Effect of concentration, method of auxin application and cultivation conditions on in vitro rooting of bog blueberry (Vaccinium uliginosum L.)
Abstract
BACKGROUND:
Bog blueberry, Vaccinium uliginosum is one of the newer fruit crops. Choosing the most appropriate methods for mass vegetative propagation of V. uliginosum cultivars, including in vitro propagation, is crucial.
OBJECTIVE:
We investigated the effect of growth regulators and light on in vitro rhizogenesis of V. uliginosum to determine the optimal method of propagation.
METHODS:
The following cultivation techniques were tested on microshoots of bog blueberry varieties ‘Golubaya rossyp’, ‘Nektarnaya’, ‘Shegarskaya’ and ‘Iksinskaya’: 1) ½ Anderson (A) medium supplemented with 3 and 10 μM indole-3-butyric acid (IBA) or indole-3-acetic acid (IAA) and 2) 24-h aqueous treatments of microshoots with 148 μM IBA or 171 μM IAA and further cultivation in ½ A medium. In addition, we tested precultivation in darkness for 3 d versus a 16/8-h photoperiod.
RESULTS:
100% rooting was observed in ‘Golubaya rossyp’ in all treatments of the experiments and with ‘Nektarnaya’ on the growth-regulator-free ½ A medium and after treating the microshoots with 171 μM IAA solution. Precultivation in darkness produced a positive effect on rooting in ‘Shegarskaya’ and ‘Iksinskaya.’
CONCLUSIONS:
Response to in vitro rooting techniques varied between cultivars of V. uliginosum. Treatment of microshoots with auxin solutions improved in vitro rooting.
1Introduction
Blueberry (genus Vaccinium L., family Ericaceae Juss.) is one of the world’s leading fruit crops. The genus includes approximately 500 species [1]. The most widespread blueberry crops are highbush blueberry (V. corymbosum L.), rabbiteye blueberry (V. virgatum A.), lowbush blueberry (V. angustifolium A.) and half-high blueberry (V. corymbosum×V. angustifolium). Russian and foreign researchers have investigated techniques to propagate the bog blueberry, V. uliginosum L. since the 1970s [2]. Development of effective methods of mass vegetative propagation of new forms and cultivars of blueberries, including tissue culture propagation, is crucial to their introduction as crop species.
The success of in vitro technologies depends on the stage of microshoot rooting at which they are applied. Root regeneration varies between cultivars and species and depends on the sensitivity of plants to rooting factors and the rooting techniques used. The process of in vitro organ regeneration is accompanied by cell differentiation and initiation of new meristematic centers [3]. Some of the exogenous and endogenous factors that regulate root formation in intact plants (e.g., chemicals, light, temperature, mycorrhization) can be used to control rhizogenesis in vitro [4, 5].
Among blueberry species, V. corymbosum and V. virgatum root well (76%) on propagation medium [6], and V. macrocarpon roots on medium without growth regulators [7], without additional stimulation of rhizogenesis. However, most plant species require specific chemicals (growth regulators, mineral components, etc.) for the initiation of cell differentiation and root meristem formation. Plant response to chemical growth regulators, particularly auxins, depends on the ability of the plant to absorb and metabolize these compounds. Furthermore, the structure of auxin receptors differs between plants [8]. Some studies have found supplementation of the culture medium with auxins to improve rooting in blueberries [9–12]. The ratio of auxins and cytokinins also affects root formation. Frett and Smagula [13] found that the transfer of highbush blueberry microshoots to a medium with a low level of cytokinin (2-isopentenyl adenine) improved rooting success.
E x vitro rooting of blueberry microshoots is effective [11, 14] and can reduce the cost of planting material [15]. In addition, rooting under non-sterile conditions contributes to the better survival of plants. For these reasons, rooting of V. corymbosum was performed by immersing shoot bases in IBA solution with subsequent placement into chopped sphagnum in growth chambers under periodic fog conditions [16]. Lirene [17] was the first to use tissue culture for the propagation of blueberry V. ashei R. (=V. virgatum A.) and to successfully root the microshoots in perlite in a growth chamber without the use of growth regulators. Meiners [11] found that microshoots of V. corymbosum and V. vitis-idaea L. successfully rooted both on media containing auxins and ex vitro without growth regulators.
Physical factors including light intensity and spectral composition directly affect organogenesis, and rhizogenesis in particular, contributing to cell differentiation and initiation of new meristematic centers [4, 18–20]. These factors also have an indirect effect on organogenesis, causing photodegradation of growth regulators in the culture medium [21, 22].
Our study aimed to investigate the effect of growth regulators and light on in vitro rhizogenesis of V. uliginosum, to contribute to development of this blueberry species as a commercial crop.
2Materials and methods
We studied four cultivars of V. uliginosum from the Central Siberian Botanical Garden of the Siberian Branch Russian Academy of Sciences (Novosibirsk, Russia): ‘Golubaya rossyp,’ ‘Nektarnaya,’ ‘Shegarskaya,’ and ‘Iksinskaya.’ Axillary buds were used as starting material for in vitro culture. The main stages of micropropagation, including surface sterilization of axillary buds and shoot multiplication, were performed using our previously published methods [23, 24].
Meristematic apices isolated from the buds were placed on Anderson’s (A) medium [25] supplemented with 5 μM 2-isopentenyl adenine (2-iP) (Sigma-Aldrich, St Louis, Missouri, USA), 200 mg l–1 casein hydrolyzate (Sigma-Aldrich, St Louis, Missouri, USA), 3% sucrose and 0,6% agar (Sigma-Aldrich, St Louis, Missouri, USA). Micropropagation was performed on A media supplemented with 5 μM 2-iP, 200 mg l–1 glutamic acid (Sigma-Aldrich, St Louis, Missouri, USA), 3% sucrose and 0,6% agar. Shoots were cultivated at 4-week intervals over 3 years. Prior to rooting, the microshoots were passaged through two subcultures on growth-regulator-free A medium with 2-fold reduced content of macro- and microelements (½ A medium) for 8 weeks. We used the following cultivation technique: ½ A medium supplemented with indole-3-butyric acid (IBA) (Sigma-Aldrich, St Louis, Missouri, USA) or indole-3-acetic acid (IAA) (Sigma-Aldrich, St Louis, Missouri, USA) at 3 or 10 μM; treatment of microshoots with aqueous solution of 148 μM IBA or 171 μM IAA for 1 d, and further cultivation on ½ A medium (according to Vysotsky [26] with modifications). The pH of the medium was adjusted to 5.7 before autoclaving at 121°C and 1.1 kg cm–2 for 20 min. Auxins were added as sterile solutions to the culture media, autoclaved and cooled to 60–65°C. In addition, precultivation was carried out for all treatments either in darkness for 3 d, followed by a 16/8-hour light/dark photoperiod or a continuous 16/8-h photoperiod (light/dark). Cultures were incubated at 24±1°C, with lighting of 54 μmol m–2 s–1. As a control, we used growth-regulator-free ½ A medium, cultivated either using the photoperiod or precultivated in darkness for 3 d. Microshoots of 10 mm length were used for rooting. Measurements were replicated twice using 100 microshoots per experiment after 40 d cultivation.
Statistical analysis was carried out in Microsoft Excel 7.0 and Statistica 6.0 (LSD-test, ANOVA). Data are presented as mean values with confidence intervals (p≤0.05): X±t×Sx, where
X is sample mean
t is Student’s criterion
Sx is sample error.
3Results
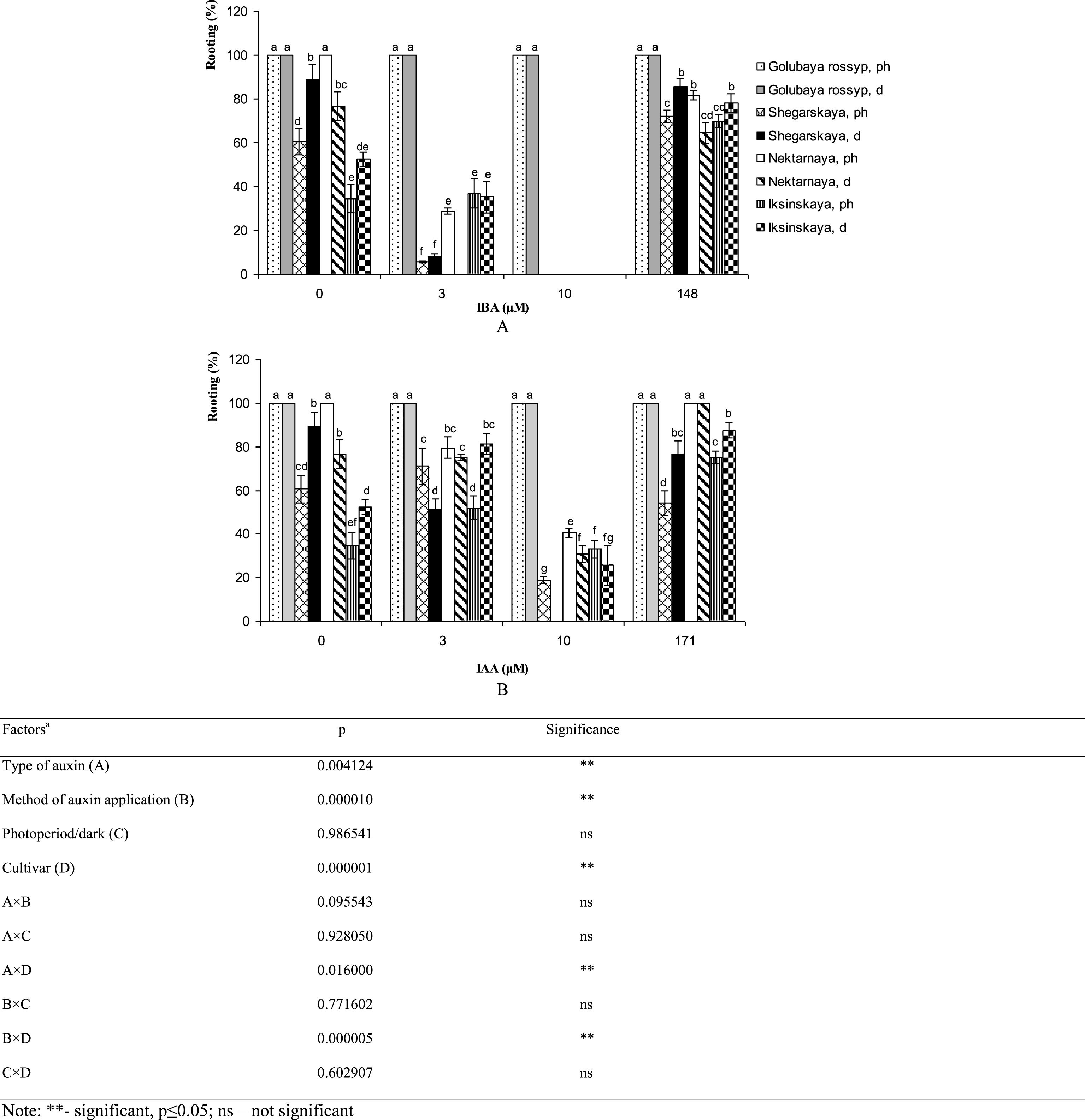
Effectiveness of techniques used for rooting microshoots varied between cultivars of V. uliginosum (Fig. 1). The highest rooting percentage (100%) among treatments was observed in ‘Golubaya rossyp’ on growth-regulator-free ½ A medium, as well as in ‘Nektarnaya’ after treating microshoots with 171 μM IAA solution. The number of rooted plants of varietals ‘Shegarskaya’, ‘Nectarnaya’ and ‘Iksinskaya’ was reduced with IBA supplementation of the culture medium. IAA addition to the medium at 10 μM concentration inhibited rhizogenesis as well. Low concentrations of IAA increased rhizogenesis in plants exposed to photoperiod from 60.5% to 71.0% in ‘Shegarskaya,’ from 34.5% to 52.0% in ‘Iksinskaya,’ and from 52.4% to 81.4% in ‘Iksinskaya’ in precultivation and under conditions of darkness. It should be noted that precultivation of these cultivars in darkness on growth-regulator-free medium and after treating the microshoots with IAA or IBA yielded better results for rooting microshoots, with rhizogenesis increased by 1.5–2.3 times. The cultivar ‘Nektarnaya’ showed good rooting on the growth-regulator-free media (100%), on media with low IAA content (79.5%) and after auxin treatment (100%) under photoperiod conditions. Better rooting was observed during continuous cultivation under photoperiod conditions in all experimental treatments.
Fig.1
In vitro rooting percentage (%) of cultivars of V. uliginosum grown with different concentrations and types of auxin, and under varying light regimes. A – IBA; B – IAA. “Ph” microshoots were grown in photoperiod; “d” microshoots in darkness. Abscissa show the concentration of auxin in ½ A culture medium (0, 3, and 10) and the concentration in a 24-hour treatment with further cultivation of microshoots on ½ A medium without plant growth regulators (148 and 171). Bars represent 95% confidence intervals. The influence of factors (a) was evaluated by ANOVA; ** – significant at p≤0.05; ns – not significant. Means followed by the same letter are not significantly different according to the LSD at p≤0.05.
The type and concentration of auxins during the final rooting stage affected the development of the root system and aerial parts of V. uliginosum (Table 1). Our findings indicate that high IBA concentrations (10 μM) completely inhibit the development of the root system in ‘Shegarskaya,’ ‘Nektarnaya’ and ‘Iksinskaya’, and a similar concentration of IAA greatly reduces rhizogenesis (by 2–3 times) in comparison with the control. The maximum number of roots, 7.6±1.3 pcs./shoot, was observed in ‘Golubaya rossyp’ after treating the microshoots in IBA solution (Figs. 2, 3). The treatment of microshoots with auxin solutions was observed to enhance initiation and development of roots across the shoot surface, and axillary bud development was often seen only after the formation of a well-developed root system (Fig. 3D, E). In general both, the application of auxins to the culture medium, and auxin treatment, stimulated root initiation and development in ‘Golubaya rossyp’ under all experimental conditions.
Table 1
Effect of cultivar features and the method of application and type of auxin on the growth and development of V. uliginosum in vitro culture (ANOVA)
| Parameters | Shoot length | Number of leaves | Number of roots | Root length |
| Method of auxin | ** | ** | ** | ** |
| application (A) | p = 0.000055 | p = 0.000001 | p = 0.001605 | p = 0.000001 |
| Type of auxin (B) | ** | ns | ns | ns |
| p = 0.071742 | p = 0.585871 | p = 0.685704 | p = 0.034096 | |
| Cultivar (C) | ** | ** | ** | ** |
| p = 0.000001 | p = 0.000001 | p = 0.000001 | p = 0.000001 | |
| A×B | ns | ** | ns | ns |
| p = 0.780207 | p = 0.008265 | p = 0.307671 | p = 0.818145 | |
| A×C | ns | ** | ns | ** |
| p = 0.181643 | p = 0.000066 | p = 0.604529 | p = 0.017171 | |
| B×C | ** | ** | ** | ** |
| p = 0.018658 | p = 0.000001 | p = 0.000001 | p = 0.000001 | |
| A×B×C | ns | ns | ** | ns |
| p = 0.534724 | p = 0.085983 | p = 0.042482 | p = 0.164238 |
Note: **- significant at p≤0.05; ns – not significant.
Fig.2
The number of roots (no. of roots) on microshoots of cultivars of V. uliginosum grown with different concentrations and types of auxin, and under varying light regimes. A – IBA; B – IAA. “Ph” microshoots were grown in photoperiod; “d” microshoots in darkness. Abscissa show the concentration of auxin in ½ A culture medium (0, 3, and 10) and the concentration in a 24-hour treatment with further cultivation of microshoots on ½ A medium without plant growth regulators (148 and 171). Bars represent 95% confidence intervals. The influence of factors (a) was evaluated by ANOVA; ** – significant at p≤0.05; ns – not significant. Means followed by the same letter are not significantly different according to the LSD at p≤0.05.
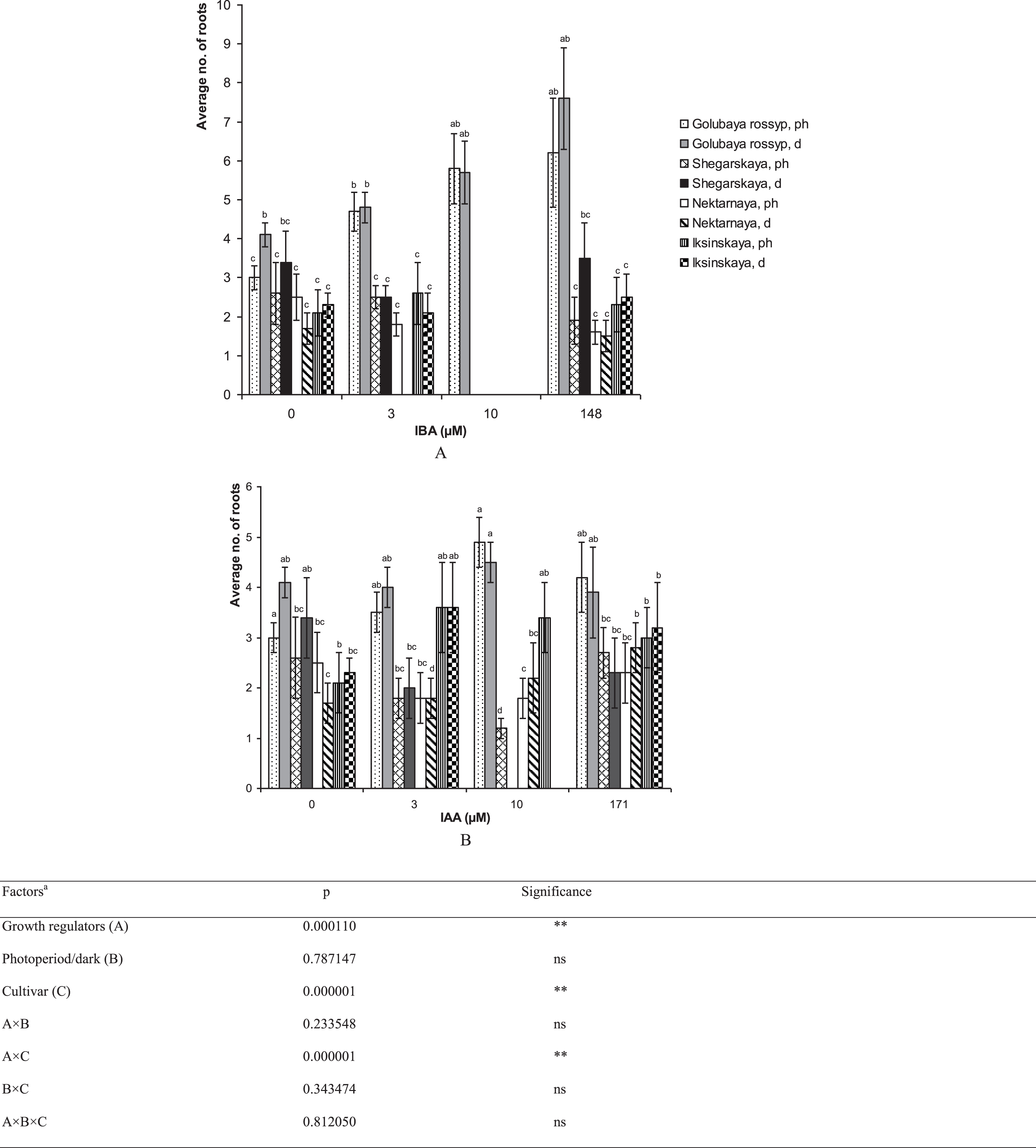
Fig.3
In vitro rooted shoots of V. uliginosum ‘Golubaya rossyp:’ A – control, without growth regulators, 40 d; B – ½ A medium supplemented with 3 μM IBA, 40 d; C – root formation on microshoots after treatment with 148 μM IBA, 30 d; D- shoot formation after treatment with 148 μM IBA, 40 d. Bar: 1 cm.

For the cultivar ‘Iksinskaya,’ cultivation on a medium supplemented with 3 μM IAA had a stimulating effect on the number of formed roots (from 2.1 to 3.6 pcs./shoot). For ‘Shegarskaya’ and ‘Nektarnaya,’ treatment with auxin reduced rhizogenesis or did not affect it (Fig. 2B).
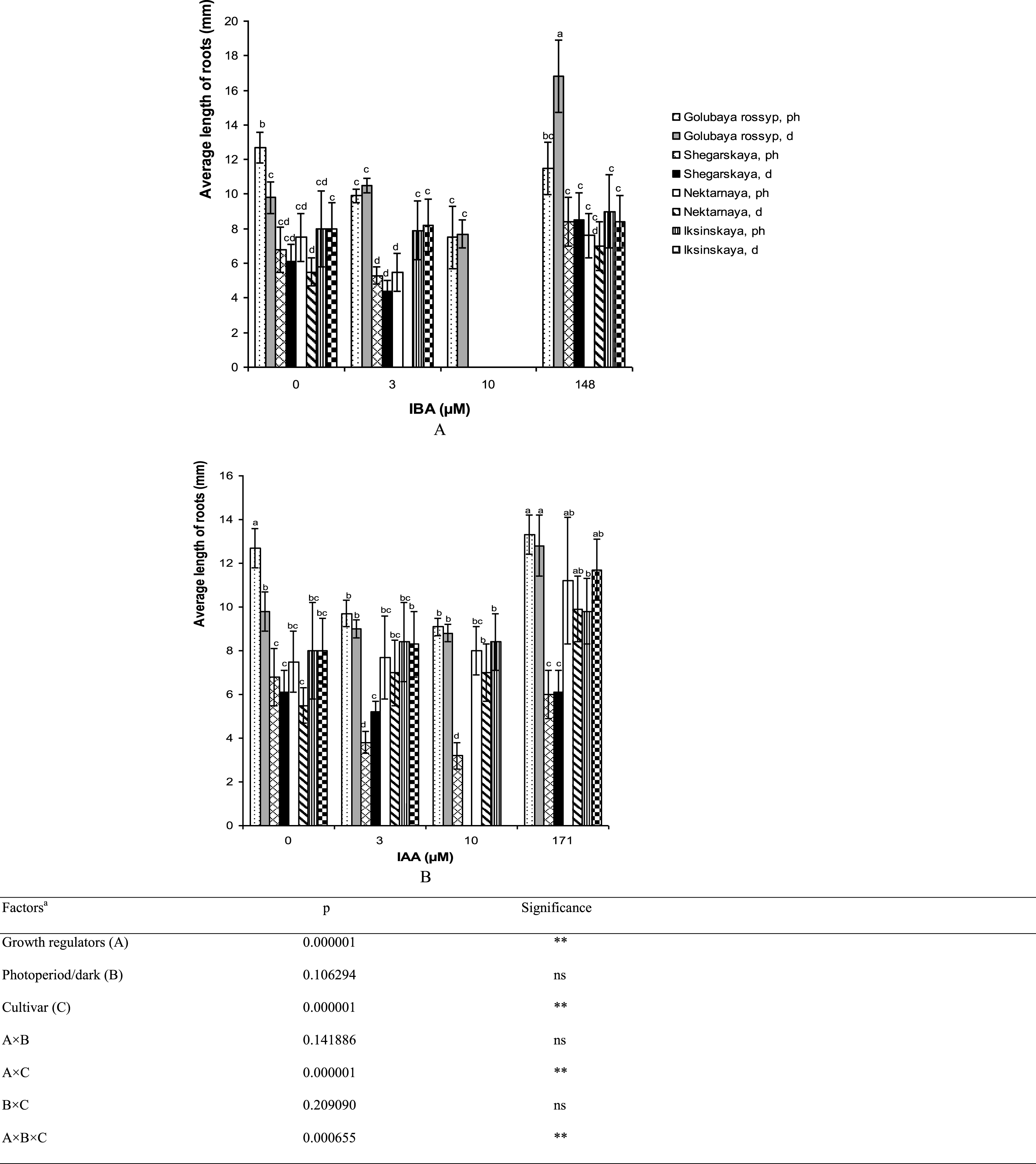
The longest average root length was observed in ‘Golubaya rossyp’ on the growth-regulator-free medium after the treatment of microshoots with IBA solution and precultivation in darkness (16.8±2.1 mm). This technique also had a positive effect on the growth of roots of the other cultivars under study (Fig. 4). The addition of 3 or 10 μM IBA to the culture medium reduced or did not affect root length in all cultivars of V. uliginosum studied (Fig. 4A). The treatment of microshoots in 171 μM IAA solution led to root elongation in ‘Iksinskaya’ from 8.0 to 9.8 mm when cultivated in the photoperiod, and up to 11.7 mm during precultivation in darkness. The same tendency was observed for ‘Nektarnaya’ (from 7.5 to 11.2 mm when cultivated in the photoperiod, from 5.5 to 9.9 mm when precultivated in darkness, Fig. 4B). In general, the addition of IAA to the culture medium resulted in reduced root length in ‘Golubaya rossyp’ and ‘Shegarskaya,’ however it had no effect on ‘Nektarnaya’ and ‘Iksinskaya.’
Fig.4
Average length of roots (mm) on microshoots of cultivars of V. uliginosum grown with different concentrations and types of auxin, and under varying light regimes. A – IBA; B – IAA. “Ph” microshoots were grown in photoperiod; “d” microshoots in darkness. Abscissa show the concentration of auxin in ½ A culture medium (0, 3, and 10) and the concentration in a 24-hour treatment with further cultivation of microshoots on ½ A medium without plant growth regulators (148 and 171). Bars represent 95% confidence intervals. The influence of factors (a) was evaluated by ANOVA; ** – significant at p≤0.05; ns – not significant. Means followed by the same letter are not significantly different according to the LSD at p≤0.05.
Varietal differences in microshoot height and the number of leaves per stem were observed on growth-regulator-free ½ A medium (Figs. 5, 6). The cultivar ‘Iksinskaya’ exhibited the shortest shoots (28.9±1.9 mm), and ‘Golubaya rossyp’ the highest shoots (53.2±6.7 mm). As for the number of leaves per stem, two groups can be distinguished: ‘Shegarskaya’ and ‘Iksinskaya’ (from 6.7 to 9.0 pcs.), and ‘Golubaya rossyp’ and ‘Nektarnaya’ (from 8.6 to 10.3 pcs.). We observed reliable differences in development of the aerial part of the bog blueberry dependent on auxin type and rooting technique (Table 1).
Fig.5
Average height of shoots (mm) of cultivars of V. uliginosum grown with different concentrations and types of auxin, and under varying light regimes. A – IBA; B – IAA. “Ph” microshoots were grown in photoperiod; “d” microshoots in darkness. Abscissa show the concentration of auxin in ½ A culture medium (0, 3, and 10) and the concentration in a 24-hour treatment with further cultivation of microshoots on ½ A medium without plant growth regulators (148 and 171). Bars represent 95% confidence intervals. The influence of factors (a) was evaluated by ANOVA; ** – significant at p≤0.05; ns – not significant. Means followed by the same letter are not significantly different according to the LSD at p≤0.05.
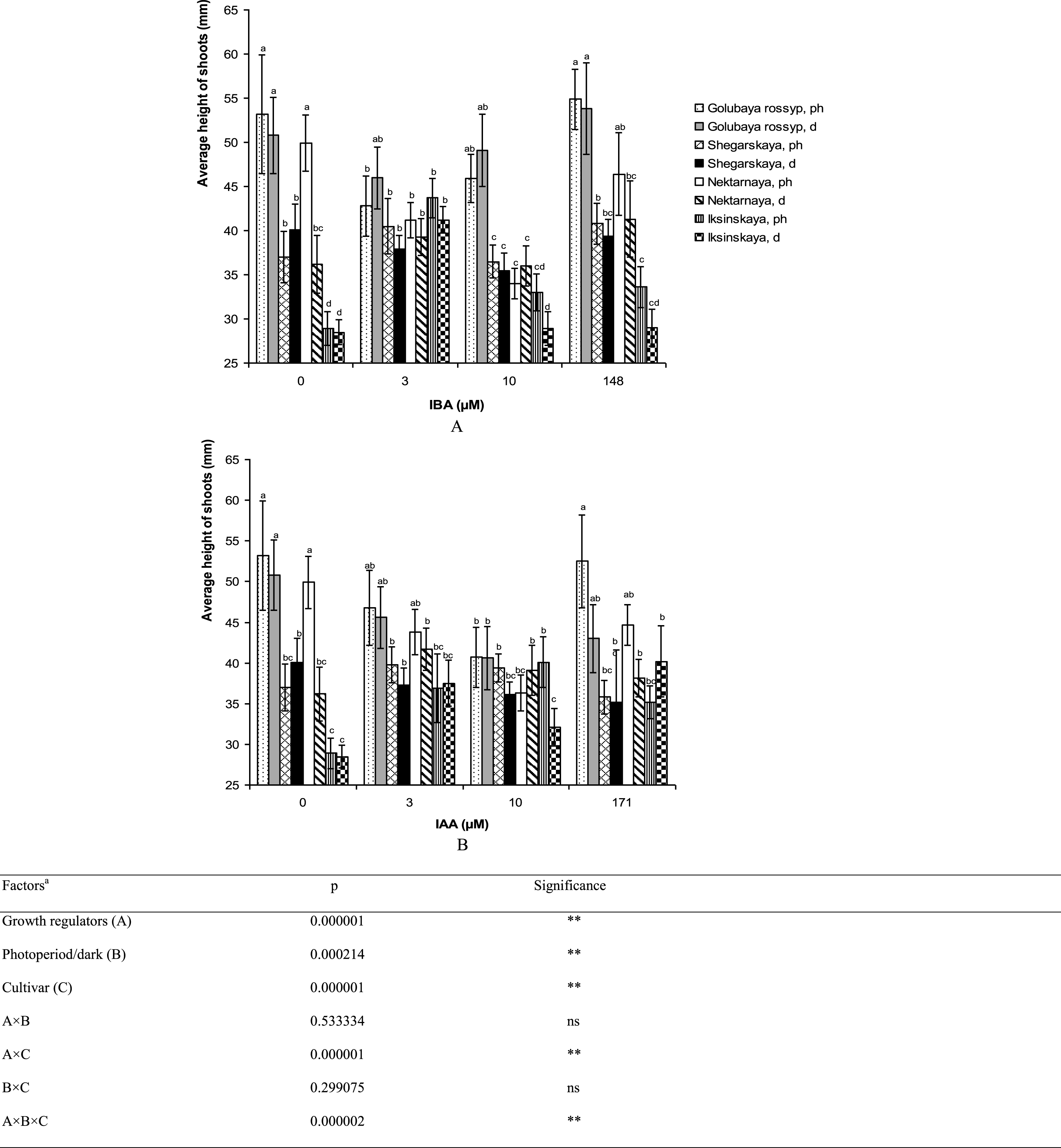
Fig.6
The number of leaves (no. of leaves) on microshoots of cultivars of V. uliginosum grown with different concentrations and types of auxin, and under varying light regimes. A – IBA; B – IAA. “Ph” microshoots were grown in photoperiod; “d” microshoots in darkness. Abscissa show the concentration of auxin in ½ A culture medium (0, 3, and 10) and the concentration in a 24-hour treatment with further cultivation of microshoots on ½ A medium without plant growth regulators (148 and 171). Bars represent 95% confidence intervals. The influence of factors (a) was evaluated by ANOVA; ** – significant at p≤0.05; ns – not significant. Means followed by the same letter are not significantly different according to the LSD at p≤0.05.
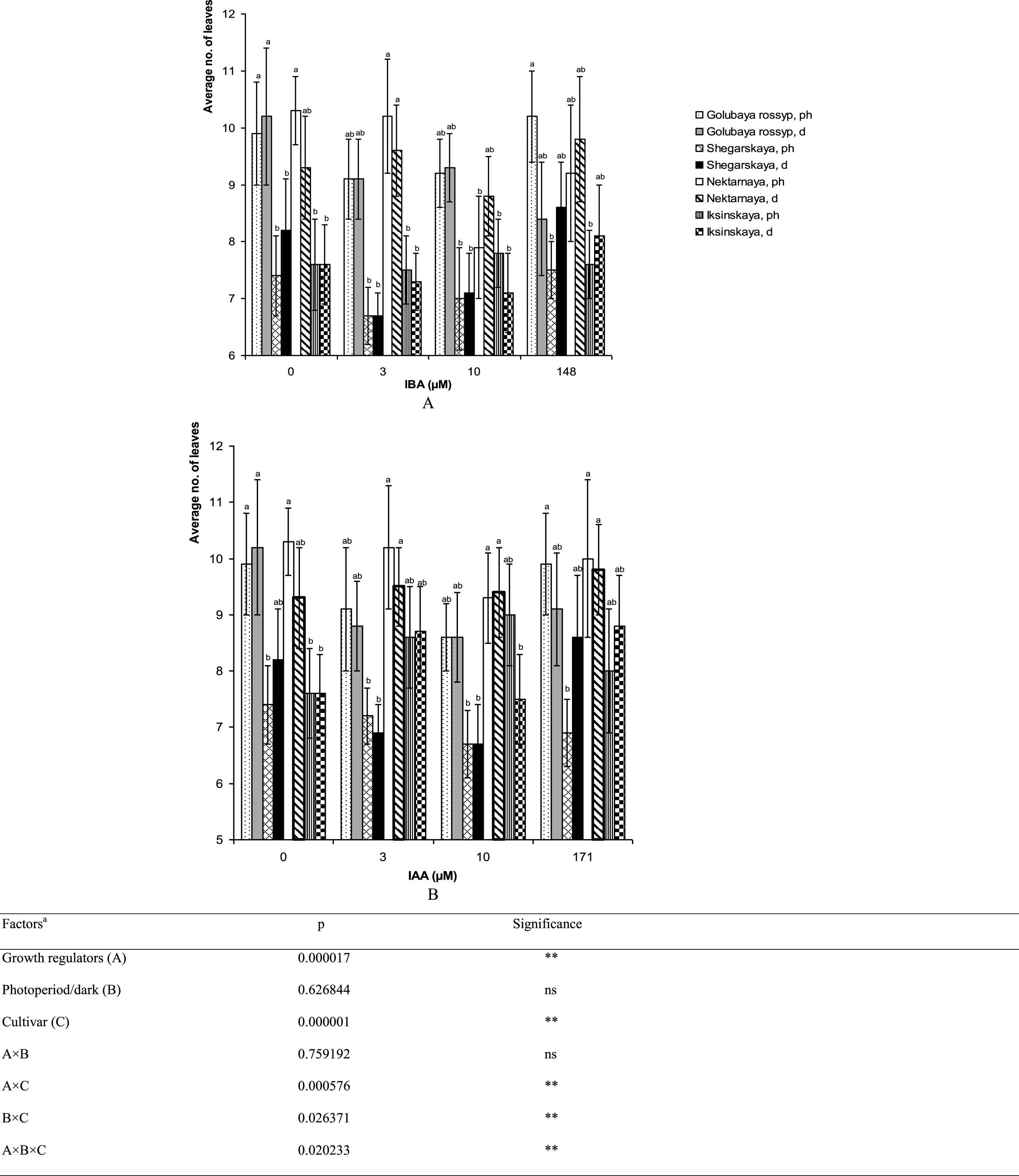
The addition of auxins to the culture media reduced the shoot height in all cultivars except ‘Iksinskaya.’ The height of shoots of this cultivar was increased by 1.5 times (up to 43.7 mm) on the medium with 3 μM IBA cultivated under photoperiod conditions, and it grew 1.4 times higher (up to 40.1 mm) on the medium containing 10 μM IAA under photoperiod conditions (Fig. 5). Reliable differences in shoot height during cultivation under photoperiod conditions and precultivation in darkness were observed for ‘Nektarnaya’ only. On ½ A medium under photoperiod conditions, the shoot height of this cultivar was significantly higher (49.9 mm under photoperiod conditions and 36.2 mm when precultivated in darkness, Fig. 5A).
4Discussion
Vegetative reproduction of many new cultivars and forms is constrained by weak rooting of cuttings, or complexity of rooting techniques. In bog blueberry, air layerage is most effective, providing 80–100% rooting. A recently developed green grafting technique [27] provides 55.0% and 64.5% rooting with good root branching with or without growth regulators (120 mg l–1 IBA), respectively [2]. In vitro provides ideal conditions to study factors influencing rhizogenesis, since experiments can be carried out under strictly controlled conditions of humidity, temperature and lighting [28].
We found the optimal technique of in vitro rooting of V. uliginosum to be the treatment of microshoots with auxin solution for 24 h, followed by transfer to growth-regulator-free media. This technique was proposed by Vysotsky [26] and successfully tested by other researchers [29, 30] for members of the genus Rhododendron L. According to our data, treatment with IBA or IAA solutions increased the percentage of bog blueberry rooting and resulted in a more developed root system. Previous research indicates that IBA is preferable for this technique, since IAA is rapidly oxidized in explant tissues [31], and hence, high concentrations of auxin are necessary. However, in our study, IBA and IAA at the same concentration resulted in similar rooting percentages of V. uliginosum cultivars (Fig. 1).
The effectiveness of auxins depends on their transport, metabolism, and affinity for auxin receptors in plants. Nissen and Sutter [21] reported that photooxidation of IAA in vitro culture occurs rapidly (50% within 24 h), but that of IBA proceeds slowly (10%). Thus, the higher effectiveness of IAA may be associated with auxin destruction after transferring crops from the darkness to the light, that is, after the initiation of root primordia and before the inhibition of root development by auxin in the culture medium. However, the range of effects observed with use of auxins may be due to another cause. As observed with other growth regulators, low auxin concentrations enhance rhizogenesis, and high (superoptimal) auxin concentrations inhibit the development of initiated root primordia [32, 33]. We found that high concentrations of auxin in the culture media (10 μM) inhibit the development of the root system in ‘Nektarnaya’, ‘Shegarskaya’ and ‘Iksinskaya’ cultivars. Low concentrations of auxins exhibited a stimulating effect on rhizogenesis in ‘Iksinskaya.’ Thus, genotypic features of the cultivars, their endogenous hormonal balance and physiological age must be crucial for the development of adventitious roots of the studied cultivars. It is known that levels of endogenous auxins in plants of different ages can result in different shoot rooting percentages under similar experimental conditions [34].
Our study did not find an effect of clear light on the rooting percentage of the studied cultivars of V. uliginosum. However, the photodegradation of IAA and IBA added to the culture medium may account for the improved root formation observed under conditions of darkness as compared to conventional lighting [22]. According to our data, ‘Shegarskaya’ and ‘Iksinskaya’ showed a high percentage of shoot rooting during precultivation in darkness after treatment with IBA (85.7% for ‘Shegarskaya’) and IAA (87.5% for ‘Iksinskaya’). Regardless of the type of exogenous auxin, darkness can also affect the different phases of auxin metabolism and rhizogenesis, by changing peroxidase activity as well as the activity of endogenous phenolic compounds [35]. In addition, light affects rhizogenesis through the phytochrome system. The participation of the phytochrome system in plant morphogenesis is well known [36], and this approach is proposed for the light induction of rhizogenesis by a number of authors [37, 38]. The effect of light on rhizogenesis induction has been found to vary in different plant species and cultivars [19]. Our studies revealed that ‘Shegarskaya’ and ‘Iksinskaya’ cultivated in growth-regulator-free media rooted better during precultivation in darkness; ‘Golubaya rossyp’ exhibited 100% rooting under all cultivation regimes, and ‘Nektarnaya’ showed better rooting when grown in a continuous photoperiod.
5Conclusions
Plant genotype was shown to have a primary effect on rooting of the cultivars of V. uliginosum in vitro culture. Rooting, development of the root system and aerial part were most successful in ‘Golubaya rossyp.’ The treatment of microshoots with auxin solutions was effective for all studied cultivars, and ‘Golubaya rossyp’ exhibited the best root system development (7.6 roots, each root 16.8 mm in length) after treatment with 148 μM IBA. Method of auxin application had a greater effect than the type of the auxin used. The addition of auxins to the culture medium decreased rhizogenesis for ‘Shegarskaya’ and ‘Nektarnaya,’ and 3 μM IAA used for ‘Iksinskaya’ increased the percentage of rhizogenesis up to 81.4%. Precultivation of microshoots in darkness had a positive effect on rooting percentage in ‘Shegarskaya’ and ‘Iksinskaya.’ The effect of light on rhizogenesis induction varied greatly between cultivars. The development of optimal rooting regimes for cultivars of V. uliginosum will improve plant survival at later stages of regenerant adaptation to ex vitro conditions.
Conflict of interest
The authors have no conflict of interest to report.
Acknowledgments
The work was carried out with financial support of the budgetary project of Central Siberian Botanical Garden, SB RAS NºAAAA-A17-117012610055-3 “Assessment of morphogenetic potential of North Asian plant populations by experimental methods” (No. 0312-2016-0001) within the framework of the State Assignment and the D. I. Mendeleev Scientific Fund Program of Tomsk State University. The authors would like to thank Prof. N. A. Vechernina for helpful suggestions and assistance in this study. We would like to extend gratitude to Mathew T. Sharples (Department of Ecology and Evolutionary Biology, University of Colorado, Boulder) and Mimi Kessler (Director, Eurasian Bustard Alliance, North Eurasian Bustard Coordinator, IUCN Bustard Specialist Group) for proofing the English of the text.
We used materials from the in vitro collection USU _440534 “Collection of living plants indoors and outdoors” of the Laboratory of Biotechnology, Central Siberian Botanical Garden.
References
[1] | Vander Kloet SP . Vaccinium Linnaeus. Flora of North America: Oxford University Press, USA; (2009) . |
[2] | Gorbunov AB , Snakina TI . Blueberries. Introduction of non-traditional fruit, berry and vegetable plants in Western Siberia. Novosibirsk: Geo; (2013) ;109–27 . |
[3] | Gahan PB . Totipotency and the cell cycle. In: Jain SM, Haggman H, editors. Protocols for micropropagation of woody trees and fruits. Springer, Dordrecht; (2007) ;3–14. |
[4] | Ragonezi C , Klimaszewska K , Castro MR , Lima M , De Oliveira P , Zavattieri MA . Adventitious rooting of conifers: Influence of physical and chemical factors. Trees. (2010) ;24: (6):975–92. doi: 10.1007/s00468-010-0488-8 |
[5] | Wiesman Z , Markus A , Wybraniec S , Schwartz L , Wolf D . Promotion of rooting and development of cuttings by plant growth factors formulated into a controlled-release system. Biol Fertil Soils. (2002) ;36: (5):330–4. doi: 10.1007/s00374-002-0553-6 |
[6] | Tetsumura T , Matsumoto Y , Sato M , Honsho Ch , Yamashita K , Komatsu H , Sugimoto Y , Kunitake H . Evaluation of basal media for micropropagation of four highbush blueberry cultivars. Sci Hortic-Amsterdam. (2007) ;119: (1):72–4. doi: 10.1016/j.scienta.2008.06.028 |
[7] | Debnath SC , McRae KB . An efficient in vitro shoot propagation of cranberry (Vaccinium macrocarpon Ait.) by axillary bud proliferation. In Vitro Cell Dev-Pl. (2001) ;37: (2):243–9. doi: 10.1007/s11627-001-0043-9 |
[8] | Hamburg KZ , Rekoslavskaya NI , Shvetsov SG . Auxins in the tissue and cells cultures of plant. Nauka: Novosibirsk; (1990) . |
[9] | Jaakola L , Tolvanen A , Laine K , Hohtola A . Effect of N6-isopentenyladenine concentration on growth initiation in vitro and rooting of bilberry and lingonberry microshoots. Plant Cell Tiss Org. (2001) ;66: (1):73–7. doi: 10.1023/A:1010602726215 |
[10] | Ostrolucka MG , Libiakova G , Ondruskova E , Gajdosova A . In vitro propagation of Vaccinium species. Acta Universitatis Latviensis, Biology. (2004) ;676: :207–12. |
[11] | Meiners J , Schwab M , Szankowski I . Efficient in vitro regeneration systems for Vaccinium species. Plant Cell Tiss Org. (2007) ;89: (2-3):169–76. doi: 10.1007/s11240-007-9230-7 |
[12] | Ruzic D , Vujovic T , Libiakova G , Cerovic R , Gajdosova A . Micropropagation in vitro of highbush blueberry (Vaccinium corymbosum L.). Journal of Berry Research. (2012) ;2: (2):97–103. doi: 10.3233/JBR-2012-030 |
[13] | Frett JJ , Smagula JM . In vitro shoot production of lowbush blueberry. Can J Plant Sci. (1983) ;63: (2):467–72. |
[14] | Song GQ , Sink KC . Agrobacterium tumefaciens-mediated transformation of blueberry (Vaccinium corymbosum L.). Plant Cell Rep. (2004) ;23: (7):475–84. doi: 10.1007/s00299-004-0842-7 |
[15] | Zimmerman RH . Micropropagation of woody plants: Post tissue culture aspects. Acta Hort. (1988) ;227: :489–99. doi: 10.17660/ActaHortic.1988.227.102 |
[16] | Zimmerman RH , Broome OC . Blueberry micropropagation. USDA-SEA Agr Res Results ARRNE. (1980) ;11: :44–7. |
[17] | Lyrene PM . Blueberry callus and shoot tip culture. Proc Fla State Hort Soc. (1978) ;91: :171–2. |
[18] | George EF . Plant Propagation by Tissue Culture. Exegetics Ltd., Basingstoke, UK; (1993) . |
[19] | Bertazza G , Baraldi R , Predieri S . Light effects on in vitro rooting of pear cultivars of different rhizogenic ability. Plant Cell Tiss Org. (1995) ;41: (2):139–43. doi: 10.1007/BF00051582 |
[20] | Tang W , Newton RJ . Micropropagation via organogenesis in slash pine. In: Jain SM, Haggman H, editors. Protocols for micropropagation of woody trees and fruits. Springer, Dordrecht, (2007) . pp. 15–22. |
[21] | Nissen SJ , Sutter EG . Stability of IAA and IBA in Nutrient Medium to Several Tissue Culture Procedures. Hort Science. (1990) ;25: (7):800–2. |
[22] | Drew RA , Simpson BW , Osborne WJ . Degradation of exogenous indole-3-butyric acid and riboflavin and their influence on rooting response of papaya in vitro. Plant Cell Tiss Org. (1991) ;26: (1):29–34. doi: 10.1007/BF00116606 |
[23] | Vechernina NA , Tavartkiladze OC , Gorbunov AB , Erst AA . Propagation in vitro of bog bilberry. Vestnik of Altai State Agrarian University. (2008) ;6: (44):21–5. |
[24] | Erst AA , Vechernina NA . Micropropagation of promising new cultivars of Vaccinium uliginosum L. Bulletin of the Kharkov National Agrarian University, Biological Series. (2010) ;2: (20):96–103. |
[25] | Anderson WC . Propagation of rhododendrons by tissue culture. I. Development of a culture medium for multiplication of shoots. Combined Proceedings of International Plant Propagators’ Society. (1975) ;25: :129–33. |
[26] | Vysotsky VA . Biotechnological methods in the system of production improved the planting material and breeding of fruit and berry plants: Author PhD diss; (1998) . |
[27] | Gorbunov AB , Voltchkov VE , Snakina TI . Green cuttings of bog bilberry. Forest Science of the XXI Century. Gomel; (2010) . pp. 520–4. |
[28] | McCown BH . Adventitious rooting of tissue cultured plants. In: Davis TD, Hassing BE and Sankhla N, editors. Adventitious Root Formation in Cuttings. Discorides Press, Portland, OR, USA; (1988) . pp. 287–302. |
[29] | Vasilieva OG . The biological and morphological base of clonal micropropagation of some species of the genus Rhododendron L.: Author PhD diss; (2009) . |
[30] | Erst AA , Karakulov AV , Erst AS . Propagation and conservation in vitro of unique genotypes of Rhododendron ledebourii Pojark. (Ericaceae DC). Adv Environ Biol. (2014) ;8: (10):276–82. |
[31] | Epstein E , Ludwig-Muller J . Indole-3-butyric acid in plants: Occurrence, synthesis, metabolism, and transport. Physiol Plant. (1993) ;88: (2):382–9. doi: 10.1111/j.1399-3054.1993.tb05513.x |
[32] | De Klerk GJ , Keppel M , Ter Brugge J , Meekes H . Timing of the phases in adventitious root formation in apple microcuttings. J Exp Bot. (1995) ;46: (8):965–72. |
[33] | De Klerk GJ , Ter Brugge J , Marinova S . Effectiveness of indoleacetic acid, indolebutyric acid and naphthaleneacetic acid during adventitious root formation in vitro in Malus ‘Jork 9’. Plant Cell Tiss Org. (1997) ;49: (1):39–44. doi: 10.1023/A:1005850222973 |
[34] | Vieitez J , Kingston DGI , Ballester A , Vieitez E . Identification of two compounds correlated with lack of rooting capacity of chestnut cuttings. Tree Physiol. (1987) ;3: (3):247, 255. |
[35] | Druart P , Kevers C , Boxus P , Gaspar T . In vitro promotion of root formation by apple shoots through darkness effect on endogenous phenols and peroxidases. Z. Pflanzenphysiol. (1982) ;108: (5):429–36. |
[36] | Behringer FJ , Davies PJ , Reid JB . Phytochrome regulation of stem growth and indole-3-acetic acid levels in the Iv and Lv genotypes of Pisum. Photochem Photobiol. (1992) ;56: (5):677–84. doi: 10.1111/j.1751-1097.1992.tb02221.x |
[37] | Pinker I , Zoglauer K , Goring H . Influence of light on adventitious root formation in birch shoot cultures in vitro. Biol Plant. (1989) ;31: :254–60. doi: 10.1007/BF02907285 |
[38] | Nemoykina AL . Influence of light and hormones on the morphogenesis of Yucca elephant in in vitro culture: Author. PhD diss; (2002) . |




