COVID-19 in a Cohort of Patients with Congenital Myasthenic Syndrome
Abstract
Congenital Myasthenic Syndromes (CMS) are a rare group of genetic disorders of neuromuscular transmission. Some subtypes of CMS can be associated with respiratory and bulbar weakness and these patients may therefore be at high risk of developing a severe disease from COVID-19. We screened 73 patients with genetically confirmed CMS who were attending the UK national referral centre for evidence of previous Severe Acute Respiratory Syndrome Corona Virus 2 infection and their clinical outcome. Of 73 patients, seven had history of confirmed COVID-19. None of the infected patients developed a severe disease, and there were no signals that CMS alone carries a high risk of severe disease from COVID-19.
INTRODUCTION
Congenital Myasthenic Syndromes (CMS) are a rare group of heterogenous inherited neuromuscular disorders caused by mutations in genes that encode proteins essential for maintaining normal neuromuscular transmission [1]. Their characteristic clinical feature is fatigable muscle weakness. Some CMS subtypes are associated with respiratory muscle and bulbar weakness causing apnoea, respiratory failure and swallowing difficulties with inhalation risk [2]. These patients could be at high risk of severe disease from COVID-19 and thus have been categorised as at increased risk.
Here we assess CMS patients seen in our UK national referral centre for evidence of previous Severe Acute Respiratory Syndrome Corona Virus 2 (SARS-CoV-2) infection.
METHOD
A cross-sectional, single-centre audit of a cohort of patients with CMS at our nationally commissioned highly specialised CMS service in Oxford, United Kingdom. When patients attended regular clinic appointments, questions on symptoms of possible COVID-19 in patient and their household were asked, along with the results of any nasopharyngeal swab polymerase chain reaction (PCR) tests or antibody tests done locally. SARS-CoV-2 anti-nucleocapsid IgG testing was also offered in Oxford and those who consented had a blood test taken locally, returned back to us in pre-paid kits and were analysed at our centre. In patients with confirmed diagnosis of COVID-19, information was also collected on household contacts as a comparator group. Medical records were reviewed for demographics, diagnosis, symptoms of CMS and recent examination findings.
RESULTS
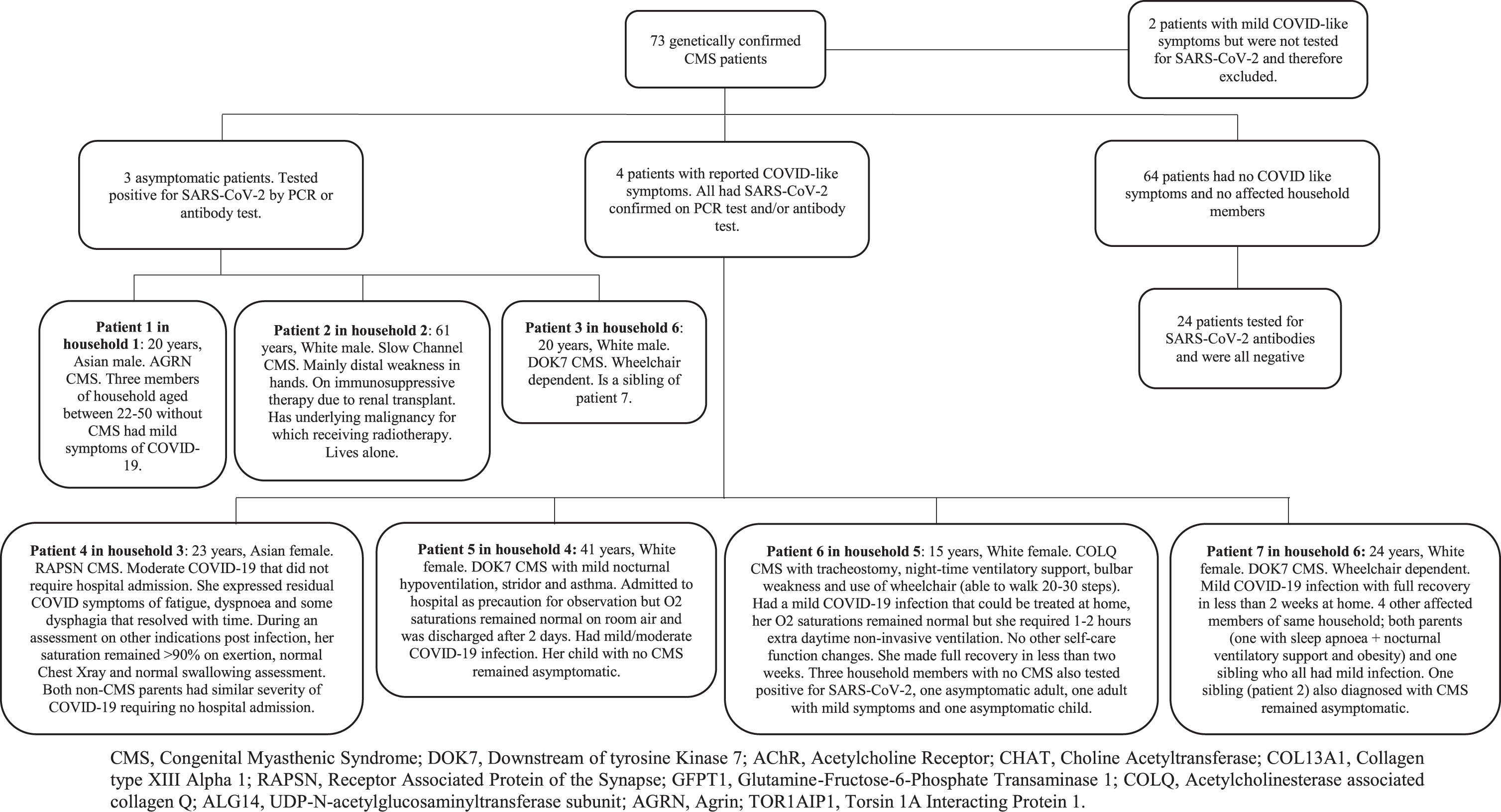
A total of 73 patients with confirmed genetic diagnosis of CMS were included. Their median age, ethnicity and distribution of CMS subtypes are summarised in Table 1. Seven patients developed evidence of SARS-CoV-2 infection, confirmed on either PCR or antibody test. Three remained asymptomatic and four developed mild to moderate COVID-19 clinical symptoms with no requirement for supplementary oxygen. Only one patient who also had asthma was admitted to hospital for observation for two days. The patient whose CMS was associated with the highest respiratory and bulbar compromise requiring nocturnal non-invasive ventilation (NIV) via tracheostomy and modified diet had a mild disease and was treated at home with temporary increase in NIV support. One patient with CMS and other serious health conditions (underlying malignancy, renal transplant with immunosuppressive therapy and dialysis, asthma and ischaemic heart disease) had an asymptomatic COVID-19.
Table 1
Descriptive of patients included in the study
| All patients. N = 73 | Patients with confirmed COVID-19. N = 7 | |
| Median age in years (range) | 32 (8–77) | 23 (15–61) |
| Subtype of CMS (N) | ||
| DOK7 | 21 | 3 |
| Fast Channel CMS | 3 | |
| Slow Channel CMS | 12 | 1 |
| CHRNE | 13 | |
| CHAT | 1 | |
| COL13A1 | 1 | |
| RAPSN | 7 | 1 |
| GFPT1 | 3 | |
| COLQ | 6 | 1 |
| ALG14 | 1 | |
| PLEC | 1 | |
| AGRN | 1 | 1 |
| TOR1AIP1 | 1 | |
| CHRNA1 | 1 | |
| MUSK | 1 | |
| Ethnicity | ||
| White | 58 | 5 |
| Asian | 14 | 2 |
| Mixed race | 1 |
SD, Standard Deviation; CMS, Congenital Myasthenic Syndrome; DOK7, Downstream Of tyrosine Kinase 7; CHRNE, Cholinergic Receptor Nicotinic Epsilon subunit; CHAT, Choline Acetyltransferase; COL13A1, Collagen type XIII Alpha 1; RAPSN, Receptor Associated Protein of the Synapse; GFPT1, Glutamine-Fructose-6-Phosphate Transaminase 1; COLQ, Acetylcholinesterase associated collagen Q; ALG14, UDP-N-acetylglucosaminyltransferase subunit; PLEC, Plectin; AGRN, Agrin; TOR1AIP1, Torsin 1A Interacting Protein 1; CHRNA1, Cholinergic Receptor Nicotinic Alpha 1 subunit; MUSK, Muscle Specific Kinase.
When compared to other non-CMS household members, the CMS patients infected with SARS-CoV-2 did not appear to develop a more severe disease. Within two families, there were three examples of age-matched siblings without CMS or additional risk factors who experienced more or equal COVID-19 symptoms. Additionally, SARS-CoV-2 infection was reported in non-CMS household members of almost all affected patients.
None reported any significant long-term worsening of their CMS. Figure 1 summarises our findings.
Fig. 1
Flowchart and summary of results.
DISCUSSION
Our report of a relatively large CMS cohort identified COVID-19 in less than 10%of our patients. However, of those affected, the majority had mild symptoms and interestingly, within infected households the CMS patients did not appear to be at increased risk of severe infection compared to age similar family members. None of the infected patients required supplementary oxygen therapy and only one patient had a short observational admission to hospital related to her underlying asthma.
Although infection can trigger worsening symptoms of CMS [3], three of our patients remained asymptomatic and the remaining four developed only a mild to moderate disease. Both patients who were considered to have the highest morbidity, including a patient who had respiratory and bulbar compromise (patient 6) and a patient who was on immunosuppressive therapy and had an active malignancy (patient 2) did remarkably well and remained either asymptomatic or had a mild self-limiting disease with full recovery. In COVID-19 affected patients, it is difficult to know if some of the COVID symptoms could be attributed to worsening of the CMS but the similar severity of COVID-19 symptoms in other family members would suggest CMS worsening was not a major feature. Of note, young children with CMS who tend to have significant worsening of their CMS with infections tend to be least likely to develop symptomatic COVID-19.
Our report is the largest study on CMS and COVID-19, and the results are in line with a previous published single case report of a mild case [4]. We were unable to assess the overall risk of developing COVID-19 in CMS as many of our patients were advised to shield. Due to the small numbers an increased risk of infection and severe disease cannot be ruled out, particularly in those with other risk factors that led to shielding (e.g., older age, other comorbidities). However, from this series there are no signals that CMS alone carries a high risk of severe COVID-19.
ETHICAL CONSIDERATIONS
The study is supported by Neuromuscular disorders research tissue bank, 21/SC/0018 and clinical audit 6073, with the title: Prevalence and clinical outcomes of COVID-19 in patients attending a nationally commissioned specialist service.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
We would like to express our special thanks to our patients and to the UK National Specialised Commissioning Team for funding of the Diagnostic and Advisory service for CMS in Oxford.
REFERENCES
[1] | Rodriguez Cruz PM , Palace J , Beeson D . Inherited disorders of the neuromuscular junction: An update. Journal of Neurology. (2014) ;261: (11):2234–43. |
[2] | Palace J , Beeson D . The congenital myasthenic syndromes. Journal of Neuroimmunology. (2008) ;201-202: :2–5. |
[3] | Abicht A , Müller JS , Lochmüller H . Congenital Myasthenic Syndromes. In: Adam MP, Ardinger HH, Pagon RA,Wallace SE, Bean LJH, Stephens K, et al., editors. GeneReviews(®). Seattle (WA): University ofWashington, Seattle Copyright© 1993–2020, University ofWashington, Seattle. GeneReviews is a registered ™ of the University of Washington, Seattle. All rights reserved.; 1993. |
[4] | Bonanno S , Maggi L . Favorable outcome of COVID-19 infection in a patient with congenital myasthenic syndrome. Neurological sciences: Official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology. (2021) ;42: (4):1253. |



