Pain Assessment in Elderly with Behavioral and Psychological Symptoms of Dementia
Abstract
Background:
Pain is under-detected and undertreated in people with dementia. The present study investigates the prevalence of pain in people with dementia hospitalized in nursing homes that are members of National Association of Third Age Residences (ANASTE) Calabria, and evaluates the association among pain, mood, and behavioral and psychological symptoms of dementia (BPSD).
Objective:
The aim of this study is to define the prevalence of pain in people with dementia in long term care facilities using scales of self-reporting and observational tools and, particularly, to study the relationship between pain and BPSD.
Methods:
A prospective observational study was carried out on 233 patients. Pain assessment was performed using self-reporting tools such as the Numeric Rating Scale (NRS) for patients with slight cognitive impairment or no cognitive impairment and observational tools such as Pain Assessment In Advanced Dementia Scale (PAINAD) for patients with moderate or severe cognitive impairment. Mood was evaluated through the Cornell Scale for Depression in Dementia (CSDD) while behavioral problems were assessed through the Cohen-Mansfield Agitation Inventory (CMAI) and Neuropsychiatric Inventory (NPI).
Results:
Only 42.5% of patients evaluated by NRS provided a reliable answer; of these, 20.4% reported no pain. The percentage of pain evaluated by PAINAD was 51.8% . Analysis of data showed a statistically significant correlation between diagnosis of pain and depressive symptoms, assessed with CSDD (p = 0.0113), as well as by single items of NPI, such as anxiety (p = 0.0362) and irritability (p = 0.0034), and F1 profile (Aggression) of CMAI (p = 0.01).
Conclusion:
This study confirms that self-report alone is not sufficient to assess pain in elderly people with dementia; the observational tool is a necessary and suitable way of assessing pain in patients with cognitive impairment. If not adequately treated, chronic pain can cause depression, agitation, and aggression in patients with dementia.
INTRODUCTION
Prevalence of chronic pain has been estimated to vary between 20% and 50% in elderly people, and it has been reported to exist in 40–80% of residents in long term care facilities (LTCF) [1]. Dementia, along with its behavioral and psychiatric symptoms, has long been the prevalent mental disorder in LTCF [2]. The presence of dementia in American nursing homes (NH) is widespread and it is in constant growth; in fact in 2010 almost 60% of residents had a diagnosis of Alzheimer’s disease (AD) or other dementia [3].
In European LTCF, the Services and Health for the Elderly in Long Term Care (SHELTER) Study showed that 37.6% of patients had mild/moderate cognitive impairment, 30.5% had severe cognitive impairment, and 27.5% showed behavioral symptoms [4]. About 78% of patients residing in Italian LTCF associated with the National Association of Third Age Residences (ANASTE) Calabria, were found to suffer from cognitive deterioration, differing in etiologies and degree; 52% of these were found to be affected by severe dementia [5].
Dementia complicates the assessment of pain, because it impairs memory and verbal communication. In patients, it produces significant functional changes in central nervous system tolerance to pain, but no pain threshold results altered [6].
To date it is still unclear if, in these patients, the experience of pain changed due to the decline of nociceptive pathways or to the progressive degeneration of the brain regions involved in the processing of nociceptive stimulus [7]. Some studies suggest that people with dementia can experience pain differently from people without dementia. Benedetti and colleagues [8] found that patients with AD had a higher pain tolerance and it was directly related to the severity of cognitive impairment. Conversely, Cole et al. [9] investigated pain-related brain activity by functional MRI and showed that perception and processing of pain decreased in AD patients.
However, there is no empirical evidence to suggest that people with dementia feel less pain: actually they might not be able to verbally communicate pain [10]. Self-reporting is often considered the ‘gold standard’ in pain assessment. A wide range of self-report scales is currently available to assess pain in the elderly, most of which have been developed and also tested on elderly people with dementia [11].
Despite recent studies supporting the reliability and validity of self-report for people with dementia, health professionals and experts recognize that pain self-report alone is not enough for these patients who are necessarily subjected to observational strategies for pain assessment [12]. In 2002, the American Geriatrics Society established comprehensive guidelines for the assessment of behavioral indicators of pain [13].
More recently, the American Society for Pain Management Nursing Task Force on the assessment of pain in patient record (including people with dementia) recommended a holistic approach that integrates hierarchical self-report and observation of pain behavior [14]. In addition, at least 15 instruments for pain observation have been developed and validated over the last decade. Assessment of Pain in Advanced Dementia Scale (PAINAD) by Warden et al. [15] was developed to assess pain in patients with advanced dementia; the test was specific for a residential setting. Behavior such as verbal complaints, negative vocalizations, sighing, moaning, agitation, crying, grimacing, rapid blinking, movement/restlessness, rubbing, strength, reinforcement, stiffness, wandering, inappropriate verbal speech, and aggression can be considered as signs of pain [16], but in patients with cognitive impairment, these signs may be taken as behavioral and psychological symptoms of dementia (BPSD) [17].
Because BPSD can confuse pain assessment, identifying pain in people with dementia becomes even more important. In fact, causes of BPSD are very complex and multifactorial and they can be an expression of unmet needs, such as pain [18]. BPSD can affect up to 80-85% of people with dementia and it significantly increases the cost of care as well as the caregiver’s burden and depression, decreasing quality of life [19]. Until now, little is known about the interaction between pain and behavioral symptoms.
The aim of this study is to define the prevalence of pain in people with dementia in LTCF using scales of self-reporting and observational tools and, particularly, to study the relationship between pain and BPSD.
METHODS
A network of LTCF consisting of Nursing Homes and Residences for Extensive Rehabilitation (RER) operates in the care of the frail elderly in Calabria. Admission to these facilities is regulated according to the guidelines provided by the Calabria Region (DGR 685/2002, DGR 695/2003, LR 29/2008, DGR 3137/1999). This is an observational and descriptive study carried out on a sample of resident people, across 10 NH associated with the ANASTE Calabria in March 2014. The present study was conducted by carrying out the customary care practice, normally provided to all patients, who belong to the LTCF ANASTE Calabria, and nursing care did not involve any different procedures. Informed consent for daily care practice and use of personal data is normally required when people are admitted to LTCF. All patients underwent multidimensional geriatric assessment. The standard protocol was submitted to the local Ethical Committee for approval.
Subjects and measurements
A sample of 233 residents, 150 females (64.4% ) and 83 males (35.6% ), was subjected to multidimensional and multidisciplinary evaluation. The clinical diagnosis of dementia was firstly investigated through a detailed interview about personal as well as familiar history and, subsequently, confirmed by the administration of psychometric tests. All patients fulfilled the criteria for dementia as described in the Diagnosticand Statistical Manual of Mental Disorders, Revised Fourth Edition (DSM IV) [20]. National Institute of Neurological and Communicative Disorders and Stroke/Alzheimer’s Disease and Related Disorders Association (NINCDS/ADRDA) [21] criteria for diagnosis of AD were used, while for vascular dementia (VaD), the criteria of the National Institute for Neurological Disorders and Stroke-Association Internationale pour la Recherche et Enseignement en Neurosciences (NINCDS-AIREN) [22] was used.
Diagnosis of chronic pain was made according to ICD9-CM Official [23]. Cognitive evaluation was conducted using Folstein’s Mini-Mental State Examination (MMSE) [24]. According to the MMSE, patients were affected by severe, moderate, or slight cognitive impairment on scores ranging from 0–10, 11–20, or 21–23, respectively. Functional state was evaluated through the Activity Daily Living scale (ADL) and Barthel Index (BI); in both scales, a lower score indicates a worse functional state [25, 26]. Comorbidity was examined through the index of severity (ICS) and index of complex comorbidity (ICC) according to the Cumulative Illness Rating Scale (CIRS), in which the higher score indicates greater comorbidity [27]. The affective state was scored through the Cornell Scale for Depression in Dementia (CSDD); a high score (>9) denoted a depressive state. The scale consists of nineteen items; the depressive symptoms were noted during the week preceding the interview and they are classified as absent, mild/intermittent, and severe. Symptoms are grouped into five main categories: mood-related signs, behavioral disturbances, physical signs, and cyclic function.
Pain assessment was performed through self-reporting Numeric Rating Scale (NRS) for patients without cognitive impairment or affected by slight cognitive impairment (MMSE >20). The NRS score was: 0 no pain, 1–3 mild pain, 4–6 moderate pain, and finally 7–10 severe pain. The NRS was presented as a horizontal line where 0 represents no pain and 10 the deepest pain, equally spaced dashes representing pain intensity rating ranging 1 to 9. The scale was printed in large and bold characters to be easily used by visually impaired patients [14].
PAINAD is an observational tool and it was used on patients affected by moderate or severe cognitive impairment (MMSE <20). PAINAD scoring is 0–1 no pain, 2–4 light pain, 5–7 moderate pain, and 7–10 severe pain [15]. The PAINAD version used consists of five types of behavior with three possible answers (0, 1, or 2). An increased score reflects a growing pain. Nurses were trained on the use of the tool. Before applying the scale, an observation period of 5 minutes was respected. The authors consider it user-friendly [11].
Behavioral disorders were subdivided as following: mild (1–48), moderate (49–96), and severe (97–100) according to the total score of Neuropsychiatric Inventory (NPI) [29]. The NPI is based on responses from an informed caregiver. It is a reliable and valid measure for behavioral symptoms in patients with dementia. Twelve neuropsychiatric symptoms were considered: Delusions, Depression, Hallucinations, Agitation/Aggression, Anxiety, Euphoria/Exaltation, Apathy/Indifference, Disinhibition, Irritability/Lability, Motor Disorder, Appetite Disorders, and Sleep. Further questions were asked to determine the severity (1 = Mild: produces little distress in the patient; 2 = Moderate: more disturbing to the patient but can be redirected by the caregiver; 3 = Severe: very disturbing to the patient and difficult to redirect) and frequency (0 = Never; 1 = Rarely: less than once per week; 2 = Sometimes: about once per week; 3 = Often: several times per week but less than every day; 4 = Very often: once or more per day) of behavioral disorders. The individual domain scores for each of the 12 neuropsychiatric symptoms were obtained by calculating the product of the frequency and severity scores of each symptom (cut-off 0–12). The sum of these domain scores produced the total NPI score for each patient [29]. For statistical analysis each NPI-domain value >0 was included.
Manifestations of agitated behavior were evaluated according to the Cohen-Mansfield Agitation Inventory (CMAI) profiles: Aggression (F1), Physical non-aggression (F2), Verbal agitation (F3). It is a specific rating scale of behavior that includes activities of self-care, cognition, mood, and objects. CMAI uses a 7-points scale to assess the frequency (from ‘never’ to ‘several times an hour’) of 29 behaviors commonly observed in residents. The behaviors are divided in four groups: verbally abusive (for example, directed at a person or an object), not verbally aggressive (not directed at a specific object or person), physically aggressive (direct), and physically non-aggressive (undirected). The total score is more commonly used to quantify behavioral disorders. The staff was trained before using the tool [30].
Statistical analysis
Descriptive statistical analysis was elaborated by qualitative variables, expressed as percentages, and quantitative variables as mean values with standard deviation. Statistical analysis was performed using univariate and bivariate analysis between pain diagnosis (ICD-9) and depressive symptoms evaluated by CSDD for single item of NPI with Odds Ratio (OR) and related confidence interval (C.I.) at 95% estimated for the primary endpoint. Adjusted prevalence of each endpoint was analyzed with logistic regression model between pain rated by PAINAD, depressive symptoms by CSDD, and aggression by CMAI profile F1. In addition, the statistical significance in the logistic regression model between the irritability/lability (dependent variable) and pain, anxiety, depression, and drugs (independent variables) was calculated. The value of the OR to measure the associations has been interpreted as follow: OR = 1, absence of association; OR <1, negative association; OR >1, positive association. For OR values greater than 1, the association was considered “stronger". The level of statistical significance was set at: *p < 0.05; **p < 0.01; ***p < 0.005. Finally, statistical analysis was performed using EpiInfo version 3.2.2 and SPSS version 19.
RESULTS
77.7% of the sample met the criteria of the DSM-IV for dementia. VaD was diagnosed in 61.9% (60% VaD probable and 40% VaD possible according to NINCDS-AIREN criteria) whereas patients with AD were 29.3% (73.6% AD probable and AD possible in the 26.4% according to NINCDS-ADRDA criteria). Characteristics of the sample are shown in Table 1. Prevalence of pain in people with dementia, according to IC D9-CM Official codes [23], was 46.4% whereas pain was present in 28.3% of those without dementia (Table 2). Based on the type and location, pain has been classified in oncological (A), nociceptive somatic musculoskeletal (B), post-traumatic (C), post-surgical (D), neuropathic (E), psychogenic (F), and mixed (G) pain (Fig. 1). Only 42.5% of patients evaluated by NRS provided a reliable answer: 20.4% of patients referred no pain, 14.4% reported mild pain, 3.9% had moderate pain and, finally, 2.9% of them reported severe pain. 97% of patients with dementia and pain (ICD-9 diagnosed) were taking analgesic medications. Among 53.5% of people with dementia without pain that was ICD-9 diagnosed, 8.2% were taking analgesic medications as well.
Prevalence of pain evaluated by PAINAD was 51.8% (34.9% had slight pain, 15.7% moderate pain, and 1.2% severe pain), while 48.2% of patients did not report pain. 49.7% did not indicate the type of pain, while in 36.5% pain was musculoskeletal, in 9.5% it was mixed, finally, 3.3% and 1.2% of patients reported neuropathic and psychogenic pain, respectively. 47.0% of these patients were taking pain medications, classified according to the Anatomical Therapeutic Chemical; 36.1% were taking benzodiazepines, 47.5% neuroleptics, and 31.5% antidepressants (Fig. 2). The evaluation of the affective state with CSDD showed that 62.8% of the sample had depressive symptoms (Table 1). NPI total score, used to evaluate the BPSD, showed that 77.3% of the sample displayed a mild behavior disorder, 22.1% presented a moderate disorder, whereas only 0.6% of patients displayed severe behavior modifications (Table 1). Behavioral symptoms evaluated through single item of NPI in patients with dementia are shown in Fig. 3. The evaluation by CMAI showed that 20.0% of the sample had F1 profile, 39.4% F2 profile, and 40.6% F3 profile (Table 1). The bivariate analysis showed a statistically significant correlation between diagnosis of pain and depressive symptoms, assessed with Cornell Scale (p = 0.0113), as well as with single item of NPI which anxiety (p = 0.0362) and irritability/lability (p = 0.0034) (Table 3). Each variable was tested in a logistic regression model to correct the influence of different factors; pain (OR: 3.025; 95% CI: 1.168–7.838; p = 0.0227), anxiety (OR: 4.536; 95% CI: 1.124–6.449; p = 0,0021), and depression (OR: 2.692; 95% CI: 1.124–6.449; p = 0.0263) were statistically significantly associated with irritability/lability (Fig. 4). Following, the univariate and bivariate analysis were carried out considering the pain evaluated according to PAINAD. New logistic regression models were obtained comparing the variable found to be associated; results show a correlation between the presence of pain, assessed by PAINAD, and depressive symptoms, Cornell Scale (p = 0.05), and CMAI profile F1 (Aggression) (p = 0.01) statistically significant (Table 4). The use of antipsychotics, anxiolytics, and sedative/hypnotic drugs does not modify the associations (Table 5).
DISCUSSION
In LTCF residents with dementia, pain is often ignored and under-treated. The under-treatment of pain was observed in all three stages of AD (early, middle, and end) and in all subtypes of dementia [31].
In our study, the prevalence of chronic pain was almost double in patients with dementia than in those without dementia. The lack of a well-defined pain assessment tool and proper documentation are the main obstacles to a successful pain management in residents with dementia. In fact, only about 40% of residents were evaluated with NRS for an answer deemed reliable. Observing behavior is a necessary and suitable measure of pain, among people with moderate and severe cognitive impairment in particular. Generally, many studies support the psychometric properties of different pain observational instruments [32].
There is also a considerable variation in the estimates of pain prevalence in people with dementia that can be attributed to differences in research methodology, setting, and characteristics of the sample studied. Similarly there is a different prevalence of VaD and AD.
In our study, we found a higher prevalence of VaD and a lower prevalence of AD. Evidence is supported by a very recent Italian study, InveCe.Ab study [33]. In fact, Guaita and colleagues have reported that the overall dementia prevalence was 3% (95% CI: 2.1–4.1% ). In particular, 38.5% of the subjects with dementia were diagnosed with AD, and 46.1% with VaD. That can be explained by the diagnostic criteria used: actually, only our study, the InveCe.Ab study, and the Conselice study used the NINDS-AIREN criteria for diagnosis of VaD. In addition, subjects born in southern Italy were shown to be at greater risk both of overall dementia and of vascular dementia in relation to the influence of socio-demographic factors.
We found another apparent discrepancy between the group with dementia and pain ICD-9-diagnosed and the group with dementia and pain PAINAD-scored, relating to prevalence of pain (46.4% and 51.8% , respectively) and the analgesic medications use (97% and 47% , respectively). Because PAINAD is used in patients with moderate-severe dementia, we can suppose that a quote of the last group of patients could have not received any diagnosis of pain, so they did not receive adequate pain treatment. The PAINAD was a valuable tool in evaluating patients with moderate and severe cognitive impairment
Some authors report that most observational tools for pain can also take into account “fear”, which could lead to false attributions of pain [19]. Even if PAINAD is a highly sensitive instrument, it has a low specificity and possibility of false positive [34]. In our study there is a significant correlation between pain and depression. A growing body of literature has focused on the interaction between depression and pain [35]. The prevalence of pain in depression cohorts and depression in pain cohorts are higher than in a single condition. This interaction has been described by some authors as “pain syndrome depression” or “depression pain dyed”, implying that the conditions often coexist, so the understanding of this relationship has become very important [35]. A recent study has shown that the circuits involving the prefrontal cortex and anterior cingolate cortex are more likely to be changed in comorbidity between chronic pain and depression [36]. Consequently, depression was interpreted as an emotional state of being rather than physical pain. These shared representations may therefore be related to the behavioral context, memory, and motivation, and may be the network link between depression and pain [37].
Pain is often communicated via behavior. Cohen-Mansfield and Creedon define the behavior pain as “nonverbal behavior observable” [38]. The behavioral symptoms related to dementia are inappropriate, repetitive, and destabilize the living and working environment. Among the symptoms of BPSD described in literature, errant behavior, aggressive behavior, and agitated behavior are the most important. Wandering occurs in approximately 40–60% whereas aggression and agitation occur in about 50% to 80% of NH residents with cognitive deficits [39]. In this framework, it is not controversial that pain can appear as a behavioral disorder, but it is still unclear what particular type of BPSD is more likely to be associated with pain. Our results correlate chronic pain with some behavioral phenotypes, such as anxiety and irritability, evaluated with the NPI, and aggression, according to CMAI. Other studies conducted in NH have shown that pain positively correlates with aggression and agitation and socially inappropriate behavior, as well as resistance to care and delusions. Moreover, it negatively correlates with wandering [40]. Pain, if not treated, can lead to agitated behavior. A study which examined the factors predicting the development of aggression in patients living in community, aged 60 + years with dementia (n = 215), found that participants suffering higher pain had increased risk for development of aggression [18]. Paradoxically, pain related agitation often leads to inadequate treatment with neuroleptics or sedatives rather than analgesic drugs, masking pain related symptoms and consequently hindering the right treatment of pain. In the present study, 46.6% of patients with dementia were treated with analgesics (Fig. 5).
Actually, the systematic use of pain observation tools has been routinely included in multidimensional assessment tests, to improve pain management for residents with dementia in NH.
However, the present study has some limitations, first and foremost, the limited population enrolled in the study. On the other hand, the integrated care pathways for chronic pain in LTCF, developed and used at regional level, would be extended on national level, in order to make specific guidelines for a better pain assessment and treatment of patients living in LTCF.
In conclusion, the present study confirms that instruments of self-report alone are not sufficient to assess pain in elderly people with dementia. Therefore, observational tools for pain should be used in combination with self-report tools in a multidimensional assessment of pain. The integration of these tools in specific pain care pathways makes health assistance more sensitive. There is a close relationship between pain and behavioral and psychological symptoms in patients with dementia. In fact, chronic pain can cause depression, agitation, and aggression in patients with dementia if not adequately treated. Further research is needed to clarify the exact pathophysiological mechanisms underlying behavior and pain. Finally, the ability to identify and treat the real cause of pain in dementia can resolve problematic behavior and improve the quality of life.
ACKNOWLEDGMENTS
This work was conducted in the following LTC Facilities: Casa Protetta “Madonna del Rosario,” Lamezia Terme (CZ); RSA “Villa Elisabetta,” Cortale (CZ); RSA “San Domenico” Lamezia Terme (CZ); Centro di Riabilitazione Estensiva a ciclo continuativo “San Domenico” Lamezia Terme (CZ); RSA “Casa Amica,” Fossato Serralta (CZ); Casa Protetta “San Domenico” Palermiti (CZ); RSA “Santa Maria del Monte” San Sostene (CZ); Casa Protetta “Villa Azzurra” Roseto Capo Spulico (CS).
Our thanks to Mrs. Rosalba Principato for revising the English version of the manuscript.
Authors’ disclosures available online (http://www.j-alz.com/manuscript-disclosures/15-0808r2).
REFERENCES
[1] | Apinis C , Tousignant M , Arcand M , Yannick Tousignant-Laflamme Y ((2014) ) Can adding a standardized observational tool to interdisciplinary evaluation enhance the detection of pain in older adults with cognitive impairments? Pain Med 15: , 32–41. |
[2] | Grabowski DC , Aschbrenner KA , Rome VF , Bartels SJ ((2010) ) Quality of mental health care for nursing home residents: A literature review. Med Care Res Rev 67: , 627–656. |
[3] | Li Q , Zheng NT , Temkin-Greener H ((2013) ) End of life quality of care among long-term nursing home decedent residents with and without dementia. J Am Geriatr Soc 61: , 1066–1073. |
[4] | Onder G , Carpenter I , Finne-Soveri H , Frijters D , Henrard JC , Nikolaus T , Topinkova E , Tosato M , Liperoti R , Landi F , Bernabei R . and the SHELTER project ((2012) ) Assessment of nursing home residents in Europe: The Services and Health for Elderly in Long TERm care (SHELTER) study. BMC Health Serv Res 12: , 5. |
[5] | Malara A , Sgrò G , Caruso C , Ceravolo F , Curinga G , Renda GF , Spadea F , Garo M , Rispoli V ((2014) ) Relationship between cognitive impairment and nutritional assessment on functional status in Calabrian long-term-care. Clin Interv Aging 9: , 105–110. |
[6] | Huffman JC , Kunick ME ((2000) ) Assessment and understanding of pain in patients with dementia. Gerontologist 40: , 574–581. |
[7] | Mcauliffe L , Brown D , Fetherstonhaugh D ((2012) ) Pain and dementia: An overview of the literature. Int J Older People Nurs 7: , 219–226. |
[8] | Benedetti F , Vighetti S , Ricco C , Lagna E , Bergamasco B , Pinessi L , Rainero I ((1999) ) Pain threshold and tolerance in Alzheimer’s disease. Pain 80: , 377–382. |
[9] | Cole LJ , Farrell MJ , Duff EP , Barber JB , Egan GF , Gibson S ((2006) ) Pain sensitivity and fMRI pain-related brain activity in Alzheimer’s disease. Brain 129: , 2957–2965. |
[10] | Bachino C , Snow AL , Kunrk ME , Cody M , Wristers K ((2001) ) Principles of pain assessment and treatment in non-communicative demented patients. Clin Gerontol 23: , 97–115. |
[11] | Zwakhalen S , Abu-Saad J , Berger HM ((2006) ) Pain in elderly people with severe dementia: A systematic review of behavioural pain assessment tools. BMC Geriatr 6: , 3. |
[12] | Pautex S , Michon A , Guedira M , Emond H , Lous P , Samaras D , Michel JP , Herrmann F , Giannakopoulos P , Gold G ((2006) ) Pain in severe dementia: Self-assessment or observational scales. J Am Geriatr Soc 54: , 1040–1045. |
[13] | American Geriatrics Society ((2009) ) AGS panel on the pharmacological management of persistent pain in older persons. JAGS 57: , 1331–1346. |
[14] | Horgas AL , Elliott AF , Marsiske M ((2009) ) Pain assessment in persons with dementia: Relationship between self-report and behavioral observation. J Am Geriatr Soc 57: , 126–132. |
[15] | Warden V , Hurley AC , Volicer L ((2003) ) Development and psychometric evaluation of the pain assessment in advanced dementia (PAINAD) Scale. J Am Med Dir Assoc 4: , 9–15. |
[16] | Burfield AH , Wan TT , Sole ML , Cooper JW ((2012) ) Behavioral cues to expand a pain model of the cognitively impaired elderly in long-term care. Clin Interv Aging 7: , 207–223. |
[17] | Horgas AL ((2008) ) Assessing pain in persons with dementia. Medsurg Nurs 16: , 207–208. |
[18] | Kunik ME , Snow AL , Davila JA , Steele AB , Balasubramanyam V , Doody RS , Schulz PE , Kalavar JS , Morgan RO ((2010) ) Causes of aggressive behavior in patients with dementia. J Clin Psychiatry 71: , 1145–1152. |
[19] | Sampson EL , White N , Lord K , Leurent B , Vickerstaff V , Scott S , Jones L ((2015) ) Pain, agitation, and behavioural problems in people with dementia admitted to general hospital wards: A longitudinal cohort study. Pain 156: , 675–683. |
[20] | American Psychiatric Association ((2000) ) Diagnostic and statistical manual of mental disorders, fourth edition, text revision (DSM-IV-TR). American Psychiatric Association, Washington, DC. |
[21] | McKhann G , Drachman D , Folstein M , Katzman R , Price D , Stadlan EM ((1984) ) Clinical diagnosis of Alzheimer’s disease: Report of the NINCDS-ADRDA Work Group under the auspices of department of health and human services task force on Alzheimer’s Disease. Neurology 34: , 934–994. |
[22] | Roman GC , Tatemichi TK , Erkinjuntti T , Cummings JL , Masdeu JC , Garcia JH , Amaducci L , Orgogozo JM , Brun A , Hofman A et al. ((1993) ) Vascular dementia: Diagnostic criteria for research studies. Report of the NINDS-AIREN International Workshop. Neurology 43: , 250–260. |
[23] | Zeisset A ((2007) ) Coding guidelines for pain. J AHIMA 3: , 78–79. |
[24] | Folstein MF , Folstein SE , McHugh PR ((1975) ) “Mini-mental state.” A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12: , 189–198. |
[25] | Katz S ((1983) ) Assessing self-maintenance: Activities of daily living, mobility, and instrumental activities of daily living. J Am Geriatr Soc 31: , 721–727. |
[26] | Mahoney FI , Barthel DW ((1965) ) Functional evaluation: The Barthel Index. Md State Med J 14: , 61–65. |
[27] | Parmelee PA , Thuras PD , Katz IR , Lawton MP ((1995) ) Validation of the cumulative illness rating scale in a geriatric residential population. J Am Geriatr Soc 43: , 130–137. |
[28] | Alexopulos GS , Abrams RC , Young BC , Shamoian CA ((1988) ) Cornell scale for depression in dementia. Biol Psychiatr 23: , 271–284. |
[29] | Cummings JL , Mega M , Gray K , Rosemberg-Thompson S , Carusi DA , Gornbei J ((1994) ) The neuropsychiatric inventory: Comprehensive assessment of psychopathology in dementia. Neurology 44: , 2308–2314. |
[30] | Cohen-Mansfield J , Marx MS , Rosenthal AS ((1989) ) A description of agitation in the nursing home. J Gerontol 44: , 77–84. |
[31] | Scherder EJA , Bouma A ((1997) ) Is decreased use of analgesics in Alzheimer disease due to a change in the affective component of pain? Alzheimer Dis Assoc Disord 11: , 171–174. |
[32] | Liu JY1 , Lai C ((2014) ) Implementation of observational pain management protocol to improve pain management for long-term institutionalized older care residents with dementia: Study protocol for a cluster-randomized controlled trial. Trials 15: , 78. |
[33] | Guaita A , Vaccaro R , Davin A , Colombo M , Vitali SF , Polito L , Abbondanza S , Valle E , Forloni G , Ferretti VV , Villani S ((2015) ) Influence of socio-demographic features and apolipoprotein E epsilon 4 expression on the prevalence of dementia and cognitive impairment in a population of 70-74-year olds: The InveCe.Ab study. Arch Gerontol Geriatr 60: , 334–343. |
[34] | Jordan A , Hughes. J , Pakresi M , Hepburn S , O’Brien JT ((2011) ) The utility of PAINAD in assessing pain in a UK population with severe dementia. Int J Geriatr Psychiatry 26: , 118–126. |
[35] | Bair MJ , Robinson RL , Katon W , Kroenke K ((2003) ) Depression and pain comorbidity: A literature review. Arch Intern Med 163: , 2433–2445. |
[36] | Doan L , Manders T , Wang J ((2015) ) Neuroplasticity underlying the comorbidity of pain and depression. Neural Plast 2015: , 504691. |
[37] | Woo CW , Koban L , Kross E , Lindquist MA , Banich MT , Ruzic L , Andrews-Hanna JR ((2014) ) Separate neural representations for physical pain and social rejection. Nat Commun 5: , 5380. |
[38] | Cohen-Mansfield. J , Creedon M ((2002) ) Nursing staff member’s perceptions of pain indicators in persons with severe dementia. Clin J Pain 18: , 64–73. |
[39] | Ahn H , Horgas A ((2013) ) The relationship between pain and disruptive behaviors in nursing home resident with dementia. BMC Geriatr 13: , 14. |
[40] | Tosato M , Lukas A , van der Roest HG , Danese P , Antocicco M , Finne-Soveri H , Nikolaus T , Landi F , Bernabei R , Onder G ((2012) ) Association of pain with behavioral and psychiatric symptoms among nursing home residents with cognitive impairment: Results from the SHELTER study. Pain 153: , 305–310. |
Figures and Tables
Fig.1
Typology of pain in people with dementia. Oncological (A); Nociceptive Somatic Musculoskeletal (B); Post traumatic (C); Post-surgical (D); Neuropathic (E); Psychogenic (F) and Mixed (G) pain.
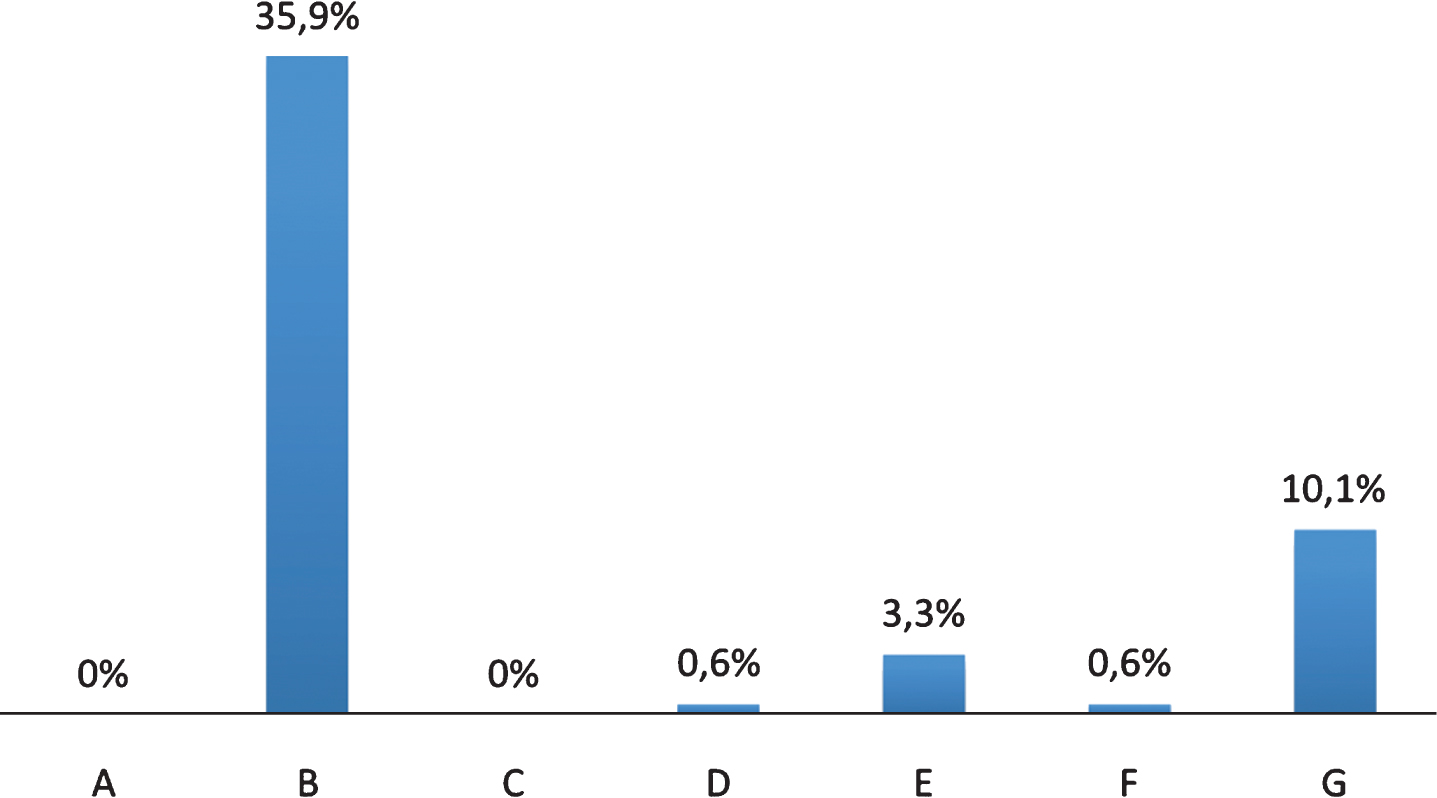
Fig.2
Drugs used in people with dementia and chronic pain. Classes of Anatomical Taxonomic Classification (ATC) Drugs: ATC N02: Analgesic Drugs; M01: FANS; N05 A: Antipsychotic drugs; N05B: Anxiolytics drugs; N05C: Hypnotic/sedative drugs; N06A: Antidepressants drugs.
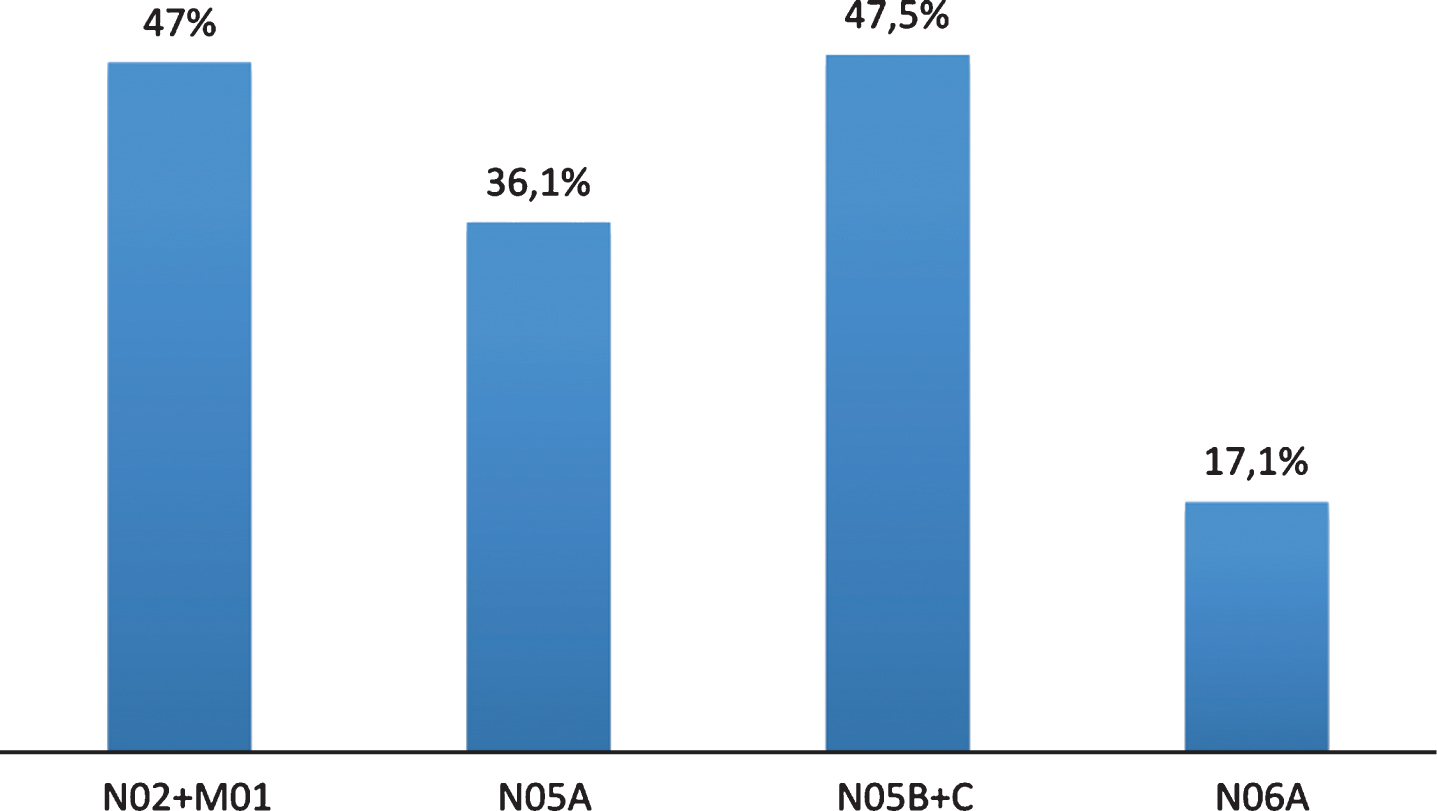
Fig.3
Behavioral symptoms evaluated through single item of NPI in people with dementia.
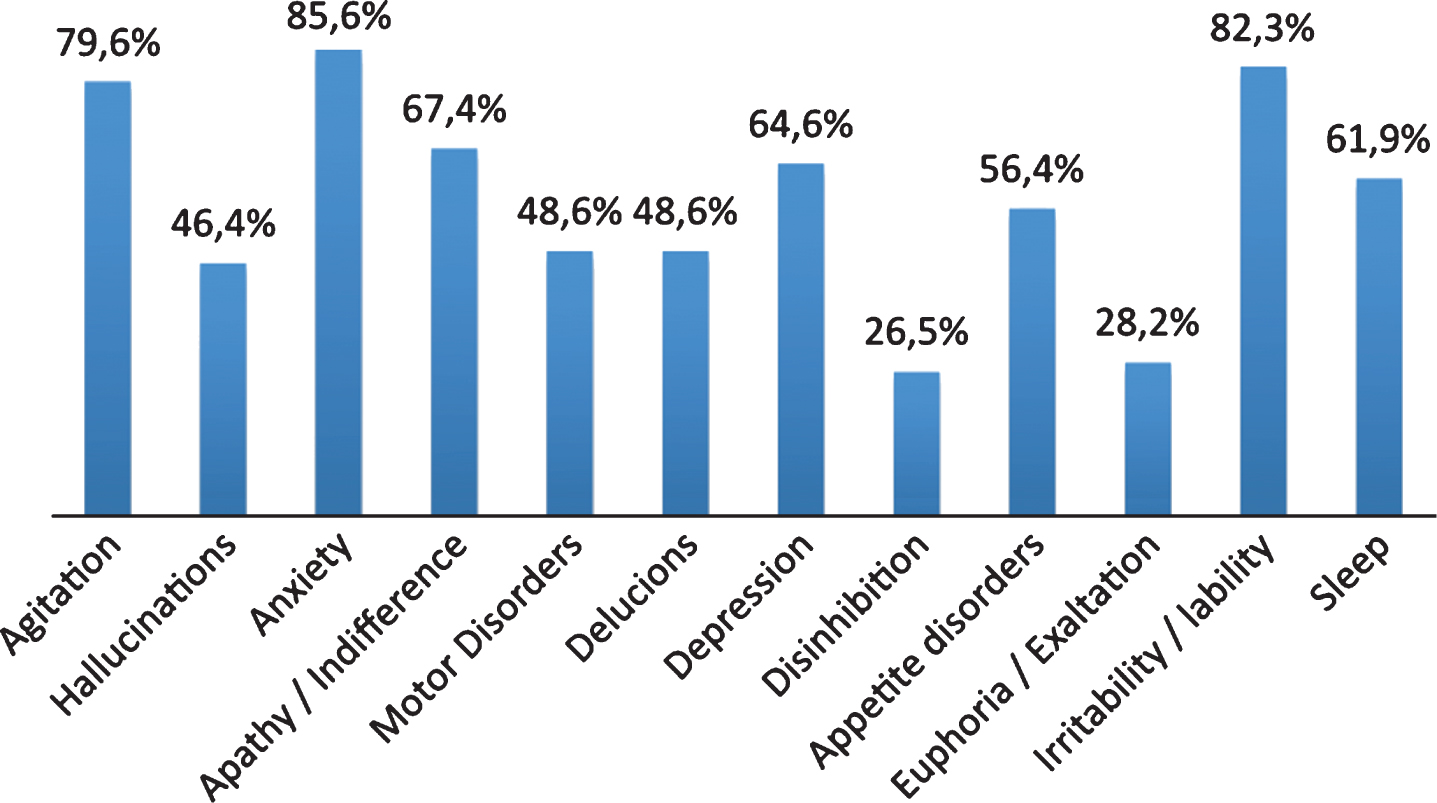
Fig.4
The Multivariate analysis. Statistical significance in the logistic regression model between the irritability/lability (dependent variable) and pain, anxiety and depression (independent variable).
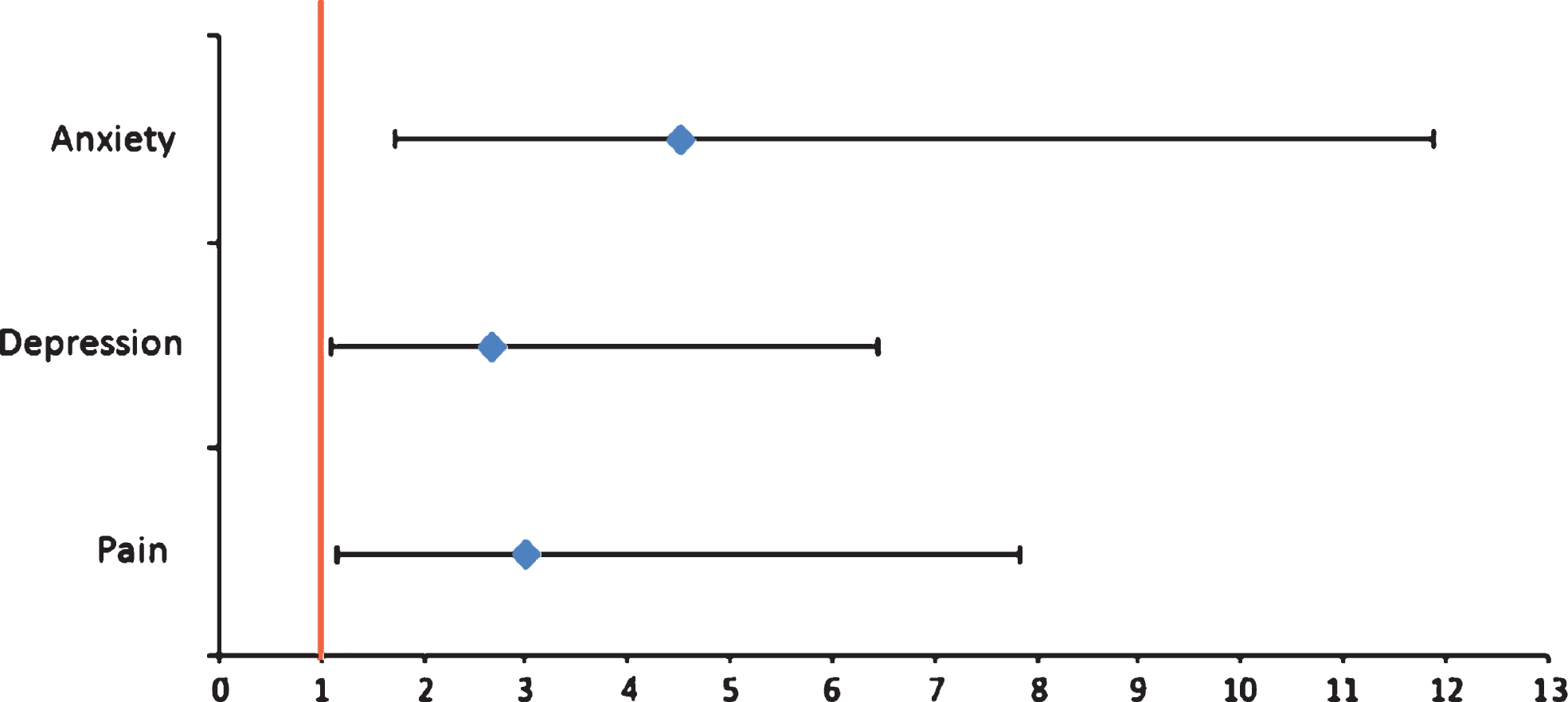
Fig.5
Analgesic drugs administered in people with dementia.
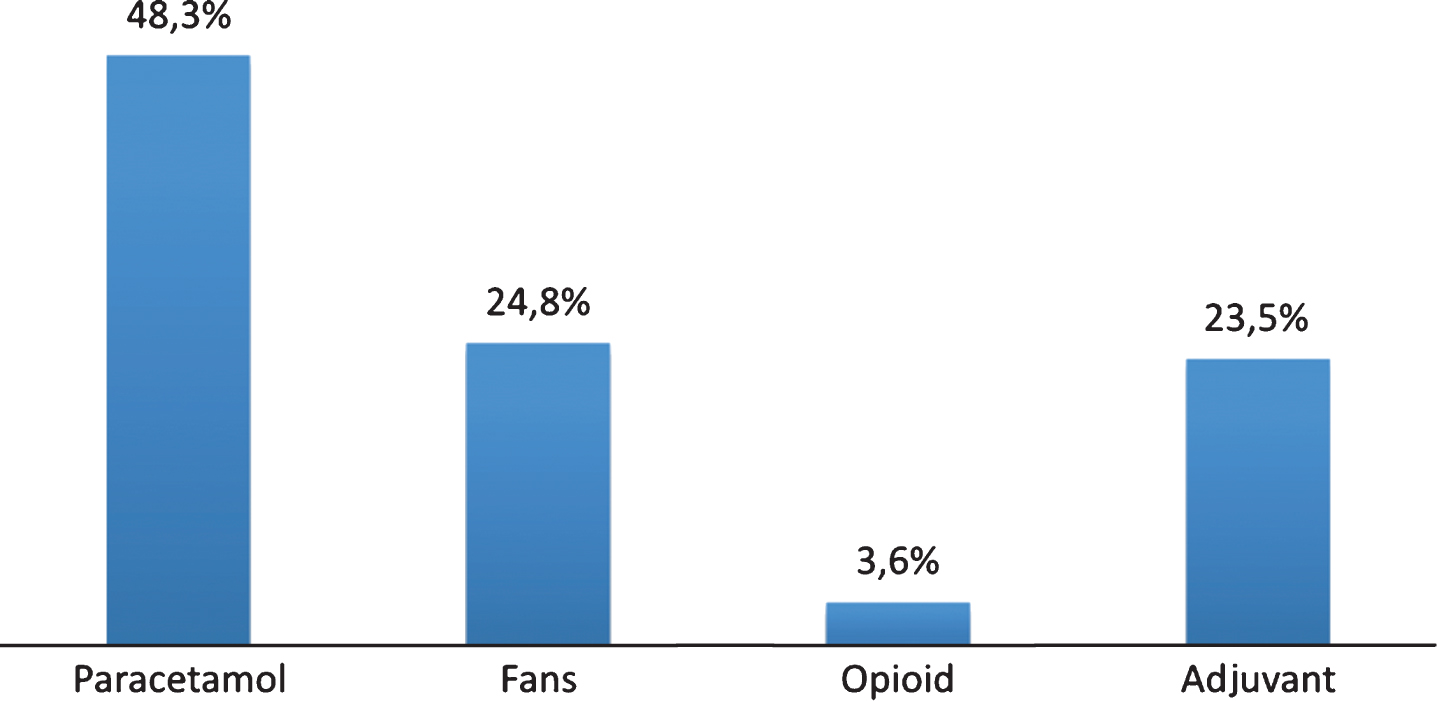
Table 1
Characteristics and measurement of multidimensional evaluation of subjects with dementia
| Subjects | N | (% ) | Mean + SD |
| Gender | |||
| Male (M) | 61 | 33.7 | |
| Female (F) | 120 | 66.3 | |
| Age (y) | |||
| Average M | 80.7±9.3 | ||
| Average F | 85.6±7.3 | ||
| NINCDS-ADRA | |||
| ADPo | 14 | 26.4 | |
| ADPr | 39 | 73.6 | |
| NINCDS-AIREN | |||
| VaDPo | 48 | 40 | |
| VaDPr | 72 | 60 | |
| CIRS | |||
| ICC | 5.3±2.5 | ||
| ICS | 2.1±0.5 | ||
| MMSE | |||
| Severe Cognitive Impairment | 112 | 61.9 | |
| Moderate Cognitive Impairment | 49 | 27.1 | |
| Slight Cognitive Impairment | 20 | 11.1 | |
| CSDD | |||
| Depressive symptoms | 113 | 62.8 | |
| No Depressive symptoms | 67 | 37.2 | |
| ADL | |||
| Functional state severely compromise | 160 | 88.4 | |
| Functional state moderately compromise | 18 | 9.9 | |
| Functional state slightly compromise | 3 | 1.7 | |
| BI | |||
| Total dependence | 83 | 45.9 | |
| Severe dependence | 60 | 33.1 | |
| Moderate dependence | 22 | 12.2 | |
| Mild dependence | 16 | 8.8 | |
| NPI | |||
| Mild behavioral disorders | 140 | 77.3 | |
| Moderate behavioral disorders | 40 | 22.1 | |
| Severe behavioral disorders | 1 | 0.6 | |
| CMAI | |||
| Aggression (F1), | 32 | 20 | |
| Physical non-aggression (F2) | 63 | 39.4 | |
| Verbal agitation (F3) | 65 | 40.6 |
ADPr, Alzheimer’s disease probable; ADPo, Alzheimer’s disease possible; VaDPr, vascular dementia probable; VaDPo, vascular dementia possible; CIRS, Comorbility Illness Rating Scale; ICC, Index of Complex Comorbility; ICS, Index of Severity Comorbility; MMSE, Mini-Mental State Examination; CSDD, Cornell Scale for Depression in Dementia; ADL, Activities of Daily Living; BI, Barthel Index; NPI, Neuropsychiatric Inventory; CMAI, Cohen-Mansfield Agitation Inventory. Data are expressed as N = number of patients and percentages, while for Age and CIRS mean±standard deviation is displayed.
Table 2
Prevalence of pain diagnosis according to ICD9-CM in subjects with dementia
| ICD9-CM Official Code | N | (% ) |
| 338.21 | 14 | 9.3 |
| 338.28 | 5 | 5.9 |
| 338.29 | 51 | 60.7 |
| 338.4 | 7 | 8.3 |
| 780.96 | 7 | 8.3 |
According to Coding Guidelines for Pain, table shows the code for each type of pain. 338.21: Chronic pain due to trauma; 338.28: Other chronic postoperative pain; 338.29: Other chronic pain; 338.4: Chronic pain syndrome; 780.96: Generalized pain (see text for reference). Data are expressed as percentages.
Table 3
The bivariate analysis between diagnosis of pain, depressive and behavioral symptoms
| Diagnosis of Pain (ICD9) | Odds Ratio | 95% | C.I. | Coefficient | E.S. | Z statistic | p value |
| Cornell_Depression | 2.2441 | 1.2007 | 4.1944 | 0.8083 | 0.3191 | 2.5332 | 0.0113* |
| Anxiety | 2.6795 | 1.0657 | 6.7371 | 0.9856 | 0.4704 | 2.0952 | 0.0362* |
| Irritability/Lability | 3.8194 | 1.5567 | 9.3714 | 1.3401 | 0.4579 | 2.9263 | 0.0034*** |
| Agitation | 1.7955 | 0.8483 | 3.8005 | 0.5853 | 0.3826 | 1.5298 | 0.1261 |
| Motor disorders | 1.0143 | 0.5654 | 1.8198 | 0.0142 | 0.2982 | 0.0478 | 0.9619 |
The bivariate analysis showed a statistically significant correlation between diagnosis of pain (ICD9) and depressive symptoms by Cornell Scale for Depression in Dementia (CSDD) and single item of NPI (Anxiety and Irritability/Lability, Agitation, Motor disorders). Level of significance: *p < 0.05, **p < 0.01, ***p < 0.005.
Table 4
Analysis of logistic regression model between pain, depressive and behavioral symptoms
| Aggression (CMAI-F1) | Depression (CSDD) | |||||||||
| Odds Ratio | 95% | C.I. | Coefficient | p value | Odds Ratio | 95% | C.I. | Coefficient | p value | |
| Independent Variable | ||||||||||
| Pain/PAINAD | 3.2571 | 1.284 | 8.2643 | 1.1808 | 0.013* | 2.7129 | 1.333 | 5.52 | 0.998 | 0.006** |
| Covariate | ||||||||||
| Agitation | 3.6987 | 0.455 | 30.0898 | 1.308 | 0.221 | 3.5626 | 1.416 | 8.964 | 1.2705 | 0.007** |
| Anxiety | 1123217.096 | 0 | >1.0E12 | 13.932 | 0.972 | 1.8348 | 0.58 | 5.804 | 0.607 | 0.302 |
Statistical significance in the logistic regression model between pain, assessed by PAINAD, depressive symptoms by CSDD and Aggression by CMAI (profile F1). Level of significance: *p < 0.05, **p < 0.01, ***p < 0.005.
Table 5
Analysis of logistic regression model pain, anxiety, depressive symptoms and drugs
| Variable | Odds Ratio | 95% | C.I. | Coefficient | E.S. | Z Statistic | p value |
| Anxiety | 4.9836 | 1.8506 | 13.4209 | 1.6062 | 0.5054 | 3.1777 | 0.0015*** |
| Depression | 2.8429 | 1.1725 | 6.8929 | 1.0448 | 0.4519 | 2.3122 | 0.0208* |
| Pain | 3.1747 | 1.2068 | 8.3516 | 1.1552 | 0.4935 | 2.3408 | 0.0192* |
| Drugs | 0.6599 | 0.2484 | 1.7531 | –0.4157 | 0.4985 | –0.8339 | 0.4043 |
Statistical significance in the logistic regression model between the Irritability/Lability (dependent variable) and Anxiety, Depression, Pain and Drugs (independent variable). The use of antipsychotics, anxiolytics and sedative/hypnotic drugs does not correlate with Irritability/Lability. Level of significance: *p < 0.05, **p < 0.01, ***p < 0.005.




