Effect of ethylene on ripening of kiwifruit stored under controlled or modified atmosphere packaging and treated with 1-methylcyclopropene
Abstract
BACKGROUND: Kiwifruit (Actinidia deliciosa (A. Chev.) C.F. Liang et A.R. Ferguson cv. ‘Hayward’) is usually harvested firm, unripe, and with a high starch content, and thus fruit do not always achieve a suitable soluble solid concentration (SSC), and soft texture when ripe. A high dry matter concentration at harvest can be used to predict fruit sweetness at ripening. However, fruit softening capacity is difficult to predict because of the interaction between postharvest practices and biochemical processes that occur at 0°C.
OBJECTIVE: To understand the effect of ethylene on restoring the lost softening capacity of kiwifruit treated with1-methylcyclopropene (1-MCP) alone or in combination with modified atmosphere packaging (MAP) or controlled atmosphere storage (CA).
METHODS: The effect of ethylene (100 μl l–1 for 12 h at 20°C) on kiwifruit harvested at commercial maturity (6.2% SSC) and treated with 1-MCP (1 μl l–1, 24 h) and packed in perforated bags (PB) or MAP for 60, 90 or 120 days at 0°C and ripened at 20°C for 17 days was determined. The effect of ethylene applied at 100 μl l–1 for 24, 48 or 72 h was also evaluated on fruit treated with 1-MCP (1 μl l–1, 24 h) subjected to 108 days of CA (2% O2 and 5% CO2) and a further 30 days in PB at 0°C.
RESULTS: Both 1-MCP and MAP reduced fruit softening after 60 and 90 days at 0°C, and delayed fruit ripening at 20°C. A synergistic effect of 1-MCP and MAP extended those results to 120 days at 0°C. Ethylene treatment restored the ripening of MA fruit but not the 1-MCP-treated fruit stored for 60 days at 0°C or when it was packed in MA bags for 90 days at 0°C. Ethylene for 48 h prevented the ‘hard core’ texture observed in CA with 1-MCP treated fruit.
CONCLUSIONS: Kiwifruit treated with 1-MCP and/or packed with MA requires a minimum time at 0°C before to induce ripening by ethylene treatment (100 μl l–1 for 12 h at 20°C). Ethylene treatment avoids ‘hard core’ development on 1-MCP treated fruit stored in CA.
1Introduction
Kiwifruit (Actinidia deliciosa (A. Chev.) C.F. Liang et A.R. Ferguson cv. ‘Hayward’) is harvested mature, firm, unripe, and with a starchy texture that allows storage at 0°C for 4 to 6 months. However, softening is the main concern for kiwifruit storage worldwide and considerable attention has been given to new technologies that maintain fruit firmness during storage. Modified atmosphere packaging [1], controlled atmosphere storage [2–4], and 1-methylcyclopropene (1-MCP) [5–10], in combination with rigorous temperature management at 0°C in an ethylene-free environment are commercially available tools that slow kiwifruit softening. However, the quality desired by consumers is not always achieved at ripening since fruit may remain hard or have irregular softening, and the fruit harvested with a low dry matter concentration will have a low sweetness.
The quality of kiwifruit on the market depends on attributes gained during fruit development and on the biochemical processes that occur during storage and subsequent ripening. These processes include the conversion of starch to sugar and the degradation of acid and depolymerization of the cell wall that results in softening and in the development of flavor. Based on the strong association, at the ripe stage, between soluble solid and dry matter (DM) concentrations, a minimum value of 16.0% fruit DM is recommended at harvest [11, 12]. Therefore, the perception of high quality kiwifruit by consumers requires a high DM concentration at harvest as well as a softening rate that ensure a ripe stage with acceptable flavor, lack of physiological disorders, and uniform softening among fruit and fruit tissues. Fruit that is harvested when immature may never soften fully and an external ethylene application may help to restore (albeit not completely) the natural ripening capacity obtained when fully mature fruit is harvested.
The application of ethylene at 100 μl l–1 for 6–12 h at 0°C has been recommended to reduce the ripening time at 20°C [13, 14], which allows the selling of ‘ready to eat’ kiwifruit after one month of storage. The process of softening after ethylene application and during fruit ripening at 20°C depends on the length of storage at 0°C prior the application, the ethylene concentration and the temperature at the time of application [15, 16].
Especially with immature fruit, the postharvest management practices, such as a storage temperature below 0°C and the length of storage may promote physiological disorders [17–20]. The application of 1-MCP, an inhibitor of ethylene action, at harvest has been demonstrated to slow softening at 0°C and maintain kiwifruit firmness especially during ripening [7–10]. A similar effect on firmness retention during storage occurs with the application of MAP and CA technologies. This effect is mainly attributed to the high CO2 concentration and less to the low O2 [1, 2].
However, there are concerns about uneven and poor ripening with the use of these technologies, e.g., a hard core is surrounded by ripe flesh tissue [21]. The objective of this work was to understand the effect of ethylene to restore the softening capacity of kiwifruit treated with 1-methylcyclopropene (1-MCP) alone or in combination with modified atmosphere packaging (MAP) or controlled atmosphere storage (CA) during ripening after different storage periods at 0°C.
2Materials and methods
2.1Plant material
Actinidia deliciosa fruit (cv. ‘Hayward’) with a mass between 100 and 130 g were harvested from two commercial orchards located in the Central Valley (Chimbarongo, VI region) and Central South Chile (Curicó, VII region). The fruit harvested from the first orchard had a 6.4% soluble solid (SS) concentration, 1.6% titratable acidity (TA), 17.2% dry matter (DM) concentration, and a 68.5 N average firmness. The maturity parameters of the fruit harvested from the second orchard were 6.2% SS, 16.4% DM, 1.2% TA and 77 N firmness. The fruit harvested from the first orchard were used to investigate the effect of ethylene on the ripening capacity of fruit treated with 1-MCP and/or packed in modified atmosphere bags. The fruit from the second orchard were used to evaluate the effect of the ethylene application period (i.e., 24, 48, 72 h) on the ripening condition of fruit previously treated with 1-MCP and subjected to commercially controlled atmosphere storage and packed in perforated bags.
2.2Treatments
To study the effect of ethylene on the ripening capacity of fruit treated with 1-MCP and/or packed in modified atmosphere bags, fruit harvested on the same day were stored at 20°C for 36 h. Then, fruit were randomly divided into two groups (see Fig. 1 for the details of amount of boxes used); one group was transferred to a 24-m3 chamber at 20°C and 1 μl l–1 of 1-MCP was released from SmartFreshTM powder (a.i. 0.14%; Agrofresh) for 24 h according to the manufacturer’s instructions. The second group remained in the 20°C chamber and was used as control. The fruit in each group were placed in 10-kg carton boxes using two types of packaging: i) a high density 10 μm, 0.3% -perforated polyethylene bag (PB) and ii) a 60- μm, low-density polyethylene modified atmosphere bag (MAP, FF Kiwi, San Jorge Packaging, Chile). The boxes were stored at 0°C for three days with the bags open (the pulp temperature was 1°C). Then, the MA bags were heat sealed and the top of the PB bags were folded and stored for 60, 90 or 120 days at 0°C (±0.5) in an ethylene-free environment produced by passing air through a potassium permanganate ethylene scrubber. The ethylene concentration was monitored every two days and was consistently <0.005 μl l–1. One 10-kg box of 100 fruit each, from a total of 4 replicates was used as an individual replicate and 15 fruit were analyzed for firmness, SS and TA immediately after removal from storage and at 3, 7, 10, 14 and 17 days at 20°C. Similar amount of fruit was used for ethylene treatment, Following storage at 0°C for 60, 90 or 120 days, half of the MA and PB boxes were transferred to a 20°C ripening room where 100 μl l–1 of ethylene obtained from a commercial mixture of 95% N2 and 5% C2H4 (Azetil, Indura, Santiago, Chile) was applied for 12 h and monitored using 108SC ethylene detector tubes. The detection limit was 1–200 ul l–1 (Kitagawa, Kawasaki, Japan). The remaining boxes were stored in a 20°C room. These treatments were performed on previously opened MA and PB bags. Fruit ripening at 20°C was monitored every 2-3 days for a period of 17 days (Fig. 1). An analysis of variance (ANOVA) was performed to determine significant differences between treatments (i.e., PB, 1-MCP, MAP, MAP + 1-MCP) and an LSD test was used to identify the differences (P < 0.05) in firmness after treatment for 60, 90 or 120 days at 0°C. The effect of ethylene was determined by comparing the standard error of the fruit flesh and core firmness after ripening at 20°C of ethylene-treated and non-treated fruit.
To study the effect of the ethylene application period on fruit treated with 1-MCP and controlled atmosphere storage (CA), kiwifruit were harvested in commercial 400-kg bins and transported to a packing house; five bins were labeled and the postharvest process was monitored. The fruit were cured for 48 h in a ventilated covered environment in an ethylene-free condition (<0.005 μl l–1 ethylene) and stored in a commercial cold storage room with a 450-ton capacity. The fruit were cooled in the bins from a temperature of 15-13°C to 1°C over a period of 7–10 days. Once the pulp temperature was between 5 and 3°C, 1 μl l–1 of 1-MCP was released for 24 h from a commercial generator (Smartfresh, Agrofresh) located inside the chamber. An atmosphere of 2% ±0.5% O2 and 5% ±1% CO2 was established in the CA treatment 15 days after harvest. After a 108-day period in CA storage, the fruit were graded, packaged in 10-kg boxes using a high density 10-μm, 0.3% perforated polyethylene bag and stored for an additional 30 days at 0°C in a normal atmosphere, simulating the transport to the market. Then, the boxes were transferred to 20°C and 100 μl l–1 of ethylene was applied for 24, 48 or 72 h and compared with fruit stored at 20°C (control treatment) for the same period of time under ethylene-free conditions. Fruit ripening at 20°C was monitored every 3-4 days for a period of 17 days (Fig. 2). Four replicates of 15 fruit each were used to compare the standard error of the whole fruit and core tissue firmness of the ethylene-treated and non-treated fruit ripened at 20°C.
A separate experiment was established to determine the relationships between the firmness of the core tissue evaluated using a penetrometer and the perception of the core firmness described by a trained group of selected soft kiwifruit (<10 N) of the CA treatment.
2.3Quality parameters evaluation
The quality evaluation of kiwifruit included the determination of whole fruit and core firmness, and the SS and TA concentration. Following skin removal, whole fruit firmness was measured on the opposite sides of the fruit using a penetrometer (FT 327; Effegy, Alfonsine, Italy) mounted on a drill base and fitted with a 7.9-mm tip. The firmness of the core was assessed by pressing the tip of the penetrometer directly on the core tissue of one of the halves after cutting the fruit equatorially. The soluble solid concentration and titratable acidity were determined on the juice collected from a composite sample of 1-cm longitudinal tissue acquired from 15 fruit; the soluble solid was measured using thermocompensated refractometer (Atago, Spectrum Technologies, Plainfield, Illinois, USA) the titratable acidity was determined by titration of 5 ml juice to pH 8.2 with 0.1 N NaOH and the percent citric acid equivalents were determined.
2.4Sensory parameters evaluation
The perception of core firmness was evaluated by eight trained panelists. Each panelist was presented with four halves of kiwifruit samples at five targeted ranges of core firmness (i.e., 17.8–22.2 N, 22.6–31.1 N, 35.5–40.0 N, 44.4–48.8 N, 53.3–66.6 N). The core firmness was previously measured on one of the halves of soft kiwifruit using a penetrometer. Within each range of penetrometer values, the panelists were asked to judge the perception of core firmness on a scale of 0 (soft) to 5 (hard). The panelists were presented with the fruit in a random order. The degree of perception was subjected to an analysis of variance (ANOVA) and the means were compared using the LSD test (P < 0.05).
3Results and discussion
The application of 1 μl–1 l of 1-MCP or packaging in a modified atmosphere bag significantly reduced kiwifruit softening after 60 and 90 days at 0°C compared with fruit packed in polyethylene perforated bags (PB, control). The highest firmness value was obtained with the treatment of 1-MCP and packaging in an MA bag. However, the minimum firmness value (35.5 N) required by some importers after 120 days at 0°C was not obtained with any of the treatments (Table 1).
The reduction of softening in kiwifruit and other species is well documented as the main physiological effect in response to a reduction in ethylene production when ethylene is inhibited at the receptor level by 1-MCP. 1-MCP also reduces directly ethylene production in kiwifruit by blocking expression of the 1-aminocyclopropane-1-carboxylic acid synthase and 1-aminocyclopropane-1-carboxylic acid oxidase genes which are responsible for ethylene biosynthesis enzymes [22]. However, the reduction in kiwifruit softening is not as marked as that reported for apple [23].
The internal concentration of CO2 and O2 in the MA bags varied between 5–8% and 10–15%, respectively (data not shown), after 120 d at 0°C. These values are in accordance with the steady state concentration reported in other publication using the same bags [1]. High CO2 concentration has a greater ability to retard kiwifruit softening compared with low O2 [2]. The main physiological effects that are attributed to a high CO2 concentration include a reduction in metabolic pH in the intercellular space, inhibition of ACC oxidase, and competitive interaction with ethylene receptors [24–27].
The effect of 1-MCP, MAP or the combination of both treatments persisted during fruit ripening. The flesh and core naturally soften when the fruit ripen, and the evaluation of the whole fruit is similar as the loss of firmness in the flesh [21]. However, the core tissue maintains greater firmness than the flesh and this difference is reduced during the ripening process at 20°C [15, 21, 28, 29]. The perception of the eating quality of ‘Hayward’ kiwifruit is a combination of flesh and core firmness, and marked differences in the firmness of these tissues may affect consumer acceptability such as the ‘hard core’ texture described previously [21]. Some standards firmness criteria have been established previously [21, 30]. Firmness of kiwifruit between 13 and 18 N are considered ‘ready to buy’ because of start of sensing soft at hand palm pressure and fruit between 4 to 13 N are ‘ready to eat’ and firmness in the range of 5.8 to 8 N is considered acceptable for the consumers.
The fruit treated with 1-MCP alone (Fig. 3, PB + 1-MCP) or in combination with MAP (Fig. 4, MAP + 1-MCP) retained a firmness value higher than 13 N for a period longer than 14 days at 20°C after 60 days at 0°C compared with control fruit (PB), which ripened within 7–10 days. The most significant effect of 1-MCP was to reduce the softening of the core, which intensified the firmness difference with the whole fruit. This effect was detected during ripening at 20°C, even after the fruit were stored for 90 days at 0°C and it was most noticeable on 1-MCP-treated fruit packed in MA bags (Figs. 3 and 4). The firmness difference of the whole fruit persisted after 10 days of ripening at 20°C on fruit stored in MAP for 120 days at 0°C (Fig. 4, MAP + 1-MCP).
The application of ethylene (100 μl l–1) for 12 h to fruit stored for 60, 90 and 120 days at 0°C reduced the ripening time at 20°C; however, this effect depended on the kiwifruit tissue and postharvest treatment used to reduce fruit softening at 0°C. Ethylene to fruit packed in perforated bags reduced firmness on fruit removed after 60 and 90 days at 0°C; however, the strongest effect was obtained on core softening, which reduced the differences in firmness between the whole fruit and core (Fig. 5). A similar effect was observed in fruit stored in MAP, where fruit treated with ethylene ripened rapidly at 20°C, which reduced the firmness of the whole fruit and especially the core (Fig. 6). This demonstrates the high sensitivity of the fruit pulp tissues to ethylene after a period of storage under high CO2 concentration.
In fruit stored in PB, the application of ethylene to 1-MCP-treated fruit did not change the softening of fruit stored for a period shorter than 120 days at 0°C although there was a significant reduction in core firmness during ripening compared with fruit not treated with ethylene on fruit stored for 60 and 90 days at 0°C (Fig. 7).
In fruit treated with 1-MCP and stored in MA bags, the ethylene treatment demonstrated that the effects of 1-MCP persisted in fruit stored for 60 or 90 days at 0°C, because the 12 h application of 100 ul l–1 of ethylene did not change the fruit softening during ripening at 20°C. In addition, the synergist effect of 1-MCP and MAP was demonstrated by the null softening produced by the ethylene on the core tissue in the first 7 days of ripening at 20°C after the fruit were stored for 90 days at 0°C (Fig. 8). Fruit maturity, length of ethylene treatments are factors that would probably change these results and requires further investigation. Soluble solid concentration was delayed by the 1-MCP and MAP treatments in concordance with the delay of ripening, but similar concentration in the range of 14.2% to 14.8% was obtained at the ‘ready to eat’ stage (<13 N). The application of ethylene did not influence greatly this result (data not shown). The TA varied from 1.6% at harvest to 1.02% –1.31% after ripening at different storage time and treatments. The highest TA values were found on fruit treated by 1-MCP, MAP or a combination of both treatments. Ethylene did not reduce the TA consistently only in fruit treated with 1-MCP and packed with MA bag got significant reduction after 60 and 90 days at 0°C. (Table 2).
The effect of ethylene application period on kiwifruit treated with 1-MCP and CA storage was evaluated in a separated experiment.
Kiwifruit treated with 1-MCP and stored for 108 days under CA and 30 days at 0°C were used to determine the effect of 24, 48 or 72 h of ethylene application on the softening of the pulp and core tissues during ripening. The application of 48 or 72 h of 100 μl l–1 of ethylene increased fruit softening compared with the 24-h treatments or fruit ripened at 20°C without ethylene (Control); however, the main effect of ethylene was on the texture of the core. A ripening period of 24, 48 or 72 h at 20°C with ethylene reduced the firmness of the core during further ripening at 20°C; the applications for 48 or 72 h were the most effective treatments (Fig. 9).
To understand the sensory perception of a hard core, a group of trained panelists were asked to differentiate kiwifruit with a range of core firmness (the core firmness of soft flesh kiwifruit (<10 N) was evaluated using a penetrometer). The panelists were able to differentiate the increasing range of core firmness detected by the penetrometer with values ranging from 0 (soft) to 5 (hard). A score value higher than 2.5 indicates fruit with a hard core, which coincides with firmness greater than 35.5 N –40 N (Fig. 10). Although a hard or fibrous core appears as description of firm fruit in the work performed by Stec et al. [21] using the same methodology, these authors did not report specific firmness values that can be associated with hard core tissue. Hard core symptoms described here resemble a physiological disorder associated to loss of ripening capacity of this specific tissue, when kiwifruit are stored for a long period of time on CA and when the ethylene perception and production have been limited by 1-MCP. This symptom was not detected on fruit treated with 1-MCP and packed in MA bag (the most effective treatment for reducing softening at 0°C). Probably the CO2/O2 concentration could explain part of the differences. However fruit growing location and pre harvest practices should be important factors to be considered. Less transcripts of 1-aminocyclopropane-1-carboxylic acid synthase gene were detected in the core than in the flesh [22] that predispose genetically to less production of ethylene and leads with the difficulties to induce normal softening under blocked ethylene production by 1-MCP together with high CO2 and low oxygen of CA treatment. More research need to be done to understand the direct effect induced on cell wall degradation enzymes associated with hard core texture and how agronomic factors differentiate the capacity of fruit to soft.
Acknowledgments
This information is part of the research program ‘Fortalecimiento de la competitividad del kiwi Chileno’ of ‘The Chilean Kiwifruit Committee’ and supported by The ‘Fundación para la Innovación Agraria’ (FIA).
References
[1] | Zoffoli JP , Rodríguez J , Levy N . Postharvest factors that influence the effectiveness of modified atmosphere packaging of kiwifruit. Acta Hort. (2007) ;753: :741–744. |
[2] | Arpaia M , Mitchell F , Kader A, Mayer G. Effects of 2% O2 and varying concentrations of CO2, with or without C2H4, on the storage performance of kiwifruit. J Amer Soc Hort Sci. (1985) ;110: :200–203. |
[3] | Antunes M , Sfakiotakis E . Ethylene biosynthesis and ripening behavior of Hayward kiwifruit subjected to some controlled atmosphere. Postharvest Biol Technol. (2002) ;26: :167–179. |
[4] | McDonald B , Harman J . Controlled-atmosphere storage of kiwifruit I. Effect on fruit firmness and storage life. Scientia Horticulturae. (1982) ;17: :113–123. |
[5] | Boquete E , Trinchero G , Fraschina A , Vilella F , Sozzi G . Ripening of “Hayward” kiwifruit treated with 1-methylcyclopropene after cold storage. Postharvest Biol Technol. (2004) ;32: :57–65. |
[6] | Crisosto C , Garner D . 1-MCP inhibits kiwifruit softening during storage. Perishables Handling Quarterly. (2001) ;108: :19–20. |
[7] | Kim H , Hewett E , Lallu N . Softening and ethylene production of kiwifruit reduced with 1-methylcyclopropene. Acta Hort. (2001) ;553: :157–170. |
[8] | Koukounaras A , Sfakiotakis E . Effect of 1-MCP prestorage treatment on ethylene and CO2 production and quality of ‘Hayward’ kiwifruit during shelf-life after short, medium and long term cold storage. Postharvest Biology and Technology. (2007) ;46: :174–180. |
[9] | Regiroli G , Vriends P . SmartfreshSM (1-methylcyclopropene) benefits for kiwifruit. Acta Hort. (2007) ;753: :745–753. |
[10] | Sfakiotakis E , Koukounaras A . Postharvest 1-MCP (Smartfresh SM) treatment prolongs storage and shelf life of kiwifruit by suppression of ethylene action and production. Acta Hort. (2007) ;753: :755–761. |
[11] | Crisosto C , Zegbe J , Hasey J , Crisosto G . Is dry matter a reliable quality index for ‘Hayward’ Kiwifruit? Acta Hort. (2011) ;913: :531–534. |
[12] | Burdon J , McLeod D , Lallu N , Gamble J , Petley M , Gunson A . Consumer evaluation of “Hayward” kiwifruit of different at-harvest dry matter contents. Postharvest Biol Technol. (2004) ;34: :245–255. |
[13] | Crisosto C . Final preconditioning guidelines for kiwifruit shippers. Central Valley Postharvest Newsletter. (1997) ;6: :1–4. |
[14] | Crisosto C , Garner D , Crisosto GM , Kaprielian R . Kiwifruit Preconditioning protocol. Acta Horticulturae. (1997) ;444: :555–559. |
[15] | Lallu N , Searle A , MacRae E . An investigation of ripening and handling strategies for early season kiwifruit (Actinidia deliciosa. cv Hayward). J Sci Food Agr. (1989) ;47: :387–400. |
[16] | Ritenour M , Crisosto C , Garner D , Cheng G , Zoffoli J . Temperature, length of cold storage and maturity influence the ripening rate of ethylene-preconditioned kiwifruit. Postharvest Biol Technol. (1999) ;15: :107–115. |
[17] | Antunes M , Sfakiotakis E . Changes in fatty acid composition and electrolyte leakage of ‘Hayward’ kiwifruit during storage at different temperaturs. Food Chemistry. (2008) ;110: :891–896. |
[18] | Lallu N . Low temperature breakdown in kiwifruit. Acta Hort. (1997) ;444: :579–586. |
[19] | Burdon J , Lallu N , Francis K , Boldingh H . The susceptibility of kiwifruit to low temperature breakdown is associated with pre-harvest temperatures and at harvest soluble solids content. Postharvest Biol Technol. (2007) ;43: :283–290. |
[20] | Sfakiotakis E , Chlioumis G , Gerasopoulus D . Preharvest chilling reduces low temperature breakdown incidence of kiwifruit. Postharvest Biol Technol. (2005) ;38: :169–174. |
[21] | Stec M , Hodgson J , MacRae E , Triggs C . Role of fruit firmness in the sensory evaluation of kiwifruit (Actinidia deliciosa cv Hayward). J Sci Food Agric. (1989) ;47: :417–33. |
[22] | Illina N , Alem H , Pagano E , Sozzi G . Suppression of ethylene perception after exposure to cooling conditions delays the progress of softening in ‘Hayward’ kiwifruit. Postharvest Biol and Technol. (2010) ;55: :160–168. |
[23] | Watkins CB . Overview of 1-methylcyclopropene trials and uses for edible horticultural crops. HortScience. (2008) ;43: :86–94. |
[24] | Burton WG . Some bio-physical principles underlying the controlled atmosphere storage of plant material. Ann Appl Biol. (1974) ;78: :149–68. |
[25] | Lange DL , Kader AA . Elevated carbon dioxide exposure alter intracellular pH and energy charge in avocado fruit tissue. J Amer Soc Hort Sci. (1997) ;122: :253–257. |
[26] | Mathooko FM . Regulation of respiratory metabolism in fruit and vegetables by carbon dioxide. Postharvest Biol Technol. (1996) ;7: :1–26. |
[27] | Mathooko FM . Regulation of ethylene biosynthesis in higher plants by carbon dioxide. Postharvest Biol Technol. (1996) ;9: :247–264. |
[28] | MacRae E , Bowen J , Stec M . Maturation of kiwifruit (Actinidia deliciosa cv. Hayward) from two orchards: Differences in composition of the tissue zones. J Sci Food Agric. (1989) ;47: :401–416. |
[29] | Jackson P , Harker F . Changes in firmness of the outer pericarp, inner pericarp, and core of Actinidia species during ripening. New Zealand Journal of Crop and Horticultural Science. (1997) ;25: :185–189. |
[30] | Crisosto C , Crisosto G . Understanding consumer acceptance of early harvested ‘Hayward’ kiwifruit. Postharvest Biology and Technology. (2001) ;22: :205–213. |
Figures and Tables
Fig.1
Amount of boxes required to investigate the effect of ethylene on the ripening capacity of fruit treated with 1-MCP and/or packed in modified atmosphere bags, a total of 96 kiwifruit boxes were used, 48 boxes on 1-MCP treatment and the other half was left as control. The 1-MCP boxes used are described for the ethylene and control treatments applied after 60, 90 and 120 days at 0°C. Four replicates of one box each was evaluated considering 15 fruit each. The evaluations were at 0, 3, 7, 10, 14 and 17 d at 20°C after 60, 90 and 120 days at 0 C. A total of 100 fruit were included in each box. Similar amount of boxes and fruit were used for the control without 1-MCP. PB: Perforated bag, MAP: Modified atmosphere packaging
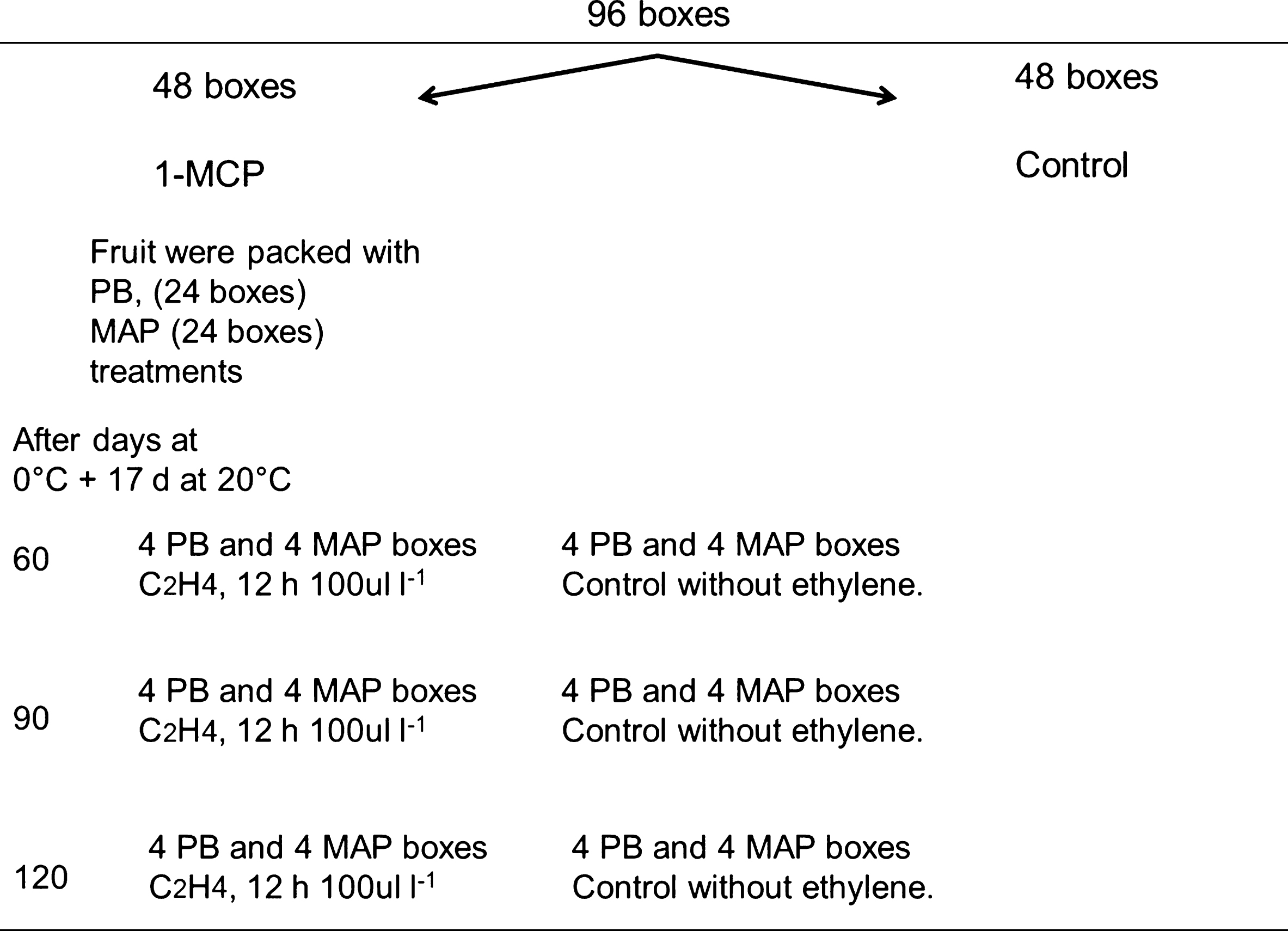
Fig.2
Amount of boxes required to investigate the effect of ethylene on the ripening capacity of fruit treated with 1-MCP (1 ul l–1) at harvest and stored in controlled atmosphere (CA), a total of 24 kiwifruit boxes were packed after 108 days storage in CA (2% O2 and 5% CO2), four boxes were treated for 24, 48 and 72 h with ethylene 100 ul l–1 and 15 fruit each were evaluated after at 0, 3, 7, 10, 14 and 17 d at 20°C. A similar amount of fruit was used as control without the application of ethylene at 20°C.
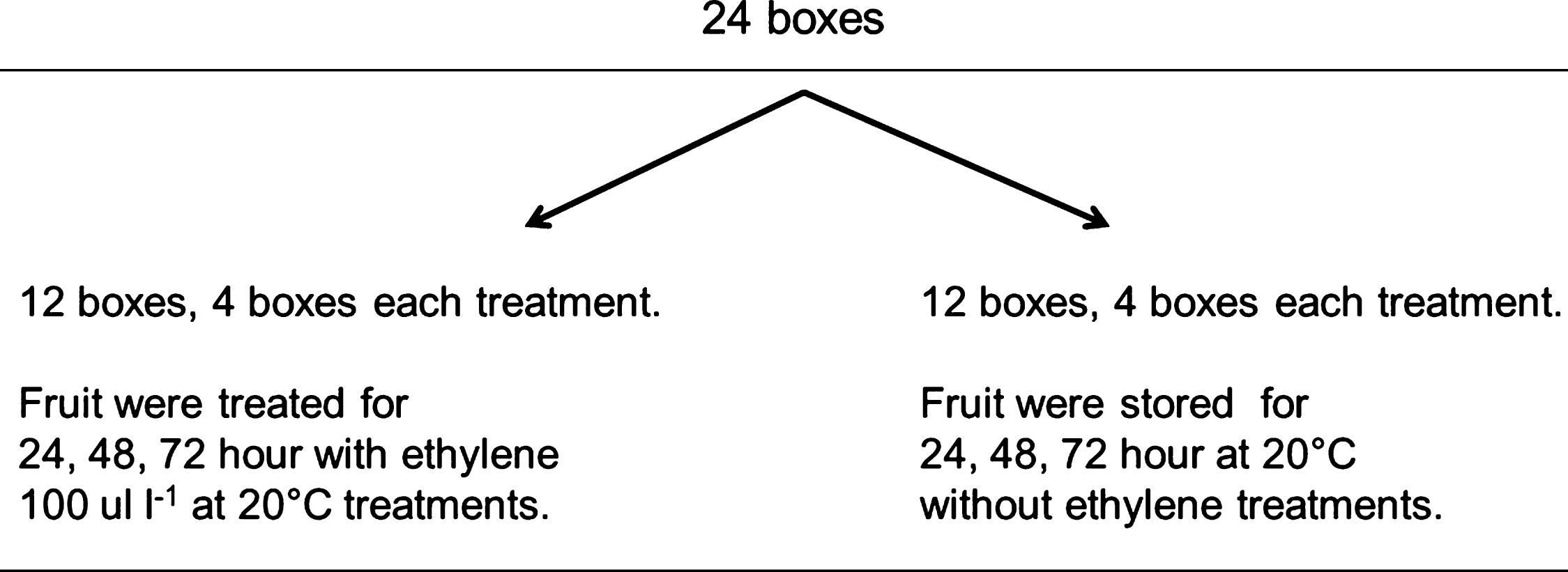
Fig.3
Effect of 1-methylcyclopropene (1-MCP), on softening of whole kiwifruit or core tissue, packed with perforated bag (PB) during ripening at 20°C after storage for 60, 90 or 120 days at 0°C. The horizontal lines indicate the ready to eat firmness value (<13 N). The vertical bar represents the standard error of the firmness mean of 4 replicates of 15 fruit each.
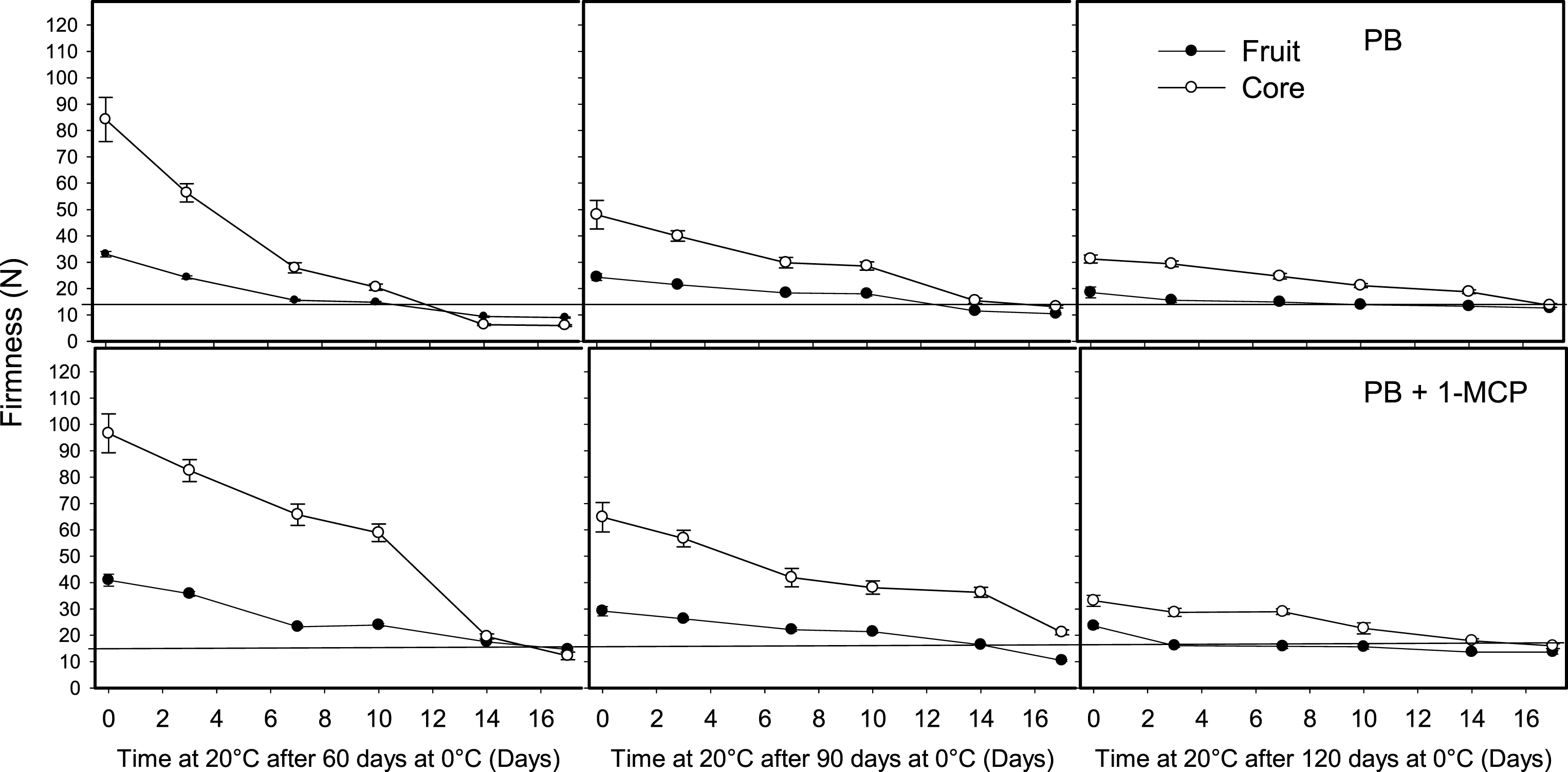
Fig.4
Effect of 1-methylcyclopropene (1-MCP) on softening of whole kiwifruit or core tissue of fruit packed on modified atmosphere packaging (MAP), during ripening at 20°C after storage for 60, 90 or 120 days at 0°C. The horizontal lines indicate the ready to eat firmness value (<13 N). The vertical bar represents the standard error of the firmness mean of 4 replicates of 15 fruit each.
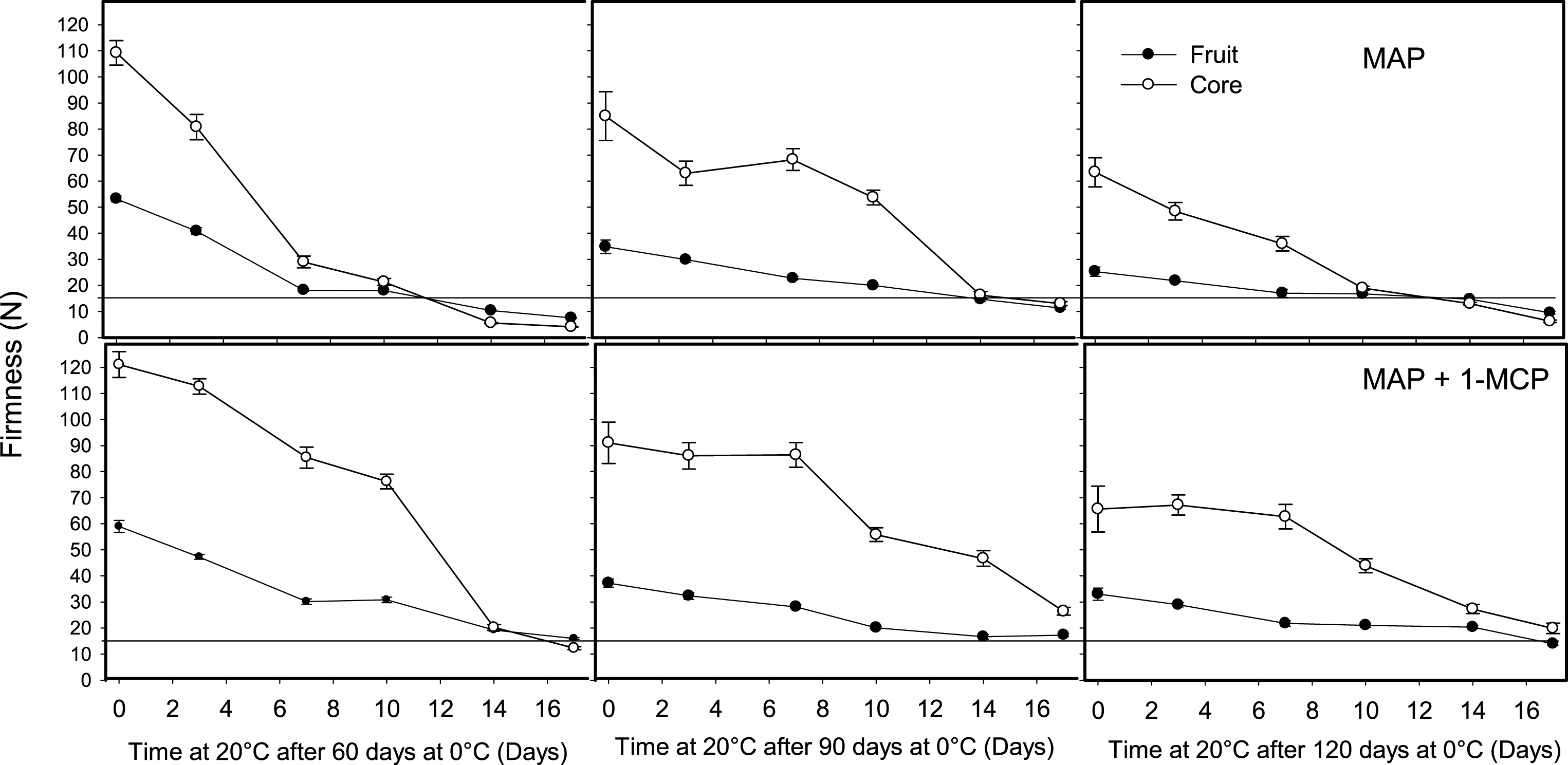
Fig.5
Effect of ethylene on softening of whole kiwifruit or core tissue during ripening at 20°C after storage for 60, 90 or 120 days at 0°C. Ethylene (100 μl l–1) was applied at 20°C for 12 h after removing fruit packed in a perforated bag (PB). The solid and dotted horizontal lines indicate the ready to eat firmness value (<13 N) and soft core firmness perception (<35.5 N) receptively. The vertical bar represents the standard error of the firmness mean of 4 replicates of 15 fruit each.
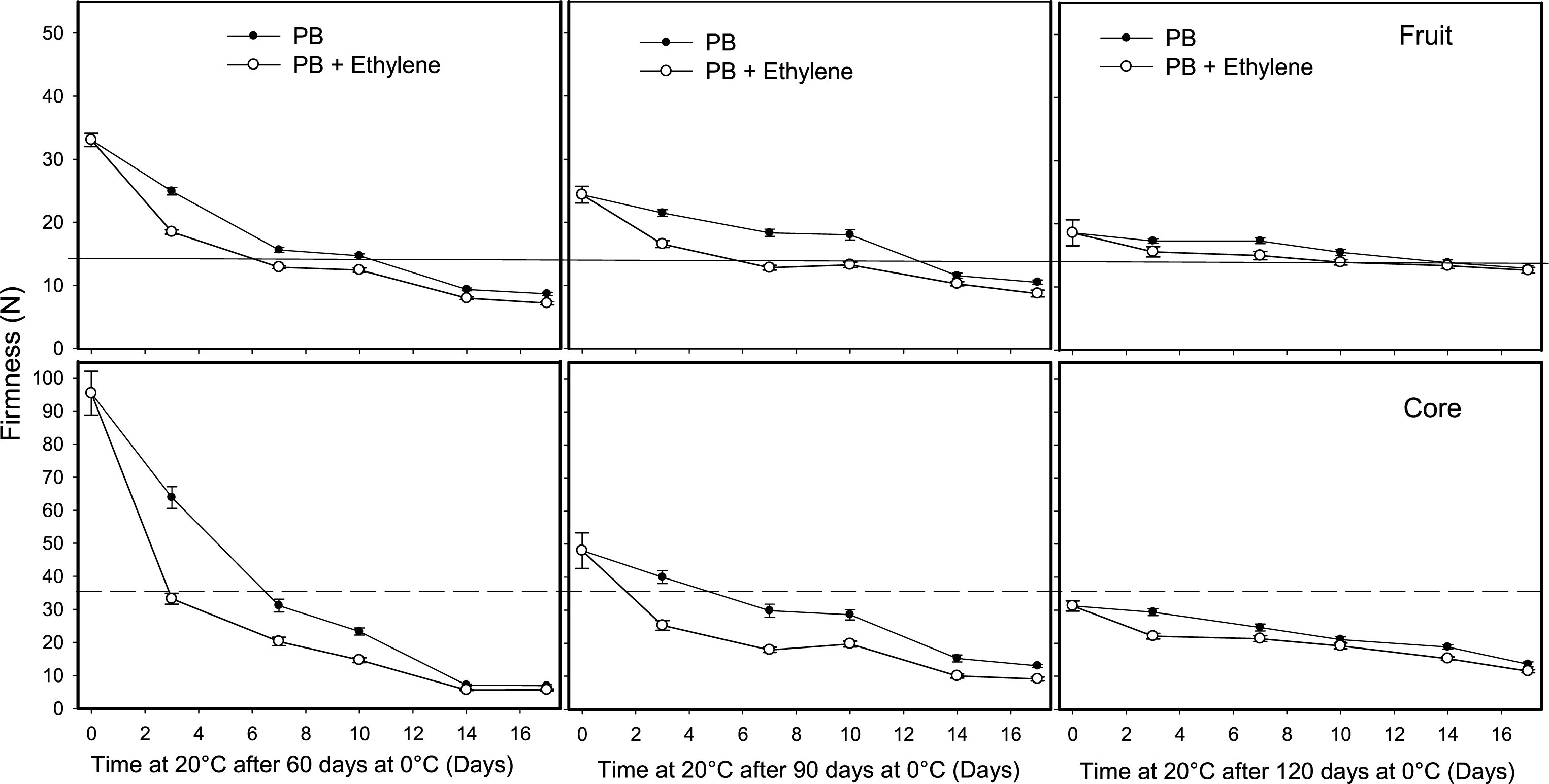
Fig.6
Effect of ethylene on softening of whole kiwifruit or core during ripening at 20°C after storage for 60, 90 or 120 days at 0°C. Ethylene (100 μl l–1) was applied at 20°C for 12 h after removing fruit packed in modified atmosphere packaging (MAP). The solid and dotted horizontal lines indicate the ready to eat firmness value (<13 N) and soft core firmness perception (<35.5 N) receptively. The vertical bar represents the standard error of the firmness mean of 4 replicates of 15 fruit each.
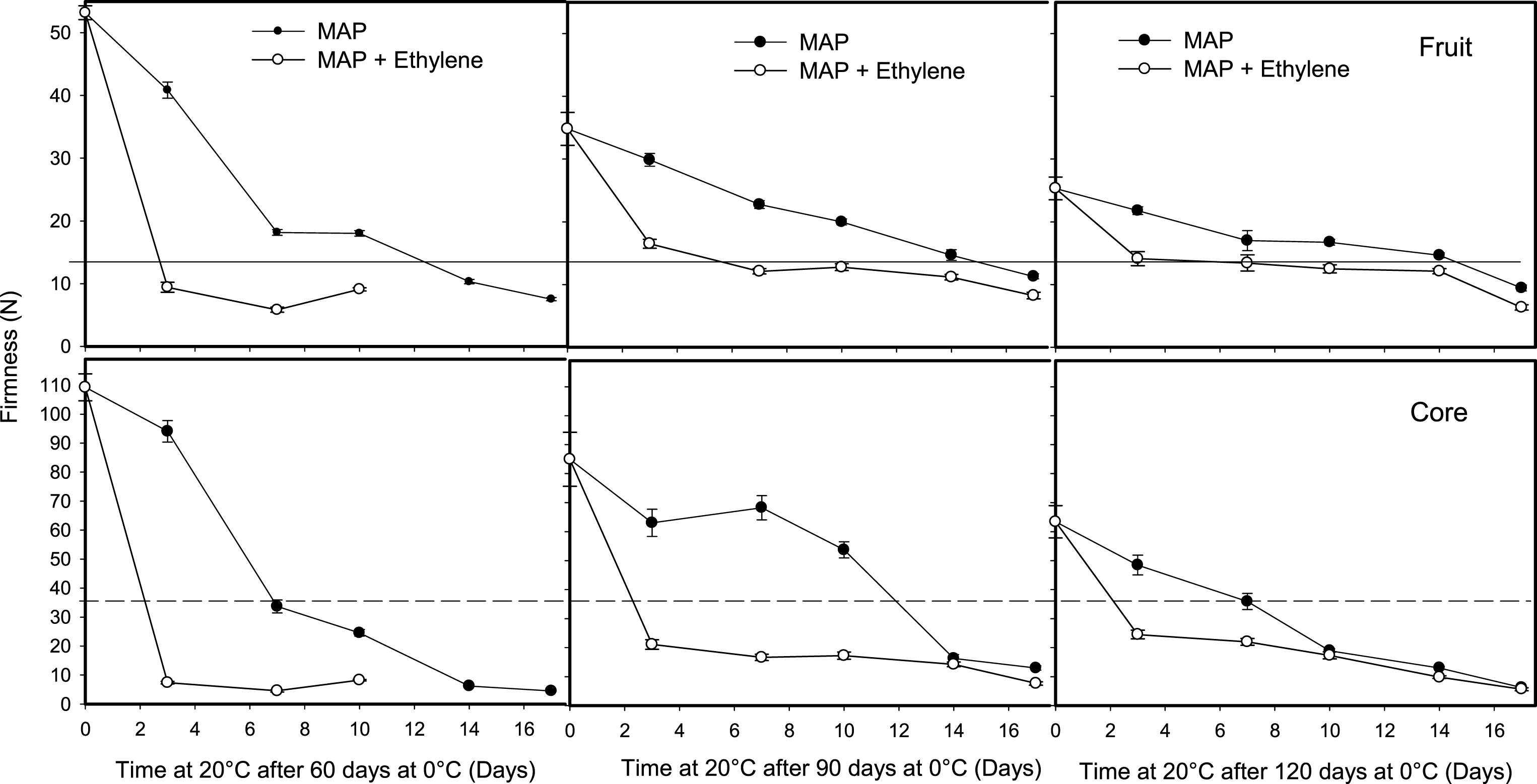
Fig.7
Effect of ethylene on softening of whole kiwifruit or core tissue during ripening at 20°C after storage for 60, 90 or 120 days at 0°C. Ethylene (100 μl l–1) was applied at 20°C for 12 h after removing fruit treated with 1 μl l–1 of 1-MCP at harvest and packed in a perforated bag (PB). The solid and dotted horizontal lines indicate the ready to eat firmness value (<13 N) and soft core firmness perception (<35.5 N) receptively. The vertical bar represents the standard error of the firmness mean of 4 replicates of 15 fruit each.
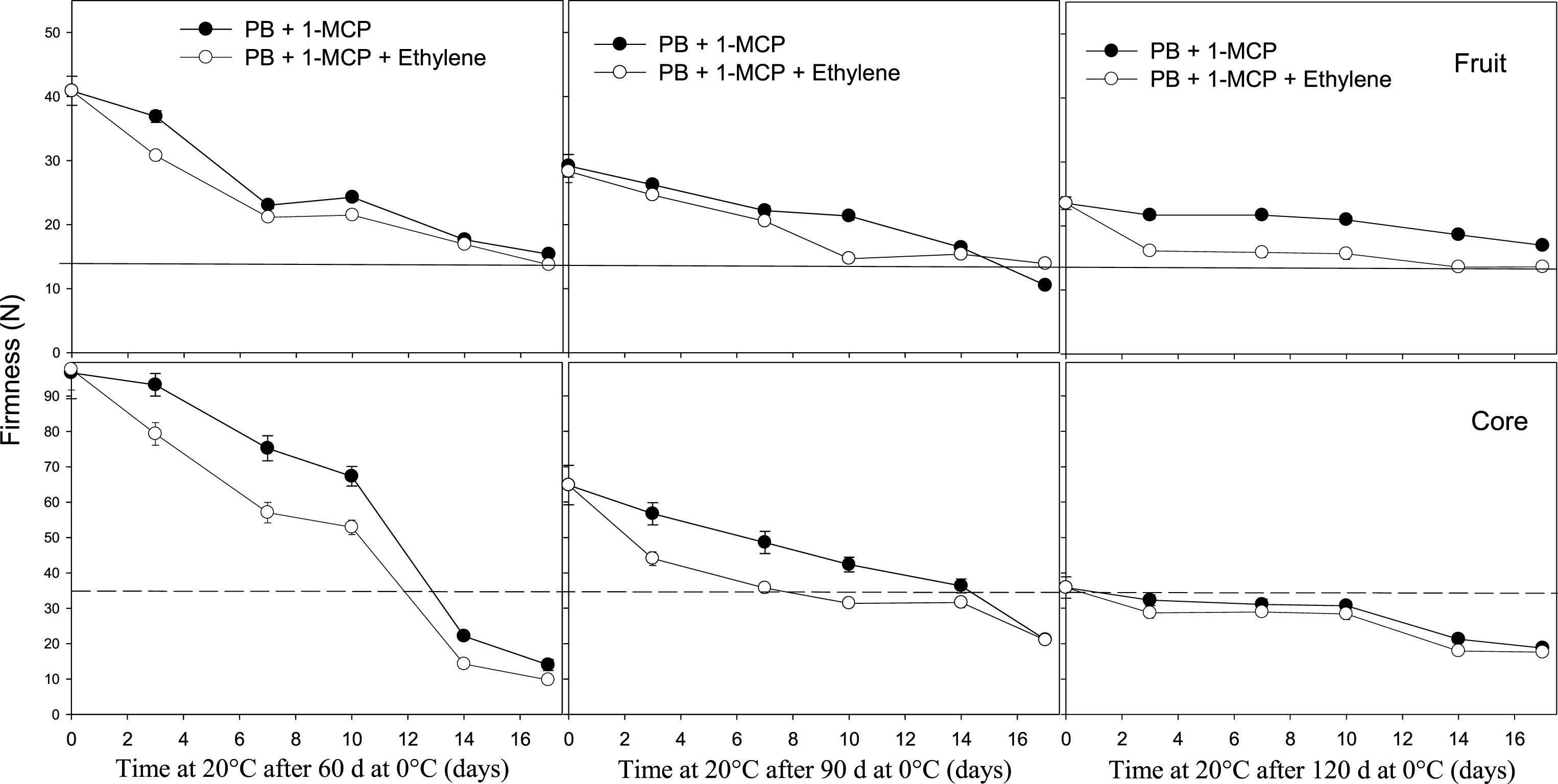
Fig.8
Effect of ethylene on softening of whole kiwifruit or core tissue during ripening at 20°C after storage for 60, 90 or 120 days at 0°C. Ethylene (100 μl l–1) was applied at 20°C for 12 h after removing fruit treated with 1 μl l–1 of 1-MCP at harvest and packed in modified atmosphere system (MAP). The solid and dotted horizontal lines indicate the ready to eat firmness value (<13 N) and soft core firmness perception (<35.5 N) receptively. The vertical bar represents the standard error of the firmness mean of 4 replicates of 15 fruit each.
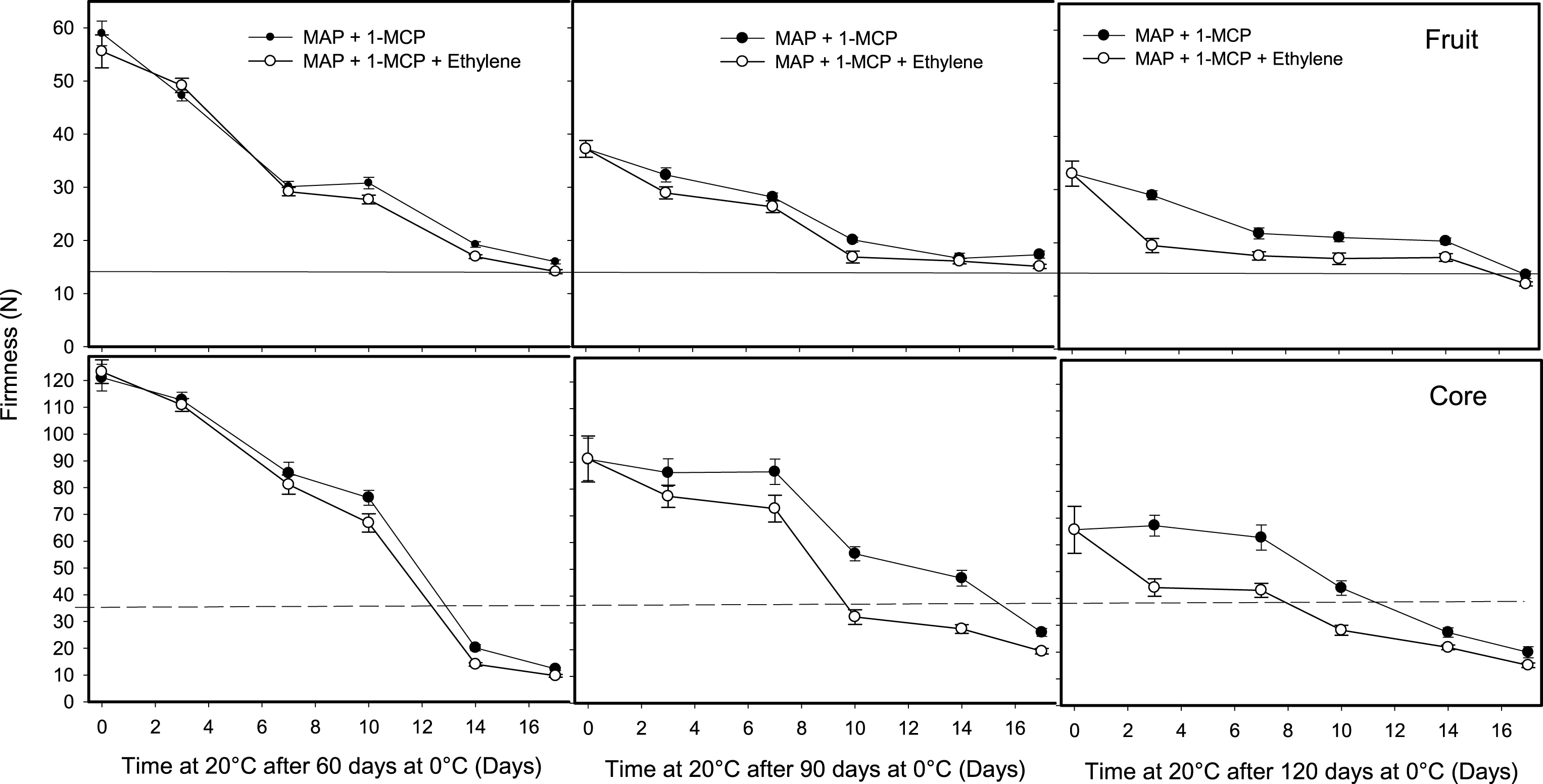
Fig.9
Effect of ethylene (100 μl l–1) application time (24, 48, 72 h) at a temperature of 20°C on kiwifruit tissue softening (whole fruit and core) during ripening at 20°C after 108 days storage in a controlled atmosphere (CA, 2% O2 and 5% CO2) and packed in perforated bags (PB) at 0°C for 30 days. The ethylene was applied after the fruit was removed from CA and further 30 days at 0°C and compared with fruit no- treated with ethylene and pre-ripened at 20°C for 24, 48 or 72 h. The solid and dotted horizontal lines indicate the ready to eat firmness value (<13 N) and soft core firmness perception (<35.5 N) receptively.
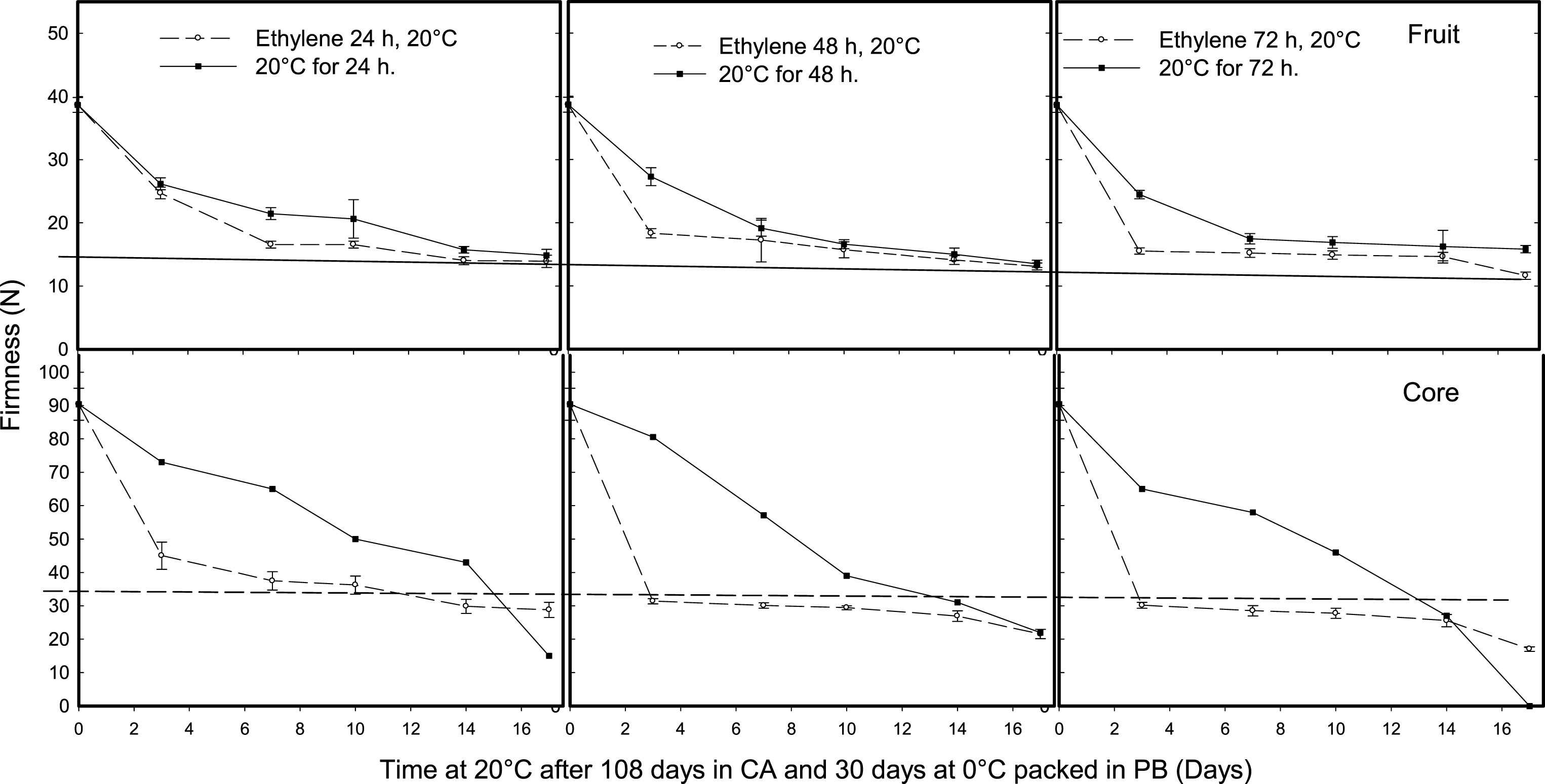
Fig.10
Firmness perception of soft kiwifruit (<10 N) with core tissue with different ranges of firmness. The means of eight replicates (panelists) followed by the same letters were not statistically significant according to Fisher’s least significant difference (LSD) test (P≤0.05). Horizontal line indicates the limit of perception of soft core tissue, firmness value <35.5 N).
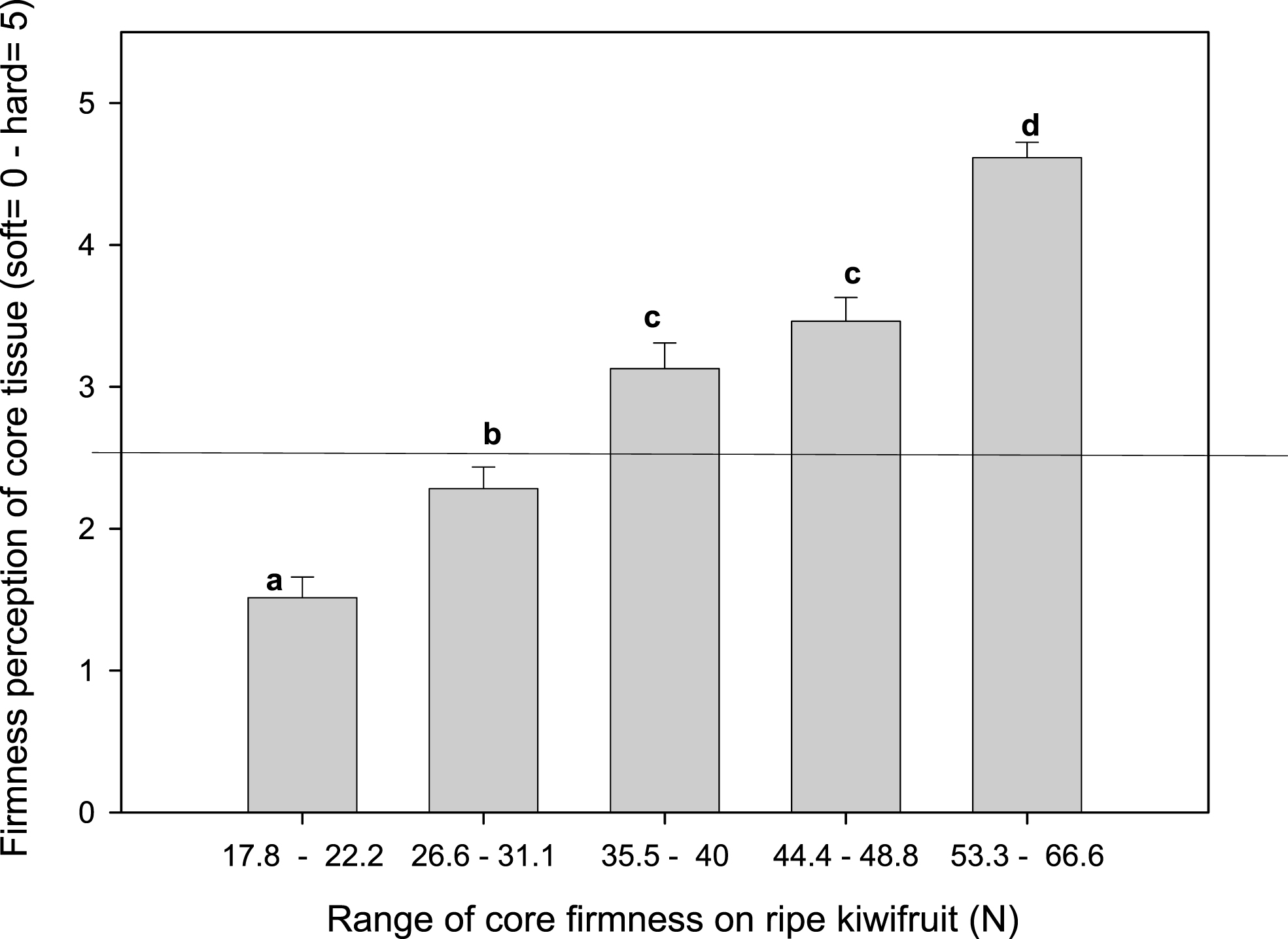
Table 1
Effect of 1-methylcyclopropene (1-MCP), modified atmosphere packaging (MAP) or combination of 1-MCP treated fruit in MAP on flesh firmness (N) of kiwifruit cv. ‘Hayward’ stored for 60, 90 or 120 d at 0°C
| Time in storage at 0°C | 1-MCP1 | Control2 | ||
| Days | PB3 | MAP4 | PB | MAP |
| 605 | 40.6 b | 57.9 d | 33.8 a | 52.1 c |
| 90 | 32.1 a | 48.1 b | 28.0 a | 34.3 a |
| 120 | 24.9 ab | 33.4 c | 19.6 a | 29.9 bc |
11-MCP (1 μl l–1) was applied in a 20°C chamber for 24 h. 2Control, the kiwifruit was maintained in a 20°C chamber without 1-MCP. 3PB, kiwifruit was packed in a high-density, 10-μ, 0.3% perforated polyethylene bag. 4MAP, fruit was packed in a low-density, 60-μ polyethylene modified atmosphere bag (MA, FF Kiwi, San Jorge Packaging). 5The means of four replicates followed by the same letters within each storage time at 0°C were not statistically significant according to Fisher's least significant difference (LSD) test (P≤0.05).
Table 2
Effect of ethylene (100 μl l–1) on titratable acidity (%) of ripe kiwifruit (<13 N) at 20°C packed in modified atmosphere packaging (MAP) or polyethylene perforated bags (PB) previously treated at harvest with 1-MCP (1 μl l–1)
| Time in storage | 1-MCP1 | Control2 | ||||||
| at 0°C (Days) | ||||||||
| PB4 | MAP5 | PB | MAP | |||||
| Ethylene3 | Control | Ethylene | Control | Ethylene | Control | Ethylene | Control | |
| 606 | 1.26 cd | 1.29 de | 1.23 bc | 1.31 e | 1.24 bc | 1.33 e | 1.02 a | 1.19 b |
| 90 | 1.37 c | 1.41 c | 1.17 a | 1.25 b | 1.19 a | 1.21 ab | 1.21 ab | 1.20 a |
| 120 | 1.30 f | 1.24 ef | 1.16 cd | 1.14 bcd | 1.20 de | 1.08 ab | 1.10 abc | 1.06 a |
1Ripe kiwifruit treated with 1-MCP and/or packed in a MA bag was evaluated after 14 days at 20°C and after 7 days for PB-packed fruit. 1-MCP, 1 μ1 l–1 was applied in a 20°C chamber for 24 h. 2Control, the kiwifruit was maintained in a 20°C chamber without 1-MCP. 3100 μ1 l–1 ethylene was applied for 12 h at 20°C. 4PB, fruit was packed in a high-density, 10-μ, 0.3% perforated polyethylene bag. 5MAP, fruit was packed in a low-density, 60-μ polyethylene modified atmosphere bag (MA, FF Kiwi, San Jorge Packaging). 6The means of four replicates followed by the same letters within each storage time at 0°C were not statistically significant according to Fisher’s least significant difference (LSD) test (P≤0.05).



