavanaugh JT , Earhart GM , Ford MP , Foreman KB , Dibble LE ((2013) ) Barriers to exercise in people with Parkinson disease. Phys Ther 93: , 628–636. |
[11] | Lord S , Godfrey A , Galna B , Mhiripiri D , Burn D , Rochester L ((2013) ) Ambulatory activity in incident Parkinson’s: More than meets the eye? J. Neurol 260: , 2964–2972. |
[12] | Cavanaugh JT , Ellis TD , Earhart GM , Ford MP , Foreman KB , Dibble LE ((2012) ) Capturing ambulatory activity decline in Parkinson’s disease. J Neurol Phys Ther 36: , 51–57. |
[13] | Washburn RA , Smith KW , Jette AM , Janney CA ((1993) ) The Physical Activity Scale for the Elderly (PASE): Development and evaluation. J Clin Epidemiol 46: , 153–162. |
[14] | Parkinson Progression Marker Initiative ((2011) ) The Parkinson Progression Marker Initiative (PPMI). Prog Neurobiol 95: , 629–635. |
[15] | Schuit AJ , Schouten EG , Westerterp KR , Saris WH ((1997) ) Validity of the Physical Activity Scale for the Elderly (PASE): According to energy expenditure assessed by the doubly labeled water method. J Clin Epidemiol 50: , 541–546. |
[16] | Washburn RA , Ficker JL ((1999) ) Physical Activity Scale for the Elderly (PASE): The relationship with activity measured by a portable accelerometer. J Sports Med Phys Fitness 39: , 336–340. |
[17] | Uc EY , Doerschug KC , Magnotta V , Dawson JD , Thomsen TR , Kline JN , Rizzo M , Newman SR , Mehta S , Grabowski TJ , Bruss J , Blanchette DR , Anderson SW , Voss MW , Kramer AF , Darling WG ((2014) ) Phase I/II randomized trial of aerobic exercise in Parkinson disease in a community setting. Neurology 83: , 413–425. |
[18] | Schenkman M , Hall DA , Barón AE , Schwartz RS , Mettler P , Kohrt WM ((2012) ) Exercise for people in early- or mid-stage Parkinson disease: A 16-month randomized controlled trial. Phys Ther 92: , 1395–1410. |
[19] | Corcos DM , Robichaud JA , David FJ , Leurgans SE , Vaillancourt DE , Poon C , Rafferty MR , Kohrt WM , Comella CL ((2013) ) A two-year randomized controlled trial of progressive resistance exercise for Parkinson’s disease. Mov Disord 28: , 1230–1240. |
[20] | Kelly NA , Ford MP , Standaert DG , Watts RL , Bickel CS , Moellering DR , Tuggle SC , Williams JY , Lieb L , Windham ST , Bamman MM ((2014) ) Novel, high-intensity exercise prescription improves muscle mass, mitochondrial function, and physical capacity in individuals with Parkinson’s disease. J Appl Physiol (1985) 116: , 582–592. |
[21] | Cavanaugh JT , Ellis TD , Earhart GM , Ford MP , Foreman KB , Dibble LE ((2015) ) Toward understanding ambulatory activity decline in Parkinson disease. Phys Ther 95: , 1142–1150. |



