Elevated Activity of the Sympathetic Nervous System Is Related to Diminished Practice Effects in Memory: A Pilot Study
Abstract
Background:
Reductions in memory practice effects have gained interest as risk factor for future cognitive decline. Practice effects vary with age and can be moderated by factors such as individual variability in arousal or stress experience acting as an additional cognitive load.
Objective:
In the current pilot study, we examined whether sympathetic nervous system activation moderates the relationship between age and practice effects.
Methods:
Thirty cognitively healthy individuals aged 40–70 years performed a mnemonic discrimination task twice. Salivary alpha amylase (sAA) samples were obtained at different time points as a proxy of sympathetic activity. Spearman correlations examined the relation between practice effects and sAA. Subsequently, age by sAA interactions on practice scores were explored with bootstrapped linear regression models. Additionally, participants were divided in learners (exhibiting practice effects) and non-learners based on the difference in mnemonic discrimination performance.
Results:
Higher age and baseline SNS activity were independently related to lower practice effects. The non-learners showed significantly higher sAA scores at all time points compared to learners. Among the learners, baseline-adjusted lower levels of sAA after encoding were associated with greater practice effects, particularly in middle-aged individuals. No such interaction was observed for non-learners.
Conclusion:
These results show that higher baseline sympathetic activation is associated with worse practice effects independently of age. Additionally, in a subgroup of middle-aged learners practice effects were observed when sympathetic activity remained low during learning. These findings suggest that elevated sympathetic nervous system activation may be a promising indicator of imminent cognitive decline.
INTRODUCTION
Reductions in memory practice effects have gained interest as risk factor for future cognitive decline [1, 2]. Practice effects are defined as improvement on a test score due to repeated performance of a test [3, 4]. For a long time, practice effects were considered a source of error in longitudinal data sets, but recently attenuation of practice effects has been shown to hold valuable information for future decline and could indicate underlying neurodegenerative changes [1, 2, 5]. During aging, memory performance as well as the functioning of our stress system deteriorate, resulting in attenuated learning and altered stress responses, respectively [6–14]. Even though practice effects vary depending on frequency of testing, cognitive domains, and individual characteristics, age has a negative relationship with practice effects [3, 15]. Given that the stress system alters during aging, individual stress vulnerability contributes to explaining individual differences in cognitive trajectories [16, 17] and that stress experiences can act as an additional cognitive load [18], we set out to investigate whether individual variability in sympathetic activity moderates the relationship between age and practice effects.
Practice effects are expected with repeated performance of cognitive tasks [19, 20], especially between the first and second trial, after which they often plateau [15, 19, 21, 22]. Although the magnitude of practice effects in older adults are smaller compared to younger adults, cognitively intact older adults commonly show practice effects at retesting [3]. Attenuated practice effects have been associated with higher Alzheimer’s disease biomarker burden in cognitively intact older adults and are further attenuated or absent in people with mild cognitive impairment or Alzheimer’s disease [1, 22, 23].
Experience of stress activates two major stress systems: the autonomic nervous system (ANS) and the hypothalamus-pituitary adrenal axis (HPA-axis). The activation of these systems leads to the release of stress hormones and neurotransmitters, including noradrenaline from the sympathetic division of the ANS [24]. Salivary alpha amylase (sAA) can be considered a proxy for sympathetic nervous system (SNS) activation [25, 26] and increases rapidly in response to physiological as well as psychosocial stress conditions [27–29]. Baseline SNS activity is elevated in older individuals [30, 31]. Interestingly the effect of sAA activity on memory performance may be dependent on age. In younger adults elevated noradrenaline levels measured with sAA after encoding was related to better pattern separation performance [32]. In older adults, higher sAA levels around learning were related to impaired recall, while younger adults show improved recall [30, 33]. However, other studies have shown attenuated or similar response of the SNS activity in older adults when compared to younger adults [14, 31]. Animal research suggests that the neural mechanism underlying the effect of SNS activity on memory performance could be related to increased excitability of the hippocampus and amygdala in response to noradrenergic activity [34, 35].
As previous studies indicated that memory performance in young but not in older individuals improves after activation of the noradrenergic system prior to learning, we administered a well-validated object mnemonic discrimination task [36, 37] that is sensitive to subtle age-related memory changes, beginning as early as in the fourth decade [38–40]. Furthermore, previous work showed that increases in performance on this task occur in concert with increases in salivary alpha-amylase [32]. Thus, this experimental paradigm allows us to examine whether activation of the noradrenergic system facilitates practice effects on a memory task, and whether this is dependent on age in a sample of older individuals between the ages of 40 to 75 years old. Ultimately, the findings of this study can contribute to the development of measurements to identify individuals at risk for cognitive decline in the future.
METHODS
Participants
A total of thirty cognitively healthy individuals between the ages of 40–75 years old were randomly included from a larger study examining the effect of age on pattern separation. All participants had to be fluent in the Dutch language and able to give informed consent. Exclusion criteria were presence of cognitive deficits on neuropsychological screening (i.e., 1.5 SD below norms for age, sex, and education), current neurological or psychiatric diseases, neuroendocrine disorder, elevated blood pressure, use of medication for psychoactive disorder or medication that affects the stress system including blood pressure medication, and alcohol or drug abuse. The study was approved by the local Ethics Review Committee Psychology and Neuroscience and written informed consent was obtained from all participants in accordance to the Declaration of Helsinki (World Medical Association, 2013).
This sample size was estimated with a power calculation based on earlier findings by Segal et al. [32] (n = 13, r = 0.75), indicated that at least 11 participants would be required (power = 0.95, alpha = 0.05) to examine the effect of sAA levels on lure discrimination performance. Given that this effect was obtained within only the sAA responders (40%of the sample), we applied a similar proportionality to our calculation indicating that at least 28 subjects would be required.
Procedure
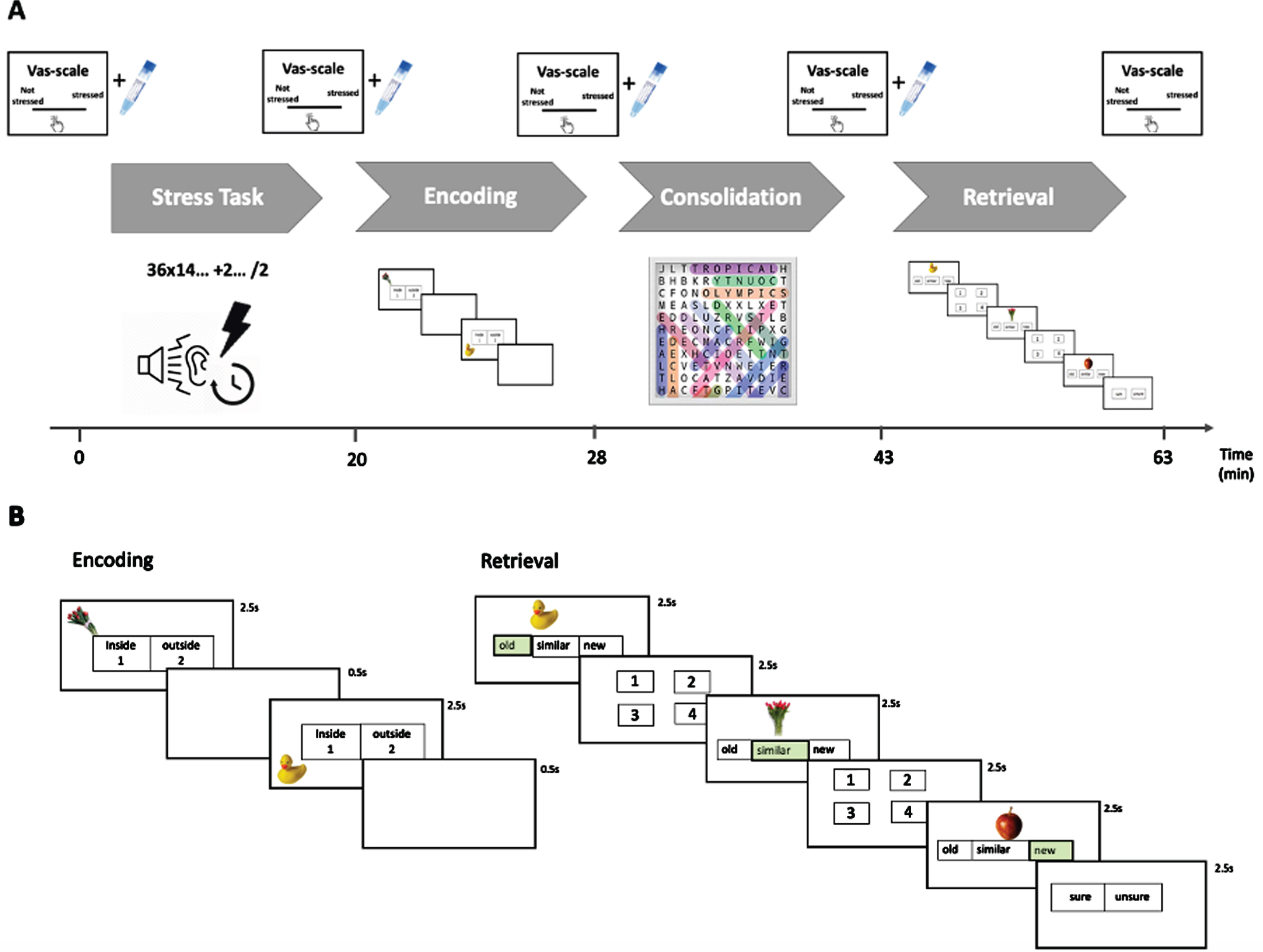
Participants were invited to Maastricht University and after obtaining informed consent they engaged in extensive neuropsychological testing and performed the first session of the mnemonic discrimination task.
Upon arrival at the university for their follow up session, informed consent for this additional study was obtained, and participants were asked to complete several computerized questionnaires and a base-line saliva sample was taken. Thereafter, participants performed a challenging arithmetic task (Markus-Peters Arithmetic (MPA) test) for 20 min. Upon completion, the second salivary sample and Visual Analogue Scales (VAS–scales) were acquired. Participants then performed an analogue similar version of the mnemonic discrimination task that was comparable in difficulty to the first test session. Two more saliva samples and VAS scales were collected, after learning and before retrieval of the mnemonic discrimination task (see Fig. 1A for timeline). At the end of the study, participants were debriefed about the goal of the study to ensure no long-term worry about their performance.
Fig. 1
A) Procedure timeline showing the MPA-task followed by the mnemonic discrimination task. Serial measurements of subjective stress (VAS) and physiologic (salivary samples) SNS levels are depicted above the timeline. B) Enlarged visualization of the mnemonic discrimination task with both the. encoding (lower left side) and retrieval (lower right side) phases. The correct answers during the retrieval stage are highlighted in green. VAS, visual analogue scale.
Measurements and tests
Neuropsychological test battery and questionnaires
The current stress and mood levels of subjects were measured via the Perceived Stress Scale [41] and the Hamilton depression rating scale [42]. Changes in subjective stress perception during the study were obtained via repeated measurements of Visual Analogue Scales (VAS). In addition, we collected information about their personality profile by administering the NEO-PI neuroticism subscale [43] which has been related to individual stress vulnerability [44–47].
The stress task
During the Markus-Peters Arithmetic (MPA) test [48], participants are asked to solve a number of mental equations under a time-constraint of 60 s. Each equation consists of three parts, starting with a difficult operation (e.g., 36×14), which is followed by two easy operations (e.g., +2 and /2). Participants have to keep the outcome of the first two operations in mind to answer the final outcome (in this example 253) presented as a multiple-choice selection during the last operation. Participants complete in total 20 successive 1 min trials. During the performance of mental equations, participants listened to industrial noise via headphones at 75 dB, 80 dB, or 85 dB. The participants were led to believe that they could manipulate the level of noise presented to them if their task performance met a set criterion. However, the criterion was calculated by taking the average time per equation needed on previous trials, and always adding one extra equation to this average. If participants failed to meet this challenging criterion always set above their ability in the previous trial, the level of noise would be determined by the computer. In earlier studies the MPA has been shown to increase psychological and physiological stress [48, 49]. During the performance of the MPA, participants were video-recorded using a camera and told that the videos would be compared with the videos of their peers. As social evaluation has been proven to enhance HPA axis activity [50]. To ensure a similar subjective experience of loudness of the noise via headphones participants were asked to rate a sound until it reached an eight in loudness on a scale from 1 to 10.
Memory mnemonic discrimination task
The mnemonic discrimination task consisted of 200 color photographs of everyday objects (see Fig. 1B). This task has been validated to assess pattern separation [36, 37]. During the encoding phase the participants memorized everyday objects in a quadrant location (150 items, for 2.5 s, ISI duration is 0.5 s). To ensure attention to the displayed objects, participants are asked to make a decision if they identify the object as an indoor or outdoor object via a button press. During the 15 min rest (termed consolidation) phase participants solved easy word finding puzzles to decrease the likelihood that they were rehearsing the learned lists. Subsequently participants performed the recognition phase, which consisted of 200 images. Participants had to indicate if an object was “old”, i.e., an exact repetition of the object seen during encoding, “similar”, i.e., a lure object which is perceptually similar, but not identical to an object seen during encoding, or “new”, thus not presented during encoding (50 old items, 100 similar items, and 50 new items; presented 2.5 s with and ISI of 0.5 s). If they indicated the object to be “old” or “similar” item, in a next step they had to indicate in which quadrant the original item was shown during encoding, to tax spatial source memory.
Pattern separation performance was assessed by calculating a lure discrimination index (LDI) score. LDI was calculated as the difference between the rate of “similar” responses given to similar lures minus “similar” responses given to the new items (p(“Similar”|lure) –p(“Similar”|new)). This was done to correct for response biases. A low LDI score is indicative of poor pattern separation abilities. Recognition performance was assessed by calculating (p(“Old”|old) –p(“old”|new)).
Salivary sampling and analysis
Alpha-amylase measures were obtained by acquiring four saliva samples. Synthetic Salivette (Sarstedt, Etten-Leur, The Netherlands) swabs were used to collect saliva samples. Participants were instructed to refrain from drinking coffee, tea, or other caffeinated drinks 4 h prior to participation and to not eat or brush their teeth 1 h before participation. Upon arrival, participants were asked to rinse their mouth with water before the first sample was taken. All participants were instructed to keep the Salivette in their mouth for 2 min to acquire enough saliva for further analysis. The saliva samples were stored at –20°C until analysis. After completion of data acquisition all samples were analyzed in one batch for sAA at the Kirschbaum lab Technical University, Dresden. Concentration of sAA in saliva was measured by an enzyme kinetic method: Saliva was processed on a Genesis RSP8/150 liquid handling system (Tecan, Crailsheim, Germany). First, saliva was diluted 1:625 with double-distilled water by the liquid handling system. Twenty microliters of diluted saliva and standard were then transferred into standard transparent 96-well microplates (Roth, Karlsruhe, Germany). Standard was prepared from “Calibrator f.a.s.” solution (Roche Diagnostics, Mannheim, Germany) with concentrations of 326, 163, 81.5, 40.75, 20.38, 10.19, and 5.01 U/l alpha-amylase, respectively, and bidest water as zero standard. After that, 80 ml substrate reagent (a-amylase EPS Sys; Roche Diagnostics, Mannheim, Germany) were pipetted into each well using a multichannel pipette. The microplate containing sample and substrate was then warmed to 37°C by incubation in a water bath for 90 s. Immediately afterward, a first interference measurement was obtained at a wavelength of 405 nm using a standard ELISA reader (Anthos Labtech HT2, Anthos, Krefeld, Germany). The plate was then incubated for another 5 min at 37°C in the water bath, before a second measurement at 405 nm was taken. Increases in absorbance were calculated for unknowns and standards. Increases of absorbance of diluted samples were transformed to alpha-amylase concentrations using a linear regression calculated for each microplate (Graphpad Prism 4.0c for MacOSX, Graphpad Software, San Diego, CA). Taken from Rohleder et al. [51].
Statistical analyses
All statistical analyses were done using R 3.5.2 [52] (http://www.R-project.org/). The median and interquartile range (IQR) of the demographic, neuropsychological characteristics, and SNS activity measures of the participants are reported due to violations of normality. Listwise deletion was applied to handle missing data. For one participant, the LDI score could not be calculated due to technical problems during the memory task on day one. For four participants, the saliva samples at least one of the saliva samples did not return enough saliva for sAA analysis, these participants were therefore not included in the analysis. For all multiple linear models bootstrapping was done using the boot function from the car package [53], using 1000 bootstrap samples, resulting in a 95%confidence interval.
Age and LDI practice effects
Individual SNS reactivity as well as subjective stress ratings were assessed by comparing saliva sAA measures and VAS scores after the MPA task to baseline with a Wilcox signed rank test. The relation between sAA levels and VAS scores and their relation with lure discrimination performance during session two or LDI difference scores were assessed using a Spearman correlation coefficient.
Spearman correlation coefficients were used to explore the relation between age and lure discrimination performance on session one, session two, and the LDI difference score (LDI session 2 –LDI session 1). A sensitivity analysis was performed with age as dichotomous variable. A possible age by sAA interaction on lure discrimination performance during session two was examined with a multiple linear model for sAA levels after MPA task (sAA2) and sAA levels after encoding (sAA3) corrected for sAA levels at baseline (sAA1) with the following models:
1. LDI score∼sAA1 + sAA2 + Age + sAA2:Age
2. LDI score∼sAA1 + sAA3 + Age + sAA3:Age.
Practice effects between the first and second mnemonic discrimination task at group level were tested with a paired Wilcoxon signed-rank test. The main effects of sAA and age on lure discrimination were tested for sAA1, sAA2 and sAA3 corrected for sAA levels at baseline (sAA1) with the following models:
3. LDI difference score∼sAA1 + Age
4. LDI difference score∼sAA1 + sAA2 + Age
5. LDI difference score∼sAA1 + sAA3 + Age
Subsequently a possible interaction effect between sAA levels and age on LDI difference-scores was evaluated with the following multiple linear regression models:
6. LDI difference score∼sAA1 + Age + sAA1:Age
7. LDI difference score∼sAA1 + sAA2 + Age + sAA2:Age
8. LDI difference score∼sAA1 + sAA3 + Age + sAA3:Age
Learners versus non-learners
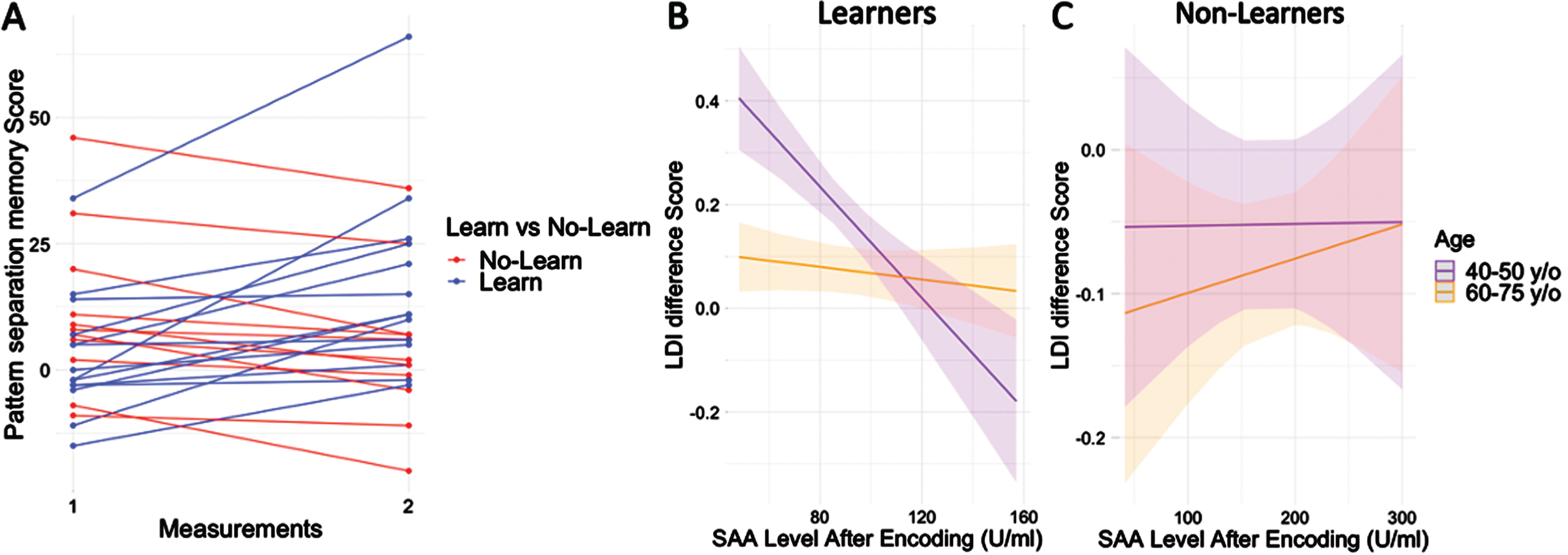
Individual changes in discrimination performance between the first and second session is visualized in Fig. 3A, and a difference-score was computed between the two LDI scores. As post-hoc analyses the LDI difference-score was used to divide the participants into learners (who improved between first and second test, n = 14) and non-learners (who did not improve between first and second test, n = 11). Wilcoxon rank sum test was used to test differences between both groups in continuous variables and sex differences were investigated using chi-square test.
Spearman correlations between sAA levels and LDI difference-scores were computed within learners as well as the non-learners. These correlations were assessed for all sAA time points. Interactions between sAA levels and age on LDI difference-scores were examined repeating equation 7 and 8 within the learning or non-learning group.
RESULTS
Demographic and descriptive statistics
A total of thirty healthy individuals participated in the current study with a median age of 56 years (IQR = [43, 68.75]), of which 43.3%was female (Table 1). On the first mnemonic discrimination task participants had a median LDI of 0.05 (IQR = [–0.03, 0.14]), on the second mnemonic discrimination task the median LDI was 0.08 (IQR = [0.01, 0.14]). There was no significant difference between the first and second LDI score (Z = –1.2, p = 0.22, r = –0.24; Supplementary Figure 1).
Table 1
Characteristics of participants
| Median [IQR] | |
| n | 30 |
| Age (y) | 56 [43.0, 68.75] |
| Sex (%female) | 13, 43.3% |
| Education level | 5 [4, 6] |
| MMSE (score) | 29 [29, 30] |
| WLT (words) | 48 [44.25, 50.75] |
| WLT delayed (words) | 10 [9.0, 11.75] |
| LDST 90 (items) | 52.5 [46, 58] |
| HDRS (score) | 1 [0, 2] |
| Perceived Stress Scale (score) | 8.5 [5.25, 11.75] |
| LDI no stress (score) | 0.05 [–0.03, 0.14] |
| LDI stress (score) | 0.08 [0.01, 0.14] |
| Recognition no stress (score) | 0.52 [0.28, 0.68] |
| Recognition stress (score) | 0.68 [0.57, 0.79] |
| SAA1 (U/ml) | 146.18 [76.2, 320.3] |
| SAA2 (U/ml) | 124.2 [69.1, 209.7] |
| SAA3 (U/ml) | 110.1 [64.5, 153.9] |
| SAA4 (U/ml) | 145.3 [77.9, 210.6]. |
| VAS 1 (score) | 5.00 [0.25, 11.50] |
| VAS 2 (score) | 46.5 [23.25, 70.25] |
| VAS 3 (score) | 12 [3.5, 22.75] |
| VAS 4 (score) | 6.00 [1.0, 15.25] |
IQR, interquartile range; MMSE, Mini-Mental State Exam; WLT, word learning task; LDST, letter digit substitution task; HDRS, Hamilton depression rating scale; LDI, lure discrimination index; SAA, salivary alpha amylase, VAS, visual analog scale.
Age and LDI practice effects
The VAS score after the MPA task was significantly elevated compared to the VAS-score at baseline for the entire group (Z = 4.73, p < 0.001, r = –0.85). We found a trend-level reduction in sAA (Z = –1.89, p = 0.058, r = –0.35) levels for the moment after the MPA task compared to baseline for the whole group. We observed no correlation between the sAA measures and VAS-scores at any time point (range: Rs = [–0.16, 0.25], p = [0.20, 0.98]). There were no significant correlations between VAS-scores and LDI scores (Supplementary Table 1). In the entire group, higher baseline sAA levels correlated with worse practice effects as measured with the LDI difference score (Rs = –0.49, p = 0.01).
There was no significant relation between age and LDI-scores on the first session (Rs = –0.15, p = 0.45; Fig. 2A). Older individuals exhibited worse LDI-scores on the second session (Rs = –0.50, p = 0.01; Fig. 2B), as well as worse LDI difference-scores (Rs = –0.42; p = 0.04; Fig. 2C), for results with age as dichotomous variable see Supplementary Figure 2. We observed a significant main effect of age (b = –0.004, t = –2.15, p = 0.04, Cohen’s f2 = 0.15, BootstrappedCI = [–0.007, –0.001]) and baseline sAA levels (b = –0.0003, t = –2.47, p = 0.02, Cohen’s f2 = 0.22, BootstrappedCI = [–0.0005, –0.0001]) on LDI difference scores. These results show that higher age as well as higher baseline sAA levels are independently related to lower LDI difference scores. There were no significant interactions between age and sAA levels on LDI difference scores at any time point (range: b = [–0.000003, 0.00003], p = [0.25, 0.91]).
Fig. 2
A) Spearman correlation between LDI score during session 1 and age (rs = –0.16). B) Spearman correlation between LDI score during session 2 and age (rs = –0.50). C) Spearman correlation between LDI difference-score between session 1 and 2 and age (rs = –0.42). The regression line estimated with a linear model.
Learners versus non-learners
Visualization of the LDI scores over time revealed a positive slope for 56%(n = 14, “learners”) of the participants and a negative slope for the remaining participants (n = 11, “non-learners”) (Fig. 3A). The time between the performance of the first and second memory mnemonic discrimination task had a median of 14.5 days and an IQR of [8.0, 27.75], with no significant difference between leaners and non-learners (Z = –1.20, p = 0.36, r = –0.22). Demographic characteristics of the groups are described in Table 2. The non-learners showed higher sAA scores at all time points compared to learners. However, subjective stress ratings were not different between the groups for any of the time points (Table 2). Within the groups of learners or non-learners, no significant correlations were found between LDI difference-score and the different sAA levels (Learners: Rs = [–0.34, 0.13], p = [0.23, 0.96]; Non-Learners: Rs = [–0.33, –0.06], p = [0.32, 0.85]).
Fig. 3
A) LDI scores over time plotted for non-leaners (red) and learners (blue). B) For learners, the interaction between age and SAA levels after encoding, showed that in younger individuals, lower sAA levels after encoding were associated with higher practice effects, whereas there was no relationship between sAA and practice effects in the older participants. C) For non-learners, no interaction was found between age and SAA levels after encoding on practice effects. In B and C, sAA at baseline was added as covariate. Age as a continuous variable is depicted in the figures with simple slopes representing the median of the middle aged (43 years old) and older adults (69 years old).
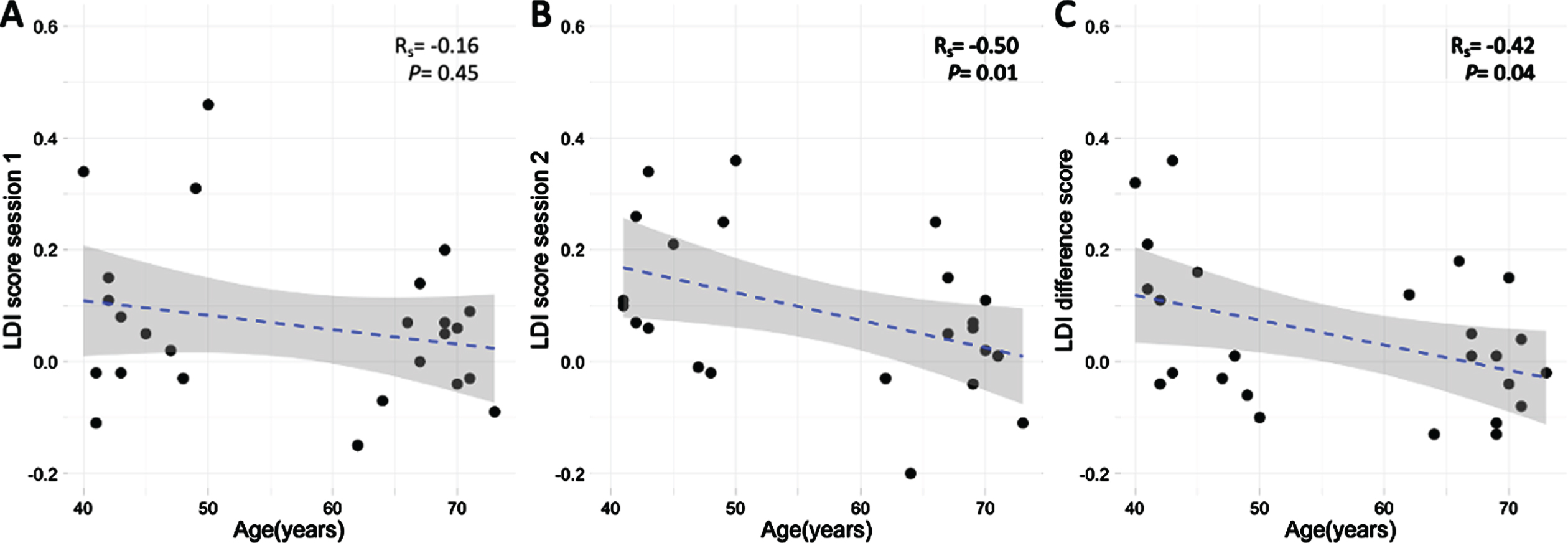
Table 2
Characteristics non learners versus learners
| Non-learners | Learners | Z (r) | |
| Median [IQR] | Median [IQR] | ||
| n | 11 | 14 | - |
| Age (y) | 64 [48, 69.5] | 55 [42.25, 67] | –1.18 (–0.24) |
| Sex (%female) | 45.5% | 35.7% | |
| Education (level) | 5 [4, 5.5] | 5 [4, 6] | –0.45 (–0.09) |
| MMSE (score) | 29 [28,29] | 29.5 [29,30] | –2.0 (–0.40) |
| WLT (words) | 48 [43.5, 49.5] | 48.5 [45, 50.75] | –0.47 (–0.09) |
| WLT delayed (words) | 11 [9.5, 11.5] | 10 [8.5, 11.75] | –0.28 (–0.06) |
| LDST 90 (items) | 48 [45.5, 54.5] | 56 [46.75, 59.50] | –1.34 (–0.27) |
| HDRS (score) | 1 [0, 2] | 0 [0, 1] | –1.05 (–0.21) |
| Perceived Stress (score) | 8 [6, 11.5] | 6 [3.50, 9.75] | –1.32 (–0.26) |
| SAA1 (U/ml) | 321.93 [214.3, 384.18] | 121.25 [70.32, 164.74] | –2.79 (–0.56)** |
| SAA2 (U/ml) | 247.25 [116.69, 327.08] | 87.85 [60.98, 153.58] | –2.49 (–0.50)* |
| SAA3 (U/ml) | 199.84 [116.26, 233.26] | 91.56 [63.07, 111.27] | –2.61 (–0.52)** |
| SAA4 (U/ml) | 185.01 [148.03, 248.08] | 98.75 [59.68, 148.88] | –2.61 (–0.52)** |
| VAS1(score) | 6.0 [1.5, 12.5] | 2.0 [0.0, 10.25] | –1.05 (–0.21) |
| VAS2 (score) | 56.0 [21.5, 72.0] | 33.5 [22.0, 68.0] | –0.33 (–0.07) |
| VAS3 (score) | 8.0 [4.5, 21.0] | 8.50 [0.75, 17.50] | –0.52 (–0.10) |
| VAS4 (score) | 2.0 [1.0, 6.5] | 6.00 [0.25, 15.00] | –0.39 (–0.08) |
| LDI no stress (score) | 0.08 [0.04, 0.16] | –0.01 [–0.03, 0.07] | –1.53 (–0.31) |
| LDI stress (score) | 0.02 [–0.03, 0.07] | 0.11 [0.05, 0.24] | –1.69 (–0.34) |
| Recognition no stress (score) | 0.46 [0.20, 0.59] | 0.64 [0.34, 0.75] | –1.34 (–0.27) |
| Recognition stress (score) | 0.62 [0.55, 0.77] | 0.73 [0.64, 0.82] | –1.32 (–0.26) |
| Nr of days | 9.0 [6.5, 24] | 20 [9.0, 38.5] | –0.69 (–0.14) |
Group differences was tested with the Wilcoxon rank sum test. *p < 0.05; **p < 0.01. MMSE, Mini-Mental State Exam; WLT, word learning task; LDST, letter digit substitution task; HDRS, Hamilton Depression Rating Scale; SAA, salivary alpha amylase, VAS, visual analog scale, LDI, lure discrimination index.
Within the group of learners, we observed a significant interaction between age and sAA levels after encoding on LDI difference scores while controlling for sAA baseline levels (b = 0.02, t = 4.35, p = 0.0019, Cohen’s f2 = 0.47, BootstrappedCI = [0.004, 0.04]; Fig. 3B). No such interaction was observed for the non-learners (b = 0.0009, t = 0.56, p = 0.6, Cohen’s f2 = 0.04, BootstrappedCI = [–0.009, 0.02]; Fig. 3C). The sAA levels after MPA task did not return a significant interaction with age for either learners (b = 0.00005, t = 1.5, p = 0.17, Cohen’s f2 = 0.09, BootstrappedCI = [–0.0005, 0.00001]) or non-learners (b = 2.56e–06, t = 0.39, p = 0.7, Cohen’s f2 = 0.02, BootstrappedCI = [–0.00007, 0.00002]. For results with age group as dichotomous variable see Supplementary Figure 3.
DISCUSSION
In the current pilot study, we examined the relation between age and practice effects and how this is modified by individual sympathetic nervous system reactivity. Previous studies demonstrated that activation of the sympathetic system, as measured with salivary alpha-amylase—a proxy for norepinephrine—is associated with better performance on a memory tasks in younger individuals. In the current study, we took a novel approach by exploring practice effects, which can be more sensitive to subtle cognitive deficits. Our results show that higher age as well as higher baseline SNS activity are related to lower LDI practice scores. Additionally, non-learners exhibited elevated sAA levels compared to the learners on all time points, possibly indicative of a perpetual physiological hyperactive SNS in the non-learners. These findings, while preliminary, suggest that hyperactivation of the SNS may be a promising factor as an early marker of imminent cognitive decline.
Consistent with other studies, we showed a negative relation between age and LDI practice effects [3, 15]. We extended these observations by showing that higher baseline SNS activity predicts independently of age lower practice effects. Higher baseline SNS activity may thus be a factor contributing to individual variability in cognitive performance [17, 54]. Furthermore, the fact that non-learners exhibited elevated sAA levels compared to the learners on all time points is indicative of a perpetual physiological hyperactive SNS in the non-learners. Interestingly, non-learners did not report higher subjective stress experiences compared to learners, suggesting that the elevated sAA levels are predominantly physiologic in nature and not related to an increased subjective feeling of stress or a heightened anticipatory stress response toward participation. These observations may signify the importance of SNS activity as predictor of cognitive decline. In particular, the locus coeruleus (LC), the major source of noradrenaline in the brain, is one of the earliest regions accumulating hyperphosphorylated tau [55], and higher LC integrity in older adults has been related to more proficient memory functioning [56]. Prior studies also demonstrated that higher noradrenaline activity was related to lower cognitive functioning in healthy older individuals [54] and patients with Alzheimer’s disease [57–60].
We did not find an interaction between age and SNS activity on practice effects in the entire group. However, further exploration revealed that predominantly the middle-aged learners benefited from lower sAA levels as reflected in their higher practice effects. Given the small sample size, we need to be cautious in interpreting possible subgroup patterns, but we speculate that in learners the magnitude of learning is modified by age and variability in SNS activation. This relation could possibly mimic the canonical Yerkes-Dodson relationship, and that the shape or width of these curves may be altered by age, individual variability in the state of the SNS system or accumulation of brain pathology. For example, younger learners with lower sAA levels are performing well because they are located at the top of their inverted U-curve [61]. In contrast, the younger learners with higher sAA levels demonstrate lower practice scores, as their sAA levels have surpassed the optimal peak. For older learners, we did not see an effect of sAA on performance, consistent with previous work [33]. We also like to note that the range of sAA was higher in non-learners as compared to learners, further exemplifying the nonlinear relationship between SNS activity and learning. Future studies in larger groups with more observations over time in combination with the use of biomarker data are warranted to substantiate this hypothesis. This will allow for measurement of intra-individual variability in SNS activity as one contributing factor to the observed variability in practice effects while using a similar a paradigm.
Our study has several limitations that are worth noting. The sample size of the study was relatively small, but we succeeded in detecting relatively large effects allowing us to better generalize our results to the population. Additionally, we did not collect saliva samples during the first part of the study and therefore we cannot explore the relation between sAA and subjective stress on lure discrimination performance during session one or confirm that the higher baseline sAA levels were also present during the first part of the study. Including a control group could further help disentangle the effect of SNS activation on practice effects, as sAA levels immediately after the arithmetic task were not related to memory performance. Furthermore, there are some limitations to interpreting findings of sAA as a measure of sympathetic system activation, as sAA might be a reflection of both sympathetic and parasympathetic activity [62]. An important factor is the parasympathetic stimulation of salivary flow rate, which can possibly bias sAA secretion. The influence of flow rate on sAA secretion might be dependent on the research condition. However, the effect of salivary flow rate under stressful conditions on sAA output is most likely small [51]. Consistent with previous work showing a lack in practice effects on lure discrimination tasks, we observed no significant changes in LDI scores at a group-level [63, 64]. However, individual variability in LDI scores over time was related to baseline sAA levels, indicating that individual factors can result in different cognitive profiles. Finally, practice scores in our study were determined over two test sessions which makes it vulnerable to measurement errors. Therefore, future research into practice effects could benefit from psychometric characterization of practice scores, ideally over more than two observations per person. The mechanism relating elevated SNS activation to attenuated practice effects needs to be further explored by collecting biochemical, imaging, and cognitive assessments.
In conclusion, this pilot study demonstrated that elevated baseline SNS activity independently of age was related to worse practice effects on a well-established mnemonic discrimination task. In a subgroup of learners, learning was predominantly observed in middle-aged individuals with lower SNS activity. Learning subsequently decreased under higher SNS activity. These findings suggest that hyperactivation of the SNS may hold promise as one potential factor to further explain individual variability in practice scores and possibly as indicator of imminent cognitive decline. More research on the interactions between the variability in SNS activation, age, and practice effects in cognition is warranted especially in the context of markers of neurodegenerative disorders, given the relevance for early detection of cognitive decline and possibly improving participant selection to clinical trials aimed at preventing dementia.
ACKNOWLEDGMENTS
We want to thank Rob Markus for sharing the MPA task software with us.
Acquisition of the data was funded by intramural support from the Faculty of Health Medicine and Life Sciences of Maastricht University to HILJ. HILJ is also supported by the NIH-National Institute on Aging grant R01AG062559. MAY is supported by US National Institute on Aging grants P50AG05146, R21AG049220, and R01AG053555.
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/20-0783r2).
SUPPLEMENTARY MATERIAL
[1] The supplementary material is available in the electronic version of this article: https://dx.doi.org/10.3233/JAD-200783.
REFERENCES
[1] | Hassenstab J , Ruvolo D , Jasielec M , Xiong C , Grant E , Morris JC ((2015) ) Absence of practice effects in preclinical Alzheimer’s disease. Neuropsychology 29: , 940–948. |
[2] | Jonaitis EM , Koscik RL , La Rue A , Johnson SC , Hermann BP , Sager MA ((2015) ) Aging, practice effects, and genetic risk in the Wisconsin Registry for Alzheimer’s Prevention. Clin Neuropsychol 29: , 426–441. |
[3] | Calamia M , Markon K , Tranel D ((2012) ) Scoring higher the second time around: Meta-analyses of practice effects in neuropsychological assessment. Clin Neuropsychol 26: , 543–570. |
[4] | McCaffrey RJ , Duff K , Westervelt HJX ((2000) ) Practitioner’s guide to evaluating change with neuropsychological assessment instruments, Springer Science & Business Media. |
[5] | Machulda MM , Hagen CE , Wiste HJ , Mielke MM , Knopman DS , Roberts RO , Vemuri P , Lowe VJ , Jack CR , Petersen RC ((2016) ) Practice effects and longitudinal cognitive change in clinically normal older adults differ by Alzheimer imaging biomarker status. Clin Neuropsychol 31: , 99–117. |
[6] | Davis H , Small S , Stern Y , Mayeux R , Feldstein S , Keller F ((2003) ) Acquisition, recall, and forgetting of verbal information in long-term memory by young, middle-aged, and elderly individuals. Cortex 39: , 1063–1091. |
[7] | Huh TJ , Kramer JH , Gazzaley A , Delis DC ((2006) ) Response bias and aging on a recognition memory task. J Int Neuropsychol Soc 12: , 1–7. |
[8] | Ponds RWHM , Commissaris KJAM , Jolles J ((1997) ) Prevalence and covariates of subjective forgetfulness in a normal population in The Netherlands. Int J Aging Hum Dev 45: , 207–221. |
[9] | Stevens FC , Kaplan CD , Ponds RW , Diederiks JP , Jolles J ((1999) ) How ageing and social factors affect memory. Age Ageing 28: , 379–384. |
[10] | Rönnlund M , Lövdén M , Nilsson L-G ((2007) ) Cross-sectional versus longitudinal age gradients of tower of hanoi performance: The role of practice effects and cohort differences in education. Aging Neuropsychol Cogn 15: , 40–67. |
[11] | Van Der Elst WIM , Van Boxtel MPJ , Van Breukelen GJP , Jolles J ((2007) ) Detecting the significance of changes in performance on the Stroop Color-Word Test, Rey’s Verbal Learning Test, and the Letter Digit Substitution Test: The regression-based change approach. J Int Neuropsychol Soc 14: , 71–80. |
[12] | Kudielka BM , Buske-Kirschbaum A , Hellhammer DH , Kirschbaum C ((2004) ) Differential heart rate reactivity and recovery after psychosocial stress (TSST) in healthy children, younger adults, and elderly adults: The impact of age and gender. Int J Behav Med 11: , 116–121. |
[13] | Kudielka BM , Hellhammer DH , Wust S ((2009) ) Why do we respond so differently? Reviewing determinants of human salivary cortisol responses to challenge. Psychoneuroendocrinology 34: , 2–18. |
[14] | Strahler J , Mueller A , Rosenloecher F , Kirschbaum C , Rohleder N ((2010) ) Salivary alpha-amylase stress reactivity across different age groups. Psychophysiology 47: , 587–595. |
[15] | Bartels C , Wegrzyn M , Wiedl A , Ackermann V , Ehrenreich H ((2010) ) Practice effects in healthy adults: A longitudinal study on frequent repetitive cognitive testing. BMC Neurosci 11: , 118. |
[16] | Aggarwal NT , Wilson RS , Beck TL , Rajan KB , Mendes de Leon CF , Evans DA , Everson-Rose SA ((2014) ) Perceived stress and change in cognitive function among adults 65 years and older. Psychosom Med 76: , 80–85. |
[17] | McEwen BS ((2002) ) Sex, stress and the hippocampus: Allostasis, allostatic load and the aging process. Neurobiol Aging 23: , 921–939. |
[18] | Stawski RS , Sliwinski MJ , Smyth JM ((2006) ) Stress-related cognitive interference predicts cognitive function in old age. Psychol Aging 21: , 535–544. |
[19] | Ivnik RJ , Smith GE , Lucas JA , Petersen RC , Boeve BF , Kokmen E , Tangalos EG ((1999) ) Testing normal older people three or four times at 1-to 2-year intervals: Defining normal variance. Neuropsychology 13: , 121. |
[20] | Benedict RH , Zgaljardic DJ ((1998) ) Practice effects during repeated administrations of memory tests with and without alternate forms. J Clin Exp Neuropsychol 20: , 339–352. |
[21] | Rienacker F , Jacobs HIL , Van Heugten CM , Van Gerven PWM ((2018) ) Practice makes perfect: High performance gains in older adults engaged in selective attention within and across sensory modalities. Acta Psychol (Amst) 191: , 101–111. |
[22] | Machulda MM , Pankratz VS , Christianson TJ , Ivnik RJ , Mielke MM , Roberts RO , Knopman DS , Boeve BF , Petersen RC ((2013) ) Practice effects and longitudinal cognitive change in normal aging vs. incident mild cognitive impairment and dementia in the Mayo Clinic Study of Aging. Clin Neuropsychol 27: , 1247–1264. |
[23] | Samaroo A , Amariglio RE , Burnham S , Sparks P , Properzi M , Schultz AP , Buckley R , Johnson KA , Sperling RA , Rentz DM , Papp KV ((2020) ) Diminished Learning Over Repeated Exposures (LORE) in preclinical Alzheimer’s disease. Alzheimers Dement (Amst) 12: , e12132. |
[24] | Sapolsky RM , Romero LM , Munck AU ((2000) ) How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocrine Rev 21: , 55–89. |
[25] | Nater UM , Rohleder N ((2009) ) Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: Current state of research. Psychoneuroendocrinology 34: , 486–496. |
[26] | Nater UM , Rohleder N , Gaab J , Berger S , Jud A , Kirschbaum C , Ehlert U ((2005) ) Human salivary alpha-amylase reactivity in a psychosocial stress paradigm. Int J Psychophysiol 55: , 333–342. |
[27] | Chatterton RT Jr , Vogelsong KM , Lu Yc , Ellman AB , Hudgens GA ((1996) ) Salivary α-amylase as a measure of endogenous adrenergic activity. Clin Physiol 16: , 433–448. |
[28] | Schoofs D , Hartmann R , Wolf OT ((2009) ) Neuroendocrine stress responses to an oral academic examination: No strong influence of sex, repeated participation and personality traits. Stress 11: , 52–61. |
[29] | Strahler J , Berndt C , Kirschbaum C , Rohleder N ((2010) ) Aging diurnal rhythms and chronic stress: Distinct alteration of diurnal rhythmicity of salivary α-amylase and cortisol. Biol Psychol 84: , 248–256. |
[30] | Hidalgo V , Almela M , Villada C , Salvador A ((2014) ) Acute stress impairs recall after interference in older people, but not in young people. Horm Behav 65: , 264–272. |
[31] | Seals DR , Dinenno FA ((2004) ) Collateral damage: Cardiovascular consequences of chronic sympathetic activation with human aging. Am J Physiol Heart Circ Physiol 287: , H1895–1905. |
[32] | Segal SK , Stark SM , Kattan D , Stark CE , Yassa MA ((2012) ) Norepinephrine-mediated emotional arousal facilitates subsequent pattern separation. Neurobiol Learn Mem 97: , 465–469. |
[33] | Hidalgo V , Pulopulos MM , Salvador A ((2019) ) Acute psychosocial stress effects on memory performance: Relevance of age and sex. Neurobiol Learn Mem 157: , 48–60. |
[34] | Joëls M , Krugers H , Karst H ((2007) ) Stress-induced changes in hippocampal function. Prog Brain Res 167: , 3–15. |
[35] | Roozendaal B , McEwen BS , Chattarji S ((2009) ) Stress, memory and the amygdala. Nat Rev Neurosci 10: , 423–433. |
[36] | Kim J , Yassa MA ((2013) ) Assessing recollection and familiarity of similar lures in a behavioral pattern separation task. Hippocampus 23: , 287–294. |
[37] | Stevenson RF , Reagh ZM , Chun AP , Murray EA , Yassa MA ((2020) ) Pattern separation and source memory engage distinct hippocampal and neocortical regions during retrieval. J Neurosci 40: , 843–851. |
[38] | Leal SL , Yassa MA ((2015) ) Neurocognitive aging and the hippocampus across species. Trends Neurosci 38: , 800–812. |
[39] | Leal SL , Yassa MA ((2018) ) Integrating new findings and examining clinical applications of pattern separation. Nat Neurosci 21: , 163–173. |
[40] | Stark SM , Yassa MA , Lacy JW , Stark CE ((2013) ) A task to assess behavioral pattern separation (BPS) in humans: Data from healthy aging and mild cognitive impairment. Neuropsychologia 51: , 2442–2449. |
[41] | Cohen S , Kamarck T , Mermelstein R (1994) Perceived stress scale. Measuring stress: A guide for health and social scientists. |
[42] | Hamilton M ((1960) ) A rating scale for depression. J Neurol Neurosurgery Psychiatry 23: , 56–62. |
[43] | Costa PT , McCrae RR (1992) Revised NEO Personality Inventory (NEO PI-R) and NEO Five-Factor Inventory (NEO_FFI): Professional manual/, Odessa, FL. |
[44] | Gallagher DJ ((1990) ) Extraversion, neuroticism and appraisal of stressful academic events. Pers Individ Dif 11: , 1053–1057. |
[45] | Luteijn F , Bouman TK ((1988) ) The concepts of depression, anxiety, and neuroticism in questionnaires. Eur J Pers 2: , 113–120. |
[46] | Wilson RS , Arnold SE , Schneider JA , Kelly JF , Tang Y , Bennett DA ((2006) ) Chronic psychological distress and risk of Alzheimer’s disease in old age. Neuroepidemiology 27: , 143–153. |
[47] | Wilson RS , Arnold SE , Schneider JA , Li Y , Bennett DA ((2007) ) Chronic distress, age-related neuropathology, and late-life dementia. Psychosom Med 69: , 47–53. |
[48] | Peters ML , Godaert GLR , Ballieux RE , van Vliet M , Willemsen JJ , Sweep FCGJ , Heijnen CJ ((1998) ) Cardiovascular and endocrine responses to experimental stress: Effects of mental effort and controllability. Psychoneuroendocrinology 23: , 1–17. |
[49] | Markus CR , Verschoor E , Firk C , Kloek J , Gerhardt CC ((2010) ) Effect of tryptophan-rich egg protein hydrolysate on brain tryptophan availability, stress and performance. Clin Nutr 29: , 610–616. |
[50] | Dickerson SS , Kemeny ME ((2004) ) Acute stressors and cortisol responses: A theoretical integration and synthesis of laboratory research. Psychol Bull 130: , 355–391. |
[51] | Rohleder N , Wolf JM , Maldonado EF , Kirschbaum C ((2006) ) The psychosocial stress-induced increase in salivary alpha-amylase is independent of saliva flow rate. Psychophysiology 43: , 645–652. |
[52] | Team RC (2016) R: A language and environment for statistical computing [Computer software manual]. Vienna, Austria. |
[53] | Fox J , Weisberg S ((2018) ) An R companion to applied regression, Sage Publications. |
[54] | Wang LY , Murphy RR , Hanscom B , Li G , Millard SP , Petrie EC , Galasko DR , Sikkema C , Raskind MA , Wilkinson CW , Peskind ER ((2013) ) Cerebrospinal fluid norepinephrine and cognition in subjects across the adult age span. Neurobiol Aging 34: , 2287–2292. |
[55] | Braak H , Thal DR , Ghebremedhin E , Del Tredici K ((2011) ) Stages of the pathologic process in Alzheimer disease: Age categories from 1 to 100 years. J Neuropathol Exp Neurol 70: , 960–969. |
[56] | Dahl MJ , Mather M , Duzel S , Bodammer NC , Lindenberger U , Kuhn S , Werkle-Bergner M ((2019) ) Rostral locus coeruleus integrity is associated with better memory performance in older adults. Nat Hum Behav 3: , 1203–1214. |
[57] | Pascualy M , Petrie EC , Brodkin K , Peskind ER , Wilkinson CW , Raskind MA ((2000) ) Hypothalamic pituitary adrenocortical and sympathetic nervous system responses to the cold pressor test in Alzheimer’s disease. Biol Psychiatry 48: , 247–254. |
[58] | Wang LY , Raskind MA , Wilkinson CW , Shofer JB , Sikkema C , Szot P , Quinn JF , Galasko DR , Peskind ER ((2018) ) Associations between CSF cortisol and CSF norepinephrine in cognitively normal controls and patients with amnestic MCI and AD dementia. Int J Geriatr Psychiatry 33: , 763–768. |
[59] | Elrod R , Peskind ER , DiGiacomo L , Brodkin KI , Veith RC , Raskind MA ((1997) ) Effects of Alzheimer’s disease severity on cerebrospinal fluid norepinephrine concentration. Am J Psychiatry 154: , 25–30. |
[60] | Jacobs HIL , Riphagen JM , Ramakers I , Verhey FRJ (2019) Alzheimer’s disease pathology: Pathways between central norepinephrine activity, memory, and neuropsychiatric symptoms. Mol Psychiatry, doi: 10.1038/s41380-019-0437-x |
[61] | Aston-Jones G , Cohen JD ((2005) ) An integrative theory of locus coeruleus-norepinephrine function: Adaptive gain and optimal performance. Annu Rev Neurosci 28: , 403–450. |
[62] | Bosch JA , Veerman EC , de Geus EJ , Proctor GB ((2011) ) alpha-Amylase as a reliable and convenient measure of sympathetic activity: Don’t start salivating just yet!. Psychoneuroendocrinology 36: , 449–453. |
[63] | Clark R , Tahan AC , Watson PD , Severson J , Cohen NJ , Voss M ((2017) ) Aging affects spatial reconstruction more than spatial pattern separation performance even after extended practice. Hippocampus 27: , 716–725. |
[64] | Liu KY , Gould RL , Coulson MC , Ward EV , Howard RJ ((2016) ) Tests of pattern separation and pattern completion in humans-A systematic review. Hippocampus 26: , 705–717. |




