Efficacy and Safety of Plasma Exchange with 5% Albumin to Modify Cerebrospinal Fluid and Plasma Amyloid-β Concentrations and Cognition Outcomes in Alzheimer’s Disease Patients: A Multicenter, Randomized, Controlled Clinical Trial
Abstract
Background: Studies conducted in animal models and humans suggest the presence of a dynamic equilibrium of amyloid-β (Aβ) peptide between cerebrospinal fluid (CSF) and plasma compartments.
Objective: To determine whether plasma exchange (PE) with albumin replacement was able to modify Aβ concentrations in CSF and plasma as well as to improve cognition in patients with mild-moderate Alzheimer’s disease (AD).
Methods: In a multicenter, randomized, patient- and rater-blind, controlled, parallel-group, phase II study, 42 AD patients were assigned (1 : 1) to PE treatment or control (sham) groups. Treated patients received a maximum of 18 PE with 5% albumin (Albutein®, Grifols) with three different schedules: two PE/weekly (three weeks), one PE/weekly (six weeks), and one PE/bi- weekly (12 weeks), plus a six-month follow-up period. Plasma and CSF Aβ1–40 and Aβ1–42 levels, as well as cognitive, functional, and behavioral measures were determined.
Results: CSF Aβ1–42 levels after the last PE compared to baseline were marginally higher in PE-treated group versus controls (adjusted means of variation: 75.3 versus –45.5 pg/mL; 95% CI: –19.8, 170.5 versus 135.1, 44.2; p = 0.072). Plasma Aβ1–42 levels were lower in the PE-treated group after each treatment period (p < 0.05). Plasma Aβ1–40 levels showed a saw-tooth pattern variation associated with PE. PE-treated patients scored better in the Boston Naming Test and Semantic Verbal Fluency (p < 0.05) throughout the study. Neuropsychiatric Inventory scores were higher in controls during the PE phase (p < 0.05).
Conclusion: PE with human albumin modified CSF and plasma Aβ1–42 levels. Patients treated with PE showed improvement in memory and language functions, which persisted after PE was discontinued.
INTRODUCTION
Alzheimer’s disease (AD) is the most frequent form of dementia in the adult, and its prevalence increases exponentially with age [1]. AD is a neurodegenerative process characterized by progressive cognitive deficits in multiple cognitive domains and impairments in activities of daily living, as well as neurological and psychiatric symptoms [2]. The initial pathological event that leads to neurodegeneration (neuronal and synaptic loss) is unknown. However, the accumulation of extracellular amyloid-β (Aβ) in the form of diffuse and neuritic plaques and intracellular accumulation of phosphorylated tau in the form of neurofibrillary tangles, neuropil threads, and dystrophic neurites constitutes the most salient neuropathological feature [3]. Only symptomatic therapies are available for the treatment of AD, and disease-modifying compounds are currently being tested, especially medications that target Aβ metabolism [4, 5].
Aggregation and accumulation of Aβ proteins in the brain has been considered a defining pathology associated with AD [3], and it has been suggested that the sporadic form of AD is related to decreased clearance of Aβ from the central nervous system (CNS) [6]. Aβ proteins are the most important component of the neuritic (senile) plaque (SNP). Aβ1–42 is the predominant form in the SNP, and is deposited first, while the Aβ1–40 is deposited later [7]. CSF Aβ1–42 levels are decreased in AD patients, while Aβ1–40 levels remain normal [8]. Similarly, Aβ proteins are detectable in plasma; some studies have shown increased Aβ1–42 levels in early AD [9], while others have shown decreased Aβ1–42 levels, and Aβ1–42/Aβ1–40 ratio [10].
Studies conducted in animal models and humans have detected the presence of a bidirectional flow of Aβ peptide between the CSF and plasma compartments [11–13]. It has been reported that direct transport of Aβ through the blood-brain barrier represents 25% of its clearance [14] in cognitively normal humans. Studies conducted in animal models showed that the elimination half-life of Aβ between CSF and plasma is about 30 minutes [12, 15, 16]. Plasma levels of Aβ decrease as the deposits of the latter within the brain increase [17]. These findings suggest that there is equilibrium between plasma and CNS Aβ, and that SNP are continuously creating a new equilibrium because Aβ not only enters the plasma but also deposits in the CNS [18]. This dynamic equilibrium between Aβ in plasma and brain can be altered with therapeutic interventions that target Aβ pathology. Peripheral sequestration of plasma Aβ resulted in a reduction of Aβ in both the CSF and in the brain of transgenic mice [19].
PE is a process used to remove patient plasma and replace it with another solution, maintaining normal volemia and osmotic balance, with the purpose of eliminating toxic substances from plasma such as autoantibodies, alloantibodies, immune complexes, proteins, and toxins [20]. To this effect, albumin is frequently used as the replacement solution in PE. By taking advantage of the fact that the vast majority of Aβ circulating in blood (around 90%) is bound to albumin in a 1:1 ratio [21], preliminary phase I studies in humans using plasma exchange (PE) with 5% human albumin (Albutein®) showed that PE was able to induce alterations in the Aβ1–42 mobilization (i.e., a trend to increase in CSF and a clear decrease in plasma after the PE period, returning to baseline levels at 6 months of follow-up) and that this finding may be related to a tendency of stabilizing the scores in cognition tests, even after 1 year of follow-up [22]. In a confirmation of the occurrence of Aβ mobilization in CSF and plasma which was associated with improved scores in cognitive tests, in this study, we further describe and test PE with albumin replacement as a novel strategy for the treatment of AD.
MATERIALS AND METHODS
This was a multicenter, randomized, patient- and rater-blind, controlled phase II trial (EudraCT number: 2007-000414-36), conducted in compliance with a clinical protocol, regulatory requirements, good clinical practice (GCP), and the ethical principles of the latest revision of the Declaration of Helsinki as adopted by the World Medical Association. The study was reviewed and approved by Independent Ethics Committees (IEC) and Institutional Review Boards (IRB).
Objectives
The primary objective was to determine whether PE treatment with 5% human albumin was able to modify the concentration of Aβ (Aβ1–40 and Aβ1–42) in CSF in patients with mild to moderate AD.
The secondary objectives were: 1) to determine whether the treatment was able to modify the concentrations of Aβ in plasma; 2) to assess the effects of PE on cognitive, functional, and behavioral outcomes; and 3) to evaluate the safety of the treatment by monitoring adverse reactions and clinically relevant changes during and after the PE procedure.
Patients
Patients were recruited from four different sites, two in Spain (Fundació ACE – Institut Catalá de Neurociències Aplicades in Barcelona, where patients from the Hospital General Universitari Vall d’Hebron in Barcelona were also included, and the Hospital General Universitario Gregorio Marañón in Madrid) and two in the USA (Alzheimer’s Research Corporation – Mid Atlantic Geriatric Association in Manchester, NJ, and Howard University in Washington, DC). The inclusion criteria were: 1) age 55–85; 2) meet the NINCDS-ADRDA criteria for probable AD [23]; 3) Mini-Mental State Examination (MMSE) [24] scores between ≥18 and ≤26); 4) be in a stable dose of a cholinesterase inhibitors for the previous three months; and 5) absence of cerebrovascular disease by clinical history and by computed axial tomography (CAT) or magnetic resonance imaging (MRI).
The exclusion criteria were: 1) Any contraindication or difficulty for PE (e.g., difficult venous access, behavioral disorders, a history of frequent adverse reactions to blood products; hypersensitivity to albumin or allergies to any of the components of human albumin 5%); 2) abnormal coagulation or laboratory parameters (e.g., hypocalcemia Ca++ <8.7 mg/dL; thrombocytopenia <100,000/μL; fibrinogen <1.5 g/L; prothrombin time [Quick] p < 60% versus control); 3) angiotensin-converting enzyme inhibitors or beta-blocker treatment; 4) bradycardia <60/min; 5) plasma creatinine >2 mg/dL; alanine aminotransferase >2.5×upper limit of normal; bilirubin >2 mg/dL; 6) uncontrolled high blood pressure; 7) heart disease; 8) participation in other clinical trials or the use of any other investigational drug in the three months prior to the start of the study; 9) fewer than six years of education; and 10) any condition that complicated adherence to the studyprotocol.
The patient and a close relative or legal representative signed the informed consent form to participate in the trial.
Study design
The time between screening and randomization (1:1 ratio to either the control group or the PE-treated group) was two weeks. The PE treatment consisted of processing approximately one whole plasma volume with simultaneous substitution by the same volume of 5% human albumin, a procedure specifically known as Total Plasma Exchange. The PE treatment was provided in three periods of six PE each: 1) Intensive treatment period: three weeks with two PE per week; 2) Maintenance treatment period I: six weeks with one PE per week; and 3) Maintenance treatment period II: 12 weeks with one PE every two weeks. The control group underwent a sham treatment (simulated PE without invasive procedures).
The patients were followed during the treatment phase of the study (first 21 weeks), and during an additional six-month period, where they had two visits every three months (weeks 33 and 44±7 days). Patients’ physical and clinical data were collected throughout the study (baseline and visits at weeks 3, 9, 21, 33, and 44). All evaluators of laboratory and psychometric testing were blinded to patient treatment.
Randomization and masking
Eligible patients were randomly assigned (1:1) to PE treatment or control (simulated plasma exchange without invasive procedures: sham) groups using a SAS computer-generated sequence that was programmed by an independent, unmasked, statistical team at a contract research organization (Trial Form Support – TFS). The randomization codes were kept in envelopes that were opened each time a patient was randomized. In sham treatment, gauze dressing was placed on the subclavicular region, affixing a dual lumen catheter of characteristics similar to the central catheters used in the treatment group, and procedures mimicking PE were carried out [22]. Likewise, these patients undertook the same visits schedule and assessments as the patients in the treatmentgroup.
All evaluators of laboratory and psychometric testing were blinded to patient treatment. Importantly, the evaluators of the neuropsychological tests did not have access to any kind of information allowing them to identify the patient’s assignment to treatment. The patient care takers or informers were instructed not to mention any details about the treatment during the neuropsychological tests.
Product and study procedures
The human albumin product for PE was Human Albumin Grifols®/Albutein® 5% (Grifols, Barcelona, Spain). Approximately one plasma volume was processed in each PE, calculated from body weight, height, and hematocrit (approximately 35–45 mL/kg, corresponding to a volume of 2500–3000 mL for a 70 kg. subject). The same volume of Albutein® 5% was administered (60–100 ml/min) as a replacement fluid to the treatment group (50 g of albumin per liter of replaced plasma). This is considered a standard PE procedure and is the same that was used in the preliminary phase I studies [22].
For each treatment period (intensive, maintenance I, maintenance II), a baseline lumbar puncture (before the start of treatment) and a lumbar puncture immediately after the end of the last PE were performed to collect CSF samples. Lumbar punctures were also performed during the follow-up period (weeks 33 and 44). Plasma samples were collected before and at the end of each PE. Aβ levels were determined from the samples. Before every PE, a physical examination was carried out.
Lumbar puncture and blood sampling were carried out following the standard techniques of each center. CSF collection (discarding the first few mL of fluid) was as follows: 3 mL for general testing (kept at room temperature, with immediate analysis); and 3 mL for Aβ determination (collected in polypropylene tubes, which could be immediately frozen at –80°C until analysis). For blood collection (with EDTA), recommendations were as follows: 5 mL for coagulation parameters and hematological tests; 10 mL for proteinogram, biochemistry, and serology; 4 mL (approximately 2 mL of plasma) for ApoE determination; and 10 mL (approximately 5 mL of plasma) in polypropylene tubes for Aβdetermination.
Efficacy variables
Efficacy outcomes included the variation of Aβ1–40 and Aβ1–42 levels in CSF during the treatment period, between baseline and at the end of the last PE, as well as the variation between the beginning and finalization of each of the three treatment periods. Based on the observations of the preliminary phase I studies [22], the difference of CSF Aβ1–42 levels between the treatment group and the control group at the end of the last PE considered to be feasible was 100 pg/ml.
CSF and plasma levels of Aβ1–40 were determined with the h Amyloid Aβ1–40 ELISA (The Genetics Company; TGC). CSF and plasma levels of Aβ1–42 were determined with the high-sensitivity Innotest β-amyloid (1 –42) (Innogenetics, now Fujirebio Diagnostic Inc.) with a determination cutoff limit of 7.8 pg/mL.
Clinical efficacy measures included global cognitive measures such as the MMSE [24], and the Alzheimer’s Disease Assessment Scale, cognitive subscale (ADAS-Cog) [25], and selected language/attention neuropsychological tests: 1) Symbol Digit Modalities Test (SDMT) [26], 2) Semantic Verbal Fluency (SVF) [27], 3) Phonemic Verbal Fluency (PVF) [27], 4) Boston Naming Test (BNT) [28], and 5) Rey Auditory Verbal Learning Test (RAVLT) [29]. Behavioral measures included: 1) the Neuropsychiatric Inventory (NPI) [30], 2) the Cornell Scale for Depression in Dementia (CSDD) [31], 3) the Overt Aggression Scale (OAS) [32], and 4) the Agitated Behavior Scale (ABS) [33]. Functional measures included: 1) the Alzheimer’s Disease Cooperative Study-Activities of Daily Living (ADCS-ADL) [34], 2) the Alzheimer’s Disease Cooperative Study – Clinical Dementia Rating – Sum of boxes (CDR-sb) [35], and 3) the Clinical Global Impression of Change (ADCS-CGIC) [36].
Safety assessments
The primary assessment of safety was the number and percentage of patients with at least one adverse effect (AE) that could be related or not to the study procedure. The AE were coded according to the classification of the World Health Organization (WHO) (MedDRA version 13.1), and were described by a synonym (Lowest Level Term) and the affected organ/system, the intensity, causality and seriousness.
Statistical analysis
A sample size of 36 patients (18 per group) was calculated for the study to have 80% power for the primary efficacy variable (variation in Aβ1–42 levels in CSF from the baseline visit to the last available measurement), a difference of 220 pg Aβ1–42/mL between means of the treatment and control groups, assuming the common standard deviation (SD) to be 217.4 pg/mL (according to data obtained in the preliminary phase 1 studies) [22], using the Student t-test with a two-sided level of significance of 5%. Assuming a global dropout rate of approximately 15%, the study had to enroll 42 patients to obtain 36 patients for evaluation.
All patients with at least three PE sessions during the intensive treatment period were considered for efficacy analysis (intention-to-treat [ITT] population). A second analysis was also conducted on the per-protocol (PP) population, which included the patients who completed the treatment without breaches in the study protocol, determined before the randomization was unblinded. All the patients included in the study and subjected to at least one PE session were included in the safetypopulation.
The analysis of the variation in Aβ1–40 and Aβ1–42 levels was carried out by an analysis of covariance (ANCOVA) with adjusted (least-squares) mean and 95% confidence intervals (CI) as output and the change from baseline in Aβ in CSF at the last available measurement as dependent variable, treatment group as a factor, and the baseline level of Aβ as a covariate. The following variables were descriptively analyzed by treatment group: variation in Aβ1–40 and Aβ1–42 levels in CSF between the end and start of each of the three treatment periods; variation in Aβ1–40 and Aβ1–42 levels in CSF between the treatment phase and follow up; and variation of Aβ1–40 and Aβ1–42 levels in plasma before and after each PE and follow up.
The clinical measures were examined with a mixed model for repeated measures between-group differences in test scores at end of PE treatment (week 21) and end of follow up (week 44). The dependent variable in each analysis was the change from baseline in test score. Fixed effects were treatment (PE-treated or control group), visit, and treatment-by-visit interaction. No adjustments for multiple comparisons or corrections for multiplicity were planned.
Data are presented as median and range or as mean±standard deviation (SD) or±95% CI, whenever appropriate. Statistical significance was set at 5% level. The SAS® software (SAS Institute Inc., Cary, NC, USA) version 9.2 was used for calculations.
RESULTS
Patients characteristics
A total of 48 patients that met the inclusion criteria were enrolled and signed the informed consent, with no screening failures. Forty-two of them were randomized, 21 to the PE-treated group and 21 to the control group with sham PE treatment (See Fig. 1). Thirty-nine patients provided baseline and longitudinal data (evaluable population), 19 patients in the treatment group and 20 patients in the control group. Twenty-nine patients completed the study and 13 dropped out: six because AEs (2 anxiety, 2 injuries due to fall, 1 hospitalization due to otitis media, and 1 death), five withdrew consent (by patient or caregiver), one protocol violation (taking angiotensin-converting enzyme inhibitor), and one lost to follow-up.
Baseline demographic, anthropometric, and clinical characteristics of patients are shown in Tables 1 and 2. No significant differences between the characteristics of the two study groups were observed. All patients received a maximum of 18 PE, with a median exposure to treatment of 155 days (range: 1–166). All subjects included in the study and subjected to at least three PE sessions during the intensive treatment period were considered for efficacy analysis (ITT population; n = 37) while the PP population was n = 35. All the 39 evaluable patients were included in the safety analysis.
Aβ levels in CSF
Variation of Aβ1–42 levels in CSF between the end of the last PE and baseline (primary efficacy variable) showed a higher adjusted (least-squares) mean for the PE-treated group compared to the control group, approaching significance (p = 0.072), with values of 75.3 pg/mL (95% CI: –19.8, 170.5) for the PE-treated group versus –45.5 pg/mL (95% CI: –135.1, 44.2) for the control group. There were not significant differences between the two groups of patients in the variation of Aβ1–40 levels in CSF between the end of the last PE and the baseline, with adjusted mean values of 135.9 pg/mL (95% CI: –954.0, 1225.8) in the PE-treated group versus 441.5 pg/mL (95% CI: –585.6, 1468.6) in the control group.
Results of the levels of Aβ1–40 and Aβ1–42 in CSF during the course of the treatment phase and follow-up are shown in Fig. 2. Aβ1–42 values of PE-treated patients were consistently higher than control group (Fig. 2B). At baseline, Aβ1–42 mean levels in the PE-treated group and the controls were, respectively, 333.2±151.8 pg/mL and 304.1±121.3 pg/mL; while at the end of last PE the levels were, respectively, 398.0±273.8 pg/mL and 256.7±100.5 pg/mL. In all the CSF Aβ measurements, results were similar when performed on the PP population (see Supplementary Tables 1 and 2).
Aβ levels in plasma
Plasma levels of Aβ1–40 and Aβ1–42 measured before and after each PE showed marked differences between groups (Fig. 3). In PE-treated patients the Aβ1–40 levels showed a saw-tooth pattern that approximately ranged from 100 to 300 pg/mL, while the Aβ1–42 levels ranged from 20 to 60 pg/mL. The plasma Aβ1–42 levels were statistically lower after each treatment period in the PE-treated group compared to the control group (See Fig. 3B). After the treatment periods, plasma Aβ1–40 and Aβ1–42 levels tended to return to baseline levels during the observational phase of the study. Results of all plasma Aβ measurements were similar when performed on the PP population (available as Supplementary Tables 3 and 4). The relationship between intensity of the treatment and plasma Aβ1–40 mobilization is also shown as Supplementary Figure 1.
Clinical efficacy outcomes
Table 3 summarizes the changes in the test scores associated with treatment (treatment effect or treatment-by-visit effect) after the treatment period and after the follow-up compared to baseline, in both patient groups.
PE-treated patients exhibited better scores than those in the control group in measures of global cognition (MMSE and ADAS-Cog) during the intervention and observational phases of the study (see Table 3 and Fig. 4), with a trend to significance (abstractly considered when p value was from ≥0.05 to <0.12) in favor of the PE-treated group with the MMSE (mean difference at week 44 : 2.6 [95% CI –0.8, 6.1]; p = 0.081), and with the ADAS-cog (mean difference at week 21: –3.6 [95% CI –8.6, 1.5]; p = 0.094).
The PE-treated group had better BNT and SVF scores throughout the study compared to the control group, and there was a statistically significant difference between groups in the treatment-by-visit effect (see Table 3 and Fig. 5). The mean differences between PE-treated and control at week 44 were 1.3 (95% CI –1.1, 3.8) for BNT (p = 0.043) and 2.5 (95% CI 0.1, 5.0) for SVF (p = 0.024).
Similarly, the PE-treated group had better RAVLT1 and RAVLT2 scores than the control group, and there was a trend to statistical significance favoring the PE-treated group throughout the study (see Table 3). The mean differences between PE-treated and control at week 44 were 1.5 (95% CI 0.1, 2.9) for RAVLT1 (p = 0.050) and 1.3 (95% CI 0.2, 2.5) for RAVLT2 (p = 0.099).
By contrast, the control group scored better than the PE-treated group in behavioral (NPI) and functional (ADCS-ADL) measures (see Table 3 and Fig. 6). The effect was more marked at the end of the treatment period (week 21), with mean differences between PE-treated and control of 9.4 (95% CI –0.2, 18.5) for NPI and –7.1 (95% CI –14.2, 0.1) for ADCS-ADL. In NPI, statistically significant differences were observed during both the treatment period (p = 0.028) and the follow-up (p = 0.048), while in ADCS-ADL the statistical significance was a trend (p = 0.050 and p = 0.119, respectively).
Safety results
The rate of AEs was similar between the two treatment groups (see Tables 5 and 6). Out of the 39 evaluable patients, 14 (70.0%) in the control group underwent at least one adverse event throughout the study (seven of them moderate to severe) whereas there were 18 (94.7%) in the PE-treated patients (nine of them moderate to severe). Five patients (12.8%) had serious AEs: two of them in the control group: cholangitis and otitis media; and three patients in the PE-treated group: hemorrhage associated with the catheter insertion, complex partial seizures (unrelated to treatment according to the investigator), and loss of consciousness (unrelated to treatment according to the investigator; the patient was admitted to the ICU and was subsequently diagnosed of a myocardial infarction, resulting in a multisystem organ failure and death 2 days later; this patient was submitted to a PE 2 days prior to the AE).
In the PE-treated group, 18 patients (97.7%) experienced any AE, seven patients (36.8%) experienced AEs related to the study intervention, and 15 patients (78.9%) experienced AEs related to study procedures (adverse reactions), in contrast to 14 (70.0%), one (5.0%) and five (25.0%) patients, respectively, in the control group (p = 0.044, p = 0.014, and p = 0.002, respectively; Table 4).
The most frequent AE were infections (in 14 patients, mostly device-related); psychiatric disorders (in 14 patients, mostly anxiety); blood and lymphatic system disorders (in 10 patients, mostly anemia); nervous system disorders (in six patients,mostly convulsion/seizure); general and medical device complications (in five patients, mostly fatigue); and injury and procedural complications (in five patients, mostly fall). Detailed results for PE-treated and control group patients are shown in Table 5.
Five (12.8%) out of the 39 evaluable patients included in the study reported abnormal laboratory values. Three of them (7.7%) had low hematocrit and hemoglobin levels. One patient had high GOT, GPT, and LDH levels, and one patient had abnormal IgG levels.
DISCUSSION
This study showed that PE treatment with fresh therapeutic albumin decreased Aβ1–42 levels in plasma, and led to borderline increased Aβ1–42 levels in CSF. This effect was associated with a trend to improvement in patients’ global cognitive measures (MMSE, ADAS-Cog) during PE treatment and post PE observational period, as well as with a sustained improvement in memory and language measures. By contrast, treated patients had worse scores in behavioral and functional measures during the PE phase, which improved in the post-PE phase.
The variation in Aβ1–42 levels in CSF between the end of the last PE and the baseline levels was the primary efficacy variable. Although the treated group had higher Aβ1–42 values than the control group through the course of the treatment phase (Fig. 2), the difference in the change at baseline versus the last PE between the two groups of patients was only close to the level of significance, a trend of increase that was also observed in the preliminary phase I studies [22]. This was even with the difference of mean Aβ1–42 levels at the end of last PE between both patient groups (around 142 pg/mL) being above the 100 pg/mL that was considered feasible. By contrast, CSF levels of Aβ1–40 did not change with PE while in plasma showed a clear saw-tooth pattern associated with PE periods in the treatment group which was not present in the control group, as it was also seen in the preliminary phase I studies [22]. This dissociation between Aβ1–42 and Aβ1–40 has been found in other studies and suggests that Aβ1–40 has a faster clearance compared to Aβ1–42 [37, 38].
Our findings are consistent with a greater effect of PE in plasma than in the CSF, which is reasonable since nearly all cells in the organism generate Aβ species. While it is thought that the Aβ in CNS originates in the CNS, the Aβ in plasma is the product of the AβPP metabolism in multiple organs, erythrocytes, and platelets [39]. Therefore, the Aβ in plasma partially reflects its metabolism in the CNS. Although the mechanisms involved in the passage of Aβ from CSF to plasma are not well understood, there are a number of receptors that are implicated (e.g., low-density lipoprotein receptor) [40], and this process seems to be modulated by APOE [41]. Findings of this study are in line with our scientific hypothesis: sequestration of plasma Aβ through PE [39] may alter the CNS/plasma equilibrium. That is, the decreased Aβ1–42 in plasma would lead to an accelerated efflux from CNS to compensate the peripheral drop, and consequently more Aβ1–42 would be released in the CNS. Whether the increased CSF Aβ1–42 is a product of the normal AβPP metabolism, the release from the SNP or both is difficult to determine.
One of the most important, and intriguing, aspects of this trial is that there was an improvement in cognitive measures during the PE treatment phase, which persisted after the treatment was discontinued. Although bordering statistical significance in some cases, the results are clinically relevant. The change from baseline scores in the MMSE and ADAS-Cog of the PE-treated group were at all time points better than those of the control group, as well as most scores in memory and language functions. A nearly identical profile was observed in the treated patients of the preliminary phase I studies [22]. This may indicate that the removal of Aβ, and perhaps other unknown proteins, by PE initiates a more durable process or processes in the CNS that have beneficial effects in cognition. There is an ongoing clinical study known as the ‘Alzheimer Management by Albumin Replacement’ (AMBAR) whose primary objective is to evaluate cognitive and functional changes in AD patients treated with PE with albumin and immunoglobulin [42].
Conversely, patients in the control group had better behavioral and functional symptoms than those in the PE group during the treatment phase. However, the statistical differences subsided during the observational phase. A possible explanation for this is that PE has a negative impact on activities of daily living during the intensive treatment phases but returns to baseline levels once the treatment is completed. Similarly, patients in the control group had better NPI scores than the PE treated group, although at the end of the observational phase the treated group had greater improvement compared to the control group. This indicates that PE can trigger psychiatric symptoms in AD patients, either related to the fact that the patients had to live with a catheter inserted in the chest, had discomfort caused by metabolic alterations related to PE, or both. Indeed, as shown in Table 5, 50% of the patients in the treated group developed psychiatric symptoms, especially anxiety.
The most frequent AE were infections (28.6% in controls versus 55.6% in PE-treated patients) and psychiatric symptoms (35.7% versus 50.0%). Although there were no statistically significant differences between groups in terms of serious AEs, the incidence of AEs related to the study product or study procedures was higher in PE-treated patients than in the controls with sham PE. This is what could be expected in patients treated with PE [43–45]. PE is safely used in some neurologic diseases [46] as well as in chronic conditions such as familial hypercholesterolemia [47]. In this study, PE showed in general a comparable safety and tolerability profile. The only fatal AE (myocardial infarction) was considered not related to the study product, but could be unlikely related to the study procedures as a worst-case scenario. It is possible that, in order to optimize the PE treatment for AD, meaning to achieve maximal Aβ mobilization with the minimal PE procedures to reduce AEs, the schedule of the maintenance period can be reduced.
Possible study limitations would include characteristics of the study product and/or procedures. Feasibility of albumin replacement treatment previously studied in vitro showed that Albutein®, the therapeutic albumin used in this study, was Aβ-free capable of binding peptides containing the primary sequence of human Aβ [48] and may also play a role in oxidation stress associated with AD [49]. However, PE removes albumin, Aβ, and many other active plasma components. Consequently, the possibility that one or more unidentified agents could play a role in the observed effects of PE on the AD patients cannot be ruled out. Indeed, studies conducted in animal models suggest a role of still-unknown plasma components in the reversal of age-related cognitive impairment [50]. On the other hand, the study population was recruited by excluding patients with any condition that could potentially interfere with PE. This could be a source of bias. In spite of the relatively small sample size, this study opens a new avenue for the treatment of AD, and expands our understanding of Aβ metabolism.
In conclusion, after PE treatment with 5% human albumin of AD patients, a measurable modification in Aβ1–42 concentration in CSF and plasma was observed, with different patterns of mobilization. PE treatment was associated with improvement in memory and language functions compared to control group patients (sham PE), who showed the cognitive decline expected in AD.
ACKNOWLEDGMENTS
This study was funded by Grifols. Jordi Bozzo PhD CMPP (Grifols) is acknowledged for medical writing and editorial assistance in the preparation of this manuscript.
Authors’ disclosures available online (http://j-alz.com/manuscript-disclosures/16-0565r1).
Appendices
The supplementary material is available in the electronic version of this article: http://dx.doi.org/10.3233/JAD-160565.
REFERENCES
[1] | Fitzpatrick AL , Kuller LH , Ives DG , Lopez OL , Jagust W , Breitner JC , Jones B , Lyketsos C , Dulberg C ((2004) ) Incidence and prevalence of dementia in the Cardiovascular Health Study. J Am Geriatr Soc 52: , 195–204. |
[2] | Becker JT , Boller F , Lopez OL , Saxton J , McGonigle KL ((1994) ) The natural history of Alzheimer’s disease. Description of study cohort and accuracy of diagnosis. Arch Neurol 51: , 585–594. |
[3] | Hyman BT , Phelps CH , Beach TG , Bigio EH , Cairns NJ , Carrillo MC , Dickson DW , Duyckaerts C , Frosch MP , Masliah E , Mirra SS , Nelson PT , Schneider JA , Thal DR , Thies B , Trojanowski JQ , Vinters HV , Montine TJ ((2012) ) National Institute on Aging-Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease. Alzheimers Dement 8: , 1–13. |
[4] | Salloway S , Sperling R , Fox NC , Blennow K , Klunk W , Raskind M , Sabbagh M , Honig LS , Porsteinsson AP , Ferris S , Reichert M , Ketter N , Nejadnik B , Guenzler V , Miloslavsky M , Wang D , Lu Y , Lull J , Tudor IC , Liu E , Grundman M , Yuen E , Black R , Brashear HR ((2014) ) Two phase 3 trials of bapineuzumab in mild-to-moderate Alzheimer’s disease. N Engl J Med 370: , 322–333. |
[5] | Doody RS , Thomas RG , Farlow M , Iwatsubo T , Vellas B , Joffe S , Kieburtz K , Raman R , Sun X , Aisen PS , Siemers E , Liu-Seifert H , Mohs R ((2014) ) Phase 3 trials of solanezumab for mild-to-moderate Alzheimer’s disease. N Engl J Med 370: , 311–321. |
[6] | Patterson BW , Elbert DL , Mawuenyega KG , Kasten T , Ovod V , Ma S , Xiong C , Chott R , Yarasheski K , Sigurdson W , Zhang L , Goate A , Benzinger T , Morris JC , Holtzman D , Bateman RJ ((2015) ) Age and amyloid effects on human central nervous system amyloid-beta kinetics. Ann Neurol 78: , 439–453. |
[7] | Iwatsubo T , Odaka A , Suzuki N , Mizusawa H , Nukina N , Ihara Y ((1994) ) Visualization of A beta 42(43) and A beta 40 in senile plaques with end-specific A beta monoclonals: Evidence that an initially deposited species is A beta 42(43). Neuron 13: , 45–53. |
[8] | Galasko D , Chang L , Motter R , Clark CM , Kaye J , Knopman D , Thomas R , Kholodenko D , Schenk D , Lieberburg I , Miller B , Green R , Basherad R , Kertiles L , Boss MA , Seubert P ((1998) ) High cerebrospinal fluid tau and low amyloid beta42 levels in the clinical diagnosis of Alzheimer disease and relation to apolipoprotein E genotype. Arch Neurol 55: , 937–945. |
[9] | Mayeux R , Tang MX , Jacobs DM , Manly J , Bell K , Merchant C , Small SA , Stern Y , Wisniewski HM , Mehta PD ((1999) ) Plasma amyloid beta-peptide 1-42 and incipient Alzheimer’s disease. Ann Neurol 46: , 412–416. |
[10] | Lui JK , Laws SM , Li QX , Villemagne VL , Ames D , Brown B , Bush AI , De RK , Dromey J , Ellis KA , Faux NG , Foster J , Fowler C , Gupta V , Hudson P , Laughton K , Masters CL , Pertile K , Rembach A , Rimajova M , Rodrigues M , Rowe CC , Rumble R , Szoeke C , Taddei K , Taddei T , Trounson B , Ward V , Martins RN , AIBL Research Group ((2010) ) Plasma amyloid-beta as a biomarker in Alzheimer’s disease: The AIBL study of aging. J Alzheimers Dis 20: , 1233–1242. |
[11] | Zlokovic BV ((2004) ) Clearing amyloid through the blood-brain barrier. J Neurochem 89: , 807–811. |
[12] | Shibata M , Yamada S , Kumar SR , Calero M , Bading J , Frangione B , Holtzman DM , Miller CA , Strickland DK , Ghiso J , Zlokovic BV ((2000) ) Clearance of Alzheimer’s amyloid-ss(1-40) peptide from brain by LDL receptor-related protein-1 at the blood-brain barrier. J Clin Invest 106: , 1489–1499. |
[13] | Marques MA , Kulstad JJ , Savard CE , Green PS , Lee SP , Craft S , Watson GS , Cook DG ((2009) ) Peripheral amyloid-beta levels regulate amyloid-beta clearance from the central nervous system. J Alzheimers Dis 16: , 325–329. |
[14] | Roberts KF , Elbert DL , Kasten TP , Patterson BW , Sigurdson WC , Connors RE , Ovod V , Munsell LY , Mawuenyega KG , Miller-Thomas MM , Moran CJ , Cross DT III , Derdeyn CP , Bateman RJ ((2014) ) Amyloid-beta efflux from the central nervous system into the plasma. Ann Neurol 76: , 837–844. |
[15] | Ghersi-Egea JF , Gorevic PD , Ghiso J , Frangione B , Patlak CS , Fenstermacher JD ((1996) ) Fate of cerebrospinal fluid-borne amyloid beta-peptide: Rapid clearance into blood and appreciable accumulation by cerebral arteries. J Neurochem 67: , 880–883. |
[16] | Poduslo JF , Curran GL , Sanyal B , Selkoe DJ ((1999) ) Receptor-mediated transport of human amyloid beta-protein 1-40 and 1-42 at the blood-brain barrier. Neurobiol Dis 6: , 190–199. |
[17] | Tapiola T , Alafuzoff I , Herukka SK , Parkkinen L , Hartikainen P , Soininen H , Pirttila T ((2009) ) Cerebrospinal fluid beta-amyloid 42 and tau proteins as biomarkers of Alzheimer-type pathologic changes in the brain. Arch Neurol 66: , 382–389. |
[18] | DeMattos RB , Bales KR , Parsadanian M , O’Dell MA , Foss EM , Paul SM , Holtzman DM ((2002) ) Plaque-associated disruption of CSF and plasma amyloid-beta (Abeta) equilibrium in a mouse model of Alzheimer’s disease. J Neurochem 81: , 229–236. |
[19] | Matsuoka Y , Saito M , LaFrancois J , Saito M , Gaynor K , Olm V , Wang L , Casey E , Lu Y , Shiratori C , Lemere C , Duff K ((2003) ) Novel therapeutic approach for the treatment of Alzheimer’s disease by peripheral administration of agents with an affinity to beta-amyloid. J Neurosci 23: , 29–33. |
[20] | Meca-Lallana JE , Rodriguez-Hilario H , Martinez-Vidal S , Saura-Lujan I , Carreton-Ballester A , Escribano-Soriano JB , Martin-Fernandez J , Genoves-Aleixandre A , Mateo-Bosch E , Fernandez-Barreiro A ((2003) ) Plasmapheresis: Its use in multiple sclerosis and other demyelinating processes of the central nervous system. An observation study. Rev Neurol 37: , 917–926. |
[21] | Kuo YM , Kokjohn TA , Kalback W , Luehrs D , Galasko DR , Chevallier N , Koo EH , Emmerling MR , Roher AE ((2000) ) Amyloid-beta peptides interact with plasma proteins and erythrocytes: Implications for their quantitation in plasma. Biochem Biophys Res Commun 268: , 750–756. |
[22] | Boada M , Ortiz P , Anaya F , Hernandez I , Munoz J , Nunez L , Olazaran J , Roca I , Cuberas G , Tarraga L , Buendia M , Pla RP , Ferrer I , Paez A ((2009) ) Amyloid-targeted therapeutics in Alzheimer’s disease: Use of human albumin in plasma exchange as a novel approach for Abeta mobilization. Drug News Perspect 22: , 325–339. |
[23] | McKhann G , Drachman D , Folstein M , Katzman R , Price D , Stadlan EM ((1984) ) Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer’s Disease. Neurology 34: , 939–944. |
[24] | Folstein MF , Folstein SE , McHugh PR ((1975) ) “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12: , 189–198. |
[25] | Rosen WG , Mohs RC , Davis KL ((1984) ) A new rating scale for Alzheimer’s disease. Am J Psychiatry 141: , 1356–1364. |
[26] | Smith A ((1973) ) Symbol Digits Modality Test, Western Psychological Services, Los Angeles. |
[27] | Benton A , Hamsher K ((1983) ) Multilingual aphasia examination, ASA Associates, Iowa City. |
[28] | Kaplan E , Goodglass H , Weintraub S ((1983) ) The Boston naming test, Lea & Febiger, Philadelphia. |
[29] | Rey A ((1964) ) L’examen clinique en psychologie, Presses Universitaires de France, Paris. |
[30] | Cummings JL , Mega M , Gray K , Rosenberg-Thompson S , Carusi DA , Gornbein J ((1994) ) The Neuropsychiatric Inventory: Comprehensive assessment of psychopathology in dementia. Neurology 44: , 2308–2314. |
[31] | Alexopoulos GS , Abrams RC , Young RC , Shamoian CA ((1988) ) Cornell Scale for Depression in Dementia. Biol Psychiatry 23: , 271–284. |
[32] | Yudofsky SC , Silver JM , Jackson W , Endicott J , Williams D ((1986) ) The Overt Aggression Scale for the objective rating of verbal and physical aggression. Am J Psychiatry 143: , 35–39. |
[33] | Corrigan JD ((1989) ) Development of a scale for assessment of agitation following traumatic brain injury. J Clin Exp Neuropsychol 11: , 261–277. |
[34] | Galasko D , Bennett D , Sano M , Ernesto C , Thomas R , Grundman M , Ferris S ((1997) ) An inventory to assess activities of daily living for clinical trials in Alzheimer’s disease. The Alzheimer’s Disease Cooperative Study. Alzheimer Dis Assoc Disord 11: (Suppl 2), S33–S39. |
[35] | Hughes CP , Berg L , Danziger WL , Coben LA , Martin RL ((1982) ) A new clinical scale for the staging of dementia. Br J Psychiatry 140: , 566–572. |
[36] | Schneider LS , Olin JT , Doody RS , Clark CM , Morris JC , Reisberg B , Schmitt FA , Grundman M , Thomas RG , Ferris SH ((1997) ) Validity and reliability of the Alzheimer’s Disease Cooperative Study-Clinical Global Impression of Change. The Alzheimer’s Disease Cooperative Study. Alzheimer Dis Assoc Disord 11: (Suppl 2), S22–S32. |
[37] | Siemers ER , Sundell KL , Carlson C , Case M , Sethuraman G , Liu-Seifert H , Dowsett SA , Pontecorvo MJ , Dean RA , Demattos R ((2015) ) Phase 3 solanezumab trials: Secondary outcomes in mild Alzheimer’s disease patients. Alzheimers Dement 12: , 110–120. |
[38] | Liu E , Schmidt ME , Margolin R , Sperling R , Koeppe R , Mason NS , Klunk WE , Mathis CA , Salloway S , Fox NC , Hill DL , Les AS , Collins P , Gregg KM , Di J , Lu Y , Tudor IC , Wyman BT , Booth K , Broome S , Yuen E , Grundman M , Brashear HR ((2015) ) Amyloid-beta 11C-PiB-PET imaging results from 2 randomized bapineuzumab phase 3 AD trials. Neurology 85: , 692–700. |
[39] | Rosenberg RN , Baskin F , Fosmire JA , Risser R , Adams P , Svetlik D , Honig LS , Cullum CM , Weiner MF ((1997) ) Altered amyloid protein processing in platelets of patients with Alzheimer disease. Arch Neurol 54: , 139–144. |
[40] | Huang Y , Potter R , Sigurdson W , Kasten T , Connors R , Morris JC , Benzinger T , Mintun M , Ashwood T , Ferm M , Budd SL , Bateman RJ ((2012) ) Beta-amyloid dynamics in human plasma. Arch Neurol 69: , 1591–1597. |
[41] | Castellano JM , Kim J , Stewart FR , Jiang H , DeMattos RB , Patterson BW , Fagan AM , Morris JC , Mawuenyega KG , Cruchaga C , Goate AM , Bales KR , Paul SM , Bateman RJ , Holtzman DM ((2011) ) Human apoE isoforms differentially regulate brain amyloid-beta peptide clearance. Sci Transl Med 3: , 89ra57. |
[42] | Boada M , Ramos-Fernández E , Guivernau B , Muñoz FJ , Costa M , Ortiz AM , Jorquera JI , Nunez L , Paez A ((2016) ) Treatment of Alzheimer’s disease using a combined therapy of plasma exchange and hemopheresis with albumin and intravenous immunoglobulin: Rationale and theraeutic approach to the AMBAR study (Alzheimer Management by Albumin Replacement). Neurología 31: , 473–481. |
[43] | Shemin D , Briggs D , Greenan M ((2007) ) Complications of therapeutic plasma exchange: A prospective study of 1,727 procedures. J Clin Apher 22: , 270–276. |
[44] | Basic-Jukic N , Kes P , Glavas-Boras S , Brunetta B , Bubic-Filipi L , Puretic Z ((2005) ) Complications of therapeutic plasma exchange: Experience with 4857 treatments. Ther Apher Dial 9: , 391–395. |
[45] | Mortzell Henriksson M , Newman E , Witt V , Derfler K , Leitner G , Eloot S , Dhondt A , Deeren D , Rock G , Ptak J , Blaha M , Lanska M , Gasova Z , Hrdlickova R , Ramlow W , Prophet H , Liumbruno G , Mori E , Griskevicius A , Audzijoniene J , Vrielink H , Rombout S , Aandahl A , Sikole A , Tomaz J , Lalic K , Mazic S , Strineholm V , Brink B , Berlin G , Dykes J , Toss F , Axelsson CG , Stegmayr B , Nilsson T , Norda R , Knutson F , Ramsauer B , Wahlstrom A ((2016) ) Adverse events in apheresis: An update of the WAA registry data. Transfus Apher Sci 54: , 2–15. |
[46] | Cortese I , Cornblath DR ((2013) ) Therapeutic plasma exchange in neurology: 2012. J Clin Apher 28: , 16–19. |
[47] | Nordestgaard BG , Chapman MJ , Humphries SE , Ginsberg HN , Masana L , Descamps OS , Wiklund O , Hegele RA , Raal FJ , Defesche JC , Wiegman A , Santos RD , Watts GF , Parhofer KG , Hovingh GK , Kovanen PT , Boileau C , Averna M , Boren J , Bruckert E , Catapano AL , Kuivenhoven JA , Pajukanta P , Ray K , Stalenhoef AF , Stroes E , Taskinen MR , Tybjaerg-Hansen A ((2013) ) Familial hypercholesterolaemia is underdiagnosed and undertreated in the general population: Guidance for clinicians to prevent coronary heart disease: Consensus statement of the European Atherosclerosis Society. Eur Heart J 34: , 3478–390a. |
[48] | Costa M , Ortiz AM , Jorquera JI ((2012) ) Therapeutic albumin binding to remove amyloid-beta. J Alzheimers Dis 29: , 159–170. |
[49] | Costa M , Horrillo R , Ortiz AM , Pérez A , Herrero P , Canela N , Boada M , Ruiz A , Hernández I , Afonso N , Torres M , Grancha S , Jorquera JI ((2015) ) Characterization of albumin oxidation status in Alzheimer’s disease patients. J Prev Alzheimers Dis 2: , 370–371. |
[50] | Villeda SA , Plambeck KE , Middeldorp J , Castellano JM , Mosher KI , Luo J , Smith LK , Bieri G , Lin K , Berdnik D , Wabl R , Udeochu J , Wheatley EG , Zou B , Simmons DA , Xie XS , Longo FM , Wyss-Coray T ((2014) ) Young blood reverses age-related impairments in cognitive function and synaptic plasticity in mice. Nat Med 20: , 659–663. |
Figures and Tables
Fig.1
Flow of patients through the study.
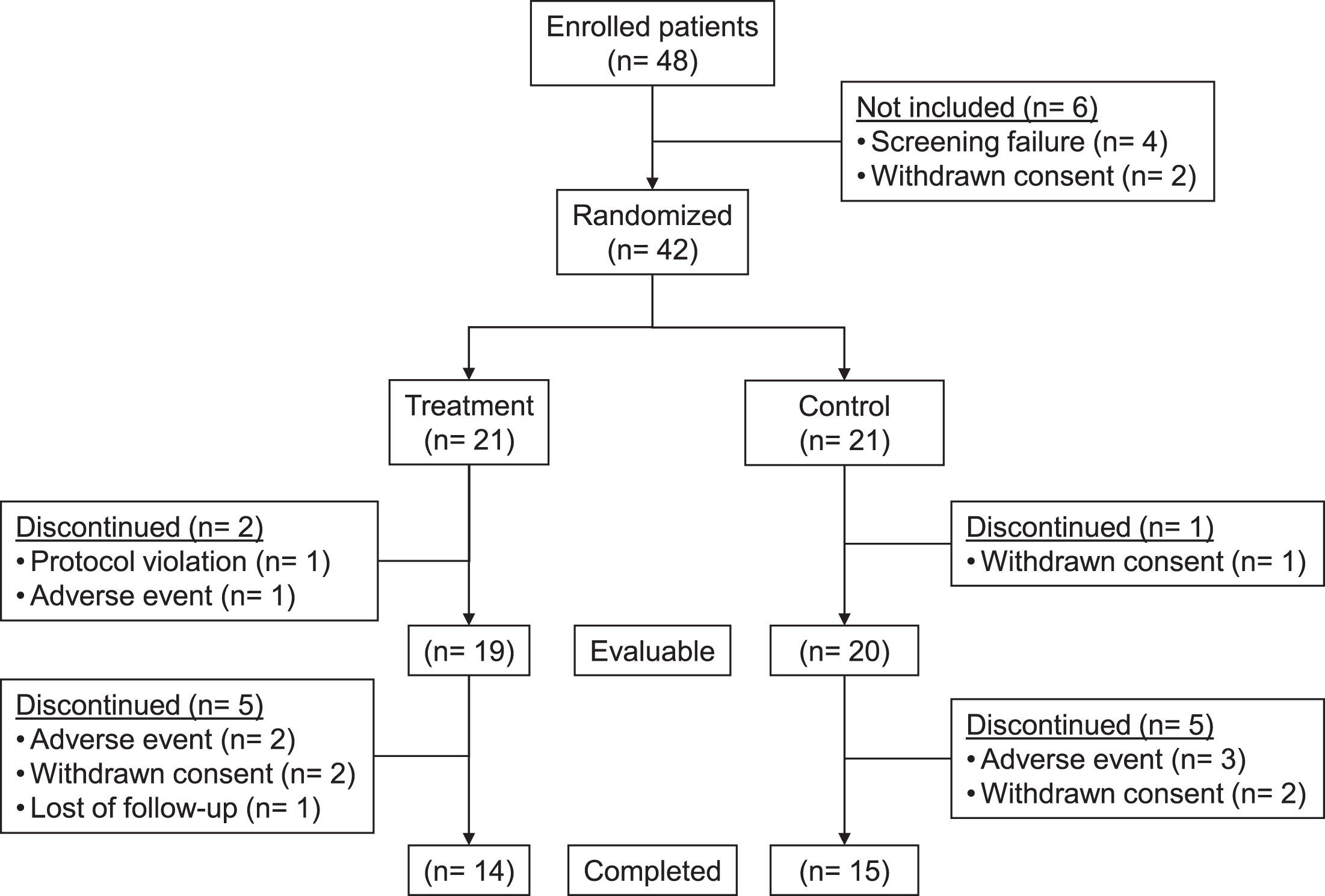
Fig.2
Levels of Aβ1–40 (A) and Aβ1–42 (B) in cerebrospinal fluid of plasma exchange (PE)-treated patients and control (sham PE) during the treatment phase and follow-up periods (Mean±SD; N = 14–19; intention-to-treat population). The p value refers to variation of adjusted (least-squares) mean of Aβ1–42 levels between the end of the last PE and baseline in the PE-treated group.
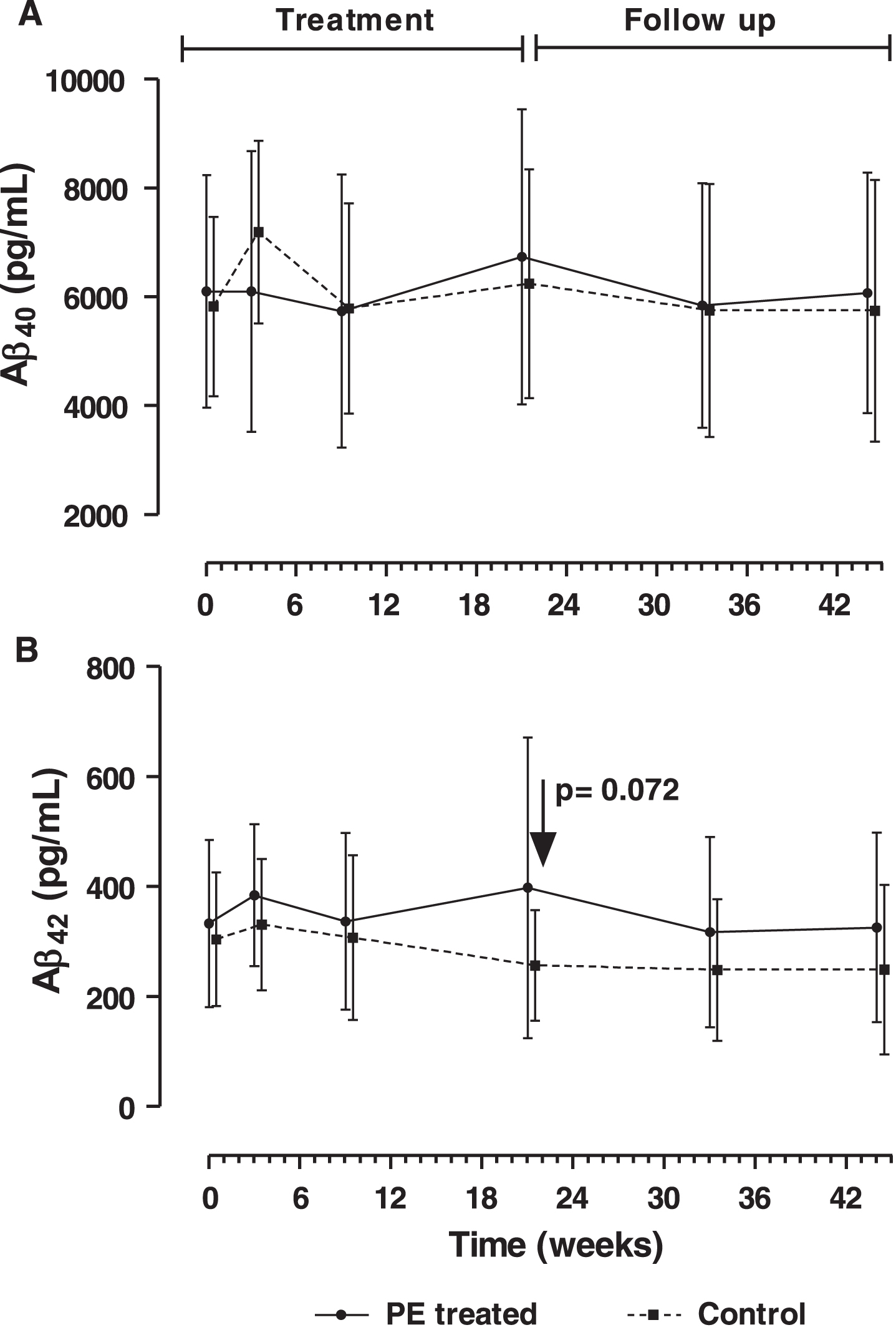
Fig.3
Levels of Aβ1–40 (A) and Aβ1–42 (B) in plasma of plasma exchange (PE)-treated patients and controls (sham PE) before and after each PE, during the three treatment periods (intensive, maintenance I and maintenance II) and follow-up (means without dispersion; N = 18–19; intention-to-treat population).
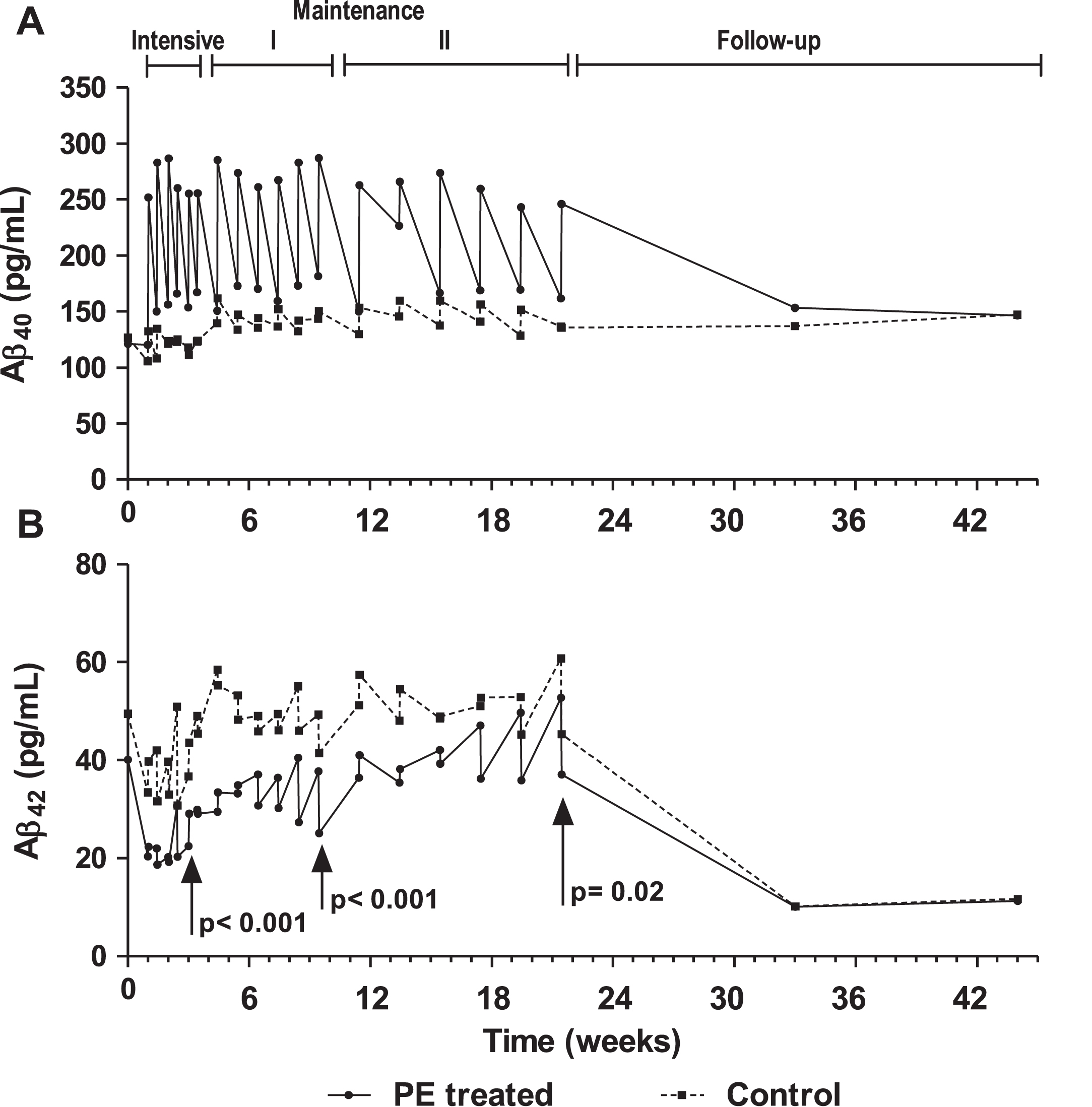
Fig.4
Score change from baseline in Mini-Mental Status Examination (MMSE) (A) and Alzheimer’s Disease Assessment Scale –Cognitive subscale (ADAS-Cog) (B) tests measured in plasma exchange (PE)-treated patients and controls (sham PE) (mean±95% CI; N = 18–19; intention-to-treat population). P-values refer to treatment effect.
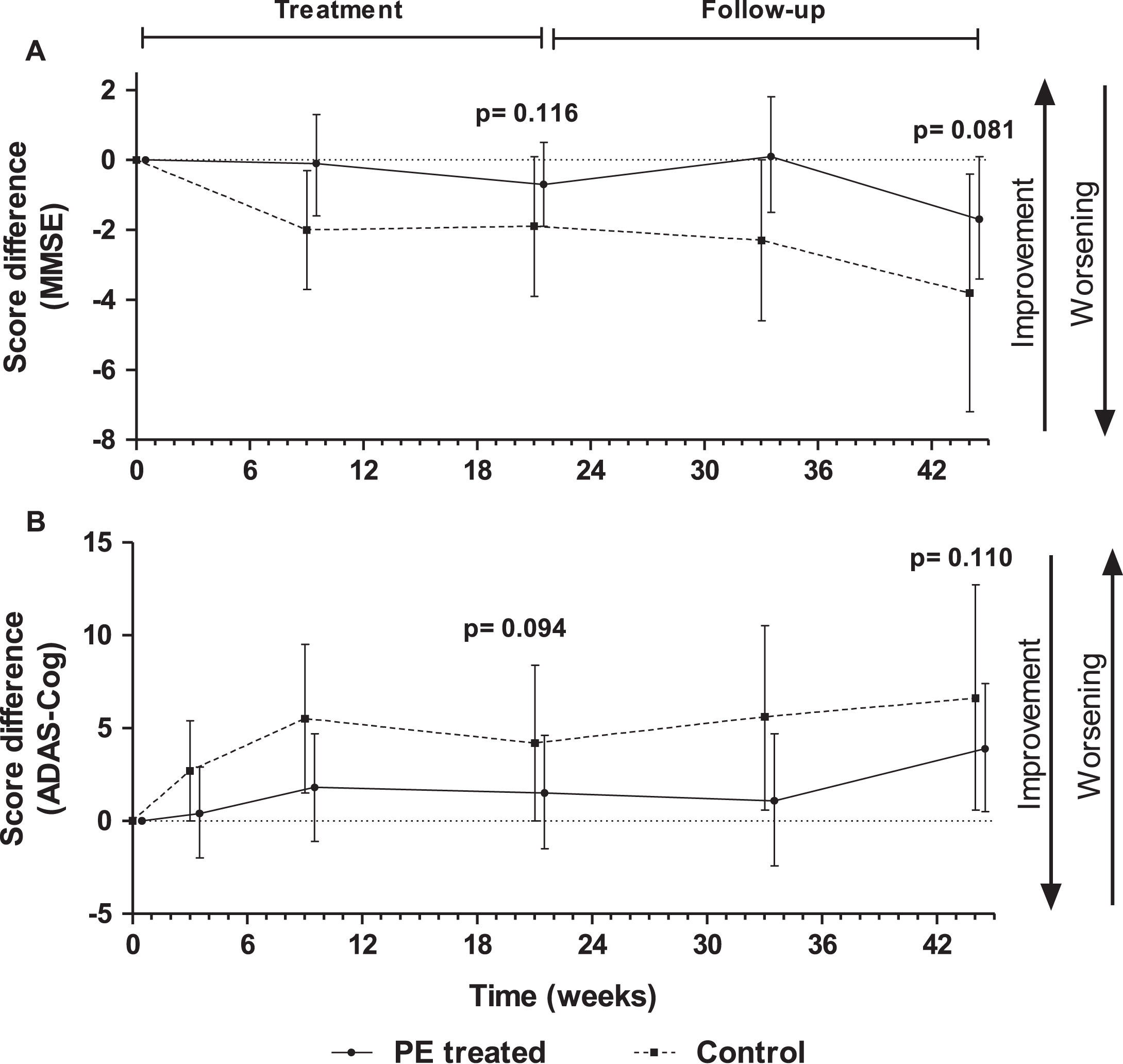
Fig.5
Score differences with respect to baseline in Boston Naming Test (BNT) (A) and Semantic Verbal Fluency (SVF) (B) tests measured in plasma exchange (PE)-treated patients and controls (sham PE) (mean±95% CI; N = 18–19; intention-to-treat population). P-values refer to treatment-by-visit effect.
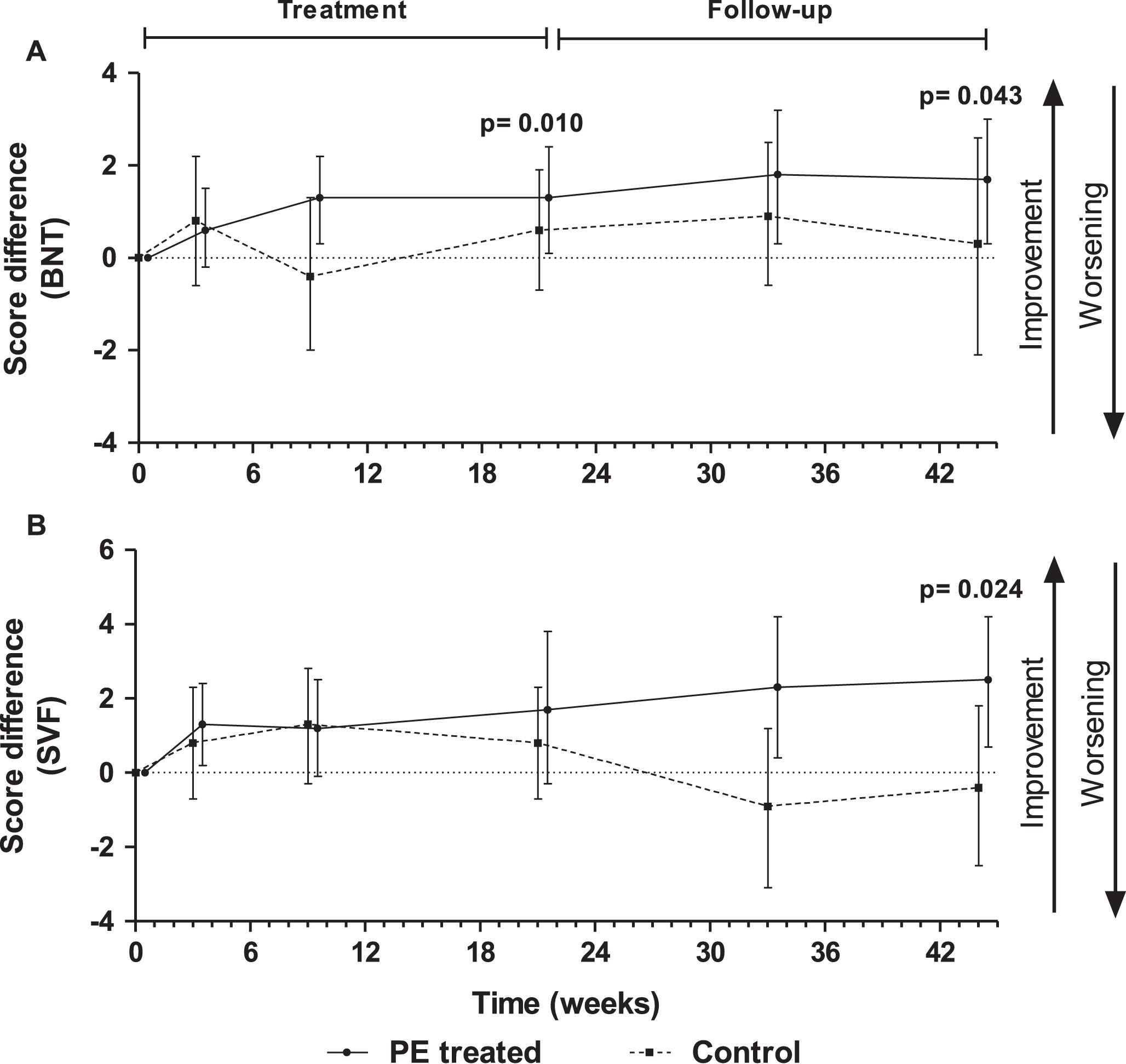
Fig.6
Score differences with respect to baseline in Neuropsychiatric Inventory (NPI) (A) and Alzheimer’s Disease Cooperative Study – Activities of Daily Living (ADCS-ADL) (B) tests measured in plasma exchange (PE)-treated patients and controls (sham PE) (mean±95% CI; N = 18–19; intention-to-treat population). P-values refer to treatment-by-visit effect in NPI and to treatment effect in ADCS-ADL.
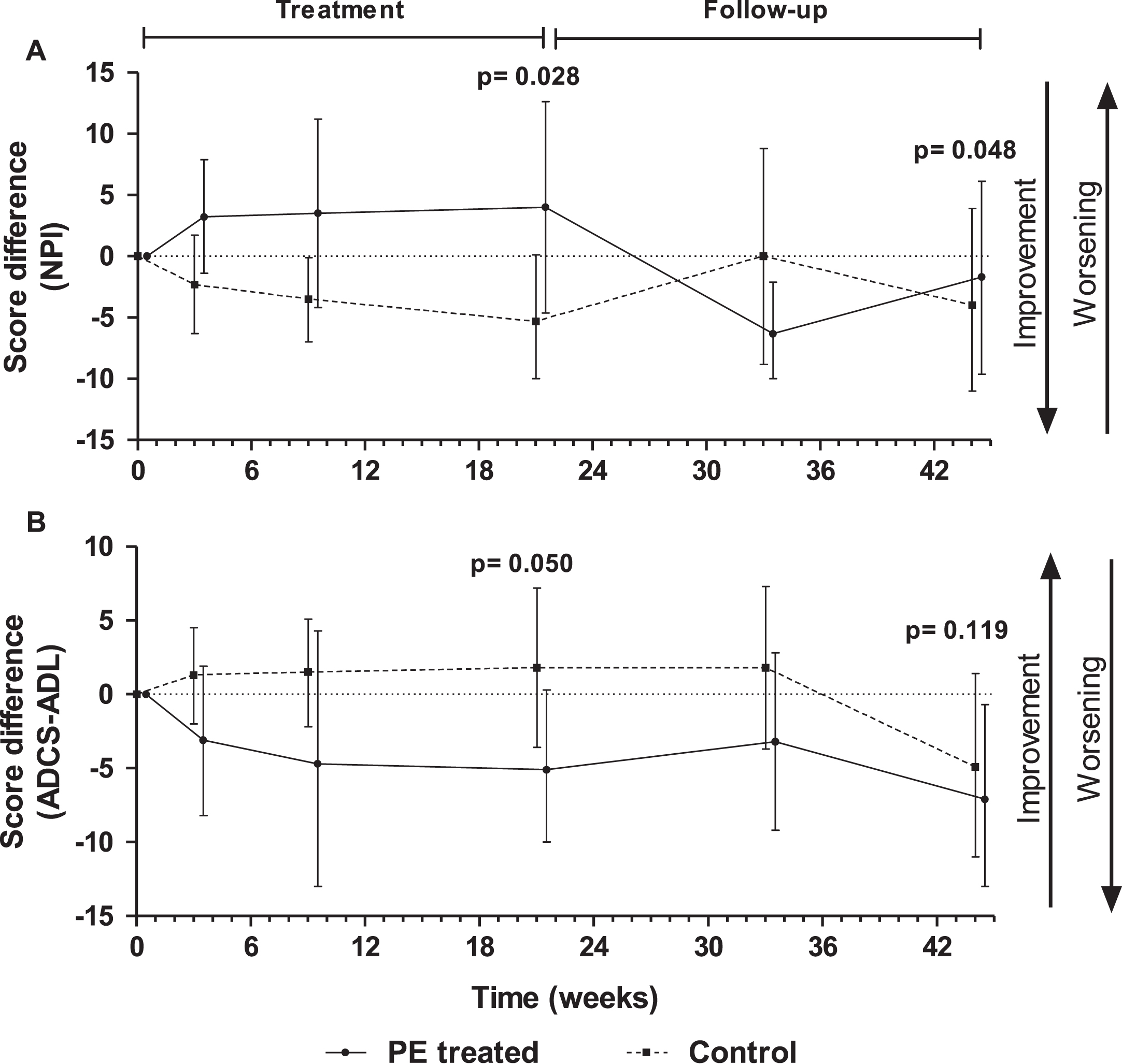
Table 1
Demographic, anthropometric and baseline neuropsychological characteristics of patients in the plasma exchange (PE)-treated and control (sham PE) groups (mean±SD or number of subjects and percentage)
| All patients | PE-treated | Control | |
| (N = 39) | (N = 19) | (N = 20) | |
| Age (years old) | 67.7±7.9 | 68.2±6.7 | 67.3±9.1 |
| Sex (females) | 30 (76.9%) | 15 (78.9%) | 15 (75.0%) |
| Height (cm) | 159.5±10.3 | 159.7±11.9 | 159.4±9.1 |
| Weight (kg) | 65.4±15.1 | 65.4±14.1 | 65.4±16.3 |
| Duration of symptoms (years) | 1.2±0.8 | 1.0±0.6 | 1.5±0.9 |
| Global cognitive measures (score) | |||
| MMSE | 21.5±2.8 | 22.6±2.7 | 20.9±3.1 |
| ADAS-Cog | 21.7±8.6 | 18.7±5.9 | 23.1±10.5 |
| Language/attention tests (score) | |||
| BNT | 8.4±3.8 | 8.8±3.7 | 8.1±3.1 |
| SVF | 9.0±3.4 | 9.8±3.7 | 8.3±3.1 |
| RAVLT immediate 1 | 2.7±1.6 | 2.8±1.5 | 2.6±1.7 |
| RAVLT immediate 2 | 3.8±2.0 | 4.3±2.4 | 3.4±1.5 |
| RAVLT immediate 3 | 4.5±2.3 | 5.2±2.3 | 3.9±2.1 |
| RAVLT immediate 4 | 5.0±2.2 | 5.9±2.1 | 4.2±2.1 |
| RAVLT immediate 5 | 5.1±2.4 | 5.7±2.7 | 4.6±1.9 |
| RAVLT delayed | 0.8±2.0 | 1.1±2.3 | 0.6±1.8 |
| SDMT | 10.3±9.3 | 11.2±9.4 | 9.5±9.3 |
| PVF (F) | 5.9±3.6 | 6.7±3.8 | 5.1±3.3 |
| PVF (A) | 5.0±3.4 | 5.9±3.3 | 4.1±3.3 |
| PVF (S) | 6.0±4.1 | 6.5±4.1 | 5.5±4.2 |
| Behavioral/functional measures (score) | |||
| NPI | 9.8±9.2 | 11.5±9.3 | 8.2±9.1 |
| ADCS-ADL | 61.7±7.6 | 62.4±5.2 | 61.1±9.5 |
| ADCS-CGIC | 3.3±0.6 | 3.1±0.5 | 3.5±0.6 |
| CDR-sb | 5.2±2.0 | 4.5±1.7 | 5.8±2.1 |
| CSDD | 3.6±4.4 | 4.4±4.2 | 2.8±4.5 |
No significant differences between the two study groups. ADAS-Cog, Alzheimer’s Disease Assessment Scale –Cognitive subscale; ADCS-ADL, Alzheimer’s Disease Cooperative Study –Activities of Daily Living; ADCS-CGIC, Alzheimer’s Disease Cooperative Study –Clinical Global Impression of Change; BNT, Boston Naming Test; CDR-sb, Clinical Dementia Rating - Sum of boxes; CSDD, Cornell Scale for Depression in Dementia; MMSE, Mini-Mental Status Examination; NPI, Neuropsychiatric Inventory, including: PVF, Phonetic Verbal Fluency; RAVLT, Rey Auditory Verbal Learning Test; SDMT, Symbol Digit Modalities Test; SVF, Semantic Verbal Fluency.
Table 2
Clinical characteristics of patients in the plasma exchange-treated and control groups at study entry
| All patients | PE-treated | Control | |
| (N = 39) | (N = 19) | (N = 20) | |
| Surgical procedures | 33 (84.6%) | 18 (94.7%) | 15 (75.0%) |
| Metabolism and nutrition disorders | 26 (66.7%) | 7 (36.8%) | 19 (95.0%) |
| Hypercholesterolemia | 23 (59.0%) | 7 (36.8%) | 16 (80.0%) |
| Psychiatric disorders | 26 (66.7%) | 12 (63.2%) | 14 (70.0%) |
| Depression | 16 (41.0%) | 7 (36.8%) | 9 (45.0%) |
| Anxiety | 2 (5.13%) | 1 (5.3%) | 1 (5.0%) |
| Musculoskeletal and connective tissue disorders | 16 (41.0%) | 9 (47.4%) | 7 (35.0%) |
| Cerebrovascular disorders | 15 (38.5%) | 10 (52.6%) | 5 (25.0%) |
| Hypertension | 9 (23.1%) | 6 (31.6%) | 3 (15.0%) |
| Diabetes | 5 (12.8%) | 2 (10.5%) | 3 (15.0%) |
| Heart disease | 1 (2.6%) | 1 (5.3%) | 0 |
| Gastrointestinal disorders | 8 (20.5%) | 4 (21.1%) | 4 (20.0%) |
| Nervous system disorders | 8 (20.5%) | 2 (10.5%) | 6 (30.0%) |
| Administration site conditions | 6 (15.4%) | 1 (5.3%) | 5 (25.0%) |
| Renal and urinary disorders | 5 (12.8%) | 5 (26.3%) | 0 |
| Infections | 5 (12.8%) | 3 (15.8%) | 2 (10.0%) |
| Renal and urinary system disorders | 5 (12.8%) | 5 (26.3%) | 0 |
| Immune system disorders | 4 (10.3%) | 4 (21.1%) | 0 |
| Other disorders (<10% patients) | 17 (43.6%) | 9 (47.4%) | 8 (40.0%) |
No significant differences between the two study groups.
Table 3
Score change from baseline to the end of the treatment and to the end of the follow-up periods in cognition tests determined in plasma exchange (PE)-treated and control (sham PE) groups. See also Figs. 4, 5, and 6
| Test | Group scoring bettera | Change from baseline (p value) | |
| Treatment-by-visit effect | Treatment effect | ||
| Global cognition | |||
| MMSE | PE-treated | NS | ≥0.05 –<0.12b,c |
| ADAS-Cog | PE-treated | NS | ≥0.05 –<0.12b,c |
| Language/attention | |||
| BNT | PE-treated | <0.05b,c | NS |
| SVF | PE-treated | <0.05c | ≥0.05 –<0.12c |
| RAVLT 1 | PE-treated | NS | ≥0.05 –<0.12c |
| RAVLT 2 | PE-treated | NS | ≥0.05 –<0.12c |
| RAVLT 3-5, delayed | None | NS | NS |
| SDMT | None | NS | NS |
| PVF (F,A,S) | None | NS | NS |
| Behavioral/functional | |||
| NPI | Control | <0.05c | <0.05b |
| ADCS-ADL | Control | NS | ≥0.05 –<0.12b,c |
| ADCS-CGIC | Control | NS | NS |
| CDR-sb | Control | NS | NS |
| CSDD | None | NS | NS |
aIn the mean score of at least 4/5 or 3/4 of the visits after baseline; bTo week 21 (treatment period); cTo week 44 (follow-up period); NS: non-significant. ADAS-Cog, Alzheimer’s Disease Assessment Scale – Cognitive subscale; ADCS-ADL, Alzheimer’s Disease Cooperative Study – Activities of Daily Living; ADCS-CGIC, Alzheimer’s Disease Cooperative Study – Clinical Global Impression of Change; BNT, Boston Naming Test; CDR-sb, Clinical Dementia Rating – Sum of boxes; CSDD, Cornell Scale for Depression in Dementia; MMSE, Mini-Mental Status Examination; NPI, Neuropsychiatric Inventory, including: PVF, Phonetic Verbal Fluency; RAVLT, Rey Auditory Verbal Learning Test; SDMT, Symbol Digit Modalities Test; SVF, Semantic Verbal Fluency.
Table 4
Summary of adverse events in the plasma exchange (PE)-treated and control (sham PE) groups
| PE-treated (N = 19) | Control (N = 20) | |
| Any AE | 18 (94.7%)* | 14 (70.0%) |
| AE related to treatment | 7 (36.8%)* | 1 (5.0%) |
| Moderate or severe AE | 9 (47.0%) | 7 (35.0%) |
| Moderate or severe AE related to treatment | 3 (15.8%) | 0 |
| AE related to study procedures | 15 (78.9%)** | 5 (25.0%) |
| Moderate or severe AE related to study procedures | 5 (26.3%) | 1 (5.0%) |
| Serious AE | 3 (15.8%) | 2 (10.0%) |
| Serious AE related to study procedures | 1 (5.3%) | 0 |
| Deceased | 1 (5.3%) | 0 |
*p < 0.05; **p < 0.01.
Table 5
Adverse events by system organ class, preferred term in the plasma exchange-treated and control (sham PE) groups
| PE-treated (N = 18) | Control (N = 14) | |
| Infections | 10 (55.6%) | 4 (28.6%) |
| Device-related infections | 5 (27.8%) | 1 (7.1%) |
| Nasopharyngitis | 2 (11.1%) | 1 (7.1%) |
| Urinary tract infection | 1 (5.6%) | 0 |
| Psychiatric symptoms | 9 (50.0%) | 5 (35.7%) |
| Aggression | 1 (5.6%) | 1 (7.1%) |
| Agitation | 0 | 1 (7.1%) |
| Anxiety | 4 (22.2%) | 2 (14.3%) |
| Depression | 1 (5.6%) | 0 |
| Blood and lymphatic system disorders | 7 (38.9%) | 3 (21.4%) |
| Anemia | 7 (38.9%) | 2 (14.3%) |
| Thrombocytopenia | 0 | 1 (7.1%) |
| CNS disorders | 4 (22.2%) | 2 (14.3%) |
| Grand mal seizures | 0 | 1 (7.1%) |
| Partial complex seizures | 1 (5.3%) | 0 |
| Injury and procedural complications | 3 (16.7%) | 2 (14.3%) |
| Fall | 2 (11.1%) | 2 (14.3%) |
| General and medical device complications | 3 (16.7%) | 2 (14.3%) |
| Fatigue/asthenia | 2 (11.1%) | 0 |
| Site hemorrhage | 1 (5.6%) | 0 |
| Site inflammation | 0 | 1 (7.1%) |
| Gastrointestinal disorders | 2 (11.1%) | 2 (14.3%) |
| Diarrhea | 1 (5.6%) | 2 (14.3%) |
| Deceased | 1 (5.6%) | 0 |




