Sex, Neuropsychiatric Profiles, and Caregiver Burden in Alzheimer’s Disease Dementia: A Latent Class Analysis
Abstract
Background:
Neuropsychiatric symptoms (NPS) in Alzheimer’s disease (AD) can be disruptive for patients and their families.
Objective:
We aimed to classify patients based on NPS and to explore the relationship of these classes with sex and with caregiver burden.
Methods:
The study cohort comprised individuals with AD dementia diagnosed at Ace Alzheimer Center in Barcelona, Spain, between 2011–2020. NPS were ascertained by using the Neuropsychiatric Inventory-Questionnaire. Latent class analysis was used to identify clusters of individuals sharing a similar NPS profile. We evaluated the caregiver burden using the Zarit Burden Interview. Multivariable regression models were used to obtain adjusted estimates of the association between sex, NPS classes, and caregiver burden.
Results:
A total of 1,065 patients with AD dementia and their primary caregivers were included. We classified patients into five different classes according to their NPS profile: “Affective”, “High-behavioral-disturbance”, “Negative-affect”, “Affective/deliriant”, and “Apathy”. We found that age, sex, and type of AD diagnosis differed greatly across classes. We found that patients from the “High-behavioral-disturbance” (OR = 2.56, 95% CI: 1.00–6.56), “Negative-affect” (OR = 2.72, 95% CI: 1.26–3.64), and “Affective/deliriant” (OR = 2.14, 95% CI: 1.26–3.64) classes were over two times more likely to have a female caregiver than those in “Apathy” class. These three classes were also the ones associated to the greatest caregiver burden in the adjusted analyses, which seems to explain the increased burden observed among female caregivers.
Conclusion:
Caregiver burden is highly dependent on the patient’s NPS profiles. Female caregivers provide care to patients that pose a greater burden, which makes them more susceptible to become overwhelmed.
INTRODUCTION
Alzheimer’s disease (AD) is currently the most frequent cause of dementia worldwide, and it is expected to affect more than 150 million people by 2050 [1]. Even though two thirds of clinically diagnosed cases of AD dementia are women [2], the importance of sex and gender related factors in this condition has traditionally been overlooked. These ignored differences include sex interactions, such as the differential metabolic effects of APOE ɛ4 carriers among females [3, 4], the increased prevalence of common risk factors, like the lower access to education among women, or the existence of sex-specific risk factors (e.g., pregnancy or menopause) [5]. This generates knowledge gaps that may have delayed advances in the detection, treatment, and care of AD patients [6].
This complex disease is characterized by the presence of cognitive symptoms, such as memory, orientation, language, or executive function impairment, along with neuropsychiatric symptoms (NPS) that can be extremely disruptive not only for the patient, but also the family and friends.
In fact, NPS place a tremendous burden on caregivers [7]. Consequently, caregivers frequently become overwhelmed, which impacts upon their own mental health and general well-being, as well as their ability to provide an adequate care to the patient. This has been described as bidirectional relationship, in which patients might experience more severe or frequent NPS as a direct consequence of the caregiver being overwhelmed [8].
Interestingly, the occurrence of NPS varies greatly between individuals with notable differences between male and female patients. For instance, apathy, agitation, and inappropriate social conduct tend to be more common among male patients, whereas depressive symptoms, emotional instability, and delirium are more frequent among females [9]. Recent neuroimaging studies have confirmed the existence of NPS-specific brain lesion patterns [10], highlighting the importance of NPS to discern different subtypes of AD. In a previous work, we used prominent NPS to determine profiles in mild cognitive impairment patients and confirmed the predictive value of these profiles on conversion to specific types of dementia [11]. NPS are always presented in a subject in a comorbid way. Two or more NPS are usually presented at the same time in the same patient. Latent class analysis (LCA) is an excellent tool to manage this, offering a more clinical and ecological framework. Given the importance of NPS in the caregiver burden of patients with dementia, we hypothesized that this burden is likely to differ depending on the patients’ NPS profiles, and that gender could play an important role in how this burden is perceived by caregivers. It is important to highlight that prior evidence shows that female caregivers experience more burden than male caregivers [12, 13] and most dementia caregivers are females [14].
We studied a cohort of AD patients and their primary caregivers from a large, specialized memory unit based in Barcelona, Spain, and classified patients according to their NPS profiles. We aimed to explore how these profiles were associated with sex of both the patient’ and the caregiver) and with the burden of the disease perceived by the caregiver as measured by Zarit Burden Interview scores [10].
METHODS
The study cohort comprised individuals with AD dementia (including AD with vascular dementia) diagnosed at Ace Alzheimer Center, Barcelona, Spain, between 2011 and 2020. AD diagnosis was based on NINCDS/ADRDA criteria [16] up to 2014 and on NIA-AA criteria [17] thereafter. Patients with potential causes of dementia other than AD were excluded (i.e., Lewy bodies, Parkinson, pure vascular dementia, and frontotemporal dementia). Only patients with a Clinical Dementia Rating (CDR) score between 1 and 3, and those who had an identified primary permanent caregiver (i.e., relative) were eligible [18]. In order to be included in the study, caregivers were also required to have a Zarit evaluation completed within 3 months of the neurological examination in which the patient’s neuropsychological symptoms were evaluated.
NPS were ascertained by using the Neuropsychiatric Inventory-Questionnaire (NPI-Q). This is a simplified clinical scale used to assess dementia-related behavioral disturbances in 12 domains [19]. In our study, NPI-Q was completed by caregivers, under the supervision of trained physicians. For each domain, a change during the last month was measured as present or absent (dichotomous variable). Psychometric properties of the NPI-Q have been shown to be robust, with high test-retest correlations between total symptom and distress scores [20].
We used LCA to identify clusters of individuals sharing a similar NPS profile. LCA provides a flexible analytical approach that allows researchers to study patterns of observations in data and to make inferences about unobserved sources of population heterogeneity [21]. The main objective of this strategy is to assign participants sharing similar characteristics (person-centered approach) to distinct profiles, based on their expressions on a number of variables that are intercorrelated [22]. LCA uses patterns of responses on dichotomous variables to estimate two different parameters, called latent class probabilities and conditional probabilities. Latent class probabilities become prevalence of each class and conditional probabilities are rates of each analyzed variable given membership in each latent class. Using these parameters, it is possible to estimate the probability an individual’s affiliation to each class, according to their pattern of symptoms and their modal class membership. Agitation/aggression (agitation), hallucinations, anxiety, apathy/indifference (apathy), aberrant motor behavior (motor), delusions, depression/dysphoria (depression), disinhibition, elation/euphoria (euphoria), irritability/lability (irritability), appetite and eating disorders (appetite), and sleep and night-time behavior (sleep) were the NPS domains included in our analysis. The final LCA model was determined using a consensus of several fit criteria including Akaike and Bayes Information Criterion (AIC and BIC, respectively) [23], Entropy value [24], Vuong-Lo-Mendell-Rubin Likelihood Ratio Test (LRT), and Lo-Mendell-Rubin adjusted LRT in order to choose the best fitting model with the smallest number of parameters (NPS profiles) [25]. An optimal application of LCA needs the consideration that variables included in the analysis are independent between them after conditional class membership is created. This assumption was tested using standardized bivariate residuals [26], contrasting the observed symptom patterns with those predicted by the model. Once LCA was performed and the most parsimonious number of classes was determined, each participant was assigned to the class according to the highest membership probability. Subsequently, we used this latent class solution as a study factor in all the analyses.
We evaluated the caregiver burden using the Zarit Burden Interview (ZBI) [15]. This scale is widely used and even though it was originally conceived to assess the burden in caregivers of dementia patients, it has been successfully applied also to caregivers of other types of patients [27, 28]. This self-administered scale is completed by the caregiver under the supervision of the social worker. It consists of 22 items addressing how the caregiver feels about the current situation covering different topics that are usually affected in the caregiver’s life such as stress level, financial strain, emotional health, etc. This scale has been translated to Spanish and previously validated in Spain showing high consistency and reliability [29]. In this study, we used the total score that results from summing up the individual scores obtained in all 22 items. This approach assumes a single dimension for the latent burden construct. However, other authors have identified an underlying structure with between two and five different components within this test. Thus, in a sensitivity analysis we also explored the use of three different factors in an alternative analysis as proposed by Martin-Carrasco et al: burden, competency, and dependence [30].
Other patients’ characteristics such as age, sex, education, Mini-Mental State Examination (MMSE) scores [31], CDR [18], and type of AD diagnosis (possible/probable) [32] were also obtained from Ace Alzheimer Center Barcelona clinical database and included in the analyses. Similarly, caregivers’ age, sex, education, and relationship with the patient were also obtained and considered in the analyses.
Statistical analyses included univariate test such as ANOVA for continuous variables and chi-square test for categorical variables. Multivariable logistic (for likelihood of having a female caregiver) and linear (for ZBI scores) regression models were used to obtain adjusted estimates of association between the study variables. Bonferroni correction was used to adjust for multiple comparisons in the main analyses. Patients’ and caregivers’ sex were explored as modifiers of the effect of NPS classes on ZBI burden by introducing interaction terms in the adjusted model. All analyses were conducted using Stata 12.1 (College Station, TX) and Mplus 8.0.
RESULTS
A total of 1,065 patients with AD dementia and their corresponding primary caregivers were included in the study. Mean age was 80.1 years (SD = 7.9) among patients and 69.0 years (SD = 12.0) among caregivers. Most patients (60.8%) and caregivers (62.9%) were females. Caregivers were mainly patient’s spouses (64.3%) and children (31.8%). Mean time from caregiver’s Zarit evaluation to the corresponding patient’s neurological examination was 12.5 days (SD = 17.0)
The most frequent NPS reported were apathy (63.6%) and irritability (53.1%) followed by anxiety (49.6%) and depression (40.3%). The least common symptoms were disinhibition (6.8%) and euphoria (1.3%) (Supplementary Tables 1 and 2). NPS more prevalent among female than among male patients include eating disorders (17.7% versus 9.4; p < 0.01), depression (45.2% versus 32.6%; p < 0.01), anxiety (54.0% versus 42.7%; p < 0.01), hallucinations (10.8% versus 6.0%; p < 0.01), and delusions (21.1% versus 14.4%; p < 0.01). Only disinhibition was significantly more frequent among male than among female patients (9.1% versus 5.2%; p = 0.01). Apathy and irritability were also more frequent among men, although the difference was not statistically significant. The final LCA solution was determined according to parameters included in Table 1 and its clinical interpretation. The 5-class model was considered to fit the best. Thus, although Akaike and BIC values of the 6-class model was the lowest, entropy value increased with the number of classes in the model up to the 5-class model, and then decreased for the 6-class model, this latter not being statistically significant in terms of the adjusted LRT.
Table 1
Latent class analysis results for increasing number of classes
| Number of latent classes | |||||
| 2 | 3 | 4 | 5 | 6 | |
| Akaike | 11144.3 | 10935.5 | 10876.8 | 10842.8 | 10817.5 |
| BIC | 11268.6 | 11124.5 | 11130.4 | 11161.1 | 11200.4 |
| Entropy | 0.70 | 0.69 | 0.71 | 0.72 | 0.69 |
| Vuong-Lo-Mendell-Rubin Likelihood Ratio Test (LRT) p | <0.001 | 0.001 | 0.005 | 0.036 | 0.405 |
| Lo-Mendell-Rubin Adjusted LRT p | <0.001 | 0.001 | 0.006 | 0.037 | 0.409 |
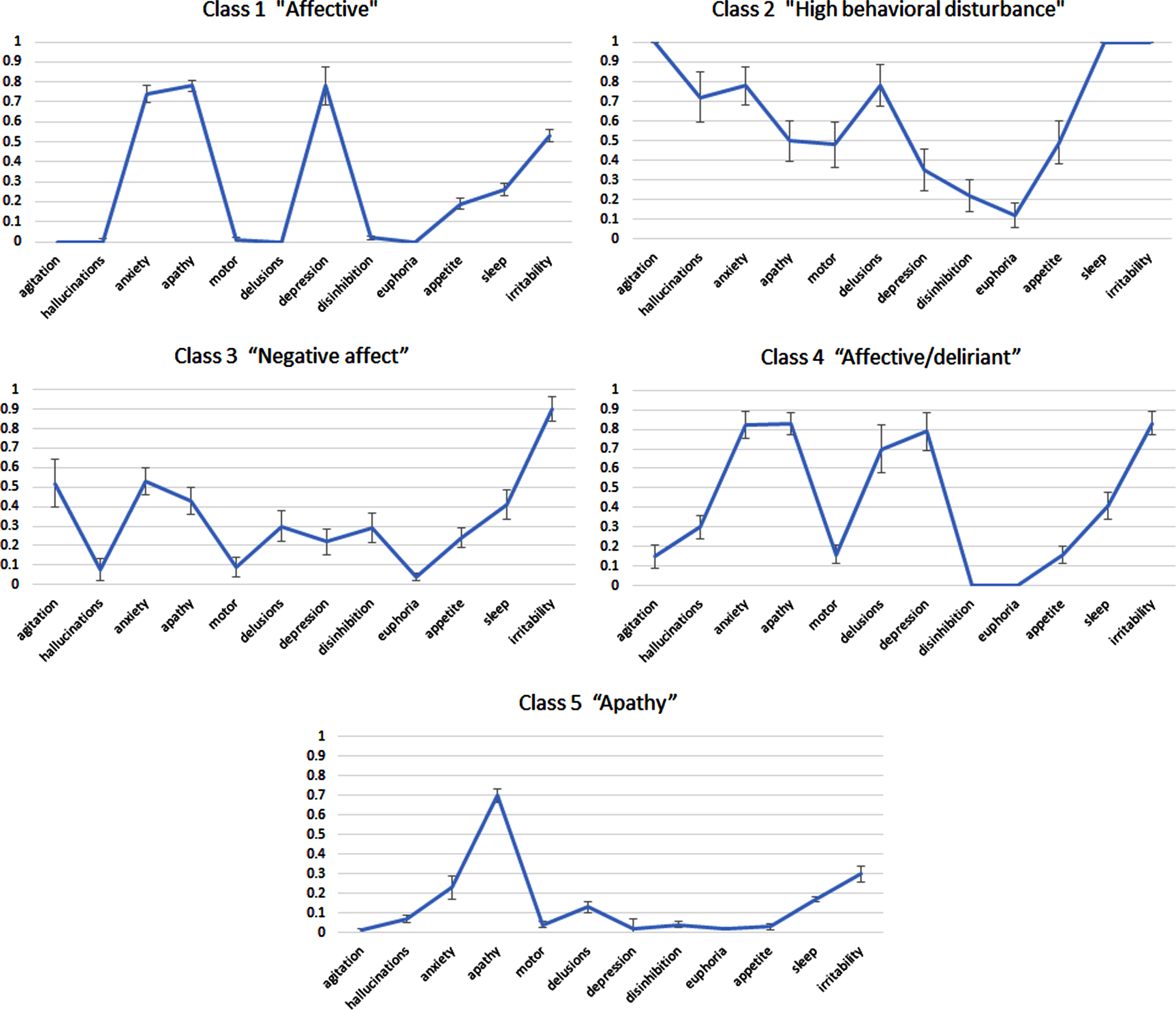
The results revealed a structure in which each class was determined by specific symptomatology, and the most preeminent symptoms were used to name each class (see Fig. 1). Thus, class 1 (“Affective”; n = 387; 36.3%) comprised patients with high prevalence of depression, anxiety and apathy; class 2 (“High behavioral disturbance”; n = 35; 3.3%) included patients with a high prevalence of almost all symptoms except for depression, disinhibition, and euphoria (the latter two, the least frequent among all patients); class 3 (“Negative affect”, n = 110, 10.3%) patients showing mostly irritability and anxiety; class 4 (“Affective/deliriant”; n = 122; 11.5%) similar to Class 1 but with higher prevalence of irritability and especially delusion; and class 5 (“Apathy”; n = 411; 38.6%), the most populous group, included patients that shared apathy as the most frequently reported symptom. In terms of clinical significance, to accept a 4-class solution (see Table 1) essentially means collapsing from the 5-class output the class 2 (“High behavioral disturbance”) and class 3 (“Negative affect”). However, to have a group with a preeminent irritable and anxiety profile or a group with high neuropsychiatric burden seems to be clinically relevant, because it means the opportunity to have, separately, one of the most disturbing groups that have to be handled in a Memory Unit (and by its caregivers). For the 5-class solution, classification probabilities for the most likely latent class membership in the case of the class 1 was 85%, for class 2 86%, 64% for class 3, 75% in the case of class 4 and 91% for class 5. Finally, among the 264 residuals generated by the 5-class model, only 4 (1.5%) had a value >1.96, suggesting conditional independence among latent class indicators.
Fig. 1
Profile plots represent estimated conditional probabilities (y-axis) observed in the latent class analysis (LCA) for the domains of the Neuropsychiatric Inventory-Questionnaire (NPI-Q; x-axis), displaying the 5-class solution.
We describe the frequency of patient’s characteristics stratified by clusters (5-class solution) in Table 2 (and Supplementary >Tables 3 and 4). In the univariate analyses there were significant differences among the five classes in most of the variables, except for education, with the frequency of patients with secondary education being around 60% in all five classes. Interestingly, females represented the larger share of patients in all classes except for class 3 (characterized as “Negative affect”) as most patients with this NPS profile were male (50.9%; p < 0.01). Regarding caregivers’ characteristics (Table 2) we found that caregivers’ age differed between the five defined classes, but again caregiver’s education did not. Finally, large differences were observed in the proportion of patients with female caregivers, that ranged from 46.6% in class 1 to 81.8% among patients from class 3. The results of the multivariable adjusted analysis showed that patients from “High behavioral disturbance”, “Negative affect”, and “Affective/deliriant” classes were over two times more likely to have a female caregiver than those in “Apathy” class (Supplementary Table 5).
Table 2
Patient and caregiver characteristics by NPS profiles
| Factors | class 1 Affective | class 2 High behavioral disturbance | class 3 Negative affect | class 4 Affective/deliriant | class 5 Apathy | Chi2/ ANOVA test |
| N = 387 | N = 35 | N = 110 | N = 122 | N = 411 | p | |
| Patient characteristics | ||||||
| Sex | ||||||
| male | 135 (34.9%) | 11 (31.4%) | 56 (50.9%) | 35 (28.7%) | 180 (43.8%) | <0.001* |
| female | 252 (65.1%) | 24 (68.6%) | 54 (49.1%) | 87 (71.3%) | 231 (56.2%) | |
| Age categories | ||||||
| 40–59 | 9 (2.3%) | 0 (0.0%) | 3 (2.7%) | 3 (2.5%) | 10 (2.4%) | <0.001* |
| 60–69 | 35 (9.0%) | 2 (5.7%) | 4 (3.6%) | 7 (5.7%) | 32 (7.8%) | |
| 70–79 | 137 (35.4%) | 3 (8.6%) | 19 (17.3%) | 40 (32.8%) | 122 (29.7%) | |
| 80–89 | 188 (48.6%) | 22 (62.9%) | 71 (64.5%) | 58 (47.5%) | 207 (50.4%) | |
| 90+ | 18 (4.7%) | 8 (22.9%) | 13 (11.8%) | 14 (11.5%) | 40 (9.7%) | |
| Mean (SD) | 79.0 (7.9) | 84.7 (6.2) | 82.1 (7.5) | 80.6 (8.0) | 80.1 (7.9) | < 0.001* |
| Education | ||||||
| primary or less | 156 (40.3%) | 14 (40.0%) | 44 (40.0%) | 55 (45.1%) | 155 (37.7%) | 0.70 |
| secondary or more | 231 (59.7%) | 21 (60.0%) | 66 (60.0%) | 67 (54.9%) | 256 (62.3%) | |
| CDR | ||||||
| 1 | 211 (54.5%) | 8 (22.9%) | 42 (38.2%) | 32 (26.2%) | 188 (45.7%) | <0.001* |
| 2 | 160 (41.3%) | 21 (60.0%) | 59 (53.6%) | 75 (61.5%) | 185 (45.0%) | |
| 3 | 16 (4.1%) | 6 (17.1%) | 9 (8.2%) | 15 (12.3%) | 38 (9.2%) | |
| MMSE | ||||||
| Mean (SD) | 18.4 (4.9) | 13.9 (5.8) | 16.5 (5.4) | 15.6 (5.3) | 17.1 (5.9) | <0.001* |
| Dementia diagnosis | ||||||
| possible | 127 (32.8%) | 17 (48.6%) | 20 (18.2%) | 41 (33.6%) | 99 (24.1%) | <0.001* |
| probable | 260 (67.2%) | 18 (51.4%) | 90 (81.8%) | 81 (66.4%) | 312 (75.9%) | |
| Caregiver characteristics | ||||||
| Sex | ||||||
| male | 168 (43.4%) | 8 (22.9%) | 20 (18.2%) | 42 (34.4%) | 157 (38.2%) | <0.001* |
| female | 219 (56.6%) | 27 (77.1%) | 90 (81.8%) | 80 (65.6%) | 254 (61.8%) | |
| Age categories | ||||||
| 30–59 | 93 (24.0%) | 9 (25.7%) | 25 (22.7%) | 41 (33.6%) | 82 (20.0%) | 0.23 |
| 60–69 | 86 (22.2%) | 13 (37.1%) | 27 (24.5%) | 29 (23.8%) | 91 (22.1%) | |
| 70–79 | 123 (31.8%) | 7 (20.0%) | 35 (31.8%) | 33 (27.0%) | 136 (33.1%) | |
| 80–89 | 82 (21.2%) | 5 (14.3%) | 22 (20.0%) | 18 (14.8%) | 96 (23.4%) | |
| 90+ | 3 (0.8%) | 1 (2.9%) | 1 (0.9%) | 1 (0.8%) | 6 (1.5%) | |
| Mean (SD) | 68.9 (12.3) | 66.3 (12.4) | 69.2 (12.2) | 65.4 (12.4) | 70.3 (11.4) | 0.001* |
| Education | ||||||
| primary or less | 226 (58.4%) | 21 (60.0%) | 79 (71.8%) | 74 (60.7%) | 254 (61.8%) | 0.16 |
| secondary or more | 161 (41.6%) | 14 (40.0%) | 31 (28.2%) | 48 (39.3%) | 157 (38.2%) |
Data are presented as mean (SD) for continuous variables and n (%) for categorical variables. Univariate test results are presented in the last column. *Statistically significant p-values after Bonferroni correction (p < 0.004).
We were able to obtain a score for the ZBI in all caregivers and studied to what extent the characteristics of the patient (notably the NPS profile) or the caregiver were related with caregiver burden as captured by this score. Thus, after adjusting for patient and caregiver characteristics we found that the lowest burden estimates corresponded to caregivers of patients from class 1 (“Affective”: ZBI = 50.6, 95% CI: 49.2–52.1) and class 5 (“Apathy”: ZBI = 48.5, 95% CI: 48.5–51.3). Caregivers of patients in class 2 (“High behavioral disturbance”: ZBI = 64.9, 95% CI: 60.0–69.7) and class 4 (“Affective/deliriant”: ZBI = 58.8, 95% CI: 56.1–61.3) experienced the greatest burden. When individual NPS were used instead of the profiles defined by the LCA, we found that only anxiety and apathy were associated with a lower burden, although far from statistical significance. In contrast, presence of hallucinations, delusion, disinhibition, and irritability symptoms were all associated with an increased burden in the adjusted analysis (Supplementary Tables 1and 2).
Among other factors, female caregivers reported slightly higher burden than males, but this difference (beta coefficient = 2.5) did not reach statistical significance (p = 0.06). As seen in Table 3, older age of the caregiver was associated with a statistically significant lower burden, but caregivers’ education, or patient age, sex and education did not affect the caregiver’s burden. However, increasing disease severity was associated with greater burden, as individuals with CDR of 2 and 3 were associated with beta coefficients of 5.50 and 6.71 respectively, compared to those with a CDR of 1 (Table 3). Finally, we found no evidence that the observed association between NPS classes and caregiver burden was modified by patient’s or caregiver’s sex (Supplementary Tables 6 and 7).
Table 3
Zarit Burden score as a function of patient/caregiver characteristics
| Factors | N | Estimated Marginal Mean (SE)† | Beta† | 95% CI† | p† |
| Patient characteristics | |||||
| Sex | |||||
| female* | 648 | 52.1 (0.7) | – | ||
| male | 417 | 52.2 (0.9) | 0.09 | (–2.45 to 2.64) | 0.94 |
| Age categories | |||||
| 40–59* | 25 | 51.0 (3.0) | – | ||
| 60–69 | 80 | 51.9 (1.8) | 0.98 | (–5.62 to 7.59) | 0.77 |
| 70–79 | 321 | 52.5 (0.9) | 1.54 | (–4.65 to 7.72) | 0.63 |
| 80–89 | 546 | 52.4 (0.7) | 1.40 | (–4.75 to 7.54) | 0.66 |
| 90+ | 93 | 50.5 (1.6) | –0.49 | (–7.29 to 6.30) | 0.89 |
| Education | |||||
| primary education or less* | 424 | 52.0 (0.8) | – | ||
| secondary education or more | 641 | 52.3 (0.6) | 0.35 | (–1.64 to 2.34) | 0.73 |
| NPI profiles | |||||
| class 1 Affective | 387 | 50.6 (0.7) | 0.76 | (–1.28 to 2.81) | 0.46 |
| class 2 High behavioral dist. | 35 | 64.9 (2.5) | 14.99 | (9.90 to 20.09) | <0.01‡ |
| class 3 Negative affect | 110 | 54.9 (1.4) | 5.03 | (1.94 to 8.11) | <0.01‡ |
| class 4 Affective/deliriant | 122 | 58.8 (1.3) | 8.89 | (5.91 to 11.87) | <0.01‡ |
| class 5 Apathy* | 411 | 49.9 (0.7) | – | ||
| CDR | |||||
| 1* | 481 | 49.1 (0.8) | – | ||
| 2 | 500 | 54.6 (0.7) | 5.50 | (3.12 to 7.88) | <0.01‡ |
| 3 | 84 | 55.8 (2.0) | 6.71 | (1.92 to 11.50) | 0.01 |
| MMSE | |||||
| per unit | – | – | –0.12 | (–0.36 to 0.13) | 0.36 |
| Dementia diagnosis | |||||
| possible* | 304 | 53.7 (0.9) | – | ||
| probable | 761 | 51.5 (0.5) | –2.20 | (–4.20 to –0.20) | 0.03 |
| Caregiver characteristics | |||||
| Sex | |||||
| male* | 395 | 50.6 (0.9) | – | ||
| female | 670 | 53.1 (0.7) | 2.52 | (–0.07 to 5.11) | 0.06 |
| Age categories | |||||
| 30–59* | 250 | 55.7 (1.0) | – | ||
| 60–69 | 246 | 55.6 (1.0) | –0.03 | (–2.79 to 2.72) | 0.98 |
| 70–79 | 334 | 49.6 (0.9) | –6.11 | (–8.99 to –3.22) | <0.01‡ |
| 80–89 | 223 | 48.7 (1.1) | –6.94 | (–10.11 to –3.77) | <0.01‡ |
| 90+ | 12 | 44.6 (4.2) | –11.04 | (–19.72 to –2.36) | 0.01 |
| Education | |||||
| primary education or less* | 654 | 51.7 (0.6) | – | ||
| secondary education or more | 411 | 52.9 (0.8) | 1.15 | (–0.92 to 3.22) | 0.28 |
*Reference category. †Estimates obtained by fitting a linear regression model with all variables in the table. ‡Statistically significant p-values after Bonferroni correction (p < 0.002).
In a sensitivity analysis we repeated the analyses separately for each one of the three components identified in ZBI by Martin-Carrasco et al. [30] instead of the total score used in the main analyses (Supplementary Tables 8–10). Qualitatively, the results of these sensitivity analyses for these three components (burden, competency, and dependence) were similar to those from the main analysis.
DISCUSSION
Our study, performed in 1,065 patients with AD dementia and their corresponding caregivers, shows that classifying patients with AD dementia according to NPS using LCA analysis is a useful tool that enables the identification of groups of individuals that share a similar profile of symptoms. We detected five groups or classes of NPS that we defined as “Affective”, “High behavioral disturbance”, “Negative affect”, “Affective/deliriant”, and “Apathy” according to the preponderant symptomatology.
Patients belonging to each NPS class differed in almost every characteristic, except for education. Individuals in classes 2 (“High behavioral disturbance”) and 3 (“Negative affect”) were the oldest. Those in the “Negative affect” class were also the most likely to be males and to have a diagnosis of ‘probable AD’. We also found that this “Negative affect” class, along with the “High behavioral disturbance”, and “Affective/deliriant” classes (all of them with high irritability component) had with the largest proportions of female caregivers. Interestingly, these three classes remained the most likely to have a female caregiver after adjusting for patient’s characteristics (including age, sex, and others). This suggests other reasons (possibly related to socio-cultural issues, such as women’s traditional role in family care along with lower access to education and labor markets) to explain why patients with this irritability component tend to have a female caregiver, irrespective of their sex.
We should keep this in mind when interpreting the results of how the caregiver burden is associated with the NPS profile of patients. We found that the strongest predictor of burden was in fact the NPS profile the patients belonged to, with the previously mentioned classes of patients with highly prevalent irritability component (classes 2–4) being those that consistently associate with a higher caregiver burden. These results are compatible with those from a recent systematic review including studies assessing individual NPS showing that irritability was associated with the greatest caregiver burden [33] Also, previous studies have already shown that female caregivers experienced greater burden [34]. However, the novelty of our study is that we found that once NPS are introduced in the adjusted model, female caregivers are no longer significantly associated with an increased burden (borderline statistical significance only). This finding suggests that the previously reported greater burden among female caregivers, is arguably explained by its relationship with patients’ NPS profiles. It is important to note that, the strong relationship between the NPS classes has a similar effect on caregiver burden irrespective of patient’s or caregiver’s sex (i.e., no evidence of effect modification). Introducing these NPS classes in the analyses helped us to confirm that the higher burden observed in unadjusted estimates among female caregivers is not due to a more negative perception of the same burden. Thus, their increased burden is likely related to the fact that females take care of patients with symptoms associated to greater burden, such as those patients in classes 2–4 in our analyses. While the ultimate reasons behind this reality are likely to have socio-cultural roots, ones that might not be easily be changed, it is important to stress that according to our data female caregivers do not simply “perceive” a greater burden, but they actually carry the greatest burden of AD dementia patients on their shoulders.
Another interesting observation is that older caregivers seem to report less burden than younger ones. Contrary to what we observed for female caregivers, this result remained significant after adjustment for all covariates. Therefore, given a patient with similar NPS profile, age, sex, etc., older caregivers consistently reported less burden. This suggest that there could be a true divergence between “perceived” and “experienced” burden. For instance, greater resignation in older caregivers who have been raised in a different social context could serve as an explanation. Alternatively, younger caregivers might have additional professional or familiar responsibilities beyond the dementia patient or have greater expectations that make them feel more overwhelmed. One could think that older caregivers, who probably face health problems of their own, could experience a greater burden from a similar patient, but the results of our study suggest the exact opposite, they tend to perceive less burden. To what extent adaptative mechanisms described by other authors could also be related to this finding is uncertain [35].
We should note that previous studies using LCA of NPS in AD have frequently employed population-based samples [36]. While this approach has other advantages, it tends to include milder patients who present with less comorbidities, and this influences the results of these analyses. In contrast, our study sample comprised patients from a memory clinic, making conclusions regarding NPS classes extremely valuable for clinicians.
This study has several limitations. Although we were able to accrue a large sample size, these numbers became smaller as we classify into NPS groups or into different strata of relevant variables. We cannot ignore the potential reverse causation in this bidirectional relationship and should not exclude that NPS occurrence is a consequence rather than a cause of caregiver burden, as previously reported [8]. Furthermore, these tests are based on perception of patient symptoms and caregiver burden, and these two might not be entirely independent. For instance, overwhelmed caregivers might tend to either overemphasize or to downplay existing NPS in the patients they take care of. Perception of burden, and gender issues in patient-caregiver relationships are rather affected by the social and cultural background. Thus, we should be cautious when interpreting the study results and be aware that some of the conclusions from this study might not be applicable to other populations. Furthermore, we restricted our study to primary caregivers, so making inference about other type of caregivers based on these data can be problematic. Finally, some of the analyses involved a large number of comparisons. However, applying Bonferroni correction did not change our conclusions.
We should also note that the use of a clustering strategy grouping individuals according to their symptoms, as in the case of this study, should be interpreted carefully. LCA does not provide exclusive groups of patients because the same symptom can be shared in two or more latent classes. For example, depression should be considered a preeminent symptom when defining classes 1 and 4, but this symptom is also present in other classes, although in a lower probability. Patients are assigned to a group or class according to a most-likely membership process. We are assuming, as in any other clustering approach, that any decision of the assignation of an individual to a class or about the results that emerge when analyzing the connection of these classes with other factors will be accompanied by some uncertainty. In the present study, as reported previously, the analysis of residuals allows us to confirm that, at least, conditional independence among latent class indicators can be consistently assumed, that is, there is no longer any association of one symptom with another and the class group can be assumed as the reason of their association
Detecting subtle behavioral changes and focusing on behavioral disturbances is a key clinical aspect in AD. This should be considered from the very first contact, as it would help professionals design an action plan to improve, throughout the clinical process, the quality of life of patients and families and to optimize treatment strategies. We have shown that identifying profiles of AD patients with dementia based on NPS is useful to predict the burden they pose on caregivers and to better understand the role other factors play. We found that NPS profiles that include irritability symptoms (“High behavioral disturbance”, “Negative affect”, and “Affective/deliriant”) are associated to increased caregiver burden. AD dementia requires a holistic approach in which the patient should be at the center, but recognizing the essential role played by the caregiver. Our study confirmed that female caregivers provide care to patients that tend to pose a greater burden. Therefore, they are more susceptible to becoming overwhelmed, and this should be considered by neurologists, psychologists and social workers involved in improving the quality of life of patients with AD dementia. Our societies should acknowledge the invaluable contribution of these female caregivers and propose mechanisms to minimize the disruption they might experience.
ACKNOWLEDGMENTS
We are grateful to the patients of Fundació ACE memory clinic and their families who contributed with significant time and effort, without which this article would not have been possible. We also thank other collaborators at Fundació ACE, the health professionals assisting our patients and the administrative team. We are also thankful to Fundació ACE’s sponsors and contributors.
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/21-5648r4).
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/JAD-215648.
REFERENCES
[1] | Patterson C ((2018) ) World Alzheimer Report 2018. The state of the art of dementia research: New frontiers. Alzheimer’s Disease International, London. |
[2] | Alzheimer’s Association ((2013) ) 2013 Alzheimer’s disease facts and figures. Alzheimers Dement 9: , 208–245. |
[3] | Hsu M , Dedhia M , Crusio WE , Delprato A ((2019) ) Sex differences in gene expression patterns associated with the APOE4 allele. F1000Res 8: , 387. |
[4] | Arnold M , Nho K , Kueider-Paisley A , Massaro T , Huynh K , Brauner B , MahmoudianDehkordi S , Louie G , Moseley MA , Thompson JW , John-Williams LS , Tenenbaum JD , Blach C , Chang R , Brinton RD , Baillie R , Han X , Trojanowski JQ , Shaw LM , Martins R , Weiner MW , Trushina E , Toledo JB , Meikle PJ , Bennett DA , Krumsiek J , Doraiswamy PM , Saykin AJ , Kaddurah-Daouk R , Kastenmüller G ((2020) ) Sex and APOE ɛ4 genotype modify the Alzheimer’s disease serum metabolome. Nat Commun 11: , 1148. |
[5] | Ferretti MT , Martinkova J , Biskup E , Benke T , Gialdini G , Nedelska Z , Rauen K , Mantua V , Religa D , Hort J , Santuccione Chadha A , Schmidt R ((2020) ) Sex and gender differences in Alzheimer’s disease: Current challenges and implications for clinical practice: Position paper of the Dementia and Cognitive Disorders Panel of the European Academy of Neurology. Eur J Neurol 27: , 928–943. |
[6] | Nebel RA , Aggarwal NT , Barnes LL , Gallagher A , Goldstein JM , Kantarci K , Mallampalli MP , Mormino EC , Scott L , Yu WH , Maki PM , Mielke MM ((2018) ) Understanding the impact of sex and gender in Alzheimer’s disease: A call to action. Alzheimers Dement 14: , 1171–1183. |
[7] | van der Lee J , Bakker TJEM , Duivenvoorden HJ , Dröes RM ((2014) ) Multivariate models of subjective caregiver burden in dementia: A systematic review. Ageing Res Rev 15: , 76–93. |
[8] | Isik AT , Soysal P , Solmi M , Veronese N ((2019) ) Bidirectional relationship between caregiver burden and neuropsychiatric symptoms in patients with Alzheimer’s disease: A narrative review. Int J Geriatr Psychiatry 34: , 1326–1334. |
[9] | Sundermann EE , Katz MJ , Lipton RB ((2017) ) Sex differences in the relationship between depressive symptoms and risk of amnestic mild cognitive impairment. Am J Geriatr Psychiatry 25: , 13–22. |
[10] | Chen Y , Dang M , Zhang Z ((2021) ) Brain mechanisms underlying neuropsychiatric symptoms in Alzheimer’s disease: A systematic review of symptom-general and -specific lesion patterns. Mol Neurodegener 16: , 38. |
[11] | Roberto N , Portella MJ , Marquié M , Alegret M , Hernández I , Mauleón A , Rosende-Roca M , Abdelnour C , de Antonio EE , Gil S , Tartari JP , Vargas L , Espinosa A , Ortega G , Pérez-Cordón A , Sanabria Á , Orellana A , de Rojas I , Moreno-Grau S , Montrreal L , Alarcón-Martín E , Ruíz A , Tárraga L , Boada M , Valero S ((2021) ) Neuropsychiatric profiles and conversion to dementiain mild cognitive impairment, a latent class analysis. Sci Rep 11: , 6448. |
[12] | D’Onofrio G , Sancarlo D , Addante F , Ciccone F , Cascavilla L , Paris F , Picoco M , Nuzzaci C , Elia AC , Greco A , Chiarini R , Panza F , Pilotto A ((2015) ) Caregiver burden characterization in patients with Alzheimer’s disease or vascular dementia. Int J Geriatr Psychiatry 30: , 891–899. |
[13] | Akpınar B , Kücükgüçlü O , Yener G ((2011) ) Effects of gender on burden among caregivers of Alzheimer’s patients. J Nurs Scholarsh 43: , 248–254. |
[14] | Cañabate P , Martínez G , Rosende-Roca M , Moreno M , Preckler S , Valero S , Sotolongo O , Hernández I , Alegret M , Ortega G , Espinosa A , Mauleón A , Vargas L , Rodríguez O , Abdelnour C , Sánchez D , Martín E , Ruiz A , Tárraga L , Boada M ((2017) ) Social representation of dementia: An analysis of 5,792 consecutivecases evaluated in a memory clinic. J Alzheimers Dis 58: , 1099–1108. |
[15] | Zarit SH , Reever KE , Bach-Peterson J ((1980) ) Relatives of the impaired elderly: Correlates of feelings of burden. Gerontologist 20: , 649–655. |
[16] | McKhann G , Drachman D , Folstein M , Katzman R , Price D , Stadlan EM ((1984) ) Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group* under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34: , 939–939. |
[17] | McKhann GM , Knopman DS , Chertkow H , Hyman BT , Jack CR Jr , Kawas CH , Klunk WE , Koroshetz WJ , Manly JJ , Mayeux R , Mohs RC , Morris JC , Rossor MN , Scheltens P , Carrillo MC , Thies B , Weintraub S , Phelps CH ((2011) ) The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7: , 263–269. |
[18] | Morris JC ((2012) ) The Clinical Dementia Rating (CDR): Current version and scoring rules. Neurology 43: , 2412. |
[19] | Boada M , Cejudo JC , Tàrraga L , López OL , Kaufer D ((2002) ) Neuropsychiatric inventory questionnaire (NPI-Q): Spanish validation of an abridged form of the Neuropsychiatric Inventory (NPI). Neurologia 17: , 317–323. |
[20] | Kaufer DI , Cummings JL , Ketchel P , Smith V , MacMillan A , Shelley T , Lopez OL , DeKosky ST ((2000) ) Validation of the NPI-Q, a brief clinical form of the Neuropsychiatric Inventory. J Neuropsychiatry Clin Neurosci 12: , 233–239. |
[21] | Miettunen J , Nordström T , Kaakinen M , Ahmed AO ((2016) ) Latent variable mixture modeling in psychiatric research–a review and application. Psychol Med 46: , 457–467. |
[22] | Berlin KS , Williams NA , Parra GR ((2014) ) An introduction to latent variable mixture modeling (part 1): Overview and cross-sectional latent class and latent profile analyses. J Pediatr Psychol 39: , 174–187. |
[23] | Schwarz G ((1978) ) Estimating the dimension of a model. Ann Stat 6: , 461–464. |
[24] | Ramaswamy V , Desarbo WS , Reibstein DJ , Robinson WT ((1993) ) An empirical pooling approach for estimating marketing mix elasticities with PIMS data. Mark Sci 12: , 103–124. |
[25] | Lo Y , Mendell NR , Rubin DB ((2001) ) Testing the number of components in a normal mixture. Biometrika 88: , 767–778. |
[26] | Andersen R ((2003) ) Jacques A. Hagenaars and Allan L. McCutcheon, eds. Applied Latent Class Analysis. Can J Sociol 28: , 584–587. |
[27] | Hagell P , Alvariza A , Westergren A , Årestedt K ((2017) ) Assessment of burden among family caregivers of people with Parkinson’s disease using the Zarit Burden Interview. J Pain Symptom Manage 53: , 272–278. |
[28] | Goncalves-Pereira M , González-Fraile E , Santos-ZorrozúaB , Martín-Carrasco M , Fernández-Catalina P , Domínguez-Panchón AI , Muñoz-Hermoso P , Ballesteros J ((2017) ) Assessment of the consequences of caregiving in psychosis: Apsychometric comparison of the Zarit Burden Interview (ZBI) and theInvolvement Evaluation Questionnaire (IEQ). Health Qual Life Outcomes 15: , 63. |
[29] | Carrasco MM ((1996) ) Adaptación para nuestro medio de la Escala de Sobrecarga del Cuidador de Zarit. Rev Multidisciplinar Gerontol 6: , 338. |
[30] | Martin-Carrasco M , Otermin P , Pérez-Camo V , Pujol J , Agüera L , Martín MJ , Gobartt AL , Pons S , Balañá M ((2010) ) EDUCAstudy: Psychometric properties of the Spanish version of the ZaritCaregiver Burden Scale. Aging Ment Health 14: , 705–711. |
[31] | Folstein MF , Folstein SE , McHugh PR ((1975) ) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12: , 189–198. |
[32] | Lopez OL , Kuller LH , Becker JT , Dulberg C , Sweet RA , Gach HM , Dekosky ST ((2007) ) Incidence of dementia in mild cognitive impairment in the cardiovascular health study cognition study. Arch Neurol 64: , 416–420. |
[33] | Terum TM , Andersen JR , Rongve A , Aarsland D , Svendsboe EJ , Testad I ((2017) ) The relationship of specific items on the Neuropsychiatric Inventory to caregiver burden in dementia: A systematic review. Int J Geriatr Psychiatry 32: , 703–717. |
[34] | Pinquart M , Sörensen S ((2003) ) Differences between caregivers and noncaregivers in psychological health and physical health: A meta-analysis. Psychol Aging 18: , 250–267. |
[35] | Seeher KM , Low LF , Reppermund S , Slavin MJ , Draper BM , Kang K , Kochan NA , Trollor JN , Sachdev PS , Brodaty H ((2014) ) Correlates of psychological distress in study partners of older people with and without mild cognitive impairment (MCI) - the Sydney Memory and Ageing Study. Aging Ment Health 18: , 694–705. |
[36] | Leoutsakos JM , Forrester SN , Corcoran CD , Norton MC , Rabins PV , Steinberg MI , Tschanz JT , Lyketsos CG ((2015) ) Latent classes of course in Alzheimer’s disease and predictors: The Cache County Dementia Progression Study. Int J Geriatr Psychiatry 30: , 824–32. |




