Vitamin D receptor polymorphisms associate with the efficacy and toxicity of radioiodine-131 therapy in patients with differentiated thyroid cancer
Abstract
BACKGROUND:
Radioiodine-131 (I-131) therapy is the common postoperative adjuvant therapy for differentiated thyroid cancer (DTC) However, methods to evaluate the efficacy and toxicity of I-131 on DTC are still lacking.
OBJECTIVE:
To evaluate the association between vitamin D receptor (VDR) gene polymorphisms and the efficacy and toxicity of I-131 in DTC patients.
METHODS:
A total of 256 DTC patients who received I-131 therapy were enrolled. The patients were divided into effective group and ineffective group. 4 single nucleotide polymorphisms (SNPs) (rs7975232, rs731236, rs1544410 and rs10735810) of VDR were analyzed by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) Cell counting kit-8 (CCK-8) and flow cytometry were used to detect the proliferation and apoptosis of thyroid cancer cells.
RESULTS:
Patients in effective group had more CC genotype of rs7975232 and GG genotype of rs10735810 compared with patients in ineffective group They were also independent factors for influencing the efficacy of I-131. PTC-1 and FTC-133 cells transfected with CC genotype of rs7975232 showed lower proliferative activity and higher apoptosis rate after being treated with I-131 In addition, patients with CC genotype at rs7975232 had fewer adverse reactions after I-131 treatment.
CONCLUSIONS:
VDR gene polymorphisms may be associated with the efficacy and toxicity of I-131 in DTC patients, which will help to personalize the treatment for patients.
1.Introduction
Thyroid cancer is a common malignant tumor of the neck, which mainly originates from thyroid follicular epithelial cells. Thyroid cancer is divided into differentiated thyroid cancer (DTC), squamous carcinoma, undifferentiated carcinoma, medullary carcinoma, of which the incidence of DTC accounts for more than 90% of the thyroid cancer [1, 2]. In the past decades, the mortality rate of thyroid cancer in China has remained the highest in the world [3]. Radioiodine-131 (I-131) can be absorbed by thyroid cancer tissues with high specificity The high-energy
At present, the pathogenesis and progression of thyroid cancer are still under discussion. Some scholars believed that chemical toxins, insulin resistance, metabolic syndrome, and vitamin D deficiency were potential risk factors for thyroid cancer [6, 7, 8]. The active form of vitamin D, 1,25-(OH)2D3, mainly binds to the nuclear vitamin D receptor (VDR) and controls the expression of more than 200 genes to exert physiological functions [9]. Previous studies have shown that VDR was closely related to thyroid cancer. The expression of VDR was increased in DTC and benign thyroid tumors compared with normal thyroid [10]. Some researchers also found that VDR knockdown could attenuate the anti-proliferation, pro-apoptosis and anti-invasion effects of vitamin D in papillary thyroid carcinoma (PTC) by activating the Wnt/
In this study, 4 key SNPs, rs7975232, rs731236, rs1544410 and rs10735810 were selected to analyze the effects of these SNPs on the clinical efficacy and side effects of I-131 in patients with DTC. The aim of this study is to provide more theoretical reference for I-131 therapy.
2.Materials and methods
2.1 Patient enrollment
All participants in this study were from the Chinese Han population. A total of 256 patients with DTC were diagnosed by pathological examination in Chenzhou first people’s Hospital. These patients underwent residual ablation of I-131 at hospital (dose of 100 mci), including 251 patients with PTC and 5 patients with FTC, 53 males and 203 females. Informed consent was obtained from hospitalized patients or their guardians, and the ethics committee of Chenzhou first people’s Hospital approved this study.
Exclusion criteria for this study were: pregnancy, hepatic or renal insufficiency, or concurrent malignancy Patients with distant metastasis. A history of radiotherapy or chemotherapy. No history of clinical examination [16].
2.2Evaluation of postoperative I-131 efficacy
Efficacy evaluation criteria [16]: Approximately 6 months after treatment with I-131, patients discontinued thyroid hormone and a low-iodine diet for 2–4 weeks and thyroid uptake of I-131 was measured. If no spots were found in the thyroid bed (I-131 uptake), the thyroid uptake rate of I-131 after 24 hours of I-131 treatment was
2.3Genotyping
In the morning, 2 ml of fasting elbow venous blood was collected into anticoagulant tubes containing ethylene diamine tetraacetic acid (EDTA). According to the DNA extraction kit (cat.no. 69504; Qiagen, Hilden, Germany) to extract genomic DNA. The genotypes of 4 SNPs (rs7975232, rs731236, rs1544410 and rs10735810) in VDR gene were analyzed by polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) [16].
2.4Cell culture
TPC-1 and FTC-133 human thyroid cancer cell lines were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). The Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; cat. no. 11965092; Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; cat. no. 16140089; Gibco) and 100 mg/ml streptomycin (cat. no. ST487; Beyotime, Shanghai, China) and 100 IU/ml penicillin (cat. no. ST486; Beyotime) at 37∘C in a 5% CO2 incubator.
2.5Plasmid construction and transfection
Referring to the methods described by previous [17]. A VDR promoter region containing the polymorphisms rs7975232 and rs10735810 were obtained by PCR. PCR products containing the VDR polymorphism were digested with Bgl II (cat. no. ER0081; Thermo Fisher Scientific, Waltham, MA, USA) and Kpn I (cat. no. ER0521; Thermo Fisher Scientific) and ligated into the vector pGL3-basic (cat. no. HG-VQP0121; HonorGene, Changsha, Hunan Province, China) to construct the rs7975232 recombinant plasmids (vector-AA and vector-CC) and rs10735810 recombinant plasmids (vector-AA and vector-GG). Lipofectamine 2000 (cat. no. 11668019; Thermo Fisher Scientific) were transfected into TPC-1 and FTC-133 cells with vectors.
2.6Cell counting kit-8 (CCK-8) assay
CCK-8 solution (cat. no. ab228554, Abcam, Burlin-game, CA, USA) was recommended to assess cell viability of TPC-1 and FTC-133 cells. The cells were inoculated into 96-well plates at a density of 1
2.7Flow cytometry
Flow cytometry was performed by using Alexa Fluor 488 Annexin V/Dead Cell Apoptosis Kit (cat. no. V13241, Thermo Fisher Scientific) following manufacturer’s instructions. TPC-1 and FTC-133 cells were exposed to 1 mCi I-131 for 12 h. For cell apoptosis analysis, cells were harvested and fixed in pre-cold 70% ethanol at 4∘C overnight. The cells were stained with Annexin V-FITC and propidium iodide (PI) and subsequently the ratio of apoptotic cells was tested by flow cytometry (BD FACS Calibur, Becton Dickinson, Franklin Lake, New Jersey, USA). Each experiment was repeated three times.
2.8Statistical analysis
Visualization and analysis of the data were performed using GraphPad Prism 6.0. An F-test was performed for normality of the measurement data, and a
Table 1
Clinical baseline information of DTC patients
| Characteristics | Effective group ( | Ineffective group ( | |
|---|---|---|---|
| Gender (male, %) | 33 (21.57) | 20 (19.42) | 0.677 |
| Age (mean | 43.64 | 42.17 | 0.335 |
| BMI | |||
| 18.5 | 11 (7.19) | 8 (7.77) | 0.084 |
| 18.5–24 | 88 (57.51) | 45 (43.69) | |
| 24 | 54 (35.30) | 50 (48.54) | |
| Serum TSH levels before therapy (mean | 79.38 | 68.76 | 0.004 |
| Serum TG levels before therapy (mean | 16.11 | 17.03 | 0.038 |
| Types of pathology | |||
| PTC | 150 (98.04) | 101 (98.06) | 0.991 |
| FTC | 3 (1.96) | 2 (1.94) | |
| TNM | |||
| Stage I | 125 (81.70) | 66 (64.08) | 0.006 |
| Stage II | 11 (7.19) | 13 (12.62) | |
| Stage III | 17 (11.11) | 24 (23.30) |
BMI, Body mass index; TSH, Thyroid stimulating hormone; TG, Thyroglobulin; PTC, Papillary thyroid carcinoma; FTC, Follicular thyroid carcinoma; TNM, Tumor node metastasis.
3.Results
3.1Clinical baseline information
The basic characteristics of all patients are summarized in Table 1, including 153 patients in the effective group and 103 patients in the ineffective group. There were no significant differences in gender, age, body mass index (BMI) and pathological type between the two groups (
3.2HWE test
The distribution of the genotypes of the 4 polymorphism sites of VDR in the study samples was tested by HWE test. The
Table 2
Association between polymorphisms of VDR gene and I-131 efficacy
| SNPs | Effective group ( | Ineffective group ( | OR | 95% CI | ||
|---|---|---|---|---|---|---|
| rs7975232 | ||||||
| AA | 7 (4.58) | 12 (11.65) | ||||
| AC | 50 (32.68) | 46 (44.66) | 0.225 | 0.899 | 0.537 | 0.195–1.480 |
| CC | 96 (62.74) | 45 (43.69) | 0.008 | 0.030 | 0.273 | 0.101–0.741 |
| CC | 146 (95.42) | 91 (88.35) | 0.034 | 0.136 | 0.364 | 0.138–0.957 |
| A | 64 (20.92) | 70 (33.98) | ||||
| C | 242 (79.08) | 136 (66.02) | 0.001 | 0.004 | 0.514 | 0.345–0.766 |
| rs731236 | ||||||
| GG | 1 (0.65) | 1 (0.97) | ||||
| AG | 17 (11.11) | 18 (17.48) | 0.968 | 3.874 | 1.059 | 0.061–18.30 |
| AA | 135 (88.24) | 84 (81.55) | 0.736 | 2.945 | 0.622 | 0.038–10.08 |
| AA | 152 (99.35) | 102 (99.03) | 0.777 | 3.110 | 0.671 | 0.042–10.85 |
| G | 19 (6.21) | 20 (9.71) | ||||
| A | 287 (93.79) | 186 (90.29) | 0.143 | 0.573 | 0.616 | 0.320–1.185 |
| rs1544410 | ||||||
| TT | 2 (1.31) | 1 (0.97) | ||||
| CT | 30 (19.61) | 20 (19.42) | 0.817 | 3.274 | 1.333 | 0.113–15.70 |
| CC | 121 (79.08) | 82 (79.61) | 0.805 | 3.218 | 1.355 | 0.121-15.19 |
| CC | 151 (98.69) | 102 (99.03) | 0.806 | 3.225 | 1.351 | 0.121–15.10 |
| T | 34 (11.11) | 22 (10.68) | ||||
| C | 272 (88.89) | 184 (89.32) | 0.878 | 3.512 | 1.045 | 0.592–1.845 |
| rs10735810 | ||||||
| AA | 22 (14.38) | 26 (25.24) | ||||
| AG | 72 (47.06) | 51 (49.52) | 0.134 | 0.534 | 0.599 | 0.306–1.173 |
| GG | 59 (38.56) | 26 (25.24) | 0.007 | 0.30 | 0.371 | 0.179–0.775 |
| GG | 131 (85.62) | 77 (74.76) | 0.029 | 0.116 | 0.497 | 0.264–0.937 |
| A | 116 (37.91) | 103 (50.00) | ||||
| G | 190 (62.09) | 103 (50.00) | 0.007 | 0.027 | 0.611 | 0.427–0.873 |
*Represents the
Table 3
Logistic regression analysis of factors associated with efficacy of I-131 in DTC patients
| Factor | B | SE | OR | 95%CI | |
|---|---|---|---|---|---|
| Serum TSH levels before therapy | 0.013 | 0.005 | 0.987 | 0.978–0.997 | 0.009 |
| Serum TG levels before therapy | 0.082 | 0.041 | 1.086 | 1.003–1.175 | 0.042 |
| TNM stage | 0.547 | 0.181 | 1.728 | 1.211–2.464 | 0.003 |
| rs7975232 | 0.567 | 0.221 | 0.567 | 0.367–0.875 | 0.010 |
| rs10735810 | 0.388 | 0.197 | 0.678 | 0.461–0.998 | 0.049 |
B, Bias; SE, Standard error; OR, Odds ratio; CI, Confidence interval; TSH, Thyroid-stimulating hormone; TG, Thyroglobulin; TNM, Tumor node metastasis.
3.3Association between polymorphisms of VDR gene and I-131 efficacy
The results of the analyses between VDR gene polymorphisms and I-131 efficacy were shown in Table 2. The distribution of CC genotype at rs7975232 was significantly increased in the effective group (CC vs AA,
3.4Logistic regression analysis of factors associated with the efficacy of I-131 treatment of DTC patients
To further clarify the independent factors influencing the efficacy of I-131, we performed multifactorial regression analyses with characteristics that differed in univariate analyses (patients’ pre-therapy serum TG and TSH levels, TNM stage, rs7975232 polymorphisms, and rs10735810) as independent variables and treatment outcome (effective or ineffective) as the dependent variable. The results showed that all of the above dependent variables were significantly associated with the efficacy of I-131 treatment (all
Figure 1.
The effect of rs7975232 on proliferation and apoptosis of thyroid cancer cells. (A–B) The proliferation activity of TPC-1 (A) and FTC-133 (B) cells in different treatment groups (rs7975232) was analyzed via CCK-8 assay. Data are represented as mean
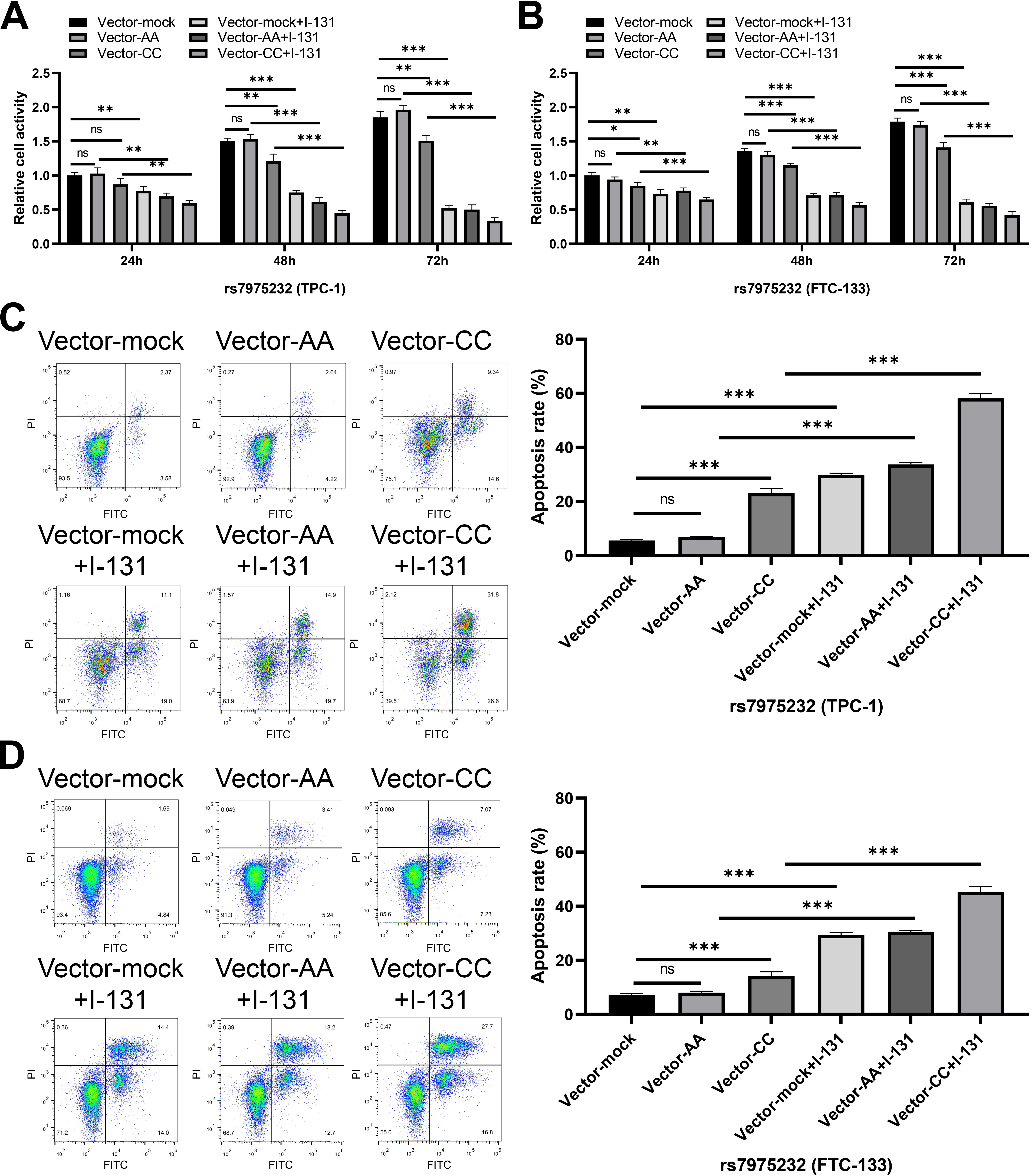
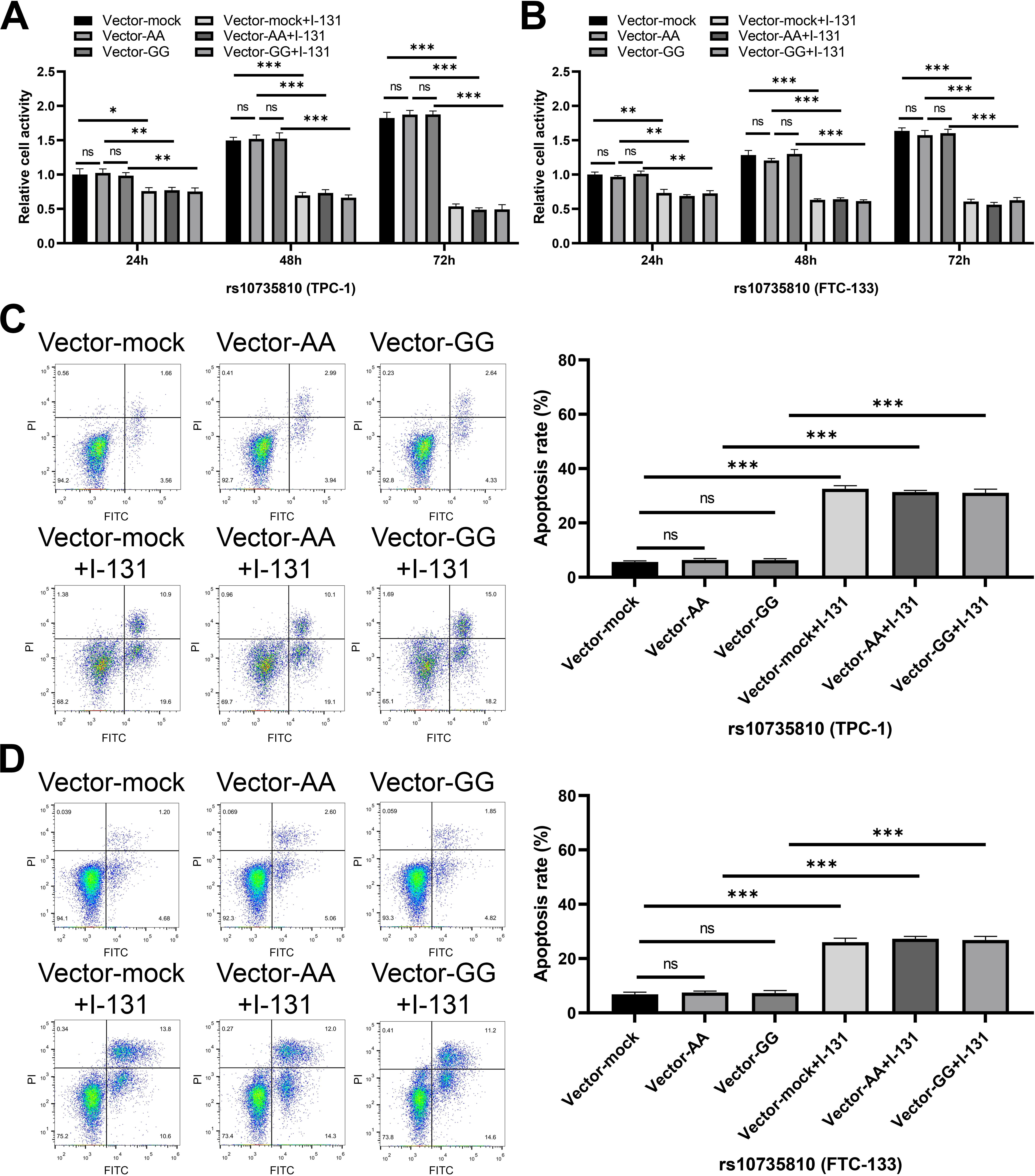
Figure 2.
The effect of rs10735810 on proliferation and apoptosis of thyroid cancer cells. (A–B) The proliferation activity of TPC-1 (A) and FTC-133 (B) cells in different treatment groups (rs10735810) was analyzed via CCK-8 assay. Data are represented as mean
3.5Proliferation and apoptotic ability of transfected TPC-1 and FTC-133 cells
In order to further clarify the effect of rs7975232 and rs10735810 on the biological function of thyroid cancer cells, functional tests were performed on transfected TPC-1 and FTC-133 cells. The results of CCK-8 assay showed that with the extension of cell culture time, the proliferation activity of TPC-1 and FTC-133 cells in CC genotype group (rs7975232) was significantly reduced compared with that in AA genotype group (Fig. 1A–B). At the same time, no matter the mock group, AA genotype group or CC genotype group, the cell proliferation activity was significantly down-regulated after I-131 treatment (Fig. 1A–B). On the other hand, AA and GG genotypes of rs10735810 could not regulate the proliferation of TPC-1 and FTC-133 cells (Fig. 2A–B). The effect of rs7975232 and rs10735810 on the apoptosis level of TPC-1 and FTC-133 cells was further explored. Apoptosis experiment also showed that the apoptosis rate of AA genotype in TPC-1 and FTC-133 cells was not significantly different from that of control cells (Fig. 1C–D). Compared with AA genotype, CC genotype could enhance the apoptosis rate of TPC-1 and FTC-133 cells (Fig. 1C–D). After exposure to I-131, apoptosis rate of thyroid cancer cells in 3 groups was significantly up-regulated (Fig. 1C–D). On the other hand, AA and GG genotypes of rs10735810 could not regulate the apoptosis rate of TPC-1 and FTC-133 cells (Fig. 2C–D). These results suggest that CC genotype of rs7975232 can inhibit the proliferation of thyroid cancer cell, which may promote the therapeutic effect of I-131.
Table 4
Relationship between polymorphisms of VDR gene and I-131 toxicity
| SNPs | Effective group | Ineffective group | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| Neck discomfort | SG side effects | GI symptoms | Total adverse reaction |
| Neck discomfort | SG side effects | GI symptoms | Total adverse reaction | |||
| rs7975232 | ||||||||||||
| AA | 7 | 2 | 0 | 1 | 3 | 0.005 | 12 | 4 | 2 | 2 | 8 | |
| AC | 50 | 4 | 0 | 1 | 5 | 46 | 2 | 1 | 1 | 4 | ||
| CC | 96 | 3 | 1 | 2 | 6 | 45 | 1 | 1 | 1 | 3 | ||
| rs731236 | ||||||||||||
| GG | 1 | 0 | 0 | 0 | 0 | 0.419 | 1 | 0 | 0 | 0 | 0 | 0.205 |
| AG | 17 | 2 | 0 | 1 | 3 | 18 | 3 | 1 | 1 | 5 | ||
| AA | 135 | 7 | 1 | 3 | 11 | 84 | 4 | 3 | 3 | 10 | ||
| rs1544410 | ||||||||||||
| TT | 2 | 1 | 0 | 0 | 1 | 0.079 | 1 | 0 | 0 | 0 | 0 | 0.917 |
| CT | 30 | 2 | 1 | 1 | 4 | 20 | 2 | 0 | 1 | 3 | ||
| CC | 121 | 6 | 0 | 3 | 9 | 82 | 5 | 4 | 3 | 12 | ||
| rs10735810 | ||||||||||||
| AA | 22 | 1 | 0 | 1 | 2 | 0.694 | 26 | 1 | 1 | 1 | 3 | 0.680 |
| AG | 72 | 5 | 1 | 2 | 8 | 51 | 4 | 3 | 2 | 9 | ||
| GG | 59 | 3 | 0 | 1 | 4 | 26 | 2 | 0 | 1 | 3 | ||
SNPs, Single nucleotide polymorphisms; SG, Salivary gland; GI, Gastrointestinal.
3.6Relationship between polymorphisms of VDR gene and I-1311 toxicity
In order to further explore whether the toxic side effects after I-131 treatment were related to VDR gene polymorphism. We collected different adverse reactions of patients, which mainly manifested as neck discomfort, gastrointestinal side effects, and salivary gland side effects. The results showed (Table 4) that patients with rs7975232 CC genotype had less adverse events (
4.Discussion
A total of 256 DTC patients who received postoperative I-131 ablation therapy were enrolled in this study. The patients were divided into effective group and ineffective group according to serum TG, TSH and imaging results. The results showed that patients with CC genotype at rs7975232 and GG genotype at rs10735810 were more sensitive to I-131 treatment. Biological experiments also verified the above conclusions, that the proliferation activity of cancer cells transfected with rs7975232 CC genotype was significantly decreased after I-131 treatment, while the level of apoptosis was significantly increased. In terms of adverse reactions, patients with CC genotype at rs7975232 locus have fewer adverse reactions after I-131 treatment. The results of this study for clinical screening potential future patients benefit from the I-131 provides a certain theoretical basis.
I-131 therapy is a commonly used adjuvant treatment for patients with DTC after resection, which can effectively remove residual thyroid tissue and play a crucial role in improving the clinical treatment effect and prognosis of patients [18]. However, some patients do not respond to I-131 therapy in clinical practice. Some reports have suggested that the efficacy of I-131 is associated with SNPs in some genes. Zhang and his partners found that cytotoxic T lymphocyte-associated antigen-4 (CTLA-4) gene polymorphisms were associated with the efficacy of I-131 in DTC patients, with a significantly greater distribution of AG
Because of the ligand-dependent nature of VDR, its biological effects are affected by gene expression, so some VDR gene polymorphisms may affect the binding and biological activity of vitamin D and VDR [27, 28]. Previous studies have indicated that the system on low VDR expression, can activate the Wnt/
In addition, although I-131 can eliminate residual thyroid tissue, its
Despite the above findings, this study has some shortcomings. Although we found through biological experiments that VDR polymorphism does affect the proliferation and apoptosis of thyroid cancer cells, the specific mechanism of the relationship between VDR polymorphism and DTC needs to be further explored In addition, this study demonstrated that CC genotype of rs7975232 could inhibit the proliferation of thyroid cancer cell in vitro. However, this conclusion has not been confirmed in vivo. Finally, the conclusions of this study are based on a small sample size and have not been validated in other sample sets or data sets. Therefore, it is necessary to verify the conclusions of this study in other sample sets in future studies.
5.Conclusion
In conclusion, CC genotype of rs7975232 and GG genotype of rs10735810 were significantly associated with better I-131 efficacy in DTC patients after surgery, and cancer cells transfected with CC genotype of rs7975232 were more sensitive to I-131 treatment. This may be of great value in evaluating the efficacy of I-131. In addition, in terms of adverse reactions, patients with CC genotype at rs7975232 locus had fewer adverse reactions after I-131 treatment. It also suggests that VDR polymorphism can be used as a potential target to predict the efficacy and toxicity of I-131 in DTC patients, which can be applied to clinical practice in the future to improve the clinical efficacy and reduce the toxicity of patients with thyroid cancer.
Author contributions
Conception: Yi Zhang. Interpretation or analysis of data: Yuanhong Deng and Ying Fu. Preparation of the manuscript: Yuanhong Deng, Ying Fu and Ganghua Feng. Revision for important intellectual content: Yuanhong Deng and Yi Zhang. Supervision: Yi Zhang.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical approval and consent to participate
The experiments were approved by the ethics committee of Chenzhou first people’s Hospital (Approval No. 2023014).
Consent for publication
Not applicable.
Acknowledgments
Not applicable.
Conflict of interest
The authors declare no conflict of interest.
References
[1] | J.A. Fagin and S.A. Wells, Jr., Biologic and Clinical Perspectives on Thyroid Cancer, N Engl J Med 375: ((2016) ), 1054–67. |
[2] | K.A. Araque, S. Gubbi and J. Klubo-Gwiezdzinska, Updates on the Management of Thyroid Cancer, Horm Metab Res 52: ((2020) ), 562–577. |
[3] | Y. Deng, H. Li, M. Wang, N. Li, T. Tian, Y. Wu, P. Xu, S. Yang, Z. Zhai, L. Zhou, Q. Hao, D. Song, T. Jin, J. Lyu and Z. Dai, Global Burden of Thyroid Cancer From 1990 to 2017, JAMA Netw Open 3: ((2020) ), e208759. |
[4] | B.R. Haugen, E.K. Alexander, K.C. Bible, G.M. Doherty, S.J. Mandel, Y.E. Nikiforov, F. Pacini, G.W. Randolph, A.M. Sawka, M. Schlumberger, K.G. Schuff, S.I. Sherman, J.A. Sosa, D.L. Steward, R.M. Tuttle and L. Wartofsky, 2015 American Thyroid Association Management Guidelines for Adult Patients with Thyroid Nodules and Differentiated Thyroid Cancer: The American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer, Thyroid 26: ((2016) ), 1–133. |
[5] | A. Ciarallo and J. Rivera, Radioactive Iodine Therapy in Differentiated Thyroid Cancer: 2020 Update, AJR Am J Roentgenol 215: ((2020) ), 285–291. |
[6] | B. Biondi, D. Arpaia, P. Montuori, G. Ciancia, S. Ippolito, G. Pettinato and M. Triassi, Under the shadow of vesuvius: a risk for thyroid cancer, Thyroid 22: ((2012) ), 1296–7. |
[7] | M. Roskies, Y. Dolev, D. Caglar, M.P. Hier, A. Mlynarek, A. Majdan and R.J. Payne, Vitamin D deficiency as a potentially modifiable risk factor for thyroid cancer, J Otolaryngol Head Neck Surg 41: ((2012) ), 160–3. |
[8] | W. Borena, T. Stocks, H. Jonsson, S. Strohmaier, G. Nagel, T. Bjørge, J. Manjer, G. Hallmans, R. Selmer, M. Almquist, C. Häggström, A. Engeland, S. Tretli, H. Concin, A. Strasak, P. Stattin and H. Ulmer, Serum triglycerides and cancer risk in the metabolic syndrome and cancer (Me-Can) collaborative study, Cancer Causes Control 22: ((2011) ), 291–9. |
[9] | P. Kmieć and K. Sworczak, Vitamin D in thyroid disorders, Exp Clin Endocrinol Diabetes 123: ((2015) ), 386–93. |
[10] | I. Clinckspoor, E. Hauben, L. Verlinden, A. Van den Bruel, L. Vanwalleghem, V. Vander Poorten, P. Delaere, C. Mathieu, A. Verstuyf and B. Decallonne, Altered expression of key players in vitamin D metabolism and signaling in malignant and benign thyroid tumors, J Histochem Cytochem 60: ((2012) ), 502–11. |
[11] | R. Pang, Y. Xu, X. Hu, B. Liu and J. Yu, Vitamin D receptor knockdown attenuates the antiproliferative, pro-apoptotic and anti-invasive effect of vitamin D by activating the Wnt/β-catenin signaling pathway in papillary thyroid cancer, Mol Med Rep 22: ((2020) ), 4135–4142. |
[12] | M. Penna-Martinez, E. Ramos-Lopez, J. Stern, N. Hinsch, M.L. Hansmann, I. Selkinski, F. Grünwald, C. Vorländer, R.A. Wahl, W.O. Bechstein, S. Zeuzem, K. Holzer and K. Badenhoop, Vitamin D receptor polymorphisms in differentiated thyroid carcinoma, Thyroid 19: ((2009) ), 623–8. |
[13] | A. Maciejewski and K. Lacka, Vitamin D-Related Genes and Thyroid Cancer-A Systematic Review, Int J Mol Sci 23: ((2022) ). |
[14] | A.M. Cocolos, A. Muresan, A. Caragheorgheopol, M. Ghemigian, D. Ioachim and C. Poiana, Vitamin D Status and VDR Polymorphisms as Prognostic Factors in Differentiated Thyroid Carcinoma, In Vivo 36: ((2022) ), 2434–2441. |
[15] | M.U.N. Iqbal and T.A. Khan, Association between Vitamin D receptor (Cdx2, Fok1, Bsm1, Apa1, Bgl1, Taq1, and Poly (A)) gene polymorphism and breast cancer: A systematic review and meta-analysis, Tumour Biol 39: ((2017) ), 1010428317731280. |
[16] | D.F. Chang, X.H. Chen, J. Huang, Y.M. Sun, D.Y. Zhu and Z.Q. Xu, CTLA-4 gene polymorphisms associate with efficacy of postoperative radioiodine-131 for differentiated thyroid carcinoma, Future Oncol 13: ((2017) ), 1057–1068. |
[17] | Y. Chen, R. Lu, H. Zheng, R. Xiao, J. Feng, H. Wang, X. Gao and L. Guo, The NFKB1 polymorphism (rs4648068) is associated with the cell proliferation and motility in gastric cancer, BMC Gastroenterol 15: ((2015) ), 21. |
[18] | P. Kumar, N.A. Damle, S. Agarwala, S.N. Dwivedi and C. Bal, Individualized dosimetry in children and young adults with differentiated thyroid cancer undergoing iodine-131 therapy, J Pediatr Endocrinol Metab ((2020) ). |
[19] | Retraction, Future Oncol 16: ((2020) ), 613. |
[20] | X.R. Han, X. Wen, S. Wang, S.H. Fan, J. Zhuang, Y.J. Wang, Z.F. Zhang, M.Q. Li, B. Hu, Q. Shan, C.H. Sun, Y.X. Bao, D.M. Wu, J. Lu and Y.L. Zheng, Correlations of CTLA-4 exon-1 49 A/G and promoter region 318C/T polymorphisms with the therapeutic efficacy of (131) I radionuclide in graves’ disease in Chinese Han population, J Cell Biochem 119: ((2018) ), 6383–6390. |
[21] | H. Zhong, R. Zhou, Y. Feng, G.X. Zheng, Y. Liang, J.Y. Zhang, X.Q. Qin, W. Chen, J.Q. Wu and Y.H. Zhong, Association of vitamin D receptor gene polymorphism with the risk of lung cancer: a meta-analysis, J Recept Signal Transduct Res 34: ((2014) ), 500–5. |
[22] | J. Li, S. Qin, S. Zhang, Y. Lu, Q. Shen, L. Cheng and R. Zhong, Serum vitamin D concentration, vitamin D-related polymorphisms, and colorectal cancer risk, Int J Cancer 153: ((2023) ), 278–289. |
[23] | L. Perna, K. Butterbach, U. Haug, B. Schöttker, H. Müller, V. Arndt, B. Holleczek, B. Burwinkel and H. Brenner, Vitamin D receptor genotype rs731236 (Taq1) and breast cancer prognosis, Cancer Epidemiol Biomarkers Prev 22: ((2013) ), 437–42. |
[24] | P. Gapska, R.J. Scott, P. Serrano-Fernandez, T. Huzarski, T. Byrski, J. Kładny, J. Gronwald, B. Górski, C. Cybulski, J. Lubinski and T. Debniak, Vitamin D receptor variants and breast cancer risk in the Polish population, Breast Cancer Res Treat 115: ((2009) ), 629–33. |
[25] | S. Beysel, N. Eyerci, F.A. Pinarli, M. Apaydin, M. Kizilgul, M. Caliskan, O. Ozcelik, S. Kan and E. Cakal, VDR gene FokI polymorphism as a poor prognostic factor for papillary thyroid cancer, Tumour Biol 40: ((2018) ), 1010428318811766. |
[26] | J.B. Pao, Y.P. Yang, C.N. Huang, S.P. Huang, T.C. Hour, T.Y. Chang, Y.H. Lan, T.L. Lu, H.Z. Lee, S.H. Juang, C.Y. Huang and B.Y. Bao, Vitamin D receptor gene variants and clinical outcomes after androgen-deprivation therapy for prostate cancer, World J Urol 31: ((2013) ), 281–7. |
[27] | H. Sun, S.R. Long, X. Li, H. Ge, X. Liu, T. Wang, F. Yu, Y. Wang, Y. Xue and W. Li, Serum vitamin D deficiency and vitamin D receptor gene polymorphism are associated with increased risk of cardiovascular disease in a Chinese rural population, Nutr Res 61: ((2019) ), 13–21. |
[28] | A.G. Uitterlinden, Y. Fang, J.B. Van Meurs, H.A. Pols and J.P. Van Leeuwen, Genetics and biology of vitamin D receptor polymorphisms, Gene 338: ((2004) ), 143–56. |
[29] | Y. Ling, F. Xu, X. Xia, D. Dai, R. Sun and Z. Xie, Vitamin D receptor regulates proliferation and differentiation of thyroid carcinoma via the E-cadherin-β-catenin complex, J Mol Endocrinol 68: ((2022) ), 137–151. |
[30] | T. Zhang, L. He, Z. Wang, W. Dong, W. Sun, Y. Qin, P. Zhang and H. Zhang, Calcitriol enhances Doxorubicin-induced apoptosis in papillary thyroid carcinoma cells via regulating VDR/PTPN2/p-STAT3 pathway, J Cell Mol Med 24: ((2020) ), 5629–5639. |
[31] | L. Yang, J. Ma, P. Lei, J. Yi, Y. Ma, Z. Huang, T. Wang, H. Ping, D. Ruan, D. Sun and H. Pan, Advances in Antioxidant Applications for Combating (131)I Side Effects in Thyroid Cancer Treatment, Toxics 11: ((2023) ). |
[32] | J. Liu, X. Tang, F. Shi, C. Li, K. Zhang, J. Liu, G. Wang, J. Yin and Z. Li, Genetic polymorphism contributes to (131)I radiotherapy-induced toxicities in patients with differentiated thyroid cancer, Pharmacogenomics 19: ((2018) ), 1335–1344. |
[33] | I.S. Carvalho, C.I. Gonçalves, J.T. Almeida, T. Azevedo, T. Martins, F.J. Rodrigues and M.C. Lemos, Association of Vitamin D Pathway Genetic Variation and Thyroid Cancer, Genes (Basel) 10: ((2019) ). |




