The changing landscape of SARS-CoV-2: Implications for the maternal-infant dyad
Abstract
The COVID-19 pandemic represents the greatest challenge to date faced by the medical community in the 21st century. The rate of rapid dissemination, magnitude of viral contagiousness, person to person transmission at an asymptomatic phase of illness pose a unique and dangerous challenge for all patients, including neonatal and obstetric patients. Although scientific understanding of the pathophysiology of the disease, nature of transmission, and efficacy of mitigation strategies is growing, neither a cure or vaccine have been developed. While COVID-19 is primarily a disease of older patients, infection is now seen across all age demographics with reports of illness in pregnant patients and infants. Altered hormone status and predominance of Th-2 immune helper cells may result in increased predisposition to SARS-CoV-2. Case reports of pregnant patients demonstrate a clinical presentation comparable to non-pregnant adults, but evidence of vertical transmission to the fetus is controversial. Neonatal reports demonstrate an inconsistent and non-specific phenotype, and it is often difficult to separate COVID-19 from the underlying conditions of prematurity or bacterial infection. The development of international registries to enable risk profiling of COVID-19 positive pregnant mothers and/or their offspring may facilitate the development of enhanced mitigation strategies, medical treatments and effective vaccinations.
1Introduction
In December of 2019 cases of an unknown viral pneumonia were reported from Wuhan, Hubei, China [1]. After extensive scientific investigation by Chinese scientists, the virus was named the 2019 novel coronavirus in January 2020, before the World Health Organization (WHO) formally renamed the disease COVID-19 [2]. COVID-19 was officially declared a pandemic on March 11, 2020 [3]. As of May12th 2020 there are over 4,250,000 cases, spanning 180 countries, with over 286,000 deaths [4]. Although much uncertainty remains, regarding the natural history and demographics of COVID-19, the virus appears to primarily cause infection in adults over 51 with case fatality rates increasing dramatically with age [5]. Appearance of symptoms, which usually include fever, cough, fatigue, and dyspnea with occasional gastrointestinal tract symptoms [6], typically occurs following five days of incubation [7]. In addition to pulmonary infection, the virus appears to have manifestations in the central nervous system [8] and heart [9].The published literature largely focuses on the biologic effects of COVID-19 in adults, the burden to adult health care systems, and repercussions on society. The impact on perinatal health care is unique; specifically, the risks to maternal health may extend not only to health care providers in the delivery room, but to the newly born infant, and neonatal intensive care unit providers. As the disease spreads and testing capabilities expand, the likelihood of encountering COVID-19 positive pregnant women and/or neonates increases. The true magnitude of the pandemic to maternal, fetal, and neonatal health will only be realized as the case mix rises. The goal of this review is to examine the patho-biology of maternal COVID disease and possible effects on the developing fetus, provide an overview of published evidence related to the mother, and to appraise strategies to modulate the burden of illness.
2Microbiology, immunology and pathophysiology
Genetic sequencing identified that the virus belongs to the coronavirus family. Coronaviridae are enveloped, positive sense, single stranded RNA viruses with a large genome [10]. Coronavirus often infects bats and other mammalian hosts but, particularly the genera betacoronavirus, has the potential to infect human populations. Severe acute respiratory syndrome (SARS-CoV) and Middle East respiratory syndrome (MERS-CoV) are betacoronaviruses that have led to recent epidemics. Sequencing has determined that the cause of COVID-19, officially named SARS-CoV-2, is also a member of the betacoronarvirus genera [11], and its genome is nearly 80% identical to SARS-CoV. SARS-CoV-2 is a novel enveloped RNA betacoronavirus, which infects host respiratory epithelial cells. It is thought to act through angiotensin-converting enzyme 2 (ACE2), a membrane-bound aminopeptidase, which functions as its putative receptor. Whilst ACE2 is predominantly expressed within type II alveolar cells of the lung, the receptor is also present in several extrapulmonary sites including the mucosa of the oral cavity [12]. Sexual diamorphism has been hypothesized based on cellular studies which reveal attenuated expression of ACE2 in females. In general, the magnitude of the physiological and mechanical changes induced by pregnancy place the mother at increased risk of infection. These are likely to be exaggerated when respiratory health is adversely compromised or in an advanced stage of pregnancy when functional residual capacity decreases steadily, due to diaphragmatic splinting by the gravid uterus. Both the Spanish (1918) and Asian (1968) Flu pandemics were characterized by increased mortality in pregnant mothers and infants less than one year. On the contrary, SARS-CoV-2 appears to be less pathogenic in these patients.
There is immunologic evidence that the pregnancy bias towards T-helper 2 (Th2) system dominance and fetus protection leaves the mother vulnerable to viral infections, which are more effectively contained by the Th1 system. In pregnancy, the attenuation in cell-mediated immunity by Th1 cells due to the physiological shift to a Th2 cell dominant environment contributes to increased maternal susceptibility to intracellular pathogens like viruses [13]. Non-pregnant patients with COVID-19 demonstrate activation of both Th1 and Th2 immunity, characterized by the presence of both microbicidal/pro-inflammatory cytokines (IFNγ and IL-1β) in addition to anti-inflammatory cytokines (IL-4 and IL-10) [1]. In pregnant mothers with COVID-19 the immune responses may be variable and characterized by early adaptive immune responses predictive of milder disease severity [14]. It has been postulated that the combined effects of changes in the hormonal milieu of pregnancy which negatively influence immunological responses to viral pathogens [15] and the biological transition to a Th2 predominant environment, result in a less severe COVID-19 clinical phenotype compared to non-pregnant female. These biologic factors may place pregnant mothers, their fetus and health care workers who take care of them at increased risk compared to individuals of a comparable age, although there is no published evidence to substantiate these concerns.
While respiratory secretions appear to be the primary means, evidence exists to suggest that transmission may occur through multiple routes, including ocular and fecal oral routes [16]. Patients with active COVID-19 have been reported to have elevated inflammatory markers, D-dimer, and proinflammatory cytokines [1]. Of particular concern are the risks of COVID-19 infection to healthcare workers and the ability of spread to occur through asymptomatic patients; for example in Lombardi, Italy up to 20% of health care workers became infected [17]. COIVD-19 is highly transmissible and infection can occur with a very short exposure time.
Diagnosis of COVID-19 has largely been based on the combination of a clinical presentation, lab data and viral testing; however, computerized tomography (CT) imaging has played an additional supportive role. The radiologic findings (Fig. 1) are non-specific and include bilateral (64 (79%) patients), peripheral (44 (54%) patients), ill-defined (66 (81%) patients), and ground-glass opacification (53 (65%) patients), and mainly involving the right lower lobes in 225 (27%) of 849 affected patients[18]. CT findings have been categorized into different phases: an early phase defined as 0–7 days from symptom onset, and a late phase defined as 7–14 days from symptom onset [6]. Early phase appearance has been categorized predominantly by bilateral peripheral ground glass opacities. As the disease progresses to the late phase, there is increased lung involvement and the development of linear opacities, ground glass opacities with reticulation (“crazy paving” pattern) and “reverse halo” sign [6].The overall sensitivity for CT to identify the signs of pneumonia is high, however, its ability to perform as a diagnostic tool specific for COVID-19 remains unknown. Data from a Chinese cohort revealed that the sensitivity, specificity, positive predictive value (PPV), and negative predictive (NPV) value of a chest CT in diagnosing COVID-19 are 97%, 25%, 65%, and 83% respectively [19]. The clinical utility of CT needs further characterization, particularly in pregnant females where the adverse effects of ionizing radiation to the mother and the fetus need to be considered.
Fig. 1
Chest radiograph (Panel A) of COVID positive 18-year-old female patient with non-specific radiographic evidence of bilateral pneumonia. Computerized Tomography (Panel B) from 4 pregnant mothers reveals diffuse reduction in brightness of both lungs and large multiple areas of ground-glass opacities or patchy shadow with an uneven density [29]. [Panel B image has been approved by Lancet Infectious Disease].
![Chest radiograph (Panel A) of COVID positive 18-year-old female patient with non-specific radiographic evidence of bilateral pneumonia. Computerized Tomography (Panel B) from 4 pregnant mothers reveals diffuse reduction in brightness of both lungs and large multiple areas of ground-glass opacities or patchy shadow with an uneven density [29]. [Panel B image has been approved by Lancet Infectious Disease].](https://content.iospress.com:443/media/npm/2020/13-3/npm-13-3-npm200460/npm-13-npm200460-g001.jpg)
3The impact of COVID-19 on the pregnant mother
In total, 55 pregnant women who have been positive for Covid-19 have been reported in the literature, all of whom were diagnosed in the third trimester [20]. Of those infected, there have been no published cases of maternal death and two cases of severe infection [20, 21]. A recent review draws attention to two maternal deaths, secondary to ARDS following delivery, reported by the Iranian Ministry of Health and Medical Education; however at the time of this publication these cases were not listed on PubMed [22]. Most evidence supports that pregnant women present with symptoms like non-pregnant adults [20]. There are, however, emerging case reports of pregnant mothers who test positive for COVID-19 infection and who remain either completely asymptomatic [23] and or manifest mild symptoms in the subsequent 24 hours following delivery. Recent data from universal screening in a New York urban center revealed that 13.5% of asymptomatic women were positive for SARS-CoV-2 [24]. In sick patients, symptoms include low grade fever and mild respiratory symptoms [25]. From a diagnostic standpoint, pregnant women appear to have similar findings on laboratory evaluation, chest x-ray and CT scan. Imaging findings include peripheral airspace shadowing, bilateral multi-lobar ground-glass opacities or consolidation [20]. Table 1 provides a summary of all case series of pregnant mother illness at the time of the current review.
Table 1
Obstetric COVID-19 reports published in the literature
| Author | Year | Location | Study | Cases | Main conclusions |
| design | (n) | ||||
| Zhu et al. [27] | 2020 | China | Case series | 9 | Notable obstetric outcomes: intrauterine fetal distress, premature labor. Neonatal outcomes: respiratory distress, thrombocytopenia, DIC &death. No vertical transmission |
| Chen et al. [26] | 2020 | China | Case series | 9 | Disproportionately affects 3rd trimester of pregnancy. Common lab abnormality: lymphopenia. COVID-19 pneumonia in pregnancy appears like other adults. No ICU admissions or maternal deaths, no evidence of SARS-CoV-2 virus in amniotic fluid, cord blood or breastmilk. No vertical transmission or adverse neonatal outcomes |
| Liu et al. [23] | 2020 | China | Case series | 15 | All patients had CT evidence of mild pneumonia with early round glass opacities. Pregnancy and delivery do not cause exacerbation of symptoms or CT findings. No adverse neonatal outcomes |
| Zhang et al. [50] | 2020 | China | Case control | 16 | No difference in rates of fetal distress, meconium, preterm birth or neonatal asphyxia. Significantly higher rates of uterine atony requiring uterotonics |
| Di Mascio et al. [33] | 2020 | Italy, USA | Metanalysis | 32 | Pooled analysis of above studies concluded significantly higher rates of preterm birth, preeclampsia and perinatal death |
| Breslin et al. [28] | 2020 | USA | Case series | 7 | Asymptomatic carriers are present in substantial numbers and respiratory deterioration is relatively rapid |
| Yu et al. [29] | 2020 | China | Case series | 7 | Symptoms included fever (n = 6), cough (n = 1), SOB (n = 1), diarrhea (n = 1). Lab abnormalities included lymphopenia, thrombocytopenia, elevated liver function tests, elevated c-reactive protein, elevated D-dimer. All delivered by cesarean section. All treated with oxygen but no ICU admissions. No adverse neonatal outcomes |
Abbreviations: DIC, disseminated intravascular coagulation; SOB, shortness of breath.
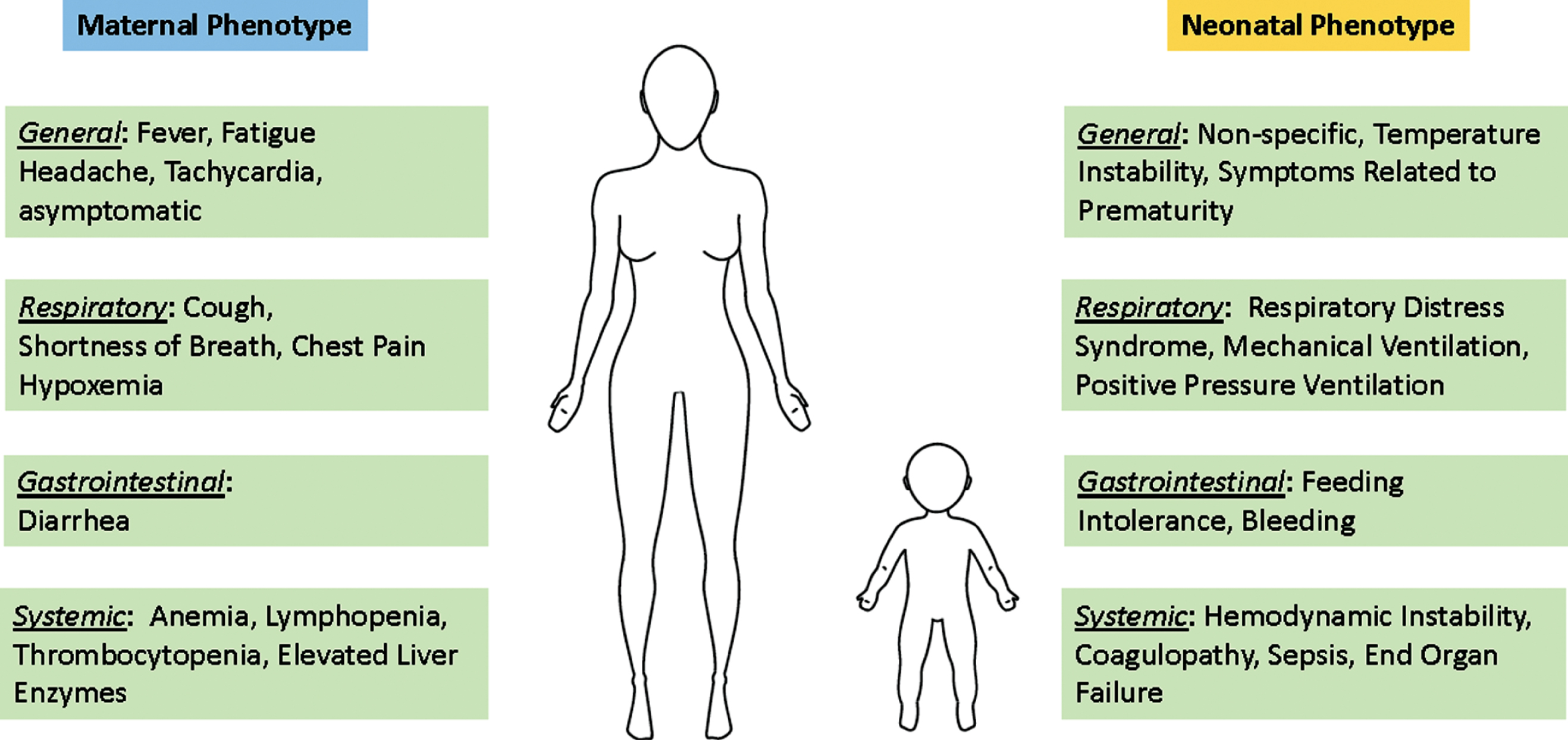
3.1Clinical presentation of the pregnant woman (Fig. 2)
A retrospective review, published in Lancet, described the clinical findings in nine COVID-19 positive women. Seven of the nine woman presented with fever, four presented with cough, but none developed severe pneumonia or required intubation. Six women developed lymphopenia and elevated C-reactive protein and three had elevation of liver enzymes [26]. Zhu et al. reported similar characteristics from nine COVID-19 positive mothers and their 10 neonates. Fever and cough were the predominant presenting symptoms, and six of the nine women had symptoms appear either before or on the day of delivery [27]. In these two reports, premature delivery was common but attributed to maternal illness. Although, four of nine [44%, range 36–36+6 weeks gestation] and six of ten [60%, range31–33+6 weeks gestation] were delivered preterm, no fetal or neonatal deaths were reported. Breslin et al. report that two of seven women presenting in New York were asymptomatic, despite positive COVID-19 testing [28]. A retrospective study by Yu et al., involving seven pregnant women admitted to Tongji hospital, reveals additional clinical information [29]. Their average age was 32 years, illness incubation period was 5 days, and gestational age at delivery was 39+1 weeks. All received oxygen via nasal cannula, however none required critical care. Symptoms included fever (n = 6), cough (n = 1) shortness of breath (n = 1) and diarrhea (n = 1). Three women had coinfection with influenza H1N1 or Legionella pneumophilia, and CT imaging for six of the women demonstrated bilateral pneumonia. All women underwent caesarian section, and all were discharged home. Only one infant tested positive. This patient was 36 hours of age at the time of diagnosis, was transferred to a local children’s hospital, remained well and was ultimately discharged home after 2 weeks and repeat negative testing[29].
3.2Implications on pregnancy outcomes
Pregnancy complications associated with COVID-19 include miscarriage (2%), intrauterine growth restriction (10%), and preterm birth (39%) [20]. There is little evidence to date regarding the impact of COVID-19 infection on the developing fetus in the 1st or early 2nd trimester. Concern exists, however, based on evidence from prior viral epidemics that early gestation may represent a period of increased fetal and maternal risk [30]. During the SARS outbreak in 2003, there were several reported first trimester spontaneous miscarriages and evidence suggests that ongoing pregnancies were complicated by intrauterine growth restriction and neonatal ischemic gut injury [31]. There is a high rate of cesarean section among those affected; however, this may relate to delivery trends in China which predated the pandemic or maternal illness. Of those who attempted vaginal delivery, there is some evidence of higher rates of fetal distress, although there is no reported increased rate of hypoxic-ischemic encephalopathy or need for therapeutic hypothermia [32].
The impact of COVID-19 on preterm birth has recently been examined in a meta-analysis of pregnant patients from three coronavirus-related outbreaks, which includes data from six studies on 41 pregnant mothers who were infected with COVID-19 [33]. The authors concluded increased risk of preterm birth in COVID-19 positive pregnant mothers. Unfortunately, the relatively high rate of preterm birth was not subdivided into spontaneous and iatrogenic preterm delivery, which is vital to discerning the role of COVID-19 on development of preterm labor. The incidence of pre-eclampsia and perinatal death was also elevated in the COVID-19 cohort, although the strength of this association was attenuated. Of note, as with the individual series mentioned previously, pooled proportions show that >90% of patients presented with some form of pneumonia and the cesarean section (CS) rate was 91%. In contrast, outcomes such as preterm premature rupture of membranes (PPROM) and fetal growth restriction (FGR) were not significantly elevated in the COVID-19 group.
3.3Pregnancy specific transmission
Neither SARS nor MERS demonstrated evidence of vertical transmission, however the mortality rate in pregnant women reached 25% and 37%, respectively [34]. The risk of vertical transmission of COVID-19, however, is biologically plausible given wide expression of the ACE2 receptor in the placenta [35]. Although clinical evidence is lacking, the case numbers to date of COVID-19 in pregnancy remain very low [32] and case reports of two neonates who tested positive for SARS-CoV-2 shortly after birth lends some credence to the concern. The methods of perinatal transmission through other maternal-fetal-neonatal exposures remain unclear. Samples of amniotic fluid, cord blood, breast milk,and neonatal throat swabs have failed to yield any viral isolates [26] and there has been no evidence of COVID-19 in placental specimens.
In summary, pregnant women represent a population at risk for the development of COVID-19 related illness. Alterations to the immune system and increased susceptibility to respiratory pathogens during pregnancy represent known risk factors for intrapartum sepsis. The magnitude of the impact of COVID-19 remains unclear, due to the limited number of cases reported, however in most cases the illness is mild. Future studies should investigate the currently presumed differential impact of SARS-CoV2 on the health of pregnant mothers, compared to prior pandemics, the effect of illness earlier in pregnancy, and long-term consequences to both maternal and infant health.
4The impact of COVID-19 to the newly born infant
As of May 12th, there has yet to be a single published report of a case of neonatal COVID-19 illness in the Unites States, although anecdotal evidence suggests otherwise. The evidence for neonatal transmission, immune susceptibility, viral penetrance, and clinical phenotype remains scarce. To date, limited case studies describe the clinical phenotype of early onset neonatal infection which would appear to be mild (Fig. 2). In one of the largest maternal case series from China, of 33 infants born to COVID-19 positive mothers only three (11%) infants tested positive [36]. All mothers had evidence of pneumonia on CT and had positive nasopharyngeal (NP) swabs for SARS-CoV-2. None required the provision of intensive care. All infants were delivered by caesarian section and noted to be appropriately grown, without dysmorphic features or malformation sequences. Nasopharyngeal and anal swabs were positive on postnatal days 2 and 4, but negative between days 6 and 7. Two of the three infants were born at term and all survived. The third infant was born by caesarian section, secondary to maternal distress, at 31 week’s gestation. The infants APGAR scores were 3, 4 and 5 at 1, 5 and 10 minutes respectively. A clinical diagnosis of respiratory distress syndrome (RDS) was made and the infant required intubation. The infant developed a sepsis-like illness with hemodynamic instability and coagulopathy. Routine blood culture was, however, positive for an Enterobacter species which was managed with antibiotics. The infant’s symptoms could not be solely attributed to COVID-19, due to concurrent prematurity, sepsis and relative asphyxia.
Fig. 2
Symptomatology of obstetric and neonatal COVID-19 infection. Maternal symptoms have been reported as like the general population. Neonatal symptoms have been reported, but it important to note that distinguishing the phenotype and separating the etiology of the symptoms as specific to COVID-19, rather than bacterial infection or prematurity, has yet to occur.
In a research letter, Dong et al. reported a possible case of vertical transmission in a 29 year old women with radiographic evidence of pneumonia and a positive NP swab for COVID-19 [37]. The mother received antibiotics, oxygen, corticosteroids, and antiviral drugs prior to delivery. The mother wore an N95 mask and did not hold the infant following the delivery. The infant was asymptomatic at birth, had Apgar scores of 9 and 10 and was immediately quarantined. Of note, the infant demonstrated elevated SARS-CoV-2 IgM and IgG levels at two hours of life, but serial NP swabs for SARS-CoV-2 were negative. At time of discharge, immunoglobin levels were still elevated. The presence of IgG levels at two hours of life could be explained by elevated maternal levels, however IgM antibodies do not freely cross the placenta [38]. It is difficult to determine whether the elevated IgM was due to fetal infection, delivery, or an unrelated process. Without testing amniotic fluid or placenta specimens, no definitive conclusions can be drawn. Increased availability of IgG and IgM testing has improved the quality of surveillance and detection of SARS-CoV-2. Zeng et al. reported immunoglobulin levels from six infants born to COVID-19 positive mothers between February 16th and March 6th [39]. All mothers underwent caesarian section and the infants were immediately isolated following delivery. Neonatal viral throat and serum swabs were all negative for SARS-CoV-2, but IgG and IgM were elevated in five and two respectively, out of the six infants. While the sample size is small and no evaluation of the placenta or amniotic fluid occurred, the report raises concern of placental involvement and fetal exposure to the virus.
In the series published by Zhu et al., the clinical status of 10 neonates were reported. None of the infants born to COVID-19 positive mothers demonstrated a positive throat swab PCR [27]. One infant, born at 34 weeks’ gestation, developed hemorrhagic shock, secondary to profound gastrointestinal bleeding and ultimately died secondary to multi end organ failure. A second infant, born at 34 week’s gestation, developed fever and gastrointestinal (GI) bleeding, but responded to supportive therapies. With negative testing, and without evaluation of placental tissue or amniotic fluid, it is difficult to draw any conclusions regarding the biologic nature of the GI symptoms or any putative relationship to COVID-19 exposure.
Clinical symptoms in neonates are non-specific and appear similar to the classic presentation of respiratory distress syndrome [40]. Additionally, radiologic imaging may demonstrate pneumonia, which is difficult to distinguish from premature lung disease. It is important to have a high degree of suspicion when managing neonates born to positive mothers, while recognizing more information is needed to truly develop a consistent neonatal phenotype.
5Strategies to modulate COVID-19 in the perinatal setting
There is currently no specific treatment for COVID-19 infection, nor access to an available vaccine. The American Academy of Pediatrics (AAP) and the Centers for Disease Control (CDC) have developed guidelines to aid in the care of infants born to COVID-19 positive mothers (Table 4). Recent guidelines prioritize universal maternal testing and recommend a shared decision-making process related to whether the infant should stay with the mother or be isolated. Treatment of patients with active SARS-CoV-2 illness has focused on the provision of supportive care according to illness severity. This may range from oxygen therapy and/or prone positioning to intubation, ICU support in patients with shock or adult respiratory distress syndrome (ARDS). Table 3 provides an overview of current strategies that have been implemented or are under investigation to either mitigate the risk of transmission or modulate the disease course.
Table 2
Neonatal COVID-19 reports published in the literature
| Author | Year | Location | Study design | Cases (n) | |
| Zhu et al. [27] | 2020 | China | Case Series | 10 | 6 COVID-19 positive cases were premature and 4 were full term. Neonates manifested shortness of breath, fever, thrombocytopenia and abnormal liver function as well as tachycardia, vomiting, pneumothorax. Pharyngeal swabs collected (n = 9) 1–9 days after birth were all negative. 5 neonates have been discharged, 1 has died, and 4 neonates remain in hospital in a stable condition. |
| Li et al. [51] | 2020 | China | Case Report | 1 | Infant born at 35 week’s gestation to COVID-19 positive mother by C/S for fetal HR 110 Multiple oropharyngeal swab specimens for SARS-CoV-2 negative. Oropharyngeal swab, blood, faeces, and urine samples remained negative (tested at 7 time-points) |
| Zeng et al. [36] | 2020 | China | Case series | 33 | 3 patients were found to be COVID-19 positive (nasopharyngeal and anal swab) on postnatal day 2 [possible vertical transmission]. All cases presented with lethargy and pneumonia; 2 of the 3 presented with fever. Symptoms were mild and none died. |
| Case 1 – born at 40 weeks’ gestation via C/S due to meconium-stained amniotic fluid and maternal COVID-19 pneumonia. Infant was admitted to NICU on day 2 with lethargy and fever. CXR showed pneumonia. Procalcitonin was elevated. Nasopharyngeal and anal swabs were positive for SARS-CoV-2 on days 2 and 4 but negative on day 6. | |||||
| Case 2 – born at 40 weeks’ gestation via C/S for maternal COVID. Infant presented with fever, lethargy, vomiting on postnatal day 2. Lab tests - leucocytosis/lymphocytopenia. CXR showed pneumonia. Nasopharyngeal/anal swabs were positive for SARS-CoV-2 on days 2, 4 but negative on day 6. | |||||
| Case 3 – born at 31 weeks’ gestation by C/S due to fetal distress and confirmed maternal COVID-19 pneumonia. Resuscitation was required. Apgar scores were 3, 4, and 5 at 1, 5, and 10 minutes. CXR revealed Respiratory Distress Syndrome and pneumonia. Symptoms resolved by postnatal day 14 after treatment with non-invasive ventilation, caffeine, and antibiotics. He also had sepsis, with an Enterobacter agglomerates–positive blood culture, leukocytosis, thrombocytopenia, and coagulopathy, which improved with antibiotic treatment. Nasopharyngeal and anal swabs were positive for SARS-CoV-2 on days 2, 4 but negative on day 7. | |||||
| Dong et al. [37] | 2020 | China | Case Report | 1 | Term infant delivered via C/S for maternal COVID. Infant showed elevated IgG and IgM to SARS-CoV-2 by 2 hours, elevated cytokines and WBC. Chest CT was normal. Infant tested negative on PCR nasopharyngeal swabs (n = 5) taken from 2 hours to day 16. [possible vertical transmission]. |
| Zeng et al. [39] | 2020 | China | Case Series | 6 | Infants born to COVID-19 positive mothers with IgG/IgM to SARS-CoV-2. Elevated IgG (n = 5) and IgM (n = 2) suggest possible vertical transmission as IgM does not typically cross the placenta. Quantitative RT-PCR for SARS-CoV-2 on neonatal serum and throat swabs were negative. |
| Chen et al. [26] | 2020 | China | Case Series | 9 | SARS-CoV-2 virus not detected in amniotic fluid, cord blood or breastmilk. No vertical transmission. No HIE or adverse neonatal outcomes. |
| Liu et al. [23] | 2020 | China | Case series | 11 | 10 infants born via C/S and 1 vaginally. No HIE or death. |
| Wang et al. [34] | 2020 | China | Case report | 1 | Infant born at 30 week’s gestation. Emergent C/S for decreased fetal movement and no fetal heart rate. Birth samples of amniotic fluid, placenta, umbilical cord blood, infant gastric juice and throat swabs were negative for SARS-CoV-2 [RT-PCR]. Repeat RT-PCR analyses (days 3, 7, 9) of neonatal throat swab and stool samples negative. |
| Yu et al. [29] | 2020 | China | Case series | 7 | Mean gestational age was 39 weeks (range 37-41 weeks). APGAR scores were normal.Four asymptomatic infants were not tested for SARS-CoV-2 and discharged. Three infants were admitted to the NICU and tested for SARS-CoV-2. Nucleic acid test of the throat swab of one neonate was positive at 36 h after birth (cord blood and placenta were negative). There was no fever and cough, but mild shortness of breath and CXR revealed mild pulmonary infection. The patient was later discharged home after testing negative. At 28 days after birth, the remaining three neonates were healthy and had no respiratory symptoms or fever. |
| Iqbal et al. [52] | 2020 | USA | Case report | 1 | Infant born at 39 week’s gestation to COVID-19 positive mother via spontaneous vaginal delivery. APGAR scores were normal. Infant showed no evidence of neonatal or intra-amniotic infection, was separated from the mother, fed formula or expressed breastmilk and was discharged on day 6. Phone follow revealed an asymptomatic infant. |
| Kamali et al. [53] | 2020 | Iran | Case Report | 1 | 5-day-old neonate was admitted with fever, lethargy, cutaneous mottling, and respiratory distress without cough. CXR was negative. Blood, urine and stool cultures were negative. Infant was treated with vancomycin, amikacin, and oseltamivir. Discharged home after 6 days. |
| Zeng et al. [54] | 2020 | China | Case report | 1 | 17-day-old ex-term neonate with one-week history of fever, sneezing, vomiting and diarrhea. CXR revealed pneumonia. Pharyngeal and anal swabs were positive for SARS-CoV-2 [RT-PCR]. Infant was discharged home. |
C/S, caesarean section; NICU, neonatal intensive care unit; CXR, chest radiograph; CT, computerized tomography; RT-PCR, Reverse transcription polymerase chain reaction.
Table 3
Therapeutic agents suggested as possible strategies for COVID-19
| Therapeutic agent | FDA approval | Mechanism | Investigational status |
| Remdesivir | No | Adenosine nucleotide analogue, inhibits viral replication through inducing early termination of RNA transcription [55] | Phase III Clinical trials |
| Chloroquine | No | Increases endosomal pH which interferes with the virus fusing to cells [55, 56] | Clinical trials, a RCT demonstrated shortening of the disease course [46] |
| Hydroxychloroquine Lopinavir | No | Antiretroviral protease inhibitor [55] | RCT, no benefit in clinical improvement demonstrated [57] |
RCT, randomized controlled trial; FDA, Food and Drug Administration.
Table 4
AAP and CDC guidelines for infant care following birth to COVID-19 positive mother
| AAP[58] | CDC[59] | |
| Delivery Room Precautions | Airborne, droplet and contact precautions should be worn when attending deliveries of COVID-19 positive mothers. | Pregnant women admitted with suspected COVID-19 or who develop symptoms concerning for suspected COVID-19 during admission should be prioritized for testing. Healthcare facilities should ensure recommended infection control practices for hospitalized pregnant patients who have confirmed COVID-19 or are PUIs are consistent with CDC recommendations for patients with confirmed Coronavirus disease or persons under investigation. |
| Separation | Pending available physical space, temporary separation of mother and newborn at birth to minimize risks of postnatal transmission is recommended. | Case by case consideration of separation factoring maternal illness and available physical space. |
| Breast Feeding | Mothers may express milk to feed to infant, no current evidence of viral presence in breast milk. | Mothers encouraged to express breastmilk to feed to the infant. In cases of direct breastfeeding, mothers should wear face masks and employ effective hand hygiene. |
| Newborn Testing | Pending testing availability, consider testing at 24 hours of infant life and repeat testing at 48 hours. Throat and NP swab recommended | No specific recommendations for neonates. For initial diagnostic testing for COVID-19, CDC recommends collecting and testing upper respiratory tract specimens (nasopharyngeal swab). CDC also recommends testing lower respiratory tract specimens, if available. A positive test result for the neonate may negate the reason to separate. |
| Newborn Discharge | Discharge of well newborns with negative testing based on center specific policies. Infants with positive testing but no symptoms may discharge home with close follow up across 14 days on a case by case basis. | Patients can be discharged from the healthcare facility whenever clinically indicated. No specific recommendations for infants born to COVID-19 positive mother. |
| Infant Visitation | COVID-19 positive mothers can visit infants after remaining afebrile for 72 hours, improvement of respiratory symptoms, and negative results from two consecutive NP swabs spaced 24 hours apart. | Considerations to discontinue temporary separation in the healthcare setting are the same as those to discontinue transmission-based precautions [Negative SARS-CoV2 test or symptom free At least 3 days (72 hours) have passed since recovery defined as resolution of fever without the use of fever-reducing medications and improvement in respiratory symptoms (e.g., cough, shortness of breath) and at least 7 days have passed since symptoms first appeared |
Abbreviations: American Academy of Pediatrics (AAP), Centers for Disease Control (CDC), Nasopharyngeal (NP).
Prevention has been used as a primary means to control the growth of COVID-19. As virus spread primarily relates to“person to person” interaction and has demonstrated familial clustering [41], limitation strategies that minimize human interaction, or social distancing, may help diminish transmission. It has been difficult to accurately determine the basic reproduction number (RO) of SARS-CoV-2, which is used to determine how quickly the virus can spread throughout a population [42], across various countries and cities with differing population densities. Currently studies indicate the virus has a RO of 2.2, meaning each infected individuals has the potential to spread the virus to 2.2 other individuals. Until the RO drops to less than 1 the outbreak is likely to continue with aggressive spread. Mitigation measures such as social distancing, school closures, quarantine of exposed individuals have been determined through simulation and observational studies to have the potential to reduce transmission of previous pandemics, although there is no published evidence of the efficacy of this strategy in the setting of the current pandemic. Vaccination research has started, with several academic centers and pharmaceutical companies having taken on the challenge, however the timeline needed to ensure patient safety remains considerable [43].
Modulation of the spread within the health care system is of vital importance. Wu et al. reported that 3.8% of their positive COVID-19 population in China were infected health care personnel [44]. The impact to health care workers in Italy is even greater; in some situations, hospitals have closed because of the magnitude of transmission among health care workers [45]. Protection of healthcare workers has been made especially challenging due to an insufficient global supply of personal protective equipment and disruptions within the supply chain. To help combat this shortage, given that pregnant mothers may be either be asymptomatic carriers or undiagnosed due to limited testing, the vague or mild nature of symptoms, andthe potential for comorbid disease states, it is incumbent on healthcare systems to ensure that neonatal teams have efficient processes to protect vulnerable front-line workers and mitigate both COVID-19 spread and access to healthcare delivery for other neonatal ICU patients.
An array of medications are in current use to treat patients with COVID-19. Anti-viral agents such as oseltamivir, ganciclovir, lopinavir/ritonavir and remdesivir have been used for critically ill patients. Remdesivir is a nucleotide analogue that interferes with viral replication. A clinical trial is currently in progress at multiple centers in Wuhan province. Additionally, the anti-malarial agent chloroquine phosphate has been used to treat pneumonia symptoms, as it possesses anti-viral and anti-inflammatory properties and has demonstrated positive clinical effect in the treatment of COVID-19 [46]. Clinical trials of the efficacy of chloroquine are also being conducted in China. Finally, lopinavir has demonstrated inhibition of protease activity in coronavirus species and is currently under investigation [47]. There are no reports of use of these agents in pregnant mothers or their infants.
Use of convalescent plasma, as a means of generating passive immunity, has been the subject of recent investigation. This therapeutic strategy was previously investigated during the SARS and MERS outbreaks; specifically, a metanalysis demonstrated a significant reduction in mortality compared to placebo [48]. In addition, clinical experience from five COVID-19 positive individuals in China with ARDS demonstrated clinical improvement following the receipt of convalescent plasma [49]. While this was not a randomized controlled study, the report is compelling and suggests biological and therapeutic plausibility. While many of the current treatments under investigation hold promise, due diligence and scientific rigor are essential to minimize the potential of unintended or unanticipated patient harm. There is currently no scientific evidence to justify any approach to treatment in pregnant mothers or in neonates. Centers should develop institutional guidelines based on best available evidence and resources available to them.
6Conclusion
The COVID-19 pandemic represents one of the greatest threats to human existence seen in the last one hundred years, with rapid spread across the globe and throughout the United States. The impact of rapid case escalation on the Italian and Spanish health care systems has forced the establishment of triaging committees to determine access to mechanical ventilation and intensive care resources. Although knowledge increases daily, it is incumbent on health care professionals, medical societies and governing bodies to establish comprehensive registries to track disease progression and enable an enhanced understanding of clinical presentation and risk profiles for those patients greatest at risk. These registries are even more relevant for obstetrics and neonatology where individual centers are likely to see low volumes of maternal cases, and even lower volumes of neonatal illness. COVID-19 does not appear to be particularly aggressive in pregnant women, neonates or children, based on the limited data that is available. Due to the dynamic nature and rapid evolution of our scientific understanding of this condition, these premises are likely to change as knowledge grows. The clinical experiences from countries (e.g. Italy, Spain, Iran) where COVID-19 has crippled ICU resources and preventative strategies from countries (e.g. South Korea, Singapore) where rapid dissemination has been limited will enable the development of standardized guidelines and frameworks for practice that may be universally adopted across the world. The ramifications of the pandemic to society have yet to be assessed, but are likely to be profound from personal, fiscal, and global health perspectives. The world beyond COVID-19 is likely to be a very different place.
Disclosure
Financial disclosure statement
No honorarium, grant, or other form of payment received to produce the manuscript.
Human research disclosure
No human research was involved in this manuscript
Animal research disclosure
No animal research was involved in this manuscript.
Acknowledgments
No additional acknowledgements outside of authorship.
References
[1] | Huang C , Wang Y , Li X , Ren L , Zhao J , Hu Y , et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. (2020) ;395: (10223):497–506. |
[2] | Sun P , Lu X , Xu C , Sun W , Pan B . Understanding of COVID-19 based on current evidence. J Med Virol. 2020. |
[3] | World Health Organization. WHO Director-General’s opening remarks at the media briefing on COVID-19 - 12 March 2020 Geneva2020 [updated March 12. Available from: https://www.who.int/dg/speeches/detail/who-director-general-s-opening-remarks-at-the-mission-briefing-on-covid-19—12-march-2020. |
[4] | Johns Hopkins University & Medicine. COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU) 2020 [updated 4/18.Available from: https://coronavirus.jhu.edu/map.html. |
[5] | Livingston E , Bucher K . Coronavirus Disease 2019 (COVID-19) in Italy. Jama. 2020. |
[6] | Zhou S , Wang Y , Zhu T , Xia L . CT Features of Coronavirus Disease 2019 (COVID-19) Pneumonia in 62 Patients in Wuhan, China. AJR American journal of roentgenology. 2020:1-8. |
[7] | Rothan HA , Byrareddy SN . The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. J Autoimmun. (2020) ;109: :102433. |
[8] | Baig AM , Khaleeq A , Ali U , Syeda H . Evidence of the COVID-19 virus targeting the CNS: Tissue distribution, host-virus interaction, and proposed neurotropic mechanisms. ACS Chem Neurosci. (2020) ;11: (7):995–8. |
[9] | Zheng YY , Ma YT , Zhang JY , Xie X . COVID-19 and the cardiovascular system. Nat Rev Cardiol. (2020) ;17: (5):259–60. |
[10] | Irigoyen N , Firth AE , Jones JD , Chung BYW , Siddell SG , Brierley I . High-resolution analysis of coronavirus gene expression by RNA sequencing and ribosome profiling. PLoS Pathog. (1005) ;12: (2):e1005473–e. |
[11] | Dong S , Sun J , Mao Z , Wang L , Lu YL , Li J . A guideline for homology modeling of the proteins from newly discovered betacoronavirus, 2019 novel coronavirus (2019-nCoV). J Med Virol. 2020. |
[12] | Xu H , Zhong L , Deng J , Peng J , Dan H , Zeng X , et al. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int J Oral Sci. (2020) ;12: (1):8. |
[13] | Nelson-Piercy C . Respiratory Diseases. Handbook of obstetric medicine. Boca Raton: CRC Press; 2015. p. 63-84. |
[14] | Thevarajan I , Nguyen THO , Koutsakos M , Druce J , Caly L , van de Sandt CE , et al. Breadth of concomitant immune responses prior to patient recovery: A case report of non-severe COVID-19. Nature Medicine. 2020. |
[15] | Littauer EQ , Esser ES , Antao OQ , Vassilieva EV , Compans RW , Skountzou I . H1N1 influenza virus infection results in adverse pregnancy outcomes by disrupting tissue-specific hormonal regulation. PLoS Pathog. (2017) ;13: (11):e1006757. |
[16] | van Doremalen N , Bushmaker T , Morris DH , Holbrook MG , Gamble A , Williamson BN , et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med. 2020. |
[17] | Remuzzi A , Remuzzi G . COVID-19 and Italy: What next? The Lancet. 2020. |
[18] | Shi H , Han X , Jiang N , Cao Y , Alwalid O , Gu J , et al. Radiological findings from 81 patients with COVID-19 pneumonia in Wuhan, China: A descriptive study. The Lancet Infectious diseases. (2020) ;20: (4):425–34. |
[19] | Ai T , Yang Z , Hou H , Zhan C , Chen C , Lv W , et al. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: A report of 1014 cases. Radiology. 2020:200642. |
[20] | Dashraath P , Jing Lin Jeslyn W , Mei Xian Karen L , Li Min L , Sarah L , Biswas A , et al. Coronavirus disease 2019 (COVID-19) pandemic and pregnancy. Am J Obstet Gynecol. 2020. |
[21] | Mullins E , Evans D , Viner RM , O’Brien P , Morris E . Coronavirus in pregnancy and delivery: Rapid review. Ultrasound Obstet Gynecol. 2020. |
[22] | Karimi-Zarchi M , Neamatzadeh H , Dastgheib SA , Abbasi H , Mirjalili SR , Behforouz A , et al. Vertical transmission of coronavirus disease 19 (COVID-19) from infected pregnant mothers to neonates:Areview. Fetal Pediatr Pathol. 2020:1-5. |
[23] | Liu D , Li L , Wu X , Zheng D , Wang J , Yang L , et al. Pregnancy and perinatal outcomes of women with coronavirus disease (COVID-19) pneumonia: A preliminary analysis. AJR Am J Roentgenol. 2020:1-6. |
[24] | Sutton D , Fuchs K , D’Alton M , Goffman D . Universal screening for SARS-CoV-2 inwomen admitted for delivery. N Engl J Med. 2020. |
[25] | Chen S , Liao E , Shao Y . Clinical analysis of pregnant women with 2019 novel coronavirus pneumonia. J Med Virol. 2020. |
[26] | Chen H , Guo J , Wang C , Luo F , Yu X , Zhang W , et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: A retrospective review of medical records. The Lancet. (2020) ;395: (10226):809–15. |
[27] | Zhu H , Wang L , Fang C , Peng S , Zhang L , Chang G , et al. Clinical analysis of 10 neonates born to mothers with 2019-nCoV pneumonia. Transl Pediatr. (2020) ;9: (1):51–60. |
[28] | Breslin N , Baptiste C , Miller R , Fuchs K , Goffman D , Gyamfi-Bannerman C , et al. COVID-19 in pregnancy: Early lessons. Am J Obstet Gynecol. 2020:100111. |
[29] | Yu N , Li W , Kang Q , Xiong Z , Wang S , Lin X , et al. Clinical features and obstetric and neonatal outcomes of pregnant patients with COVID-19 in Wuhan, China: A retrospective, single-centre, descriptive study. Lancet Infect Dis. 2020. |
[30] | Jiao J . Under the epidemic situation of COVID-19, should special attention to pregnant women be given? J Med Virol. 2020. |
[31] | Li AM , Ng PC . Severe acute respiratory syndrome (SARS) in neonates and children. Arch Dis Child Fetal Neonatal Ed. (2005) ;90: (6):F461–F5. |
[32] | Schwartz DA . An analysis of 38 pregnant women with COVID-19, their newborn infants, and maternal-fetal transmission of SARS-CoV-2: Maternal coronavirus infections and pregnancy outcomes. Arch Pathol Lab Med. 2020. |
[33] | Di Mascio D , Khalil A , Saccone G , Rizzo G , Buca D , Liberati M , et al. Outcome of coronavirus spectrum infections (SARS, MERS, COVID 1 -19) during pregnancy: A systematic review andmeta-analysis. AmJ Obstet Gynecol. 2020:100107. |
[34] | Wang S , Guo L , Chen L , Liu W , Cao Y , Zhang J , et al. A case report of neonatal COVID-19 infection in China. Clin Infect Dis. 2020. |
[35] | Levy A , Yagil Y , Bursztyn M , Barkalifa R , Scharf S , Yagil C . ACE2 expression and activity are enhanced during pregnancy. Am J Physiol Regul Integr Comp Physiol. (2008) ;295: (6):R1953–61. |
[36] | Zeng L , Xia S , Yuan W , Yan K , Xiao F , Shao J , et al. Neonatal early-onset infection With SARS-CoV-2 in 33 neonates born to mothers with COVID-19 in Wuhan, China. JAMA Pediatrics. 2020. |
[37] | Dong L , Tian J , He S , Zhu C , Wang J , Liu C , et al. Possible vertical transmission of SARS-CoV-2 from an infected mother to her newborn. Jama. 2020. |
[38] | Palmeira P , Quinello C , Silveira-Lessa AL , Zago CA , Carneiro-Sampaio M . IgG placental transfer in healthy and pathological pregnancies. Clin Dev Immunol. (2012) ;2012: :985646. |
[39] | Zeng H , Xu C , Fan J , Tang Y , Deng Q , Zhang W , et al. Antibodies in infants born to mothers with COVID-19 pneumonia. JAMA. 2020. |
[40] | Wang L , Shi Y , Xiao T , Fu J , Feng X , Mu D , et al. Chinese expert consensus on the perinatal and neonatal management for the prevention and control of the 2019 novel coronavirus infection (First edition). Ann Transl Med. (2020) ;8: (3):47. |
[41] | Chan JF-W , Yuan S , Kok K-H , To KK-W , Chu H , Yang J , et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. The Lancet. (2020) ;395: (10223):514–23. |
[42] | Prem K , Liu Y , Russell TW , Kucharski AJ , Eggo RM , Davies N , et al. The effect of control strategies to reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China: A modelling study. Lancet Public Health. 2020. |
[43] | Prompetchara E , Ketloy C , Palaga T . Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol. (2020) ;38: (1):1–9. |
[44] | Wu Z , McGoogan JM . Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020. |
[45] | Anelli F , Leoni G , Monaco R , Nume C , Rossi RC , Marinoni G , et al. Italian doctors call for protecting healthcare workers and boosting community surveillance during covid-19 outbreak. BMJ (Clinical research ed). (2020) ;368: :m1254. |
[46] | Gao J , Tian Z , Yang X . Breakthrough: Chloroquine phosphate has shown apparent efficacy in treatment of COVID-19 associated pneumonia in clinical studies. Biosci Trends. (2020) ;14: (1):72–3. |
[47] | Yao TT , Qian JD , Zhu WY , Wang Y , Wang GQ . A systematic review of lopinavir therapy for SARS coronavirus and MERS coronavirus-A possible reference for coronavirus disease-19 treatment option. J Med Virol. 2020. |
[48] | Mair-Jenkins J , Saavedra-Campos M , Baillie JK , Cleary P , Khaw F-M , Lim WS , et al. The effectiveness of convalescent plasma and hyperimmune immunoglobulin for the treatment of severe acute respiratory infections of viral etiology: A systematic review and exploratory meta-analysis. J Infect Dis. (2015) ;211: (1):80–90. |
[49] | Shen C , Wang Z , Zhao F , Yang Y , Li J , Yuan J , et al. Treatment of 5 critically ill patients with COVID-19 with convalescent plasma. JAMA. 2020. |
[50] | Zhang L , Jiang Y , Wei M , Cheng BH , Zhou XC , Li J , et al. [Analysis of the pregnancy outcomes in pregnant women with COVID-19 in Hubei Province]. Zhonghua Fu Chan Ke Za Zhi. (2020) ;55: (0):E009. |
[51] | Li Y , Zhao R , Zheng S , Chen X , Wang J , Sheng X , et al. Lack of vertical transmission of severe acute respiratory syndrome coronavirus 2, China. Emerg Infect Dis. (2020) ;26: (6). |
[52] | Iqbal SN , Overcash R , Mokhtari N , Saeed H , Gold S , Auguste T , et al. An Uncomplicated delivery in a patient with Covid-19 in the United States. N Engl J Med. 2020. |
[53] | Kamali Aghdam M , Jafari N , Eftekhari K . Novel coronavirus in a 15-day-old neonate with clinical signs of sepsis, a case report. Infectdis. (London, England). 2020:1-3. |
[54] | Zeng LK , Tao XW , Yuan WH , Wang J , Liu X , Liu ZS . [First case of neonate infected with novel coronavirus pneumonia in China]. Zhonghua er ke za zhi=Chinese journal of pediatrics. (2020) ;58: (0):E009. |
[55] | Lu CC , Chen MY , Chang YL . Potential therapeutic agents against COVID-19: What we know so far. J Chin Med Assoc: JCMA. 2020. |
[56] | Mauthe M , Orhon I , Rocchi C , Zhou X , Luhr M , Hijlkema K-J , et al. Chloroquine inhibits autophagic flux by decreasing autophagosome-lysosome fusion. Autophagy. (2018) ;14: (8):1435–55. |
[57] | Cao B , Wang Y , Wen D , Liu W , Wang J , Fan G , et al.. A Trial of lopinavir–ritonavir in adults hospitalized with severe Covid-19. N Engl J Med. 2020. |
[58] | Karen M . Kimberlin, James Cummings. AAP News. AAp Publications [Internet]. 2020 April 02. Available from: https://www.aappublications.org/news/2020/04/02/infantcovidguidance040220?cct=2287. |
[59] | Centers for Disease Control and Prevention. Considerations for inpatient obstetric healthcare settings: U.S. Department of Health and Human Services; 2020 [updated April 4. Available from: https://www.cdc.gov/coronavirus/2019-ncov/hcp/inpatient-obstetric-healthcare-guidance.html#f1 |




