The Synaptic Function of α-Synuclein
Abstract
α-Synuclein is an abundant neuronal protein which localizes predominantly to presynaptic terminals, and is strongly linked genetically and pathologically to Parkinson’s disease and other neurodegenerative diseases. While the accumulation of α-synuclein in the form of misfolded oligomers and large aggregates defines multiple neurodegenerative diseases called “synucleinopathies”, its cellular function has remained largely unclear, and is the subject of intense investigation. In this review, I focus on the structural characteristics of α-synuclein, its cellular and subcellular localization, and discuss how this relates to its function in neurons, in particular at the neuronal synapse.
HISTORY
α-Synuclein was named after its localization on synaptic vesicles and on nuclear envelopes isolated from the Torpedo electric organ [1]. In parallel, α-synuclein was identified as the non-amyloid-β component (NAC) found in amyloid plaques of Alzheimer’s disease patients [2]. The discovery of α-synuclein was soon followed by the identification of its close homologs β- and γ-synuclein [3–6]. Since then, α-synuclein has been linked to various devastating diseases, including Parkinson’s disease [7, 8], dementia with Lewy bodies [7, 8], multiple system atrophy [9–11], Alzheimer’s disease [12, 13], pantothenate kinase-associated neurodegeneration (PKAN; a.k.a. neurodegeneration with brain iron accumulation type I; formerly Hallervorden-Spatz syndrome) [14–16], Pick’s disease [17], diffuse Lewy body disease [18], Lewy body variant of Alzheimer’s disease [19], amyotrophic lateral sclerosis (ALS) [20, 21], ALS-Parkinsonism-dementia complex of Guam [22, 23], pure autonomic failure [24], frontotemporal dementia [25, 26], progressive supranuclear palsy [27, 28], corticobasal degeneration [29], and Krabbe disease [30], collectively termed “synucleinopathies”. In addition, genome-wide association studies have identified a higher risk of sporadic Parkinson’s disease for individuals with variations in the SNCA gene [31], highlighting α-synuclein’s genetic link to the disease. The physiological function of α-synuclein, however, has remained enigmatic.
α-SYNUCLEIN EXPRESSION & LOCALIZATION
α-Synuclein is a protein of 140 residues that is predominantly and ubiquitously expressed in the brain [4], in particular throughout the neocortex, hippocampus, olfactory bulb, striatum, thalamus, and cerebellum in the rat brain [32]. While initially described as a nuclear protein [33, 34], these reports have not been consistent. In contrast, the presynaptic localization of α-synuclein has become well established (see below). Yet, although α-synuclein is highly enriched in synaptic boutons which sprout from axons of different neurochemical phenotypes, α-synuclein is not present in all synaptic terminals, and, curiously, not all terminals accumulate the protein in neurodegenerative disorders [35], suggesting selective expression, targeting, and pathogenic vulnerability in certain neuronal populations. Furthermore, although highly enriched in the nervous system [2, 4], its expression is not limited to nervous tissues: significant amounts of α-synuclein have been detected in red blood cells [36], and low levels of expression have been found at mRNA and/or protein level also in other tissues [37–43], suggesting more general cellular functions in addition to its activity in the brain.
Out of the three synuclein family members, β-synuclein reveals the most brain-specific expression [44], and γ-synuclein the least [5]. Similar to α-synuclein, β- and γ-synucleins localize to synaptic terminals [4, 45, 46], and overlap with expression of α-synuclein in certain brain areas [5, 44, 47]. Although β- and γ-synuclein are absent from Lewy bodies, they co-localize with α-synuclein in spheroid-like neuronal inclusions in Parkinson’s disease, dementia with Lewy bodies and PKAN [7, 15]. The identification of polymorphisms in β- and γ-synuclein that predispose to dementia with Lewy bodies and diffuse Lewy body disease [18, 48], neurodegeneration in mutant β- and wild-type γ-synuclein transgenic mice [49–51], co-occurrence of β-synuclein in α-synuclein-containing Pick bodies in frontotemporal dementia [17], and the link of γ-synuclein to ALS, Gaucher’s disease, and Alzheimer’s disease [52–54], suggests that all synucleins may be involved in neurodegenerative diseases.
Within the nervous system, the expression of α-synuclein is developmentally regulated. α-Synuclein mRNA expression begins in late embryonic stages in rodents, reaches a peak in the first few postnatal weeks, and is then reduced [55, 56]. α-Synuclein protein levels increase during development and remain high during adulthood [56, 57], suggesting post-transcriptional regulation of its levels. α-Synuclein distributes from the soma to presynaptic terminals during early weeks of development in rodents [58, 59] and in humans [60, 61], where it associates with synaptic vesicles [1, 62]. Although it is still unclear how α-synuclein reaches the synapse, its preference for synaptic vesicle membranes [1, 62], and its affinity for the vesicular SNARE protein synaptobrevin-2 [63], synapsin III [64], or rab3A [65], may target it to presynaptic boutons. Strikingly, while highly concentrated in presynaptic terminals, α-synuclein is among the last proteins to reach the synapse [58, 66]. Together with its presence only in vertebrates [67], this suggests that α-synuclein has an activity required for a more complex cellular function that is not essential for basic neurotransmitter release or synapse development.
STRUCTURE OF α-SYNUCLEIN
α-Synuclein has a remarkable and unique structure (Fig. 1). Its N-terminal sequence is divided into seven 11-mer repeats with a KTKGEV consensus sequence (residues 1–95), which, similar to apolipoproteins, form an amphipathic alpha-helix with 3 turns, and mediate association of α-synuclein with lipid membranes [68–72]. This region contains also the NAC domain (residues 60–95), an area believed to be responsible for α-synuclein aggregation [2] and sensing of lipid properties [73]. Curiously, all identified mutations associated with synucleinopathies are located in this region: A30P, E46K, H50Q, G51D, A53E, and A53T [74–80], five of which cluster within eight residues, suggesting that lipid binding or lack thereof may be linked to α-synuclein pathology. The C-terminus of α-synuclein (residues 96–140) is highly acidic and largely unstructured [68, 69, 81], target of various post-translational modifications [82], and believed to be responsible for (i) interactions with proteins (see below), (ii) ion, polycation and polyamine binding [83–86], (iii) modulation of membrane binding of α-synuclein [87, 88], and for (iv) protection of α-synuclein from aggregation [89–91].
INTRACELLULAR POOLS OF α-SYNUCLEIN
α-Synuclein exists in a dynamic equilibrium between a soluble state and a membrane-bound state, with its secondary structure depending on its environment. The interaction between α-synuclein and lipid surfaces is believed to be key feature for mediating its cellular functions (Fig. 2). Soluble cytosolic α-synuclein is intrinsically unstructured and behaves like a natively unfolded protein [71, 92–95]. A debate has recently developed around α-synuclein’s soluble state, due to a proposed metastable tetrameric form of α-synuclein [96, 97]. While other studies have demonstrated that no such cytosolic tetramer exists in the central nervous system, in erythrocytes, mammalian cells, and in E.coli [94, 95, 98, 99], binding to cellular factors, such as lipids or membranes, can induce and stabilize such multimers [100], as endogenous multimers become unstable as the protein approaches purity [101].
In presence of lipid membranes, such as artificial liposomes, lipid droplets and lipid rafts, the N-terminal residues of α-synuclein adopt an alpha-helical structure which mediates binding of α-synuclein to membranes [68–71, 102–104]. Membrane binding is likely a cooperative effect of the 11-mer sequences, as truncation of the N-terminal domain reduces lipid binding drastically, and requires acidic head groups [102–106], such as phosphatidylethanolamine, phosphatidylserine or phosphatidylinositol. This suggests an interaction of the membrane headgroups with lysines found on opposite sides of the α-synuclein helix. Both, a single elongated alpha-helix, and a broken alpha-helix have been reported, depending on membrane curvature [68, 71, 72], and α-synuclein is able to transition between these two states [81, 107]: Upon binding to membranes with larger diameter (∼100 nm), α-synuclein adopts an elongated helix [68, 108–111]. In contrast, in presence of small and highly curved vesicles, α-synuclein adopts a broken helix conformation [71, 81, 112, 113], likely to adapt to the smaller liposome area. α-Synuclein preferentially binds to vesicles of smaller diameter [69, 114], and as such associates with synaptic vesicles in the brain [1, 62].
Recently, it was found that α-synuclein is N-terminally acetylated, mediated by attachment of an acetyl group to the alpha amino group of the first amino acid of α-synuclein [94, 95, 115, 116]. N-terminal acetylation of α-synuclein is seen both in healthy and Parkinson’s disease individuals, and increases its helical folding propensity, its affinity for membranes, and its resistance to aggregation [115–118], suggesting that N-terminal acetylation of α-synuclein could have important implications for both the native and pathological structures and functions of α-synuclein [119]. In addition, phosphorylation of α-synuclein regulates its structure, membrane binding, protein interactions, oligomerization, fibril formation, and neurotoxicity [120–125], although the exact kinases and phosphatases regulating (de)phosphorylation of α-synuclein remain unknown. Other post-translational modifications, such as ubiquitination [126, 127], sumoylation [128, 129], glycation [130–132], glycosylation [133, 134], nitration [135–137], and proteolysis [12, 89, 138, 139], can result in changes in protein charge and structure. This may lead to altered binding affinities with other proteins and lipids, but their functional significance remains unknown and controversial.
α-Synuclein folding stabilizes and protects its target membrane [140], and membrane-binding protects α-synuclein from aggregation [141–144], although membrane binding has also been reported to accelerate aggregation under oxidative stress [145]. Recently, alpha-helical multimers of α-synuclein have been reported upon binding of α-synuclein to membranes, which are required for its physiological function at the synapse, and protect α-synuclein from aggregation [100, 142, 146]. In contrast to these physiological conformations, in its pathologically relevant state, alpha -synuclein adopts a beta-sheet rich conformation which is accompanied by aggregation and fibril formation, and deposition into Lewy bodies [147–151]. These cytosolic aggregates are likely derived from the less stable, natively unfolded conformations of cytosolic α-synuclein [142].
α-SYNUCLEIN FUNCTION AT THE SYNAPSE
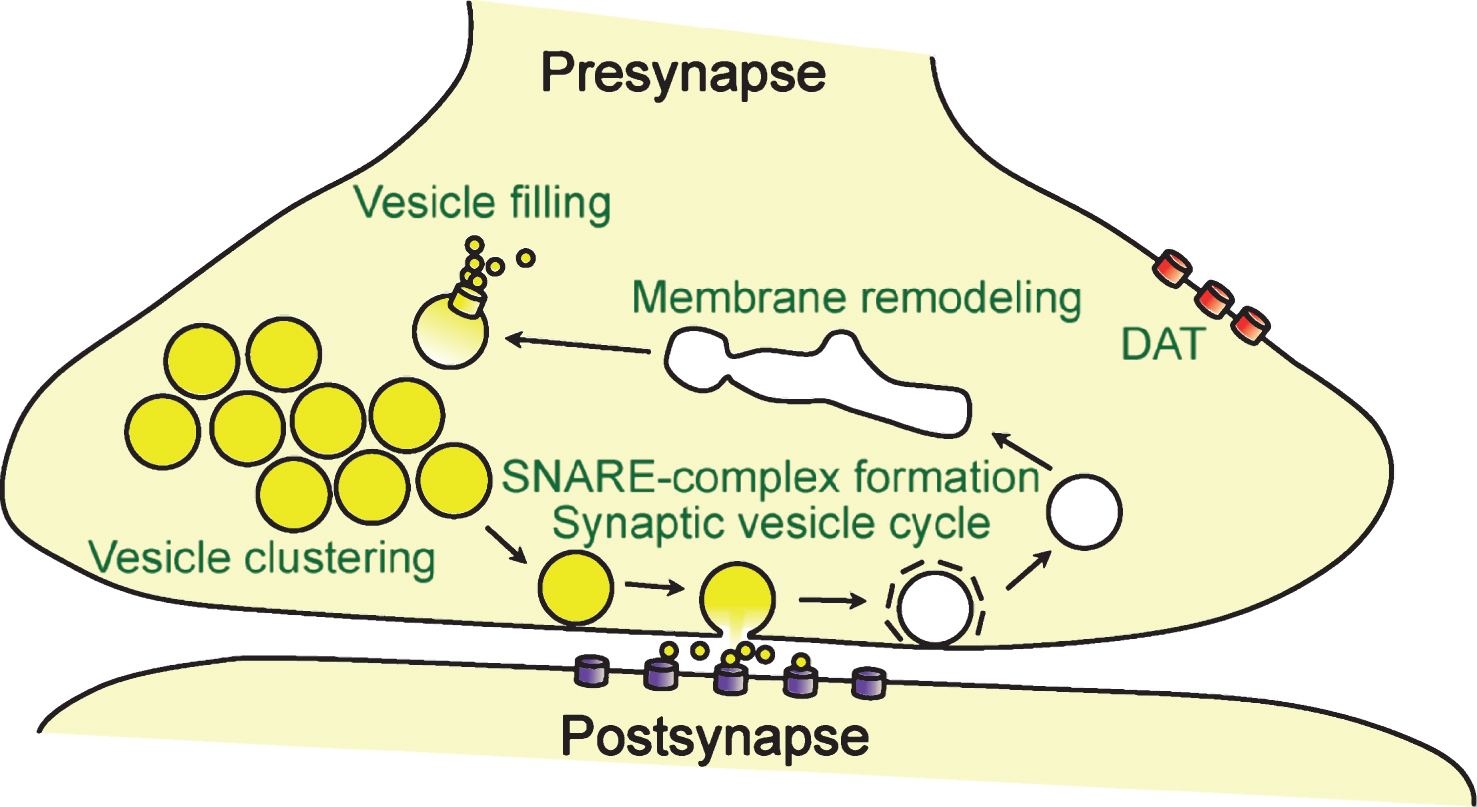
The normal function of α-synuclein remains enigmatic, despite more than 25 years of research. Assessing the normal function of α-synuclein has been challenging, because: (i) α-Synuclein is an intrinsically unstructured protein that cycles between a natively unfolded state in cytosol, and a helical multimeric state on membranes [71, 92–95, 100]; (ii) Overexpression of α-synuclein triggers toxic effects in humans [152, 153] and in animal models [154–156], that are much worse than the effects caused by loss of α-synuclein [157, 158]. This disconnection of the pathogenic activity of α-synuclein from its physiological function [159] complicates findings in overexpression models; (iii) Potential compensation of α-synuclein function by its isoforms β- and γ-synuclein complicate findings in knockout animals and necessitate simultaneous knockout of all isoforms or acute manipulation, such as done via viral injections. However, α-synuclein’s presynaptic localization and its interaction with highly curved membranes and synaptic proteins strongly suggests a regulatory function associated with the synapse, such as synaptic activity, synaptic plasticity, learning, neurotransmitter release, dopamine metabolism, synaptic vesicle pool maintenance, and/or vesicle trafficking (Fig. 3).
Protein interactions
α-Synuclein has been reported to interact with and affect a variety of proteins, mostly at the presynaptic terminal. This includes a controversial binding of phospholipase D [160–163], regulation of the membrane interaction of the G-protein rab3 [65], binding to the SNARE-protein synaptobrevin-2 and chaperoning SNARE-complex assembly [63, 159], binding and modulation of synapsin III [64], binding of VMAT2 [164], dopamine and serotonin transporters [165–167], and regulation of tyrosine hydroxylase [168–170]. While these interactions are compatible with a function at the presynaptic terminal, the reported localization of α-synuclein to mitochondria [171–173], endoplasmic reticulum [174, 175], Golgi [174, 175], and nuclei [1, 176] may arise from an altered subcellular distribution or spillover to other membranes, due to overexpression or during cell disruption. Overall, the functional significance of most of these findings remains unclear.
Lipid transport, lipid packing and membrane biogenesis
The similarity of α-synuclein with class A2 apolipoproteins and decreased brain palmitate, phosphatidylglycerol and cardiolipin metabolism in absence of α-synuclein [177–179] suggest a role in lipid metabolism, although lipidomic profiling of brains from synuclein transgenic and knockout mice revealed minimal effects of synuclein on lipid metabolism [180]. α-Synuclein has been reported to bind to fatty acids [181], and may thus serve as a fatty acid transporter between the cytosol and membrane compartments, while other studies suggest the contrary [182]. Furthermore, α-synuclein has been shown to induce membrane curvature and convert large vesicles into highly curved membrane tubules, cylindrical micelles and vesicles [183–187], driven by binding affinity, partition depth, and interleaflet order asymmetry [188]. In addition, α-synuclein has been reported to organize membrane components [189], to modulate phospholipid composition [190], and to be a specific inhibitor of phospholipase D1 and D2 in vitro andin vivo [160–162]. This suggests that α-synuclein may be involved in cleavage of membrane lipids and membrane biogenesis. Yet, the data on α-synuclein and phospholipase D inhibition are controversial [163]. Last, α-synuclein has been reported to sense lipid packing defects and to affect lipid packing [191, 192], and binding of α-synuclein to synaptic vesicles may stabilize them via stabilizing their intrinsically tight curvature [193].
Impact on dopamine metabolism and dopaminergic neurons
While many types of neurons are affected in Parkinson’s disease [194–196], a remarkable sign is the loss of dopaminergic neurons in the substantia nigra, and the resulting deficiency of dopamine signaling [197–199]. Despite tremendous strides in the understanding of α-synuclein function and dysfunction, the increased vulnerability of dopaminergic neurons to α-synuclein pathology remains unclear at the mechanistic level. α-Synuclein has been proposed to regulate homeostasis of monoamines in synapses, via interaction with the serotonin transporter [165]. It binds to and regulates the targeting and the activity of the dopamine transporter DAT [166, 167, 200], although its mode of action remains controversial [201–203]. α-Synuclein inhibits dopamine synthesis by inhibiting the expression and activity of tyrosine hydroxylase [154, 168–170, 204], likely via reducing the phosphorylation state of tyrosine hydroxylase and stabilizing dephosphorylated inactive tyrosine hydroxylase [168, 205–207]. In agreement, aging-related increases in α-synuclein expression in the substantia nigra negatively correlate to the expression of tyrosine hydroxylase [57]. In addition, α-synuclein affects the vesicular dopamine transporter VMAT2: Knockdown of alpha -synuclein increased the density of VMAT2 molecules per vesicle, while overexpression inhibits VMAT2 activity, interrupting dopamine homeostasis by causing increased cytosolic dopamine levels [164]. In agreement with a function in dopamine metabolism, absence of α-synuclein causes decreased reuptake of dopamine in the dorsal striatum [208], a 36% reduction in striatal dopamine, accompanied by a reduction in tyrosine hydroxylase-positive fibers in the striatum, decreased striatal levels of tyrosine hydroxylase and dopamine transporter [209], and a decrease in the number of dopaminergic neurons in the substantia nigra [210, 211]. In addition, α/β-synuclein double knockout mice display 20% reduced dopamine levels, with no change in dopamine uptake and release [212], a two-fold increase in extracellular dopamine levels upon striatal stimulation, and hyperactivity in a novel environment, which is reminiscent of mice expressing reduced levels of the dopamine transporter [213]. Overall, this suggests that dopaminergic neurons may have both, a higher need for α-synuclein function, and a higher susceptibility to α-synuclein dysfunction. Yet, the presence of α-synuclein in cells other than dopaminergic neurons suggests a more general activity in neuronal function.
Molecular chaperone activity
The biochemical structure of α-synuclein predicts a function as a molecular chaperone capable of binding to other intracellular proteins. This hypothesis was strengthened by three observations: First, alpha -synuclein shares structural and functional homology with the 14-3-3 family of molecular chaperone proteins [214]. Second, via its C-terminal domain, α-synuclein suppresses the aggregation of thermally denatured proteins [215–219], and overexpression of α-synuclein protects dopaminergic neurons from oxidative stress and apoptosis [220, 221]. Third, α-synuclein rescues the lethal neurodegeneration caused by knockout of the co-chaperone CSPα in mice by chaperoning assembly of synaptic SNARE-complexes [63, 222]. This function of α-synuclein is essential for long term functioning of neurons, since α-, β-, γ-synuclein triple-knockout mice have reduced SNARE-complex assembly, show neuropathological signs and reveal shortened survival [63, 223, 224]. This chaperone function is consistent with the lack of an acute effect of α-synuclein on cell survival and neurotransmitter release, and may become particularly important under stressful conditions and during the long life of a neuron.
Neurotransmitter release and synaptic plasticity
The presynaptic localization of α-synuclein, its interaction with synaptic vesicles [1, 62] and synaptobrevin-2 [63], its SNARE-complex chaperoning activity [63], and its changes during periods of song-acquisition-related synaptic rearrangements in birds [225] strongly argues for a role in neurotransmitter release and synaptic plasticity, although its precise function remains unclear. Yet, absence of α-synuclein in worms, flies and yeast suggests that α-synuclein is not required for synaptic transmission or membrane trafficking in general. In agreement, knockout of α-, α/β-, α/γ-, or α/β/γ-synucleins does not induce morphological changes in the brain [63, 157, 212, 224], although changes in synaptic protein levels [63, 212], changes in synapse structure and size [223], and impairments in survival [63, 223] have been reported in synuclein triple knockout mice. Together with neuromuscular pathology in mice lacking α-synuclein [226], and reduced working and spatial memory learning in α-synuclein knockout mice [227, 228], this suggests that α-synuclein contributes to the long-term operation of a neuron.
The effect of α-synuclein on neurotransmission and synaptic plasticity has been investigated both in knockout and under overexpressing conditions, where α-synuclein has been reported to both promote and inhibit neurotransmitter release, or have no effect at all. While some studies reported a lack of effect of α-synuclein on neurotransmitter release [63, 212, 229], others revealed an enhancement of synaptic transmission [223, 224, 230–234], or a decrease in release [157, 158, 213, 223, 235–237]. Two recent studies have reported an inhibitory effect of α-synuclein on synaptic vesicle endocytosis during intense stimulation, but not under basal levels [238, 239], while another study reported an enhancement of clathrin-mediated endocytosis by α-synuclein in neuronal and non-neuronal cells [240]. Whether the inconsistent results obtained for the effects of α-synuclein on neurotransmission and synaptic plasticity could be ascribed to the experimental models used and the investigated brain regions, needs to be determined. It seems to be clear, though, that α-synuclein is not required for basal neurotransmission, but plays an important role in maintaining neurons during intense neuronal activity and over their long lifetime.
How does α-synuclein exert its effect on the neurotransmission machinery? Within the presynaptic terminal, α-synuclein is highly mobile, as shown by photo-bleaching experiments, and α-synuclein disperses from synaptic vesicles upon stimulation [241, 242], similar to synapsin I [243]. Facilitated by its dynamic membrane-binding, this suggests that α-synuclein can be recruited to the site of high membrane-fusion activity, and that neural activity controls the normal function of α-synuclein at the nerve terminal. Indeed, α-synuclein attenuates the mobility of synaptic vesicle pools between presynaptic boutons and maintains the overall size of the recycling pools at individual synapses [244].
In vitro, α-synuclein inhibits docking of synaptic vesicle mimics with plasma membrane mimics [245, 246]. This inhibition is not caused by interfering with the fusion process itself, but is due to clustering of synaptic vesicle mimics, a process strongly dependent on α-synuclein’s ability to associate with lipids and synaptobrevin-2 [246]. α-Synuclein driven vesicle clustering has been initially reported in yeast [247, 248]. Recently, α-synuclein has been reported to cluster synaptic vesicles in neurons [146], which is likely mediated by α-synuclein’s ability to form multimers on the vesicle surface [100, 146]. This clustering activity of α-synuclein restricts synaptic vesicle motility [146], and thereby likely affects the kinetics of neurotransmitter release. Supportively, α-synuclein associates with specific subpopulations of synaptic vesicles [100, 249], and cooperatively regulates synaptic function with synapsin III in dopaminergic neurons [64]. In addition, α-synuclein knockout synapses reveal a selective deficiency of undocked vesicles without affecting docked vesicles [158], and knockdown of α-synuclein leads to a significant reduction in the distal pool of synaptic vesicles [66].
How does clustering of synaptic vesicles by α-synuclein multimers relate to increased SNARE-complex levels? α-Synuclein induced vesicle clustering may increase the local concentration of synaptic vesicles and thereby of the SNARE protein synaptobrevin-2. This clustering of synaptic vesicles at the active zone would promote the formation of neuronal SNARE-complexes by constraining additional synaptic vesicles close to the active zone. Supportively, the SNARE-complex assembly deficit in α/β/γ-synuclein triple knockout mice aggravates with increased synaptic activity [63].
Overall, the effect of α-synuclein on neurotransmitter release is likely not mediated by directly acting on the release machinery, but by affecting the spatial organization of distinct synaptic vesicle pools within the presynaptic terminal, possibly via α-synuclein multimerization, which is triggered by membrane binding and potentiates SNARE-complex assembly [100]. This activity of α-synuclein contributes to the long-term operation of the nervous system, suggesting that alterations in the physiological function of α-synuclein could promote the development of neuropathology in Parkinson’s disease and related disorders.
CONCLUSION
α-Synuclein is important for the normal function and integrity of synapses, and in the aging nervous system, dysfunction of α-synuclein becomes a predisposing factor for synaptic dysfunction and the development of neuropathology. Overexpression of α-synuclein triggers redistribution of the SNARE proteins SNAP-25, syntaxin-1 and synaptobrevin-2 in an age-dependent manner in the striatum [250], impairs proper vesicle trafficking and recycling [175, 248, 251, 252], and large α-synuclein oligomers inhibit SNARE-mediated vesicle fusion in vitro [253]. Furthermore, misfolded α-synuclein, in the form of oligomers and aggregates, is believed to be toxic [254, 255], and recent studies have revealed propagation of misfolded α-synuclein between neurons [256–259]. However, many questions remain unclear, including the causes of the selective vulnerability of dopaminergic neurons in Parkinson’s disease, the triggers for α-synuclein aggregation and pathology, and the role of aging in the pathogenesis of Parkinson’s disease. Understanding how α-synuclein localizes to and functions at the synapse, will provide a biological context to how it misfolds, which species of α-synuclein are toxic, how these species are released and taken up by neurons, and how they nucleate new aggregates in a healthy cell. It is clear that either too little or too much α-synuclein is deleterious for the brain. Thus, aiming at maintaining a healthy balance of α-synuclein in the brain is a worthwhile target for preventing synucleinopathies, while maintaining its normal brain function.
CONFLICT OF INTEREST
The author has none to declare.
ACKNOWLEDGMENTS
I thank Dr. Manu Sharma for critical review of this manuscript. This work was supported by an award from the Leon Levy Foundation, and by the American Parkinson Disease Association.
REFERENCES
1 | Maroteaux L, Campanelli JT, Scheller RH (1988) Synuclein: A neuron-secific protein localized to the nucleus and presynaptic nerve terminal J Neurosci 8: 2804 2815 |
2 | Ueda K, Fukushima H, Masliah E, Xia Y, Iwai A, Yoshimoto M, Otero DA, Kondo J, Ihara Y, Saitoh T (1993) Molecular cloning of cDNA encoding an unrecognized component of amyloid in Alzheimer disease Proc Natl Acad Sci U S A 90: 11282 11286 |
3 | Nakajo S, Omata K, Aiuchi T, Shibayama T, Okahashi I, Ochiai H, Nakai Y, Nakaya K, Nakamura Y (1990) Purification and characterization of a novel brain-specific 14-kDa protein J Neurochem 55: 2031 2038 |
4 | Jakes R, Spillantini MG, Goedert M (1994) Identification of two distinct synucleins from human brain FEBS Lett 345: 27 32 |
5 | Lavedan C, Leroy E, Dehejia A, Buchholtz S, Dutra A, Nussbaum RL, Polymeropoulos MH (1998) Identification, localization and characterization of the human gamma-synuclein gene Hum Genet 103: 106 112 |
6 | Buchman VL, Hunter HJ, Pinon LG, Thompson J, Privalova EM, Ninkina NN, Davies AM (1998) Persyn, a member of the synuclein family, has a distinct pattern of expression in the developing nervous system J Neurosci 18: 9335 9341 |
7 | Galvin JE, Uryu K, Lee VM, Trojanowski JQ (1999) Axon pathology in Parkinson’s disease and Lewy body dementia hippocampus contains alpha-, beta-, and gamma-synuclein Proc Natl Acad Sci U S A 96: 13450 13455 |
8 | Spillantini MG, Crowther RA, Jakes R, Hasegawa M, Goedert M (1998) alpha-Synuclein in filamentous inclusions of Lewy bodies from Parkinson’s disease and dementia with lewy bodies Proc Natl Acad Sci U S A 95: 6469 6473 |
9 | Spillantini MG, Crowther RA, Jakes R, Cairns NJ, Lantos PL, Goedert M (1998) Filamentous alpha-synuclein inclusions link multiple system atrophy with Parkinson’s disease and dementia with Lewy bodies Neurosci Lett 251: 205 208 |
10 | Tu PH, Galvin JE, Baba M, Giasson B, Tomita T, Leight S, Nakajo S, Iwatsubo T, Trojanowski JQ, Lee VM (1998) Glial cytoplasmic inclusions in white matter oligodendrocytes of multiple system atrophy brains contain insoluble alpha-synuclein Ann Neurol 44: 415 422 |
11 | Gai WP, Power JH, Blumbergs PC, Blessing WW (1998) Multiple-system atrophy: A new alpha-synuclein disease? Lancet 352: 547 548 |
12 | Lewis KA, Su Y, Jou O, Ritchie C, Foong C, Hynan LS, White CL3rd, Thomas PJ, Hatanpaa KJ (2010) Abnormal neurites containing C-terminally truncated alpha-synuclein are present in Alzheimer’s disease without conventional Lewy body pathology Am J Pathol 177: 3037 3050 |
13 | Hamilton RL (2000) Lewy bodies in Alzheimer’s disease: A neuropathological review of 145 cases using alpha-synuclein immunohistochemistry Brain Pathol 10: 378 384 |
14 | Saito Y, Kawai M, Inoue K, Sasaki R, Arai H, Nanba E, Kuzuhara S, Ihara Y, Kanazawa I, Murayama S (2000) Widespread expression of alpha-synuclein and tau immunoreactivity in Hallervorden-Spatz syndrome with protracted clinical course J Neurol Sci 177: 48 59 |
15 | Galvin JE, Giasson B, Hurtig HI, Lee VM, Trojanowski JQ (2000) Neurodegeneration with brain iron accumulation, type 1 is characterized by alpha-, beta-, and gamma-synuclein neuropathology Am J Pathol 157: 361 368 |
16 | Arawaka S, Saito Y, Murayama S, Mori H (1998) Lewy body in neurodegeneration with brain iron accumulation type 1 is immunoreactive for alpha-synuclein Neurology 51: 887 889 |
17 | Mori F, Hayashi S, Yamagishi S, Yoshimoto M, Yagihashi S, Takahashi H, Wakabayashi K (2002) Pick’s disease: Alpha- and beta-synuclein-immunoreactive Pick bodies in the dentate gyrus Acta Neuropathol 104: 455 461 |
18 | Nishioka K, Wider C, Vilarino-Guell C, Soto-Ortolaza AI, Lincoln SJ, Kachergus JM, Jasinska-Myga B, Ross OA, Rajput A, Robinson CA, Ferman TJ, Wszolek ZK, Dickson DW, Farrer MJ (2010) Association of alpha-, beta-, and gamma-Synuclein with diffuse lewy body disease Arch Neurol 67: 970 975 |
19 | Wirths O, Weickert S, Majtenyi K, Havas L, Kahle PJ, Okochi M, Haass C, Multhaup G, Beyreuther K, Bayer TA (2000) Lewy body variant of Alzheimer’s disease: Alpha-synuclein in dystrophic neurites of A beta plaques Neuroreport 11: 3737 3741 |
20 | Doherty MJ, Bird TD, Leverenz JB (2004) Alpha-synuclein in motor neuron disease: An immunohistologic study Acta Neuropathol 107: 169 175 |
21 | Takei Y, Oguchi K, Koshihara H, Hineno A, Nakamura A, Ohara S (2013) alpha-Synuclein coaggregation in familial amyotrophic lateral sclerosis with SOD1 gene mutation Hum Pathol 44: 1171 1176 |
22 | Yamazaki M, Arai Y, Baba M, Iwatsubo T, Mori O, Katayama Y, Oyanagi K (2000) Alpha-synuclein inclusions in amygdala in the brains of patients with the parkinsonism-dementia complex of Guam J Neuropathol Exp Neurol 59: 585 591 |
23 | Forman MS, Schmidt ML, Kasturi S, Perl DP, Lee VM, Trojanowski JQ (2002) Tau and alpha-synuclein pathology in amygdala of Parkinsonism-dementia complex patients of Guam Am J Pathol 160: 1725 1731 |
24 | Arai K, Kato N, Kashiwado K, Hattori T (2000) Pure autonomic failure in association with human alpha-synucleinopathy Neurosci Lett 296: 171 173 |
25 | Wilhelmsen KC, Forman MS, Rosen HJ, Alving LI, Goldman J, Feiger J, Lee JV, Segall SK, Kramer JH, Lomen-Hoerth C, Rankin KP, Johnson J, Feiler HS, Weiner MW, Lee VM, Trojanowski JQ, Miller BL (2004) 17q-linked frontotemporal dementia-amyotrophic lateral sclerosis without tau mutations with tau and alpha-synuclein inclusions Arch Neurol 61: 398 406 |
26 | Yancopoulou D, Xuereb JH, Crowther RA, Hodges JR, Spillantini MG (2005) Tau and alpha-synuclein inclusions in a case of familial frontotemporal dementia and progressive aphasia J Neuropathol Exp Neurol 64: 245 253 |
27 | Judkins AR, Forman MS, Uryu K, Hinkle DA, Asbury AK, Lee VM, Trojanowski JQ (2002) Co-occurrence of Parkinson’s disease with progressive supranuclear palsy Acta Neuropathol 103: 526 530 |
28 | Mori H, Oda M, Komori T, Arai N, Takanashi M, Mizutani T, Hirai S, Mizuno Y (2002) Lewy bodies in progressive supranuclear palsy Acta Neuropathol 104: 273 278 |
29 | Yamashita S, Sakashita N, Yamashita T, Tawara N, Tasaki M, Kawakami K, Komohara Y, Fujiwara Y, Kamikawa M, Nakagawa T, Hirano T, Maeda Y, Hasegawa M, Takeya M, Ando Y (2014) Concomitant accumulation of alpha-synuclein and TDP-43 in a patient with corticobasal degeneration J Neurol 261: 2209 2217 |
30 | Smith BR, Santos MB, Marshall MS, Cantuti-Castelvetri L, Lopez-Rosas A, Li G, van Breemen R, Claycomb KI, Gallea JI, Celej MS, Crocker SJ, Givogri MI, Bongarzone ER (2014) Neuronal inclusions of alpha-synuclein contribute to the pathogenesis of Krabbe disease J Pathol 232: 509 521 |
31 | Simon-Sanchez J, Schulte C, Bras JM, Sharma M, Gibbs JR, Berg D, Paisan-Ruiz C, Lichtner P, Scholz SW, Hernandez DG, Kruger R, Federoff M, Klein C, Goate A, Perlmutter J, Bonin M, Nalls MA, Illig T, Gieger C, Houlden H, Steffens M, Okun MS, Racette BA, Cookson MR, Foote KD, Fernandez HH, Traynor BJ, Schreiber S, Arepalli S, Zonozi R, Gwinn K, van der Brug M, Lopez G, Chanock SJ, Schatzkin A, Park Y, Hollenbeck A, Gao J, Huang X, Wood NW, Lorenz D, Deuschl G, Chen H, Riess O, Hardy JA, Singleton AB, Gasser T (2009) Genome-wide association study reveals genetic risk underlying Parkinson’s disease Nat Genet 41: 1308 1312 |
32 | Iwai A, Masliah E, Yoshimoto M, Ge N, Flanagan L, de Silva HA, Kittel A, Saitoh T (1995) The precursor protein of non-A beta component of Alzheimer’s disease amyloid is a presynaptic protein of the central nervous system Neuron 14: 467 475 |
33 | Mori F, Tanji K, Yoshimoto M, Takahashi H, Wakabayashi K (2002) Immunohistochemical comparison of alpha- and beta-synuclein in adult rat central nervous system Brain Res 941: 118 126 |
34 | Yu S, Li X, Liu G, Han J, Zhang C, Li Y, Xu S, Liu C, Gao Y, Yang H, Ueda K, Chan P (2007) Extensive nuclear localization of alpha-synuclein in normal rat brain neurons revealed by a novel monoclonal antibody Neuroscience 145: 539 555 |
35 | Totterdell S, Hanger D, Meredith GE (2004) The ultrastructural distribution of alpha-synuclein-like protein in normal mouse brain Brain Res 1004: 61 72 |
36 | Nakai M, Fujita M, Waragai M, Sugama S, Wei J, Akatsu H, Ohtaka-Maruyama C, Okado H, Hashimoto M (2007) Expression of alpha-synuclein, a presynaptic protein implicated in Parkinson’s disease, in erythropoietic lineage Biochem Biophys Res Commun 358: 104 110 |
37 | Askanas V, Engel WK, Alvarez RB, McFerrin J, Broccolini A (2000) Novel immunolocalization of alpha-synuclein in human muscle of inclusion-body myositis, regenerating and necrotic muscle fibers, and at neuromuscular junctions J Neuropathol Exp Neurol 59: 592 598 |
38 | Ltic S, Perovic M, Mladenovic A, Raicevic N, Ruzdijic S, Rakic L, Kanazir S (2004) Alpha-synuclein is expressed in different tissues during human fetal development J Mol Neurosci 22: 199 204 |
39 | Tamo W, Imaizumi T, Tanji K, Yoshida H, Mori F, Yoshimoto M, Takahashi H, Fukuda I, Wakabayashi K, Satoh K (2002) Expression of alpha-synuclein, the precursor of non-amyloid beta component of Alzheimer’s disease amyloid, in human cerebral blood vessels Neurosci Lett 326: 5 8 |
40 | Shin EC, Cho SE, Lee DK, Hur MW, Paik SR, Park JH, Kim J (2000) Expression patterns of alpha-synuclein in human hematopoietic cells and in Drosophila at different developmental stages Mol Cells 10: 65 70 |
41 | Kim S, Jeon BS, Heo C, Im PS, Ahn TB, Seo JH, Kim HS, Park CH, Choi SH, Cho SH, Lee WJ, Suh YH (2004) Alpha-synuclein induces apoptosis by altered expression in human peripheral lymphocyte in Parkinson’s disease FASEB J 18: 1615 1617 |
42 | Hashimoto M, Yoshimoto M, Sisk A, Hsu LJ, Sundsmo M, Kittel A, Saitoh T, Miller A, Masliah E (1997) NACP, a synaptic protein involved in Alzheimer’s disease, is differentially regulated during megakaryocyte differentiation Biochem Biophys Res Commun 237: 611 616 |
43 | Li QX, Campbell BC, McLean CA, Thyagarajan D, Gai WP, Kapsa RM, Beyreuther K, Masters CL, Culvenor JG (2002) Platelet alpha- and gamma-synucleins in Parkinson’s disease and normal control subjects J Alzheimers Dis 4: 309 315 |
44 | Lavedan C, Leroy E, Torres R, Dehejia A, Dutra A, Buchholtz S, Nussbaum RL, Polymeropoulos MH (1998) Genomic organization and expression of the human beta-synuclein gene (SNCB) Genomics 54: 173 175 |
45 | Nakajo S, Tsukada K, Omata K, Nakamura Y, Nakaya K (1993) A new brain-specific 14-kDa protein is a phosphoprotein. Its complete amino acid sequence and evidence for phosphorylation Eur J Biochem 217: 1057 1063 |
46 | Ninkina N, Peters OM, Connor-Robson N, Lytkina O, Sharfeddin E, Buchman VL (2012) Contrasting effects of alpha-synuclein and gamma-synuclein on the phenotype of cysteine string protein alpha (CSPalpha) null mutant mice suggest distinct function of these proteins in neuronal synapses J Biol Chem 287: 44471 44477 |
47 | Ninkina NN, Alimova-Kost MV, Paterson JW, Delaney L, Cohen BB, Imreh S, Gnuchev NV, Davies AM, Buchman VL (1998) Organization, expression and polymorphism of the human persyn gene Hum Mol Genet 7: 1417 1424 |
48 | Ohtake H, Limprasert P, Fan Y, Onodera O, Kakita A, Takahashi H, Bonner LT, Tsuang DW, Murray IV, Lee VM, Trojanowski JQ, Ishikawa A, Idezuka J, Murata M, Toda T, Bird TD, Leverenz JB, Tsuji S, La Spada AR (2004) Beta-synuclein gene alterations in dementia with Lewy bodies Neurology 63: 805 811 |
49 | Fujita M, Sugama S, Sekiyama K, Sekigawa A, Tsukui T, Nakai M, Waragai M, Takenouchi T, Takamatsu Y, Wei J, Rockenstein E, Laspada AR, Masliah E, Inoue S, Hashimoto M (2010) A beta-synuclein mutation linked to dementia produces neurodegeneration when expressed in mouse brain Nat Commun 1: 110 |
50 | Ninkina N, Peters O, Millership S, Salem H, van der Putten H, Buchman VL (2009) Gamma-synucleinopathy: Neurodegeneration associated with overexpression of the mouse protein Hum Mol Genet 18: 1779 1794 |
51 | Peters OM, Millership S, Shelkovnikova TA, Soto I, Keeling L, Hann A, Marsh-Armstrong N, Buchman VL, Ninkina N (2012) Selective pattern of motor system damage in gamma-synuclein transgenic mice mirrors the respective pathology in amyotrophic lateral sclerosis Neurobiol Dis 48: 124 131 |
52 | Rockenstein E, Hansen LA, Mallory M, Trojanowski JQ, Galasko D, Masliah E (2001) Altered expression of the synuclein family mRNA in Lewy body and Alzheimer’s disease Brain Res 914: 48 56 |
53 | Myerowitz R, Mizukami H, Richardson KL, Finn LS, Tifft CJ, Proia RL (2004) Global gene expression in a type 2 Gaucher disease brain Mol Genet Metab 83: 288 296 |
54 | Peters OM, Shelkovnikova T, Highley JR, Cooper-Knock J, Hortobagyi T, Troakes C, Ninkina N, Buchman VL (2015) Gamma-synuclein pathology in amyotrophic lateral sclerosis Ann Clin Transl Neurol 2: 29 37 |
55 | Kholodilov NG, Neystat M, Oo TF, Lo SE, Larsen KE, Sulzer D, Burke RE (1999) Increased expression of rat synuclein in the substantia nigra pars compacta identified by mRNA differential display in a model of developmental target injury J Neurochem 73: 2586 2599 |
56 | Petersen K, Olesen OF, Mikkelsen JD (1999) Developmental expression of alpha-synuclein in rat hippocampus and cerebral cortex Neuroscience 91: 651 659 |
57 | Chu Y, Kordower JH (2007) Age-associated increases of alpha-synuclein in monkeys and humans are associated with nigrostriatal dopamine depletion: Is this the target for Parkinson’s disease? Neurobiol Dis 25: 134 149 |
58 | Withers GS, George JM, Banker GA, Clayton DF (1997) Delayed localization of synelfin (synuclein, NACP) to presynaptic terminals in cultured rat hippocampal neurons Brain Res Dev Brain Res 99: 87 94 |
59 | Hsu LJ, Mallory M, Xia Y, Veinbergs I, Hashimoto M, Yoshimoto M, Thal LJ, Saitoh T, Masliah E (1998) Expression pattern of synucleins (non-Abeta component of Alzheimer’s disease amyloid precursor protein/alpha-synuclein) during murine brain development J Neurochem 71: 338 344 |
60 | Bayer TA, Jakala P, Hartmann T, Egensperger R, Buslei R, Falkai P, Beyreuther K (1999) Neural expression profile of alpha-synuclein in developing human cortex Neuroreport 10: 2799 2803 |
61 | Galvin JE, Schuck TM, Lee VM, Trojanowski JQ (2001) Differential expression and distribution of alpha-, beta-, and gamma-synuclein in the developing human substantia nigra Exp Neurol 168: 347 355 |
62 | Kahle PJ, Neumann M, Ozmen L, Muller V, Jacobsen H, Schindzielorz A, Okochi M, Leimer U, van Der Putten H, Probst A, Kremmer E, Kretzschmar HA, Haass C (2000) Subcellular localization of wild-type and Parkinson’s disease-associated mutant alpha -synuclein in human and transgenic mouse brain J Neurosci 20: 6365 6373 |
63 | Burré J, Sharma M, Tsetsenis T, Buchman V, Etherton MR, Sudhof TC (2010) Alpha-synuclein promotes SNARE-complex assembly in vivo and in vitro Science 329: 1663 1667 |
64 | Zaltieri M, Grigoletto J, Longhena F, Navarria L, Favero G, Castrezzati S, Colivicchi MA, Corte LD, Rezzani R, Pizzi M, Benfenati F, Spillantini MG, Missale C, Spano P, Bellucci A (2015) Alpha-synuclein and synapsin III cooperatively regulate synaptic function in dopamine neurons J Cell Sci 128: 2231 2243 |
65 | Chen RH, Wislet-Gendebien S, Samuel F, Visanji NP, Zhang G, Marsilio D, Langman T, Fraser PE, Tandon A (2013) alpha-Synuclein membrane association is regulated by the Rab3a recycling machinery and presynaptic activity J Biol Chem 288: 7438 7449 |
66 | Murphy DD, Rueter SM, Trojanowski JQ, Lee VM (2000) Synucleins are developmentally expressed, and alpha-synuclein regulates the size of the presynaptic vesicular pool in primary hippocampal neurons J Neurosci 20: 3214 3220 |
67 | George JM (2002) The synucleins Genome Biol 3: REVIEWS3002 |
68 | Bussell RJr, Eliezer D (2003) A structural and functional role for 11-mer repeats in alpha-synuclein and other exchangeable lipid binding proteins J Mol Biol 329: 763 778 |
69 | Davidson WS, Jonas A, Clayton DF, George JM (1998) Stabilization of alpha-synuclein secondary structure upon binding to synthetic membranes J Biol Chem 273: 9443 9449 |
70 | Eliezer D, Kutluay E, Bussell RJr, Browne G (2001) Conformational properties of alpha-synuclein in its free and lipid-associated states J Mol Biol 307: 1061 1073 |
71 | Chandra S, Chen X, Rizo J, Jahn R, Sudhof TC (2003) A broken alpha -helix in folded alpha -Synuclein J Biol Chem 278: 15313 15318 |
72 | Bussell RJr, Ramlall TF, Eliezer D (2005) Helix periodicity, topology, and dynamics of membrane-associated alpha-synuclein Protein Sci 14: 862 872 |
73 | Fusco G, De Simone A, Gopinath T, Vostrikov V, Vendruscolo M, Dobson CM, Veglia G (2014) Direct observation of the three regions in alpha-synuclein that determine its membrane-bound behaviour Nat Commun 5: 3827 |
74 | Appel-Cresswell S, Vilarino-Guell C, Encarnacion M, Sherman H, Yu I, Shah B, Weir D, Thompson C, Szu-Tu C, Trinh J, Aasly JO, Rajput A, Rajput AH, Jon Stoessl A, Farrer MJ (2013) Alpha-synuclein p. H50Q, a novel pathogenic mutation for Parkinson’s disease Mov Disord 28: 811 813 |
75 | Kruger R, Kuhn W, Muller T, Woitalla D, Graeber M, Kosel S, Przuntek H, Epplen JT, Schols L, Riess O (1998) Ala30Pro mutation in the gene encoding alpha-synuclein in Parkinson’s disease Nat Genet 18: 106 108 |
76 | Lesage S, Anheim M, Letournel F, Bousset L, Honore A, Rozas N, Pieri L, Madiona K, Durr A, Melki R, Verny C, Brice A (2013) G51D alpha-synuclein mutation causes a novel parkinsonian-pyramidal syndrome Ann Neurol 73: 459 471 |
77 | Polymeropoulos MH, Lavedan C, Leroy E, Ide SE, Dehejia A, Dutra A, Pike B, Root H, Rubenstein J, Boyer R, Stenroos ES, Chandrasekharappa S, Athanassiadou A, Papapetropoulos T, Johnson WG, Lazzarini AM, Duvoisin RC, Di Iorio G, Golbe LI, Nussbaum RL (1997) Mutation in the alpha-synuclein gene identified in families with Parkinson’s disease Science 276: 2045 2047 |
78 | Proukakis C, Dudzik CG, Brier T, MacKay DS, Cooper JM, Millhauser GL, Houlden H, Schapira AH (2013) A novel alpha-synuclein missense mutation in Parkinson disease Neurology 80: 1062 1064 |
79 | Zarranz JJ, Alegre J, Gomez-Esteban JC, Lezcano E, Ros R, Ampuero I, Vidal L, Hoenicka J, Rodriguez O, Atares B, Llorens V, Gomez Tortosa E, del Ser T, Munoz DG, de Yebenes JG (2004) The new mutation, E46K, of alpha-synuclein causes Parkinson and Lewy body dementia Ann Neurol 55: 164 173 |
80 | Pasanen P, Myllykangas L, Siitonen M, Raunio A, Kaakkola S, Lyytinen J, Tienari PJ, Poyhonen M, Paetau A (2014) Novel alpha-synuclein mutation A53E associated with atypical multiple system atrophy and Parkinson’s disease-type pathology Neurobiol Aging 35: 2180 e2181 e2185 |
81 | Ulmer TS, Bax A, Cole NB, Nussbaum RL (2005) Structure and dynamics of micelle-bound human alpha-synuclein J Biol Chem 280: 9595 9603 |
82 | Oueslati A, Fournier M, Lashuel HA (2010) Role of post-translational modifications in modulating the structure, function and toxicity of alpha-synuclein: Imlications for Parkinson’s disease pathogenesis and therapies Prog Brain Res 183: 115 145 |
83 | Fernandez CO, Hoyer W, Zweckstetter M, Jares-Erijman EA, Subramaniam V, Griesinger C, Jovin TM (2004) NMR of alpha-synuclein-polyamine complexes elucidates the mechanism and kinetics of induced aggregation EMBO J 23: 2039 2046 |
84 | Brown DR (2007) Interactions between metals and alpha-synuclein–function or artefact? FEBS J 274: 3766 3774 |
85 | Paik SR, Shin HJ, Lee JH, Chang CS, Kim J (1999) Copper(II)-induced self-oligomerization of alpha-synuclein Biochem J 340: Pt 3 821 828 |
86 | Nielsen MS, Vorum H, Lindersson E, Jensen PH (2001) Ca2+binding to alpha-synuclein regulates ligand binding and oligomerization J Biol Chem 276: 22680 22684 |
87 | Bodner CR, Dobson CM, Bax A (2009) Multiple tight phospholipid-binding modes of alpha-synuclein revealed by solution NMR spectroscopy J Mol Biol 390: 775 790 |
88 | Sevcsik E, Trexler AJ, Dunn JM, Rhoades E (2011) Allostery in a disordered protein: Oxidative modifications to alha-synuclein act distally to regulate membrane binding J Am Chem Soc 133: 7152 7158 |
89 | Hoyer W, Cherny D, Subramaniam V, Jovin TM (2004) Impact of the acidic C-terminal region comprising amino acids 109-140 on alpha-synuclein aggregation in vitro Biochemistry 43: 16233 16242 |
90 | Park SM, Jung HY, Kim TD, Park JH, Yang CH, Kim J (2002) Distinct roles of the N-terminal-binding domain and the C-terminal-solubilizing domain of alpha-synuclein, a molecular chaperone J Biol Chem 277: 28512 28520 |
91 | Crowther RA, Jakes R, Spillantini MG, Goedert M (1998) Synthetic filaments assembled from C-terminally truncated alpha-synuclein FEBS Lett 436: 309 312 |
92 | Weinreb PH, Zhen W, Poon AW, Conway KA, Lansbury PTJr (1996) NACP, a protein implicated in Alzheimer’s disease and learning, is natively unfolded Biochemistry 35: 13709 13715 |
93 | Kim J (1997) Evidence that the precursor protein of non-A beta component of Alzheimer’s disease amyloid (NACP) has an extended structure primarily composed of random-coil Mol Cells 7: 78 83 |
94 | Fauvet B, Mbefo MK, Fares MB, Desobry C, Michael S, Ardah MT, Tsika E, Coune P, Prudent M, Lion N, Eliezer D, Moore DJ, Schneider B, Aebischer P, El-Agnaf OM, Masliah E, Lashuel HA (2012) alpha-Synuclein in central nervous system and from erythrocytes, mammalian cells, and Escherichia coli exists predominantly as disordered monomer J Biol Chem 287: 15345 15364 |
95 | Burré J, Vivona S, Diao J, Sharma M, Brunger AT, Sudhof TC (2013) Properties of native brain alpha-synuclein Nature 498: E4 E6 discussion E6-7 |
96 | Bartels T, Choi JG, Selkoe DJ (2011) alpha-Synuclein occurs physiologically as a helically folded tetramer that resists aggregation Nature 477: 107 110 |
97 | Wang W, Perovic I, Chittuluru J, Kaganovich A, Nguyen LT, Liao J, Auclair JR, Johnson D, Landeru A, Simorellis AK, Ju S, Cookson MR, Asturias FJ, Agar JN, Webb BN, Kang C, Ringe D, Petsko GA, Pochapsky TC, Hoang QQ (2011) A soluble alpha-synuclein construct forms a dynamic tetramer Proc Natl Acad Sci U S A 108: 17797 17802 |
98 | Binolfi A, Theillet FX, Selenko P (2012) Bacterial in-cell NMR of human alpha-synuclein: A disordered monomer by nature? Biochem Soc Trans 40: 950 954 |
99 | Smaldone G, Diana D, Pollegioni L, Di Gaetano S, Fattorusso R, Pedone E (2015) Insight into conformational modification of alpha-synuclein in the presence of neuronal whole cells and of their isolated membranes FEBS Lett 589: 798 804 |
100 | Burré J, Sharma M, Sudhof TC (2014) alpha-Synuclein assembles into higher-order multimers upon membrane binding to promote SNARE complex formation Proc Natl Acad Sci U S A 111: E4274 E4283 |
101 | Luth ES, Bartels T, Dettmer U, Kim NC, Selkoe DJ (2015) Purification of alpha-synuclein from human brain reveals an instability of endogenous multimers as the protein approaches purity Biochemistry 54: 279 292 |
102 | Middleton ER, Rhoades E (2010) Effects of curvature and composition on alpha-synuclein binding to lipid vesicles Biophys J 99: 2279 2288 |
103 | Jo E, McLaurin J, Yip CM, St George-Hyslop P, Fraser PE (2000) alpha-Synuclein membrane interactions and lipid specificity J Biol Chem 275: 34328 34334 |
104 | Perrin RJ, Woods WS, Clayton DF, George JM (2000) Interaction of human alpha-Synuclein and Parkinson’s disease variants with phospholipids. Structural analysis using site-directed mutagenesis J Biol Chem 275: 34393 34398 |
105 | Kubo S, Nemani VM, Chalkley RJ, Anthony MD, Hattori N, Mizuno Y, Edwards RH, Fortin DL (2005) A combinatorial code for the interaction of alpha-synuclein with membranes J Biol Chem 280: 31664 31672 |
106 | Bisaglia M, Schievano E, Caporale A, Peggion E, Mammi S (2006) The 11-mer repeats of human alpha-synuclein in vesicle interactions and lipid composition discrimination: A cooperative role Biopolymers 84: 310 316 |
107 | Georgieva ER, Ramlall TF, Borbat PP, Freed JH, Eliezer D (2010) The lipid-binding domain of wild type and mutant alpha-synuclein: Comactness and interconversion between the broken and extended helix forms J Biol Chem 285: 28261 28274 |
108 | Trexler AJ, Rhoades E (2009) Alpha-synuclein binds large unilamellar vesicles as an extended helix Biochemistry 48: 2304 2306 |
109 | Jao CC, Der-Sarkissian A, Chen J, Langen R (2004) Structure of membrane-bound alpha-synuclein studied by site-directed spin labeling Proc Natl Acad Sci U S A 101: 8331 8336 |
110 | Georgieva ER, Ramlall TF, Borbat PP, Freed JH, Eliezer D (2008) Membrane-bound alpha-synuclein forms an extended helix: Long-distance pulsed ESR measurements using vesicles, bicelles, and rodlike micelles J Am Chem Soc 130: 12856 12857 |
111 | Jao CC, Hegde BG, Chen J, Haworth IS, Langen R (2008) Structure of membrane-bound alpha-synuclein from site-directed spin labeling and computational refinement Proc Natl Acad Sci U S A 105: 19666 19671 |
112 | Borbat P, Ramlall TF, Freed JH, Eliezer D (2006) Inter-helix distances in lysophospholipid micelle-bound alpha-synuclein from pulsed ESR measurements J Am Chem Soc 128: 10004 10005 |
113 | Drescher M, Veldhuis G, van Rooijen BD, Milikisyants S, Subramaniam V, Huber M (2008) Antiparallel arrangement of the helices of vesicle-bound alpha-synuclein J Am Chem Soc 130: 7796 7797 |
114 | Nuscher B, Kamp F, Mehnert T, Odoy S, Haass C, Kahle PJ, Beyer K (2004) Alpha-synuclein has a high affinity for packing defects in a bilayer membrane: A thermodynamics study J Biol Chem 279: 21966 21975 |
115 | Kang L, Moriarty GM, Woods LA, Ashcroft AE, Radford SE, Baum J (2012) N-terminal acetylation of alpha-synuclein induces increased transient helical propensity and decreased aggregation rates in the intrinsically disordered monomer Protein Sci 21: 911 917 |
116 | Maltsev AS, Ying J, Bax A (2012) Impact of N-terminal acetylation of alpha-synuclein on its random coil and lipid binding properties Biochemistry 51: 5004 5013 |
117 | Dikiy I, Eliezer D (2014) N-terminal acetylation stabilizes N-terminal helicity in lipid- and micelle-bound alpha-synuclein and increases its affinity for physiological membranes J Biol Chem 289: 3652 3665 |
118 | Bartels T, Kim NC, Luth ES, Selkoe DJ (2014) N-alpha-acetylation of alpha-synuclein increases its helical folding propensity, GM1 binding specificity and resistance to aggregation PLoS One 9: e103727 |
119 | Trexler AJ, Rhoades E (2012) N-Terminal acetylation is critical for forming alpha-helical oligomer of alpha-synuclein Protein Sci 21: 601 605 |
120 | Fujiwara H, Hasegawa M, Dohmae N, Kawashima A, Masliah E, Goldberg MS, Shen J, Takio K, Iwatsubo T (2002) alpha-Synuclein is phosphorylated in synucleinopathy lesions Nat Cell Biol 4: 160 164 |
121 | McFarland MA, Ellis CE, Markey SP, Nussbaum RL (2008) Proteomics analysis identifies phosphorylation-dependent alpha-synuclein protein interactions Mol Cell Proteomics 7: 2123 2137 |
122 | Chen L, Feany MB (2005) Alpha-synuclein phosphorylation controls neurotoxicity and inclusion formation in a Drosophila model of Parkinson disease Nat Neurosci 8: 657 663 |
123 | Paleologou KE, Oueslati A, Shakked G, Rospigliosi CC, Kim HY, Lamberto GR, Fernandez CO, Schmid A, Chegini F, Gai WP, Chiappe D, Moniatte M, Schneider BL, Aebischer P, Eliezer D, Zweckstetter M, Masliah E, Lashuel HA (2010) Phosphorylation at S87 is enhanced in synucleinopathies, inhibits alpha-synuclein oligomerization, and influences synuclein-membrane interactions J Neurosci 30: 3184 3198 |
124 | Chen L, Periquet M, Wang X, Negro A, McLean PJ, Hyman BT, Feany MB (2009) Tyrosine and serine phosphorylation of alpha-synuclein have opposing effects on neurotoxicity and soluble oligomer formation J Clin Invest 119: 3257 3265 |
125 | Paleologou KE, Schmid AW, Rospigliosi CC, Kim HY, Lamberto GR, Fredenburg RA, Lansbury PTJr, Fernandez CO, Eliezer D, Zweckstetter M, Lashuel HA (2008) Phosphorylation at Ser-129 but not the phosphomimics S129E/D inhibits the fibrillation of alpha-synuclein J Biol Chem 283: 16895 16905 |
126 | Nonaka T, Iwatsubo T, Hasegawa M (2005) Ubiquitination of alpha-synuclein Biochemistry 44: 361 368 |
127 | Rott R, Szargel R, Haskin J, Shani V, Shainskaya A, Manov I, Liani E, Avraham E, Engelender S (2008) Monoubiquitylation of alpha-synuclein by seven in absentia homolog (SIAH) promotes its aggregation in dopaminergic cells J Biol Chem 283: 3316 3328 |
128 | Dorval V, Fraser PE (2006) Small ubiquitin-like modifier (SUMO) modification of natively unfolded proteins tau and alpha-synuclein J Biol Chem 281: 9919 9924 |
129 | Krumova P, Meulmeester E, Garrido M, Tirard M, Hsiao HH, Bossis G, Urlaub H, Zweckstetter M, Kugler S, Melchior F, Bahr M, Weishaupt JH (2011) Sumoylation inhibits alpha-synuclein aggregation and toxicity J Cell Biol 194: 49 60 |
130 | Munch G, Luth HJ, Wong A, Arendt T, Hirsch E, Ravid R, Riederer P (2000) Crosslinking of alpha-synuclein by advanced glycation endproducts–an early pathophysiological step in Lewy body formation? J Chem Neuroanat 20: 253 257 |
131 | Shaikh S, Nicholson LF (2008) Advanced glycation end products induce in vitro cross-linking of alpha-synuclein and accelerate the process of intracellular inclusion body formation J Neurosci Res 86: 2071 2082 |
132 | Padmaraju V, Bhaskar JJ, Prasada Rao UJ, Salimath PV, Rao KS (2011) Role of advanced glycation on aggregation and DNA binding properties of alpha-synuclein J Alzheimers Dis 24: Suppl 2 211 221 |
133 | Alfaro JF, Gong CX, Monroe ME, Aldrich JT, Clauss TR, Purvine SO, Wang Z, Camp DG2nd, Shabanowitz J, Stanley P, Hart GW, Hunt DF, Yang F, Smith RD (2012) Tandem mass spectrometry identifies many mouse brain O-GlcNAcylated proteins including EGF domain-specific O-GlcNAc transferase targets Proc Natl Acad Sci U S A 109: 7280 7285 |
134 | Marotta NP, Cherwien CA, Abeywardana T, Pratt MR (2012) O-GlcNAc modification prevents peptide-dependent acceleration of alpha-synuclein aggregation Chembiochem 13: 2665 2670 |
135 | Giasson BI, Duda JE, Murray IV, Chen Q, Souza JM, Hurtig HI, Ischiropoulos H, Trojanowski JQ, Lee VM (2000) Oxidative damage linked to neurodegeneration by selective alpha-synuclein nitration in synucleinopathy lesions Science 290: 985 989 |
136 | Hodara R, Norris EH, Giasson BI, Mishizen-Eberz AJ, Lynch DR, Lee VM, Ischiropoulos H (2004) Functional consequences of alpha-synuclein tyrosine nitration: Diminished binding to lipid vesicles and increased fibril formation J Biol Chem 279: 47746 47753 |
137 | Yamin G, Uversky VN, Fink AL (2003) Nitration inhibits fibrillation of human alpha-synuclein in vitro by formation of soluble oligomers FEBS Lett 542: 147 152 |
138 | Li W, West N, Colla E, Pletnikova O, Troncoso JC, Marsh L, Dawson TM, Jakala P, Hartmann T, Price DL, Lee MK (2005) Aggregation promoting C-terminal truncation of alpha-synuclein is a normal cellular process and is enhanced by the familial Parkinson’s disease-linked mutations Proc Natl Acad Sci U S A 102: 2162 2167 |
139 | Murray IV, Giasson BI, Quinn SM, Koppaka V, Axelsen PH, Ischiropoulos H, Trojanowski JQ, Lee VM (2003) Role of alpha-synuclein carboxy-terminus on fibril formation in vitro Biochemistry 42: 8530 8540 |
140 | Lokappa SB, Suk JE, Balasubramanian A, Samanta S, Situ AJ, Ulmer TS (2014) Sequence and membrane determinants of the random coil-helix transition of alpha-synuclein J Mol Biol 426: 2130 2144 |
141 | Zhu M, Fink AL (2003) Lipid binding inhibits alpha-synuclein fibril formation J Biol Chem 278: 16873 16877 |
142 | Burré J, Sharma M, Sudhof TC (2015) Definition of a molecular pathway mediating alpha-synuclein neurotoxicity J Neurosci 35: 5221 5232 |
143 | Jo E, Darabie AA, Han K, Tandon A, Fraser PE, McLaurin J (2004) alpha-Synuclein-synaptosomal membrane interactions: Implications for fibrillogenesis Eur J Biochem 271: 3180 3189 |
144 | Narayanan V, Scarlata S (2001) Membrane binding and self-association of alpha-synucleins Biochemistry 40: 9927 9934 |
145 | Lee HJ, Choi C, Lee SJ (2002) Membrane-bound alpha-synuclein has a high aggregation propensity and the ability to seed the aggregation of the cytosolic form J Biol Chem 277: 671 678 |
146 | Wang L, Das U, Scott DA, Tang Y, McLean PJ, Roy S (2014) alpha-synuclein multimers cluster synaptic vesicles and attenuate recycling Curr Biol 24: 2319 2326 |
147 | Conway KA, Harper JD, Lansbury PT (1998) Accelerated in vitro fibril formation by a mutant alpha-synuclein linked to early-onset Parkinson disease Nat Med 4: 1318 1320 |
148 | El-Agnaf OM, Jakes R, Curran MD, Middleton D, Ingenito R, Bianchi E, Pessi A, Neill D, Wallace A (1998) Aggregates from mutant and wild-type alpha-synuclein proteins and NAC peptide induce apoptotic cell death in human neuroblastoma cells by formation of beta-sheet and amyloid-like filaments FEBS Lett 440: 71 75 |
149 | Conway KA, Harper JD, Lansbury PTJr (2000) Fibrils formed in vitro from alpha-synuclein and two mutant forms linked to Parkinson’s disease are typical amyloid Biochemistry 39: 2552 2563 |
150 | Uversky VN (2007) Neuropathology, biochemistry, and biophysics of alpha-synuclein aggregation J Neurochem 103: 17 37 |
151 | Lashuel HA, Petre BM, Wall J, Simon M, Nowak RJ, Walz T, Lansbury PTJr (2002) Alpha-synuclein, especially the Parkinson’s disease-associated mutants, forms pore-like annular and tubular protofibrils J Mol Biol 322: 1089 1102 |
152 | Singleton AB, Farrer M, Johnson J, Singleton A, Hague S, Kachergus J, Hulihan M, Peuralinna T, Dutra A, Nussbaum R, Lincoln S, Crawley A, Hanson M, Maraganore D, Adler C, Cookson MR, Muenter M, Baptista M, Miller D, Blancato J, Hardy J, Gwinn-Hardy K (2003) alpha-Synuclein locus triplication causes Parkinson’s disease Science 302: 841 |
153 | Ibanez P, Bonnet AM, Debarges B, Lohmann E, Tison F, Pollak P, Agid Y, Durr A, Brice A (2004) Causal relation between alpha-synuclein gene duplication and familial Parkinson’s disease Lancet 364: 1169 1171 |
154 | Masliah E, Rockenstein E, Veinbergs I, Mallory M, Hashimoto M, Takeda A, Sagara Y, Sisk A, Mucke L (2000) Dopaminergic loss and inclusion body formation in alpha-synuclein mice: Implications for neurodegenerative disorders Science 287: 1265 1269 |
155 | Kahle PJ, Neumann M, Ozmen L, Haass C (2000) Physiology and pathophysiology of alpha-synuclein. Cell culture and transgenic animal models based on a Parkinson’s disease-associated protein Ann N Y Acad Sci 920: 33 41 |
156 | Rockenstein E, Mallory M, Hashimoto M, Song D, Shults CW, Lang I, Masliah E (2002) Differential neuropathological alterations in transgenic mice expressing alpha-synuclein from the platelet-derived growth factor and Thy-1 promoters J Neurosci Res 68: 568 578 |
157 | Abeliovich A, Schmitz Y, Farinas I, Choi-Lundberg D, Ho WH, Castillo PE, Shinsky N, Verdugo JM, Armanini M, Ryan A, Hynes M, Phillips H, Sulzer D, Rosenthal A (2000) Mice lacking alpha-synuclein display functional deficits in the nigrostriatal dopamine system Neuron 25: 239 252 |
158 | Cabin DE, Shimazu K, Murphy D, Cole NB, Gottschalk W, McIlwain KL, Orrison B, Chen A, Ellis CE, Paylor R, Lu B, Nussbaum RL (2002) Synaptic vesicle depletion correlates with attenuated synaptic responses to prolonged repetitive stimulation in mice lacking alpha-synuclein J Neurosci 22: 8797 8807 |
159 | Burré J, Sharma M, Sudhof TC (2012) Systematic mutagenesis of alpha-synuclein reveals distinct sequence requirements for physiological and pathological activities J Neurosci 32: 15227 15242 |
160 | Gorbatyuk OS, Li S, Nguyen FN, Manfredsson FP, Kondrikova G, Sullivan LF, Meyers C, Chen W, Mandel RJ, Muzyczka N (2010) alpha-Synuclein expression in rat substantia nigra suppresses phospholipase D2 toxicity and nigral neurodegeneration Mol Ther 18: 1758 1768 |
161 | Jenco JM, Rawlingson A, Daniels B, Morris AJ (1998) Regulation of phospholipase D2: Selective inhibition of mammalian phospholipase D isoenzymes by alpha- and beta-synucleins Biochemistry 37: 4901 4909 |
162 | Payton JE, Perrin RJ, Woods WS, George JM (2004) Structural determinants of PLD2 inhibition by alpha-synuclein J Mol Biol 337: 1001 1009 |
163 | Rappley I, Gitler AD, Selvy PE, LaVoie MJ, Levy BD, Brown HA, Lindquist S, Selkoe DJ (2009) Evidence that alpha-synuclein does not inhibit phospholipase D Biochemistry 48: 1077 1083 |
164 | Guo JT, Chen AQ, Kong Q, Zhu H, Ma CM, Qin C (2008) Inhibition of vesicular monoamine transporter-2 activity in alpha-synuclein stably transfected SH-SY5Y cells Cell Mol Neurobiol 28: 35 47 |
165 | Wersinger C, Rusnak M, Sidhu A (2006) Modulation of the trafficking of the human serotonin transporter by human alpha-synuclein Eur J Neurosci 24: 55 64 |
166 | Butler B, Goodwin S, Saha K, Becker J, Sambo D, Davari P, Khoshbouei H (2015) Dopamine Transporter Activity Is Modulated by alpha-synuclein J Biol Chem |
167 | Swant J, Goodwin JS, North A, Ali AA, Gamble-George J, Chirwa S, Khoshbouei H (2011) alpha-Synuclein stimulates a dopamine transporter-dependent chloride current and modulates the activity of the transporter J Biol Chem 286: 43933 43943 |
168 | Perez RG, Waymire JC, Lin E, Liu JJ, Guo F, Zigmond MJ (2002) A role for alpha-synuclein in the regulation of dopamine biosynthesis J Neurosci 22: 3090 3099 |
169 | Baptista MJ, O’Farrell C, Daya S, Ahmad R, Miller DW, Hardy J, Farrer MJ, Cookson MR (2003) Co-ordinate transcriptional regulation of dopamine synthesis genes by alpha-synuclein in human neuroblastoma cell lines J Neurochem 85: 957 968 |
170 | Yu S, Zuo X, Li Y, Zhang C, Zhou M, Zhang YA, Ueda K, Chan P (2004) Inhibition of tyrosine hydroxylase expression in alpha-synuclein-transfected dopaminergic neuronal cells Neurosci Lett 367: 34 39 |
171 | Devi L, Raghavendran V, Prabhu BM, Avadhani NG, Anandatheerthavarada HK (2008) Mitochondrial import and accumulation of alpha-synuclein impair complex I in human dopaminergic neuronal cultures and Parkinson disease brain J Biol Chem 283: 9089 9100 |
172 | Li WW, Yang R, Guo JC, Ren HM, Zha XL, Cheng JS, Cai DF (2007) Localization of alpha-synuclein to mitochondria within midbrain of mice Neuroreport 18: 1543 1546 |
173 | Nakamura K, Nemani VM, Wallender EK, Kaehlcke K, Ott M, Edwards RH (2008) Optical reporters for the conformation of alpha-synuclein reveal a specific interaction with mitochondria J Neurosci 28: 12305 12317 |
174 | Thayanidhi N, Helm JR, Nycz DC, Bentley M, Liang Y, Hay JC (2010) Alpha-synuclein delays endoplasmic reticulum (ER)-to-Golgi transport in mammalian cells by antagonizing ER/Golgi SNAREs Mol Biol Cell 21: 1850 1863 |
175 | Cooper AA, Gitler AD, Cashikar A, Haynes CM, Hill KJ, Bhullar B, Liu K, Xu K, Strathearn KE, Liu F, Cao S, Caldwell KA, Caldwell GA, Marsischky G, Kolodner RD, Labaer J, Rochet JC, Bonini NM, Lindquist S (2006) Alpha-synuclein blocks ER-Golgi traffic and Rab1 rescues neuron loss in Parkinson’s models Science 313: 324 328 |
176 | Kontopoulos E, Parvin JD, Feany MB (2006) Alpha-synuclein acts in the nucleus to inhibit histone acetylation and promote neurotoxicity Hum Mol Genet 15: 3012 3023 |
177 | Golovko MY, Faergeman NJ, Cole NB, Castagnet PI, Nussbaum RL, Murphy EJ (2005) Alpha-synuclein gene deletion decreases brain palmitate uptake and alters the palmitate metabolism in the absence of alpha-synuclein palmitate binding Biochemistry 44: 8251 8259 |
178 | Ellis CE, Murphy EJ, Mitchell DC, Golovko MY, Scaglia F, Barcelo-Coblijn GC, Nussbaum RL (2005) Mitochondrial lipid abnormality and electron transport chain impairment in mice lacking alpha-synuclein Mol Cell Biol 25: 10190 10201 |
179 | Barcelo-Coblijn G, Golovko MY, Weinhofer I, Berger J, Murphy EJ (2007) Brain neutral lipids mass is increased in alpha-synuclein gene-ablated mice J Neurochem 101: 132 141 |
180 | Rappley I, Myers DS, Milne SB, Ivanova PT, Lavoie MJ, Brown HA, Selkoe DJ (2009) Lipidomic profiling in mouse brain reveals differences between ages and genders, with smaller changes associated with alpha-synuclein genotype J Neurochem 111: 15 25 |
181 | Sharon R, Goldberg MS, Bar-Josef I, Betensky RA, Shen J, Selkoe DJ (2001) alpha-Synuclein occurs in lipid-rich high molecular weight complexes, binds fatty acids, and shows homology to the fatty acid-binding proteins Proc Natl Acad Sci U S A 98: 9110 9115 |
182 | Lucke C, Gantz DL, Klimtchuk E, Hamilton JA (2006) Interactions between fatty acids and alpha-synuclein J Lipid Res 47: 1714 1724 |
183 | Varkey J, Isas JM, Mizuno N, Jensen MB, Bhatia VK, Jao CC, Petrlova J, Voss JC, Stamou DG, Steven AC, Langen R (2010) Membrane curvature induction and tubulation are common features of synucleins and apolipoproteins J Biol Chem 285: 32486 32493 |
184 | Westphal CH, Chandra SS (2013) Monomeric synucleins generate membrane curvature J Biol Chem 288: 1829 1840 |
185 | Jiang Z, de Messieres M, Lee JC (2013) Membrane remodeling by alpha-synuclein and effects on amyloid formation J Am Chem Soc 135: 15970 15973 |
186 | Mizuno N, Varkey J, Kegulian NC, Hegde BG, Cheng N, Langen R, Steven AC (2012) Remodeling of lipid vesicles into cylindrical micelles by alpha-synuclein in an extended alpha-helical conformation J Biol Chem 287: 29301 29311 |
187 | Shi Z, Sachs JN, Rhoades E, Baumgart T (2015) Biophysics of alpha-synuclein induced membrane remodelling Phys Chem Chem Phys 17: 15561 15568 |
188 | Braun AR, Lacy MM, Ducas VC, Rhoades E, Sachs JN (2014) alpha-Synuclein-induced membrane remodeling is driven by binding affinity, partition depth, and interleaflet order asymmetry J Am Chem Soc 136: 9962 9972 |
189 | Madine J, Doig AJ, Middleton DA (2006) A study of the regional effects of alpha-synuclein on the organization and stability of phospholipid bilayers Biochemistry 45: 5783 5792 |
190 | Adamczyk A, Kacprzak M, Kazmierczak A (2007) Alpha-synuclein decreases arachidonic acid incorporation into rat striatal synaptoneurosomes Folia Neuropathol 45: 230 235 |
191 | Kamp F, Beyer K (2006) Binding of alpha-synuclein affects the lipid packing in bilayers of small vesicles J Biol Chem 281: 9251 9259 |
192 | Ouberai MM, Wang J, Swann MJ, Galvagnion C, Guilliams T, Dobson CM, Welland ME (2013) alpha-Synuclein senses lipid packing defects and induces lateral expansion of lipids leading to membrane remodeling J Biol Chem 288: 20883 20895 |
193 | Braun AR, Sevcsik E, Chin P, Rhoades E, Tristram-Nagle S, Sachs JN (2012) alpha-Synuclein induces both positive mean curvature and negative Gaussian curvature in membranes J Am Chem Soc 134: 2613 2620 |
194 | Braak H, Sastre M, Bohl JR, de Vos RA, Del Tredici K (2007) Parkinson’s disease: Lesions in dorsal horn layer I, involvement of parasympathetic and sympathetic pre- and postganglionic neurons Acta Neuropathol 113: 421 429 |
195 | Baba T, Kikuchi A, Hirayama K, Nishio Y, Hosokai Y, Kanno S, Hasegawa T, Sugeno N, Konno M, Suzuki K, Takahashi S, Fukuda H, Aoki M, Itoyama Y, Mori E, Takeda A (2012) Severe olfactory dysfunction is a prodromal symptom of dementia associated with Parkinson’s disease: A 3 year longitudinal study Brain 135: 161 169 |
196 | Sulzer D, Surmeier DJ (2013) Neuronal vulnerability, pathogenesis, and Parkinson’s disease Mov Disord 28: 715 724 |
197 | Fahn S, Sulzer D (2004) Neurodegeneration and neuroprotection in Parkinson disease NeuroRx 1: 139 154 |
198 | Riederer P, Wuketich S (1976) Time course of nigrostriatal degeneration in parkinson’s disease. A detailed study of influential factors in human brain amine analysis J Neural Transm 38: 277 301 |
199 | Bernheimer H, Birkmayer W, Hornykiewicz O, Jellinger K, Seitelberger F (1973) Brain dopamine and the syndromes of Parkinson and Huntington. Clinical, morphological and neurochemical correlations J Neurol Sci 20: 415 455 |
200 | Oaks AW, Frankfurt M, Finkelstein DI, Sidhu A (2013) Age-dependent effects of A53T alpha-synuclein on behavior and dopaminergic function PLoS One 8: e60378 |
201 | Wersinger C, Sidhu A (2003) Attenuation of dopamine transporter activity by alpha-synuclein Neurosci Lett 340: 189 192 |
202 | Wersinger C, Prou D, Vernier P, Sidhu A (2003) Modulation of dopamine transporter function by alpha-synuclein is altered by impairment of cell adhesion and by induction of oxidative stress FASEB J 17: 2151 2153 |
203 | Lee FJ, Liu F, Pristupa ZB, Niznik HB (2001) Direct binding and functional coupling of alpha-synuclein to the dopamine transporters accelerate dopamine-induced apoptosis FASEB J 15: 916 926 |
204 | Li YH, Gao N, Ye YW, Li X, Yu S, Yang H, Ueda K, Chan P (2011) Alpha-synuclein functions as a negative regulator for expression of tyrosine hydroxylase Acta Neurol Belg 111: 130 135 |
205 | Peng X, Tehranian R, Dietrich P, Stefanis L, Perez RG (2005) Alpha-synuclein activation of protein phosphatase 2A reduces tyrosine hydroxylase phosphorylation in dopaminergic cells J Cell Sci 118: 3523 3530 |
206 | Wu B, Liu Q, Duan C, Li Y, Yu S, Chan P, Ueda K, Yang H (2011) Phosphorylation of alpha-synuclein upregulates tyrosine hydroxylase activity in MN9D cells Acta Histochem 113: 32 35 |
207 | Lou H, Montoya SE, Alerte TN, Wang J, Wu J, Peng X, Hong CS, Friedrich EE, Mader SA, Pedersen CJ, Marcus BS, McCormack AL, Di Monte DA, Daubner SC, Perez RG (2010) Serine 129 phosphorylation reduces the ability of alpha-synuclein to regulate tyrosine hydroxylase and protein phosphatase 2A in vitro and in vivo J Biol Chem 285: 17648 17661 |
208 | Chadchankar H, Ihalainen J, Tanila H, Yavich L (2011) Decreased reuptake of dopamine in the dorsal striatum in the absence of alpha-synuclein Brain Res 1382: 37 44 |
209 | Al-Wandi A, Ninkina N, Millership S, Williamson SJ, Jones PA, Buchman VL (2010) Absence of alpha-synuclein affects dopamine metabolism and synaptic markers in the striatum of aging mice Neurobiol Aging 31: 796 804 |
210 | Garcia-Reitboeck P, Anichtchik O, Dalley JW, Ninkina N, Tofaris GK, Buchman VL, Spillantini MG (2013) Endogenous alpha-synuclein influences the number of dopaminergic neurons in mouse substantia nigra Exp Neurol 248: 541 545 |
211 | Robertson DC, Schmidt O, Ninkina N, Jones PA, Sharkey J, Buchman VL (2004) Developmental loss and resistance to MPTP toxicity of dopaminergic neurones in substantia nigra pars compacta of gamma-synuclein, alpha-synuclein and double alpha/gamma-synuclein null mutant mice J Neurochem 89: 1126 1136 |
212 | Chandra S, Fornai F, Kwon HB, Yazdani U, Atasoy D, Liu X, Hammer RE, Battaglia G, German DC, Castillo PE, Sudhof TC (2004) Double-knockout mice for alpha- and beta-synucleins: Effect on synaptic functions Proc Natl Acad Sci U S A 101: 14966 14971 |
213 | Senior SL, Ninkina N, Deacon R, Bannerman D, Buchman VL, Cragg SJ, Wade-Martins R (2008) Increased striatal dopamine release and hyperdopaminergic-like behaviour in mice lacking both alpha-synuclein and gamma-synuclein Eur J Neurosci 27: 947 957 |
214 | Ostrerova N, Petrucelli L, Farrer M, Mehta N, Choi P, Hardy J, Wolozin B (1999) alpha-Synuclein shares physical and functional homology with 14-3-3 proteins J Neurosci 19: 5782 5791 |
215 | Kim TD, Paik SR, Yang CH, Kim J (2000) Structural changes in alpha-synuclein affect its chaperone-like activity Protein Sci 9: 2489 2496 |
216 | Ahn M, Kim S, Kang M, Ryu Y, Kim TD (2006) Chaperone-like activities of alpha-synuclein: Alha-synuclein assists enzyme activities of esterases Biochem Biophys Res Commun 346: 1142 1149 |
217 | Rekas A, Adda CG, Andrew Aquilina J, Barnham KJ, Sunde M, Galatis D, Williamson NA, Masters CL, Anders RF, Robinson CV, Cappai R, Carver JA (2004) Interaction of the molecular chaperone alphaB-crystallin with alpha-synuclein: Effects on amyloid fibril formation and chaerone activity J Mol Biol 340: 1167 1183 |
218 | Kim TD, Paik SR, Yang CH (2002) Structural and functional implications of C-terminal regions of alpha-synuclein Biochemistry 41: 13782 13790 |
219 | Kim TD, Choi E, Rhim H, Paik SR, Yang CH (2004) Alpha-synuclein has structural and functional similarities to small heat shock proteins Biochem Biophys Res Commun 324: 1352 1359 |
220 | Kanda S, Bishop JF, Eglitis MA, Yang Y, Mouradian MM (2000) Enhanced vulnerability to oxidative stress by alpha-synuclein mutations and C-terminal truncation Neuroscience 97: 279 284 |
221 | da Costa CA, Ancolio K, Checler F (2000) Wild-type but not Parkinson’s disease-related ala-53 –>Thr mutant alpha -synuclein protects neuronal cells from apoptotic stimuli J Biol Chem 275: 24065 24069 |
222 | Chandra S, Gallardo G, Fernandez-Chacon R, Schluter OM, Sudhof TC (2005) Alpha-synuclein cooperates with CSPalpha in preventing neurodegeneration Cell 123: 383 396 |
223 | Greten-Harrison B, Polydoro M, Morimoto-Tomita M, Diao L, Williams AM, Nie EH, Makani S, Tian N, Castillo PE, Buchman VL, Chandra SS (2010) alphabetagamma-Synuclein triple knockout mice reveal age-dependent neuronal dysfunction Proc Natl Acad Sci U S A 107: 19573 19578 |
224 | Anwar S, Peters O, Millership S, Ninkina N, Doig N, Connor-Robson N, Threlfell S, Kooner G, Deacon RM, Bannerman DM, Bolam JP, Chandra SS, Cragg SJ, Wade-Martins R, Buchman VL (2011) Functional alterations to the nigrostriatal system in mice lacking all three members of the synuclein family J Neurosci 31: 7264 7274 |
225 | George JM, Jin H, Woods WS, Clayton DF (1995) Characterization of a novel protein regulated during the critical period for song learning in the zebra finch Neuron 15: 361 372 |
226 | Pelkonen A, Yavich L (2011) Neuromuscular pathology in mice lacking alpha-synuclein Neurosci Lett 487: 350 353 |
227 | Kokhan VS, Van’kin GI, Ninkina NN, Shelkovnikova TA, Bachurin SO (2011) Impaired spatial and working memory in ageing mice with targeted inactivation of alpha-synuclein gene Dokl Biol Sci 441: 354 356 |
228 | Kokhan VS, Afanasyeva MA, Van’kin GI (2012) alpha-Synuclein knockout mice have cognitive impairments Behav Brain Res 231: 226 230 |
229 | Watson JB, Hatami A, David H, Masliah E, Roberts K, Evans CE, Levine MS (2009) Alterations in corticostriatal synaptic plasticity in mice overexpressing human alpha-synuclein Neuroscience 159: 501 513 |
230 | Steidl JV, Gomez-Isla T, Mariash A, Ashe KH, Boland LM (2003) Altered short-term hippocampal synaptic plasticity in mutant alpha-synuclein transgenic mice Neuroreport 14: 219 223 |
231 | Liu S, Ninan I, Antonova I, Battaglia F, Trinchese F, Narasanna A, Kolodilov N, Dauer W, Hawkins RD, Arancio O (2004) alpha-Synuclein produces a long-lasting increase in neurotransmitter release EMBO J 23: 4506 4516 |
232 | Gureviciene I, Gurevicius K, Tanila H (2007) Role of alpha-synuclein in synaptic glutamate release Neurobiol Dis 28: 83 89 |
233 | Gureviciene I, Gurevicius K, Tanila H (2009) Aging and alpha-synuclein affect synaptic plasticity in the dentate gyrus J Neural Transm 116: 13 22 |
234 | Martin ED, Gonzalez-Garcia C, Milan M, Farinas I, Cena V (2004) Stressor-related impairment of synaptic transmission in hippocampal slices from alpha-synuclein knockout mice Eur J Neurosci 20: 3085 3091 |
235 | Nemani VM, Lu W, Berge V, Nakamura K, Onoa B, Lee MK, Chaudhry FA, Nicoll RA, Edwards RH (2010) Increased expression of alpha-synuclein reduces neurotransmitter release by inhibiting synaptic vesicle reclustering after endocytosis Neuron 65: 66 79 |
236 | Wu N, Joshi PR, Cepeda C, Masliah E, Levine MS (2010) Alpha-synuclein overexpression in mice alters synaptic communication in the corticostriatal pathway J Neurosci Res 88: 1764 1776 |
237 | Larsen KE, Schmitz Y, Troyer MD, Mosharov E, Dietrich P, Quazi AZ, Savalle M, Nemani V, Chaudhry FA, Edwards RH, Stefanis L, Sulzer D (2006) Alpha-synuclein overexpression in PC12 and chromaffin cells impairs catecholamine release by interfering with a late step in exocytosis J Neurosci 26: 11915 11922 |
238 | Busch DJ, Oliphint PA, Walsh RB, Banks SM, Woods WS, George JM, Morgan JR (2014) Acute increase of alpha-synuclein inhibits synaptic vesicle recycling evoked during intense stimulation Mol Biol Cell 25: 3926 3941 |
239 | Vargas KJ, Makani S, Davis T, Westphal CH, Castillo PE, Chandra SS (2014) Synucleins regulate the kinetics of synaptic vesicle endocytosis J Neurosci 34: 9364 9376 |
240 | Ben Gedalya T, Loeb V, Israeli E, Altschuler Y, Selkoe DJ, Sharon R (2009) Alpha-synuclein and polyunsaturated fatty acids promote clathrin-mediated endocytosis and synaptic vesicle recycling Traffic 10: 218 234 |
241 | Fortin DL, Nemani VM, Voglmaier SM, Anthony MD, Ryan TA, Edwards RH (2005) Neural activity controls the synaptic accumulation of alpha-synuclein J Neurosci 25: 10913 10921 |
242 | Fortin DL, Nemani VM, Nakamura K, Edwards RH (2010) The behavior of alpha-synuclein in neurons Mov Disord 25: Suppl 1 S21 S26 |
243 | Tao-Cheng JH (2006) Activity-related redistribution of presynaptic proteins at the active zone Neuroscience 141: 1217 1224 |
244 | Scott D, Roy S (2012) alpha-Synuclein inhibits intersynaptic vesicle mobility and maintains recycling-pool homeostasis J Neurosci 32: 10129 10135 |
245 | Lai Y, Kim S, Varkey J, Lou X, Song JK, Diao J, Langen R, Shin YK (2014) Nonaggregated alpha-synuclein influences SNARE-dependent vesicle docking via membrane binding Biochemistry 53: 3889 3896 |
246 | Diao J, Burré J, Vivona S, Cipriano DJ, Sharma M, Kyoung M, Sudhof TC, Brunger AT (2013) Native alpha-synuclein induces clustering of synaptic-vesicle mimics via binding to phospholipids and synaptobrevin-2/VAMP2 Elife 2: e00592 |
247 | Soper JH, Roy S, Stieber A, Lee E, Wilson RB, Trojanowski JQ, Burd CG, Lee VM (2008) Alpha-synuclein-induced aggregation of cytoplasmic vesicles in Saccharomyces cerevisiae Mol Biol Cell 19: 1093 1103 |
248 | Gitler AD, Bevis BJ, Shorter J, Strathearn KE, Hamamichi S, Su LJ, Caldwell KA, Caldwell GA, Rochet JC, McCaffery JM, Barlowe C, Lindquist S (2008) The Parkinson’s disease protein alpha-synuclein disrupts cellular Rab homeostasis Proc Natl Acad Sci U S A 105: 145 150 |
249 | Lee SJ, Jeon H, Kandror KV (2008) Alpha-synuclein is localized in a subpopulation of rat brain synaptic vesicles Acta Neurobiol Exp (Wars) 68: 509 515 |
250 | Garcia-Reitbock P, Anichtchik O, Bellucci A, Iovino M, Ballini C, Fineberg E, Ghetti B, Della Corte L, Spano P, Tofaris GK, Goedert M, Spillantini MG (2010) SNARE protein redistribution and synaptic failure in a transgenic mouse model of Parkinson’s disease Brain 133: 2032 2044 |
251 | Lee HJ, Kang SJ, Lee K, Im H (2011) Human alpha-synuclein modulates vesicle trafficking through its interaction with prenylated Rab acceptor protein 1 Biochem Biophys Res Commun 412: 526 531 |
252 | Gosavi N, Lee HJ, Lee JS, Patel S, Lee SJ (2002) Golgi fragmentation occurs in the cells with prefibrillar alpha-synuclein aggregates and precedes the formation of fibrillar inclusion J Biol Chem 277: 48984 48992 |
253 | Choi BK, Choi MG, Kim JY, Yang Y, Lai Y, Kweon DH, Lee NK, Shin YK (2013) Large alpha-synuclein oligomers inhibit neuronal SNARE-mediated vesicle docking Proc Natl Acad Sci U S A 110: 4087 4092 |
254 | Kayed R, Head E, Thompson JL, McIntire TM, Milton SC, Cotman CW, Glabe CG (2003) Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis Science 300: 486 489 |
255 | Volles MJ, Lansbury PTJr (2003) Zeroing in on the pathogenic form of alpha-synuclein and its mechanism of neurotoxicity in Parkinson’s disease Biochemistry 42: 7871 7878 |
256 | Desplats P, Lee HJ, Bae EJ, Patrick C, Rockenstein E, Crews L, Spencer B, Masliah E, Lee SJ (2009) Inclusion formation and neuronal cell death through neuron-to-neuron transmission of alpha-synuclein Proc Natl Acad Sci U S A 106: 13010 13015 |
257 | Rey NL, Petit GH, Bousset L, Melki R, Brundin P (2013) Transfer of human alpha-synuclein from the olfactory bulb to interconnected brain regions in mice Acta Neuropathol 126: 555 573 |
258 | Luk KC, Kehm V, Carroll J, Zhang B, O’Brien P, Trojanowski JQ, Lee VM (2012) Pathological alpha-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice Science 338: 949 953 |
259 | Volpicelli-Daley LA, Luk KC, Patel TP, Tanik SA, Riddle DM, Stieber A, Meaney DF, Trojanowski JQ, Lee VM (2011) Exogenous alpha-synuclein fibrils induce Lewy body pathology leading to synaptic dysfunction and neuron death Neuron 72: 57 71 |
Figures and Tables
Fig.1
α-Synuclein domain structure. Upon binding to lipid membranes, the N-terminal domain of α-synuclein folds into two amphipathic helices; the C-terminal tail of α-synuclein does not contribute to membrane binding. The lipid binding domain can be divided into seven highly conserved 11-mer sequences. Helix 2 contains the aggregation-prone NAC-domain. All disease-linked mutations of α-synuclein are located in the second and fourth 11-mer stretch.
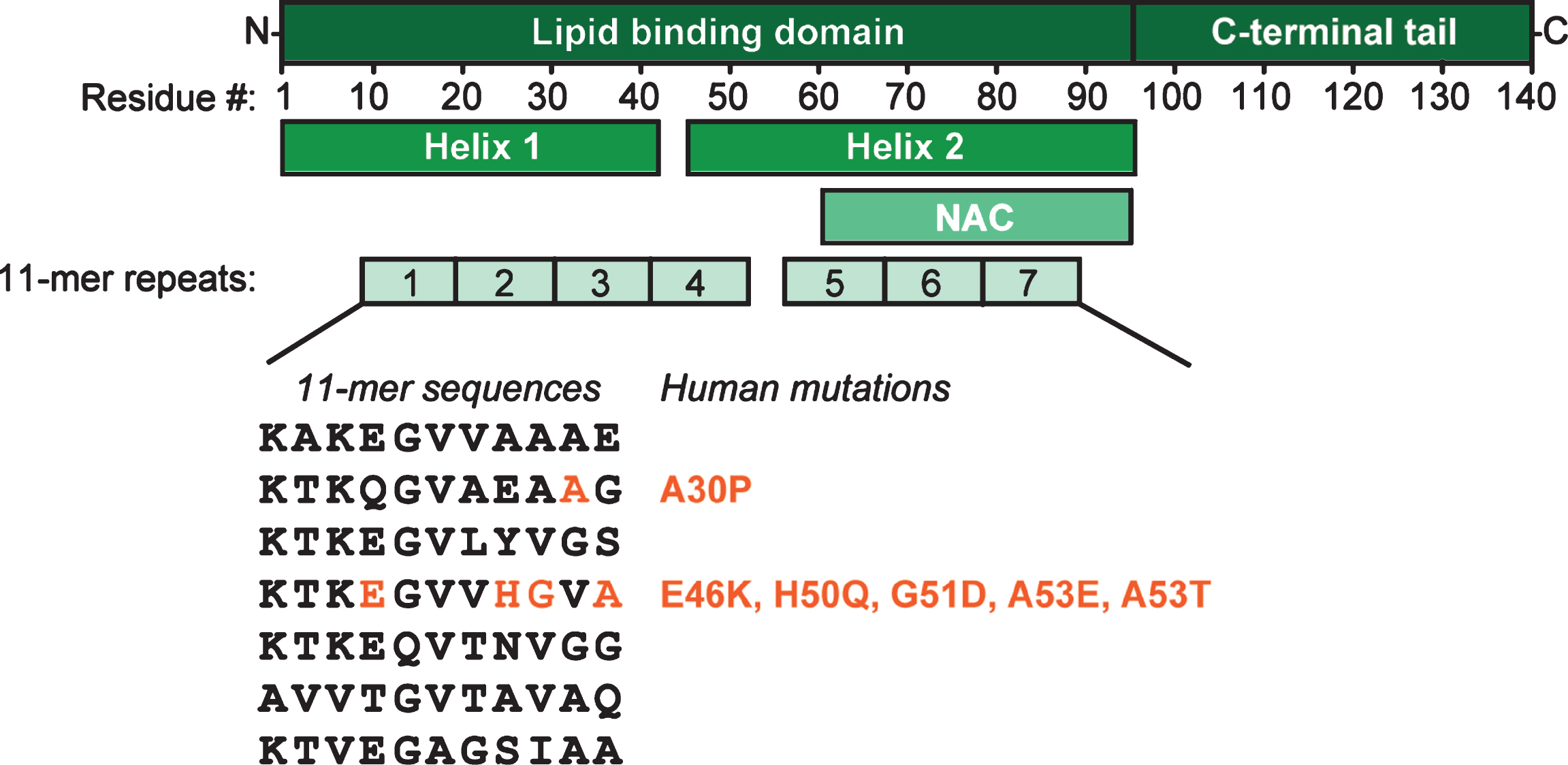
Fig.2
Physiological and pathological conformations of α-synuclein at the synapse. Cytosolic α-synuclein is monomeric and natively unfolded. Upon binding to synaptic vesicles, the N-terminal residues of α-synuclein adopt a helical structure. Membrane binding of α-synuclein is associated with its multimerization, which is essential for its physiological function at the synapse. Pathologically, unfolded α-synuclein in the cytosol can convert into β-sheet containing oligomers (protofibrils) which eventually form amyloid fibrils.
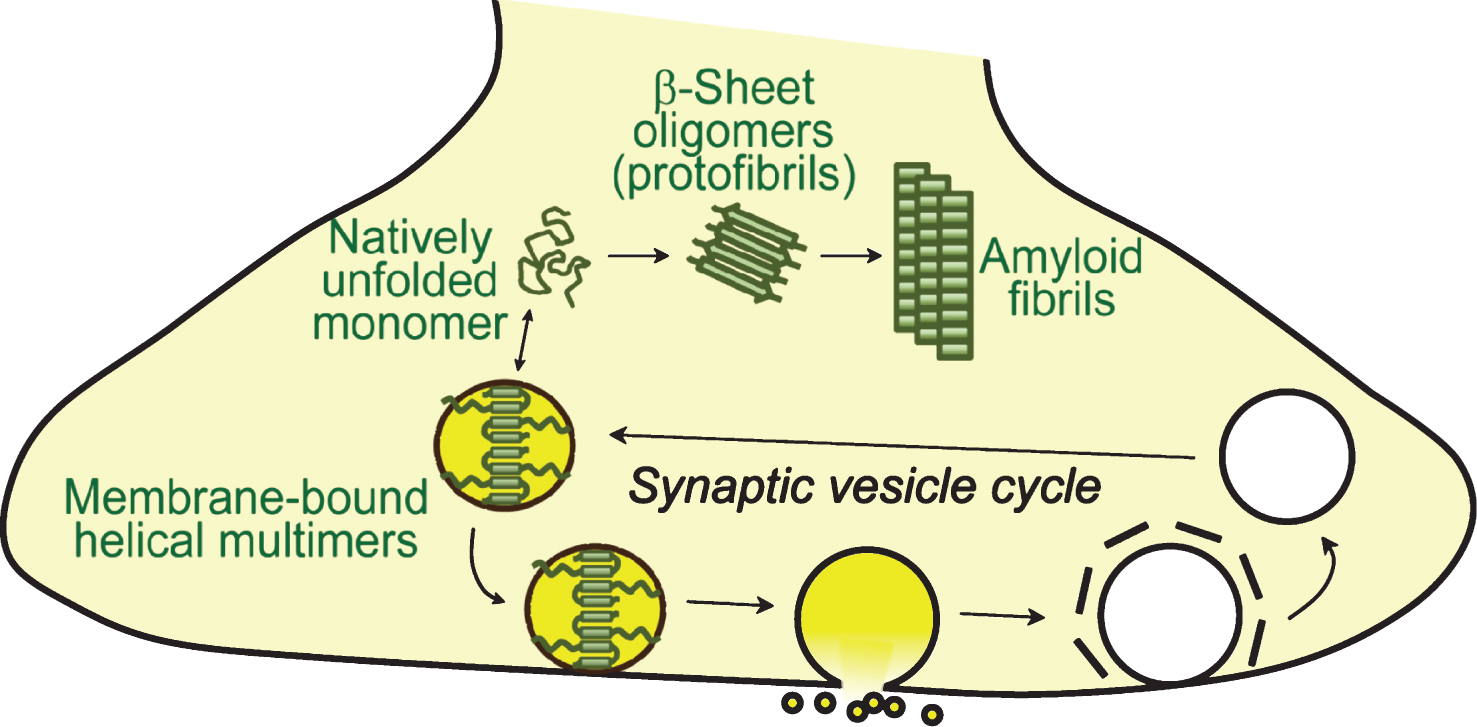
Fig.3
Function of α-synuclein at the synapse. Shown are the synaptic processes that α-synuclein has been reported to affect, including membrane remodeling, modulation of the dopamine transporter DAT and vesicular monoamine transporter VMAT2, clustering of synaptic vesicles and maintaining synaptic vesicle pools, promoting SNARE-complex assembly, and modulating the release cycle of synaptic vesicles.