Ethical Issues in the Treatment of Late-Stage Alzheimer’s Disease
Abstract
There is hope that the continuing efforts of researchers will yield a disease-modifying drug for Alzheimer’s disease. Such a drug is likely to be capable of halting, or significantly slowing, the underlying pathological processes driving cognitive decline; however, it is unlikely to be capable of restoring brain function already lost through the pathological process. A therapy capable of halting Alzheimer’s disease, while not providing restoration of function, may prompt serious ethical questions. For example, is there a stage in the disease process when it becomes too late for therapeutic intervention to commence? And who bears the responsibility of making such a decision? Conversations regarding the ethics of treating neurodegenerative conditions with non-restorative drugs have been largely absent within both clinical and research communities. Such discussions are urgently required to ensure that patients’ rights and well-being are protected when such therapeutic options become available.
Approximately 50 million people around the world are currently living with a dementia diagnosis [1]. By 2050, the number of people with dementia is projected to rise to 135 million, with Alzheimer’s disease (AD) accounting for up to 80% of these cases [2]. Over the past two decades, a concerted effort has been made toward developing disease-modifying drugs that are capable of preventing or halting the underlying pathology of AD. Despite their limited success to date, it is hoped that continued efforts in the field will ultimately yield an effective treatment for AD. The development of this drug would be a significant breakthrough in medical research and would provide the much needed first steps to be taken toward reducing the burden of this insidious disease. Concurrent efforts to identify sensitive and reliable preclinical biomarkers of the disease would also likely impact the efficacy of such a compound and enable preventative or prophylactic treatment options for those found to be at risk.
To date, therapeutic strategies have largely focused on targeting the accumulation of amyloid-β (Aβ) peptides in the brain, a pathological hallmark of AD. These strategies have involved targeting the peptide directly, or through mediators (e.g., β- and γ-secretase inhibition) to inhibit its overproduction and aggregation and/or increase its clearance from the brain. More recently the tau protein, the other pathological hallmark of the disease, has been investigated as a potential target [3]. However, regardless of which pathological target eventually yields the successful treatment, the drug is likely to have the following two traits: 1) Be capable of preventing, halting, or significantly slowing the underlying pathological processes driving cognitive decline; 2) Not be capable of restoring brain function or tissue, already lost through the pathological process.
A therapy capable of halting AD, while not providing restoration of function, raises a number of ethical questions that require careful consideration. For example, what is the goal of treatment in late-stage AD? Are there stages in the disease process when it becomes too late for this type of effective therapeutic intervention to commence? And who bears the burden of responsibility for making these decisions? To date, such conversations have been largely absent within clinical, research, and consumer communities; however, such discussions are required if we are to ensure that the rights and well-being of those affected are protected when disease-modifying therapeutic options become available.
TREATING PRECLINICAL, MILD-MODERATE, AND LATE STAGE ALZHEIMER’S DISEASE
In the pre-clinical phase of AD, secondary prevention by use of a disease-modifying therapy would provide substantial benefit for those individuals found to be at risk of developing the disease. In such cases, the administration of a disease-modifying drug may prevent the symptomatic onset of the disease entirely, making it imperative to diagnose and initiate therapy as early as possible. Such an approach may require extended courses of treatment to achieve the desired outcome and would be premised on the assumption that approved therapies were safe over the long-term. Prophylactic treatment in these cases would be akin to the use of statins to lower the incidence of cardiac disease in at-risk populations.
In mild to moderate AD, beneficial results may be seen as the burdens of inflammation and neurotoxicity are lifted. Additional cognitive improvements may also occur over time, as disease progression halts and the remaining neural compensatory mechanisms [4] enable some functional restoration to occur. This functional restoration could be further assisted through non-pharmacological interventions such as exercise and other lifestyle changes that have been reported to have positive effects on cognition [5] and increase circulating levels of brain-derived neurotrophic factor [6]. Unfortunately, they are unlikely to substantially recoup the volumetric loss of cerebral tissue that has occurred throughout the disease process. Importantly, individuals with mild to early moderate stage dementia, particularly those with a higher degree of insight into their condition, may be competent to make or contribute to the treatment decisions [7] even if they are unable to make the final treatment decision [8].
Fig. 1.
The progression of Alzheimer’s disease. As the pathological burden of Alzheimer’s disease increases there may come a time when it becomes medically futile to commence disease-modifying therapies. Diagnostic designations include mild cognitive impairment (MCI), and Alzheimer’s disease (AD).
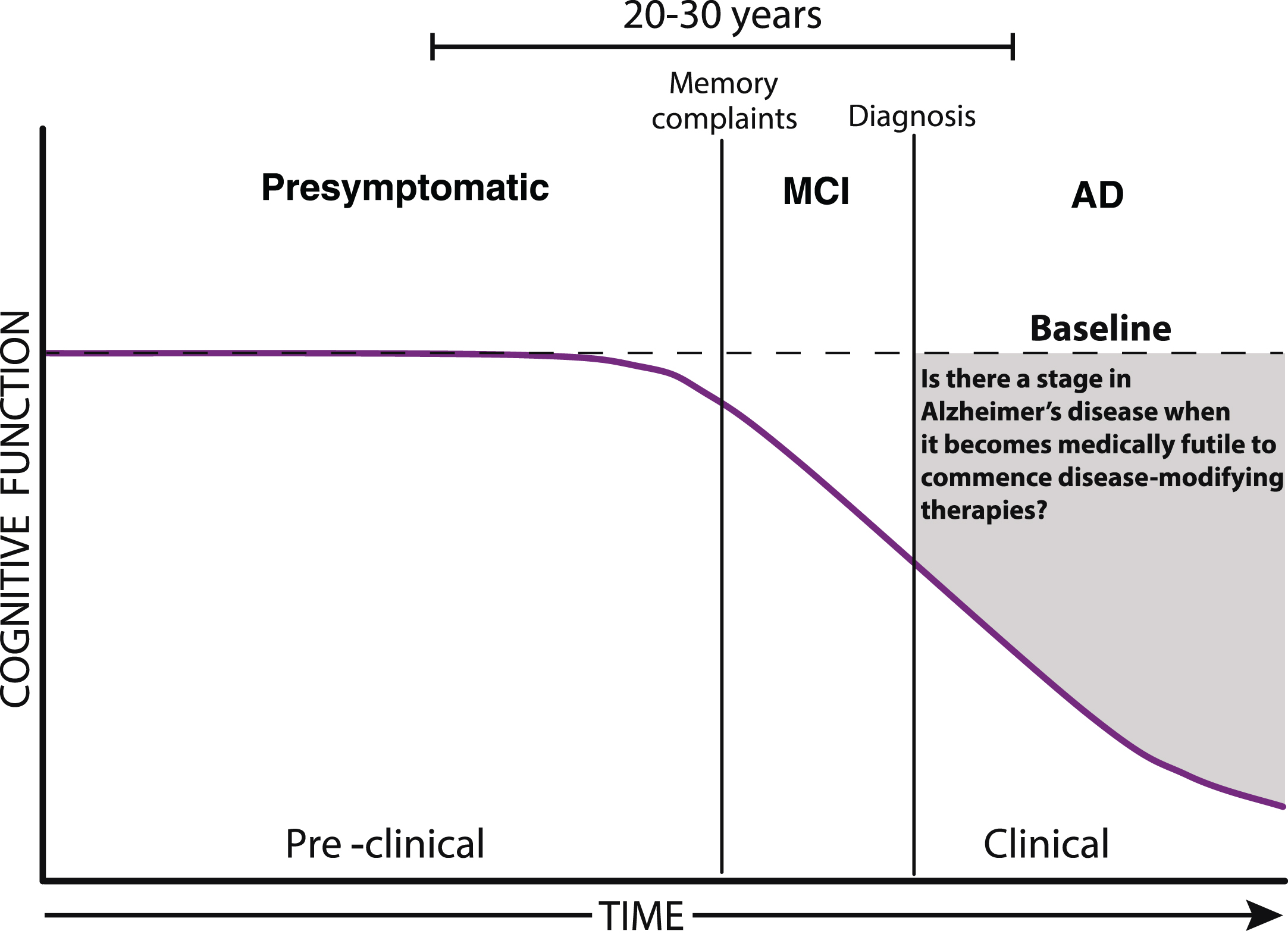
In late-stage or severe AD, affected individuals will have lost the ability to make treatment decisions, and the administration of a disease-modifying therapy is unlikely to result in a significant reduction of disease burden. The administration of such a drug may instead result in a perpetuated state of severe cognitive disability, where despite the absence of progressive neurological decline, individuals would arguably have little quality of life and very low likelihood of significantly regaining lost function.
From a research standpoint, this drug would be successful due to its ability to target and halt the underlying disease processes occurring in the brain. Nonetheless, when viewed from the perspective of those diagnosed with dementia and/or their families, an effective treatment may be more likely to be construed as synonymous with a cure; that is, a drug that restores function to a pre-diagnosis baseline (Fig. 1). This discrepancy raises the concern that certain eventualities of treatment may not have been adequately considered. In particular, the possibility that despite treatment affected individuals will continue to suffer from an ongoing cognitive disability and associated high-levels of required care. If such outcomes were not adequately discussed, caregivers would be left ill-prepared to tackle the additional emotional and financial challenges of the extended care that would subsequently ensue. The potential disconnect between clinicians and primary care teams highlight the importance of appropriately informing patients and caregivers of the likely outcomes of late-stage treatment interventions. This is of particular importance given the often over-reaching nature of media reports on the subject. Open and honest discussions are a defining characteristic of positive clinician-patient relationships. Given the high cost of miscommunication in the treatment of individuals with late-stage disease, it serves to highlight the importance of such frontline discussions. To paraphrase Dunn et al. [9], fulfilling goals of care that honor the rights and attend to the quality of life of people living with AD while balancing the ethical principles of autonomy, non-maleficence, and beneficence is extremely challenging in these clinical settings.
ETHICAL ISSUES IN TREATING ALZHEIMER’S DISEASE
The ethical implications of preventative treatments begin to arise in the preclinical and mild stages of AD. For example, what clinical measures provide a deterministic rather than probabilistic diagnosis of preclinical disease? At what stage should such interventions occur given the uncertain probability of transition to overt or more significant clinical disease and the possible side effects that may arise? And who should be involved in this decision-making process? Such discussions are of the utmost importance and require careful consideration. This commentary will, however, focus on the ethical issues that arise in late-stage disease.
Ethical decision making in Alzheimer’s disease
Individuals living with a dementia diagnosis and/or their caregivers, herein referred to as the primary care team, are currently charged with considering the ethical implications of non-disease modifying interventions, such as the use of enteral intubation to provide nutrition and hydration to individuals who are otherwise unable to consume sufficient food and water (see [10] for a review). Anecdotally, such interventions are thought to prolong life for individuals with late-stage dementia; however, a growing body of evidence indicates that no significant differences in survival rates occur between those who undergo enteral intubation and those who do not [11, 12]. By comparison, the administration of a disease-modifying therapeutic would alleviate disease-associated mortality, potentially adding years to an individual’s lifespan with little or no subsequent restoration in cognition. While parallels can be drawn between the ethical dilemmas faced in late-stage AD and other terminal diseases, such as cancer, HIV/AIDs, or renal disease [13–15], the confounding impact of the individual’s inability to participate in the decision-making progress creates additional challenges when treating dementia. More apt comparisons are perhaps made with the dilemmas arising in the treatment of severe stroke which may involve the administration of life sustaining treatments, such as mechanical ventilation, in the absence of restorative therapies [16]. Here, despite declining mortality [17], approximately 40% of patients die within the 12 months following a severe stroke [18, 19]. As in late stage AD, treatment decisions following severe stroke are complex and are often made with incomplete prognostic information.
Supporting ethical decisions in late-stage Alzheimer’s disease
In the treatment of late-stage AD, ethical decision making is not limited to issues surrounding patient autonomy. Such discussion must include consideration of the central tenets of medical ethics: beneficence (actions should benefit the patient), non-maleficence (actions should not harm the patient), justice (patients should be treated equally), and respect for autonomy; in addition to the patient’s best interests and professional integrity [20, 21].
A treatment that increases lifespan without enhancing quality of life does not necessarily harm the patient but neither does it necessarily benefit them. Such treatments raise the concept of medical futility (interventions that are unlikely to produce meaningful benefit to the patient). Futility may be defined as being either quantitative or qualitative; quantitative futility refers to a situation where the likelihood that an intervention will provide benefit to the patient is very low, while qualitative futility refers to a situation where the quality of the benefit provided to the patient by the treatment is very low [22, 23]. Sustaining physiological function in individuals with end-stage neurodegenerative conditions may appear futile [24] and clinicians are not obligated to provide treatments that are thought to be ineffective [25]. This may particularly be the case for patients receiving palliative care as such treatments would run counter to the underlying principles of palliation [26]. However, treatment decisions should be weighed judiciously and compassionately in accordance with the primary care team and the patient’s prior wishes. While most people agree that futile interventions should not be administered, different definitions on what constitutes a futile intervention can present challenges in clinical settings [24, 27].
It is also important to note that while restorative treatments are not available at present, they may become available within a patient’s lifespan. Thus, a treatment that halts the pathological progression of AD while preserving other physiological function would enable an individual to take advantage of future restorative therapies should they become available.
Where does the burden of responsibility fall?
Whether an individual in the late stages of AD commences treatment is a decision that is likely to occur at the level of the primary care team through consultation with clinicians. Input from both industry and government are also likely.
Placing decisions at the level of the primary care team and their clinicians, while crucial, will place a considerable burden of responsibility on frontline clinical staff who must ensure that primary caregivers are fully informed of the prognostic likelihood and probable long-term outcome of any given strategy. Clinicians must be explicit in explaining the non-restorative nature of these treatments and outline the potential consequences of treating individuals with late-stage disease. This will not be an easy task. For the individual or immediate family, this decision will be even more loaded with consideration of the potential for extended high level care. Such a decision will be highly dependent on the context of those affected, for some, a medication that helps someone in late-stage disease regain independent toileting function may be more valuable than one that assists someone with mild disease regain some instrumental activity of daily living (e.g., driving, food preparation, or financial management). For others the opposite may be true. For primary care teams, the competing viewpoints of individuals within the group will need to be weighed against the views of the affected individual, if they are known. Such considerations will also need to account for the logistical challenges that come with providing extended high-level care to individuals once treatment is commenced. For example, whether supporting the individual within the family home or care facility will be financially sustainable. It is equally important to consider the emotional and mental health outcomes of such decisions. For example, significant levels of guilt may be felt regardless of whether treatment was commenced or not (e.g., following an individuals’ death or subsequent to their prolonged disability). In other cases, individuals may report that their role as caregiver provides meaning to their lives and strengthens their relationships with family and friends [28, 29]. In either case, the inclusion of allied health professionals (e.g., psychologists, specialist counsellors) in the process may be of benefit in the decision-making process.
Late-stage treatment decisions may be further complicated by the input of regulatory bodies such as the United States Food and Drug Administration. Such bodies could approve a disease-modifying therapeutic while stipulating that they only be prescribed to individuals in the pre-clinical and/or early stages of the disease. This would require somewhat arbitrary clinical demarcation of the stages of AD resulting in a binary decision with a cut-off representing treatment worthy and non-treatment worthy patient populations. Such a distinction would raise additional ethical questions, regarding where the cut-off is made and how to ensure that any regulatory guidelines were adequate to make such demarcations. It should be noted that, once the drug received regulatory approval, there would be little to stop clinicians from prescribing it for off-label usage at later stages of the disease. These prescriptions could, however, be limited by only approving government, or insurance, co-payments for prescriptions provided to individuals in the early stages of the disease. This would significantly lessen off-label prescriptions; however, it could also place an undue financial strain if treatment is paid for out-of-pocket, particularly if the family is then left with the subsequent challenge of prolonged high-level care.
On a corporate level, employees and representatives of listed companies, such as those in the pharmaceutical and/or medical insurance industries, may be driven by internal moral or ethical frameworks that seek to end the suffering associated with dementia. The institutions themselves, however, lack any such internal ethical structure, and are instead constrained by their fiduciary responsibilities to target the largest consumer market and maximize profits for shareholders. The global market for an AD treatment is estimated to reach US$13.3 billion by 2023 [30]. The publicly provided information from such companies regarding both the efficacy and function of therapeutics for AD should therefore be carefully monitored and given the appropriate weight in the ensuing discussion.
Advance care planning
In some instances, an advance directive or living will may have been prepared prior to the onset of cognitive decline. These are initiated by approximately 40% of people with cognitive impairments and AD in the five years following diagnosis [31]. Advance directives aim to outline the care preferences of the individual living with dementia on issues including intubation, medication, and palliation. Without knowing the specific details of the treatment options that would become available, directives outlining an individual’s preferences for treatment in the advent of disease-modifying therapeutics would be open to scrutiny regarding what and who defines a ‘successful disease-modifying agent’. The legal status and regulation of directives differs across jurisdictions and their value has been questioned due to ongoing controversies regarding whether changes in ones interests and personal identity following the clinical onset of dementia should be valued above those outlined prior to symptom onset [32]. However, regardless of the legal requirements for the implementation of the directive, the inclusion of disease-modifying treatments in advance care planning may provide an overview of the individuals preferences that assists their primary care team in selecting the most appropriate treatment option.
CONCLUSION
The ethical questions raised by the treatment of late-stage AD are likely to extend beyond those described here. Some of these will be manageable through simple measures, such as the provision of educational materials for clinicians and consumers to assist in guiding which treatment strategies are appropriate for each stage of the disease. Others, however, may require government oversight to ensure that those living with the disease are protected and informed as they assess their treatment options. No specific ethical framework or particular position is intended herein; rather we seek to facilitate discussion on how best to ensure the highest level of appropriate care for individuals affected by all stages of AD when a disease-modifying treatment becomes available.
ACKNOWLEDGMENTS
We acknowledge the Victorian Government’s Operational Infrastructure Support Program. SC is supported in part by a NHMRC Practitioner Fellowship (#APP1105784).
Authors’ disclosures available online (https://www.j-alz.com/manuscript-disclosures/18-0865r1).
REFERENCES
[1] | Wimo A , Guerchet M , Ali G-C , Wu Y-T , Prina AM , Win-blad B , Jonsson L , Liu Z , Prince M The worldwide costs of dementia 2015 and comparisons with 2010. Alzheimers Dement 13: , 1–7. |
[2] | Alzheimer’s Disease International (2015) The Global Impact of Dementia: An analysis of prevalence, incidence, cost and trends. London, UK. |
[3] | Gotz J , Ittner A , Ittner LM ((2012) ) Tau-targeted treatment strategies in Alzheimer’s disease. Br J Pharmacol 165: , 1246–1259. |
[4] | Elman JA , Oh H , Madison CM , Baker SL , Vogel JW , Marks SM , Crowley S , O'Neil JP , Jagust WJ ((2014) ) Neural compensation in older people with brain amyloid-[beta] deposition. Nat Neurosci 17: , 1316–1318. |
[5] | Groot C , Hooghiemstra AM , Raijmakers PG , van Berckel BN , Scheltens P , Scherder EJ , van der Flier WM , Ossenkop-pele R ((2016) ) The effect of physical activity on cognitive function in patients with dementia: A meta-analysis of randomized control trials. Ageing Res Rev 25: , 13–23. |
[6] | Coelho FG , Vital TM , Stein AM , Arantes FJ , Rueda AV , Camarini R , Teodorov E , Santos-Galduroz RF ((2014) ) Acute aerobic exercise increases brain-derived neurotrophic factor levels in elderly with Alzheimer’s disease. J Alzheimers Dis 39: , 401–408. |
[7] | Karlawish JHT , Casarett DJ , James BD , Xie SX , Kim SYH ((2005) ) The ability of persons with Alzheimer disease (AD) to make a decision about taking an AD treatment. Neurology 64: , 1514–1519. |
[8] | Hirschman KB , Joyce CM , James BD , Xie SX , Karlawish JH ((2005) ) Do Alzheimer’s disease patients want to participate in a treatment decision, and would their caregivers let them? Gerontologist 45: , 381–388. |
[9] | Dunn LB , Alici Y , Roberts LW ((2015) ) Ethical challenges in the treatment of cognitive impairment in aging. Curr Behav Neurosci Rep 2: , 226–233. |
[10] | Johnson RA , Karlawish J ((2015) ) A review of ethical issues in dementia. Int Psychogeriatr 27: , 1635–1647. |
[11] | Goldberg LS , Altman KW ((2014) ) The role of gastrostomy tube placement in advanced dementia with dysphagia: A critical review. Clin Interv Aging 9: , 1733–1739. |
[12] | Brooke J , Ojo O ((2015) ) Enteral nutrition in dementia: A systematic review. Nutrients 7: , 2456–2468. |
[13] | Macklin R ((1984) ) Ethical issues in treatment of patients with end-stage renal disease. Soc Work Health Care 9: , 11–20. |
[14] | Simonds A ((2003) ) Ethics and decision making in end stage lung disease. Thorax 58: , 272–277. |
[15] | Hlongwa P ((2016) ) Current ethical issues in HIV/AIDS research and HIV/AIDS care. Oral Dis 22: , 61–65. |
[16] | Creutzfeldt CJ , Holloway RG ((2012) ) Treatment decisions after severe stroke. Uncertain Biases 43: , 3405–3408. |
[17] | Benjamin EJ , Blaha MJ , Chiuve SE , Cushman M , Das SR , Deo R , de Ferranti SD , Floyd J , Fornage M , Gillespie C , Isasi CR , Jimenez MC , Jordan LC , Judd SE , Lackland D , Lichtman JH , Lisabeth L , Liu S , Longenecker CT , Mackey RH , Matsushita K , Mozaffarian D , Mussolino ME , Nasir K , Neumar RW , Palaniappan L , Pandey DK , Thiagarajan RR , Reeves MJ , Ritchey M , Rodriguez CJ , Roth GA , Rosamond WD , Sasson C , Towfighi A , Tsao CW , Turner MB , Virani SS , Voeks JH , Willey JZ , Wilkins JT , Wu JH , Alger HM , Wong SS , Muntner P ((2017) ) Heart Disease and Stroke Statistics-2017 Update: A report from the American Heart Association. Circulation 135: , e146–e603. |
[18] | Roberts SE , Thorne K , Akbari A , Samuel DG , Williams JG ((2015) ) Mortality following stroke, the weekend effect and related factors: Record linkage study. PLoS One 10: , e0131836. |
[19] | Eriksson H , Milberg A , Hjelm K , Friedrichsen M ((2016) ) End of life care for patients dying of stroke: A comparative registry study of stroke and cancer. PLoS One 11: , e0147694. |
[20] | Smith ML ((2013) ) Ethical issues in stroke patients. In Handbook of Clinical Nutrition and Stroke, Corrigan ML, Escuro AA, Kirby DF, eds. Humana Press, Totowa, NJ, pp. 269–280. |
[21] | Gillon R ((1994) ) Medical ethics: Four principles plus attention to scope. BMJ 309: , 184–188. |
[22] | Schneiderman LJ , Jecker NS , Jonsen AR ((1990) ) Medical futility: Its meaning and ethical implications. Ann Intern Med 112: , 949–954. |
[23] | Schneiderman LJ ((2011) ) Defining medical futility and improving medical care. J Bioeth Inq 8: , 123–131. |
[24] | Low J , Ho E ((2017) ) Managing ethical dilemmas in end-stage neurodegenerative diseases. Geriatrics 2: , 8. |
[25] | Kasman DL ((2004) ) When is medical treatment futile?: A guide for students, residents, and physicians. J Gen Intern Med 19: , 1053–1056. |
[26] | Volicer L , Simard J ((2015) ) Palliative care and quality of life for people with dementia: Medical and psychosocial interventions. Int Psychogeriatr 27: , 1623–1634. |
[27] | Aghabarary M , Dehghan Nayeri N ((2016) ) Medical futility and its challenges: A review study. J Med Ethics Hist Med 9: , 11. |
[28] | Schulz R , Sherwood PR ((2008) ) Physical and mental health effects of family caregiving. Am J Nurs 108: , 23–27. |
[29] | Schulz R , Newsom J , Mittelmark M , Burton L , Hirsch C , Jackson S ((1997) ) Health effects of caregiving: The caregiver health effects study: An ancillary study of the Cardiovascular Health Study. Ann Behav Med 19: ,110–116. |
[30] | GlobalData Healthcare ((2015) ) PharmaPoint: Alzheimer’s Disease - Global Drug Forecast and Market Analysis to 2023. |
[31] | Garand L , Dew MA , Lingler JH , DeKosky ST ((2011) ) Incidence and predictors of advance care planning among persons with cognitive impairment. Am J Geriatr Psychiatry 19: , 712–720. |
[32] | Porteri C ((2018) ) Advance directives as a tool to respect patients' values and preferences: Discussion on the case of Alzheimer’s disease. BMC Med Ethics 19: , 9. |




