Initiation of Levodopa-Carbidopa Intestinal Gel Infusion Using Telemedicine (Video Communication System) Facilitates Efficient and Well-Accepted Home Titration in Patients with Advanced Parkinson’s Disease
Abstract
Background:
Levodopa-carbidopa intestinal gel (LCIG; Duodopa®) is used for continuous infusion in advanced Parkinson’s disease. To achieve optimal effect, the LCIG dose is individually titrated, traditionally conducted during hospitalization in Sweden. However, dose adjustment depends on surrounding conditions, physical activity, and emotional stress, which is why titration at home could be beneficial. Telemedicine (TM) using a video communication system offers alternative titration procedures, allowing LCIG initiation at home.
Objective:
Study objectives were to show the feasibility of TM for LCIG home titration, evaluate resource use, and assess patient, neurologist, and nurse satisfaction.
Methods:
Four clinics enrolled 15 patients to observe efficiency and feasibility of TM-based monitoring.
Results:
Patient median (range) age was 67 (52–73) years and time since diagnosis was 10 (7–23) years. Median time between LCIG initiation and end of TM-assisted titration was 2.8 (2.0–13.8) days. Median time required for home titration by neurologists, nurses, and patients was (hours:minutes) 1 : 14 (0 : 29–1 : 52), 5 : 49 (2 : 46–10 : 3), and 8 : 53 (4 : 11–14 : 11), respectively. Neurologists and nurses considered this to be less time than required for hospital titration. TM allowed patients 92% free time from start to end of titration. Technical problems associated with TM contacts were rare, mostly related to digital link, and quickly resolved. Patients, neurologists, and nurses were satisfied using TM. No serious adverse events were reported; there was one device complaint (tube occlusion).
Conclusions:
In this study, TM-assisted LCIG titration at home was resource-efficient, technically feasible, well-accepted and was deemed satisfactory by patients, neurologists, and nurses.
INTRODUCTION
Parkinson’s disease (PD) is a progressive neurodegenerative disorder leading to increasing impairments of mobility and quality of life (QoL) over time [1, 2]. Levodopa, a dopamine precursor, is the most effective symptomatic therapy for PD [3]. Nevertheless, complications such as motor and non-motor response fluctuations progressively develop over time, becoming a major source of disability, and substantially interfere with daily activities and social interactions [4]. Continuous duodenal/jejunal infusion of levodopa-carbidopa helps stabilize the levodopa plasma concentration compared with oral levodopa therapy [5].
Levodopa-carbidopa intestinal gel (LCIG) is a stable gel suspension of levodopa-carbidopa (20/5 mg/mL) for continuous delivery in patients with advanced PD via a portable pump into the duodenum through a percutaneous endoscopic gastrostomy with a duodenal/jejunal extension tube (PEG-J). Motor symptoms can be better controlled with infusion compared with conventional oral treatment, and LCIG represents an effective option for advanced PD [5]. LCIG infusion has been shown to reduce motor fluctuations, dyskinesia, and non-motor symptoms and increase QoL in randomized controlled, [7] open-label, [8–10] and long-term studies [11, 12].
Until June 2014, the EU LCIG summary of product characteristics (SmPC) required a positive response when testing LCIG using a nasojejunal tube prior to placement of a permanent PEG-J. This NJ titration is the clinical practice in Sweden as well as several other European countries. The length of this test infusion and titration period, varies from 3 to 14 days, with an estimated average of 6 to 8 days [8, 11]. In a common titration scheme, patients come to the hospital on Monday and stay for active titration until Friday. Individual dose titration at initiation of LCIG treatment depends on daily physical activity, emotional state, and stress level, which may be different when patients are admitted to the hospital compared with home. As a consequence, patients may require further LCIG dose adjustments when they return home. Therefore, initiation and titration of LCIG infusion at home could be beneficial and more convenient for patients, shorten the overall time for dose adjustments, and reduce healthcare resource utilization (and thereby costs). In addition, waiting lists for hospital beds and requirement of specific resources for an inpatient stay may delay initiation of LCIG treatment. Telemedicine (TM) represents an opportunity to individualize patient support and care while the patient is at home. It benefits patients and relatives living far away from the hospital and the hospital in lower costs and simpler overall logistics when the patient does not need to be hospitalized. It also keeps the risks of infections low. The use of interactive video conferencing was reported in the 1990s [13, 14], for online treatment and care of nursing home residents with PD [15, 16], for office-based and home web-based clinical assessments for PD [17, 18], and recent reviews summarizing the experience of TM in care of PD patients [19, 20].
Telemedicine offers an alternative procedure for initiation of LCIG treatment at home for motivated patients [21]. The objectives of this study were to show the feasibility of TM for LCIG home titration, evaluate use of resources, and assess patient, neurologist, and nurse satisfaction.
MATERIAL AND METHODS
This observational study was conducted at 4 Swedish University Hospitals. The protocol, patient information, and informed consent were approved by the regional ethical review board in Stockholm. Patients with advanced PD and motor complications eligible for LCIG treatment according to the Duodopa SmPC were included. Patients had to be motivated and confident to test LCIG home titration using TM and able to handle the infusion pump and the TM equipment by themselves or with assistance. Patients with dementia, cognitive decline, or ongoing symptomatic depression, hallucinations, or other psychotic behaviors without adequate treatment were excluded. Demographic data and medical history (concomitant PD treatments, vital signs) were recorded at inclusion. Serious adverse events and device complaints were recorded throughout the LCIG titration period.
Patients received the NJ tube (Covidien®, Dual Port Feeding Tube with Flow-Through Stylet, Kendall Entriflex) using fluoroscopic passive/active positioning in the hospital according to regular Swedish clinical practice. When the J-tube position was confirmed radiologically, the patient returned home accompanied by the DNS. Dose titration was performed according to clinical practice, essentially as described in Fernandez et al. [22]. Oral daytime levodopa was immediately replaced by LCIG. Individually-optimized dosing of LCIG was delivered over a 16-h period, administered as a morning bolus followed by continuous infusion, and if needed, intermittent extra doses (patient-initiated based on symptom experience). The volume of the morning bolus was individualized for each patient based initially on the previous total oral levodopa dose (Levodopa dose equivalents). The total morning dose was usually 5–10 mL, corresponding to 100–200 mg levodopa, and did not exceed 20 mL (400 mg levodopa). Extra doses were adjusted individually during the titration period. End of titration was defined as the time when the investigator decided on PEG/J placement. After this time point there was no study record of dose adjustments or communication. In clinical practice, dose changes +/–10% are fairly common during the first month after titration.
Telemedicine was based on video communication. Doctors’ equipment included a Cisco C40 codec with premium resolution and dual monitor options, an Atea 12” iCtrl w Duodopa GUI touch screen, 2 NEC Multisync EA273WM monitors, a Canton Media active speaker, and a Cisco PHD1080P camera with 4x zoom. Patient equipment included a Cisco SX20 codec, an Atea 10” iCtrl w Duodopa GUI touch screen, a NEC Multisync EA273WM monitor, a Canton Media active speaker, and a Cisco PHD1080P camera with 12x zoom. For Internet access, patient TM systems included a Dovado 4G router and an ICON 8014G/LTE USB modem. The Duodopa nurse specialist (DNS) TM system included a Cisco Jabber PC client, a Cisco Telepresence Personal Cam, and Jabber headset. Estimated total cost of this specific system is approximately 24 000 EUR (2014 prices). TM systems were installed by a technician before the LCIG titration period at each patient’s home and dismounted thereafter. Patient instructions were provided by the technician during TM system setup. If required, further support was provided by technicians or DNS during the home titration period.
The TM-assisted LCIG home titration period was defined as the start of the pump to the decision for permanent PEG-J surgery or termination of LCIG treatment. No assessments after this time point were recorded in the study. Assessed resource utilizations included the overall time for TM required by patients, neurologist, DNS, and technician and the number and duration of contacts per type of contacts (e.g., patient/neurologist, patient/DNS, patient/neurologist/DNS, and patient/technician). The time required by the technician to install and dismount the TM system at a patient’s home as well as pre-start training by DNS was recorded separately. Travel time for home visits conducted by DNS and technicians was assessed separately. Patients recorded time for independent pump handling in a diary. Patient free time was calculated as time from start to end of study and subtraction of all time required for contact with doctor, nurse, or technician and time for independent pump handling. Based on these data, the duration of patient free time and the percentage of patient free time during the LCIG titration period were calculated.
To assess technical feasibility, all technical events and their consequences were reported. If a scheduled TM contact failed, a TM contact was delayed, re-establishment of connection was required, or a TM session had to be replaced by a phone call, the underlying technical events were allocated to one of the following categories: TM digital link problems (failure to establish connection, interruptions, transmission quality, sound quality, image quality, or other), TM equipment (mishandling, failure to answer video call, or other), technical problems (mechanical, electrical, or other) or intentional misuse (intentional failure to answer video call or other).
Clinical Global Impression of Improvement (CGI-I) between start of LCIG infusion and end of TM home titration was evaluated by the neurologist using a 7-point Likert scale. Experiences of the patient and caregiver (when existing, either spouse [11] or professional care at a residential care facility [1]) with LCIG home titration using TM was collected using a semi-structured interview (4 questions and 12 items with a 7-point Likert scale and additional yes/no questions). The neurologist and DNS reported their experiences in a web-based questionnaire (4 questions and 8 and 9 items, respectively, with a 7-point Likert scale and additional yes/no questions). Additional free-text qualitative comments were documented separately.
Statistical analyses were performed using SAS® version 9.3 (SAS Institute, Cary, NC). Continuous data were summarized using descriptive statistics (mean, standard deviation, minimum, median and maximum) and categorical data were presented using frequency and percentage. Percentages were based on number of non-missing observations. Results are presented as medians (minimum-maximum), as values showed no normal distribution.
RESULTS
15 out of 48 eligible patients were enrolled in the study. Enrollment was “consecutive after interest”, i.e., motivation for participation was crucial. The most common reasons for exclusion from study participation were cognitive decline, low autonomy, too severe illness and lack of motivation to test TM. The first TM patient was enrolled in September 2013; through November 2014. Demographics and medical history of the 15 patients (13 males and 2 females) are summarized in Table 1. The median (minimum-maximum) age was 67 (52–73) years and median time since PD diagnosis was 10 (7–23) years.
Table 1
Demographics and medical history
| Demographics, n (%) | |
| Gender | |
| Female | 2 (13.3) |
| Male | 13 (86.7) |
| Age, years | 67 (52–73) |
| Time since PD diagnosis (years) | 10 (7–23) |
| Hoehn and Yahr (OFF stage), n (%) | |
| 1 | 0 |
| 2 | 1 (6.7) |
| 3 | 9 (60.0) |
| 4 | 4 (26.7) |
| 5 | 1 (6.7) |
| Caregiver during home titration | |
| No | 3 (20.0) |
| Yes | 12 (80.0) |
Data presented in median (minimum-maximum) or number (%).
The number and duration of contacts and overall time required by patients, neurologists, DNS, and technician for TM-assisted LCIG home titration is shown in Table 2. The median total time for any kind of contact required by patients, neurologists, and DNS during TM-assisted LCIG home titration was (hours:minutes) 8 : 53 (4 : 11–14 : 38), 1 : 14 (0 : 29–1 : 52), and 5 : 49 (2 : 46–10 : 03), respectively. The median total number of contacts per patient was 19 (12–37): 13 (68.4%) by TM, 1 (5.3%) by phone, and 5 (26.3%) home visits. The median number or TM contacts between neurologist and patient without DNS were 2. The DNS executed the majority of contacts using the TM, some by home visits and occasionally by phone.
Table 2
Time and contacts required for TM-assisted home titration
| Total TM Time | Total | per Type of contact | ||
| TM | Telephone | Home visit | ||
| Patient | ||||
| Total time (hours:minutes) | 8 : 53 (4 : 11–14 : 38) | 1 : 40 (0 : 43–2 : 51) | 0 : 16 (0 : 5–1 : 20) | 5 : 45 (2 : 45–11 : 17) |
| Neurologist | ||||
| Total time (hours:minutes) | 1 : 14 (0 : 29–1 : 52) | 0 : 57 (0 : 29–1 : 52) | 0 : 37 (0 : 37–0 : 37) | N/A |
| DNS | ||||
| Total time (hours:minutes) | 5 : 49 (2 : 46–10 : 03) | 1 : 19 (0 : 41–2 : 12) | 0 : 16 (0 : 01–0 : 50) | 4 : 41 (1 : 15–7 : 17) |
| TM Contacts | Total | per Type of contact | ||
| TM | Telephone | Home visit | ||
| Patient and neurologist | ||||
| Number of contacts | 3 (0–5) | 2 (0–5) | 0 (0–3) | 0 (0-0) |
| Overall duration (hours:minutes) | 0 : 29 (0 : 3–0 : 39) | 0 : 22 (0 : 3–0 : 39) | 0 : 30 (0 : 30–0 : 03) | N/A |
| Duration per contact (hours:minutes) | 0 : 08 (0 : 03–0 : 16) | 0 : 06 (0 : 03–0 : 16) | 0 : 10 (0 : 10-0 : 10) | N/A |
| Patient and DNS | ||||
| Number of contacts1 | 9 (5–22) | 5 (2–12) | 1 (0–7) | 3 (1–4) |
| Overall duration(1hours:minutes) | 5 : 17 (2 : 10–9 : 18) | 0 : 23 (0 : 10–1 : 37) | 0 : 16 (0 : 1–0 : 46) | 4 : 41 (1 : 15–7 : 17) |
| Duration per contact (hours:minutes) | 0 : 34 (0 : 13–0 : 54) | 0 : 7 (0 : 3–0 : 14) | 0 : 5 (0 : 1–0 : 8) | 1 : 29 (0 : 40–2 : 26) |
| Patient, neurologist, and DNS | ||||
| Number of contacts | 5 (2–11) | 5 (2–11) | 0 (0-1) | 0 (0-0) |
| Overall duration (hours:minutes) | 0 : 45 (0 : 22–1 : 17) | 0 : 42 (0 : 22–1 : 17) | 0 : 07 (0 : 07-0 : 07) | N/A |
| Duration per contact (hours:minutes) | 0 : 09 (0 : 06–0 : 14) | 0 : 09 (0 : 05–0 : 14) | 0 : 7 (0 : 07-0 : 07) | N/A |
| Patient and technician | ||||
| Number of contacts | 2 (1–3) | 0 (0-1) | 0 (0-1) | 2 (1–3)2 |
| Overall duration (hours:minutes) | 1 : 40 (1 : 0–4 : 0) | 0 : 02 (0 : 02–0 : 02) | 0 : 04 (0 : 04–0 : 04) | 1 : 40 (1 : 0–4 : 0)2 |
| Duration per contact (hours:minutes) | 60 (44–105) | 2 (2-2) | 4 (4-4) | 63.2 (45–105) |
| Patient (all contacts): | ||||
| Total number of contacts | 19 (12–37) | 13 (7–23) | 1 (0–11) | 5 (3–6) |
| Duration per contact (hours:minutes) | 0 : 24 (14–43) | 0 : 07 (5–12) | 0 : 05 (2–8) | 1 : 16 (0 : 46–2 : 9) |
| Travel and visit time for installing, training, technical support and dismounting TM | ||||||
| Before TM | During TM | After TM | ||||
| Technician Travel | Installment | Visit | Technical Travel3 | Support Visit3 | Demounting Travel | Visit |
| Time | 1 : 10 | 0 : 15 | 0 : 0 | 0 : 0 | 1 : 20 | 0 : 30 |
| (hours:minutes) | (0 : 0– 9 : 0) | (0 : 0– 2 : 30) | (0 : 0– 6 : 0) | (0 : 0– 1 : 20) | (0 : 0– 9 : 0) | (0 : 0– 1 : 0) |
| DNS | TM training Travel | Visit4 | Home visit Travel | Visit | ||
| Time | 1 : 20 | 0 : 30 | 2 : 10 | 3 : 58 | ||
| (hours:minutes) | (0 : 0– 6 : 5) | (0 : 0– 3 : 43) | (0 : 0– 7 : 0) | (1 : 5– 6 : 39) | ||
Data presented in median and minimum-maximum numbers and hours: minutes. DNS, Duodopa nurse specialist; TM, telemedicine. 1Contacts and time required before and during home titration. 2Contacts and time required for installing and dismounting TM system at patient’s home. 3One home support visit by technician: 360 minutes travelling and 80-minute visit. 4TM training by DNS.
The number of contacts and time required by patients, neurologists, and DNS per day and per contact and time for installing, training and dismounting the TM equipment are shown in Fig. 1a and Table 2. When comparing to their own experience from previous titrations, 60% of the neurologists and 93% of the nurses considered the time spent for patient contact during TM-assisted titration to be less than during hospital titration.
Fig.1
Use of video communication system. (a) Total time required per day by patient, neurologist, and DNS during TM home titration and number of patients on TM-assisted home titration over time. (b) Type and number of consequences of recorded technical events. DNS, Duodopa nurse specialist; TM, telemedicine.
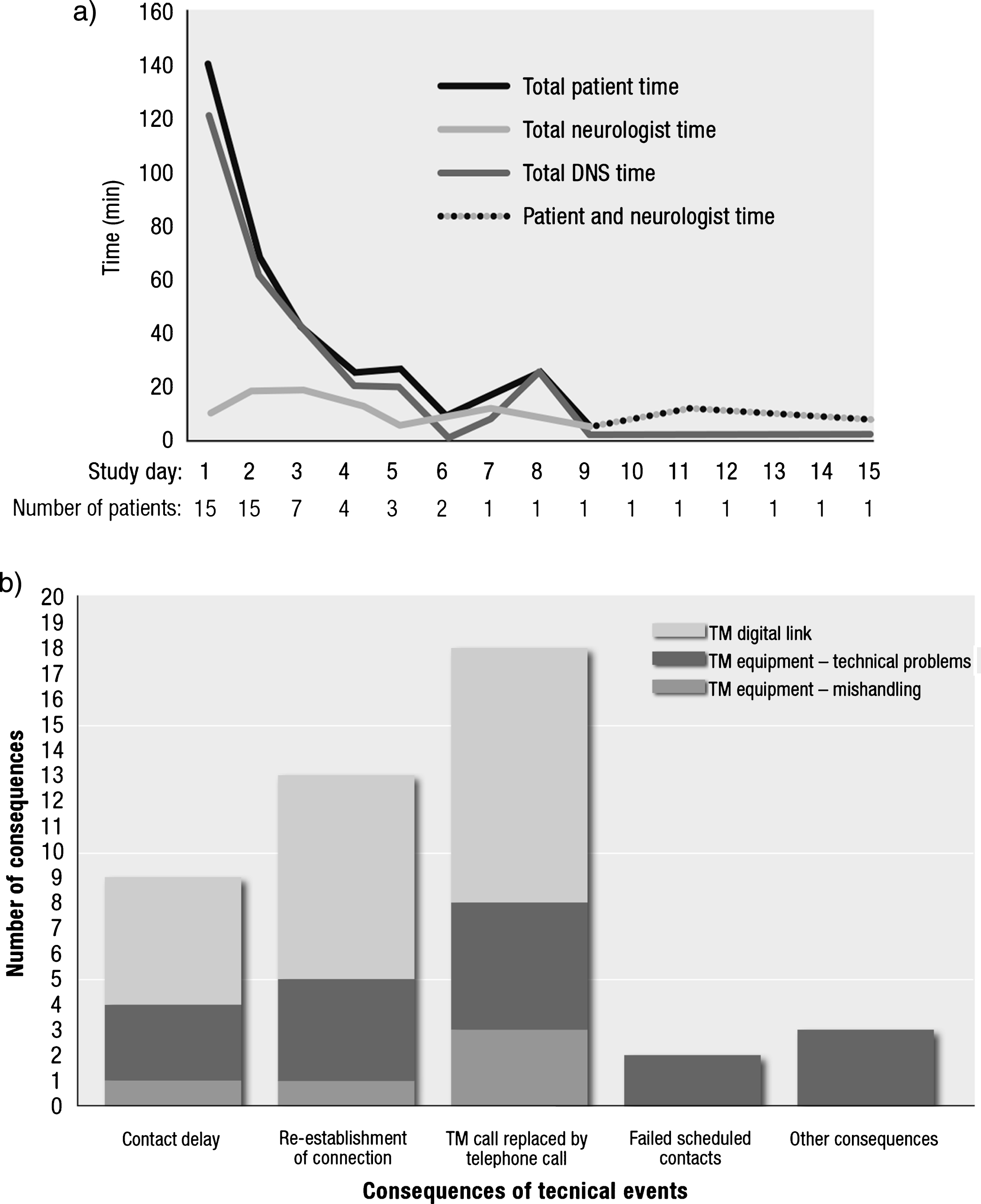
Within 3 days, LCIG home titration was completed by 8 patients. Only 1 patient required more than 6 days for home titration (Fig. 1a). The median total time for the LCIG titration period was 2.8 (2.0–13.8) days. Median total free time without contact with any healthcare professional (HCP) and without individual pump handling was 2.7 (1.7–9.5) days. The median percentage of patient total free time during TM-assisted LCIG titration period at home was 92% (69% –96%). Concomitant PD-medication varied between patients. 14 had adjunctive therapy other than levodopa at baseline and 10 patients kept at least one levodopa tablet, mainly before bedtime. 5 patients reduced or removed rotigotine, pramipexole or entacapone during the titration period. The median total time for titration for these patients was 3 (2.0–13.8) days.
Of the 183 TM contacts, 7% were associated with a technical event, affecting 11 patients (73%). The most common consequence of a technical event was replacement of TM session by phone call (18), reestablishment of connection (13), or delayed contact (9) (Fig. 1b). Only 2 of the scheduled contacts failed to take place as scheduled. The most common root-causes of the technical problems related to the digital link, such as failure to establish connection or low sound quality. Furthermore, 13 technical problems with equipment, 3 non-intentional mishandlings of equipment, and 4 “other” events were reported. No intentional misuse of TM equipment was recorded.
Overall, using TM for LCIG home titration was positive for patients, neurologists, and DNSs. Fourteen of the 15 patients consented to be interviewed and all study neurologists and DNS answered web-based questionnaires. Results of items assessed are shown in Fig. 2. Overall, 86% of patients were very satisfied (6 or 7 on the 7-point Likert scale) using TM for LCIG home titration, whereas 93% of neurologists and 80% of DNS were very satisfied with how the TM method met their clinical needs. Eighty-seven percent of patients were very satisfied with the communication with neurologist and DNS, and with the ease of using the equipment. In addition 93% said that they felt very secure being titrated at home using TM. Most (93% and 87%) of the DNSs and neurologists, respectively, stated that they were very satisfied with the communication and 87% and 80% were very satisfied with the clinical assessment (Fig. 2). Yes/no questions revealed that the majority of patients (92.9%) and caregivers (88.9%) were able to do things at home that they could not have done if the patient was hospitalized. After the experience of LCIG home titration, 92.9% of the patients and 100% of the caregivers preferred that the patient had not been hospitalized to start LCIG treatment. Themajority of the neurologists (73.3%) and DNSs (60.0%) felt no important limitations for clinical assessment by using TM, compared to hospital titration, since rigidity and balance could be estimated visually when the patient was moving in front of the camera. However, both neurologists and DNS commented in free text on the questionnaires on the lack of reliable means for determining rigidity, physical examination, and the pull-test for postural instability.
Fig.2
Experience of LCIG home titration using TM by (a) patients, (b) neurologists, and (c) DNS (DNS, Duodopa nurse specialist; LCIG, levodopa-carbidopa intestinal gel; TM, telemedicine).

Time for patient contact (real communicationtime) and time to set up for patient (booked communication time) were considered to be less by the neurologists (60.0% and 66.6%, respectively) and by DNS (93.3% and 60.0%, respectively), while time for other tasks between contacts were considered to be more for both neurologists (53.3%) and DNSs (93.3%).
Using TM home titration for start of LCIG infusion, 14 patients (93%) decided to undergo a PEG-J implantation and continue LCIG infusion, which is in the same range as previously reported [22, 23]. One patient did not continue to PEG/J surgery, since the neurologist deemed the efficacy too low.
Improvement on the CGI-I was reported for all 14 patients that chose to continue with PEG-J surgery; 10 patients (71.4%) were “much improved” and 4 patients (28.6%) were “very much improved” at the end of titration. No serious adverse drug reactions and only one device complaint (nasojejunal tube occlusion) was reported.
DISCUSSION
This study represents the first systematic evaluation of LCIG initiation and individual titration at home using a TM approach in patients with advanced PD, with the aim of evaluating the feasibility of TM, resource utilization, and patient, neurologist, and DNS satisfaction. Demographics and PD characteristics were similar to cohorts described in other studies of LCIG in routine care, [8–11] except that markedly more men (87%) than women were participating. In comparison, 68% of patients completing titration during this time period in Sweden were men. The higher gender imbalance in the study may have been due to the eligibility criteria to include only technically-motivated patients confident to test LCIG home titration using TM and to be able to handle infusion pump and TM equipment by themselves, reflecting a potential selection basis. However, considering the age, overall health status, and expected limited familiarity with TM technology in Parkinson’s population, not all patients are expected to be willing or able to use Internet-based video-communication technology. However, 3 patients lived at home without caregivers and managed to perform the LCIG home titration using TM. The patient who required 15 days of TM titration period to decide whether to continue LCIG infusion had the most advanced PD in the study, and was the only patient living in residential care. This illustrates the importance of finding appropriate and motivated patients to use TM for home titration. Nevertheless, this patient had a chance to undergo an LCIG run-in phase with the temporary nasointestinal tube at home for 14 days without beinghospitalized.
The essential outcomes of the study are the relatively low number of contacts, and the short median time required for LCIG home titration using TM. Total time spent on contacts was approximately 9 hours for patients (including training and technician visits), 1 hour for neurologists, and 6 hours for DNS (including travel time for home visits). In the post-patient questionnaire, neurologists and nurses considered this to be less time than required for hospital titration. The median period of LCIG home titration, from pump start to the decision on PEG-J implantation was 2.8 days. This is shorter than “traditional” titrations in many Swedish hospitals according to clinical experience. From a quality-of-life perspective, neurologists and nurses commented that the “normal activity level at home” and less stress in a well-known surrounding were important, contributors for “making it easier and quicker to find the right dose.” According to the neurologist judgement, the communication was focused and time efficient. Also, one neurologist commented that no time was required for within-hospital transportation and change of clothing. Other comments were that the level of independence and self-management can be expected to be high in patients titrated at home [8]. Dose adjustments during titration may, in general, affect the duration of titration. However, no apparent differences could be seen between patients with tapering and those with stable dosing during titration.
Home titration may reduce healthcare cost by reducing the need for in-ward bed occupancy as well as HCP and patient time, including hospitalization and travel. The approximate costs for equipment per clinic was 24 000 EUR plus 500 EUR per patient for technical assistance. The savings from reduced need for hospital admission was 5 200 EUR per patient (estimated on 7 nights admission) as well as the reduced time for neurologist, DNS and patient (including travel). After titrating approximately 4 patients using TM the method was regarded as cost-effective at clinic level in Sweden in 2013. However, hospital organizations, reimbursement systems, titration rate and technological possibilities are country specific and changing with time.
The most crucial benefit from the patient’s perspective may be 92% free time to do preferential things such as meeting friends or any other home-based or outbound activities, without contact with any HCP or independent pump handling during LCIG titration using TM. Importantly, fine-tuning of dose may be required during the first month of treatment independent of mode for titration (e.g., hospital outpatient or home titration).
Success factors for TM are appropriate Internet connection and reliable technical equipment, as well as proper training of patients and HCPs. The home titration could always be continued despite any technical problems, and all 15 patients succeeded to manage LCIG titration at home.
Overall, the use of TM and its feasibility for LCIG home titration using this innovative approach was highly appreciated by all involved parties. Interestingly, patient and caregiver satisfaction with TM for home titration were higher compared with ratings of neurologists and DNS. This likely reflects the overallsatisfaction of patients (and caregivers) to be able to stay at home for LCIG titration (instead of being hospitalized). However, the satisfaction is likely also influenced by the new treatment itself. Potentially, the professional expectations of neurologists and DNS might have been higher in terms of reliability of digital links and quality of picture and sound. Evaluation of PD-specific symptoms, such as rigidity, could not be fully assessed, only observed visually and communicated by the patient. An interesting opportunity would be to further develop TM for self-recording by patient at any time (e.g., stiffness or particular motor performance events during home titration), offering the opportunity to show and discuss this video sequence with the physician at a subsequently scheduled TM session.
This observational study has some design-related limitations: no control group to allow for direct comparisons, the open-label nature, a selection bias towards autonomous and technically confident patients, and a small patient population, overall making it difficult to separate TM benefits from treatment benefits. However, the detailed recordings of each type of contact and its duration, the systematic reporting of each technical problem and its consequence, and a standardized interview procedure and questionnaire using a Likert scale to assess patients, neurologists, and DNS experience allowed the thorough evaluation of technical feasibility, efficiency, and user satisfaction of TM for LCIG home titration. This study in routine care represents “real-life” data; these experiences will enable further implementation of alternative LCIG home titration in a larger patient population.
TM was appreciated by all users: patients, neurologists, and DNS. It was resource-efficient in terms of time required by patients, neurologists, and DNS, and the overall duration of titration. Importantly, it allowed patients 92% free time without HCP contact or independent pump handling. This study shows that LCIG titration at home using TM is technically feasible, offering a well-accepted alternative procedure for initiation of LCIG.
CONFLICT OF INTEREST
Thomas Willows was the principal neurologist of this study and is site neurologist for another AbbVie-sponsored clinical trial. Dag Nyholm has received lecture fees from AbbVie; has received research support from AbbVie and has until 2012 received compensation from AbbVie for serving as a consultant; has received royalties from Liber AB; has served as a consultant to Sensidose AB and OrbiMed Advisors LLC; has received honoraria from H. Lundbeck AB, Movement Disorders Society, NordicInfu Care and The National Board of Health and Welfare; NordicInfu Care; Ipsen, Selanders Foundation, Swedish Knowledge Foundation, Swedish Parkinson’s Disease Foundation, Swedish Research Council and VINNOVA Sweden’s innovation agency; is co-founders and stock owner in Jemardator AB; receive remuneration from the website netdoktor.se for participation in an expert panel; and received institutional support from Uppsala University Hospital. Nil Dizdar have received lecture fees from AbbVie and have been site neurologists for AbbVie-sponsored clinical trials. Håkan Widner, Peter Grenholm, and Ursula Schmiauke have been site neurologists for AbbVie-sponsored clinical trials. Kristina Groth, Jörgen Larsson, and Johan Permert are employed and/or affiliated with Karolinska University Hospital and Karolinska Institutet. Susanna Kjellander and Anna Urbom are employed by and consultant to AbbVie, respectively.
ACKNOWLEDGMENTS
The authors wish to acknowledge the Innovation Center and Neurology department at Karolinska University Hospital for setting the care chain and providing technical equipment; Atea for providing of technical equipment, installation, and support; and Johanna Tilly and Kerstin Wiklund at Pharma Consulting Group AB, for statistical analyses. Medical writing support was provided by Urs E. Gasser at ClinResearch Ltd, Switzerland. This work was funded by AbbVie AB. AbbVie participated in the study design, research, data collection, analysis and interpretation of data, and writing, reviewing, and approving the publication.
REFERENCES
[1] | Jankovic J ((2008) ) Parkinson’s disease: Clinical features and diagnosis. J Neurol Neurosurg Psychiatry, 79: , 368–376. |
[2] | Chapuis S , Ouchchane L , Metz O , Gerbaud L , & Durif F ((2005) ) Impact of the motor complications of Parkinson’s disease on the quality of life. Mov Disord, 20: , 224–230. |
[3] | Schapira HA ((2007) ) Treatment options in the modern management of Parkinson disease. Arch Neurol, 64: , 1083–1088. |
[4] | Calne SM ((2003) ) The psychosocial impact of late-stage Parkinson’s disease. J Neurosci Nurs, 35: , 306–313. |
[5] | Nyholm D , Askmark H , Gomes-Trolin C , Knutson T , Lennernäs H , Nyström C , & Aquilonius SM ((2003) ) Optimizing levodopa pharmacokinetics: Intestinal infusion versus oral sustained-release tablets. Clin Neuropharmacol, 26: , 156–163. |
[6] | Nyholm D , Nilsson Remahl AI , Dizdar N , Constantinescu R , Holmberg B , Jansson R , Aquilonius SM , & Askmark H ((2005) ) Duodenal levodopa infusion monotherapy vs oral polypharmacy in advanced Parkinson disease. Neurology, 64: , 216–223. |
[7] | Olanow CW , Kieburtz K , Odin P , Espay AJ , Standaert DG , Fernandez HH , Vanagunas A , Othman AA , Widnell KL , Robieson WZ , Pritchett Y , Chatamra K , Benesh L , Lenz RA , Antonini A , & for the LCIG Horizon Study Cohort ((2013) ) Continuous intrajejunal infusion of levodopa-carbidopa intestinal gel for patients with advanced Parkinson’s disease: A randomised, controlled, double-blind, double-dummy study. Lancet Neurol, 13: , 141–149. |
[8] | Antonini A , Yegin A , Preda C , & Poewe W ((2015) ) Global long-term study on efficacy and safety of DUODOPA in patients with advanced Parkinson’s disease in routine care; 12-month interim outcomes. Parkinsonism Relat Disord, 21: , 231–235. |
[9] | Antonini A , Calandrella D , Mancini F , Odin P , Lopiano L , Zibetti M , Tomantschger V , Pacchetti C , Minafra B , Bertaina I , Pickut B , De Deyn P , Cras C , Gasser UE , Wolf E , Spielberger S , & Poewe W ((2012) ) Effect and safety of duodenal levodopa infusion in advanced Parkinson’s disease: A retrospective multicenter outcome assessment in patient routine care. J Neural Transm, 120: , 1553–1558. |
[10] | Fernandez HH , Standaert DG , & Hauser RA ((2015) ) Levodopa-carbidopa intestinal gel in advanced Parkinson’s disease: Final 12-month, open-label results. Mov Disord, 30: , 500–509. |
[11] | Pålhagen SE , Dizdar N , Hauge T , Holmberg R , Jansson R , Linder J , Nyholm D , Sydow O , Wainwright M , Widner H , & Johansson A ((2012) ) Interim analysis of long-term intraduodenal levodopainfusion in advanced Parkinsondisease. Acta NeurolScand, 126: , 29–33. |
[12] | Zibetti M , Merola A , Ricchi V , Marchisio A , Artusi CA , Rizzi L , Montanaro E , Reggio D , De Angelis C , Rizzone M , & Lopiano L ((2013) ) Long-term duodenal levodopa infusion in Parkinson’s disease: A 3-year motor and cognitive follow-up study. J Neurol, 260: , 105–114. |
[13] | Hubble JP , Pahwa R , Michalek DK , Thomas C , & Koller WC ((1993) ) Interactive video conferencing: A means ofproviding interim care to Parkinson’s disease patients. Mov Disord, 8: , 380–382. |
[14] | Samii A , Ryan-Dykes P , Tsukuda RA , Zink C , Franks R , & Nichol WP ((2006) ) Telemedicine for delivery of health care in Parkinson’s disease. J Telemed Telecare, 12: , 16–18. |
[15] | Constantinescu G , Theodoros D , Russell T , Ward E , Wilson S , & Wootton R ((2011) ) Treating disordered speech and voice in Parkinson’s disease online: A randomized controlled non-inferiority trial. Int J Lang Commun Disord, 46: , 1–16. |
[16] | Biglan KM , Voss TS , Deuel LM , Miller D , Eason S , Fagnano M , George BP , Appler A , Polanowicz J , Viti L , Smith S , Joseph A , & Dorsey EE ((2009) ) Telemedicine for the care of nursing home residents with Parkinson’s disease. Mov Disord, 24: , 1073–1076. |
[17] | Cubo E , Gabriel-Galan JM , Martinez JS , lcubilla CR , Yang C , Arconada OF , & Pérez NM ((2012) ) Comparison of office-based versus home Web-based clinical assessments for Parkinson’s disease. Mov Disord, 27: , 08–311. |
[18] | Dorsey ER , Deuel LM , Voss TS , Finnigan K , George BP , Eason S , Miller D , Reminick JL , Appler A , Polanowicz J , Viti L , Smith S , Joseph A , & Biglan KM ((2010) ) Increasing access to specialty care: A pilot, randomized controlled trial of telemedicine for Parkinson’s disease. Mov Disord, 25: , 1652–1659. |
[19] | Rubin MN , Wellik KE , hanner DD , & Demaerschalk BM ((2012) ) Systematic review of teleneurology: Methodology. Front Neurol, 3: , 156. |
[20] | Wechsler LR ((2015) ) Advantages and limitations of teleneurology. JAMA Neurol, 72: , 349–354. |
[21] | Willows T , Groth K , Bjorkehag J , Andersson M , Larsson J , & Permert J ((2015) ) Moving health care to the patients’ home: An innovative approach to introduce levodopa-carbidopa intestinal gel (LCIG) treatment. Mov Disord, 30: (Suppl 1), 1123. |
[22] | Fernandez HH , Vanagunas A , Odin P , Espay AJ , Hauser RA , Standaert DG , Chatamra K , Benesh J , Pritchett Y , Hass SL , & Lenz RA ((2013) ) Levodopa-carbidopa intestinal gel in advanced Parkinson’s disease open-label study: Interim results. Parkinsonism Relat Disord, 2013: , 339–345. |
[23] | Nyholm D , Lewander T , Johansson A , Lewitt PA , Lundqvist C , & Aquilonius SM ((2008) ) Enteral levodopa/carbidopa infusion in advanced Parkinson disease: Long-term exposure. Clin Neuropharmacol, 31: , 63–73. |




