Vibrating Tail, Digging, Body/Face Interaction, and Lack of Barbering: Sex-Dependent Behavioral Signatures of Social Dysfunction in 3xTg-AD Mice as Compared to Mice with Normal Aging
Abstract
Modeling of Alzheimer’s disease (AD), classically focused on the subject-environment interaction, foresees current social neuroscience efforts as improving the predictive validity of new strategies. Here we studied social functioning among congeners in 13–14-month-old mice with normal aging in naturalistic and experimental conditions and depicted behavioral signatures of dysfunction in age-matched 3xTg-AD mice. The most sensitive variables were vibrating tail, digging, body/face and self-grooming, that can be easily used in housing routines and the assessment of strategies. Sex-specific signatures (vibrating tail, digging, and grooming) defined female 3xTg-AD mice ethogram. All animals sleep huddled while barbering was only found in females with normal aging.
INTRODUCTION
While personalized medicine urges the depiction of the nuances of individual variability that underlies epidemiology, basic research classically focused on the individual specimen as the subject of study [1], now opens to the higher level of complexity of social behavior and communication.
Social behavior is a fundamental characteristic modulated by endogenous, genetic, and environmental factors. Deficits in social interaction are defined by endophenotypes and are present in various psychiatric disorders such as autism, schizophrenia, depression, and Alzheimer’s disease (AD) with a strong impact on daily life of patients and caregivers [2–5]. Conversely, social factors are considered not only modulators of mental and physical health of people but also risk factors for symptoms of dementia [6]. Although social interaction is considered a phenotype or endophenotype of AD, the underlying mechanisms and causes remain complex [7].
At the translational level, modeling behavioral and psychological symptoms of dementia (BPSD) in murine models of AD has been addressed using methodological approaches based on the interaction of the subject with the environment, but few involve the relation with other individuals [8]. Furthermore, social neuroscience efforts to fill the gap are also foreseen as important to improve the predictive validity of new preventive and/or therapeutic strategies for AD [9].
Therefore, the present work is aimed to explore social functioning (from affiliative behaviors and dominance to aggressive behaviors) among congeners in 13-14-month-old male and female mice with normal aging in naturalistic and experimental conditions, as well as to depict behavioral signatures of social dysfunction as a consequence of genetically-induced amyloid-β and tau pathologies in age-matched 3xTg-AD mice. At these ages, the 3xTg-AD model has extensively been described mimicking advanced stages of disease with hallmark amyloid and tau pathologies [10]. For that purpose, we evaluated behavioral interactions with congeners (group-sleeping, body/face, ano-genital, aggression), with the context (horizontal and vertical exploratory activities, digging) and self-actions (self-grooming and vibrating tail) that can be easily used in housing routines and the assessment of strategies.
MATERIALS AND METHODS
Animals
A total of forty-six 13–14-month-old homozygous male and female 3xTg-AD and their non-transgenic (NTg) mice, 7th breeding generations derived from the same genetic background (C57BL/6×129 sv), engineered at the University of California Irvine were used [10]. Subjects (group, n, mean age in months±S.D.: male NTg, n = 10, 13±0; male 3xTg-AD, n = 8, 13±0; female NTg, n = 14, 13.57±0.94; female 3xTg-AD, n = 14, 13.71±0.94) were housed three to four per cage and maintained in Macrolon cages (35 x 35 x 25 cm) under standard laboratory conditions of food (A03 diet for breeding; A04 diet for maintenance, Panlab, SL, Barcelona) and water ad libitum, 22±2 °C, a 12 h light:dark cycle and relative humidity 50–60%. Behavioral assessments were performed blind to the experiment, in a counterbalanced manner. All procedures were in accordance with Spanish legislation on ‘Protection of Animals Used for Experimental and Other Scientific Purposes’ and the EU Directive (2010/63/UE) on this subject. The study complies with the ARRIVE guidelines developed by the NC3Rs and the aim to reduce the number of animals used.
Home cage sleeping behavior
Home cage group sleeping behavior was registered during 3 consecutive days and reported as the incidence of animals sleeping together, huddled (entire bodies being pressed up against each other) and/or dog-piled (similarly to dog-piling), in the same quadrant of their home cage [11].
Social interaction test
A dyad of two unfamiliar mice of the same genotype and sex were introduced in a standard home cage and video recorded for 5 min (modification of [12]). Behaviors were classified into social (social investigation, aggression, vibrant tail) and non-social (exploring, digging, self-grooming) interactions. We also scored the total number of episodes and their total duration. In order to depict the sequence of behavioral events, here we propose to plot the latencies for each genotype and sex as an ethogram tablature. There, self-grooming and vibrating tail are plotted as self-actions.
Barbering or Dalila effect
Barbering or Dalila effect, alopecia due to social interaction between animals, was scored according to 5-scores Kalueff scale [13]. The incidence per cage and the affected body area was also recorded in cm2 [14, 15].
Tube-dominance test
In order to know if the Dalila effect was related to dominance, the social range of dyads was assessed in the Tube-dominance test [11]. Animals were placed at the opposite ends of a translucent acrylic tube (diameter: 4 cm, length: 25.5 cm), allowed to advance through it for a maximum of 120 s or until one of them (“dominant”) forced the other (“subordinate”) to get out of the tube.
Statistical analysis
Results are expressed as mean±S.E.M. or incidence. Statistics using SPSS 15.0 as detailed in the legends, with significance considered at p < 0.05.
RESULTS
Home cage sleeping behaviors
All mice (100%) slept huddled, curled together, close to each other, in a quadrant of the cage, independently of the genotype or sex.
Social interaction test
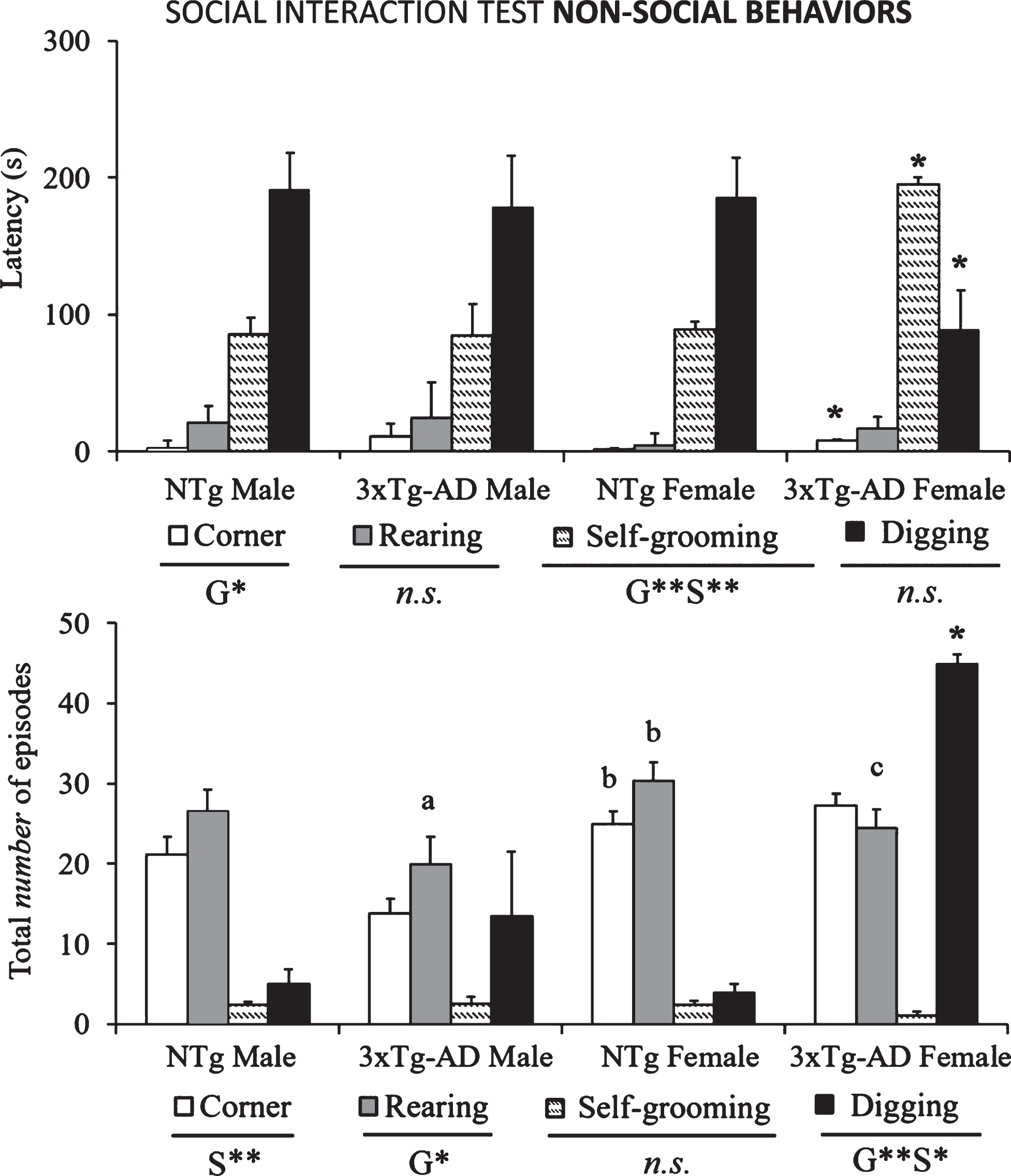
The ethogram tablature (Fig. 1) showed a consistent sequence of actions in the NTg mice, with the only distinction of vibrating tail that was anticipated in the females. The ethogram started with exploration (horizontal and vertical), followed by first body/face social interaction done before the end of the first minute. In the second minute, ano-genital interaction and self-grooming were observed. Digging appeared around the third minute of the test, followed in males or preceded in females by vibrating tail. In the 3xTg-AD mice, the tablature showed sex and genotype differences. As compared to their NTg counterparts, males were delayed in the elicitation of social interactions, while vibrating tail was advanced similarly to NTg females. In contrast, female 3xTg-AD showed a very early elicitation of vibrating tail and an anticipated digging that was performed before the elicitation of social behaviors. Body/face and ano-genital interactions were consecutively performed, and grooming was delayed to the third minute of the test, as compared to all the other groups. These differences were found reflected in the respective variables of total duration and number of episodes of social (Fig. 2) and non-social (Fig. 3) behaviors elicited among congeners in the social interaction test (SIT). Incidence of aggression was 1/8 in male 3xTg-AD mice with a latency of 213 s, and absent in the other experimental groups. Overall, the most sensitive variables were vibrating tail (duration, episodes, and latency), digging (latency and episodes), body/face (episodes, duration, and latency), and self-grooming (latency) (all F > 3.289, p < 0.001).
Fig.1
Social interaction test ethogram tablature of 14-month-old male and female 3xTg-AD mice at advanced-stages of the disease and age- and sex-matched NTg mice with normal aging. Results are mean±SEM of latency of events shown by NTg (male, n = 10; female, n = 14) and 3xTg-AD mice (male, n = 8; female, n = 14) in the social interaction test. Multivariate General Linear Modelling: G, genotype effect and S, sex effect *p<0.05 and **p<0.01, followed by Duncan’s post-hoc test comparisons ap < 0.05 different all group and bp < 0.05 different to genotype. Student’s t-test ***p < 0.001, **p < 0.01, and *p < 0.05 versus NTg mice. Student’s t-test &p < 0.05 versus male NTg mice and, #p < 0.05 versus male 3xTg-AD mice.
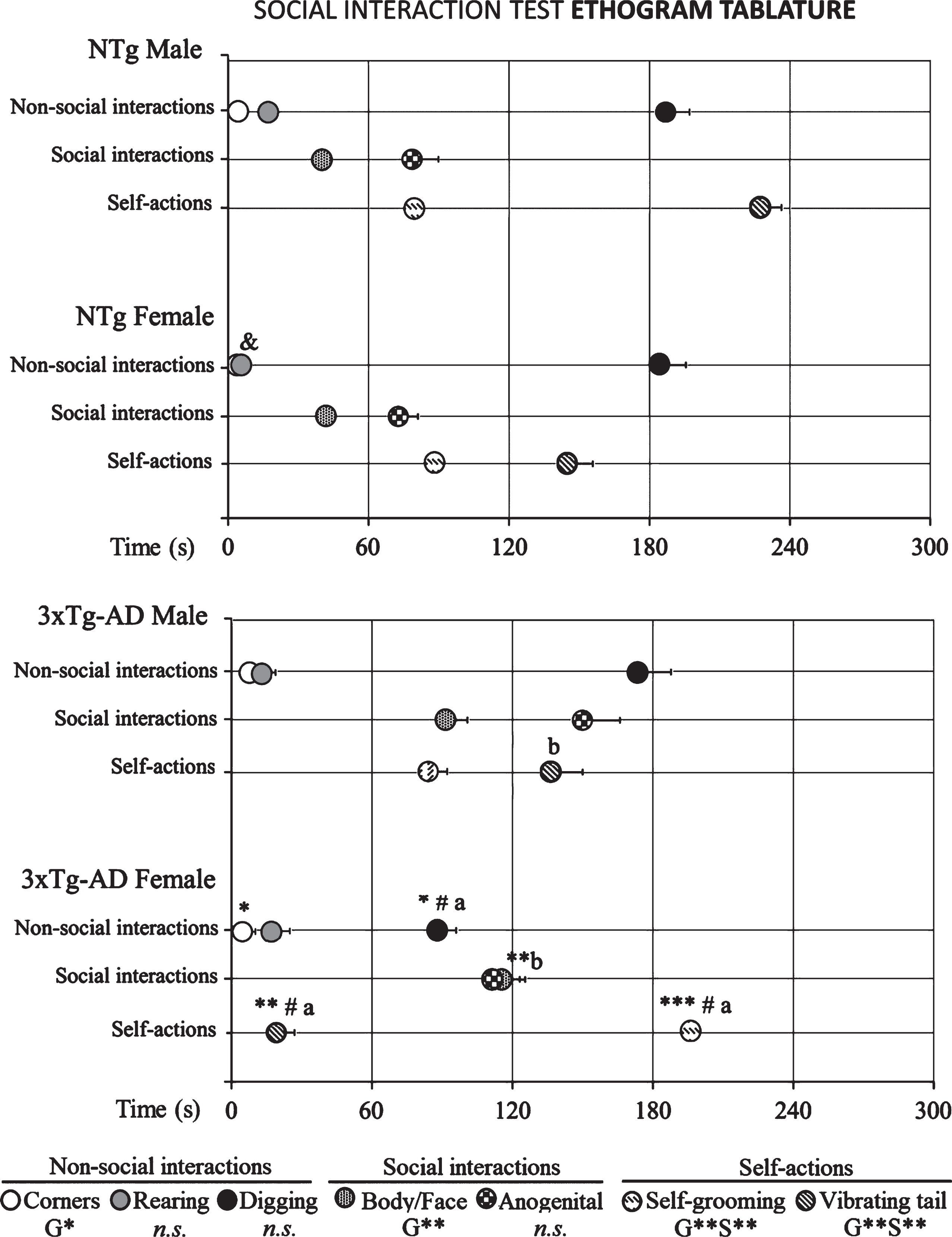
Fig.2
Social interactions (body/face and ano-genital contacts) and vibrating tail shown in the SIT by 14-month-old male and female 3xTg-AD mice at advanced-stages of the disease and age- and sex-matched NTg mice with normal aging. Results are mean±SEM of latency of events, total duration and total number of episodes. NTg (male, n = 10; female, n = 14) and 3xTg-AD mice (male, n = 8; female, n = 14) in the social interaction test. Multivariate General Linear Modelling: G, genotype effect and S, sex effect *p < 0.05 and **p < 0.01 and followed by post-hoc Duncan’s test comparisons *p < 0.05 versus all the other groups, ap < 0.05 versus the NTg mice, same sex, bp < 0.05 versus male counterpart, cp < 0.05 versus male NTg mice.
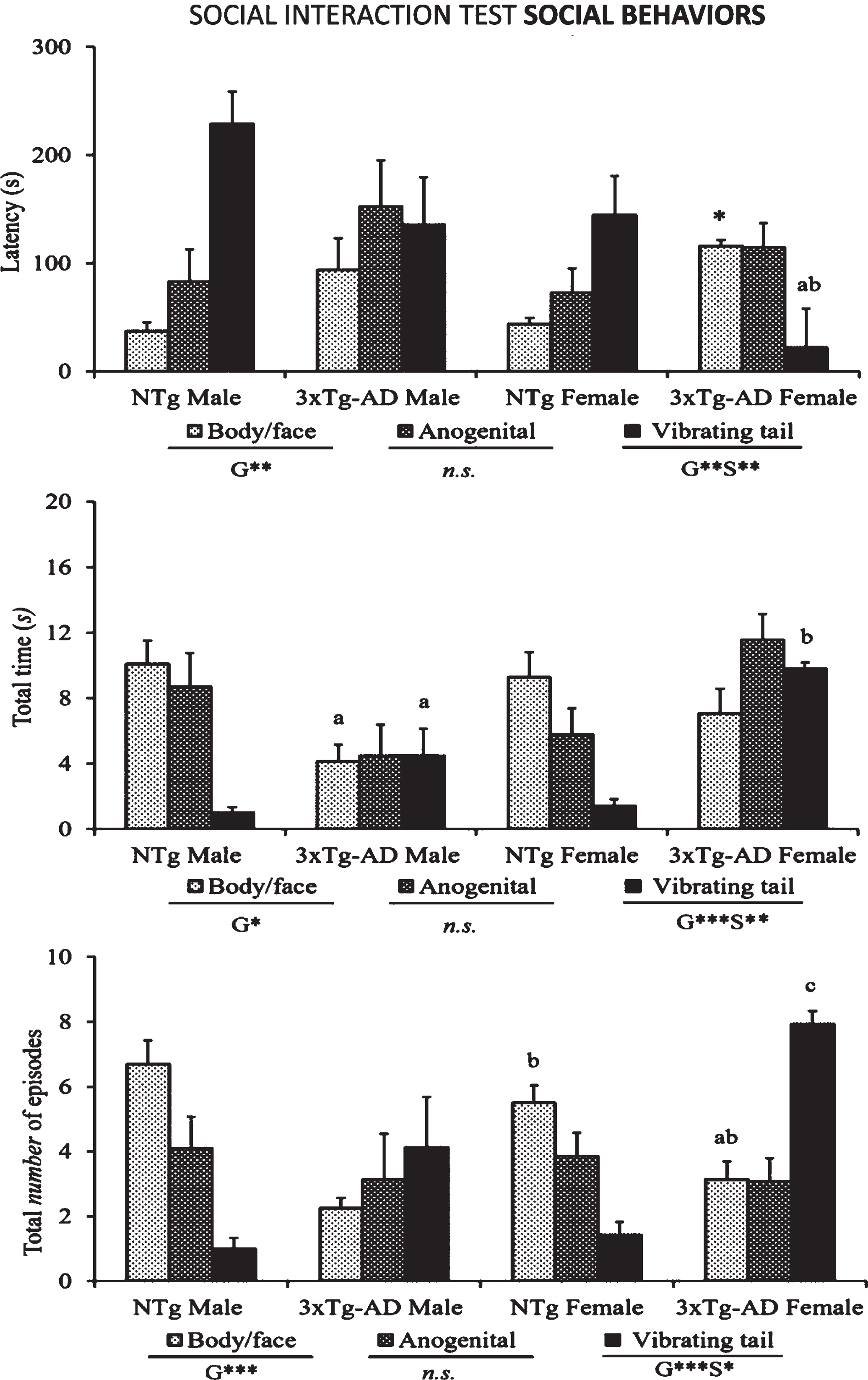
Fig.3
Non-social interactions (rearings, corners, self-grooming, and digging) shown in the SIT by 14-month-old male and female 3xTg-AD mice at advanced-stages of the disease and age- and sex-matched NTg mice with normal aging. Results are mean±SEM of latency of events and total number of episodes. NTg (male, n = 10; female, n = 14) and 3xTg-AD mice (male, n = 8; female, n = 14) in the social interaction test. Multivariate General Linear Modelling: G, genotype effect and S, sex effect *p < 0.05 and **p < 0.01 and followed by post-hoc Duncan’s test comparisons *p < 0.05 versus all the other groups, ap < 0.05 versus the NTg mice, same sex, bp < 0.05 versus male counterpart, cp < 0.05 versus male NTg mice.
Barbering or Dalila effect
Dalila effect was present only in the female NTg group, with an incidence of 0.40±0.16, a severity score of 0.80±0.35 and 0.25±0.12 cm2 of body affected area. The three variables showed significant differences as compared to null presence in 3xTg-AD (all F < 2.170, p > 0.05).
Tube-dominance test
First contact was faster in 3xTg-AD mice (1.21±1.21 s versus. NTg: 2.00±0.00 s, p < 0.05) but no differences were shown in the latency of withdrawn mice (1.42±0.13 s versus NTg: 3.42±2.33 s) or total duration of the test (16.00±1.19 s versus NTg: 11.42±2.06 s).
Discussion
The present work identifies, for the first time, vibrating tail, digging, body/face interaction, and lack of barbering as variables that can be used as behavioral signatures of AD-social phenotype in the 3xTg-AD mice, with sex-specificity and their integration with non-social behaviors. We assessed a rank of social aspects, from affiliative behaviors and dominance to aggression, using experimental and naturalistic conditions that can be used for behavioral monitoring. Thus, we used social interaction and tube-dominance tests but also we recorded affiliative behaviors developed in the home cage such as sleeping behavior and barbering.
The quantitative and qualitative analysis of the ethogram elicited in the SIT evidenced that the expression of social behaviors (body/face contact and ano-genital contact, no aggressions) was very consistent in NTg mice, independently of sex. The behavioral repertoire started with a neophobia response toward the environment (horizontal and vertical exploration of the new cage), followed by the interaction with conspecifics, and afterwards by the expression of behaviors related to enhanced emotionality such as grooming behavior and digging. In contrast, the ‘vibrating tail’ appeared as a distinctive profile for female sex, as it was earlier expressed. This behavior, when elicited in rodents by systemic administration of glutamatergic agonist NMDA, is related to increased excitability [16]. In agreement with the increased vulnerability of 3xTg-AD to the occurrence of anxious-like behaviors in new environments [8], the ethogram in the SIT was found disrupted in the 3xTg-AD mice. The ‘vibrating tail’, also the most distinctive behavior of the female sex profile, was significantly enhanced and advanced on time. Thus, it was early expressed, in more occasions (episodes) and had longer total duration. These variables entitle vibrating tail as a selective and sensitive tool for sex-dependent expression of the social anxiety-like profile in 3xTg-AD mice.
Digging in the sawdust was sensitive to genotype and sex. In a precedent work, we showed that 12-month-old male 3xTg-AD mice showed increased marble-burying that was related to neophobia, modulable by handling, reversed by chronic treatment with risperidone [17] and modified by chronic caffeine [18]. This behavior has been considered to serve to measure digging rather than evoke burying [19], while Deacon [20] typified marble burying and digging in mice as simple methods for in vivo identification of biological impacts.
Self-grooming was also affected in the SIT, in agreement with delayed grooming in 15-month-old male and female 3xTgAD assessed in an open-field test [21] but advanced in females when engaged in active copying-with-stress strategies [22]. Here, latency of self-grooming allowed to distinguish the female 3xTg-AD genotype. According to the ‘action programs’ described by Lat [23] for mice and rats when they habituate in the open-field, self-grooming emerges once the other behaviors or actions (fear-explorations) have been completed. Therefore, when the previous actions are altered, as it is the case here, the latency of self-grooming reflects the previous sequence of behaviors, and could be consider less ‘specific’ by itself. For this reason, grooming was not chosen among the behaviors cited as relevant for the behavioral signature. With regards to social grooming in home-cage conditions, mutual grooming is considered an affiliative behavior. However, presence of barbering described as Dalila effect, more prevalent in females, illustrates a unidirectional behavior resulting of barber whiskers by plucking [15] done by one mouse (Dalila) that remains with its fur intact. There are several hypotheses about this phenomenon in rodents such as genetic, environmental, social hierarchies, stress, and certain disorders of obsessive-compulsive spectrum [13]. Here, the Dalila effect was present only in the NTg females, while 3xTg-AD females exhibited similar performances in the tube-dominance test.
Common genetic mechanisms link sensorimotor gating deficits with abnormal social behavior. For instance, Dvl1 knock out mice modeling psychotic disorders show differences in whisker trimming, deficits in nest-building, less huddling sleeping, subordinate responses, and deficits in acoustic and tactile prepulse inhibition [11]. In the case of 3xTgAD mice, this spectrum is only partially observed as impaired individual [24] and group [25] nesting, some deficits in their prepulse startle response [26], social alterations when interacting with NTg mice [27], and here we report reduced social interaction with congeners in SIT and lack of whisker trimming. However, sleeping behavior was found intact.
These differences in social behavior of 3xTg-AD mice may be associated not only to their prominent anxious-like profile exhibited since young adulthood [8]. Several authors postulate it can be influenced by apathy, agitation, possible deficits social memory, all considered as first clinically symptomatic manifestations of AD [6, 28]. Bories et al. [27], the first to report both sex- and age-dependent alterations in social behavior in the 3xTg-AD mice, evaluated the deficits in terms of normal/enhanced/reduced patterns measured as sniffing, crawling over or under, following, escape behavior, and grooming the partner. In their social interaction paradigm, they tested 3xTg-AD versus NTg pairs of aged- and sex-matched animals, minimizing the occurrence of aggressive behaviors and anxiogenic situations, as also shown by no differences in grooming behavior observed during the SIT. In their work, the authors reported that male dyads of one 3xTg-AD mice with NTg partners showed similar number of social events in the SIT at 12 months of age but social disinhibition at 18 months. In contrast, females showed an advanced pattern with disinhibition followed by decreased sociability. Most importantly, as shown by correlation analysis, social changes were not related to genetically-induced amyloid-β or tau neuropathological hallmarks of disease. Instead, social changes were found correlated to an increase of both excitatory and inhibitory basal synaptic activity in the medial prefrontal cortex. Studies with other animal models such as mice APPswe/PS1 have also shown impairment in social interaction in the 3-chamber test at 6 months of age, with mutants being less willing to engage in social interaction than wild-type, whereas being hyperactive in the open-field and elevated plus-maze. They associated it with deficits in social memory [29, 30]. At the same age, male Thy1-hAPPLond/Swe + mice assessed in this test also displayed reduced social interest, interpreted by decreased social memory or discrimination [31]. 23-month-old female Tg2576 mice are also impaired in social memory and open-field habituation tests [32].
The present work using a social paradigm of genotype and sex-matched dyads contributes to the growing efforts to study social behaviors in rodents [1,9] with special attention to its complexity in models of AD (i.e., [25, 27, 29–31]). This is important because several studies have shown that social environment (rearing conditions, enrichment, cohabitation, isolation, psychosocial stress) can be determinant and exert distinct impact in cognitive and BPSD-like symptoms as well as underlying neurobiological substrates and AD neuropathology [30, 33–35]. Finally, we propose that the identification of genotype- and sex-specific variables of behavior that can be used as signatures are of special interest for behavioral monitoring of animals in husbandry routines as well as in the follow-up during the assessment of preventive/therapeutic strategies, mainly in female sex which is the one at higher risk of developing the disease [36].
ACKNOWLEDGMENTS
We thank Prof. Frank M LaFerla Institute for Memory Impairments and Neurological Disorders, Department of Neurobiology and Behavior, University of California, Irvine, USA, for kindly providing the progenitors of the Spanish colonies of 3xTg-AD and NTg mice. The work has been supported by Instituto de Salud Carlos III – Subdirección General de Evaluación y Fomento de la Investigación (AES-PI10/00283), co-financed by the European Regional Development Fund (ERDF), 2017-SGR-1468 and ArrestAD H2020 Fet-OPEN-1-2016-2017-737390 to LGL.
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/19-0253r2).
REFERENCES
[1] | Kondrakiewicz K , Kostecki M , Szadzińska W , Knapska E ((2019) ) Ecological validity of social interaction tests in rats and mice. Genes Brain Behav 18: , e12525. |
[2] | Crawley JN ((2004) ) Designing mouse behavioral tasks relevant to autistic-like behaviors. Ment Retard Dev Disabil Res Rev 10: , 248–258. |
[3] | Riedel G , Kang SH , Choi DY , Platt B ((2009) ) Scopolamine-induced deficits in social memory in mice: Reversal by donepezil. Behav Brain Res 204: , 217–225. |
[4] | Dyck BA , Skoblenick KJ , Castellano JM , Ki K , Thomas N , Mishra RK ((2009) ) Behavioral abnormalities in synapsin II knockout mice implicate a causal factor in schizophrenia. Synapse 63: , 662–672. |
[5] | Cummings JL ((2000) ) Cognitive and behavioral heterogeneity in Alzheimer’s disease: Seeking the neurobiological basis. Neurobiol Aging 21: , 845–861. |
[6] | Andrew MK , Mitnitski AB , Rockwood K ((2008) ) Social vulnerability, frailty and mortality in elderly people. PLoS One 3: , e2232. |
[7] | Rockwood K , Graham JE , Fay S , ACADIE Investigators ((2002) ) Goal setting and attainment in Alzheimer’s disease patients treated with donepezil. J. Neurol Neurosurg Psychiatry 73: , 500–507. |
[8] | Giménez-Llort L , Blázquez G , Cañete T , Johansson B , Oddo S , Tobeña A , LaFerla FM. , Fernandez-Teruel A ((2007) ) Modeling behavioral and neuronal symptoms of Alzheimer’s disease in mice: A role for intraneuronal amyloid. Neurosci Biobehav Rev 31: , 125–147. |
[9] | Holmes A ((2019) ) The neuroscience and genomics of social behavior. Genes Brain Behav 18: , e12551. |
[10] | Belfiore R , Rodin A , Ferreira E , Velazquez R , Branca C , Caccamo A , Oddo S ((2019) ) Temporal and regional progression of Alzheimer’s disease-like pathology in 3xTg-AD mice. Aging Cell 18: , e12873. |
[11] | Lijam N , Paylor R , McDonald MP , Crawley JN , Deng CX , Herrup K , Tweedie D , Perry T , Mattson MP , Kapogiannis D , Sambamurti K , Lahiri DK , Greig NH ((1997) ) Social interaction and sensorimotor gating abnormalities in mice lacking Dvl1. Cell 90: , 895–905. |
[12] | Venerosi A , Valanzano A , Alleva E , Calamandrei G ((2001) ) Prenatal exposure to anti-HIV drugs: Neurobehavioral effects of zidovudine (AZT)+lamivudine (3TC) treatment in mice. Teratology 63: , 26–37. |
[13] | Kalueff AV , Minasyan A , Keisala T , Shah ZH , Tuohimaa P ((2006) ) Hair barbering in mice: Implications for neurobehavioural research. Behav Processes 71: , 8–15. |
[14] | Garner JP , Weisker SM , Dufour B , Mench JA ((2004) ) Barbering (fur and whisker trimming) by laboratory mice as a model of human trichotillomania and obsessive-compulsive spectrum disorders. Comp Med 54: , 216–224. |
[15] | Sarna JR , Dyck RH , Whishaw IQ ((2000) ) The dalila effect: C57BL6 mice barber whiskers by plucking. Behav Brain Res 108: , 39–45. |
[16] | Giménez-Llort L , Martínez E , Camón L , de Vera N ((1997) ) Changes in brain putrescine concentration associated with nonconvulsant behavioral patterns induced by systemic N-methyl-D-aspartate injection. Mol Chem Neuropathol 30: , 289–302. |
[17] | Torres-Lista V , López-Pousa S , Giménez-Llort L ((2015) ) Marble-burying is enhanced in 3xTg-AD mice, can be reversed by risperidone and it is modulable by handling. Behav Processes 116: , 69–74. |
[18] | Baeta-Corral R , Johansson B , Giménez-Llort L ((2018) ) Long-term treatment with low-dose caffeine worsens BPSD-like profile in 3xTg-AD mice model of Alzheimer’s disease and affects mice with normal aging. Front Pharmacol 9: , 79. |
[19] | Deacon RM ((2006) ) Digging and marble burying in mice: Simple methods for in vivo identification of biological impacts. Nat Protoc 1: , 122–124. |
[20] | Gyertyán I ((1995) ) Analysis of the marble burying response: Marbles serve to measure digging rather than evoke burying. Behav Pharmacol 6: , 24–31. |
[21] | Giménez-Llort L , Arranz L , Maté I , De la Fuente M ((2008) ) Gender-specific neuroimmunoendocrine aging in a triple-transgenic 3xTg-AD mouse model for Alzheimer’s disease and its relation with longevity. Neuroimmunomodulation 15: , 331–343. |
[22] | Jiménez-Altayó F , Sánchez-Ventura J , Vila E , Giménez-Llort L ((2018) ) Crosstalk between peripheral small vessel properties and anxious-like profiles: Sex, genotype, and interaction effects in mice with normal aging and 3×Tg-AD mice at advanced stages of disease. J Alzheimers Dis 62: , 1531–1538. |
[23] | Lát J ((1973) ) The analysis of habituation. Acta Neurobiol Exp 33: , 771–789. |
[24] | Torres-Lista V , Giménez-Llort L ((2013) ) Impairment of nesting behaviour in 3xTg-AD mice. Behav Brain Res 247: , 153–157. |
[25] | Van der Jeugd A , Parra-Damas A , Baeta-Corral R , Soto-Faguás CM , Ahmed T , LaFerla FM , Giménez-Llort L , D’Hooge R , Saura CA ((2018) ) Reversal of memory and neuropsychiatric symptoms and reduced tau pathology by selenium in 3xTg-AD mice. Sci Rep 8,: , 6431. |
[26] | Garcia-Mesa Y , Lopez-Ramos JC , Giménez-Llort L , Revilla S. Guerra R , Gruart A , Laferla FM , Cristofol R , Delgado-Garcia JM , Sanfeliu C ((2011) ) Physical exercise protects against Alzheimer’s disease in 3xTg-AD mice. J Alzheimers Dis 24: , 421–454. |
[27] | Bories C , Guitton MJ , Julien C , Tremblay C , Vandal M , Msaid M , De Koninck Y , Calon F ((2012) ) Sex-dependent alterations in social behaviour and cortical synaptic activity coincide at different ages in a model of Alzheimer’s disease. PLoS One 7: , e46111. |
[28] | Norton MC , Dew J , Smith H , Fauth E , Piercy KW , Breitner JC , Tschanz J , Wengreen H , Welsh-Bohmer K , Cache County Investigators ((2012) ) Lifestyle behavior pattern is associated with different levels of risk for incident dementia and Alzheimer’s disease: The cache county study. J Am Geriatr Soc 60: , 405–412. |
[29] | Filali M , Lalonde R , Rivest S ((2011) ) Anomalies in social behaviors and exploratory activities in an APPswe/PS1 mouse model of Alzheimer’s disease. . Physiol Behav 104: , 880–885. |
[30] | Huang H , Liang K , Ke H , Chang Y , Hsieh-Li HM ((2011) ) Long-term social isolation exacerbates the impairment of spatial working memory in APP/PS1 transgenic mice. Brain Res 1371: , 150–160. |
[31] | Faizi M , Bader PL , Saw N , Nguyen TV , Beraki S , Wyss-Coray T , Longo FM , Shamloo M ((2012) ) Thy1-hAPP(Lond/Swe+) mouse model of Alzheimer’s disease displays broad behavioral deficits in sensorimotor, cognitive and social function. Brain Behav 2: , 142–154. |
[32] | Deacon RM , Koros E , Bornemann KD , Rawlins JN ((2009) ) Aged Tg2576 mice are impaired on social memory and open field habituation tests. Behav Brain Res 197: , 466–468. |
[33] | Pietropaolo S , Sun Y , Li R , Brana C , Feldon J , Yee BK ((2009) ) Limited impact of social isolation on Alzheimer-like symptoms in a triple transgenic mouse model. Behav Neurosci 123: , 181–195. |
[34] | Rothman SM , Herdener N , Camandola S , Texel SJ , Mughal MR , Cong WN , Martin B , Mattson MP ((2012) ) 3xTgAD mice exhibit altered behavior and elevated Aβ after chronic mild social stress. Neurobiol Aging 33: , 830.e1–12. |
[35] | Hsiao YH , Hung HC , Chen SH , Gean PW ((2014) ) Social interaction rescues memory deficit in an animal model of Alzheimer’s disease by increasing BDNF-dependent hippocampal neurogenesis. J Neurosci 34: , 16207–16219. |
[36] | Azad N , Al Bugami M , Loy-English I ((2007) ) Gender differences in dementia risk factors. Gender Med 4: , 120–129. |




