Amyloid-Tau-Neurodegeneration Profiles and Longitudinal Cognition in Sporadic Young-Onset Dementia
Abstract
We examined amyloid-tau-neurodegeneration biomarker effects on cognition in a Southeast-Asian cohort of 84 sporadic young-onset dementia (YOD; age-at-onset <65 years) patients. They were stratified into A+N+, A– N+, and A– N– profiles via cerebrospinal fluid amyloid-β1–42 (A), phosphorylated-tau (T), MRI medial temporal atrophy (neurodegeneration– N), and confluent white matter hyperintensities cerebrovascular disease (CVD). A, T, and CVD effects on longitudinal Mini-Mental State Examination (MMSE) were evaluated. A+N+ patients demonstrated steeper MMSE decline than A– N+ (β = 1.53; p = 0.036; CI 0.15:2.92) and A– N– (β = 4.68; p = 0.001; CI 1.98:7.38) over a mean follow-up of 1.24 years. Within A– N+, T– CVD+ patients showed greater MMSE decline compared to T+CVD– patients (β = – 2.37; p = 0.030; CI – 4.41:– 0.39). A+ results in significant cognitive decline, while CVD influences longitudinal cognition in the A– sub-group.
INTRODUCTION
The Amyloid-Tau-Neurodegeneration (ATN) classification enables an unbiased biomarker-based diagnosis of Alzheimer’s disease (AD), independent of cognitive status and temporal ordering of AD pathogenic mechanisms [1]. ATN biomarker status is defined by: A, amyloid-β deposition (amyloid-PET; cerebrospinal fluid [CSF]); T, tauopathy (tau-PET; CSF phosphorylated-tau); N, neurodegeneration (MRI; FDG-PET; CSF total-tau) [1–3]. Examining ATN biomarker profiles across diverse age-groups and geographies in clinical settings will inform model generalizability and need for revisions [4, 5]. Biomarker profiles may differ widely between young and elderly, given higher mixed brain pathology with increasing age [6]. While prior studies have illustrated greater amyloid-β deposition in young-onset compared to late-onset dementia, differences in biomarker-related cognitive trajectory in sporadic young-onset dementia need to be explored further, especially in Asian cohorts [7, 8]. Recent studies also indicate a low prevalence of APOE4 in Asia which in its association with amyloid positivity, may contribute to differences in biomarker-related disease trajectory and prevalence [9]. Moreover, a recent meta-analysis on the global prevalence of young-onset dementia illustrates the lack of data from Asia including southeast Asia, thus indicating the need for more studies from the region [10].
Indeed, AD pathology can co-exist with cerebrovascular disease (CVD) pathologies [11, 12]. Findings from our group demonstrate a high prevalence of CVD in Asians and hence the impact of CVD on biomarker influence on dementia is important to elucidate [13]. Additionally, prior studies show accelerated cognitive decline in A–N+ subjects as well as A+N– and A+N+ groups, but have largely focused on older individuals [14, 15]. Examining biomarker distribution among young-onset patients with age-at-onset below 65 years will help understand effects of age-at-onset on pathology and provide important insights into the ATN framework [4, 12].
Here, in a Southeast-Asian memory clinic cohort, we report preliminary ATN profiles and examine the contribution of A, T, and CVD on cognitive decline in sporadic young-onset dementia. Based on our prior work, we demonstrated that CVD moderated the influence of A on cognitive outcomes, but does not have a direct effect on cognition [16]. We thus hypothesized that while A+ patients will show the greatest cognitive decline, presence of CVD+ and T+ among A– patients will also contribute to cognitive decline.
METHODS
Participants and study design
Study patients were recruited from the Singapore Young Onset Dementia Cohort and Neurocognition, Imaging, and Biomarker Longitudinal Study in SingaporE research studies at the National Neuroscience Institute, Singapore between August 2016 and November 2020. Only consecutive patients who underwent structural MRI and consented for lumbar puncture for CSF Amyloid-β (1–42), phosphorylated tau (phospho-tau), and total tau (t-tau) were included for the present study. Clinical diagnosis of dementia was made by cognitive neurologists based on established diagnostic criteria [17–24]. Dementia patients included amnestic and non-amnestic dementia presentation and had a Clinical Dementia Rating of 1-2. Amnestic dementia included patients reporting a predominant impairment in memory function. Non-amnestic dementia included participants presenting with predominant language impairment, behavioral changes, or visual agnosia. Exclusion criteria included a history of alcohol or drug abuse, a current or known history of major depression, presence of comorbid neurodegenerative disease such as Parkinson’s disease, history of stroke, and presence of contraindications to MRI. In total, 84 patients were included in our study. All participants were aged below 65 years at symptom onset and classified as young-onset dementia [25].
Informed consent was sought from each patient according to the Declaration of Helsinki and local clinical research regulations. The study was granted approval by the Singhealth Centralized Review Board.
Neuropsychological assessment
Patients underwent a standardized battery of neuropsychological assessments administered by trained staff. Measures of global cognition included the Mini-Mental State Examination (MMSE) [26]. Of the 84 patients, 69 had at least one follow-up MMSE available, with the mean number of follow-up MMSE scores being 3.85 (median 3). Mean MMSE follow-up time was 1.24 years (median follow-up = 1.08 years). The remaining 15 patients were lost to follow-up either due to not being within the duration of study, change of doctor or passing away and thus did not have any follow-up MMSE available.
Imaging
Patients underwent neuroimaging assessments using either 1.5T MRI scanner (Philips Ingenia) or 3T Siemens Prisma Fit (Siemens, Erlangen, Germany). T1-weighted and FLAIR scans were used for visual rating of scans. Subject FLAIR scans were visually rated based on the modified Fazekas scale for white matter hyperintensity (WMH) severity [27]. Patients were classified as having confluent WMH and non-confluent WMH based on the Staals criteria [28]: a WMH rating of 3 in either periventricular and/or a rating of 2 or 3 in deep white matter regions in either hemisphere were assigned as confluent. Raters doing the MRI visual ratings were blinded to diagnosis and clinical information.
The neurodegeneration (N) profile was assigned based on the medial temporal lobe atrophy (MTA) scores based on the Schelten’s scale [29].
Cerebrospinal fluid analysis
All patients underwent a lumbar puncture for CSF collection and tested for levels of amyloid-β, total tau, and phosphorylated-tau proteins. ELISA immunoassays were used to process the CSF specimens collected in polypropylene tubes, in accordance with prescribed protocol and requirements (INNOTEST tTau Ag, INNOTEST PHOSPHO-TAU(181) and INNOTEST-AMYLOID(1–42); Innogenetics Inc., Alpharetta, GA) [30, 31].
APOE4 genotyping
Genomic DNA was extracted from peripheral blood using QIAamp® DNA Blood Maxi Kit (Qiagen GmbH, Hilden, Germany) as per standard protocol. Genotyping for APOE isoforms [rs429358 (ABI assay ID:C_3084793_20) was performed using TaqMan SNP genotyping assays on ABI 7900HT PCR system (Applied Biosystems, Foster City, CA). APOE genotype assignments were performed as described [32].
Classification of ATN profiles
ATN classification was assigned as CSF amyloid-β (1–42) (A), phosphorylated (phospho)-tau (T), and MTA (N) measurements using cut-offs of CSF amyloid-β (1–42)<550 pg/ml for A+, CSF phospho-tau>60 pg/ml for T+ [29, 33] and age-specific MTA positivity (N+) cut-offs based on previous studies with N+ defined as average MTA of ≥1 for individuals aged <65 [34]. Patients were CVD+ if their WMH visual ratings indicated presence of confluent WMH.
Statistical analyses
Demographic features involving continuous variables are described as mean (SD) and categorical variables are described as percentages. Comparison tests comprised independent samples T-test for continuous variables and Pearson’s chi-square analysis for categorical variables. Comparisons were carried out between the A+N+, A–N+, and A–N– groups.
We used linear mixed-effects models to assess the influence of pathological burden on the association between biomarkers (amyloid-β, MTA) and cognitive decline in MMSE scores.
In the first analysis set, longitudinal MMSE scores were the key dependent variable. The key independent variables comprised biomarker status comprising amyloid and neurodegeneration status as a categorical variable (A+N+, A–N+, A–N–), age-at-onset and an ANStatus * Time interaction term.
As our first set of analyses demonstrated that T had no effect on longitudinal cognition, in the second analysis, we assessed the specific influence of A+CVD–T± and CVD+A–T± on longitudinal cognition. Here, longitudinal MMSE scores were the key dependent variable and BiomarkerStatus * Time was the interaction term.
In an additional analysis, we specifically evaluated the effects of CVD and T among A–N+ patients on longitudinal MMSE scores. The key independent variables comprised biomarker status within A–N+ group, i.e., T+CVD–, T–CVD+ or Others (T–CVD–), age-at-onset and a A–N+Group*Time interaction term.
Baseline age, sex, education years, and baseline MMSE were included as covariates for all analyses. The random intercept and slopes were modelled at the individual subject level.
All statistical analyses were performed using R 3.6.3 (R Core Team, 2014) with Rstudio (Rstudio Team, 2012) with the lme4, lmertest, sjPlot, ggforce, ggraph, ggthemes, and ggplot2 packages.
RESULTS
ATN biomarker characteristics in sporadic young onset dementia
Key summary characteristics of the cohort are provided in Table 1.
Table 1
Patient characteristics of sporadic young-onset dementia
| A+N+ | A–N+ | A–N– | p | |
| (N = 29) | (N = 47) | (N = 8) | ||
| Age (mean, SD) | 57.7 (4.8) | 57.0 (5.5) | 56.5 (6.3) | 0.806 |
| Range: 35.4–64.5 years | ||||
| Sex (F %) | 19 (65.5%) | 20 (42.6%) | 4 (50%) | 0.150 |
| Education years (mean, SD, n = 82) | 12.1 (4.2) | 11.4 (4.2) | 13.3 (6.4) | 0.539 |
| Range: 1–25 years | ||||
| Clinical Dementia Diagnosis: | 0.200 | |||
| Amnestic | 23 (79.3%) | 28 (59.6%) | 5 (62.5%) | |
| Non-amnestic | 6 (20.7%) | 19 (40.4%) | 3 (37.5%) | |
| Confluent WMH (n %) | 11/29 (37.9%) | 26/47 (55.3%) | 4/7 (57.1%) | 0.304 |
| APOE4 carriers (n = 63, %) | 9/22 (40.9%) | 7/37 (18.9%) | 0/4 (0%) | 0.083 |
| Baseline MMSE (mean, SD, n = 81) | 18.3 (6.2) | 21.9 (6.7) | 21.7 (6.5) | 0.068 |
| Range: 6–30 | ||||
| CSF amyloid-β levels | 391.1 (78.7)b,c | 899.3 (315.3) | 944.5 (409.2) | <0.001 |
| CSF phospho-tau levels | 98.5 (62.8)b | 54.9 (33.2) | 65.3 (35.5) | 0.001 |
| CSF total-tau levels | 827.2 (466.5)b | 486.5 (444.5) | 566.5 (450.2) | 0.008 |
Values represent mean (SD) unless otherwise stated. Superscript letters indicate whether group mean was significantly different compared with bA–N+, cA–N– groups. A, amyloid; T, tau; N, neurodegeneration; F, Female; WMH, white matter hyperintensities; APOE4, apolipoprotein epsilon 4; MMSE, Mini-Mental State Examination; CSF, cerebrospinal fluid.
Based on the A and N status, patient breakdown included A+N+ (n = 29, 34.5%), A–N+ (n = 47, 55.9%), and A–N– (n = 8, 9.6%). Within the A–N+ group, sixteen patients (34.0%) were T+, nineteen (40.4%) were CVD+ and twelve (25.6%) were neither CVD+ nor T+.
A+ in amnestic dementia was 41.1% while in non-amnestic dementia was 21.4%. Among non-amnestic dementia, 35.7% were T+ and 57.1% were CVD+. Eight patients were A–N–, three of whom had elevated total-tau, five were T+, and four were CVD+. The proportion of APOE4 carriers was highest in the A+N+ group at 40.9%.
Association between ATN with cognitive decline in sporadic young-onset dementia
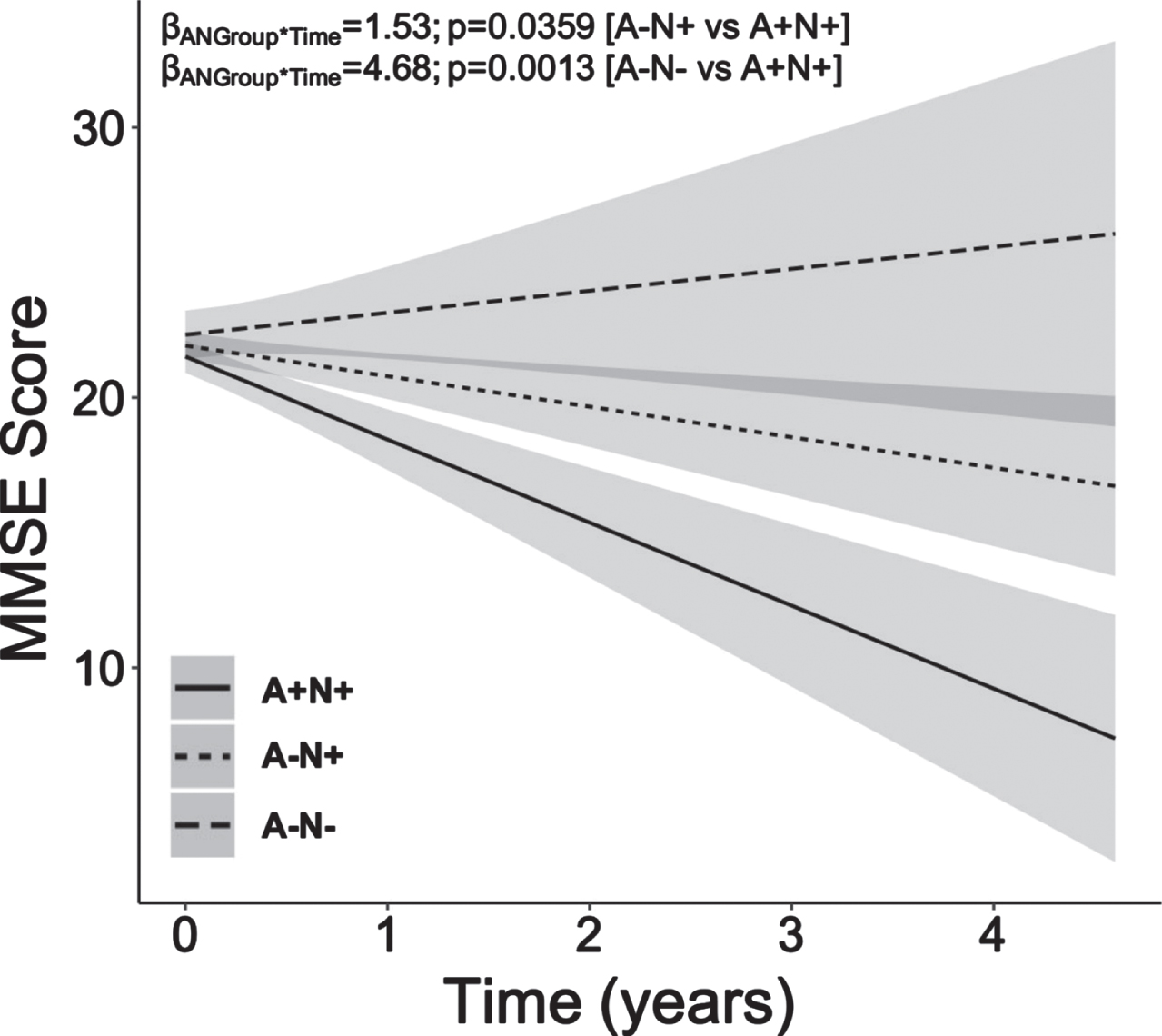
Young-onset A+N+ patients showed steeper decline in MMSE scores over mean follow-up time of 1.24 years (Fig. 1; median follow-up = 1.08 years; follow-up visits range = 2 to 9; average follow-up visits = 3.85) compared to both A–N+ (ANGroup*Time: β = 1.53, p = 0.0359, CI 0.15:2.92) and A–N– counterparts (ANGroup*Time: β = 4.68, p = 0.0013, CI 1.98:7.38).
Fig. 1
Young-onset amyloid-positive neurodegeneration-positive dementia patients exhibit greater longitudinal decline in MMSE scores compared to young-onset amyloid-negative neurodegeneration-positive and amyloid-negative neurodegeneration-negative dementia patients. Young-onset patients with high amyloid-β burden (A+) and neurodegeneration (N+), that is, high MTA burden, showed steeper decline in MMSE scores over time when compared to their young-onset A–N+ and A–N– counterparts. Baseline age, sex, education years and baseline MMSE were included as covariates. MMSE, Mini-Mental State Examination; A, amyloid-β; N, neurodegeneration; MTA, medial temporal atrophy.
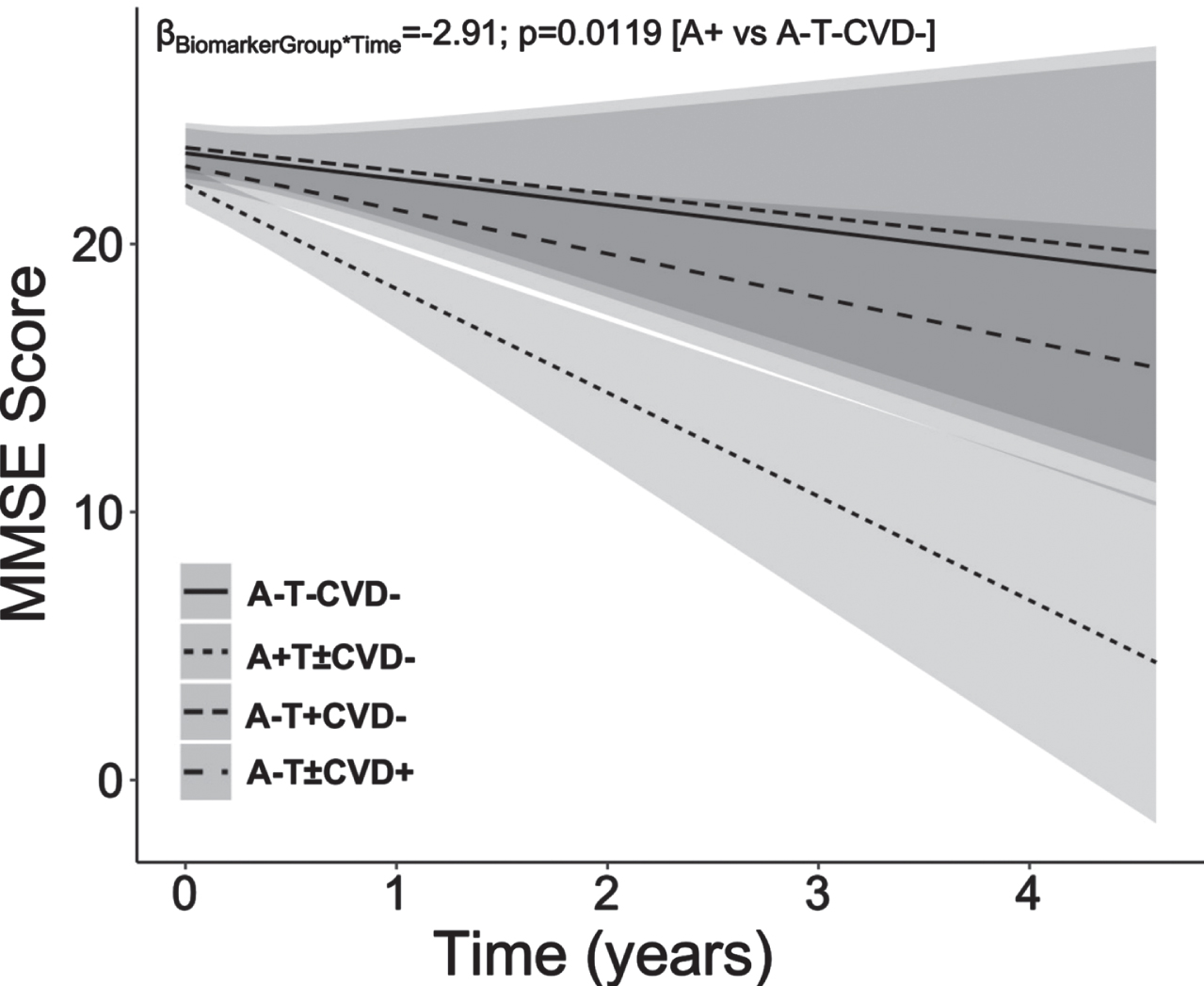
In the second analyses set, young-onset A+(T±CVD–) patients showed the steepest decline in MMSE scores compared to A–T– CVD– patients (BiomarkerStatus*Time: β = –2.91, p = 0.0119, CI – 5.03:– 0.79; Fig. 1). CVD+(A–T±) patients, also showed MMSE decline, although this did not reach significance. T+(A–CVD–) patients demonstrated an MMSE trajectory comparable to that of A–T– CVD– patients (Fig. 2).
Fig. 2
Young-onset sporadic dementia with amyloid-β pathology show steepest decline in MMSE scores compared to patients without amyloid-β, phosphorylated tau or cerebrovascular disease pathology. A+ Young-onset dementia patients showed significant decline in MMSE score than their A–T– CVD– counterparts. However, predominant CVD pathology and predominant T pathology did not significantly influence cognitive decline in comparison to normal biomarker A–T– CVD– patients. Baseline age, sex, education years and baseline MMSE were included as covariates. MMSE, Mini-Mental State Examination; A, amyloid-β; CVD, cerebrovascular disease; T, phospho-tau.
Further sub-group analyses within A–N+ revealed that CVD+T– patients showed steeper longitudinal MMSE decline compared to T+CVD– counterparts (A–N+Group*Time: β = –2.37, p = 0.0298, CI –4.41:–0.39). There were no differences in MMSE decline between CVD+ and others (T–CVD–)group.
DISCUSSION
In this study of sporadic young onset dementia patients, we demonstrate that A–N+ accounted for the largest ATN group with A+ present only in 34.5%. Our findings further demonstrate that among those with A–N+ profile, CVD + accounted for the largest group. Amyloid pathology resulted in significant cognitive decline in this young sporadic dementia cohort, with CVD only influencing longitudinal cognition among patients without amyloid-β pathology. Phospho-tau pathology in the absence of amyloid-β or CVD pathology had no impact on longitudinal cognition.
The overall prevalence of A+ in our cohort was 34.5% which was lower in comparison to other worldwide cohorts, wherein A+ frequency among dementia patients ranged between 66% and 96.9% [5, 35]. Importantly, prior memory clinic cohorts comprised predominantly of older patients with an average age between 63.3 and 75.6 years, while our cohort had an average age of 57.2 years. Prior studies have indeed shown linear increases in amyloid-β burden with increasing age [36, 37]. While one may argue that younger patients are more likely to have non-amnestic forms of dementia and hence would be less likely to be A+, we demonstrate in our cohort that even among young patients with amnestic dementia, A+ frequency was only 41.1%. In addition to the lower average age, the frequency of APOE4 carriers in our cohort was low at 25.4% compared to 55.2% in a Korean memory clinic cohort and 46.9% in a European memory clinic study [5, 35]. In line with our findings, past studies have shown lower APOE4 frequency among Southeast Asians [9]. Since APOE4 has been correlated to A+, a lower APOE4 frequency among Asians may reflect a genetic contribution to lower frequency of A+ among Southeast-Asian patients with dementia [36]. Thus, further studies with broader age ranges and varied ethnicities are required to understand the contribution of APOE4 on A+ rates in sporadic young onsetdementia.
Additionally, A–N+ often referred to as suspected non-Alzheimer’s pathology (SNAP) was the largest group (55.9%) in our young cohort. Compared to other older Asian and western cohorts, our cohort showed a higher prevalence of A–N+ patients [5, 35]. In the Alzheimer’s Biomarkers in Daily Practice cohort with mean age of 63.3, the prevalence of A–N+ in dementia patients was 28%, while in the Samsung Medical Centre study, with mean age of 75.6, the prevalence of A–N+ in dementia patients was 1.5% [5, 35]. Our findings highlight the importance of non-amyloid pathologies in furthering our understanding of the pathobiology and trajectory in younger dementia patients [5, 35, 38]. Within this group of A–N+, we demonstrate that CVD+ patients comprised the largest group and displayed MMSE decline. While CVD is often reported to be more prevalent in older dementia populations, notably, we report that CVD is prevalent in young A– patients. In this study, CVD+ was defined by the presence of confluent WMH. Confluent WMH has been shown to correlate to lacune count and suggested to represent active on-going occlusion of arterioles with greater clinical and cognitive consequences compared to non-confluent WMH [28, 39–41]. Confluent WMH has also been shown to be associated with functional connectivity changes and greater cognitive decline even in older patients [42, 43]. Additionally, prior studies have illustrated that WMH predict longitudinal increases in CSF tau [44]. Recent findings in dementia patients provide evidence for influence of CVD pathology on AD expression, likely through promotion of tau pathology, independent of amyloid-β [45]. In this regard, our findings of CVD+ dementia patients showing worse cognitive decline compared to T+ patients in the absence of amyloidosis may reflect a stage wherein CVD has not resulted in significant tau pathology. The lack of tau effects on cognitive decline in young-onset dementia were evident through our findings of no negative effects on MMSE decline. Indeed, while previous studies have found tau pathology to be associated with cognitive decline, this was not the case in our study in comparison to A+ subjects. We offer two possible explanations for our finding. Firstly, most prior studies that showed a relationship between tau pathology and cognitive decline were in the setting of amyloid positivity, wherein amyloid positivity was a pre-requisite for tau to impact cognition [46, 47]. Previous studies indicate that cognitive decline seen in primary tauopathy is usually in the setting of tau and CVD [48, 49]. In our study since we separated the presence of tau pathology from the presence of CVD, our findings are consistent with reports that show that pure tauopathy results in less several cognitive decline compared to presence of AD neuropathology [49, 50]. Therefore, this may underlie the absence of significant cognitive decline in our sample of tau positive dementiapatients.
Our findings demonstrate that young-onset dementia patients show steeper decline in global cognition in the presence of elevated amyloid-β and neurodegeneration burden when compared to A– counterparts. Such associations may be reflective of the pathological effect of amyloid-β burden in young patients [8]. Importantly, our findings of steeper cognitive decline in A+ young-onset patients are also in line with studies indicating that young-onset AD is associated with a more aggressive disease course [51, 52]. Possible mechanisms underlying steeper cognitive decline in A+ young-onset patients may involve greater neuronal loss and hypometabolism [53, 54]. Younger patients may be particularly sensitive to neural insults as a result of aggressive increases in elevated amyloid-β levels.
The limitations of this study include the relatively small cohort size; however, this being a report of ATN in young sporadic dementia, and first from Southeast Asia adds important biomarker knowledge in this population. Another limitation is the relatively short follow-up period, however even with the existing follow-up duration, clear evidence of differential cognitive trajectories based on ATN profiles were demonstrated. The use of visual ratings for CVD burden instead of quantitative WMH methods is also a limitation of our study. Additionally, we were not able to quantify TDP or alpha-synuclein pathology and did not have complete information on duration ofdisease.
In summary, in a cohort of sporadic young-onset dementia, we show a high prevalence of non-amyloid pathology (A–N+), a majority of which had high CVD pathology and elevated phospho-tau. Nonetheless, A+N+ patients showed steepest cognitive decline compared to their A–N+ and A–N– counterparts. Within the A–N+ group, presence of CVD resulted in poor cognitive outcomes. Our findings highlight the need for further studies of ATN in diverse age groups and ethnicities.
ACKNOWLEDGMENTS
This study is supported by the Ministry of Education, Singapore, under its MOE AcRF Tier 3 Award MOE2017-T3-1-002, National Medical Research Council (NMRC), Singapore, under its Clinician Scientist Award (MOH-CSAINV18nov-0007) and Clinician Scientist Individual Research Grant (NMRC/CIRG/14MAY025).
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/22-0448r1).
REFERENCES
[1] | Jack CR , Bennett DA , Blennow K , Carrillo MC , Feldman HH , Frisoni GB , Hampel H , Jagust WJ , Johnson KA , Knopman DS , Petersen RC , Scheltens P , Sperling RA , Dubois B ((2016) ) A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 87: , 539–547. |
[2] | Jack CR , Bennett DA , Blennow K , Carrillo MC , Dunn B , Haeberlein SB , Holtzman DM , Jagust W , Jessen F , Karlawish J , Liu E , Molinuevo JL , Montine T , Phelps C , Rankin KP , Rowe CC , Scheltens P , Siemers E , Snyder HM , Sperling R , Elliott C , Masliah E , Ryan L , Silverberg N ((2018) ) NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement 14: , 535–562. |
[3] | Nelson PT , Head E , Schmitt FA , Davis PR , Neltner JH , Jicha GA , Abner EL , Smith CD , Van Eldik LJ , Kryscio RJ , Scheff SW ((2011) ) Alzheimer’s disease is not “brain aging”: Neuropathological, genetic, and epidemiological human studies. Acta Neuropathol 121: , 571–587. |
[4] | Knopman DS , Haeberlein SB , Carrillo MC , Hendrix JA , Kerchner G , Margolin R , Maruff P , Miller DS , Tong G , Tome MB , Murray ME , Nelson PT , Sano M , Mattsson N , Sultzer DL , Montine TJ , Jack CR , Kolb H , Petersen RC , Vemuri P , Canniere MZ , Schneider JA , Resnick SM , Romano G , van Harten AC , Wolk DA , Bain LJ , Siemers E ((2018) ) The National Institute on Aging and the Alzheimer’s Association Research Framework for Alzheimer’s disease: Perspectives from the Research Roundtable. Alzheimers Dement 14: , 563–575. |
[5] | Altomare D , De Wilde A , Ossenkoppele R , Pelkmans W , Bouwman F , Groot C , Van Maurik I , Zwan M , Yaqub M , Barkhof F , Van Berckel BN , Teunissen CE , Frisoni GB , Scheltens P , Van Der Flier WM ((2019) ) Applying the ATN scheme in a memory clinic population: The ABIDE project. Neurology 93: , e1635–e1646. |
[6] | Schneider JA , Arvanitakis Z , Leurgans SE , Bennett DA ((2009) ) The neuropathology of probable Alzheimer disease and mild cognitive impairment. Ann Neurol 66: , 200–208. |
[7] | Cho H , Seo SW , Kim JH , Suh MK , Lee JH , Choe YS , Lee KH , Kim JS , Kim GH , Noh Y , Ye BS , Kim HJ , Yoon CW , Chin J , Na DL ((2013) ) Amyloid deposition in early onset versus late onset Alzheimer’s disease. J Alzheimers Dis 35: , 813–821. |
[8] | Marshall GA , Fairbanks LA , Tekin S , Vinters HV , Cummings JL ((2007) ) Early-onset Alzheimer’s disease is associated with greater pathologic burden. J Geriatr Psychiatry Neurol 20: , 29–33. |
[9] | Gao Q , Gwee X , Feng L , Nyunt MSZ , Feng L , Collinson SL , Chong MS , Lim WS , Lee TS , Yap P , Yap KB , Ng TP ((2018) ) Mild cognitive impairment reversion and progression: Rates and predictors in community-living older persons in the Singapore Longitudinal Ageing Studies Cohort. Dement Geriatr Cogn Dis Extra 8: , 226–237. |
[10] | Hendriks S , Peetoom K , Bakker C , Van Der Flier WM , Papma JM , Koopmans R , Verhey FRJ , De Vugt M , Köhler S , Withall A , Parlevliet JL , Uysal-Bozkir Ö , Gibson RC , Neita SM , Nielsen TR , Salem LC , Nyberg J , Lopes MA , Dominguez JC , De Guzman MF , Egeberg A , Radford K , Broe T , Subramaniam M , Abdin E , Bruni AC , Di Lorenzo R , Smith K , Flicker L , Mol MO , Basta M , Yu D , Masika G , Petersen MS , Ruano L ((2021) ) Global Prevalence of young-onset dementia: A systematic review and meta-analysis. JAMA Neurol 78: , 1080–1090. |
[11] | Vemuri P , Lesnick TG , Przybelski SA , Knopman DS , Lowe VJ , Graff-Radford J , Roberts RO , Mielke MM , Machulda MM , Petersen RC , Jack CR ((2017) ) Age, vascular health, and Alzheimer disease biomarkers in an elderly sample. Ann Neurol 82: , 706–718. |
[12] | Gauthier S , Zhang H , Ng KP , Pascoal TA , Rosa-Neto P ((2018) ) Impact of the biological definition of Alzheimer’s disease using amyloid, tau and neurodegeneration (ATN): What about the role of vascular changes, inflammation, Lewy body pathology? Transl Neurodegener 7: , 12. |
[13] | Lam BYK , Yiu B , Ampil E , Chen CLH , Dikot Y , Dominguez JC , Ganeshbhai PV , Hilal S , Kandiah N , Kim SY , Lee JY , Ong AP , Senanarong V , Leung KT , Wang H , Yang YH , Yong T , Arshad F , Alladi S , Wong S , Ko H , Lau AYL , Mok VCT ((2021) ) High burden of cerebral white matter lesion in 9 Asian cities. Sci Rep 11: , 11587. |
[14] | Vos SJB , Verhey F , Frölich L , Kornhuber J , Wiltfang J , Maier W , Peters O , Rüther E , Nobili F , Morbelli S , Frisoni GB , Drzezga A , Didic M , Van Berckel BNM , Simmons A , Soininen H , Kłoszewska I , Mecocci P , Tsolaki M , Vellas B , Lovestone S , Muscio C , Herukka SK , Salmon E , Bastin C , Wallin A , Nordlund A , De Mendonça A , Silva D , Santana I , Lemos R , Engelborghs S , Van Der Mussele S , Freund-Levi Y , Wallin ÅK , Hampel H , Van Der Flier W , Scheltens P , Visser PJ ((2015) ) Prevalence and prognosis of Alzheimer’s disease at the mild cognitive impairment stage. Brain 138: , 1327–1338. |
[15] | Burnham SC , Bourgeat P , Doré V , Savage G , Brown B , Laws S , Maruff P , Salvado O , Ames D , Martins RN , Masters CL , Rowe CC , Villemagne VL ((2016) ) Clinical and cognitive trajectories in cognitively healthy elderly individuals with suspected non-Alzheimer’s disease pathophysiology (SNAP) or Alzheimer’s disease pathology: A longitudinal study. Lancet Neurol 15: , 1044–1053. |
[16] | Yatawara C , Ng KP , Cristine Guevarra A , Wong B , Yong TT , Kandiah N ((2020) ) Small vessel disease and associations with cerebrospinal fluid amyloid, tau, and neurodegeneration (ATN) biomarkers and cognition in young onset dementia. J Alzheimers Dis 77: , 1305–1314. |
[17] | Petersen RC ((2004) ) Mild cognitive impairment as a diagnostic entity. J Intern Med 256: , 183–194. |
[18] | McKhann GM , Knopman DS , Chertkow H , Hyman BT , Jack CR , Kawas CH , Klunk WE , Koroshetz WJ , Manly JJ , Mayeux R , Mohs RC , Morris JC , Rossor MN , Scheltens P , Carrillo MC , Thies B , Weintraub S , Phelps CH ((2011) ) The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7: , 263–269. |
[19] | McKeith IG , Dickson DW , Lowe J , Emre M , O’Brien JT , Feldman H , Cummings J , Duda JE , Lippa C , Perry EK , Aarsland D , Arai H , Ballard CG , Boeve B , Burn DJ , Costa D , Del Ser T , Dubois B , Galasko D , Gauthier S , Goetz CG , Gomez-Tortosa E , Halliday G , Hansen LA , Hardy J , Iwatsubo T , Kalaria RN , Kaufer D , Kenny RA , Korczyn A , Kosaka K , Lee VMY , Lees A , Litvan I , Londos E , Lopez OL , Minoshima S , Mizuno Y , Molina JA , Mukaetova-Ladinska EB , Pasquier F , Perry RH , Schulz JB , Trojanowski JQ , Yamada M ((2005) ) Diagnosis and management of dementia with Lewy bodies: Third report of the DLB consortium. Neurology 65: , 1863–1872. |
[20] | Neary D , Snowden JS , Gustafson L , Passant U , Stuss D , Black S , Freedman M , Kertesz A , Robert PH , Albert M , Boone K , Miller BL , Cummings J , Benson DF ((1998) ) Frontotemporal lobar degeneration: A consensus on clinical diagnostic criteria. Neurology 51: , 1546–1554. |
[21] | Román GC , Tatemichi TK , Erkinjuntti T , Cummings JL , Masdeu JC , Garcia JH , Amaducci L , Orgogozo JM , Brun A , Hofman A , Moody DM , O’Brien MD , Yamaguchi T , Grafman J , Drayer BP , Bennett DA , Fisher M , Ogata J , Kokmen E , Bermejo F , Wolf PA , Gorelick PB , Bick KL , Pajeau AK , Bell MA , Decarli C , Culebras A , Korczyn AD , Bogousslavsky J , Hartmann A , Scheinberg P ((1993) ) Vascular dementia: Diagnostic criteria for research studies: Report of the NINDS-AIREN International Workshop. Neurology 43: , 250–260. |
[22] | Albert MS , DeKosky ST , Dickson D , Dubois B , Feldman HH , Fox NC , Gamst A , Holtzman DM , Jagust WJ , Petersen RC , Snyder PJ , Carrillo MC , Thies B , Phelps CH ((2011) ) The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7: , 270–279. |
[23] | Gorno-Tempini ML , Hillis AE , Weintraub S , Kertesz A , Mendez M , Cappa SF , Ogar JM , Rohrer JD , Black S , Boeve BF , Manes F , Dronkers NF , Vandenberghe R , Rascovsky K , Patterson K , Miller BL , Knopman DS , Hodges JR , Mesulam MM , Grossman M ((2011) ) Classification of primary progressive aphasia and its variants. Neurology 76: , 1006–1014. |
[24] | Boeve BF , Lang AE , Litvan I ((2003) ) Corticobasal degeneration and its relationship to progressive supranuclear palsy and frontotemporal dementia. Ann Neurol 54: (Suppl 5), S15–S19. |
[25] | Alzheimer’s Association ((2020) ) 2020 Alzheimer’s disease facts and figures. Alzheimers Dement 16: , 391–460. |
[26] | Feng L , Chong MS , Lim WS , Ng TP ((2012) ) The modified mini-mental state examination test: Normative data for Singapore Chinese older adults and its performance in detecting early cognitive impairment. Singapore Med J 53: , 458–462. |
[27] | Fazekas F , Chawluk JB , Alavi A ((1987) ) MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging. Am J Neuroradiol 149: , 351–356. |
[28] | Staals J , Makin SDJ , Doubal FN , Dennis MS , Wardlaw JM ((2014) ) Stroke subtype, vascular risk factors, and total MRI brain small-vessel disease burden. Neurology 83: , 1228–1234. |
[29] | Scheltens P , Kuiper M , Ch Wolters E , Barkhof F , Valk J , Weinsten HC , Leys D , Vermersch P , Huglo D , Steinling M ((1992) ) Atrophy of medial temporal lobes on MRI in “probable” Alzheimer’s disease and normal ageing: Diagnostic value and neuropsychological correlates. J Neurol Neurosurg Psychiatry 55: , 967–972. |
[30] | Vanmechelen E , Vanderstichele H , Davidsson P , Van Kerschaver E , Van Der Perre B , Sjögren M , Andreasen N , Blennow K ((2000) ) Quantification of tau phosphorylated at threonine 181 in human cerebrospinal fluid: A sandwich ELISA with a synthetic phosphopeptide for standardization. Neurosci Lett 285: , 49–52. |
[31] | Vanderstichele H , Van Kerschaver E , Hesse C , Davidsson P , Buyse MA , Andreasen N , Minthon L , Wallin A , Blennow K , Vanmechelen E ((2000) ) Standardization of measurement of β-amyloid (1-42) in cerebrospinal fluid and plasma. Amyloid 7: , 245–258. |
[32] | Koch W , Ehrenhaft A , Griesser K , Pfeufer A , Müller J , Schömig A , Kastrati A ((2002) ) TaqMan systems for genotyping of disease-related polymorphisms present in the gene encoding apolipoprotein E. Clin Chem Lab Med 40: , 1123–1131. |
[33] | Blennow K , Chen C , Cicognola C , Wildsmith KR , Manser PT , Sanabria Bohorquez SM , Zhang Z , Xie B , Peng J , Hansson O , Kvartsberg H , Portelius E , Zetterberg H , Lashley T , Brinkmalm G , Kerchner GA , Weimer RM , Ye K , Höglund K ((2020) ) Cerebrospinal fluid tau fragment correlates with tau PET: A candidate biomarker for tangle pathology. Brain 143: , 650–660. |
[34] | Rhodius-Meester HFM , Benedictus MR , Wattjes MP , Barkhof F , Scheltens P , Muller M , van der Flier WM ((2017) ) MRI visual ratings of brain atrophy and white matter hyperintensities across the spectrum of cognitive decline are differently affected by age and diagnosis. Front Aging Neurosci 9: , 117. |
[35] | Lee J , Jang H , Kang SH , Kim J , Kim JS , Kim JP , Kim HJ , Seo SW , Na DL ((2020) ) Cerebrospinal fluid biomarkers for the diagnosis and classification of Alzheimer’s disease spectrum. J Korean Med Sci 35: , e361. |
[36] | Jansen WJ , Ossenkoppele R , Knol DL , Tijms BM , Scheltens P , Verhey FRJ , Visser PJ , Aalten P , Aarsland D , Alcolea D , Alexander M , Almdahl IS , Arnold SE , Baldeiras I , Barthel H , Van Berckel BNM , Bibeau K , Blennow K , Brooks DJ , Van Buchem MA , Camus V , Cavedo E , Chen K , Chetelat G , Cohen AD , Drzezga A , Engelborghs S , Fagan AM , Fladby T , Fleisher AS , Van Der Flier WM , Ford L , Forster S , Fortea J , Foskett N , Frederiksen KS , Freund-Levi Y , Frisoni GB , Froelich L , Gabryelewicz T , Gill KD , Gkatzima O , Gomez-Tortosa E , Gordon MF , Grimmer T , Hampel H , Hausner L , Hellwig S , Herukka SK , Hildebrandt H , Ishihara L , Ivanoiu A , Jagust WJ , Johannsen P , Kandimalla R , Kapaki E , Klimkowicz-Mrowiec A , Klunk WE , Kohler S , Koglin N , Kornhuber J , Kramberger MG , Van Laere K , Landau SM , Lee DY , De Leon M , Lisetti V , Lleo A , Madsen K , Maier W , Marcusson J , Mattsson N , De Mendonca A , Meulenbroek O , Meyer PT , Mintun MA , Mok V , Molinuevo JL , Mollergard HM , Morris JC , Mroczko B , Van Der Mussele S , Na DL , Newberg A , Nordberg A , Nordlund A , Novak GP , Paraskevas GP , Parnetti L , Perera G , Peters O , Popp J , Prabhakar S , Rabinovici GD , Ramakers IHGB , Rami L , De Oliveira CR , Rinne JO , Rodrigue KM , Rodriguez-Rodriguez E , Roe CM , Rot U , Rowe CC , Ruther E , Sabri O , Sanchez-Juan P , Santana I , Sarazin M , Schroder J , Schutte C , Seo SW , Soetewey F , Soininen H , Spiru L , Struyfs H , Teunissen CE , Tsolaki M , Vandenberghe R , Verbeek MM , Villemagne VL , Vos SJB , Van Waalwijk Van Doorn LJC , Waldemar G , Wallin A , Wallin AK , Wiltfang J , Wolk DA , Zboch M , Zetterberg H ((2015) ) Prevalence of cerebral amyloid pathology in persons without dementia: A meta-analysis. JAMA 313: , 1924–1938. |
[37] | Rodrigue KM , Kennedy KM , Devous MD , Rieck JR , Hebrank AC , Diaz-Arrastia R , Mathews D , Park DC ((2012) ) β-amyloid burden in healthy aging: Regional distribution and cognitive consequences. Neurology 78: , 387–395. |
[38] | Mohamed Lafirdeen AS , Cognat E , Sabia S , Hourregue C , Lilamand M , Dugravot A , Bouaziz-Amar E , Laplanche JL , Hugon J , Singh-Manoux A , Paquet C , Dumurgier J ((2019) ) Biomarker profiles of Alzheimer’s disease and dynamic of the association between cerebrospinal fluid levels of β-amyloid peptide and tau. PLoS One 14: , e0217026. |
[39] | Van Dijk EJ , Prins ND , Vrooman HA , Hofman A , Koudstaal PJ , Breteler MMB ((2008) ) Progression of cerebral small vessel disease in relation to risk factors and cognitive consequences: Rotterdam scan study. Stroke 39: , 2712–2719. |
[40] | Ghaznawi R , Geerlings MI , Jaarsma-Coes MG , Zwartbol MHT , Kuijf HJ , van der Graaf Y , Witkamp TD , Hendrikse J , de Bresser J ((2019) ) The association between lacunes and white matter hyperintensity features on MRI: The SMART-MR study. J Cereb Blood Flow Metab 39: , 2486–2496. |
[41] | Silbert LC , Dodge HH , Perkins LG , Sherbakov L , Lahna D , Erten-Lyons D , Woltjer R , Shinto L , Kaye JA ((2012) ) Trajectory of white matter hyperintensity burden preceding mild cognitive impairment. Neurology 79: , 741–747. |
[42] | Kumar D , Vipin A , Wong B , Ng KP , Kandiah N ((2020) ) Differential effects of confluent and nonconfluent white matter hyperintensities on functional connectivity in mild cognitive impairment. Brain Connect 10: , 547–554. |
[43] | Heng LC , Lim SH , Foo H , Kandiah N ((2021) ) Confluent white matter in progression to alzheimer dementia. Alzheimer Dis Assoc Disord 35: , 8–13. |
[44] | Tosto G , Zimmerman ME , Hamilton JL , Carmichael OT , Brickman AM ((2015) ) The effect of white matter hyperintensities on neurodegeneration in mild cognitive impairment. Alzheimers Dement 11: , 1510–1519. |
[45] | Kim HJ , Park S , Cho H , Jang YK , Lee JS , Jang H , Kim Y , Kim KW , Ryu YH , Choi JY , Moon SH , Weiner MW , Jagust WJ , Rabinovici GD , DeCarli C , Lyoo CH , Na DL , Seo SW ((2018) ) Assessment of extent and role of tau in subcortical vascular cognitive impairment using 18F-AV1451 positron emission tomography imaging. JAMA Neurol 75: , 999–1007. |
[46] | Bejanin A , Schonhaut DR , La Joie R , Kramer JH , Baker SL , Sosa N , Ayakta N , Cantwell A , Janabi M , Lauriola M , O’Neil JP , Gorno-Tempini ML , Miller ZA , Rosen HJ , Miller BL , Jagust WJ , Rabinovici GD ((2017) ) Tau pathology and neurodegeneration contribute to cognitive impairment in Alzheimer’s disease. Brain 140: , 3286–3300. |
[47] | Chen Y-H , Lin R-R , Huang H-F , Xue Y-Y , Tao Q-Q ((2022) ) Microglial activation, tau pathology, and neurodegeneration biomarkers predict longitudinal cognitive decline in Alzheimer’s disease continuum. Front Aging Neurosci 14: , 848180. |
[48] | Iida MA , Farrell K , Walker JM , Richardson TE , Marx GA , Bryce CH , Purohit D , Ayalon G , Beach TG , Bigio EH , Cortes EP , Gearing M , Haroutunian V , McMillan CT , Lee EB , Dickson DW , McKee AC , Stein TD , Trojanowski JQ , Woltjer RL , Kovacs GG , Kofler JK , Kaye J , White CL , Crary JF ((2021) ) Predictors of cognitive impairment in primary age-related tauopathy: An autopsy study. Acta Neuropathol Commun 9: , 134. |
[49] | Teylan M , Mock C , Gauthreaux K , Chen YC , Chan KCG , Hassenstab J , Besser LM , Kukull WA , Crary JF ((2020) ) Cognitive trajectory in mild cognitive impairment due to primary age-related tauopathy. Brain 143: , 611. |
[50] | Crary JF , Trojanowski JQ , Schneider JA , Abisambra JF , Abner EL , Alafuzoff I , Arnold SE , Attems J , Beach TG , Bigio EH , Cairns NJ , Dickson DW , Gearing M , Grinberg LT , Hof PR , Hyman BT , Jellinger K , Jicha GA , Kovacs GG , Knopman DS , Kofler J , Kukull WA , Mackenzie IR , Masliah E , McKee A , Montine TJ , Murray ME , Neltner JH , Santa-Maria I , Seeley WW , Serrano-Pozo A , Shelanski ML , Stein T , Takao M , Thal DR , Toledo JB , Troncoso JC , Vonsattel JP , White CL , Wisniewski T , Woltjer RL , Yamada M , Nelson PT ((2014) ) Primary age-related tauopathy (PART): A common pathology associated with human aging. Acta Neuropathol 128: , 755–66. |
[51] | Jacobs D , Sano M , Marder K , Bell K , Bylsma F , Lafleche G , Albert M , Brandt J , Stern Y ((1994) ) Age at onset of Alzheimer’s disease: Relation to pattern of cognitive dysfunction and rate of decline. Neurology 44: , 1215–1220. |
[52] | Smits LL , Pijnenburg YAL , van der Vlies AE , Koedam ELGE , Bouwman FH , Reuling IEW , Scheltens P , van der Flier WM ((2015) ) Early onset APOE E4-negative Alzheimer’s disease patients show faster cognitive decline on non-memory domains. Eur Neuropsychopharmacol 25: , 1010–1017. |
[53] | Migliaccio R , Agosta F , Possin KL , Canu E , Filippi M , Rabinovici GD , Rosen HJ , Miller BL , Gorno-Tempini ML ((2015) ) Mapping the progression of atrophy in early- and late-onset alzheimer’s disease. J Alzheimers Dis 46: , 351–364. |
[54] | Lehmann M , Ghosh PM , Madison C , Laforce R , Corbetta-Rastelli C , Weiner MW , Greicius MD , Seeley WW , Gorno-Tempini ML , Rosen HJ , Miller BL , Jagust WJ , Rabinovici GD ((2013) ) Diverging patterns of amyloid deposition and hypometabolism in clinical variants of probable Alzheimer’s disease. Brain 136: , 844–858. |



