Why Is Amyloid-β PET Requested After Performing CSF Biomarkers?
Abstract
Background:
Amyloid-β positron emission tomography (PET) and cerebrospinal fluid (CSF) Aβ42 are considered interchangeable for clinical diagnosis of Alzheimer’s disease.
Objective:
To explore the clinical reasoning for requesting additional amyloid-β PET after performing CSF biomarkers.
Methods:
We retrospectively identified 72 memory clinic patients who underwent amyloid-β PET after CSF biomarkers analysis for clinical diagnostic evaluation between 2011 and 2019. We performed patient chart reviews to identify factors which led to additional amyloid-β PET. Additionally, we assessed accordance with appropriate-use-criteria (AUC) for amyloid-β PET.
Results:
Mean patient age was 62.0 (SD = 8.1) and mean Mini-Mental State Exam score was 23.6 (SD = 3.8). CSF analysis conflicting with the clinical diagnosis was the most frequent reason for requesting an amyloid-β PET scan (n = 53, 74%), followed by incongruent MRI (n = 16, 22%), unusual clinical presentation (n = 11, 15%) and young age (n = 8, 11%). An amyloid-β PET scan was rarely (n = 5, 7%) requested in patients with a CSF Aβ+/tau+ status. Fifteen (47%) patients with a post-PET diagnosis of AD had a predominantly non-amnestic presentation. In n = 11 (15%) cases, the reason that the clinician requested amyloid-β was not covered by AUC. This happened most often (n = 7) when previous CSF analysis did not support current clinical diagnosis, which led to requesting amyloid-β PET.
Conclusion:
In this single-center study, the main reason for requesting an amyloid-β PET scan after performing CSF biomarkers was the occurrence of a mismatch between the primary clinical diagnosis and CSF Aβ/tau results.
INTRODUCTION
Two methods are currently employed in the clinic to capture in vivo amyloid-β pathology, a pathological hallmark of Alzheimer’s disease (AD) [1, 2]. Aβ42 levels in cerebrospinal fluid (CSF) reflect the soluble amyloid-β pool that has been shown to correlate with amyloid-β depositions in the brain [3]. Alternatively, amyloid-β positron emission tomography (PET) can be employed to directly visualize parenchymal fibrillary amyloid-β depositions [4]. Although CSF and PET yield conflicting results in 10–20% of patients [5–7], they are nonetheless considered interchangeable for clinical use [8].
In our center, all patients are offered CSF biomarker analysis. However, despite the availability of CSF biomarkers, occasionally amyloid-β PET-scans are requested. The diagnostic value of amyloid-β PET to a standard dementia screening has been established in many studies [9–11], but few studies have included subgroups of people with available CSF biomarkers. Reported reasons for performing amyloid-β PET in such cases included incongruent CSF biomarkers in patients with suspicion of AD or atypical clinical presentation [12, 13]. We aimed to elucidate this practice by exploring the clinical reasoning for requesting amyloid-β PET after CSF biomarkers were disclosed, and to characterize the population that received amyloid-β PET after CSF examination.
Additionally, appropriate use criteria (AUC) for amyloid-β PET have been published to support the implementation of clinical amyloid-β PET, advocating use in three groups most likely to benefit: patients with an atypical clinical presentation or mixed etiology, persistent unexplained mild cognitive impairment (MCI), and unexplained dementia in young patients [14]. As a secondary goal, we aimed to compare our clinical practice against current amyloid-β PET AUC.
METHODS
Patient inclusion
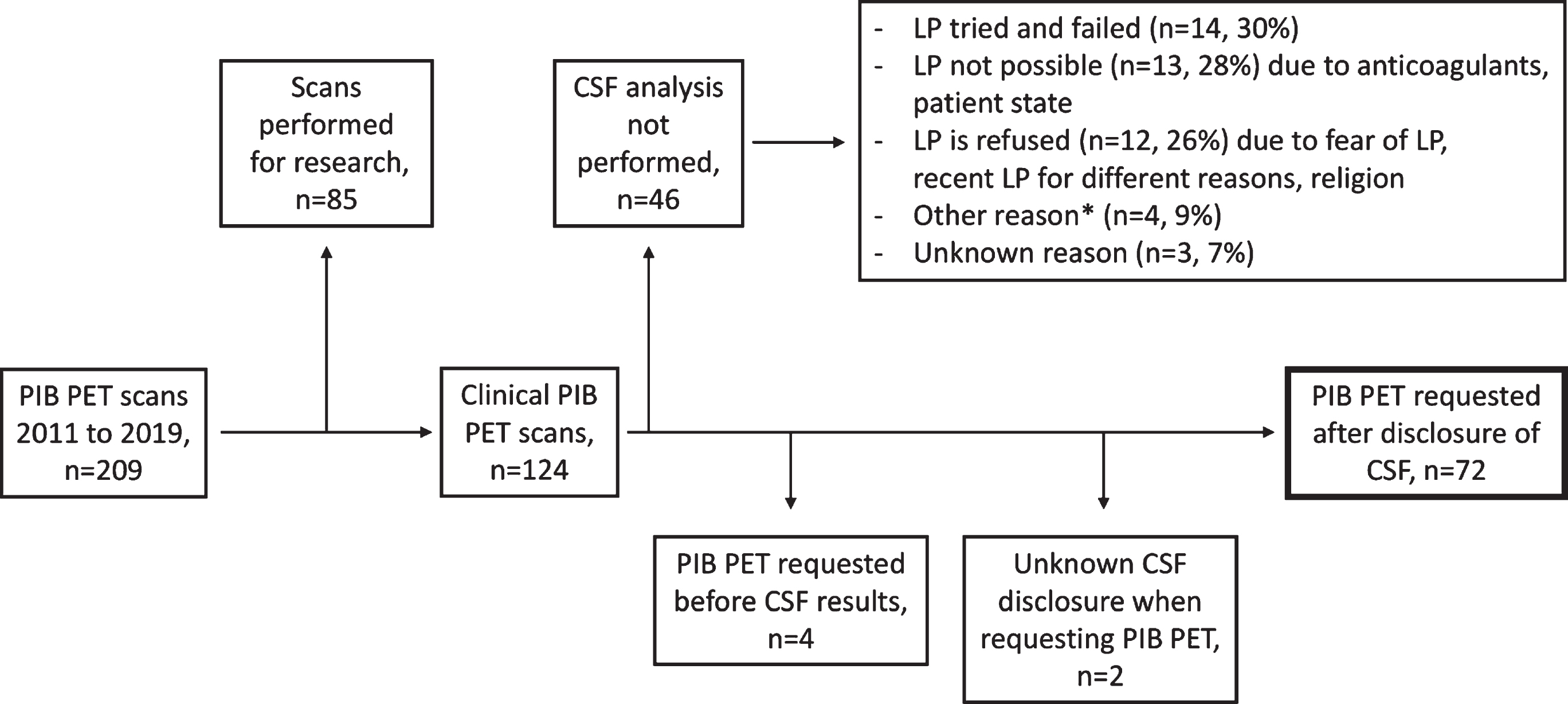
We identified 209 cases from the Amsterdam Dementia Cohort with a [11C]-Pittsburgh Compound B (PIB) PET scan between 2011 and 2019 (Fig. 1). We excluded n = 85 cases who underwent PIB-PET for research purposes, n = 4 cases with CSF analysis performed after amyloid-β PET, and n = 2 cases with unknown CSF disclosure. Although lumbar puncture (LP) is offered to all patients visiting our center, it was not performed for n = 46 patients, most often because LP was either not successful (n = 14, 30%), not possible (n = 13, 28%), or refused by the patient (n = 12, 26%). Finally, we included 72 cases with a clinically requested amyloid-β PET scan performed after CSF biomarkers examination for our analysis.
Fig.1
Flowchart for patient inclusion. *Other reasons include normal pressure hydrocephalus, increased certainty received from amyloid-β PET imaging, and imaging having a greater influence on convincing patients. CSF, cerebrospinal fluid; LP, lumbar puncture; PIB PET, positron emission tomography with 11C-Pittsburgh compound B.
Cerebrospinal fluid
CSF was obtained by LP, using a 25-gauge needle and a syringe [15]. Samples were collected in polypropylene collection tubes and centrifuged at 1800 g for 10 min at 4°C. Thereafter, samples were frozen at –20°C until routine biomarker analysis. Manual analyses of Aβ42, total tau, and phosphorylated tau (p-tau) were performed using sandwich ELISAs (Innotest assays: β-amyloid1-42, tTAU-Ag and PhosphoTAU-181p; Fujirebio) in the Neurochemistry Laboratory of the Department of Clinical Chemistry of Amsterdam UMC. In a few cases, CSF analysis was performed using automated assays for Aβ42 (n = 9), t-tau (n = 1), and p-tau (n = 1), due to change in routine methods (Elecsys CSF, Roche Diagnostics GmbH) [16]. Additionally, in n = 12 cases analyses were performed in the Department of Laboratory Medicine in Radboud UMC prior to referral to our center.
The clinical cut-off values for CSF Aβ42 have repeatedly been changed over the years due to the gradual upward drift of median CSF Aβ42 values observed in our cohort, possibly due to changes in ELISA kits and/or calibration data that are influenced by the presence of Aβ42 aggregates [17]. In order to pool all available CSF values (both local and external) in relation to different cut-offs, we created standardized values by calculating, per patient, the percentage of the CSF value relative to its concurrent cut-off. For example, a value of 150 would represent a normal CSF Aβ42 being 50% higher than the cut-off, whereas a value of 80 represented a pathologically decreased CSF Aβ42 being 20% below the cut-off value.
Positron emission tomography
Amyloid-β PET is not part of the standard diagnostic process in the Amsterdam Dementia Cohort, therefore most of the amyloid-β PET scans in our center are performed for research purposes. We only included scans with [11C]-PIB, as clinically requested PET scans in our center are routinely performed using [11C]-PIB as the radiotracer. These scans were performed using the following PET scanners: ECAT EXACT HR+ scanner (Siemens Healthcare, Germany) and Gemini TF PET/CT or Ingenuity TF PET-CT (Philips Medical Systems, the Netherlands). PET scans were performed within a median of 140 [IQR = 67, 260] days after LP. PET scans were rated as positive or negative based on visual read by an expert nuclear medicine physician [11]. Although intra-rater agreement was not available for this sample, in previous work using [11C]-PIB PET, our nuclear medicine physician showed excellent (Fleiss k = 0.88) and good to moderate (Fleiss k = 0.59 and 0.68) inter-reader agreement for standardized uptake value (SUV), SUV ratio and non-displaceable binding potential images, respectively [18].
Magnetic resonance imaging (MRI)
MRI was performed as described previously [19]. The scans were visually assessed by a neuroradiologist on three different image planes for posterior cortical atrophy [20], medial temporal atrophy [21], and global cortical atrophy [22], which were thereafter age-normalized [23]. The extent of white matter hyperintensities was rated according to the Fazekas scale [24]. Additionally, the scans were assessed for the existence of lacunes and microbleeds. An external scan was used in n = 19 cases, and MRI was not available in n = 2 cases (n = 1 with available computed tomography).
Neuropsychological testing
Patients underwent extensive neuropsychological testing as part of their diagnostic process. We used Mini-Mental State Examination (MMSE) scores to measure global cognition. Additionally, we derived z-scores of various neuropsychological tests using the mean and standard deviation values from a group of healthy controls (n = 360), whose mean age was 57.8 (standard deviation [SD] = 8.3) and mean MMSE was 28.2 (SD = 1.9). Thereafter, we compiled composite scores for five cognitive domains (memory, language, attention, executive, and visuospatial) [25].
Reasons for requesting amyloid-β PET
Our main objective was to explore the clinical reasoning for requesting an amyloid-β PET after disclosure of CSF biomarkers. Therefore, J.R. and F.Bo. performed patient chart reviews to retrieve the clinical reasoning for requesting the amyloid-β PET scan. Patients were divided into two groups (AD versus non-AD) based on the most likely etiological diagnosis prior to performing a PIB PET scan. For both diagnostic groups we listed characteristics that were recorded as not compatible with the current etiological diagnosis, therefore leading to additional amyloid-β PET. Listed reasons included incongruent findings from biomarkers (CSF, imaging, EEG) or patient history and presentation, as well as other supporting factors such as age, patient wish and implementation of a new CSF assay. For example, in the AD group we labelled CSF as a reason for additional amyloid-β PET when a normal CSF analysis or a CSF analysis with isolated low Aβ42 or high tau/p-tau did not support the clinical diagnosis. Similarly, we labelled MRI findings as a reason for additional amyloid-β PET in the non-AD group when a normal MRI or pronounced hippocampal atrophy decreased confidence in the current clinical diagnosis.
Accordance to amyloid-β PET appropriate use criteria
Previously published appropriate use criteria support amyloid-β imaging in case of 1) progressive unexplained MCI, 2) possible AD with atypical or etiologically mixed presentation, and 3) progressive dementia at an early age, usually defined as below the age of 65 [14]. Based on examining patient charts, we determined for each case accordance with the PET appropriate use criteria.
Patient population
For all patients, we determined an initial available etiological diagnosis as the first diagnosis, the first available diagnosis after amyloid-β PET as the last diagnosis, and diagnostic change as the difference between the two. In n = 23 (32%) cases, CSF analysis was performed prior to referral to our center (n = 14 with Aβ+/tau– or Aβ–/tau+ based on CSF) and n = 3 (4%) patients had undergone a previous amyloid-β PET scan.
We present our patient population by the binarized status based on CSF Aβ42 and total tau. We chose to use CSF total tau instead of p-tau in order to closely resemble clinical decision-making. Our data showed that in case of dubious diagnosis, increased levels of either CSF total tau or p-tau facilitated further diagnostics and there were more patients with an isolated increase of CSF tau (n = 8) than p-tau (n = 3). CSF tau and p-tau status were identical in n = 61 (85%) of patients.
Statistical analysis
Statistical analysis was performed using R software (Version 3.4.4) [26–29]. We compared patient features using Chi-squared tests, two samples t-tests, Wilcoxon Rank-Sum tests and linear regression models. Cognitive scores were compared while adjusting for age, sex, and education.
RESULTS
Demographics
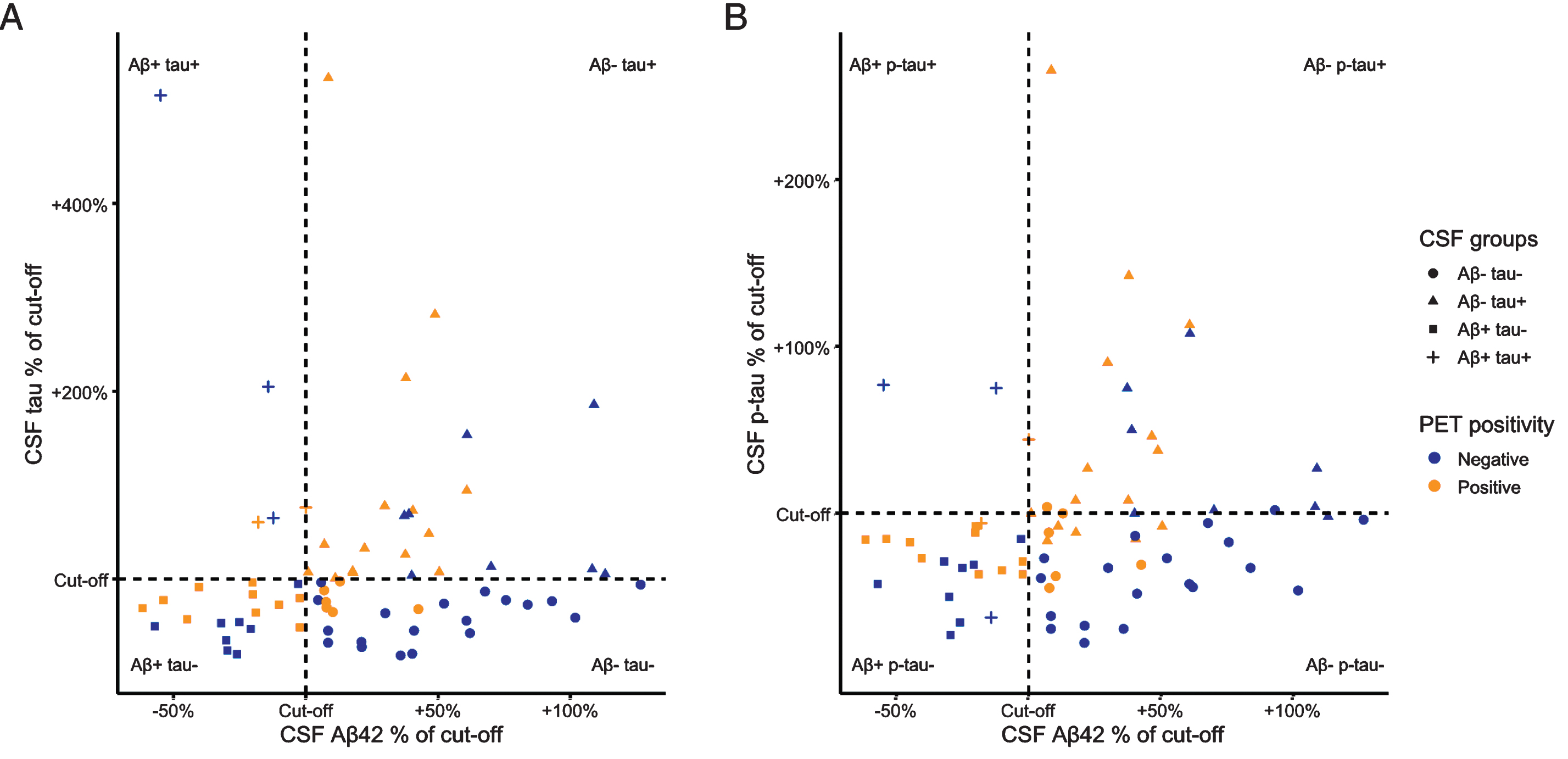
We included n = 5 (7%) patients with subjective cognitive decline (SCD), n = 3 (4%) whose symptoms were mainly associated with a psychiatric condition, n = 16 (22%) with MCI, and n = 48 (67%) with dementia. The average age in our patient cohort was 62.0 (standard deviation [SD] = 8.1), n = 46 (64%) of patients were male and average MMSE was 23.6 (SD = 3.8) (Table 1). Most patients where a clinical PIB-scan was requested were Aβ–/tau– (n = 25, 35%), Aβ–/tau+ (n = 23, 32%), or Aβ+/tau– (n = 19, 26%), while only a minority 5 (7%) had an Aβ+/tau+ status based on CSF (Fig. 2). In total, n = 34 (47%) patients were amyloid-positive based on PET (compared to n = 24 (33%) based on CSF Aβ42). Amyloid-β CSF and PET status were discordant in n = 32 (44%) cases. Amyloid-β PET positivity was lower in the Aβ–/tau– group (n = 6, 24%) compared to the Aβ–/tau+ (n = 15, 65%; p = 0.01) and Aβ+/tau– (n = 11, 58%; p = 0.048) groups. We found no significant differences in cognitive scores and MRI measures between the groups.
Table 1
Patient population stratified by binarized CSF Aβ42 and tau status
| TOTAL | Normal CSF | Conflicting CSF Aβ and tau | AD-like CSF | ||
| Aβ–/tau– | Aβ–/tau+ | Aβ+/tau– | Aβ+/tau+ | ||
| n (%) | 72 | 25 (35) | 23 (32) | 19 (26%) | 5 (7) |
| Sex, male (%) | 46 (64) | 17 (68) | 14 (61) | 12 (63) | 3 (60) |
| Age (mean (SD)) | 62.0 (8.1) | 63.9 (6.3) | 62.9 (9.5) | 60.5 (6.4) | 54.5 (11.2) |
| Education (median [IQR]) | 5 [5, 6] | 5 [5, 6] | 5 [5, 6] | 5 [5, 6] | 5 [4, 6] |
| APOE ɛ4 carriership (%) | 33 (55) | 14 (64) | 11 (55) | 8 (50) | 0/2 (0) |
| Change in diagnosis (%) | 37 (51) | 20 (80)BCD | 11 (48)A | 5 (26)A | 1 (20)A |
| CSF as a reason for amyloid-β PET (%) | 53 (74) | 15 (60) | 18 (78) | 17 (89) | 3 (60) |
| Amyloid-β PET positivity (%) | 34 (47) | 6 (24)BC | 15 (65)A | 11 (58)A | 2 (40) |
| PET according to AUC (%) | 61 (85) | 22 (88) | 19 (83) | 15 (79) | 5 (100) |
| CSF-PET time difference, | |||||
| days (median [IQR]) | 140 [67, 260] | 140 [75, 261] | 162 [78, 304] | 124 [61, 204] | 109 [34, 260] |
| MRI as a reason for amyloid-β PET (%) | 16 (22) | 6 (24) | 3 (13) | 5 (26) | 2 (40) |
| MTA positivity (%) | 32 (46) | 14 (56) | 8 (40) | 8 (42) | 2 (40) |
| PCA positivity (%) | 26 (54) | 10 (59) | 8 (67) | 7 (47) | 1 (25) |
| GCA positivity (%) | 22 (46) | 8 (47) | 5 (42) | 8 (53) | 1 (25) |
| Fazekas positivity (%) | 9 (13) | 3 (12) | 5 (23) | 1 (5) | 0 (0) |
| Lacune positivity (%) | 4 (6) | 1 (4) | 1 (5) | 2 (11) | 0 (0) |
| Microbleed positivity (%) | 5 (8) | 0 (0) | 3 (14) | 2 (11) | 0 (0) |
| MMSE (mean (SD)) | 23.6 (3.8) | 22.8 (3.6) | 23.9 (4.1) | 24.4 (3.9) | 23.7 (2.5) |
| Memory z-score (mean (SD)) | –3.20 (2.87) | –3.41 (2.22) | –3.19 (4.04) | –2.98 (2.30) | –2.74 (1.14) |
| Language z-score (mean (SD)) | –1.13 (1.39) | –1.30 (1.84) | –1.09 (1.27) | –0.87 (0.79) | –1.81 (1.47) |
| Attention z-score (mean (SD)) | –1.10 (1.09) | –1.25 (1.01) | –0.95 (1.11) | –0.92 (1.14) | –2.57 (0.08) |
| Executive z-score (mean (SD)) | –1.56 (1.41) | –1.82 (1.32) | –1.61 (1.69) | –1.05 (1.13) | –2.07 (1.17) |
| Visuospatial z-score (mean (SD)) | –0.90 (1.42) | –1.02 (1.98) | –0.75 (1.00) | –0.77 (0.78) | –2.07 (1.00) |
Education is staged by Verhage classification (1–7) [32]. MRI scans were regarded 1) medial temporal atrophy (MTA)-positive if the left-right averaged MTA ≥1 for a patient under the age of 65, or ≥1.5 for patient age between 65 and 75, 2) posterior cortical atrophy (PCA)-positive if the left-right averaged PCA ≥1 for a patient under the age of 65, and 3) global cortical atrophy (GCA)-positive if the GCA ≥1 for a patient under the age of 65 [23]. Cognitive domain z-scores were derived using the mean and standard deviation values from a group of healthy controls. A, B, C, D indicate difference (p < 0.05) from other groups: A) difference from Aβ–/tau–; B) difference from Aβ–/tau+; C) difference from Aβ+/tau–; D) difference from Aβ+/tau+.
Fig.2
CSF Aβ42, and tau/p-tau values relative to their cut-offs. We present standardized CSF values, created by calculating the percentage of the CSF value relative to its concurrent cut-off. Values of <100% represent pathologically decreased CSF Aβ42 and values of >100% indicate pathologically increased CSF tau (A) and p-tau (B).
Reasons for amyloid-β PET after CSF
To explore the clinical reasoning for amyloid-β PET, patients were divided into two groups (AD, n = 41 and non-AD, n = 31) based on the most likely etiological diagnosis prior to performing a PIB PET scan. More than one reason for amyloid-β PET was reported for n = 33 (46%) cases. Conflicting information from CSF analysis (either not supporting the current diagnosis or with discordant Aβ/tau status) was the most frequent reason for requesting an amyloid-β PET scan (n = 53/72; 74%), being more prevalent in patients with the main suspected etiological diagnosis of AD (n = 36/41, 88%) than in patients with a non-AD suspected etiological diagnosis (n = 17/31, 55%, p = 0.004) (Fig. 3). Other factors contributing to the request of a clinical amyloid-β PET scan after CSF included MRI not supporting the clinical diagnosis (n = 16/72, 22%), unusual clinical presentation (n = 11/72, 15%) and young age (n = 8/72, 11%). In some cases (n = 5/72, 7%, all with Aβ+/tau–), inexperience in interpreting the results of a new CSF Aβ42 assay (Elecsys CSF, Roche Diagnostics GmbH) contributed to diagnostic uncertainty leading to PIB PET scan. A reason for requesting a PET scan was not recorded in the patient chart for n = 2/72 (3%) cases.
Fig.3
Clinical reasons for requesting additional amyloid-β PET after CSF. Patients are grouped based on most likely diagnosis prior to an amyloid-β PET scan. For both diagnosis groups we list characteristics that were recorded as being not compatible with the current main diagnosis, therefore leading to additional amyloid-β PET imaging. Reasons for the amyloid-β PET scan are grouped as biomarkers (red), patient history and presentation (blue), and external (purple). More than one reason is possible per patient. CSF, cerebrospinal fluid; MRI, magnetic resonance imaging; EEG, electroencephalography; PET, positron emission tomography; FDG, [18F]fluorodeoxyglucose; SPECT, single-photon emission computed tomography.
![Clinical reasons for requesting additional amyloid-β PET after CSF. Patients are grouped based on most likely diagnosis prior to an amyloid-β PET scan. For both diagnosis groups we list characteristics that were recorded as being not compatible with the current main diagnosis, therefore leading to additional amyloid-β PET imaging. Reasons for the amyloid-β PET scan are grouped as biomarkers (red), patient history and presentation (blue), and external (purple). More than one reason is possible per patient. CSF, cerebrospinal fluid; MRI, magnetic resonance imaging; EEG, electroencephalography; PET, positron emission tomography; FDG, [18F]fluorodeoxyglucose; SPECT, single-photon emission computed tomography.](https://content.iospress.com:443/media/jad/2020/73-2/jad-73-2-jad190836/jad-73-jad190836-g003.jpg)
Accordance to amyloid-β PET AUC
In most cases (n = 61, 85%), amyloid-β PET scans were performed in compliance with the AUC. Our clinical practice was not covered by the AUC in n = 7 (n = 3 amyloid-negative based on PET) as the clinical findings were suspicious of amnestic AD, but conflicting information from CSF (n = 6) or a previously negative amyloid-β PET combined with normal CSF (n = 1) led to an amyloid-β PET scan. Finally, an amyloid-β PET scan was requested for n = 3 patients without objective cognitive decline, who had decreased Aβ42 in the CSF analysis, lowering diagnostic confidence; and for n = 1 patient with a known PSEN1 mutation to define the stage of pathological disease progression.
Change in diagnosis
Of the n = 42 patients with AD as their initial etiological diagnosis, n = 15 (36%) had a predominant non-amnestic presentation (either language-AD (n = 6), visuospatial (n = 5), behavioral/dysexecutive (n = 3), or corticobasal syndrome (n = 1)) (Fig. 4). Likewise, in patients with a final diagnosis of AD, about half (n = 15/32, 44%) had a non-amnestic presentation. Twenty-two patients (52%) with an initial clinical diagnosis of AD had amyloid-β positivity based on PET and n = 11 (26%) based on CSF Aβ42. Of the patients with AD as the final etiological diagnosis, n = 32 (100%) had amyloid-β pathology based on PET and 13 (41%) based on CSF Aβ42.
Fig.4
Etiological diagnosis in relation to CSF Aβ/tau status and amyloid-β PET. A Sankey diagram showing 1) the distribution of baseline diagnoses to groups based on CSF Aβ/tau status, 2) the percentage of amyloid-β PET positivity by CSF Aβ/tau groups, and 3) the correlation of final diagnosis to amyloid-β PET positivity. DLB, dementia with Lewy bodies; Psych, psychiatric disorder; SCD, subjective cognitive decline; FTD, frontotemporal dementia; PPA, primary progressive aphasia.
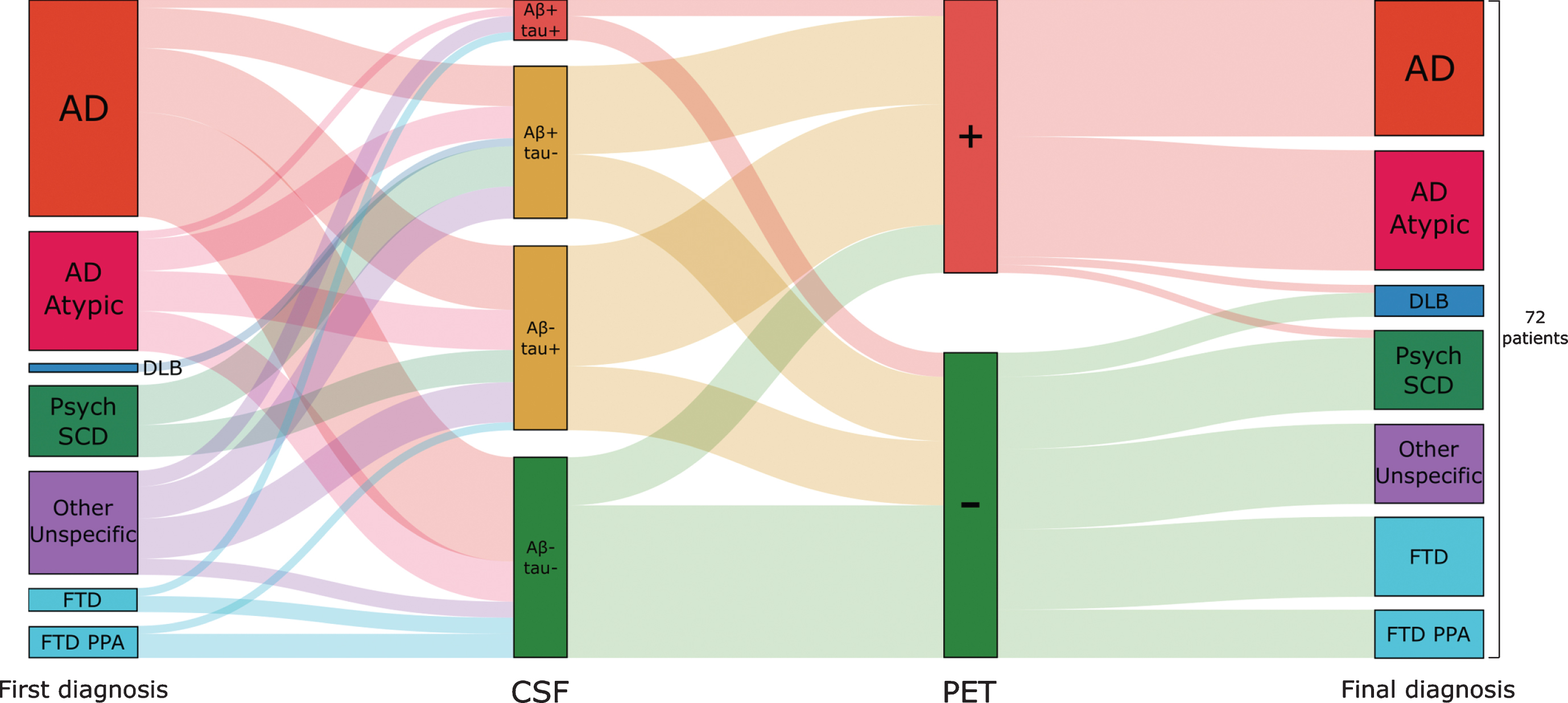
Overall, change in diagnosis occurred in n = 37 (51%) of cases. Diagnosis changed more often in the Aβ–/tau– group (n = 20, 80%) compared to the Aβ–/tau+ (n = 11, 48%; p = 0.04), Aβ+/tau– (n = 5, 26%; p < 0.01), and Aβ+/tau+ (n = 1, 20%; p = 0.03) groups.
Final diagnoses of the five cases in the CSF Aβ+/tau+ group were 1) early-onset AD (age: 48 years) with a negative family history, 2) autoimmune encephalitis, 3) corticobasal syndrome due to AD, 4) logopenic variant primary progressive aphasia with a previously negative amyloid-β PET scan, and 5) a suspected genetic variant of frontotemporal dementia. To further illustrate the diagnostic process, we present a small case series including one patient from each of the four CSF Aβ/tau groups (Fig. 5).
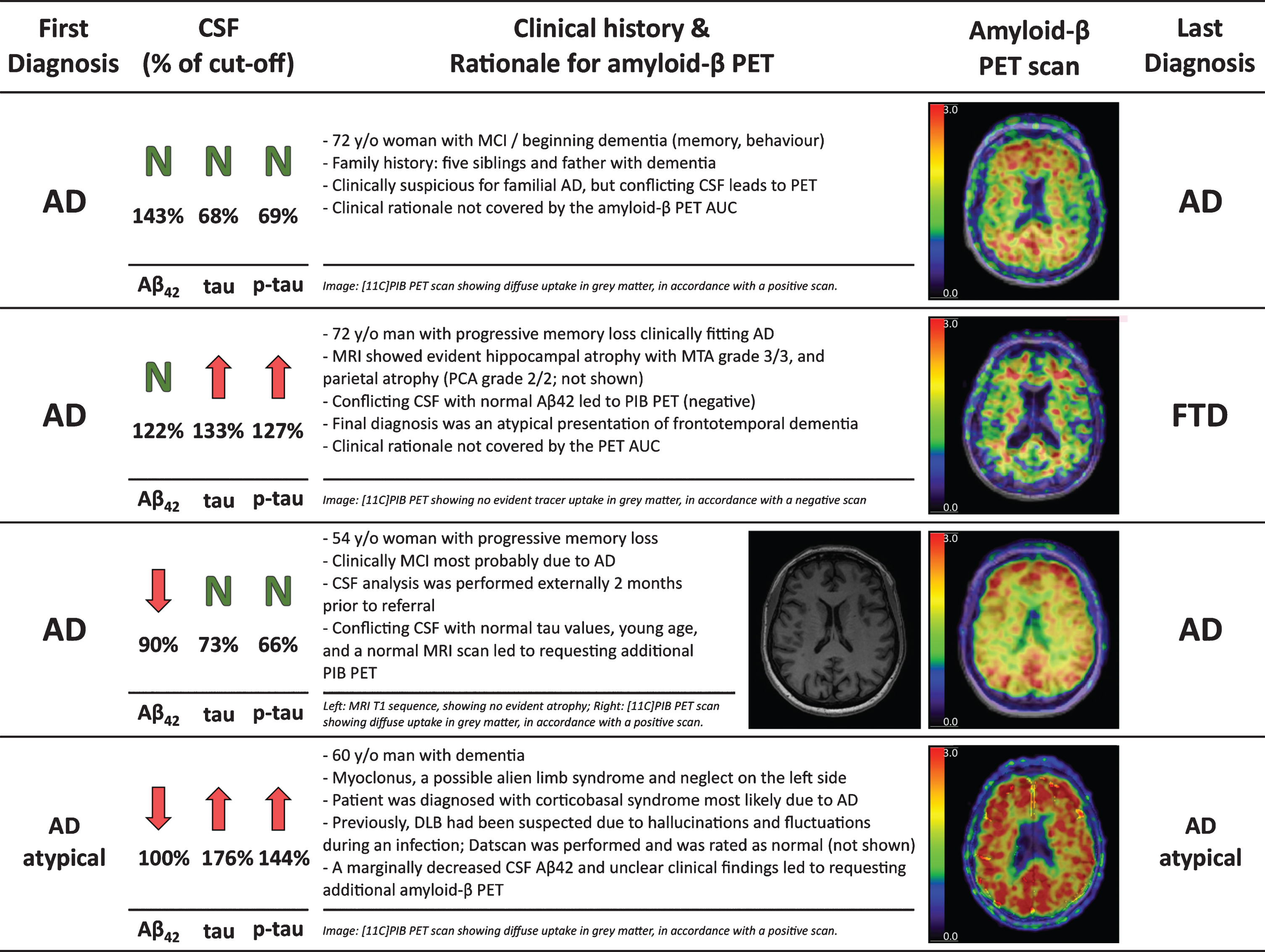
Fig.5
Four case reports illustrating the clinical reasoning for requesting an additional amyloid-β PET.
DISCUSSION
We investigated the clinical reasoning behind requesting an amyloid-β PET scan after disclosure of CSF biomarkers in a clinical cohort. Our main finding was that in most cases CSF biomarkers conflicting with the clinical diagnosis contributed to diagnostic uncertainty, which led to the request of an amyloid-β PET scan to support the clinical diagnostic process. This was reinforced by the observation that an additional amyloid-β PET scan was rarely requested in patients with a CSF Aβ+/tau+ status. Second, we found that an amyloid-β PET was requested in patients, that were relatively young, often had an atypical presentation of AD and often showed a change in diagnosis. Third, we observed that although amyloid-β PET scans were usually requested according to the AUC, our clinical practice was not wholly covered by these criteria, as it was often driven by inconclusive CSF biomarker results. Our results support previous work that CSF biomarkers that conflict with the clinical diagnosis often lead to additional amyloid-β PET [12, 13].
In our cohort, an amyloid-β PET was most often requested due to inconclusive results from the CSF biomarkers. This occurred in cases when diagnostic confidence was low due to inconclusive CSF Aβ/tau status, or when a clinical diagnosis of AD was contradicted by a non-pathologic CSF analysis, which is largely in agreement with previous findings [13]. In fact, there were few patients with CSF Aβ+/tau+ status in our cohort, indicating that patients with a clinical suspicion of AD supported by low Aβ42 and high tau in the CSF analysis usually do not need further confirmation with amyloid-β PET. This is also evident in the current guidelines for both clinical practice [8] and research [1], which advocate CSF and PET as parallel options to support the diagnosis of AD. However, our results show that in complicated cases clinicians valued the information from an additional amyloid-β PET.
Although clinical diagnosis conflicting with CSF biomarkers contributed to requesting amyloid-β PET scans in most patients, overall clinical rationale was more complex. This was illustrated by a variety of other factors, often in combination with each other, that decreased clinical diagnostic confidence and led to additional amyloid-β PET imaging. Most prominently, incongruent imaging (MRI, FDG PET, DaTscan SPECT) findings or an unusual clinical presentation contributed to decreased confidence in the clinical diagnosis despite CSF findings. The added value of amyloid-β PET imaging, in particular in atypical clinical presentations of AD, has also been shown previously [12, 30]. Our results also support younger patient age being a factor for requesting an amyloid-β PET scan, as indicated in the PET AUC [14]. This is related to younger patients more often having a non-amnestic clinical presentation, in addition to the diagnosis of AD being rare and potentially having a higher impact at a younger age. Finally, we observed that a clinician’s decision can also be influenced by external reasons, such as patient wish or decreased confidence in CSF results due to the initiation of a new CSF assay. It is also possible that clinicians as well as patients might also be inclined to have more confidence in PET imaging due to the visual aspects of a PET scan, and that clinicians’ biases and prior experiences might play a role when deciding whether to use additional amyloid-β PET diagnostics.
Amyloid-β PET was usually, but not always, requested in accordance with the PET appropriate use criteria [14]. Some differences between clinical practice and the AUC were not unexpected as the AUC were designed to build an initial framework for clinical amyloid-β PET, and were also published during the time course of our study. When clinical practice was not covered by the AUC, a PIB scan was requested in patients with no objective cognitive decline due to decreased Aβ42 values in the CSF, or due to decreased diagnostic confidence arising from inconclusive or normal CSF biomarker (or prior amyloid-β PET) results in patients with a clinical syndrome suggestive of AD. Although the recently published AUC for CSF also include performing CSF analysis in SCD [31], neither the AUC for CSF nor the AUC for PET describe the diagnostic setting, where information about amyloid-β status is already available. As amyloid-β biomarkers are increasingly integrated into clinical practice, the number of such cases is likely to increase over time. The value of an additional amyloid-β PET is likely highest in patients with a CSF analysis conflicting with the clinical diagnosis of AD, as a negative amyloid-β PET scan can refute the diagnosis [11]. In cases with a non-AD diagnosis combined with a decreased Aβ42 and/or an increased tau in the CSF, the added value of an amyloid-β PET scan is hindered by the possibility of amyloid-β as a secondary pathology, especially in older populations. Therefore, our results combined with previous work from other centers [12, 13] suggest a group of patients (i.e., clinically diagnosed with AD without an AD-like CSF biomarker signature) might benefit from being included in updated amyloid PET AUC. However, these findings must be confirmed by larger prospective multi-center studies.
The main strength of the present study is the description of the clinical practice in a tertiary memory clinic, where both CSF biomarkers are regularly used for clinical practice and there is good access to amyloid-β PET if needed. By excluding cases where an amyloid-β PET scan was performed due to involvement in research, we were able to minimize the bias caused by research and to concentrate on the clinical decision-making process. In addition, our study has some limitations. Composition of our sample resulted in some inherit biases, caused by the infrastructure of our memory clinic. For example, all patients in our center are offered CSF biomarkers analysis, many patients (often with prior CSF analysis available) are referred to us due to a diagnostic dilemma, and referred patients are generally relatively young. Additionally, all patients are assessed by five neurologists in our center, who might share similar views on the application of biomarkers. While these sample characteristics might reduce overall generalizability, we believe our findings are likely to be generalizable to other memory clinic settings where CSF analysis is commonly used and represent a relevant clinical question. Additionally, due to the retrospective nature of the study, some of the data were retrieved from patient charts. In cases of incomplete or ambiguous descriptions, some degree of subjective judgement on the part of the investigators was unavoidable. Our center has also been involved in several amyloid-β PET studies [9–11], which recruited patients from clinical practice. Therefore, our cohort may have missed cases where amyloid-β PET was deemed clinically useful from that period of time. Finally, due to the longitudinal upward drift of the median CSF Aβ42 values in our centers, the cut-off values for CSF Aβ42 in our center have been changing over time [17]. Therefore, it is feasible that the CSF Aβ42 cut-offs did not always best represent the underlying amyloid-β status.
To conclude, we presented data from a single memory clinic where CSF biomarkers are commonly used. During the period of our study, the main reason for requesting an amyloid-β PET scan was the occurrence of a mismatch between the primary clinical diagnosis and CSF Aβ/tau results. Future work is necessary to confirm similar clinical reasoning in other cohorts and to consider whether such practice should be represented in guidelines.
ACKNOWLEDGMENTS
The institutional review board of the VU University Medical Center approved all studies from which the current data was gathered and retrospectively analyzed. All patients provided written informed consent for their data to be used for research purposes.
The Alzheimer Center Amsterdam is supported by Alzheimer Nederland and Stichting VUmc fonds. Research performed at the Alzheimer Center Amsterdam is part of the neurodegeneration research program of Amsterdam Neuroscience. The clinical database structure was developed with funding from Stichting Dioraphte.
J.R. would like to thank the International Atomic Energy Agency (IAEA) and the North Estonia Medical Centre for their contribution to his professional development.
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/19-0836r2).
REFERENCES
[1] | Jack CR , Bennett DA , Blennow K , Carrillo MC , Dunn B , Haeberlein SB , Holtzman DM , Jagust W , Jessen F , Karlawish J , Liu E , Molinuevo JL , Montine T , Phelps C , Rankin KP , Rowe CC , Scheltens P , Siemers E , Snyder HM , Sperling R , Elliott C , Masliah E , Ryan L , Silverberg N ((2018) ) NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimers Dement 14: , 535–562. |
[2] | Ossenkoppele R , Jansen WJ , Rabinovici GD , Knol DL , van der Flier WM , van Berckel BNM , Scheltens P , Visser PJ , Verfaillie SCJ , Zwan MD , Adriaanse SM , Lammertsma AA , Barkhof F , Jagust WJ , Miller BL , Rosen HJ , Landau SM , Villemagne VL , Rowe CC , Lee DY , Na DL , Seo SW , Sarazin M , Roe CM , Sabri O , Barthel H , Koglin N , Hodges J , Leyton CE , Vandenberghe R , van Laere K , Drzezga A , Forster S , Grimmer T , Sánchez-Juan P , Carril JM , Mok V , Camus V , Klunk WE , Cohen AD , Meyer PT , Hellwig S , Newberg A , Frederiksen KS , Fleisher AS , Mintun MA , Wolk DA , Nordberg A , Rinne JO , Chételat G , Lleo A , Blesa R , Fortea J , Madsen K , Rodrigue KM , Brooks DJ ((2015) ) Prevalence of amyloid PET positivity in dementia syndromes: A meta-analysis. JAMA 313: , 1939–1949. |
[3] | Strozyk D , Blennow K , White LR , Launer LJ ((2003) ) CSF Abeta 42 levels correlate with amyloid-neuropathology in a population-based autopsy study. Neurology 60: , 652–656. |
[4] | Klunk WE , Engler H , Nordberg A , Wang Y , Blomqvist G , Holt DP , Bergström M , Savitcheva I , Huang GF , Estrada S , Ausén B , Debnath ML , Barletta J , Price JC , Sandell J , Lopresti BJ , Wall A , Koivisto P , Antoni G , Mathis CA , Långström B ((2004) ) Imaging brain amyloid in Alzheimer’s disease with Pittsburgh Compound-B. Ann Neurol 55: , 306–319. |
[5] | Blennow K , Mattsson N , Schöll M , Hansson O , Zetterberg H ((2015) ) Amyloid biomarkers in Alzheimer’s disease. Trends Pharmacol Sci 36: , 297–309. |
[6] | Zwan MD , Rinne JO , Hasselbalch SG , Nordberg A , Lleo A , Herukka SK , Soininen H , Law I , Bahl JMC , Carter SF , Fortea J , Blesa R , Teunissen CE , Bouwman FH , Van Berckel BNM , Visser PJ ((2016) ) Use of amyloid-PET to determine cutpoints for CSF markers: A multicenter study. Neurology 86: , 50–58. |
[7] | de Wilde A , Reimand J , Teunissen CE , Zwan M , Windhorst AD , Boellaard R , van der Flier WM , Scheltens P , van Berckel BNM , Bouwman F , Ossenkoppele R ((2019) ) Discordant amyloid-β PET and CSF biomarkers and its clinical consequences. Alzheimers Res Ther 11: , 78. |
[8] | McKhann GM , Knopman DS , Chertkow H , Hyman BT , Jack CR , Kawas CH , Klunk WE , Koroshetz WJ , Manly JJ , Mayeux R , Mohs RC , Morris JC , Rossor MN , Scheltens P , Carrillo MC , Thies B , Weintraub S , Phelps CH ((2011) ) The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 7: , 263–269. |
[9] | de Wilde A , van der Flier WM , Pelkmans W , Bouwman F , Verwer J , Groot C , van Buchem MM , Zwan M , Ossenkoppele R , Yaqub M , Kunneman M , Smets EMA , Barkhof F , Lammertsma AA , Stephens A , van Lier E , Biessels GJ , van Berckel BN , Scheltens P ((2018) ) Association of amyloid positron emission tomography with changes in diagnosis and patient treatment in an unselected memory clinic cohort. JAMA Neurol 75: , 1062–1070. |
[10] | Zwan MD , Bouwman FH , Konijnenberg E , van der Flier WM , Lammertsma AA , Verhey FRJ , Aalten P , van Berckel BNM , Scheltens P ((2017) ) Diagnostic impact of [18F]flutemetamol PET in early-onset dementia. Alzheimers Res Ther 9: , 2. |
[11] | Ossenkoppele R , Prins ND , Pijnenburg YAL , Lemstra AW , Van Der Flier WM , Adriaanse SF , Windhorst AD , Handels RLH , Wolfs CAG , Aalten P , Verhey FRJ , Verbeek MM , Van Buchem MA , Hoekstra OS , Lammertsma AA , Scheltens P , Van Berckel BNM ((2013) ) Impact of molecular imaging on the diagnostic process in a memory clinic. Alzheimers Dement 9: , 414–421. |
[12] | Ceccaldi M , Jonveaux T , Verger A , Krolak-Salmon P , Houzard C , Godefroy O , Shields T , Perrotin A , Gismondi R , Bullich S , Jovalekic A , Raffa N , Pasquier F , Semah F , Dubois B , Habert MO , Wallon D , Chastan M , Payoux P , Ceccaldi M , Guedj E , Felician O , Didic M , Gueriot C , Koric L , Kletchkova-Gantchev R , Godefroy O , Andriuta D , Devendeville A , Dupuis D , Binot I , Barbay M , Meyer ME , Moullard V , Magnin E , Chamard L , Haffen S , Morel O , Drouet C , Boulahdour H , Goas P , Querellou-Lefranc S , de la Sayette V , Cogez J , Branger P , Agostini D , Manrique A , Rouaud O , Bejot Y , Jacquin-Piques A , Dygai-Cochet I , Berriolo-Riedinger A , Moreaud O , Sauvee M , Crépin CG , Pasquier F , Bombois S , Lebouvier T , Mackowiak-Cordoliani MA , Deramecourt V , Rollin-Sillaire A , Cassagnaud-Thuillet P , Chen Y , Semah F , Petyt G , Krolak-Salmon P , Federico D , Danaila KL , Guilhermet Y , Magnier C , Makaroff Z , Rouch I , Xie J , Roubaud C , Coste MH , David K , Sarciron A , Waissi AS , Scheiber C , Houzard C , Gabelle-Deloustal A , Bennys K , Marelli C , Touati L , Mariano-Goulart D , de Verbizier-Lonjon D , Jonveaux T , Benetos A , Kearney-Schwartz A , Perret-Guillaume C , Verger A , Vercelletto M , Boutoleau-Bretonniere C , Pouclet-Courtemanche H , Wagemann N , Pallardy A , Hugon J , Paquet C , Dumurgier J , Millet P , Queneau M , Dubois B , Epelbaum S , Levy M , Habert MO , Novella JL , Jaidi Y , Papathanassiou D , Morland D , Belliard S , Salmon A , Lejeune F , Hannequin D , Wallon D , Martinaud O , Zarea A , Chastan M , Pariente J , Thalamas C , Galitzky-Gerber M , Tricoire Ricard AM , Calvas F , Rigal E , Payoux P , Hitzel A , Delrieu J , Ousset PJ , Lala F , Sastre-Hengan N , Stephens A , Guedj E ((2018) ) Added value of 18F-florbetaben amyloid PET in the diagnostic workup of most complex patients with dementia in France: A naturalistic study. Alzheimers Dement 14: , 293–305. |
[13] | Leuzy A , Savitcheva I , Chiotis K , Lilja J , Andersen P , Bogdanovic N , Jelic V , Nordberg A ((2019) ) Clinical impact of [18F]flutemetamol PET among memory clinic patients with an unclear diagnosis. Eur J Nucl Med Mol Imaging 46: , 1276–1286. |
[14] | Johnson KA , Minoshima S , Bohnen NI , Donohoe KJ , Foster NL , Herscovitch P , Karlawish JH , Rowe CC , Carrillo MC , Hartley DM , Hedrick S , Pappas V , Thies WH ((2013) ) Appropriate use criteria for amyloid PET: A report of the Amyloid Imaging Task Force, the Society of Nuclear Medicine and Molecular Imaging, and the Alzheimer’s Association. J Nucl Med 54: , 476–490. |
[15] | Engelborghs S , Niemantsverdriet E , Struyfs H , Blennow K , Brouns R , Comabella M , Dujmovic I , van der Flier W , Frölich L , Galimberti D , Gnanapavan S , Hemmer B , Hoff E , Hort J , Iacobaeus E , Ingelsson M , Jan de Jong F , Jonsson M , Khalil M , Kuhle J , Lleó A , de Mendonça A , Molinuevo JL , Nagels G , Paquet C , Parnetti L , Roks G , Rosa-Neto P , Scheltens P , Skårsgard C , Stomrud E , Tumani H , Visser PJ , Wallin A , Winblad B , Zetterberg H , Duits F , Teunissen CE ((2017) ) Consensus guidelines for lumbar puncture in patients with neurological diseases. Alzheimers Dement (Amst) 8: , 111–126. |
[16] | Willemse EAJ , van Maurik IS , Tijms BM , Bouwman FH , Franke A , Hubeek I , Boelaarts L , Claus JJ , Korf ESC , van Marum RJ , Roks G , Schoonenboom N , Verwey N , Zwan MD , Wahl S , van der Flier WM , Teunissen CE ((2018) ) Diagnostic performance of Elecsys immunoassays for cerebrospinal fluid Alzheimer’s disease biomarkers in a nonacademic multicenter memory clinic cohort: The ABIDE project. Alzheimers Dement (Amst) 10: , 563–572. |
[17] | Schindler SE , Sutphen CL , Teunissen C , McCue LM , Morris JC , Holtzman DM , Mulder SD , Scheltens P , Xiong C , Fagan AM ((2018) ) Upward drift in cerebrospinal fluid amyloid-β 42 assay values for more than 10 years. Alzheimers Dement 14: , 62–70. |
[18] | Zwan MD , Ossenkoppele R , Tolboom N , Beunders AJM , Kloet RW , Adriaanse SM , Boellaard R , Windhorst AD , Raijmakers P , Adams H , Lammertsma AA , Scheltens P , van der Flier WM , van Berckel BNM ((2014) ) Comparison of simplified parametric methods for visual interpretation of 11C-Pittsburgh compound-B PET images. J Nucl Med 55: , 1305–1307. |
[19] | Van Der Flier WM , Pijnenburg YAL , Prins N , Lemstra AW , Bouwman FH , Teunissen CE , Van Berckel BNM , Stam CJ , Barkhof F , Visser PJ , Van Egmond E , Scheltens P ((2014) ) Optimizing patient care and research: The Amsterdamdementia cohort. J Alzheimers Dis 41: , 313–327. |
[20] | Koedam ELGE , Lehmann M , Van Der Flier WM , Scheltens P , Pijnenburg YAL , Fox N , Barkhof F , Wattjes MP ((2011) ) Visual assessment of posterior atrophy development of a MRI rating scale. Eur Radiol 21: , 2618–2625. |
[21] | Scheltens P , Leys D , Barkhof F , Huglo D , Weinstein HC , Vermersch P , Kuiper M , Steinling M , Wolters EC , Valk J ((1992) ) Atrophy of medial temporal lobes on MRI in “probable” Alzheimer’s disease and normal ageing: Diagnostic value and neuropsychological correlates. J Neurol Neurosurgery Psychiatry 55: , 967–972. |
[22] | Pasquier F , Leys D , Weerts JGE , Mounier-Vehier F , Barkhof F , Scheltens P ((1996) ) Inter-and intraobserver reproducibility of cerebral atrophy assessment on MRI scans with hemispheric infarcts. Eur Neurol 36: , 268–272. |
[23] | Rhodius-Meester HFM , Benedictus MR , Wattjes MP , Barkhof F , Scheltens P , Muller M , Van Der Flier WM ((2017) ) MRI visual ratings of brain atrophy and white matter hyperintensities across the spectrum of cognitive decline are differently affected by age and diagnosis. Front Aging Neurosci 9: , 1–12. |
[24] | Fazekas F , Chawluk JB , Alavi A , Hurtig HI ZR ((1987) ) MR signal abnormalities at 1.5 T in Alzheimer’s dementia and normal aging deficiency. AJR Am J Roentgenol 149: , 351–356. |
[25] | Groot C , Van Loenhoud AC , Barkhof F , Van Berckel BNM , Koene T , Teunissen CC , Scheltens P , Van Der Flier WM , Ossenkoppele R ((2018) ) Differential effects of cognitive reserve and brain reserve on cognition in Alzheimer disease. Neurology 90: , e149–e156. |
[26] | R Core Team ((2018) ) R: A language and environment for statistical computing. https://www.R-project.org/. |
[27] | Wickham H ((2017) ) tidyverse: Easily Install and Load the “Tidyverse.”. |
[28] | Yoshida K , Bohn J ((2018) ) tableone: Create “Table 1” to Describe Baseline Characteristics. |
[29] | Gandrud C , Allaire JJ , Russell K , Yetman CJ ((2017) ) networkD3: D3 JavaScript Network Graphs from R. |
[30] | Weston PSJ , Paterson RW , Dickson J , Barnes A , Bomanji JB , Kayani I , Lunn MP , Mummery CJ , Warren JD , Rossor MN , Fox NC , Zetterberg H , Schott JM ((2016) ) Diagnosing dementia in the clinical setting: Can amyloid PET provide additional value over cerebrospinal fluid? J Alzheimers Dis 54: , 1297–1302. |
[31] | Shaw LM , Arias J , Blennow K , Galasko D , Molinuevo JL , Salloway S , Schindler S , Carrillo MC , Hendrix JA , Ross A , Illes J , Ramus C , Fifer S ((2018) ) Appropriate use criteria for lumbar puncture and cerebrospinal fluid testing in the diagnosis of Alzheimer’s disease. Alzheimers Dement 14: , 1505–1521. |
[32] | Verhage F ((1964) ) Intelligence and Age: Study with Dutch people aged 12 to 77 (Thesis). Koninklijke Van Gorcum, Groningen. |




