Health-Related Quality of Life in FKRP-Related Limb-Girdle Muscular Dystrophy R9
Abstract
Background:
Limb-girdle muscular dystrophy R9 (LGMDR9) is a chronic progressive hereditary muscle disease, related to the Fukutin Related Protein (FKRP) gene, that may cause major disabilities, cardiomyopathy, and ventilatory failure. Knowledge of how LGMDR9 affects health-related quality of life (HRQoL) is relevant in treatment and care.
Objective:
To investigate HRQoL in the Norwegian LGMDR9 population over 14 months and relation to fatigue and sleep quality.
Methods:
Participants (16+ years) of the Norwegian LGMDR9 cohort study completed two HRQoL measures, i.e., Individualized Neuromuscular Quality of Life questionnaire (INQoL) and the 36-item Short Form (SF-36) at baseline, 8, and 14 months and measures of fatigue and sleep quality at 9 months.
Results:
HRQoL response rate was 84/90 (75 c.826 C > A homozygotes and nine c.826 C > A compound heterozygotes). Compared to Norwegian normative data, all SF-36 domain scores were impaired (p≤0.006) except mental health in males (p = 0.05) and pain scores. During 14 months, perceived muscle weakness and the INQoL index (disease burden) worsened in c.826 C > A homozygotes. Compound heterozygotes reported more dysphagia and physical difficulties than homozygotes and showed a tendency towards worsening in weakness over time but some improvement on the INQoL index. Homozygous females reported generally poorer HRQoL and a higher burden than males. The INQoL index was related to perceived muscle weakness and fatigue, and fatigue to myalgia and mental distress. The prevalence of fatigue and poor sleep was 40% and 49%, respectively.
Conclusions:
The 14-month follow-up period shows a worsening of perceived weakness and burden in c.826 C > A homozygotes, which can then be expected. The prevalence and impact of fatigue indicate a need for awareness and treatment of fatigue. Myalgia and mental distress are potential targets in the treatment of fatigue, which future studies need to establish. Sleep issues and gender-specific care needs also require attention in LGMDR9.
INTRODUCTION
Limb-girdle muscular dystrophy type R9 (LGMDR9) is a rare autosomal recessive disease caused by pathogenic variants in the Fukutin Related Protein gene (FKRP) [1]. It is more frequent in Northern European populations, with the highest prevalence recorded in Norway; 2.84/100,000 [2]. In European populations, FKRP c.826 C > A is the most common disease-causing variant, and c.826 C > A homozygotes tend to have a milder disease progression than c.826 C > A compound heterozygotes [3–5]. The disease typically presents with slowly progressive proximal lower limb weakness at a young age and commonly leads to wheelchair dependency, cardiomyopathy, and eventually ventilatory failure [1, 2, 5].
While no disease-modifying or curative therapy is currently available for LGMDR9, affected individuals are provided supportive care. This may include symptom-relieving medications, cardiorespiratory-, physical-, occupational, or cognitive-behavioral therapy, providing access to user-controlled personal assistance, and efforts to facilitate social participation [6, 7]. Patient Reported Outcome Measures (PROM), such as Health-Related Quality of Life (HRQoL), are important tools to understand the patients' experiences and provide the necessary supportive care. Despite some conceptual and methodological challenges [8], HRQoL is considered important endpoints in clinical research and health planning [8, 9] and increasingly emphasized as evidence of utility or treatment success. HRQoL measures encompass both generic and disease-specific versions. Generic HRQoL enable comparisons between heterogeneous diagnostic groups and between patients and general populations and may be used to calculate quality-adjusted life years for cost-utility analyses. Disease-specific measures target disease-related symptoms, effects, and burdens and may be more relevant in clinical research aimed at monitoring disease progression or treatment-related benefits. Most HRQoL instruments measure self-perceived health status in physical, mental, and social aspects of health, whereas some also include personal evaluations by the respondents [10] to better reflect the «true» level of HRQoL.
A systematic literature review reports strong evidence for disease severity, fatigue, pain, and mood as important predictors of HRQoL in muscle diseases, moderate evidence for the role of female gender, advancing age, and poor sleep, and weak evidence for disease duration and employment [11]. A recent study indicated that LGMDR9 is associated with considerable symptoms of pain [12]. We have found no study reporting HRQoL in LGMDR9 patients. In all, only a handful of qualitative [6, 7] and quantitative studies [13, 14] have been published on how LGMD impacts daily life.
In the present study, we investigated the magnitude of changes in HRQoL in a Norwegian LGMDR9 cohort during a 14-month period and explored health areas with potential for improvement, subgroups at heightened risk for poor HRQoL, and symptoms of particular importance to patients. Additionally, we investigated the prevalence and correlates of fatigue and poor sleep quality.
MATERIALS AND METHODS
Participants
The present study is a part of “The Norwegian LGMDR9 cohort study” at the National Neuromuscular Centre Norway (NMK), University Hospital of North Norway (UNN). A total of 153 subjects (135 adults, i.e., 16+ years) in Norway with clinically and genetically confirmed LGMDR9 were invited to the cohort study. Of these, 101 subjects gave consent and participated [2]. The participants completed a study-specific questionnaire, and patient notes were collected from the specialist health care. All adults (16+ years) in the cohort (90/101) were invited to the present study of HRQoL.
Instruments
The 36-Item Short-Form Health Survey (SF-36) version 1
The Norwegian version of SF-36 v. 1 [15] is a generic profile-based HRQoL measure that yields nine 0-100-point subscales (from lowest to highest HRQoL): physical functioning, role limitations due to physical problems, bodily pain, general health, vitality, social functioning, role limitations due to emotions, mental health, and change in health over the past year. As in the SF-36 algorithm [16], missing values were replaced with the subjects’ mean score for the completed items on the respective scale given a minimum of 50% subscale completion. SF-36 is extensively validated [17, 18] and widely used including on patient populations with muscle diseases [11].
The Individualized Neuromuscular Quality of Life queastionnaire (INQoL) version 2.0
INQoL [19] v. 2.0 is a disease specific measure comprising seven symptom domains (i.e., muscle weakness, myalgia, fatigue, myotonia, diplopia, ptosis, and dysphagia), five functional or “life domains” (i.e., activities related to daily living/leisure/work, independence, social relationships, emotions, and body image), and two treatment domains (i.e., perceived and expected treatment effects). The myotonia, diplopia, ptosis, and treatment domains were not included in this study as they were considered irrelevant. Myotonia, diplopia, and ptosis are not reported in LGMDR9. Each domain contains items of severity, impact degree, and impact importance. The item scales are 7-point Likert type and categorical combined as each number also has a categorical description, from either nothing or very little, to extreme. Raw scores are transformed into domain scores ranging between 0 (no burden) and 100 (maximal burden) according to a weighted scoring algorithm. Additionally, the items on impact and impact importance of each life domain are aggregated into one sum score - the INQoL index - as a proxy of disease burden or total impact on HRQoL. Missing data were replaced according to the scoring algorithm.
INQoL is the first HRQoL instrument developed for neuromuscular disorders and has been translated to multiple languages [20]. The Norwegian version is the result of a linguistic validation methodology with forward-backward translation and tested on 100 outpatients with 70 respondents at NMK with various neuromuscular diseases proving satisfactory internal validity as well as concurrent validity with the SF-36 (unpublished data).
The Pittsburgh Sleep Quality Index (PSQI)
PSQI assesses self-reported sleep quality [21]. The Norwegian version [22] was used. PSQI has proven good reliability and validity, it is widely used in screening for sleep disturbances in both clinical and non-clinical populations [23], and has been applied in populations with other types of muscular dystrophies [24, 25]. Based on the items, seven component scores (range 0-no to 3-severe difficulty) are created and summed to one global score (range 0–21) with a cut-off score of > 5 indicating significant sleep disturbance. Missing data were not imputed.
The Fatigue Severity Scale (FSS)
FSS [26] indexes the level of fatigue. It is a generic psychometric measure validated across multiple populations including a Norwegian general population [27]. It consists of nine items rated on a 7-point Likert scale, and the FSS score is calculated as the average of the item scores [26]. The revised cut-off score of≥5 (from≥4) was recommended to indicate severe fatigue in a Norwegian FSS validation study [27]. Missing data were not imputed.
Procedure
The HRQoL instruments were distributed in paper versions by regular mail to adult participants of «The Norwegian LGMDR9 cohort study» at three consecutive time points: 2020-June, 2021-February, and 2021-August, respectively. One reminder was sent per administration. In addition, FSS and PSQI were administered one month after the second HRQoL collection. The second HRQoL collection was used for correlation analyses between HRQoL, FSS, and PSQI. Baseline HRQoL was used for correlation analyses between INQoL and SF-36 and for comparison with reference data and of subgroups except genotype subgroups (c.826 C > A homozygotes vs c.826 C > A compound heterozygotes). To enable inclusion of all compound heterozygotes, who were few, the first responses of each unique participant was used rather than the baseline HRQoL. Repeated measurements of HRQoL were applied to evaluate the level of fluctuations in cross-sectional measurements over time and to measure longitudinal changes at the individual level and the rank-order stability of the subscales (Sect. “Statistical analyses”). Social and clinical background data were collected from the questionnaire of the main study and included marital status, living situation, work status, educational achievement, activities of daily living (ADL), ambulation, use of Positive Airway Pressure (PAP) therapy (i.e., mask-based therapy for e.g., sleep apnea or hypoventilation), cardiac involvement, Percutaneous Endoscopic Gastrostomy (PEG), and consumption of pain medication for myalgia. Higher education was defined as at least one year at university or college. Age at disease onset, for calculation of disease duration, was primarily collected from patient notes. Disease onset was defined as the first clinical sign or symptom of muscle disease, such as muscle weakness, myalgia, or myoglobinuria and did not include incidental finding of elevated muscle enzymes. Norwegian normative SF-36 data, used as reference, were obtained from published material [28, 29], and social population statistics from Statistics Norway [30].
Statistical analyses
Data were analyzed using IBM SPSS Statistics for Windows (Version 27.0. Armonk, NY: IBM Corp.). Distribution of continuous variables are described using mean and standard deviation (±SD), or median and interquartile range (IQR), as appropriate. For simple group comparisons, independent t-test with bootstrapping or Kruskal-Wallis (multiple subgroups) was applied. Bootstrapping was always performed with 5000 resamples. In comparisons with normative SF-36 data, independent t-test was found acceptable given the sample sizes. In genotype comparison, multiple linear regression (MLR) with bootstrapping was applied to adjust for sex and age. To assess the change in INQoL domains from baseline to 14 months, paired t-test with bootstrapping (homozygotes), or paired Wilcoxon-signed rank test (compound heterozygotes due to a very limited sample size) was used. Categorical variables are presented as frequencies. Pearson chi-square was used for variable cell comparisons, and effect size is presented as odds ratio (OR) with confidence interval (CI). Bivariate correlations were examined with Pearson correlation (r) on normal distributions, otherwise with Spearman rank correlation (rs). Within the context of the current study, we considered correlation coefficients < 0.40 as weak, 0.40–0.69 as moderate,≥0.70 as strong, and≥0.90 as very strong.
For repeated measurements, we used generalized linear mixed models (GLMM) as it is a flexible regression analysis with regarding handling of missing data and data dependencies. The rank-order stability of the HRQoL domain scores was estimated as an Intraclass Correlation Coefficient (ICC) as the ratio between the intersubject variance (random intercept) and the total variance in a model without any fixed effects. ICC values < 0.50, 0.50 –0.75, 0.75 –0.90, > 0.90 may be considered as poor, moderate, good, or a high degree of rank-order stability, respectively. A high ICC thus means that if changes in HRQoL occur, the participants show the same change pattern with low variability in change across time. In order to regress the independent variables on the repeatedly measured dependent continuous variables of interest (perceived muscle weakness and the INQoL index), GLMM regression was applied. Choice of covariance structure for the residual matrix was decided based upon which structure that yielded the lowest Bayesian Information Criterion value. All GLMM analyses were performed with robust estimation of the standard errors. To identify predictors of dependent continuous variables measured once (FSS and PSQI), MLR with backward elimination was applied. The selection of independent variables in GLMM, or MLR before backward elimination, was based on the p-values of simple regression analyses of the variable of interest (p < 0.20 considered relevant). Age and sex were included as control variables. Assumptions of normality of the MLR residuals were checked using Q-Q plots, skewness, and kurtosis, homoscedasticity by scatter plotting predicted versus residual values, and influential cases by Cook's distance. The alpha level was set to p < 0.05. Considering the exploratory nature of the study and small subgroups (low power), correction for multiple testing was not used in this study [31].
Approvals and patient consent
All participants provided a written informed consent for the collection and use of clinical data. The study was approved by the Regional Committee for medical and health research ethics of Northern Norway (2018/1968/REK nord), and by the Data Protection Officer at UNN. The Norwegian version of SF-36 v. 1 was freely distributed by Knowledge Centre for the Health Services, Norwegian Institute of Public Health [32]. Permission to use the INQoL [20], the PSQI [33], and the FSS [34], respectively, was obtained.
RESULTS
Participants
The response rates are shown in Fig. 1. The overall HRQoL response rate was 84/90 (93%). Of all HRQoL respondents, 59 (70%) participated all three times, 16 (19%) twice, and 9 (11%) participated only once. Baseline characteristics of the sample comprising 75 c.826 C > A homozygotes and nine c.826 C > A compound heterozygotes are provided in Table 1. The compound heterozygotes had comparable disease duration to homozygotes but were younger (median 31 vs 49 years, p = 0.033). The status regarding work, educational achievement, ADL, ambulation, PAP therapy, and PEG, was relatively poorer among compound heterozygotes than homozygotes, whereas the frequency of cardiac involvement was comparable and the consumption of pain medication relatively higher among homozygotes. The subgroup of homozygotes was gender balanced, and males and females had comparable age and disease duration. Among homozygotes, females showed relatively poorer ADL and ambulation, a higher rate of PAP therapy, and a greater consumption of pain medication compared to males, whereas a higher proportion of males reported cardiac involvement. Comparison of compound heterozygous males and females was not applicable due to low sample size, heterogeneous genotypes, and unequal age distributions (Table 1 and 2).
Fig. 1
Flow chart.
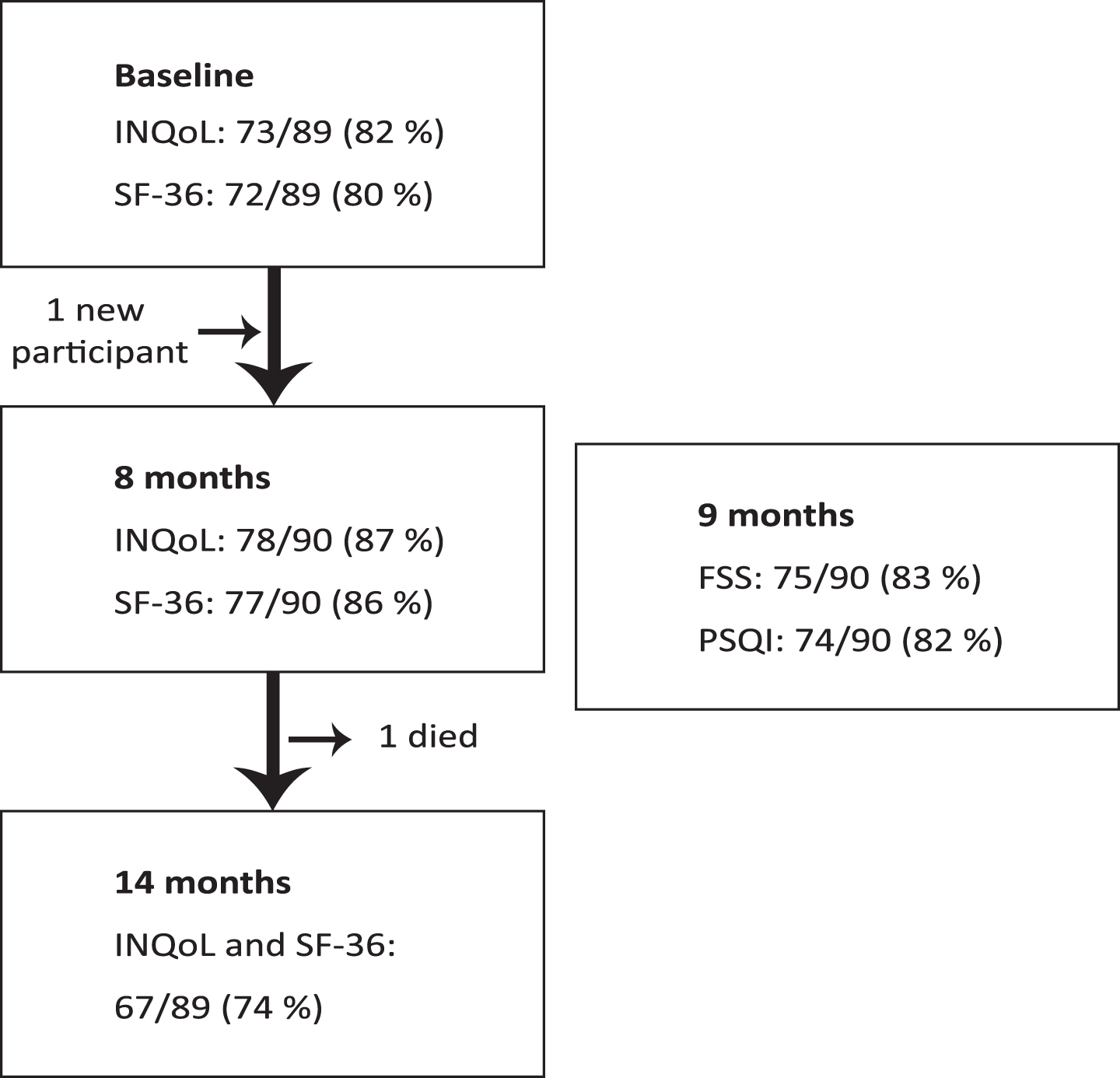
Table 1
Sociodemographic and clinical background data in all participants
| FKRP c.826 C > A homozygotes | FKRP c.826 C > A compound heterozygotes | |||||
| All | Females | Males | All | Females | Males | |
| M (IQR); n/n (%) | M (IQR); n/n (%) | M (IQR); n/n (%) | M (IQR); n/n (%) | M (IQR); n/n (%) | M (IQR); n/n (%) | |
| N | 75/75 (100) | 35/75 (47) | 40/75 (53) | 9/9 (100) | 5/9 (56) | 4/9 (44) |
| Age (years) | 49 (33–58) | 47 (33–57) | 50 (34–61) | 31 (28–36) | 31 (28–35) | 33 (25–56) |
| Range | 16–75 | 16–71 | 16–75 | 25–63 | 25–35 | 25–63 |
| Disease duration (years) | 31 (18–47) | 30 (19–45) | 32 (18–49) | 27 (16–33) | 27 (16–30.5) | 29 (15–55) |
| Spouse/ cohabitant (Age≥25 years) | 43/67 (64) | 20/31 (64) | 23/36 (64) | 4/9 (44) | 2/5 (40) | 2/4 (50) |
| Living alone | 23/72 (32) | 10/34 (29) | 13/38 (34) | 2/9 (22) | 1/5 (20) | 1/4 (25) |
| ADL | ||||||
| Independent | 31/75 (41) | 11/35 (31) | 20/40 (50) | 2/9 (22) | 1/5 (20) | 1/4 (25) |
| P.A. | 15/75 (20) | 11/35 (31) | 4/40 (10) | 6/9 (67) | 4/5 (80) | 2/4 (50) |
| Home care | 3/75 (4) | 3/35 (9) | 0/40 (0) | 0/9 (0) | 0/5 (0) | 0/4 (0) |
| Institution | 2/75 (3) | 2/35 (6) | 0/40 (0) | 0/9 (0) | 0/5 (0) | 0/4 (0) |
| Work/studies | ||||||
| Yes, 100% | 26/61 (43) | 9/30 (30) | 17/31 (55) | 1/8 (13) | 1/5 (20) | 0/3 (0) |
| Partial incapacity | 13/61 (21) | 8/30 (27) | 5/31 (16) | 0/8 (0) | 0/5 (0) | 0/3 (0) |
| 100% incapacity | 19/61 (31) | 11/30 (37) | 8/31 (26) | 6/8 (75) | 3/5 (60) | 3/3 (100) |
| Incapacity, other cause | 2/61 (3) | 1/30 (3) | 1/31 (3) | 1/8 (13) | 1/5 (20) | 0/3 (0) |
| (Age 18-67 years) | ||||||
| Higher educationa | 37/68 (54) | 18/32 (56) | 19/36 (53) | 3/9 (33) | 3/5 (60) | 0/4 (0) |
| (Age≥25 years) | ||||||
| Ambulation | ||||||
| Independent | 40/75 (53) | 13/35 (37) | 27/40 (68) | 3/9 (33) | 1/5 (20) | 2/4 (50) |
| Walking aids | 13/75 (17) | 7/35 (20) | 6/40 (15) | 0/9 (0) | 0/5 (0) | 0/4 (0) |
| Wheelchair | 22/75 (29) | 15/35 (43) | 7/40 (18) | 6/9 (67) | 4/5 (80) | 2/4 (50) |
| PAP | ||||||
| All | 22/75 (29) | 15/35 (43) | 7/40 (18) | 3/9 (33) | 1/5 (20) | 2/4 (50) |
| Day and night | 2/75 (3) | 2/35 (13) | 0/40 (0) | 3/9 (33) | 1/5 (20) | 2/4 (50) |
| CM | 16/73 (22) | 4/34 (12) | 12/39 (31) | 2/9 (22) | 1/5 (20) | 1/4 (25) |
| PEG | 1/75 (1) | 1/35 (3) | 0/40 (0) | 3/9 (33) | 1/5 (20) | 2/4 (50) |
| Analgesics (myalgia) | ||||||
| Never | 51/75 (68) | 18/35 (51) | 33/40 (83) | 4/8 (50) | 1/5 (20) | 3/3 (100) |
| Daily/almost daily | 4/75 (5) | 4/35 (11) | 0/40 (0) | 0/8 (0) | 0/5 (0) | 0/3 (0) |
(a) ≥ 1 year in college or university. M = median, IQR = inter-quartile range, ADL = activities of daily living, P.A. = user-controlled personal assistance, PAP = Positive Airway Pressure support, CM = cardiomyopathy (self-reported), PEG = Percutaneous Endoscopic Gastrostomy.
Table 2
Genotypes and natural history of FKRP c.826 C > A compound heterozygous participants
| No. | Genotype FKRP c.826 C > A / | Sex | Age (years) | Age at onset (years) | Age at W/C (years) | Age at CM (years) | Age at PAP (years) | PEG |
| 1. a | 962 C > A, (Ala321Glu) | F | 30 | 12 | 23 | 24 | – | – |
| 2. a | F | 35 | 8 | 20 | – | – | – | |
| 3. a † | M | 36 | 2 | 7 | 18 | 23 | + | |
| 4. | F | 34 | 3 | 14 | 31 | 27 | + | |
| 5. | M | 63 | 1 | 12 | – | 31 | + | |
| 6. | 899T > C, (Val300Ala) | F | 25 | 11 | – | – | – | – |
| 7. | 1323T > G (Phe441Leu) | F | 31 | 1 | 28 | 28 | – | – |
| 8. | 328 C > T, (Arg110Trp) | M | 30 | 7 | – | – | – | – |
| 9. | 166T > A, (Phe56Ile) | M | 25 | 13 | – | 20 | – | – |
(a) Siblings, (†) died during the study. W/C = wheelchair dependency, CM = cardiomyopathy (here: LGMDR9-related abnormalities on echocardiography according to medical records), PAP = Positive Airway Pressure support, PEG = Percutaneous Endoscopic Gastrostomy, F = female, M = male. Adapted table from a previous study [2] (Table A).
Internal validity
Global scores for the PSQI, the FSS, and the HRQoL could be calculated for 89%, 97%, and 96–100% of the responses, respectively. SF-36 showed considerable floor effects on physical functioning, ceiling effects on social relationships, and both floor- and ceiling effects on role physical and role emotions (Table 3). The INQoL scale was reverse scored and had negligible ceiling effects but considerable floor effects on myalgia, fatigue, and dysphagia (Table 4). ICCs were moderate to high (0.62–0.85) on all SF-36 subscales except role emotions and change (Table 3). INQoL showed overall relatively higher ICCs (0.73–0.91) (Table 4) indicating higher rank-order stability across time. There were moderate to strong correlations between SF-36 and INQoL subscales sharing conceptual similarity (Table 5).
Table 3
SF-36 measurements
| Baseline, 8 and 14 months | Baseline | First responses | ||||
| SF-36 subscales | Scores (n = 66–76) Mean SD | ICC | Floor/Ceiling % | Below reference All F /M (n = 36/36) p | Homoz. F/M (n = 31/34) p | Homoz/Comp (n = 75/9) pa |
| Physical functioning | 37.934.1 33.732.0 35.8 . 34.4 | 0.85 | 19–23/ 3–6 | */ * | 0.076 (F worse) | 0.37 (Comp worse) |
| Role physical | 46.843.9 44.141.8 47.043.3 | 0.61 | 33–39/ 28–35 | */ * | 0.75 (F worse) | 0.51 (Homoz worse) |
| Bodily pain | 68.025.5 66.425.8 71.322.5 | 0.75 | 0/ 18–20 | 0.36 / - (above reference; 0.85) | 0.040 F worse | 0.56 (Comp worse) |
| General health | 48.824.7 47.325.9 48.622.3 | 0.80 | 1–4/ 0–1 | */ * | 0.18 (F worse) | 1.0 |
| Vitality | 44.523.3 45.224.2 51.023.6 | 0.83 | 3–5/ 0 | */ 0.018 | 0.008 F worse | 0.85 (Homoz worse) |
| Social functioning | 67.027.8 66.927.2 66.626.9 | 0.66 | 0–1/ 18–28 | */ 0.0005 | 0.025 F worse | 0.39 (Homoz worse) |
| Role emotional | 65.740.6 65.340.7 62.243.8 | 0.48 | 19–27/ 52–57 | * / 0.0073 | 0.11 (F worse) | 0.55 (Homoz worse) |
| Mental health | 73.316.7 74.218.3 75.916.4 | 0.79 | 0/ 0–1 | 0.006/ 0.050 | 0.18 (F worse) | 0.24 (Homoz worse) |
| Change | 38.518.3 37.720.1 43.218.9 | 0.45 | 2–8/ 1–4 | N.D. | 0.46 (F worse) | 0.65 (Homoz worse) |
(a) Age- and sex adjusted p-value. (*) p < 0.0001. P-values < 0.05 are highlighted. SD = standard deviation, ICC = intraclass correlation coefficient, homoz = FKRP c.826 C > A homozygotes, compound = FKRP c.826 C > A compound heterozygotes, F = females, M = males, N.D. = no data. The SF-36 subscales range 0–100 (lowest to highest HRQoL). Since only 7/9 compound heterozygotes participated at baseline, the first responses, rather than only the baseline responses, were included for comparison with homozygotes.
Table 4
INQoL measurements
| Baseline, 8 and 14 months | Baseline and 14 months | Baseline | First responses | ||||
| INQoL domains | Scores (n = 67–73) | ICC | Floor/ Ceiling | Increase | Homoz F/M (n = 31/35) | Homoz/ Comp (n = 75/9) | |
| Mean SD | % | Mean SD (median) | p | pb | |||
| Homoz (n = 55) | Compa (n = 5) | ||||||
| Weakness | 62.124.5 63.023.4 64.523.9 | 0.91 | 5–7/ 1–5 | 4.79.8 (5.3) (p = 0.001) | 2.113.7 (5.3) (p = 0.49) | 0.39 (F worse) | 0.11 (Comp worse) |
| Myalgia | 29.826.2 31.027.3 25.126.0 | 0.81 | 30–36/ 0 | –2.017.9 (0) (p = 0.41) | –3.04.7 (0) (p = 0.18) | 0.38 (F worse) | 0.89 (Comp worse) |
| Fatigue | 39.928.0 43.228.3 36.431.5 | 0.81 | 19–33/ 1–3 | –1.018.2 (0) (p = 0.68) | 6.323.1 (0) (p = 0.59) | 0.71 (F worse) | 0.62 (Comp worse) |
| Dysphagia | 8.216.7 7.815.0 4.812.0 | 0.81 | 11–82/ 0 | –0.28.7 (0) (p = 0.87) | –11.614.1 (–5.3) (p = 0.11) | 0.51 (F worse) | 0.004 Comp worse |
| Activities | 55.823.4 59.923.8 57.024.7 | 0.91 | 0–1/ 0–3 | 3.311.7 (1.9) (p = 0.051) | 0.913.7 (–0.9) (p = 0.89) | 0.044 F worse | 0.14 (Comp worse) |
| Indepen-dence | 40.929.7 43.630.5 41.229.5 | 0.91 | 9–10/ 1–3 | 1.314.9 (0) (p = 0.52) | –1.11.5 (0) (p = 0.16) | 0.004 F worse | 0.0002 Comp worse |
| Relationships | 34.222.1 35.021.0 34.021.4 | 0.73 | 7–8/ 0 | 1.418.4 (0) P = 0.57) | 5.620.4 (–2.3) (p = 1.0) | 0.73 (F worse) | 0.64 (Homoz worse) |
| Emotions | 38.121.5 39.423.5 34.520.9 | 0.81 | 3–8/ 0 | 0.314.6 (0) (p = 0.88) | –6.114.6 (–5.6) (p = 0.36) | 0.064 (F worse) | 0.30 (Homoz worse) |
| Body image | 43.026.0 44.925.9 44.226.0 | 0.74 | 3–9/ 0–3 | 2.920.2 (0) (p = 0.29) | –6.123.7 (0) (p = 1.0) | 0.40 (F worse) | 0.15 (Comp worse) |
| INQoL index | 47.318.0 48.318.0 48.2 19.6 | 0.81 | 0–2/ 0 | 4.812.3 (3.9) (p = 0.010) | –4.49.1 (–6.1) (p = 0.34) | 0.15 (F worse) | 1.0 |
(a) Patient No. 1, 2, and 5–7, Table 2, (b) Age- and sex-adjusted p-value. P-values < 0.05 are highlighted. SD = standard deviation, ICC = intraclass correlation coefficient of repeated measurements homoz = FKRP c.826 C > A homozygotes, compound = FKRP c.826 C > A compound heterozygotes, F = females, M = males. The INQoL domain scores range 0–100 (no burden to maximal burden). Since only 7/9 compound heterozygotes participated at baseline, first response of each individual was included for comparison with homozygotes. Four zero-scores of muscle weakness out of 218 registrations were not included due to clear inconsistency from the other registrations of the respective respondent.
Table 5
Bivariate correlations between selected subscales from INQoL and SF-36
| INQoL | SF-36 | rs (n = 69–72) | 95% CI |
| Muscle weakness | Physical functioning | –0.56 * | (–0.71, –0.37) |
| Fatigue | Vitality | –0.70 * | (–0.81, –0.55) |
| Myalgia | Bodily pain | –0.77 * | (–0.86, –0.66) |
| Activity | Physical functioning | –0.64 * | (–0.76, –0.46) |
| Role physical | –0.44 * | (–0.62, –0.23) | |
| Independence | Physical functioning | –0.69 * | (–0.80, –0.54) |
| Relationships | Social functioning | –0.73 * | (–0.83, –0.60) |
| Emotions | Mental health | –0.67 * | (–0.79, –0.51) |
| Role emotional | –0.49 * | (–0.65, –0.29) |
(*) p < 0.001, rs = Spearman's rho. Note: INQoL and SF-36 have inverse directions on the scales, hence negative correlation coefficients.
HRQoL over time
The measures of HRQoL at the three time points showed comparable mean scores (Table 3 and 4). The greatest difference between the highest and the lowest mean score was found on the subscales of bodily pain (4.9 points) and vitality (6.5 points) of the SF-36 (Table 3) and, correspondingly, myalgia (5.9 points) and fatigue (6.8 points) of the INQoL (Table 4). The change in the INQoL subscale scores from baseline to 14 months showed, in homozygotes, a worsening in muscle weakness and the INQoL index and a borderline significant worsening in activities (Table 4). Correspondingly, GLMM regression, including all three measurements of the homozygotes, showed an increase in muscle weakness (Table 6A), and an incremental tendency in the INQoL index (p = 0.08) (Table 6B). Five compound heterozygotes, all wheelchair dependent, completed the INQoL at baseline and 14 months. They showed a tendency towards worsening in muscle weakness but, more strongly, an improvement of the INQoL index (Table 4). On the particular SF-36 item concerning change in general health over the past year, worsening was reported by 42–57% of the homozygotes at the three respective measurements, compared to 17–29% of the compound heterozygotes. Among the homozygotes, 2–9% reported severe worsening, and 4–12% reported improvement. None of the compound heterozygotes reported severe worsening or any improvement.
Table 6
Multivariate regression models (generalized linear mixed models) of INQoL scores
| Independent variables | pa | Beta | p | 95% CI |
| A) Muscle weakness (c.826 C > A homozygotes) | ||||
| Age (years) | 0.01 | 0.44 | 0.008 | (0.1, 0.8) |
| Female (vs male) | 0.21 | 7.19 | 0.14 | (–2.3, 16.6) |
| Time (months) | 0.002 | – | 0.003 | – |
| 0 | –4.22 | 0.001 | (–6.8, –1.6) | |
| 8 | –3.50 | 0.003 | (–5.8, –1.2) | |
| 14 | – | – | – | |
| B) INQoL index (c.826 C > A homozygotes) | ||||
| Age (years) | 0.04 | 0.26 | 0.03 | (–0.03, 0.50) |
| Female (vs male) | 0.03 | 9.18 | 0.02 | (1.6, 16.8) |
| Time (months) | 0.07 | – | 0.08 | – |
| 0 | –3.57 | 0.03 | (–6.73, –0.41) | |
| 8 | –1.46 | 0.29 | (–4.15, 1.24) | |
| 14 | – | – | – | |
| C) INQoL index (All participants) | ||||
| Age (years) | 0.02 | 0.001 | 0.99 | (–0.20, 0.21) |
| Female (vs male) | 0.06 | 5.25 | 0.06 | (–0.23, 10.74) |
| Higher education (vs non) | 0.14 | 4.08 | 0.14 | (–1.28, 9.44) |
| Weakness (0–100) | <0.001 | 0.37 | <0.001 | (0.25, 0.49) |
| Fatigue (0–100) | <0.001 | 0.10 | 0.006 | (0.03, 0.18) |
| Myalgia (0–100) | <0.001 | 0.04 | 0.47 | (–0.06, 0.13) |
| Dysphagia (0–100) | 0.17 | –0.03 | 0.75 | (–0.22, 0.16) |
| PAP (vs non) | 0.045 | 1.15 | 0.70 | (–4.80, 7.09) |
| Time | 0.13 | – | 0.84 | – |
14 months = reference category for the variable Time. pa = p-value before mutual adjustment for the other independent variables. CI = confidence interval, PAP = Positive Airway Pressure support. Dependent variable: (A) Perceived muscle weakness from FKRP c.826 C > A homozygotes. (B) The INQoL index from FKRP c.826 C > A homozygotes. (C) The INQoL index from all participants. Variables not included in Table 6C due to a pa≥0.20 were genotype (pa = 0.30), cardiomyopathy (pa = 0.60), and PSQI (pa = 0.33). Level of ambulation was significant (pa < 0.001) but not included due to substantial similarity with muscle weakness.
HRQoL compared to normative data, and gender differences
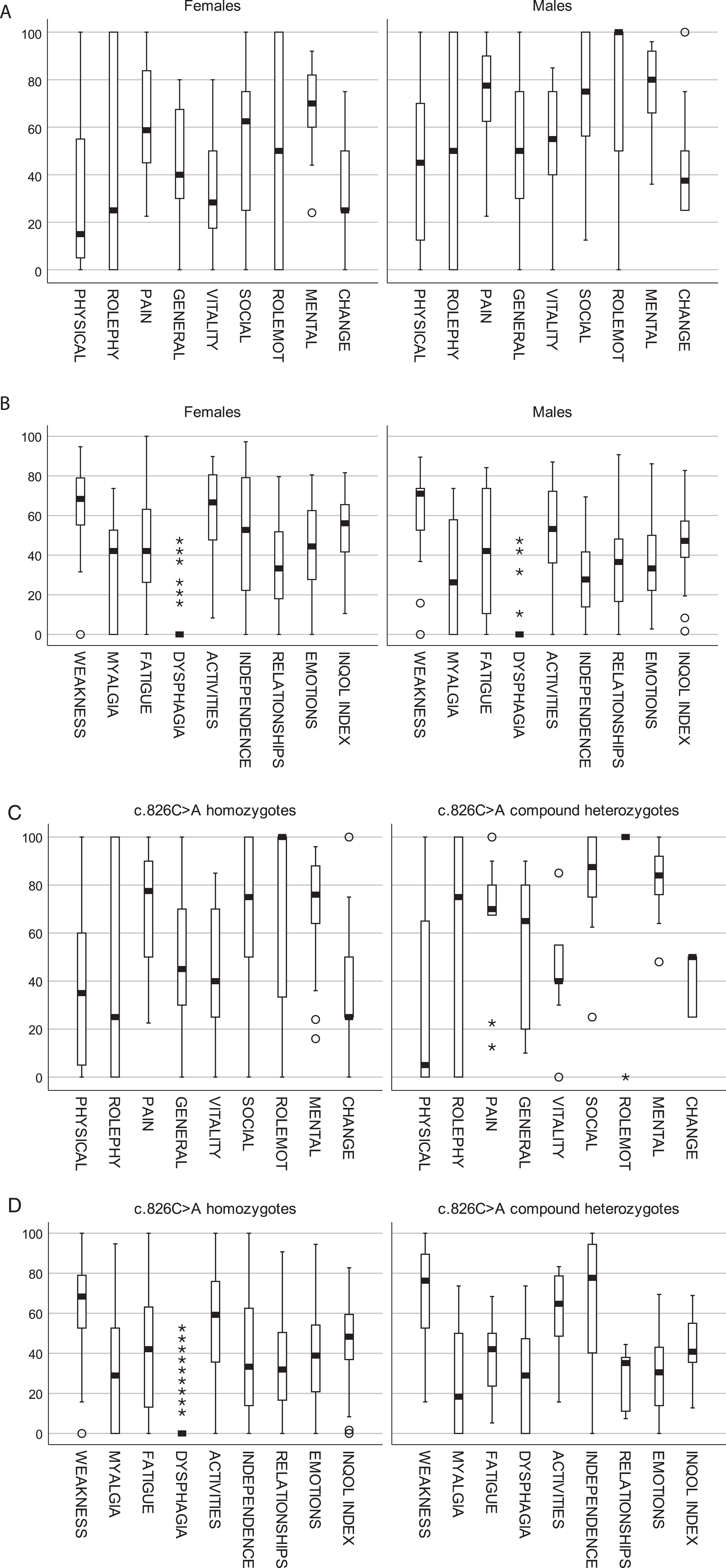
Compared to the SF-36 normative data, the present study population showed significant impairments on all subscales except bodily pain, and the impairment of mental health in males was borderline significant (Table 3). Females showed relatively poorer scores both in the reference- and the present study population (not shown), but the female impairments were more pronounced among our participants compared to reference population (Table 3). Overall, homozygous females reported lower levels of HRQoL than homozygous males (Fig. 2A) and significantly on bodily pain, vitality, and social relationships (Table 3). Moreover, they showed a higher burden on several aspects of INQoL (Fig. 2B) and significantly on activities and independence (Table 4). GLMM analyses, including all three measurements of the homozygotes, showed that females tended to have more severe muscle weakness (Table 6A) and a significantly worse INQoL index than males (Table 6B).
HRQoL and genotypes
SF-36 showed no significant differences in HRQoL between the two genotype groups but a tendency towards poorer physical functioning, yet better social and mental HRQoL among the compound heterozygotes (Table 3, Fig. 2C). On INQoL, the compound heterozygotes reported significantly higher burden on the domains of dysphagia and independence (Table 4, Fig. 2D). The prevalence of dysphagia was 6/9 (67%) in compound heterozygotes compared to 14/80 (18%) in homozygotes.
Fig. 2
Box plot showing (A) SF-36 scores in FKRP c.826 C > A homozygous females vs males. (B) INQoL scores in FKRP c.826 C > A homozygous females vs males. (C) SF-36 scores in FKRP c.826 C > A homozygotes vs c.826 C > A compound heterozygotes. (D) INQoL scores in FKRP c.826 C > A homozygotes vs c.826 C > A compound heterozygotes. Notes: SF-36 scales range from lowest (0) to highest (100) HRQoL. The INQoL subscales range from no (0) to maximal (100) burden. FKRP c.826 C > A compound heterozygotes are significantly younger than c.826 C > A homozygotes (Table 1).
HRQoL and age/ disease duration
In homozygotes, both age and disease duration correlated positively with the following INQoL subscales: muscle weakness (p = 0.017 and p = 0.004), fatigue (p = 0.038 and p = 0.004), dysphagia (p = 0.030 and p < 0.001), activities (p = 0.036 and p = 0.011), independence (p = 0.002 and p < 0.001), and the INQoL index (p = 0.039 and p = 0.039) (rs = 0.24–0.44). Duration was also positively correlated with the subscale of relationships (rs = 0.31, p = 0.008). Myalgia, emotions, and body image did not correlate with age or disease duration in homozygotes. In compound heterozygotes, age and disease duration did not correlate with any of the INQoL subscales.
HRQoL and LGMDR9-related symptoms/ involvements
The frequency of any level of muscle weakness was 93–97%, fatigue 67–82%, myalgia 63–68%, and dysphagia 17–28% at the three respective measurements. Multivariate regression showed that muscle weakness and fatigue were independent predictors of the INQoL index (Table 6C). Comparing of the INQoL index of the independent walkers and the wheelchair and walking aid subgroups showed that the INQoL index was associated with wheelchair dependency (p = 0.002) and more strongly with walking aids (p < 0.0001) but did not differ significantly between the wheelchair and walking aid subgroups (p = 0.10).
Fatigue and subjective sleep quality
The FSS cut-off score gave a fatigue prevalence of 41% (14/34) in females and 38% (15/39) in males. FSS correlated positively with age, PSQI, and the INQoL domains of emotions, myalgia, and muscle weakness but was unrelated to sex. Only emotions (p = 0.007) and myalgia (p = 0.009) were identified as independent correlates (Supplementary Table 1). Median FSS was relatively higher in those using walking aids (5.3) compared to wheelchair (4.2) and independent walkers (4.3), but the subgroup differences were not significant. FSS correlated strongly with the fatigue scale of INQoL (rs = 0.76, p < 0.0001), moderately with the INQoL index (r = 0.46, p < 0.0001) and, as expected, negatively with the vitality scale of SF-36 (rs = –0.68, p < 0.0001).
Applying the PSQI cut-off score, 19/34 (56 %) of females and 21/37 (43 %) males were classified as poor sleepers. PSQI was related to myalgia (p = 0.003) but not age, sex, PAP therapy, wheelchair dependency, or the INQoL domains of muscle weakness or emotions (Supplementary Table 2).
HRQoL and socideomographic variables
The proportion of participants 30–69 years old living alone was 18/62 (29%), which is comparable to corresponding age groups in the general population (21–26%) [30]. Among participants age 25–39 years, 9/14 (64%) females and 5/14 (36%) males had higher education, which are relatively similar to the 60% of females and 40% of males reported in the general population [30].
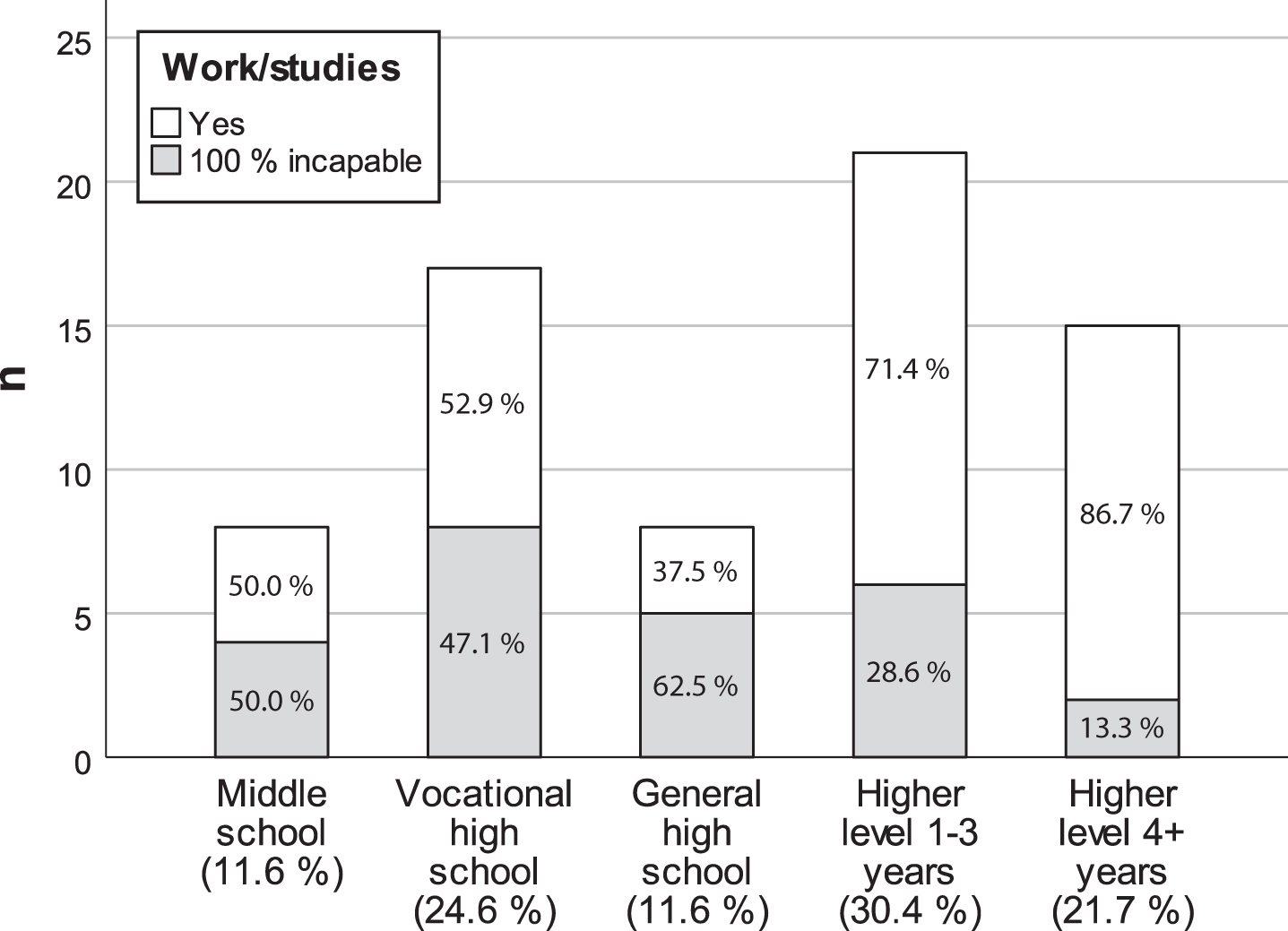
The participants living alone did not differ on any aspect of HRQoL from those living with a partner or family. Nevertheless, there was a negative trend between living alone and most aspects of HRQoL except independence. In participants 25+ years, those with higher education showed poorer HRQoL on mental health (p = 0.047) and marginally for social function (p = 0.068) as well. The INQoL index was unrelated to educational achievement (Table 6C). Median age of participants younger than 67 years with long-term or permanent 100% incapacity for work or studies was 54.5 years (range 24–66). In the age group 25–67 years, 100% study/work incapacity was associated with lower level of education (OR 3.72, 95% CI: 1.31–10.53, p = 0.011) (Fig. 3), more severe bodily pain (p = 0.021) and myalgia (p = 0.007), and poorer general health (p = 0.035), role emotions (p = 0.018), activities (p = 0.006), and independence (p < 0.001). However, the INQoL index was unrelated to study/work incapacity.
Fig. 3
Bar chart showing the distribution of participants 25–67 years old with 100% long-term sickness absence or incapacity for work or studies, according to the highest educational level achieved (n = 69).
Supportive care
In total, 17/40 (43%) females and 21/44 (45%) males received physiotherapy treatment regularly. Reasons provided for not having physiotherapy included absent treatment effect (15/46), absent needs (11/46), costs (8/46), school/work (6/46), lack of available services (4/46), transportation/ travel difficulties (3/46), and 13/46 reported other various factors. Mental health treatment was received by 3/40 females and 3/43 males while 9/40 females and 4/43 males did not receive although they reported needs for mental health treatment.
DISCUSSION
This was an exploratory study of HRQoL in a Norwegian sample with LGMDR9 comprising 75 FKRP c.826 C > A homozygotes and nine FKRP c.826 C > A compound heterozygotes. We found that both physical, social, and emotional aspects of HRQoL are compromised. Over 14 months, the homozygotes showed worsening in perceived weakness and disease burden and tentatively in activities, whereas compound heterozygotes had a tendency towards worsening in weakness but surprisingly some relief in their disease burden. Compound heterozygotes reported a higher burden on dysphagia and physical aspects of INQoL compared to homozygotes. Among homozygotes, females reported poorer HRQoL on aspects of bodily pain, social relationships, and emotions and a higher physical burden and overall burden compared to males. Disease burden was predicted by perceived muscle weakness and fatigue. Fatigue was prevalent and related to myalgia and emotions. Poor sleep was also frequent and related to myalgia.
Despite only a 14-month follow-up period, we observed a subjective worsening in muscle weakness and disease burden among c.826 C > A homozygotes. This was unexpected considering the slowly progressive nature of LGMDR9 in general and for this genotype in particular [5]. The paradoxical tendency towards a relief in burden of disease among the c.826 C > A compound heterozygotes, who generally have a more progressive phenotype, is likely related to the late disease stage of the participants. All compound heterozygous participants in the prospective study had entered a wheelchair-dependent stage, where progression may not be that easily perceived or stressful as before transition to wheelchair. This corresponds with the findings of a relatively higher disease burden and levels of fatigue in subjects with walking aids compared to wheelchair-dependent subjects. Furthermore, compared to homozygotes, compound heterozygotes also showed tendencies towards better scores on mental and social aspects despite significantly worse physical scores. These observations align with the “disability paradox”, which is the phenomenon where the perceived burden is disproportionate to the level of disability because of mental and environmental adaptations [35].
The relatively poorer HRQoL observed in females corresponds with previous studies on muscle diseases [11] but also with overall tendencies observed in normative data [28, 36]. Background clinical characteristics suggest that sex differences in disease involvement may be a contributing factor. Similar sex differences were also evident and discussed in our natural history study [2] but need further validation. Nevertheless, regardless of the causality, the current findings indicate more unmet care needs among females.
Most domains of INQoL worsened by age and disease duration. This highlights the importance of preventive and promotional care. Myalgia, body image, and emotions were unrelated to age and duration, suggesting that the potential burden of these aspects should be addressed at an early stage in the treatment.
Fatigue was predictive of disease burden, which concurs with published findings in other muscle diseases [11, 13, 37]. Applying the FSS cut-off value of≥5, 40% of the patients had clinical levels of fatigue, which is relatively high compared to 23.1% reported in a previous Norwegian general population study [27]. Fatigue is a heterogeneous and multifactorial phenomenon [38]. In our sample, fatigue severity was related to myalgia and negative emotions. In the published literature, there are strong evidence for an interrelationship between fatigue, musculoskeletal pain, and psychological distress and explanatory theories include neurotransmitter imbalances, hypothalamic-pituitary-adrenal axis dysregulation, inflammation, central sensitization, and genetic factors [39]. A previous prospective observational study on ambulatory patients with NMD suggested that fatigue also relates to the decline in physical activity that follows the progressive weakness [40]. Currently, there are accumulating evidence that moderate-intensity aerobic exercise in NMD may alleviate fatigue, and even some evidence of disease-modifying effects [41, 42], and anti-inflammatory and epigenetic mechanisms have been suggested [43]. Our findings suggest that myalgia and mental distress should be addressed in fatigued patients and both cognitive behavioral therapy and exercise therapy could be relevant approaches. However, NMD seem to respond differently to exercise and long-term data are limited [43, 44]. In this regard, both studies on short- and long-term effects of exercise therapy and comparative trials on fatigue management in LGMDR9 are required to provide evidence-based recommendations.
Myalgia was frequently reported. Nevertheless, bodily pain was comparable to the reference population, the reported consumption of pain medication for myalgia was low, and the INQoL index was unrelated to myalgia. Comparatively, in a study of Facioscapulohumeral muscular dystrophy (FSHD), a related condition well known to be associated with pain, the burden of myalgia was relatively higher (median 44.8 vs mean 25.1–31.0 of 100) and affected the INQoL index [45]. Myalgia did thus not emerge as a major issue in our LGMDR9 cohort, which is contrary to a previous study [12]. However, myalgia was associated with fatigue and poor sleep, in which myalgia may play a perpetuating role.
Subjective sleep quality did not impact disease burden. Nevertheless, the high frequency of poor-quality sleep suggests that sleep may be an overlooked aspect of care. The frequency in females (56%) and males (43%), respectively, was relatively high compared to general population studies in Germany (42.5% of females and 28.7% of males) [46] or Austria (36.5 % of females and 26.6 % of males) [47]. The overall frequency (49%) was comparable to findings in FSHD (50%) [24] and adult patients with Duchenne muscular dystrophy (55%) [25]. Although we identified a relationship with myalgia, poor sleep is unspecific and may have various underlying issues and sleep disorders that should be addressed.
The additional finding of relatively lower levels of long-term sickness absence or incapacity among participants with higher education aligns with the general pattern [48, 49]. Lastly, there were some limitations in the access to qualified physiotherapy and mental health care. These are also potential areas for improvement.
Strengths and limitations
We believe the findings are representative of the Norwegian LGMDR9 population 16+ years given the nation-wide recruitment, high response rates, and gender balance. Measurement at three time points provided insight into the longitudinal stability and change at cohort and individual level. INQoL provided information about expression of disease-related symptoms, burden, and relationships with burden. Our analyses indicated good internal validity. The use of SF-36 enabled comparison with a reference population, which was useful in the interpretations of the INQoL with regard to impaired areas of HRQoL.
This study also has some important limitations. Due to the exploratory design we accepted a higher risk of conducting type I error (false positive) and urge for caution in interpreting p-values particularly in the zone of 0.01 to 0.05 as significant. Findings that are flagged as significant should be replicated in future studies. The sample size of c.826 C > A compound heterozygotes was low, which clearly limits the statistical power. The study was conducted during the COVID-19 pandemic, which may have introduced period-specific results generalizing less well to post-COVID life. The increase in perceived muscle weakness observed in homozygotes could potentially result from inactivity due to restrictions [50]. Nevertheless, since all data collections were performed in comparable and relatively COVID-19 free periods, the cross-sectional measurements were comparable, and no longitudinal change was detected in the aspects of relationships, myalgia, fatigue, or emotions at the individual level, we believe the pandemic had minor effects on the results. Lastly, interpretations of the prospective study were also hampered by the fact that INQoL has not been validated on sensitivity in LGMD. Neither a 6-year follow-up study of patients with Dystrophia myotonica type 1 [51] nor a 20-month follow-up study of patients with Oculopharyngeal muscular dystrophy [52] detected deterioration in the INQoL measurements despite objective progression of muscle weakness.
Conclusions
This study provided insight into the impacts of LGMDR9 on HRQoL, subjective changes over a 14-months period, and relationships with disease burden. Both physical, emotional, and social aspects of HRQoL were impaired, which advocates for a multi-disciplinary care. In c.826 C > A homozygotes, perceived muscle weakness and disease burden deteriorated during the study, which is relevant information for clinical trials. However, validation of sensitivity of the INQoL measure in LGMD remains and potential COVID-19 effects should be considered. Among homozygotes, females reported a higher burden, which indicates the need for a gender-specific perspective in treatment and care. Muscle weakness and fatigue proved particularly important where fatigue is amenable to treatment. Associations suggest that myalgia and mental distress are potential targets for the treatment of fatigue, but further studies are needed to establish evidence for effective interventions. Poor sleep was frequent and will be investigated further. The combined use of a generic and a disease-specific HRQoL is particularly useful for research purposes. However, a generic HRQoL that is brief to minimize patient burden and optimize response rate, constructed for utility analyses, and with accessible norms may be preferable. Adding an item of overall QoL or happiness would have provided insight into the significance of the disease in the patient' lives as a whole and can be considered in future research.
ACKNOWLEDGMENTS
We would like to thank the participants for sharing their personal information, Jorun Willumsen and Cecilie Hæstad for administration of the instruments, Clinical Research Department, UNN, for technical and statistical support, and Geir Ringvold for assistance with figure formatting. Some of the authors are members of European Reference Network through Oslo University Hospital.
FUNDING
Financial support was received from Northern Norway Regional Health Authority.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/JND-221629.
REFERENCES
[1] | Brockington M , Yuva Y , Prandini P , Brown SC , Torelli S , Benson MA ,et al. Mutations in the fukutin-related protein gene (FKRP) identify limb girdle muscular dystrophy 2I as a milder allelic variant of congenital muscular dystrophy MDC1C, Hum Mol Genet (2001) ;10: (25):2851–9. doi: 10.1093/hmg/10.25.2851. |
[2] | Jensen SM , Müller KI , Mellgren SI , Bindoff LA , Rasmussen M , Ørstavik K ,et al. Epidemiology and natural history in 101 subjects with FKRP-related limbgirdle muscular dystrophy R9. The Norwegian LGMDR9 cohort study (2020). Neuromuscular Disorders. 2022. doi: 10.1016/j.nmd.2022.11.005. |
[3] | Sveen ML , Schwartz M , Vissing J High prevalence and phenotype-genotype correlations of limb girdle muscular dystrophy type 2I in Denmark, Ann Neurol (2006) ;59: (5):808–15. doi: 10.1002/ana.20824. |
[4] | Stensland E , Lindal S , Jonsrud C , Torbergsen T , Bindoff LA , Rasmussen M ,et al. Prevalence, mutation spectrum and phenotypic variability in Norwegian patients with Limb Girdle Muscular Dystrophy 2I, Neuromuscul Disord (2011) ;21: (1):41–6. doi: 10.1016/j.nmd.2010.08.008. |
[5] | Murphy LB , Schreiber-Katz O , Rafferty K , Robertson A , Topf A , Willis TA ,et al. Global FKRP Registry: Observations in more than 300 patients with Limb Girdle Muscular Dystrophy R9, Ann Clin Transl Neurol (2020) ;7: (5):757–66. doi: 10.1002/acn3.51042. |
[6] | Aho AC , Hultsjö S , Hjelm K Young adults’ experiences of living with recessive limb-girdle muscular dystrophy from a salutogenic orientation: An interview study, Disabil Rehabil (2015) ;37: (22):2083–91. doi: 10.3109/09638288.2014.998782. |
[7] | Hunter M , Heatwole C , Wicklund M , Weihl CC , Mozaffar T , Statland JM ,et al. Limb-girdle muscular dystrophy: A perspective from adult patients on what matters most, Muscle Nerve (2019) ;60: (4):419–24. doi: 10.1002/mus.26636. |
[8] | Haraldstad K , Wahl A , Andenæs R , Andersen JR , Andersen MH , Beisland E ,et al. A systematic review of quality of life research in medicine and health sciences, Qual Life Res (2019) ;28: (10):2641–50. doi: 10.1007/s11136-019-02214-9.2019;28(10):2641-50. |
[9] | Megari K Quality of Life in Chronic Disease Patients, Health Psychol Res. (2013) ;1: (3):23e27. doi: 10.4081/hpr.2013.e27. |
[10] | Karimi M , Brazier J Health, Health-Related Quality of Life, and Quality of Life: What is the Difference? Pharmacoeconomics (2016) ;34: (7):645–9. doi: 10.1007/s40273-016-0389-9. |
[11] | Graham CD , Rose MR , Grunfeld EA , Kyle SD , Weinman J A systematic review of quality of life in adults with muscle disease, J Neurol (2011) ;258: (9):1581–92. doi: 10.1007/s00415-011-6062-5. |
[12] | Richardson M , Mayhew A , Muni-Lofra R , Murphy LB , Straub V Prevalence of Pain within Limb Girdle Muscular Dystrophy R9 and Implications for Other Degenerative Diseases, J Clin Med (2021) ;10: (23):5517. doi: 10.3390/jcm10235517. |
[13] | Peric M , Peric S , Stevanovic J , Milovanovic S , Basta I , Nikolic A ,et al. Quality of life in adult patients with limb-girdle muscular dystrophies, Acta Neurol Belg (2018) ;118: (2):243–50. doi: 10.1007/s13760-017-0857-9. |
[14] | Kovalchick LV , Bates K , Statland J , Weihl C , Kang PB , Lowes LP ,et al. Patient reported quality of life in limb girdle muscular dystrophy, Neuromuscul Disord (2022) ;32: (1):57–64. doi: 10.1016/j.nmd.2021.11.002. |
[15] | Ware JE Jr , Sherbourne CD The MOS 36-item short-form health survey (SF-36), I. Conceptual framework and item selection. Med Care (1992) ;30: (6):473–83. |
[16] | Ware JE , Snow KK , Kosinski M , Gandek B Sf-36 Health Survey: Manual and Interpretation Guide. Boston: New England Medical Center, the Health Institute; 1993. |
[17] | Ware JE , Gandek B Overview of the SF-36 Health Survey and the International Quality of Life Assessment (IQOLA) Project, J Clin Epidemiol (1998) ;51: (11):903–12. doi: 10.1016/s0895-4356(98)00081-x. |
[18] | Contopoulos-Ioannidis DG , Karvouni A , Kouri I , Ioannidis JP Reporting and interpretation of SF-36 outcomes in randomised trials: Systematic review, Bmj (2009) ;338: :a3006. 10.1136/bmj.a3006. |
[19] | Vincent KA , Carr AJ , Walburn J , Scott DL , Rose MR Construction and validation of a quality of life questionnaire for neuromuscular disease (INQoL), Neurology (2007) ;68: (13):1051–7. doi: 10.1212/01.wnl.0000257819.47628.41. |
[20] | Mapi Research Trust [homepage on the Internet] [cited 2022 Nov 11]. Available from: https://eprovide.mapi-trust.org/instruments/individualized-neuromuscular-quality-of-life-questionnaire |
[21] | Buysse DJ , Reynolds CF , 3rd Monk TH , Berman SR , Kupfer DJ The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research, Psychiatry Res (1989) ;28: (2):193–213. doi: 10.1016/0165-1781(89)90047-4. |
[22] | Pallesen S , Nordhus I , Omvik S , Sivertsen B , Matthiesen SB , Bjorvatn B Pittsburgh Sleep Quality Index, Tidsskrift for Norsk psykologforening (2005) ;42: :714–7. |
[23] | Mollayeva T , Thurairajah P , Burton K , Mollayeva S , Shapiro CM , Colantonio A The Pittsburgh sleep quality index as a screening tool for sleep dysfunction in clinical and non-clinical samples: A systematic review and meta-analysis, Sleep Med Rev (2016) ;25: :52–73. doi: 10.1016/j.smrv.2015.01.009. |
[24] | Della Marca G , Frusciante R , Vollono C , Dittoni S , Galluzzi G , Buccarella C ,et al. Sleep quality in Facioscapulohumeral muscular dystrophy, J Neurol Sci (2007) ;263: (1-2):1549–53. doi: 10.1016/j.jns.2007.05.028. |
[25] | Crescimanno G , Greco F , Abbate A , Canino M , Bertini M , Marrone O Subjective sleep quality in adult patients affected by Duchenne muscular dystrophy, Beyond nocturnal hypoventilation. Sleep Medicine (2020) ;69: :168–71. doi: 10.1016/j.slee2020.01.025. |
[26] | Krupp LB , LaRocca NG , Muir-Nash J , Steinberg AD The fatigue severity scale, Application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol (1989) ;46: (10):1121–3. doi: 10.1001/archneur.1989.00520460115022. |
[27] | Lerdal A , Wahl A , Rustøen T , Hanestad BR , Moum T Fatigue in the general population: A translation and test of the psychometric properties of the Norwegian version of the fatigue severity scale, Scand J Public Health (2005) ;33: (2):123–30. doi: 10.1080/14034940410028406. |
[28] | Jacobsen EL , Bye A , Aass N , Fosså SD , Grotmol KS , Kaasa S ,et al. Norwegian reference values for the Short-Form Health Survey 36: Development over time, Qual Life Res (2018) ;27: (5):1201–12. doi: 10.1007/s11136-017-1684-4. |
[29] | Jacobsen EL , Bye A , Aass N , Fosså SD , Grotmol KS , Kaasa S ,et al. Correction to: Norwegian reference values for the Short-Form Health Survey 36: Development over time, Qual Life Res (2018) ;27: (5):1213–5. doi: 10.1007/s11136-017-1708-0. Erratum for: Qual Life Res. 2018;27(5):1201-1212 |
[30] | Statistics Norway [homepage on the Internet] [cited 2022 June 25]. Available from: www.ssb.no/en. |
[31] | Althouse AD Adjust for Multiple Comparisons? It’s Not That Simple, Ann Thorac Surg (2016) ;101: (5):1644–5. doi: 10.1016/j.athoracsur.2015.11.024. PMID: 27106412 |
[32] | Norwegian Institute of Public Health [homepage on the Internet] [cited 2022 Nov 02]. Available from: www.fhi.no/en/. |
[33] | PSQI developed by Buysse DJ , Reynolds CF , Monk TH , Berman SR , Kupfer DJ of the University of Pittsburgh using National Institute of Mental Health Funding. Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ: Psy-chiatry Research. (1989) ;28: , 193–213. |
[34] | FSS© 1985 Lauren B. Krupp. Reproduced with permission from the author. |
[35] | Albrecht GL , Devlieger PJ The disability paradox: High quality of life against all odds, Soc Sci Med (1999) ;48: (8):977–88. doi: 10.1016/s0277-9536(98)00411-0. |
[36] | Garratt AM , Hansen TM , Augestad LA , Rand K , Stavem K Norwegian population norms for the EQ-5D-5L: Results from a general population survey, Qual Life Res (2022) ;31: (2):517–26. doi: 10.1007/s11136-021-02938-7. |
[37] | van der Sluijs BM , Knoop H , Bleijenberg G , van Engelen BG , Voermans NC The Dutch patients’ perspective on oculopharyngeal muscular dystrophy: A questionnaire study on fatigue, pain and impairments, Neuromuscul Disord (2016) ;26: (3):221–6. doi: 10.1016/j.nmd.2015.12.009. |
[38] | Finsterer J , Mahjoub SZ Fatigue in Healthy and Diseased Individuals, Am J Hosp Palliat Care (2014) ;31: (5):562–75. doi: 10.1177/1049909113494748. |
[39] | Løke D , Løvstad M , Andelic N , Andersson S , Ystrom E , Vassend O The role of pain and psychological distress in fatigue: A co-twin and within-person analysis of confounding and causal relations, Health Psychol Behav Med (2022) ;10: (1):160–79. doi: 10.1080/21642850.2022.2033121. |
[40] | Kalkman JS , Schillings ML , van der Werf SP , Padberg GW , Zwarts MJ , van Engelen BG ,et al. Experienced fatigue in facioscapulohumeral dystrophy, myotonic dystrophy, and HMSN-I, J Neurol Neurosurg Psychiatry (2005) ;76: (10):1406–9. doi: 10.1136/jnn2004.050005. |
[41] | Janssen B , Voet N , Geurts A , van Engelen B , Heerschap A Quantitative MRI reveals decelerated fatty infiltration in muscles of active FSHD patients, Neurology (2016) ;86: (18):1700–7. doi: 10.1212/WNL.0000000000002640. |
[42] | Heskamp L , Okkersen K , Nimwegen Mv ,et al. Quantitative Muscle MRI Depicts Increased Muscle Mass after a Behavioral Change in Myotonic Dystrophy Type 1, Radiology (2020) ;297: (1):132–42. doi: 10.1148/radiol.2020192518. |
[43] | Voet NBM Exercise in neuromuscular disorders: A promising intervention, Acta Myol (2019) ;38: (4):207–14. PMID: 923 31970319. |
[44] | Sheikh AM , Vissing J Exercise therapy for muscle and lower motor neuron diseases, Acta Myol (2019) ;38: (4):215–32. |
[45] | Morís G , Wood L , FernáNdez-Torrón R , González Coraspe JA , Turner C , Hilton-Jones D ,et al. Chronic pain has a strong impact on quality of life in facioscapulohumeral muscular dystrophy, Muscle Nerve (2018) ;57: (3):380–7. doi: 10.1002/mus.25991. |
[46] | Hinz A , Glaesmer H , Brähler E , Löffler M , Engel C , Enzenbach C ,et al. Sleep quality in the general population: Psychometric properties of the Pittsburgh Sleep Quality Index, derived from a German community sample of 9284 people, Sleep Med (2017) ;30: :57–63. doi: 10.1016/j.slee2016.03.008. |
[47] | Tribl GG , Schmeiser-Rieder A , Rosenberger A , Saletu B , Bolitschek J , Kapfhammer G ,et al. Sleeping habits in the Austrian population, Sleep Med (2002) ;3: (1):21–8. doi: 10.1016/s1389-9457(01)00117-4. |
[48] | Seglem KB , Ørstavik R , Torvik FA , Røysamb E , Vollrath M Education differences in sickness absence and the role of health behaviors: A prospective twin study, BMC Public Health (2020) ;20: (1):1689. doi: 10.1186/s12889-020-09741-y. |
[49] | Baumberg B From Impairment to Incapacity –Educational Inequalities in Disabled People’s Ability to Work, Social Policy & Administration (2015) ;49: (2):182–98. doi: 10.1111/spol.12118. |
[50] | Roberts-Lewis SF , Ashworth M , White CM , Rose MR COVID-19 lockdown impact on the physical activity of adults with progressive muscle diseases, BMJ Neurol Open (2021) ;3: (1):e000140. doi: 10.1136/bmjno-2021-000140. |
[51] | Peric S , Heatwole C , Durovic E , Kacar A , Nikolic A , Basta I ,et al. Prospective measurement of quality of life in myotonic dystrophy type 1, Acta Neurol Scand. (2017) ;136: (6):694–7. doi: 10.1111/ane.12788. |
[52] | Kroon RHMJM , Kalf JG , de Swart BJM , van der Sluijs BM , Glennon JC , Raz V ,et al. Longitudinal Assessment of Strength, Functional Capacity, Oropharyngeal Function, and Quality of Life in Oculopharyngeal Muscular Dystrophy, Neurology (2021) ;11: (15):e1475–83. doi: 10.1212/WNL.0000000000012640. |




