Elevated expression patterns of P-element Induced Wimpy Testis (PIWI) transcripts are potential candidate markers for Hepatocellular Carcinoma
Abstract
BACKGROUND:
P-Element-induced wimpy testis (PIWI) proteins, when in combination with PIWI-interacting RNA (piRNA), are engaged in the epigenetic regulation of gene expression in germline cells. Different types of tumour cells have been found to exhibit abnormal expression of piRNA, PIWIL-mRNAs, and proteins. We aimed to determine the mRNA expression profiles of PIWIL1, PIWIL2, PIWIL3, & PIWIL4, in hepatocellular carcinoma patients, and to associate their expression patterns with clinicopathological features.
METHODS:
The expression patterns of PIWIL1, PIWIL2, PIWIL3, PIWIL4 mRNA, was assessed via real-time quantitative polymerase chain reaction (RT-QPCR), on tissue and serum samples from HCC patients, their impact for diagnosis was evaluated by ROC curves, prognostic utility was determined, and In Silico analysis was conducted for predicted variant detection, association with HCC microRNAs and Network Analysis.
RESULTS:
Expression levels were significantly higher in both HCC tissue and serum samples than in their respective controls (
CONCLUSION:
PIWIL mRNAs are overexpressed in HCC tissue and serum samples, the expression patterns could be valuable molecular markers for HCC, due to their association with age, tumour grade and pattern. To the best of our knowledge, our study is the first to report the expression levels of all PIWIL mRNA and to suggest their remarkable values as diagnostic and prognostic biomarkers, in addition to their correlation to HCC development. Additionally, a therapeutic opportunity might be also suggested through in silico miRNA prediction for HCC and PIWIL genes through DDX4 and miR-124-3p.
1.Introduction
Liver cancer continues to be a global health problem, with global rates increasing [1, 2]. By 2025, the disease is predicted to affect over 1 million people [3]. Hepatocellular Carcinoma (HCC) is the most common type of primary liver cancer, accounting for around 90% of cases. HCC is seen as a problematic health problem in Egypt for example, with the number of patients doubling over the last decade [4]. Several risk factors are recognised for the development and progression of HCC, but the most prominent ones are viral infections, i.e., Hepatitis B virus (HBV) and hepatitis C virus (HCV). Cirrhosis is also considered at a risk factor developing HCC [3]. HCC aetiology is additionally correlated with mutational changes ascribed from exposure to tobacco and aristolochic acid (AA), and non-alcoholic steatohepatitis (NASH), all of which were determined as probable pathogenetic cofactors in HCC [5]. There is a widely accepted protocol for diagnosing chronic liver disorders (CLD), which involves evaluating liver function using a series of serum-level enzyme assays and a significant tumour marker,
Recent studies have shown that P-element-induced wimpy testis (PIWI) proteins could be used as markers for their diagnostic and prognostic values [7, 8]. Early Carcinogenesis has been linked to multiple epigenetic abnormal events such as; global hypomethylation of DNA, post-transcriptional changes of histones, dysregulation of noncoding RNAs (ncRNAs), and reactivation of transposable elements (TE) [9, 10, 11, 12, 13, 14]. PiRNAs (P-element induced wimpy testis (PIWI)-interacting RNAs) are short single-stranded ncRNAs, typically 25–33 nucleotides, and interact with PIWI proteins of the Argonaute family. PIWI proteins are involved in the synthesis of piRNAs and assemble ribonucleoproteins known as PiRNA-induced silencing complexes (pi-RISCs) in the cytoplasmic perinuclear foci or ‘nuage’, the mechanism operates at the transcriptional and post-transcriptional stages, and is based on complementarity with short RNA strands (piRNAs, miRNAs, and siRNAs). PIWI proteins and piRNAs were first thought to be implicated in germline and stem cells, with involvements in development, gametogenesis, proliferation differentiation, and maintenance of its integrity and stability via the inhibition of transposable elements’ (TEs) activation [15, 16, 17, 18, 19]. Their roles were also identified for self-renewal, fertilisation, organogenesis and epigenetic activation, expression of genes and proteins, maturation and plasticity of the brain, pancreas functions, and even fat metabolism [20, 21]. Emerging evidence has found their role in carcinogenesis and are related with prominent cancer hallmarks [5]. In humans, the PIWI protein family consists of four proteins: PIWIL1/HIWI, PIWIL2/HILI, PIWIL3, and PIWIL4/HIWI2 [22], belong to the class of cancer/testis antigens (CTAs), and their dysregulation is associated with cancer cell maintenance of proliferative signalling, apoptosis, stemness, genomic integrity, activating invasion, metastasis, mediating genomic instability, and boosting cell growth, to mention a few [23]. The abnormal expression of PIWIs in cancer were first discovered in 2011 [24] and the molecular mechanisms underlying PIWI’s oncogenic actions are controversial. Studies have also supported that PIWI proteins can be utilized cancer prognosis, and in combination with piRNAs they could also be employed for diagnosis [25]. Overexpression of PIWIL1/HIWI gene is seen in various cancers, including seminoma cell hyperplasia [25], oesophageal squamous cell carcinoma, gastric cancer [26] and pancreatic adenocarcinoma [27]. PIWIL2 gene variants transcribed by intragenic promoters, and shorter mRNAs were implicated in various cancers due to their carcinogenic characteristics. Thus, altered PIWI proteins and their variations seen in somatic malignant tumours may serve as diagnostic and prognostic biomarkers and therapeutic targets [28, 29]. Due to inconsistent findings in the literature, we examined the expression levels of the four human members of the PIWI family, both RNA levels by quantitative RT-PCR, in HCC patients (
2.Methods
2.1Patients and sampling
The present study was conducted at Theodor Bilharz Research Institute (TBRI), Egypt. Patients who were diagnosed with HCC by multi-slice triphasic CT and increased alpha fetoprotein levels were selected. Institutional Approval was acquired from the Research Institute Board office (IRB) (NHTMRI-IRB) (Serial: 2-2019), and Theodor Bilharz Research Institute (TBRI-IRB). The research was conducted according to the declaration of Helsinki for human subject research guidelines (2013). Prior to enrolment, all patients and volunteers signed an informed consent form. Participants’ information was collected in strict confidence. 50 patients undergoing liver resection were sampled for tumour and tumour-adjacent samples, the surgery for liver resection was conducted within the department of surgery NHTMRI Hospital, Egypt, matching blood samples were also collected, and blood samples from 25 healthy volunteers were used as controls. Individuals suffering from other liver diseases (e.g., Autoimmune hepatitis, Hemochromatosis, Schistosoma), or diseases such as HIV, and ischemic heart diseases were excluded. Patients with HCV who were taking immunomodulatory interferon therapy were also excluded.
2.2Sample processing
Blood samples were allowed to clot, centrifuged at 500 xg for 10 minutes, serum was collected, centrifuged, aliquoted, and stored at
2.3Biochemical parameters
Laboratory tests including alanine aminotransferase (ALT), aspartate aminotransferase (AST), Bilirubin, albumin (ALB) and alpha-fetoprotein (AFP) were performed for all subjects as routine tests upon admission, AFP was also measured by ELISA using a commercially available kit (ABCAM, AB79801, Cambridge, UK).
2.4RNA extraction and cDNA synthesis
Total RNA was extracted using the miRNeasy extraction kit (Qiagen, Valencia, CA) was used to according to the manufacturer’s instructions for both tissue and serum samples. Samples were extracted in duplicates, then the quality and concentration of the samples were measured using a NanoDrop-1000c spectrophotometer (Thermo-Fisher Scientific, Cinisello Balsamo, Italy). For transcription of the mRNA samples into cDNA The QuantiTect Reverse Transcription Kit (Qiagen, Valencia, CA), was used according the manufacturer’s instructions, with 1
2.5Real-time quantitative polymerase chain reaction (RT-QPCR)
The selected primers included the four isoforms PIWIL1/HIWI; PIWIL2/HILI; PIWIL3, PIWIL4 and all assays were acquired from Qiagen, and were performed according to the manufacturer’s instructors. Quantitative values were respective to the Cycle number (Ct Value), where fluorescence was directly proportional to growth of PCR products, this was performed by QuantiTect SYBR Green PCR Kits (Qiagen, Valencia, CA). Glyceraldehyde 3-phosphate dehydrogenase, (GAPDH), was also used, as an endogenous control because of its transcripts’ prevalence, to normalize each sample. All reactions were run in duplicates. Finally, the
2.6In Silico variant detection for PIWIL genes and Network Analysis
Hepatocellular Carcinoma primary cancer database (
2.7Statistical
All statistical analyses were performed using statistical software SPSS (Statistical Package for Social Science) statistical program version 21.0. A Power test, indicated that the standard deviation of control is 0.8 and the standard deviation for the regression errors will be 1.9. Regression was at 1.1, and that 50 study subjects and 25 normal controls will be an appropriate representation via regression with a probability of 85%, and Type I error was 0.05. as adapted from HCC molecular marker research [33]. Normality tests determined continuous variables, described as mean
Table 1
Demographics and Clinico-pathological characteristics for HCC patients
| Clinico-pathological characteristics | Total number of patients |
|---|---|
| Age (Mean | 57.2 |
| Sex | |
| Female | 22 (44.0) |
| Male | 28 (56.0) |
| ALT | |
| 7–55 U/L | 61.4 |
| AST | |
| 8–33 U/L | 65.8 |
| Alb | |
| 3.4–5.4 g/dL | 2.3 |
| Bilirubin | |
| 0.3–1.2 mg/dL | 2.9 |
| AFP | |
| 0 ng/mL–40 ng/mL | 75.0 (40.0–150.0) |
| | |
| S. Creatinine | |
| M: 0.74–1.35 mg/dL | 0.9 |
| F: 0.59–1.04 mg/dL | |
| No. of masses | 1.1 |
| Tumour size | 2.25 (0.75–4.25) |
| Tumour grade | |
| I | 29 (38.7) |
| II | 11 (14.7) |
| III | 10 (13.3) |
| Pattern | |
| Acinar | 30 (60.0) |
| Solid | 17 (34.0) |
| Acinar/Solid | 3 (6.0) |
| Steatosis | 0.02 (0.02–0.04) |
| Stage | |
| Fibrosis | 8 (16.0) |
| Cirrhosis | 42 (84.0) |
| HAI | |
| A1 | 15 (30.0) |
| A2 | 35 (70.0) |
| A3 | 0 (0.0) |
| Hepatomegaly | |
| Negative | 41 (82.0) |
| Positive | 9 (18.0) |
| Ascites | |
| Negative | 30 (60.0) |
| Positive | 20 (40.0) |
| Splenomegaly | |
| Negative | 21 (42.0) |
| Positive | 29 (58.0) |
| Oedema lower limbs | |
| Negative | 37 (74.0) |
| Positive | 13 (26.0) |
Alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin (Alb) and alpha-fetoprotein (AFP). No. of masses are represented as Mean and SD. But Alpha feto-protein, Tumour size, and Steatosis (Fatty degeneration of hepatocytes (% of cells)) are represented as Median and Interquartile Range IQR (25%–75%). While Sex, Grade, Pattern, Stage, HAI (Hepatitis Activity Index (grade of hepatitis), Hepatomegaly, Ascites, Splenomegaly, and oedema Lower Limbs are represented as Frequency and percent.
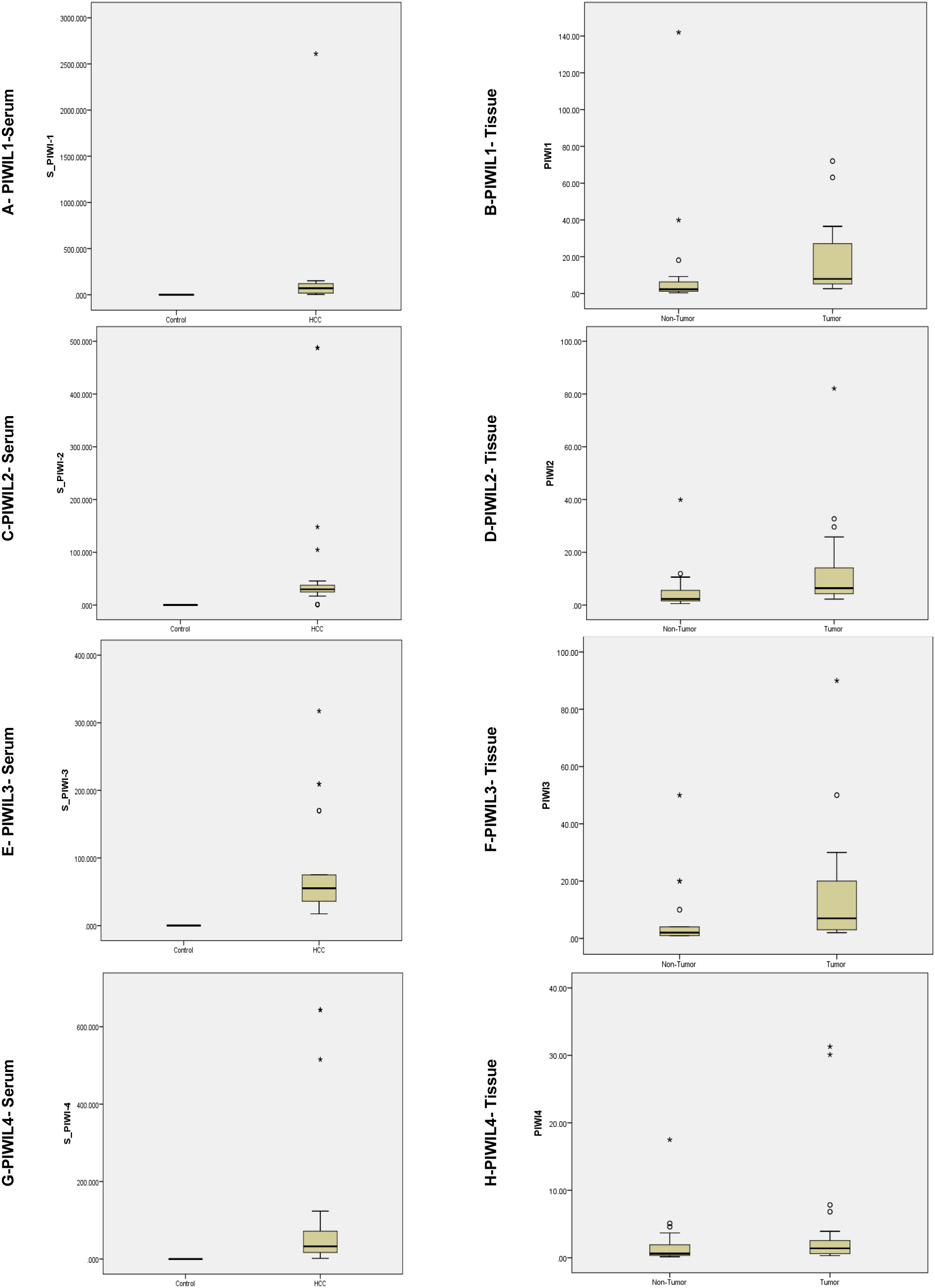
Figure 1.
Relative expression representation for PIWIL mRNA in HCC (
3.Results
3.1Patient characteristics
A total of 50 HCC participants were recruited in this study, the patients included 28 males (56%) and 22 females (44%), with a mean age of 57.2
Table 2
Clinico-pathological characteristics for HCC patients and their association with PIWIL mRNA expression
| Clinico-pathological parameters | No. of cases | Tissue expression | Serum expression | ||||||
|---|---|---|---|---|---|---|---|---|---|
| PIWIL1 | PIWIL2 | PIWIL3 | PIWIL4 | PIWIL1 | PIWIL2 | PIWIL3 | PIWIL4 | ||
| Tumour Grade | 0.2 | 0.7 | 0.4 | 0.2 | 0.8 | 0.9 | 0.9 | 0.2 | |
| I | 29 (38.7) | 6.2 (4.4–13.4) | 6.8 (3.5–25.8) | 6.0 (3.5–9.0) | 1.7 (0.8–2.9) | 71.0 (18.2–100.3) | 29.6 (24.8–41.6) | 55.3 (29.0–122.5) | 32.7 (17.0–97.8) |
|
| 0.9 | 0.5 | 0.5 | 0.1 | 0.6 | 0.9 | 0.5 | ||
| II | 11 (14.7) | 28.0 (5.9–36.5) | 6.4 (4.6–9.9) | 9.0 (3.0–20.0) | 1.0 (0.6–7.8) | 60.1 (18.2–120.2) | 29.6 (16.8–37.5) | 55.3 (36.0–170.0) | 22.8 (11.8–39.7) |
| 0.9 | 0.9 |
| 0.2 | 0.7 | 0.3 | 0.2 | |||
| III | 10 (13.3) | 10.1 (5.8–30.0) | 6.2 (4.6–8.3) | 7.0 (2.8–20.0) | 0.7 (0.4–2.6) | 100.3 (49.3–128.2) | 30.1 (22.9–39.5) | 189.7 (32.5–236.3) | 39.7 (15.7–182.9) |
| Pattern | 0.3 | 0.6 | 1 | 0.5 | 0.6 | 0.3 | 0.6 | 0.7 | |
| Acinar | 30 (60.0) | 7.9 (5.1–13.5) | 6.4 (3.8–25.8) | 6.0 (3.0–11.8) | 1.7 (0.6–2.2) | 65.6 (18.2–90.4) | 30.1 (24.9–39.5) | 55.3 (37.5–179.8) | 34.9 (17.0–84.9) |
| 0.3 | 0.3 | 0.5 | 0.4 | 0.7 | 0.5 | 0.9 | 0.8 | ||
| Solid | 17 (34.0) | 7.9 (4.1–28.8) | 6.1 (4.1–6.7) | 9.0 (3.0–20.0) | 1.0 (0.5–5.9) | 71.5 (18.2–152.2) | 29.6 (8.9–35.3) | 55.3 (22.0–189.7) | 22.8 (11.8–55.9) |
| 0.4 |
| 0.2 | 0.2 | 0.6 | 0.1 | 0.3 | 0.1 | ||
| Acinar/Solid | 3 (6.0) | 31.3 (6.2–31.3) | 10.9 (9.9–10.9) | 20.0 (7.0–20.0) | 6.8 (1.2–6.8) | 80.4 (3.1–80.4) | 37.5 (0.9–37.5) | 75.0 (22.0–75.0) | 32.7 (11.8–32.7) |
| Stage | |||||||||
| Fibrosis | 8 (16.0) | 0.7 | 0.5 | 0.4 | 0.8 |
| 0.8 | 0.7 | 0.9 |
| Cirrhosis | 42 (84.0) | 7.9 (5.3–12.9) | 8.3 (3.5–25.8) | 6.0 (3.3–9.0) | 1.9 (0.5–3.5) | 8.0 (4.9–28.0) | 6.2 (4.2–14.1) | 8.0 (3.0–20.0) | 1.4 (0.6–3.1) |
The studied genes are represented as Median and Interquartile Range IQR (25%–75%); the data were analysed by Mann-Whitney U test. *p < 0.05, **p< 0.001. Grade, Pattern, Stage, Hepatomegaly, Ascites, Splenomegaly, and oedema Lower Limbs are represented as Frequency and percent.
Table 3
Clinico-pathological characteristics for HCC patients and their correlation with PIWIL mRNA expression
| Clinico-pathological carameters | No. of cases | Serum | Tissue | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PIWIL1 | PIWIL2 | PIWIL3 | PIWIL4 | PIWIL1 | PIWIL2 | PIWIL3 | PIWIL4 | ||||||||||
|
|
|
|
|
|
|
|
| ||||||||||
| Age (Mean | 57.2 | 0.178 | 0.514 | 0.042* | 0.35 | 0.41 | 0.132 | 0.204 | 0.934 | 0.01* | 0.89 | 0.031* | 0.93 | 0.615 | 0.385 | 0.006* | |
| Sex | |||||||||||||||||
| Female | 22 (44.0) | 0.031 | 0.15 | 0.047 | 0.741 | 0.207 | 0.173 | 0.027 | 0.852 | 0.172 | 0.233 | 0.142 | 0.441 | 0.155 | 0.427 | 0.085 | 0.061 |
| Male | 28 (56.0) | 0.053 | 0.24 | 0.072 | 0.35 | 0.106 | 0.099 | 0.506 | 0.245 | 0.631 | 0.387 | 0.545 | |||||
| ALT | |||||||||||||||||
| 7–55 U/L | 61.4 | 0.03 | 0.838 | 0.274 | 0.611 | 0.087 | 0.546 | 0.784 | 0.891 | 0.725 | 0.558 | ||||||
| AST | |||||||||||||||||
| 8–33 U/L | 65.8 | 0.015 | 0.917 | 0.414 | 0.439 | 0.022 | 0.88 | 0.348 | 0.688 | 0.213 | 0.179 | ||||||
| Alb | |||||||||||||||||
| 3.4–5.4 g/dL | 2.3 | 0.048 | 0.742 | 0.076 | 0.601 | 0.049 | 0.748 | 0.672 | 0.091 | 0.528 | 0.097 | 0.503 | 0.088 | 0.542 | 0.151 | 0.296 | |
| Bilirubin | |||||||||||||||||
| 0.3–1.2 mg/dL | 2.9 | 0.031 | 0.829 | 0.047 | 0.745 | 0.207 | 0.173 | 0.027 | 0.852 | 0.172 | 0.233 | 0.142 | 0.324 | 0.115 | 0.428 | 0.165 | 0.252 |
| AFP | |||||||||||||||||
| 0 ng/mL–40 ng/mL | 75.0 (40.0–150.0) | 0.703 | 0.86 | 0.106 | 0.099 | 0.506 | 0.245 | 0.623 | 0.387 | 0.534 | |||||||
| | |||||||||||||||||
| S. Creatinine | |||||||||||||||||
| M: 0.74–1.35 mg/dL | 0.9 | 0.591 | 0.62 | 0.037 | 0.81 | 0.183 | 0.204 | 0.034 | 0.814 | 0.03 | 0.835 | 0.616 | 0.352 | ||||
| F: 0.59–1.04 mg/dL | |||||||||||||||||
| No. of masses | 1.1 | 0.767 | 0.481 | 0.415 | 0.136 | 0.348 | 0.366 | 0.879 | 0.377 | 0.578 | |||||||
| Tumour size | 2.25 (0.75–4.25) | 0.782 | 0.556 | 0.685 | 0.064 | 0.682 | 0.354 | 0.416 | 0.281 | 0.182 | |||||||
| Steatosis | 0.02 (0.02–0.04) | 0.273 | 0.848 | 0.336 | 0.189 | 0.69 | 0.738 | 0.559 | 0.554 | ||||||||
Alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin (Alb) and alpha-fetoprotein (AFP). No. of masses are represented as Mean and SD. But Alpha feto-protein, Tumour size, and Steatosis (Fatty degeneration of hepatocytes (% of cells)) are represented as Median and Interquartile Range IQR (25%–75%). Sex, represented as Frequency and percent
15 (30%) patients were rated as AI, 35 (70%) were A2 while none of the patients were A3. Abdominal ultrasounds detected hepatomegaly in 9 (18%) patients, ascites in 20 (40%) patients and splenomegaly in 29 (58%) patients and finally 13 (26%) out of the 50 HCC patients were detected with oedema lower limbs (Table 1).
Figure 2.
ROC curves of the studied PIWIL mRNA in (a) serum and (b) tissue samples. (C & D) are combined curves for serum and tissue, respectively. (E) ROC curve for AFP measured by ELISA in serum samples of HCC patients. Graphs depict the diagnostic performance of all PIWIL mRNAs in terms of specificity & sensitivity.
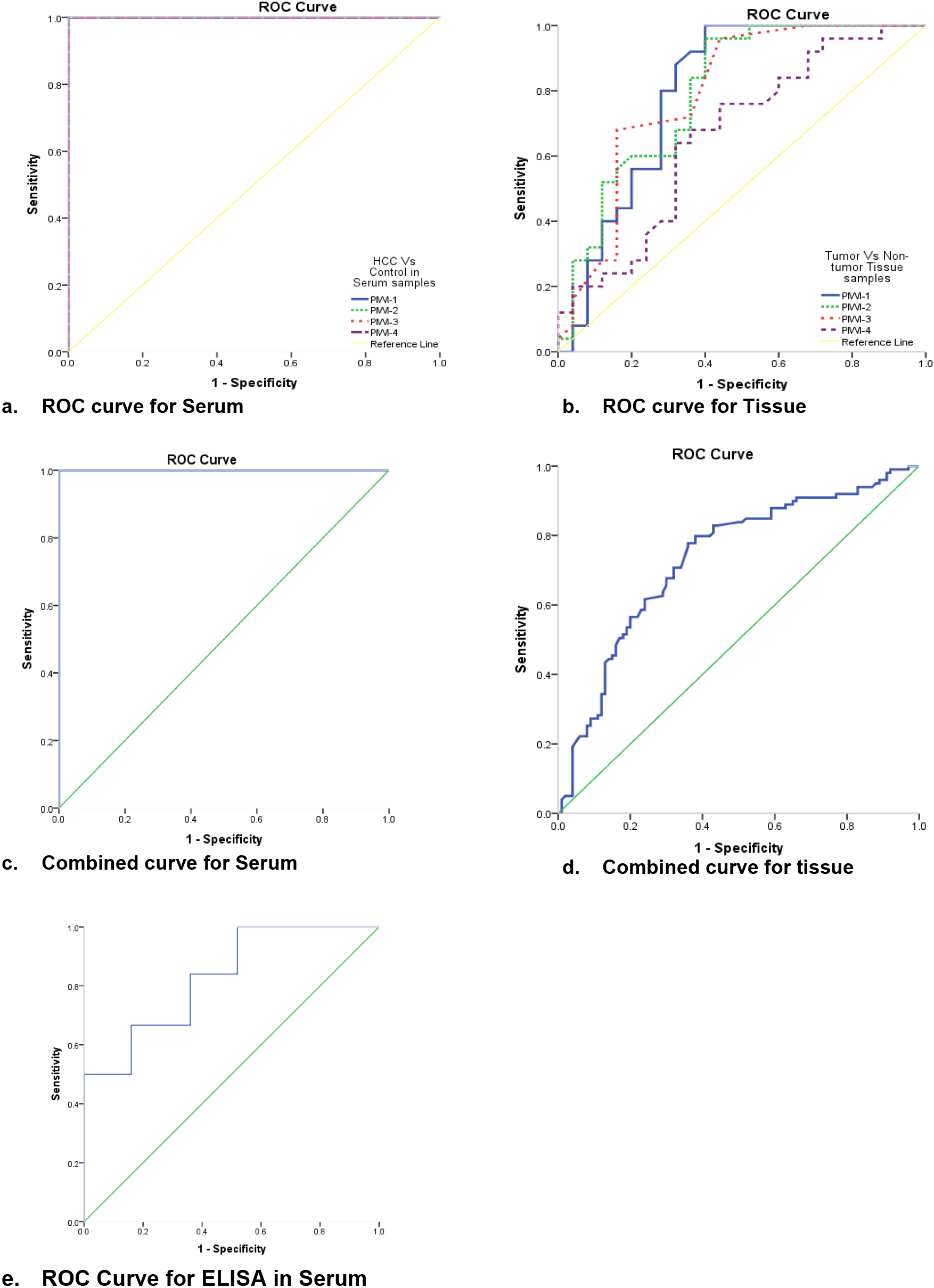
Table 4
ROC curve analysis for diagnostic performance for each of the studied PIWIL mRNAs in serum and tissue samples
| Groups | Cut-off | Sensitivity | Specificity | AUC | 95% C.I | |||
|---|---|---|---|---|---|---|---|---|
| Lower bound | Upper bound | |||||||
| Serum | PIWIL1 | 100.0 | 100.0 | 1.0 | 1.0 | 1.0 |
| |
| PIWIL2 | 100.0 | 100.0 | 1.0 | 1.0 | 1.0 |
| ||
| PIWIL3 | 100.0 | 100.0 | 1.0 | 1.0 | 1.0 |
| ||
| PIWIL4 | 100.0 | 100.0 | 1.0 | 1.0 | 1.0 |
| ||
| ELISA/AFP | 84.0 | 64.0 | 0.828 | 0.746 | 0.910 |
| ||
| Serum | Combined | 100.0 | 100.0 | 1.0 | 1.0 | 1.0 |
| |
| Tissue | PIWIL1 | 80.0 | 72.0 | 0.80 | 0.71 | 0.89 |
| |
| PIWIL2 | 84.0 | 64.0 | 0.80 | 0.71 | 0.89 |
| ||
| PIWIL3 | 68.0 | 84.0 | 0.79 | 0.70 | 0.88 |
| ||
| PIWIL4 | 64.0 | 68.0 | 0.66 | 0.56 | 0.77 |
| ||
| Tissue | Combined | 70.7 | 68.0 | 0.733 | 0.683 | 0.783 |
| |
Sn: Sensitivity, Sp: Specificity, AUC Area under curve and C.I: 95% Confidence Interval. * p< 0.05, ** p< 0.001.
3.2Expression of PIWIL mRNA transcripts in serum & tissue
RT-QPCR results showed mRNA levels were upregulated for PIWIL1, PIWIL2, PIWIL3 and PIWIL4 in serum & tissue samples at (
3.3Association of clinicopathological features with the expression PIWIL mRNA transcripts
Tumour grades of patients were associated with expression of PIWIL1-4. No association was found for serum, but for tissue samples, a significant association for PIWIL1 (
Table 5
HCC risk results of PIWIL mRNA in HCC patients
| HCC risk determination (logistic regression) | Serum groups | Tissue groups | ||||||
|---|---|---|---|---|---|---|---|---|
| PIWIL1 | PIWIL2 | PIWIL3 | PIWIL4 | PIWIL1 | PIWIL2 | PIWIL3 | PIWIL4 | |
| OR (95% C.I) | 3.87 (1.23–8.36) | 4.21 (1.04–7.23) | 3.26 (1.14–8.21) | 2.99 (1.57–9.23) | 2.35 (0.17–5.32) | 2.21 (0.15–4.24) | 2.46 (0.23–6.27) | 1.65 (0.19–3.14) |
|
|
|
|
| 0.01* | 0.01* | 0.01* | 0.01* | |
OR: Odds Ratio; C.I: Confidence Interval
3.4Diagnostic performance
Receiver Operating characteristic (ROC) analysis of PIWIL1, PIWIL2, PIWIL3 and PIWIL4 in both serum, and tissue samples of HCC patients. They showed a sensitivity of 100%, specificity of 100%, area under curve (AUC) of 1.00 (
3.5Logistic regression analysis of PIWIL’s
To assess the relative risk of HCC presented from PIWIL mRNA, logistic regression analysis model was performed (Table 5). It revealed that PIWIL1-4 is significantly associated with increased risk for HCC in serum (
3.6In Silico analysis for PIWIL genes
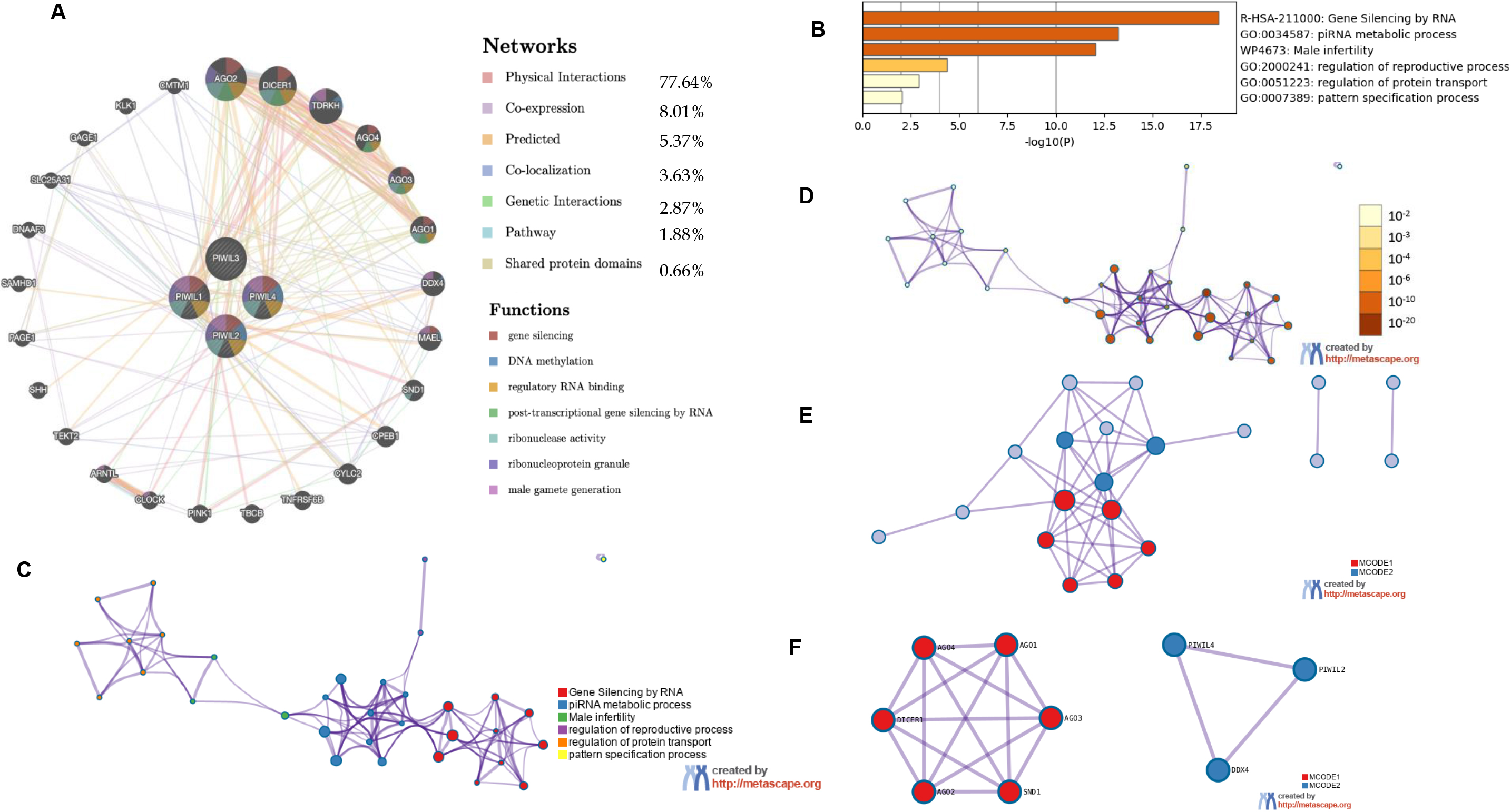
In Fig. 3B the role of PIWIL genes in HCC progression is indicated, where the highest implication being for PIWIL2 at 4% structural variant detection with an association for HCC; Fig. 3B shows the frequency of PIWI mutations according to HCC types; miRNet prediction was also employed for HCC association with PIWIL genes (Fig. 3A) and a direct association was detected between PIWIL2, PIWIL4 and MiR-124-3p.1, a known potential tumour suppressor associated with diverse processes including proliferation, apoptosis, and metastasis. The Frequency of PIWIL mutations were caried out through TCGA; via BioPortal (Fig. 3C), the results indicated that the highest frequency for reported mutations were for PIWIL2 at 4%, and the type of reported alterations were deep deleted deletions, remaining PIWIL variants had 1.4% which also had missense mutations as alterations. We identified the top 24 neighbouring genes with the highest frequency association with differential expressed PIWIL targets. The functions of these related were predicted using Metascape. The top 29 GO enrichment were described in (Fig. 4), which mainly included gene silencing by RNA, lncRNA, siRNA biogenesis and RISC complex assembly; Pathway enrichment analysis represented pathways a strong associated between PIWIL2, PIWIL4 and DDX4, DEAD-box helicase 4 (DDX4) and DDX39 was previously found to be upregulated in HCC, and that knocking down the DDX4 significantly decreased tumour formation in vivo and in vitro, as well as reduces tumour metastasis in vivo [34, 35] (Fig. 4F); indicating a relationship with HCC progression. The PPI network and MCODE components are shown in (Fig. 4C–F).
4.Discussion
PIWIL1, PIWIL2, PIWIL3 and PIWIL4, are designated as catalytic elements of the pi-RISCs complexes, the have implications in piRNAs’ biogenesis on the basis of complementarity [35, 36, 37]. Their roles vary at transcriptional and post-transcriptional epigenetic regulation. PIWIL1-2-3-4 RNAs were strongly expressed in HCC tissue, and circulating sera compared to the controls. The unexpected role of the PIWI-piRNA pathway has led to distinct functions of human PIWI proteins and mRNA in various cancer types [38, 39, 40]. Their involvement in multiple cancer hallmarks, has led to a possible representation as diagnostic and prognostic biomarkers [41, 42]. There are conflicting statements on the expression patterns of PIWIL1/PIWIL2/PIWIL3/PIWIL4, their prognostic, and predictive values, in addition to a complete absence for hepatocellular carcinoma in terms of mRNA transcript accumulation, and associated piRNAs, with recent studies on identifying novel piRNAs [43, 44, 45].
In terms of demographic data for the patients of the current study, the mean age was 57.2
Figure 3.
(A) miRNet prediction of PIWIL and HCC. (B) Reported cases with mutated PWIL genes in Liver cancer (C) Frequency of mutated PIWIL genes and their predicted variants’ implication for HCC through TCGA; via BioPortal for Cancer Genomics (https://www.cbioportal.org).
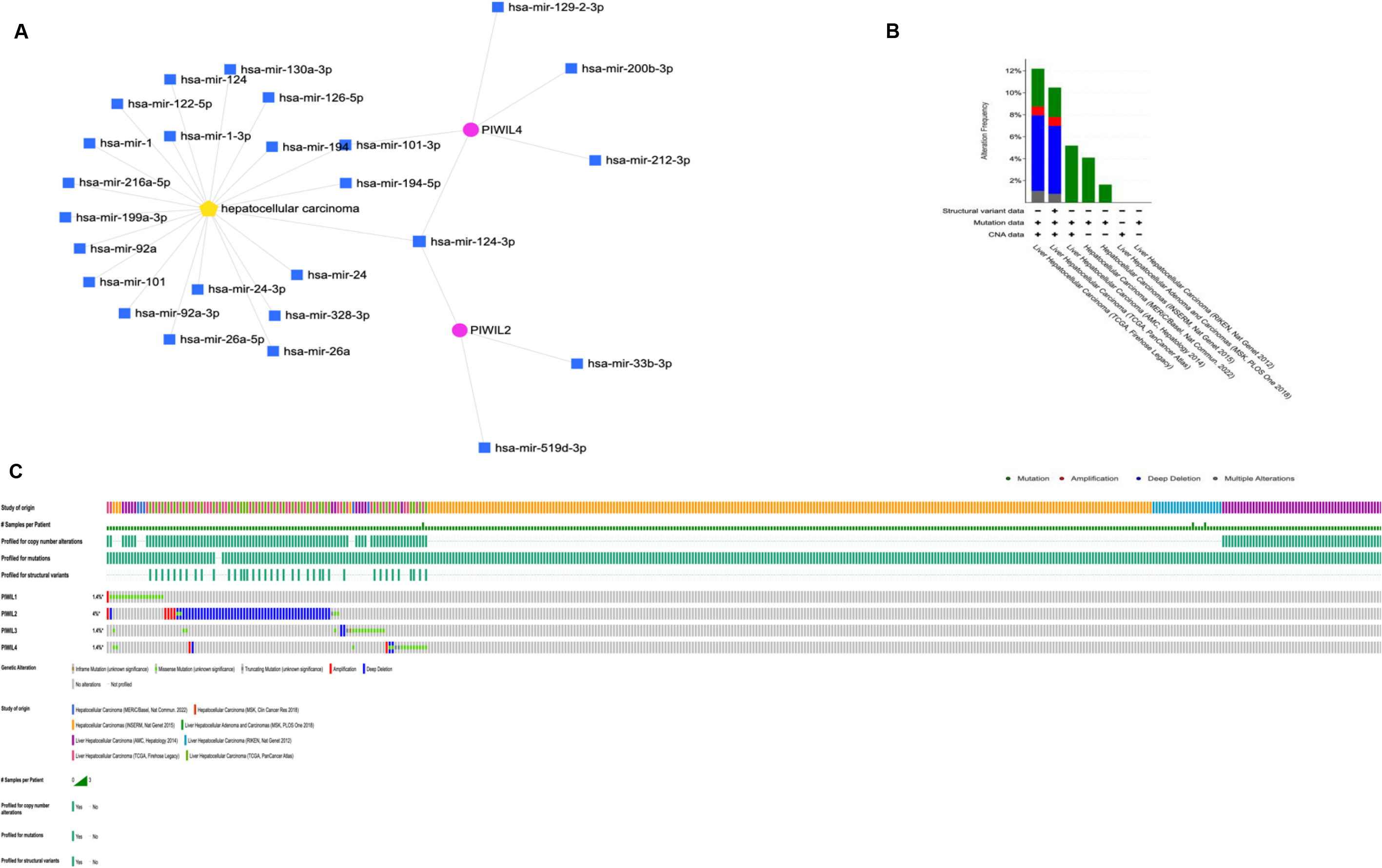
Figure 4.
(A) Genemania generated Network for PIWIL1, PIWIL2, PIWIL3, PIWIL4 and function predictions (B) Metascape results for Enriched ontology clusters. (C) PIWIL Subset network with force-directed clustering (D) Enriched network according to statistical significance, the darker the color, the more statistically significant the node is. (E) protein-protein interactions’ network from PIWIL genes. (F) Results for MCODE algorithm to identify neighborhoods where proteins are densely connected.
& PIWIL4 had significant difference with tissue expression, which was confirmed in a study by Litwin et al. for PIWIL1, & PIWIL2 in breast cancer [49].
Recently, several reports have indicated that aberrant expression of PIWI at the mRNA and protein levels occurs in various types of tumours [50, 51]. PIWIL1/HIWI was previously linked with several types of cancers, with a pattern of overexpression, moreover, it was correlated with tumour grading and staging [51, 52]. Our findings confirmed the apparent role of PIWIL mRNAs for HCC, with a pattern observed of overexpression for all PIWIL isoforms. Previously, both PIWIL1 & PIWIL2 was identified as overly expressed in colorectal, prostate, breast, cervical, gastric and bladder cancer [51], PIWIL1, 2 & 4 was observed as downregulated in renal cell carcinoma [50]. Meseure et al. conducted an investigation on the bio-pathological significance of the PIWI-PiRNA pathway through PIWIL1-2-3-4 mRNA expression levels in a panel of normal tissues and corresponding malignant tumours, and detected variable levels of expression across malignancies [53]. In addition, the relative expression of PIWIL2 mRNA was previously found to be higher in HCC tissues compared with adjacent normal liver tissues. A positive correlation was found between PIWIL2 expression and piR-Hep1 level according to Pearson’s correlation analysis [37, 38]. PIWIL2 acts as an oncogene by activating the STAT3/Bcl-xl cell signalling pathway through endogenous RNAi mechanism, hence inhibiting cell apoptosis and promoting cell proliferation. To the best of our knowledge, our study is the first to report the expression levels of all PIWIL mRNAs in serum and tissue, and to suggest their possible roles as diagnostic and prognostic biomarkers, in addition to their correlation to HCC development.
The involvement of PIWI proteins was linked with multiple hallmarks of cancer including invasion, apoptosis evasion, metastasis and cell proliferation, as such they possess prospective diagnostic factors and biomarkers for cancer prognosis [25, 54]. Significant increased level of PIWIL1 was reported for colon, bladder, and hepatocellular carcinoma. Expression of the four members of the PIWI proteins was viewed as distinct in tumour tissue when compared with the adjacent non-tumorous tissue [22, 55]. PIWIL3 and PIWIL1 were assessed for relative expression levels by [56] in colorectal cancer, and had non-significant expression statistically. Among all PIWIL genes, those assessed for expression and correlated with overall survival and recurrence-free survival were PIWIL3 & PIWIL4, in invasive urothelial bladder cancer [49]. Erber et al. reported a limitation of their study was not assessing the expression of PIWIL1 and PIWIL2 at the mRNA level, and depending on Immunohistochemistry, but they reported higher expression levels [57]. In the current study, expression of all PIWIL was assessed using real-time PCR in HCC patients. The levels of mRNA transcripts expression are reported for the first time for PIWIL1, PIWIL2, PIWIL3, PIWIL4 in tumour and nontumorous adjacent tissue, and matching serum samples HCC patients and healthy controls, the expression was correlated with clinical data. When compared to adjacent non-cancerous tissues, we found a significantly elevated expression for PIWIL1, PIWIL2, PIWIL3, &, PIWIL4 in HCC samples (
These findings are consistent with prior findings in colorectal cancer, in which PIWIL1 mRNA levels in non-cancerous tissue were low or undetectable, but were dramatically raised in malignant tissue [58]. Additionally, PIWIL1/HIWI was reported to have marked expression levels in HCC tissue, for patients who had undergone curative resection [42]. In breast cancer, PIWIL1 and PIWIL3 gene expressions were reported to be upregulated, whereas PIWIL2 and PIWIL4 were downregulated compared with normal breast tissue [53]. It is noteworthy to state that our research detected significant association between PIWIL1 & PIWIL4 expression with increasing tumour grade (
In terms of diagnostic performance, our findings showed that serum had an overall sensitivity and specificity of 100%, and an AUC of 1.0, in comparison to normal serum, which shows an indication of disease prevalence but should be adapted for clinical settings i.e., patients with chronic liver diseases. Tissue samples exhibited sensitivity of 70%, specificity of 68%, and AUC of 0.733. Both of which were significant
PIWIL genes have several transcripts, some of which appear to be transcribed by putative intragenic promoters rather than a canonical promoter, which was associated with tumorigenesis [65]. PIWIL expression was revealed to have a direct predicted influence on HCC progression, through PIWIL2, and PIWIL4, this was found through two novel associations DDX4 (Fig. 4E), and miR-124-3p.1 (Fig. 3A), Recent studies have shown that downregulation of miR-124-3p.1 was associated with poor survival, early recurrence and sorafenib sensitivity in HCC patients [42], and our findings have demonstrated a novel direct interplay between PIWIL2, PIWIL4 and miR-124-3p.1, the pathway additionally indicates a several target miRNAs which could be used as possible therapeutic targets for PIWIL2 miR-33b-3p and miR-519d-3p were identified, while for PIWIL4, miR-129-2-3p, miR-200b-3p, and miR-212-3p were found, all of which are related to miR-124-3p.1, a microRNA which when upregulated, negative affects proliferation and migration in hepatocellular carcinoma via a phosphoinositide 3-kinase catalytic subunit alpha (PIK3CA) pathway [68]. and miR-101-3p was additionally detected for PIWIL4, and was reported to negatively influence HCC proliferation and metastasis through the HGF/c-Met pathway [70]. Thus, aberrant expression of PIWIL2 and PIWIL4 is a possible mechanism of tumour suppressor inactivation Fig. 3A, and the associated miRNAs could represent attractive therapeutic targets for combined therapies where a specific antibody for PIWIL2 or the PIWI/miRNA RISC complex could be targeted [71]. PIWIL expression is also shown have a direct predicted influence on HCC progression, through PIWIL2, and PIWIL4, through association with DDX4 (Fig. 4). Studies have shown that elevated levels of DDX4, indicates its ability to promote the stemness of breast cancer stem cells by regulating the expression of proteins such as Oct3/4 and Sox-2 and promoting disease progression [72]. In this regard, its upregulation or overexpression promotes proliferation, suggesting an oncogenic role, its association through the PPI network (Fig. 4), shows a novel interaction with PIWIL2 and PIWIL4 and further analysis into PIWIL2 and PIWIL4 expression and silencing is recommended.
5.Conclusion
Finally, PIWI mRNA, PIWI proteins, and piRNAs were identified as germline markers. DNA methylation, histone methylation, histone acetylation, and histone ubiquitination not only play significant transcriptional regulatory roles, but PIWI family proteins can break mRNA under the supervision of piRNA, suggesting a post-transcriptional regulatory function. However, the method of control of PIWI/piRNAs in cancer appears to be unique. Most studies have established that PIWI mRNA, proteins, and piRNA appear to govern tumours as two distinct entities rather than as a unified entity. As a result, it is critical to investigate how the PIWI protein regulates tumours independently of piRNA. The specific molecular biological mechanism behind the effect of PIWIL on the occurrence, progression, and prognosis of HCC is currently unknown and deserves additional investigation. Our findings provide a unique viewpoint on the activities of PIWIL at the mRNA level in HCC development, as well as a different pattern of overexpression that provides potential candidates for HCC disease progression and risk assessment. PIWIL mRNAs are overexpressed in HCC tissue and serum samples, the expression patterns could be valuable molecular markers for HCC, due to their association with age, tumour grade and pattern. To the best of our knowledge, our study is the first to report the expression levels of all PIWIL mRNA and to suggest their remarkable values as diagnostic and prognostic biomarkers, in addition to their correlation to HCC development. Additionally, a therapeutic opportunity might be also suggested through in silico miRNA prediction for HCC and PIWIL genes through DDX4 and miR-124-3p. The epigenetic regulation of the identified changes in PIWIL1, PIWIL2, PIWIL3, and PIWIL4 at the transcriptional and protein levels warrants additional investigation, which could have significant clinical relevance, and should be examined for therapeutic roles. A large sample size investigation will aid in analysing the link between PIWIL expression/co-expression and HCC prognosis. This can serve as the preliminary foundation for PIWIL as molecular markers of early-stage diagnostic and prognostic evaluation, as well as targeted cancer therapies.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-230134.
References
[1] | H. Rumgay, J. Ferlay, C. de Martel, D. Georges, A.S. Ibrahim, R. Zheng, W. Wei, V. Lemmens and I. Soerjomataram, Global, regional and national burden of primary liver cancer by subtype, Eur J Cancer 161: ((2022) ), 108–118. |
[2] | A. Villanueva, Hepatocellular carcinoma, N Engl J Med 380: ((2019) ), 1450–1462. |
[3] | J.M. Llovet, R.K. Kelley, A. Villanueva, A.G. Singal, E. Pikarsky, S. Roayaie, R. Lencioni, K. Koike, J. Zucman-Rossi and R.S. Finn, Hepatocellular carcinoma, Nat Rev Dis Primers 7: ((2021) ), 6. |
[4] | W.M. Rashed, M.A.M. Kandeil, M.O. Mahmoud and S. Ezzat, Hepatocellular Carcinoma (HCC) in Egypt: A comprehensive overview, J Egypt Natl Canc Inst 32: ((2020) ), 5. |
[5] | K. Schulze, S. Imbeaud, E. Letouze, L.B. Alexandrov, J. Calderaro, S. Rebouissou, G. Couchy, C. Meiller, J. Shinde, F. Soysouvanh, A.L. Calatayud, R. Pinyol, L. Pelletier, C. Balabaud, A. Laurent, J.F. Blanc, V. Mazzaferro, F. Calvo, A. Villanueva, J.C. Nault, P. Bioulac-Sage, M.R. Stratton, J.M. Llovet and J. Zucman-Rossi, Exome sequencing of hepatocellular carcinomas identifies new mutational signatures and potential therapeutic targets, Nat Genet 47: ((2015) ), 505–511. |
[6] | P. Wang, F. Nie, T. Dong, D. Yang, T. Liu and G. Wang, Diagnostic Value of CEUS LI-RADS Version 2017 in Differentiating AFP-Negative Hepatocellular Carcinoma from Other Primary Malignancies of the Liver, Diagnostics (Basel) 11: ((2021) ). |
[7] | K. Hanusek, S. Poletajew, P. Kryst, A. Piekielko-Witkowska and J. Boguslawska, piRNAs and PIWI proteins as diagnostic and prognostic markers of genitourinary cancers, Biomolecules 12: ((2022) ). |
[8] | K. Jiang, T. Ye, J. Du, L. Tang, X. Chen, F. Sun and X. Sun, Elevated p-element-induced wimpy-testis-like protein 1 expression predicts unfavorable prognosis for patients with various cancers, J Oncol 2021: ((2021) ), 9982192. |
[9] | D.N. Cox, A. Chao, J. Baker, L. Chang, D. Qiao and H. Lin, A novel class of evolutionarily conserved genes defined by piwi are essential for stem cell self-renewal, Genes Dev 12: ((1998) ), 3715–3727. |
[10] | T.A. Farazi, S.A. Juranek and T. Tuschl, The growing catalog of small RNAs and their association with distinct Argonaute/Piwi family members, Development 135: ((2008) ), 1201–1214. |
[11] | A. Girard, R. Sachidanandam, G.J. Hannon and M.A. Carmell, A germline-specific class of small RNAs binds mammalian Piwi proteins, Nature 442: ((2006) ), 199–202. |
[12] | J.D. Klein, C. Qu, X. Yang, Y. Fan, C. Tang and J.C. Peng, c-fos repression by piwi regulates drosophila ovarian germline formation and tissue morphogenesis, PLoS Genet 12: ((2016) ), e1006281. |
[13] | E. Stuwe, K.F. Toth and A.A. Aravin, Small but sturdy: Small RNAs in cellular memory and epigenetics, Genes Dev 28: ((2014) ), 423–431. |
[14] | D. Voller, L. Linck, A. Bruckmann, J. Hauptmann, R. Deutzmann, G. Meister and A.K. Bosserhoff, Argonaute family protein expression in normal tissue and cancer entities, PLoS One 11: ((2016) ), e0161165. |
[15] | L.T. Gou, J.Y. Kang, P. Dai, X. Wang, F. Li, S. Zhao, M. Zhang, M.M. Hua, Y. Lu, Y. Zhu, Z. Li, H. Chen, L.G. Wu, D. Li, X.D. Fu, J. Li, H.J. Shi and M.F. Liu, Ubiquitination-deficient mutations in human piwi cause male infertility by impairing histone-to-protamine exchange during spermiogenesis, Cell 169: ((2017) ), 1090–1104 e13. |
[16] | Z. Kamaliyan, S. Pouriamanesh, M. Amin-Beidokhti, A. Rezagholizadeh and R. Mirfakhraie, HIWI2 rs508485 polymorphism is associated with non-obstructive azoospermia in iranian patients, Rep Biochem Mol Biol 5: ((2017) ), 108–111. |
[17] | C.D. Malone, J. Brennecke, M. Dus, A. Stark, W.R. McCombie, R. Sachidanandam and G.J. Hannon, Specialized piRNA pathways act in germline and somatic tissues of the Drosophila ovary, Cell 137: ((2009) ), 522–535. |
[18] | E.F. Roovers, D. Rosenkranz, M. Mahdipour, C.T. Han, N. He, S.M. Chuva de Sousa Lopes, L.A. van der Westerlaken, H. Zischler, F. Butter, B.A. Roelen and R.F. Ketting, Piwi proteins and piRNAs in mammalian oocytes and early embryos, Cell Rep 10: ((2015) ), 2069–2082. |
[19] | D.T. Yin, Q. Wang, L. Chen, M.Y. Liu, C. Han, Q. Yan, R. Shen, G. He, W. Duan, J.J. Li, A. Wani and J.X. Gao, Germline stem cell gene PIWIL2 mediates DNA repair through relaxation of chromatin, PLoS One 6: ((2011) ), e27154. |
[20] | S. Kuramochi-Miyagawa, T. Kimura, K. Yomogida, A. Kuroiwa, Y. Tadokoro, Y. Fujita, M. Sato, Y. Matsuda and T. Nakano, Two mouse piwi-related genes: Miwi and mili, Mech Dev 108: ((2001) ), 121–133. |
[21] | A. Lingel and M. Sattler, Novel modes of protein-RNA recognition in the RNAi pathway, Curr Opin Struct Biol 15: ((2005) ), 107–115. |
[22] | Y. Yu, J. Xiao and S.S. Hann, The emerging roles of PIWI-interacting RNA in human cancers, Cancer Manag Res 11: ((2019) ), 5895–5909. |
[23] | J.F. Quinn, T. Patel, D. Wong, S. Das, J.E. Freedman, L.C. Laurent, B.S. Carter, F. Hochberg, K. Van Keuren-Jensen, M. Huentelman, R. Spetzler, M.Y. Kalani, J. Arango, P.D. Adelson, H.L. Weiner, R. Gandhi, B. Goilav, C. Putterman and J.A. Saugstad, Extracellular RNAs: Development as biomarkers of human disease, J Extracell Vesicles 4: ((2015) ), 27495. |
[24] | M. Esteller, Non-coding RNAs in human disease, Nat Rev Genet 12: ((2011) ), 861–874. |
[25] | E.C. Han, S.B. Ryoo, J.W. Park, J.W. Yi, H.K. Oh, E.K. Choe, H.K. Ha, B.K. Park, S.H. Moon, S.Y. Jeong and K.J. Park, Oncologic and surgical outcomes in colorectal cancer patients with liver cirrhosis: A propensity-matched study, PLoS One 12: ((2017) ), e0178920. |
[26] | Y. Liu, Serum proteomic pattern analysis for early cancer detection, Technol Cancer Res Treat 5: ((2006) ), 61–66. |
[27] | L.F. Grochola, T. Greither, H. Taubert, P. Moller, U. Knippschild, A. Udelnow, D. Henne-Bruns and P. Wurl, The stem cell-associated Hiwi gene in human adenocarcinoma of the pancreas: Expression and risk of tumour-related death, Br J Cancer 99: ((2008) ), 1083–1088. |
[28] | E.J. Lee, S. Banerjee, H. Zhou, A. Jammalamadaka, M. Arcila, B.S. Manjunath and K.S. Kosik, Identification of piRNAs in the central nervous system, RNA 17: ((2011) ), 1090–1099. |
[29] | S. Sivagurunathan, K. Palanisamy, J.P. Arunachalam and S. Chidambaram, Possible role of HIWI2 in modulating tight junction proteins in retinal pigment epithelial cells through Akt signaling pathway, Mol Cell Biochem 427: ((2017) ), 145–156. |
[30] | R.S. O’Neill and A. Stoita, Biomarkers in the diagnosis of pancreatic cancer: Are we closer to finding the golden ticket? World J Gastroenterol 27: ((2021) ), 4045–4087. |
[31] | M.L. Wong and J.F. Medrano, Real-time PCR for mRNA quantitation, Biotechniques 39: ((2005) ), 75–85. |
[32] | Y. Zhou, B. Zhou, L. Pache, M. Chang, A.H. Khodabakhshi, O. Tanaseichuk, C. Benner and S.K. Chanda, Metascape provides a biologist-oriented resource for the analysis of systems-level datasets, Nat Commun 10: ((2019) ), 1523. |
[33] | H. Xie, H. Ma and D. Zhou, Plasma HULC as a promising novel biomarker for the detection of hepatocellular carcinoma, Biomed Res Int 2013: ((2013) ), 136106. |
[34] | Y. Liu, X. Liu, Y. Gu and H. Lu, A novel RNA binding protein-associated prognostic model to predict overall survival in hepatocellular carcinoma patients, Medicine 100: ((2021) ), e26491. |
[35] | C. Noyes, S. Kitajima, F. Li, Y. Suita, S. Miriyala, S. Isaac, N. Ahsan, E. Knelson, A. Vajdi, T. Tani, T.C. Thai, D. Xu, J. Murai, N. Tapinos, C. Takahashi, D.A. Barbie and M. Yajima, The germline factor DDX4 contributes to the chemoresistance of small cell lung cancer cells, Commun Biol 6: ((2023) ), 65. |
[36] | B. Czech, M. Munafo, F. Ciabrelli, E.L. Eastwood, M.H. Fabry, E. Kneuss and G.J. Hannon, piRNA-guided genome defense: From biogenesis to silencing, Annu Rev Genet 52: ((2018) ), 131–157. |
[37] | G. Zeng, D. Zhang, X. Liu, Q. Kang, Y. Fu, B. Tang, W. Guo, Y. Zhang, G. Wei and D. He, Co-expression of Piwil2/Piwil4 in nucleus indicates poor prognosis of hepatocellular carcinoma, Oncotarget 8: ((2017) ), 4607–4617. |
[38] | C. Yuan, H. Qin, M. Ponnusamy, Y. Chen and Z. Lin, PIWI-interacting RNA in cancer: Molecular mechanisms and possible clinical implications (Review), Oncol Rep 46: ((2021) ), 209. |
[39] | X. Qu, J. Liu, X. Zhong, X. Li and Q. Zhang, PIWIL2 promotes progression of non-small cell lung cancer by inducing CDK2 and Cyclin A expression, J Transl Med 13: ((2015) ), 301. |
[40] | C. Su, Z.J. Ren, F. Wang, M. Liu, X. Li and H. Tang, PIWIL4 regulates cervical cancer cell line growth and is involved in down-regulating the expression of p14ARF and p53, FEBS Lett 586: ((2012) ), 1356–1362. |
[41] | H. Zhang, Y. Ren, H. Xu, D. Pang, C. Duan and C. Liu, The expression of stem cell protein Piwil2 and piR-932 in breast cancer, Surg Oncol 22: ((2013) ), 217–223. |
[42] | Y.M. Zhao, J.M. Zhou, L.R. Wang, H.W. He, X.L. Wang, Z.H. Tao, H.C. Sun, W.Z. Wu, J. Fan, Z.Y. Tang and L. Wang, HIWI is associated with prognosis in patients with hepatocellular carcinoma after curative resection, Cancer 118: ((2012) ), 2708–2717. |
[43] | D.I. Jacobs, Q. Qin, M.C. Lerro, A. Fu, R. Dubrow, E.B. Claus, A.T. DeWan, G. Wang, H. Lin and Y. Zhu, PIWI-Interacting RNAs in Gliomagenesis: Evidence from Post-GWAS and Functional Analyses, Cancer Epidemiol Biomarkers Prev 25: ((2016) ), 1073–1080. |
[44] | Y. Li, X. Wu, H. Gao, J.M. Jin, A.X. Li, Y.S. Kim, S.K. Pal, R.A. Nelson, C.M. Lau, C. Guo, B. Mu, J. Wang, F. Wang, J. Wang, Y. Zhao, W. Chen, J.J. Rossi, L.M. Weiss and H. Wu, Piwi-Interacting RNAs (piRNAs) Are Dysregulated in Renal Cell Carcinoma and Associated with Tumor Metastasis and Cancer-Specific Survival, Mol Med 21: ((2015) ), 381–388. |
[45] | S.S. Liu, N. Liu, M.Y. Liu, L. Sun, W.Y. Xia, H.M. Lu, Y.J. Fu, G.L. Yang, J.J. Bo, X.X. Liu, H. Feng, H. Wu, L.F. Li and J.X. Gao, An unusual intragenic promoter of PIWIL2 contributes to aberrant activation of oncogenic PL2L60, Oncotarget 8: ((2017) ), 46104–46120. |
[46] | H. Taubert, S. Wach, R. Jung, M. Pugia, B. Keck, S. Bertz, E. Nolte, R. Stoehr, J. Lehmann, C.H. Ohlmann, M. Stockle, B. Wullich and A. Hartmann, Piwil 2 expression is correlated with disease-specific and progression-free survival of chemotherapy-treated bladder cancer patients, Mol Med 21: ((2015) ), 371–380. |
[47] | O. Al-Janabi, S. Wach, E. Nolte, K. Weigelt, T.T. Rau, C. Stohr, W. Legal, S. Schick, T. Greither, A. Hartmann, B. Wullich and H. Taubert, Piwi-like 1 and 4 gene transcript levels are associated with clinicopathological parameters in renal cell carcinomas, Biochim Biophys Acta 1842: ((2014) ), 686–690. |
[48] | D. Li, X. Sun, D. Yan, J. Huang, Q. Luo, H. Tang and Z. Peng, Piwil2 modulates the proliferation and metastasis of colon cancer via regulation of matrix metallopeptidase 9 transcriptional activity, Exp Biol Med (Maywood) 237: ((2012) ), 1231–1240. |
[49] | M. Litwin, A. Szczepanska-Buda, D. Michalowska, J. Grzegrzolka, A. Piotrowska, A. Gomulkiewicz, A. Wojnar, P. Dziegiel and W. Witkiewicz, Aberrant expression of PIWIL1 and PIWIL2 and their clinical significance in ductal breast carcinoma, Anticancer Res 38: ((2018) ), 2021–2030. |
[50] | R. Suzuki, S. Honda and Y. Kirino, PIWI expression and function in cancer, Front Genet 3: ((2012) ), 204. |
[51] | P. Dong, Y. Xiong, Y. Konno, K. Ihira, D. Xu, N. Kobayashi, J. Yue and H. Watari, Critical roles of PIWIL1 in human tumors: Expression, functions, mechanisms, and potential clinical implications, Front Cell Dev Biol 9: ((2021) ), 656993. |
[52] | F. Yang and J. Li, WHO classification of tumors of the breast, Zhonghua Wai Ke Za Zhi 52: ((2014) ), 1–3. |
[53] | P. Krishnan, S. Ghosh, K. Graham, J.R. Mackey, O. Kovalchuk and S. Damaraju, Piwi-interacting RNAs and PIWI genes as novel prognostic markers for breast cancer, Oncotarget 7: ((2016) ), 37944–37956. |
[54] | D. Meseure, S. Vacher, S. Boudjemaa, M. Lae, A. Nicolas, R. Leclere, W. Chemlali, G. Champenois, A. Schnitzler, L. Lesage, T. Dubois and I. Bieche, Biopathological significance of PIWI-piRNA pathway deregulation in invasive breast carcinomas, Cancers (Basel) 12: ((2020) ). |
[55] | Y. Liu, M. Dou, X. Song, Y. Dong, S. Liu, H. Liu, J. Tao, W. Li, X. Yin and W. Xu, The emerging role of the piRNA/piwi complex in cancer, Mol Cancer 18: ((2019) ), 123. |
[56] | A. Cai, Y. Hu, Z. Zhou, Q. Qi, Y. Wu, P. Dong, L. Chen and F. Wang, PIWI-Interacting RNAs (piRNAs): Promising applications as emerging biomarkers for digestive system cancer, Front Mol Biosci 9: ((2022) ), 848105. |
[57] | J. Feng, M. Yang, Q. Wei, F. Song, Y. Zhang, X. Wang, B. Liu and J. Li, Novel evidence for oncogenic piRNA-823 as a promising prognostic biomarker and a potential therapeutic target in colorectal cancer, J Cell Mol Med 24: ((2020) ), 9028–9040. |
[58] | R. Erber, J. Meyer, H. Taubert, P.A. Fasching, S. Wach, L. Haberle, P. Gass, R. Schulz-Wendtland, L. Landgraf, S. Olbricht, R. Jung, M.W. Beckmann, A. Hartmann and M. Ruebner, PIWI-Like 1 and PIWI-Like 2 Expression in Breast Cancer, Cancers (Basel) 12: ((2020) ). |
[59] | Y. Zeng, L.K. Qu, L. Meng, C.Y. Liu, B. Dong, X.F. Xing, J. Wu and C.C. Shou, HIWI expression profile in cancer cells and its prognostic value for patients with colorectal cancer, Chin Med J (Engl) 124: ((2011) ), 2144–2149. |
[60] | A. Fu, D.I. Jacobs, A.E. Hoffman, T. Zheng and Y. Zhu, PIWI-interacting RNA 021285 is involved in breast tumorigenesis possibly by remodeling the cancer epigenome, Carcinogenesis 36: ((2015) ), 1094–1102. |
[61] | X. Gu, C. Wang, H. Deng, C. Qing, R. Liu, S. Liu and X. Xue, Exosomal piRNA profiling revealed unique circulating piRNA signatures of cholangiocarcinoma and gallbladder carcinoma, Acta Biochim Biophys Sin (Shanghai) 52: ((2020) ), 475–484. |
[62] | D. Mai, Y. Zheng, H. Guo, P. Ding, R. Bai, M. Li, Y. Ye, J. Zhang, X. Huang, D. Liu, Q. Sui, L. Pan, J. Su, J. Deng, G. Wu, R. Li, S. Deng, Y. Bai, Y. Ligu, W. Tan, C. Wu, T. Wu, J. Zheng and D. Lin, Serum piRNA-54265 is a New Biomarker for early detection and clinical surveillance of Human Colorectal Cancer, Theranostics 10: ((2020) ), 8468–8478. |
[63] | A. Qu, W. Wang, Y. Yang, X. Zhang, Y. Dong, G. Zheng, Q. Wu, M. Zou, L. Du, Y. Wang and C. Wang, A serum piRNA signature as promising non-invasive diagnostic and prognostic biomarkers for colorectal cancer, Cancer Manag Res 11: ((2019) ), 3703–3720. |
[64] | N.A. Sabbah, W.M. Abdalla, W.A. Mawla, N. AbdAlMonem, A.F. Gharib, A. Abdul-Saboor, A.S. Abdelazem and N. Raafat, piRNA-823 is a unique potential diagnostic non-invasive biomarker in colorectal cancer patients, Genes (Basel) 12: ((2021) ). |
[65] | H. Tosun, A. Demirtas, G. Sonmez, S.T. Tombul, H. Akalin and Y. Ozkul, Can the expression level of PIWIL 2 gene be a serum marker for prostate cancer? A single-center prospective study, Turk J Urol 45: ((2019) ), S22–S25. |
[66] | F. Rizzo, A. Rinaldi, G. Marchese, E. Coviello, A. Sellitto, A. Cordella, G. Giurato, G. Nassa, M. Ravo, R. Tarallo, L. Milanesi, A. Destro, G. Torzilli, M. Roncalli, L. Di Tommaso and A. Weisz, Specific patterns of PIWI-interacting small noncoding RNA expression in dysplastic liver nodules and hepatocellular carcinoma, Oncotarget 7: ((2016) ), 54650–54661. |
[67] | F. Muscari and C. Maulat, Preoperative alpha-fetoprotein (AFP) in hepatocellular carcinoma (HCC): Is this 50-year biomarker still up-to-date? Transl Gastroenterol Hepatol 5: ((2020) ), 46. |
[68] | I. Riquelme, P. Perez-Moreno, P. Letelier, P. Brebi and J.C. Roa, The Emerging Role of PIWI-Interacting RNAs (piRNAs) in Gastrointestinal Cancers: An updated perspective, Cancers (Basel) 14: ((2021) ). |
[69] | Z. Dong, H. Wu, Y. He, Z. Huang, Y. Weng, H. Li, C. Liang, W. Yu and W. Chen, MiRNA-124-3p.1 sensitizes hepatocellular carcinoma cells to sorafenib by regulating FOXO3a by targeting AKT2 and SIRT1, Cell Death Dis 13: ((2022) ), 35. |
[70] | Y. Liu, J. Tan, S. Ou, J. Chen and L. Chen, MicroRNA-101-3p suppresses proliferation and migration in hepatocellular carcinoma by targeting the HGF/c-Met pathway, Invest New Drugs 38: ((2020) ), 60–69. |
[71] | Z. Xiao, J. Shen, L. Zhang, M. Li, W. Hu and C. Cho, Therapeutic targeting of noncoding RNAs in hepatocellular carcinoma: Recent progress and future prospects (Review), Oncol Lett, (2018) . |
[72] | P. Apostolou, M. Toloudi and I. Papasotiriou, Identification of genes involved in breast cancer and breast cancer stem cells, Breast Cancer: Targets and Therapy 7: ((2015) ), 183. |




