Hemisphere Asymmetry of Response to Pharmacologic Treatment in an Alzheimer’s Disease Mouse Model
Abstract
The aim of this study was to examine hemisphere asymmetry of response to pharmacologic treatment in an Alzheimer’s disease mouse model using cilostazol as a chemical stimulus. Eight-month-old mice were assigned to vehicle or cilostazol treatment for three months and hemispheres were analyzed using quantitative proteomics. Bioinformatics interpretation showed that following treatment, aggregation of blood platelets significantly decreased in the right hemisphere whereas neurodegeneration significantly decreased and synaptic transmission increased in the left hemisphere only. Our study provides novel evidence on cerebral laterality of pharmacologic activity, with important implications in deciphering regional pharmacodynamic effects of existing drugs thus uncovering novel hemisphere-specific therapeutic targets.
INTRODUCTION
Structural asymmetry in the human brain was first described by Broca in the 19th century [1]. Brain asymmetries have also been described in other mammals, including mice [2]. The asymmetry between cerebral hemispheres encompasses functional and neurochemical aspects [3]. Data from gene expression studies in humans demonstrated lateralization of gene transcription involved in synaptic transmission and neuronal electrophysiology [4].
Cerebral laterality has been implicated in the pathophysiology of Alzheimer’s disease [5]. However, whether therapeutic agents used for Alzheimer’s disease exert hemisphere-specific pharmacologic effects has yet to be elucidated.
Pharmacoproteomics, as a subdiscipline of proteomics, examines how a pharmacologic agent perturbs a system of proteins that map to canonical or novel biological pathways. Such a discourse provides a more unbiased and holistic approach to understanding pharmacologic on-target or off-target effects at the protein level [6–9].
We have shown that treatment with cilostazol, a cyclic nucleotide phosphodiesterase III inhibitor, prevented cognitive decline as a result of amyloid-β (Aβ) deposition in the brain of Tg-SwDI mice [10]. Our hypothesis was that pharmacologic treatment of Alzheimer’s disease exerts hemisphere asymmetry.
MATERIALS AND METHODS
Transgenic mouse model
To ensure reproducibility of the pharmacologic treatment and subsequent proteomic analysis the experiment was performed in duplicate. For experiments A and B, male, eight-month-old, homozygous Tg-SwDI mice (n = 6 per experiment, n = 12 in total) on a pure C57BL/6 background were obtained from Jackson Laboratories. These mice express low levels of human Swedish/Dutch/Iowa mutant AβPP in neurons under the control of the mouse Thy1.2 promoter. Mice were randomly assigned to two groups and fed with pelleted chow containing 0.3% cilostazol (n = 3 per experiment, n = 6 in total) or standard pelleted chow only (n = 3 per experiment, n = 6 in total) for 3 months. Cilostazol was supplied by Otsuka PharmaceuticalCo. Ltd.
A previous study reported that in rats treated with 0.1% cilostazol, the plasma cilostazol concentration was approximately 1 μM [11]. Based on the in vitro IC50 value, this concentration is estimated to sufficiently inhibit phosphodiesterase III activity [12]. Additionally, studies have shown that treatment with 0.1-0.3% cilostazol in rats positively affected vascular function [12, 13]. Based on this evidence, a 0.3% cilostazol treatment was used in the present study.
Mice were perfused intracardially with phosphate buffer saline, cerebral hemispheres removed and snap frozen in liquid nitrogen. All experimental procedures were approved by the Institutional Animal Care and Use Committee at the National Cerebral and Cardiovascular Center, Japan.
Proteomic analysis
For experiments A and B, respectively, cerebral hemispheres were suspended in dissolution buffer (0.5 M triethylammonium bicarbonate, 0.05% sodium dodecyl sulfate) and homogenized using the FastPrep system (Savant Bio, Cedex, Fr) followed by pulsed probe sonication (Misonix, Farmingdale, NY, USA). Lysates were subjected to centrifugation (16,000 g, 10 min, 4°C) and supernatants measured for protein content using the Direct DetectTM system (Merck Millipore, Darmstadt, Germany). For experiments A and B separately, protein extracts were pooled from each hemisphere of the three vehicle treated mice (33.3 μg from each lysate giving 100 μg final protein content). Left and right hemispheres of cilostazol treated mice were individually analyzed (100 μg from each lysate). All samples were subjected to reduction, alkylation, trypsin proteolysis, eight-plex iTRAQ labeling and two-dimensional liquid chromatography, tandem mass spectrometry analysis as described previously [8, 14, 15] (Fig. 1A).
Unprocessed raw files were submitted to Proteome Discoverer 1.4 for target decoy searching with SequestHT [8, 14]. Quantification ratios were median-normalized and log2 transformed. After treatment, a protein was considered differentially expressed in the left compared to the right hemisphere relative to its respective control when its two-group T-Test p-value across both experiments was equal to or below 0.05.
Principal component analysis (using the iTRAQ ratios of all analyzed proteins for experiments A and B) was performed using BioConductor-R (version 2.15.1) and g-plots in R (version 3.1.2). Heatmap construction of differentially expressed proteins between the two hemispheres was generated using Gene Cluster (version 3.0) and Java Treeview (version 1.1.6r4). Ingenuity Pathway Analysis (Qiagen, Venlo, Netherlands) was applied to identify canonical pathways and biological processes enriched in the differentially expressed proteins between right and left cerebral hemispheres. An |activation z-score |≥2.0 and a p-value ≤ 0.05 were considered significant.
RESULTS
A total of 9,116 proteins were quantitatively analyzed in experiment A (p < 0.05) and another 7,418 proteins in experiment B (p < 0.05), of which 6,137 were reproducibly profiled in both experiments. The average coefficient variation (CV) for the iTRAQ ratios of all proteins profiled across biological replicates was determined to be 24% and 23% for the right and left hemispheres, respectively, of cilostazol treated mice compared to control in experiment A and 15% and 16% for the right and left hemispheres, respectively, of cilostazol treated mice compared to control in experiment B. Analogous CV values between biological replicates were reported by the authors using similar proteomic methodologies [14, 15]. Principal component analysis of the reporter ion ratios of proteins profiled in each experiment showed that the right hemispheres clustered separately from the left hemispheres of cilostazol treated mice (Fig. 1B). Following treatment with cilostazol, 765 proteins were differentially expressed between the right and left cerebral hemispheres across both proteomic experiments (p≤0.05) (Fig. 1C) (Supplementary Table 1).
Bioinformatics interpretation of proteomic results
Ingenuity Pathway Analysis (IPA) using the average reporter ion ratios across both experiments of differentially expressed proteins between right and left hemisphere following cilostazol treatment showed that the G beta-gamma (Gβγ) signaling pathway significantly decreased in the right hemisphere of cilostazol treated mice (p = 1.16E-2; activation z-score = –2.646) whereas it significantly increased in the left hemisphere of cilostazol treated mice (p = 1.16E-2; activationz-score = 2.646) (Fig. 1D).
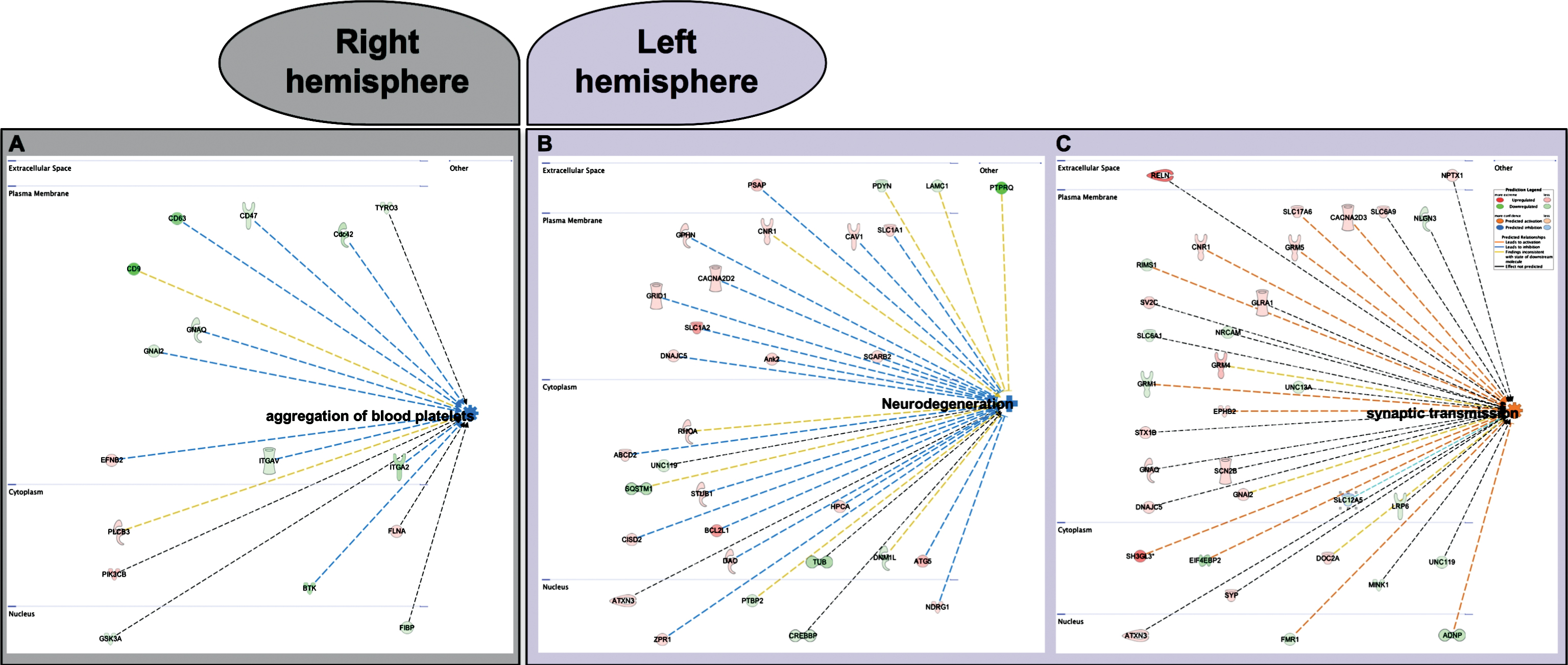
IPA also showed that aggregation of blood platelets significantly decreased in the right hemisphere only of cilostazol treated mice (p = 3.98E-3; activation z-score = –2.231) (Fig. 2A) whereas neurodegeneration significantly decreased (p = 7.7E-6; activation z-score = –2.071) (Fig. 2B) and synaptic transmission significantly increased (p = 8.62E-6; activation z-score = 2.028) (Fig. 2C) specifically in the left hemisphere of cilostazol treated mice.
DISCUSSION
This study provided novel protein level evidence on the hemisphere-specific pharmacologic effects of cilostazol, a drug that has been reported to ameliorate the symptoms of Alzheimer’s disease [16]. This observation may have important implications for an ongoing clinical trial assessing the efficacy of cilostazol in mild cognitive impairment (COMCID trial, ClinicalTrials.gov identifier: NCT02491268) but may also be extended to other pharmacologic agents.
There are only few studies examining cerebral laterality in the pathophysiology of Alzheimer’s disease. Douaud et al. [17] suggested that neurodegenerative pathology may develop asymmetrically in humans, with left hemisphere atrophy being an important predictor of progression from mild cognitive impairment to Alzheimer’s disease. Singh et al. [5] have found that atrophy of the cerebral cortex occurred earlier and progressed faster in the left compared to the right hemisphere in patients with Alzheimer’sdisease. Their findings confirmed previous observations by Chetelat et al. [18] on left lateralization of temporal changes occurring during mild cognitive impairment to Alzheimer’s disease transition. Thompson et al. [19] examined the dynamics of grey matter loss in Alzheimer’s disease and concluded that shifting deficits were asymmetric and predominantly occurring in the left compared to the right hemisphere.
Our results showed that cilostazol asymmetrically affected the two hemispheres of a transgenic Alzheimer’s disease mouse model. In the right hemisphere, aggregation of blood platelets was found to significantly decrease following cilostazol treatment (Fig. 2A). Cerebral amyloid angiopathy (CAA), characterized by the deposition of amyloid-beta in the walls of cerebral arteries plays an important role in the pathogenesis of Alzheimer’s disease. Recent evidence suggests that platelet activation may also contribute to the development of CAA [20, 21]. Targeting blood platelets has been examined as a new venue for the treatment of Alzheimer’s disease and cilostazol is awell-established antiplatelet agent currently used for the secondary prevention of cerebral infarction [22, 23].
In the left hemisphere, cilostazol decreased neurodegeneration (Fig. 2B) and increased synaptictransmission (Fig. 2C), both suggestive of animprovement to the neurodegenerative phenotype. Furthermore, Gβγ signaling was asymmetrically increased in the left and decreased in the right cerebral hemisphere following cilostazol treatment (Fig. 1D). G-protein coupled receptors are highly diverse membrane proteins that participate in the transduction of external signals to various subcellular compartments via trimetric G-proteins. G-protein signaling in the central nervous system has been implicated in nuclear gene expression and cytoskeletal reorganization, processes that significantly contribute to synaptic plasticity and memory [24]. To further investigate whether these processes were on- or off-target effects of cilostazol was beyond the scope of the present proof-of-concept study.
In conclusion, our pharmacoproteomic study provides novel endophenotypic evidence on the hemisphere-specific pharmacologic effects of cilostazol. Future studies should account for hemisphere laterality with important implications in deciphering regional pharmacodynamic effects of existing drugs thus uncovering new hemisphere-specific therapeutic targets.
Some limitations of the present study include the non-validated proteomic findings using an alternative technique (e.g., immunohistochemistry). Furthermore, sub-regions of the cerebral hemispheres (e.g., hippocampus, cortex, cerebellum) were not studied separately to identify regional asymmetric patterns of pharmacologic response to treatment. These constitute objectives in future studies.
ACKNOWLEDGMENTS
We are indebted to Mr. Roger Allsopp, Mr. Derek Coates, and Hope for Guernsey for their fund raising and vision in establishing a clinical mass spectrometry laboratory at the University of Southampton. This study was funded by the BBSRC, Rosetrees Trust and the Wessex Cancer Trust and Medical Research, UK. The authors are grateful to the support of King Saud University, Deanship of Scientific Research Chair, Prince Mutaib Bin Abdullah Chair for Biomarkers of Osteoporosis, College of Science, as well as the Visiting Professor Program of King Saud University, Riyadh, Saudi Arabia.
Authors’ disclosures available online (http://j-alz.com/manuscript-disclosures/15-1078).
Appendices
The supplementary material is available in the electronic version of this article: http://dx.doi.org/10.3233/JAD-151078.
REFERENCES
1 | Broca MP ((1861) ) Remarques sur le siège de la faculté du langage articulé, suivies d’une observation d’aphémie (perte de la parole). Bull Soc Anatomique 6: , 330–357. |
2 | Sun T , Walsh CA ((2006) ) Molecular approaches to brain asymmetry and handedness. Nat Rev Neurosci 7: , 655–662. |
3 | Toga AW , Thompson PM ((2003) ) Mapping brain asymmetry. Nat Rev Neurosci 4: , 37–48. |
4 | Karlebach G , Francks C ((2015) ) Lateralization of gene expression in human language cortex. Cortex 67: , 30–36. |
5 | Singh V , Chertkow H , Lerch JP , Evans AC , Dorr AE , Kabani NJ ((2006) ) Spatial patterns of cortical thinning in mild cognitive impairment and Alzheimer’s disease. Brain 129: , 2885–2893. |
6 | D’Alessandro A , Zolla L ((2010) ) Pharmacoproteomics: A chess game on a protein field. Drug Discov Today 15: , 1015–1023. |
7 | Roumeliotis TI , Halabalaki M , Alexi X , Ankrett D , Giannopoulou EG , Skaltsounis AL , Sayan BS , Alexis MN , Townsend PA , Garbis SD ((2013) ) Pharmacoproteomic study of the natural product Ebenfuran III in DU-145 prostate cancer cells: The quantitative and temporal interrogation of chemically induced cell death at the protein level. J Proteome Res 12: , 1591–1603. |
8 | White CH , Johnston HE , Moesker B , Manousopoulou A , Margolis DM , Richman DD , Spina CA , Garbis SD , Woelk CH , Beliakova-Bethell N ((2015) ) Mixed effects of suberoylanilide hydroxamic acid (SAHA) on the host transcriptome and proteome and their implications for HIV reactivation from latency. Antiviral Res 123: , 78–85. |
9 | Delehouzé C , Godl K , Loaëc N , Bruyère C , Desban N , Oumata N , Galons H , Roumeliotis TI , Giannopoulou EG , Grenet J , Twitchell D , Lahti J , Mouchet N , Galibert MD , Garbis SD , Meijer L ((2014) ) CDK/CK1 inhibitors roscovitine and CR8 downregulate amplified MYCN in neuroblastoma cells. Oncogene 33: , 5675–5687. |
10 | Maki T , Okamoto Y , Carare RO , Hase Y , Hattori Y , Hawkes CA , Saito S , Yamamoto Y , Terasaki Y , Ishibashi-Ueda H , Taguchi A , Takahashi R , Miyakawa , Kalaria RN , Lo EH , Arai K , Ihara M ((2014) ) Phosphodiesterase III inhibitor promotes drainage of cerebrovascular β-amyloid. Ann Clin TranslNeurol 1: , 519–533. |
11 | Miyamoto N , Tanaka R , Shimura H , Watanabe T , Mori H , Onodera M , Mochizuki H , Hattori N , Urabe T ((2010) ) Phosphodiesterase III inhibition promotes differentiation and survival of oligodendrocyte progenitors and enhances regeneration of ischemic white matter lesions in the adult mammalian brain. J Cereb Blood Flow Metab 30: , 299–310. |
12 | Oyama N , Yagita Y , Kawamura M , Sugiyama Y , Terasaki Y , Omura-Matsuoka E , Sasaki T , Kitagawa K ((2011) ) Cilostazol, not aspirin, reduces ischemic brain injury via endothelial protection in spontaneously hypertensive rats. Stroke 42: , 2571–2577. |
13 | Fujita Y , Lin JX , Takahashi R , Tomimoto H ((2008) ) Cilostazol alleviates cerebral small-vessel pathology and white-matter lesions in stroke-prone spontaneously hypertensive rats. Brain Res 1203: , 170–176. |
14 | Manousopoulou A , Woo J , Woelk CH , Johnston HE , Singhania A , Hawkes C , Garbis SD , Carare RO ((2015) ) Are you also what your mother eats? Distinct proteomic portrait as a result of maternal high-fat diet in the cerebral cortex of the adult mouse. Int J Obes (Lond) 39: , 1325–1328. |
15 | Papachristou EK , Roumeliotis TI , Chrysagi A , Trigoni C , Charvalos E , Townsend PA , Pavlakis K , Garbis SD ((2013) ) The shotgun proteomic study of the human ThinPrep cervical smear using iTRAQ mass-tagging and 2D LC-FT-Orbitrap-MS: The detection of the human papillomavirus at the protein level. J Proteome Res 12: , 2078–2089 . |
16 | Ihara M , Nishino M , Taguchi A , Yamamoto Y , Hattori Y , Saito S , Takahashi Y , Tsuji M , Kasahara Y , Takata Y , Okada M ((2014) ) Cilostazol add-on therapy in patients with mild dementia receiving donepezil: A retrospective study. PloS One 9: , e89516. |
17 | Douaud G , Menke RA , Gass A , Monsch AU , Rao A , Whitcher B , Zamboni G , Matthews PM , Sollberger M , Smith S ((2013) ) Brain microstructure reveals early abnormalities more than two years prior to clinical progression from mild cognitive impairment to Alzheimer’s disease. J Neurosci 33: , 2147–2155. |
18 | Chételat G , Desgranges B , De La Sayette V , Viader F , Eustache F , Baron JC ((2002) ) Mapping gray matter loss with voxel-based morphometry in mild cognitive impairment. Neuroreport 13: , 1939–1943. |
19 | Thompson PM , Hayashi KM , de Zubicaray G , Janke AL , Rose SE , Semple. J , Herman D , Hong MS , Dittmer SS , Doddrell DM , Toga AW ((2003) ) Dynamics of gray matter loss in Alzheimer’s disease. J Neurosci 23: , 994–1005. |
20 | Jarre A , Gowert NS , Donner L , Münzer P , Klier M , Borst O , Schaller M , Lang F , Korth C , Elvers M ((2014) ) Pre-activated blood platelets and a pro-thrombotic phenotype in APP23 mice modeling Alzheimer’s disease. Cell Signal 26: , 2040–2050. |
21 | Kniewallner KM , Ehrlich D , Kiefer A , Marksteiner J , Humpel C ((2015) ) Platelets in the Alzheimer’s disease brain: Do they play a role in cerebral amyloid angiopathy?. Curr Neurovasc Res 12: , 4–14 . |
22 | Shi L , Pu J , Xu L , Malaguit J , Zhang J , Chen S ((2014) ) The efficacy and safety of cilostazol for the secondary prevention of ischemic stroke in acute and chronic phases in Asian population–an updated meta-analysis. BMC Neurol 14: , 251. |
23 | Shinohara Y , Katayama Y , Uchiyama S , Yamaguchi T , Handa S , Matsuoka K , Ohashi Y , Tanahashi N , Yamamoto H , Genka C , Kitagawa Y , Kusuoka H , Nishimaru K , Tsushima M , Koretsune Y , Sawada T , Hamada C ((2010) ) Cilostazol for prevention of secondary stroke (CSPS 2): An aspirin-controlled, double-blind, randomised non-inferiority trial. Lancet Neurol 9: , 959–968, pp. |
24 | El Far O , Betz H ((2002) ) G-protein-coupled receptors for neurotransmitter amino acids: C-terminal tails, crowded signalosomes. Biochem J 365: , 329–336. |
Figures and Tables
Fig.1
A) Experimental workflow. B) Principal component analysis for each experiment showed separate clustering for the right and left hemispheres of cilostazol treated mice compared to controls suggesting hemisphere asymmetry of pharmacologic response to cilostazol treatment. C) Hierarchical clustering analysis of differentially expressed proteins between the right and left hemispheres in both experiments visualized in heatmap format. D) Canonical pathway analysis of differentially expressed proteins between right and left hemisphere following cilostazol treatment showed that the G beta-gamma (Gβγ) signaling pathway significantly decreased in the right (p = 1.16E-2; activation z-score = –2.646) whereas it significantly increased in the left hemisphere of cilostazol treated mice (p = 1.16E-2; activation z-score = 2.646).
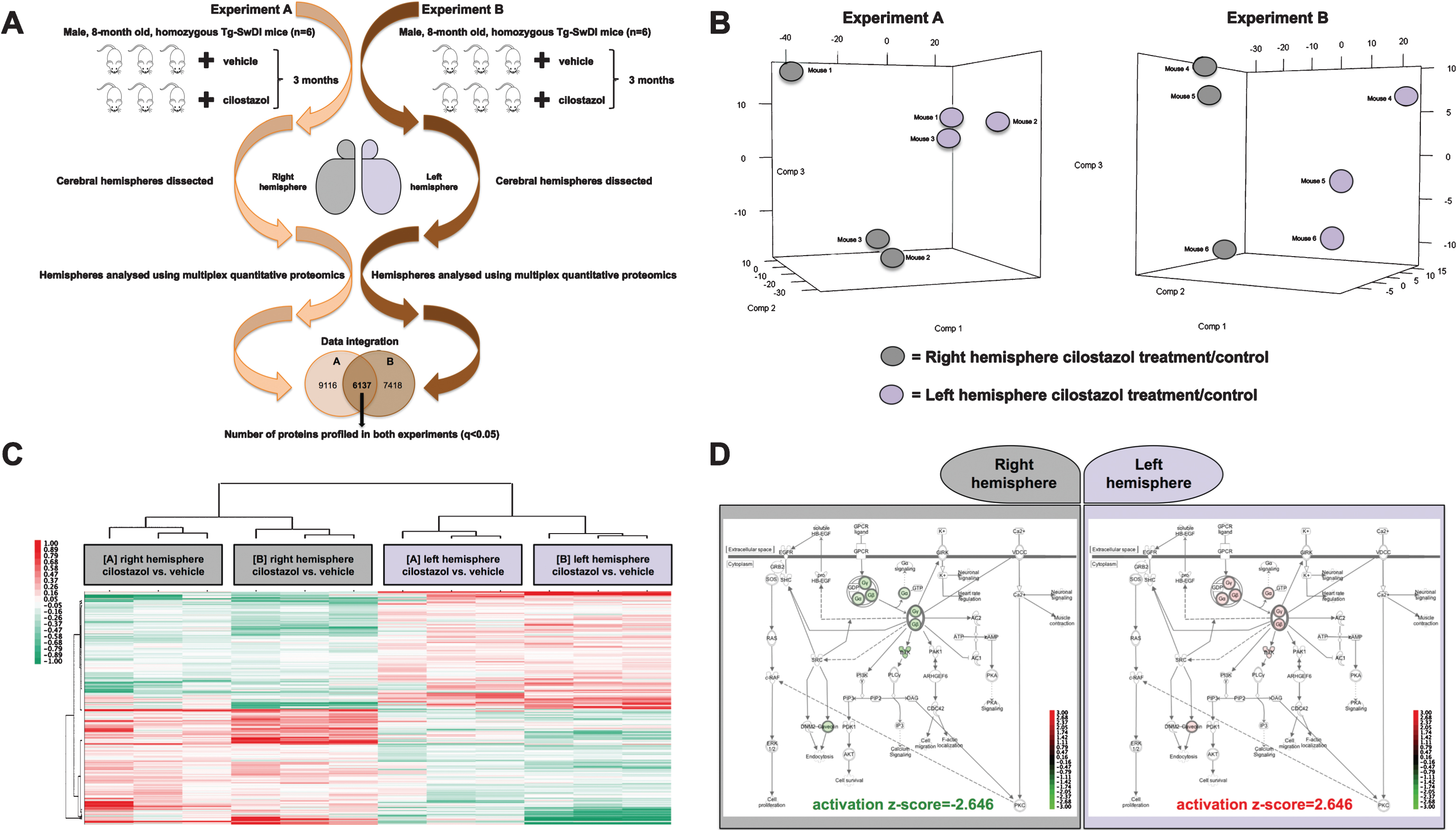
Fig.2
Bioinformatics interpretation of the results showed that (A) aggregation of blood platelets significantly decreased in the right hemisphere (p = 3.98E-3; activation z-score=–2.231) whereas (B) neurodegeneration significantly decreased (p = 7.7E-6; activation z-score = –2.071) and (C) synaptic transmission significantly increased (p = 8.62E-6; activation z-score = 2.028) in the left hemisphere of cilostazol treated mice.