miR-370 regulates ISG15 expression and influences IFN-α sensitivity in hepatocellular carcinoma cells
Abstract
BACKGROUND:
Interferon-
METHODS:
We performed loss-of-function and gain-of-function experiments to examine the role of ISG15 in the IFN-
RESULTS:
The overexpression of ISG15 reduced apoptosis in Huh7 and LH86 cells in the presence of IFN-
CONCLUSIONS:
Our findings suggest that miR-370 functions as an HCC tumor suppressor and regulator of IFN-
1.Introduction
Hepatocellular carcinoma (HCC) ranks as the second and sixth leading world-wide cause of cancer-related death in men and women, respectively [1]. About 80% to 90% of HCC patients have advanced disease at the time of diagnosis, and are thus ineligible for surgical intervention [2]. Therefore, the development of effective chemotherapy strategies for HCC is a critical research objective. Studies of melanoma and cancer of the breast, stomach, pancreas and bladder have reported the beneficial effects of using IFN-
The majority of HCC cases are chronically infected with the hepatitis B virus (HBV) or hepatitis C virus (HCV) [9]. Although the induction of microRNA (miRNA) expression by type I IFNs contributes to the regulation of cellular antiviral defenses [10], altered miRNA expression has also been reported in various types of cancer in humans [11]. One such miRNA is miR-370, which has been shown to suppress HBV replication [12]. Aberrant miR-370 expression has been reported in esophageal squamous cell carcinoma [13], laryngeal squamous cell carcinoma [14], gastric carcinoma [15], and prostate cancer [16], as well as in cholangiocarcinoma cell lines [17, 18]. Although miR-370 serves as a tumor suppressor in malignant cholangiocytes [19] and laryngeal carcinoma cells [14], the overexpression of miR-370 contributes to the progression of gastric carcinoma [15], prostate cancer [16], and acute myeloid leukemia [20]. The biological mechanisms through which miR-370 is involved in tumorigenesis in this wide range of tissue types remains unclear.
The expression of IFN-stimulated gene 15 (ISG15) and ISG15-mediated conjugation have been implicated in malignancies of the breast [21], prostate [22], bladder cancer [23], oral cavity [24], esophagus [25], stomach [26], pancreas [27], colon [28], and liver [29], but how ISG15 influences tumorigenesis is unclear. Although ISG15, a ubiquitin-like protein, forms conjugates with various cellular proteins via a pathway similar to that used by ubiquitin, conjugation by ISG15 does not induce proteasome-dependent degradation of the conjugated protein [30, 31], and the biological consequences of ISG15 conjugation are unknown. The expression of ISG15 is induced by IFN-
In our previous study, we found that ISG15 is a novel prognostic biomarker for HCC in patients with chronic HBV infection [34]. In our current study, we performed ISG15 loss-of-function and gain-of-function experiments to examine its role in the sensitivity of various HCC cell lines to treatment with IFN-
2.Materials and methods
2.1Tissues, cell lines and antibodies
The Hunan Provincial Cancer Hospital Review Board approved the protocol for the analysis of HCC tumor and noncancerous liver tissue specimens. The HCC tumor tissues and adjacent noncancerous tissue samples were collected at the Hunan Provincial Tumor Hospital (Changsha, China). Informed written consent was obtained from all patients prior to collection. The human HCC cell lines, HLCZ01, LH86, LO2, Huh7 and SMMC7721 were obtained from the Translational Medicine Research Center at Hunan University, and were grown in Dulbecco Modified Eagle Medium (DMEM, Life Technologies, Carlsbad, CA, USA) with 10% fetal bovine serum at a temperature of 37
2.2Plasmids, oligonucleotides and reagents
Transfections for the knock down of ISG15 were performed using the siPORT NeoFx reagent (ThermoFisher, Waltham, MA, USA). The anti-ISG15 siRNA and randomized negative control RNA oligonucleotide were purchased from Santa Cruz Biotechnology. The pre-miR miRNA precursor molecule and anti-miR miRNA inhibitor for miRNA-370 (henceforth referred to as miR-370 and anti-miR-370, respectively) and the corresponding negative control RNAs were purchased from Ambion (ThermoFisher). The pFLAG-ISG15 plasmid was constructed by inserting the ISG15 cDNA into the multiple cloning site of the pFLAG-CMV-2 plasmid (Sigma-Aldrich), which allowed the expression of FLAG-tagged ISG15 protein in transfected cells. For siRNA knock down of ISG15 expression, the pSilencer-shISG15 plasmid was constructed based on the pSilencer system (ThermoFisher), which expressed a 21-nt short hairpin RNA with a 9-nt loop. The ISG15 shRNA-coding sequences used to construct pSilencer-ISG15 were as follows: 5’-GATCCCCGCACCTACGAGGTACGGCTTTCAAGA GAAGCCGTACCTCGTAGGTGCTTTTTA-3’ (sense); 5’-AGCTTAAAAAGCACCTACGAGGTACGGCTTCTCTTGAAAGCCGTACCTCGTAGGTGCGGG-3’ (antisense).
2.3Cell viability assay
We examined the IFN-
2.4Western blotting
Cells were washed twice with phosphate-buffered saline (PBS), and lysed in buffer containing 137 mM NaCl, 2 mM EDTA, 20 mM Tris-HCl (pH 7.4), 10% glycerin, 1% Triton X-100 and a protein inhibitor cocktail (Roche Diagnostics) for 30 min on ice. The lysate was centrifuged at 14,000
2.5Flow cytometry
Apoptosis in the LH86 and Huh7 cell lines was assessed using flow cytometry after transfection. Vehicle controls were added to maintain equivalent transfectant volumes, and 2,000 IU/mL IFN-
2.6miRNA target prediction
To investigate the mechanisms involved in the repression of ISG15 in IFN-
2.7Relative quantification of miRNA
Relative quantification of the level of miR-370 in human tumor tissues; the LH86, HLCZ01, L02, SMMC-7721, and Huh7 cell lines; and LH86- and Huh7-derived xenograft tumors was performed using qRT-PCR. Total RNA was isolated from tissues using the MagMAX mirVana Total RNA Isolation Kit (ThermoFisher), and miRNA was isolated from cultured cells using the TaqMan MicroRNA Cells-to-C
2.8Fluorescence microscopy
Apoptosis in the LH86 and Huh7 cell lines was assessed using fluorescence microscopy after transfection with the following: IFN-
2.9Tumor xenografting
Our animal protocols were approved by the Hunan Provincial Cancer Hospital Review Board (Changsha, China) and the Hunan Province Animal Care and Use Committee, and all of the animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals (National Institutes of Health, USA). All animals were housed under standard conditions in a pathogen-free, temperature and light-controlled facility. The LH86 and Huh7 cell lines were infected with adenovirus expressing miR-370 (Ad-miR-370; Vector Biolabs, Malvern, PA, USA) or control adenovirus expressing green fluorescent protein (Ad-GFP; Vector Biolabs). After antibiotic selection for 10 days, 5
2.10Statistical analysis
All statistical analyses were performed using SPSS software, version 17.0 (IBM, Armonk, NY, USA). Student
3.Results
3.1IFN-α
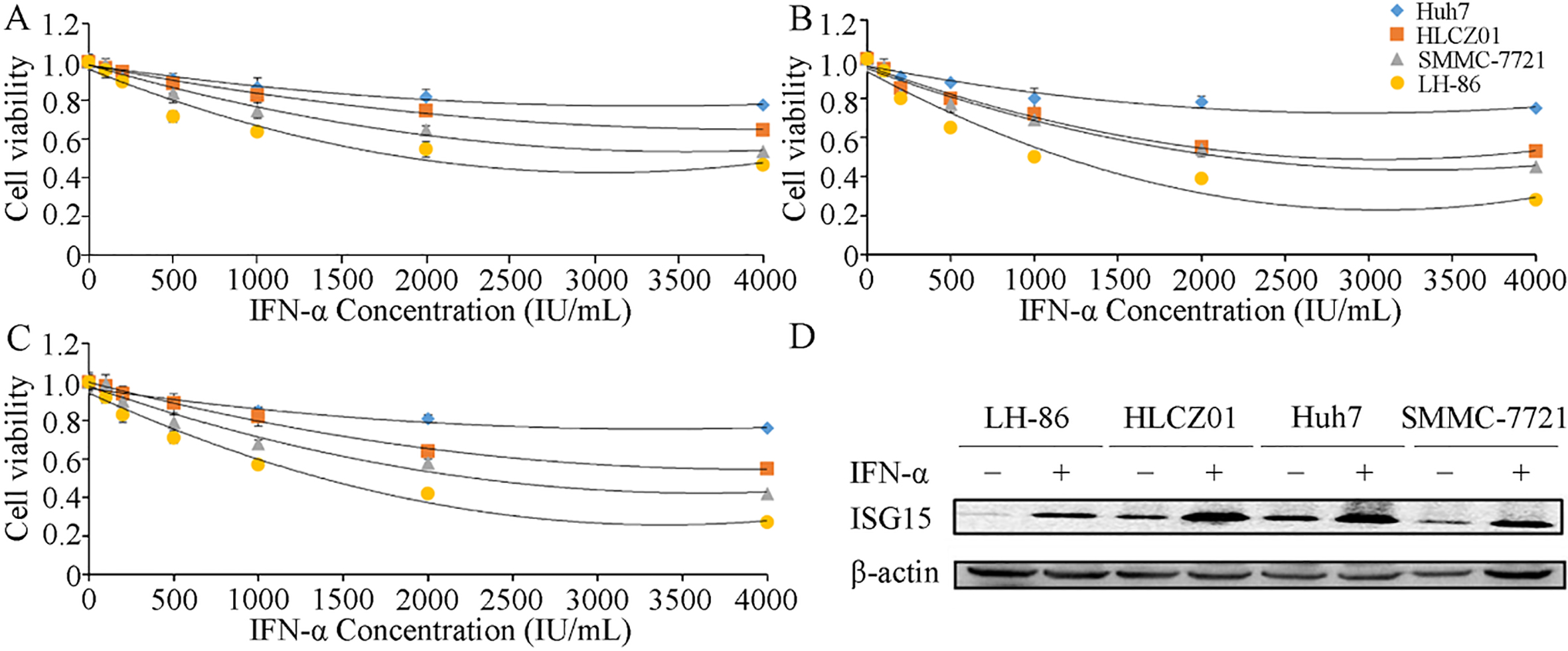
The expression of ISG15 is associated with the tumor grade, metastasis and survival in HCC patients [29]. Therefore, we evaluated ISG15 expression and apoptosis in LH86, HLCZ01, SMMC-7721 and Huh7 cells after treatment with various concentrations of IFN-
Figure 1.
Effects of IFN-
3.2ISG15 regulates the sensitivity of HCC cells to IFN-α
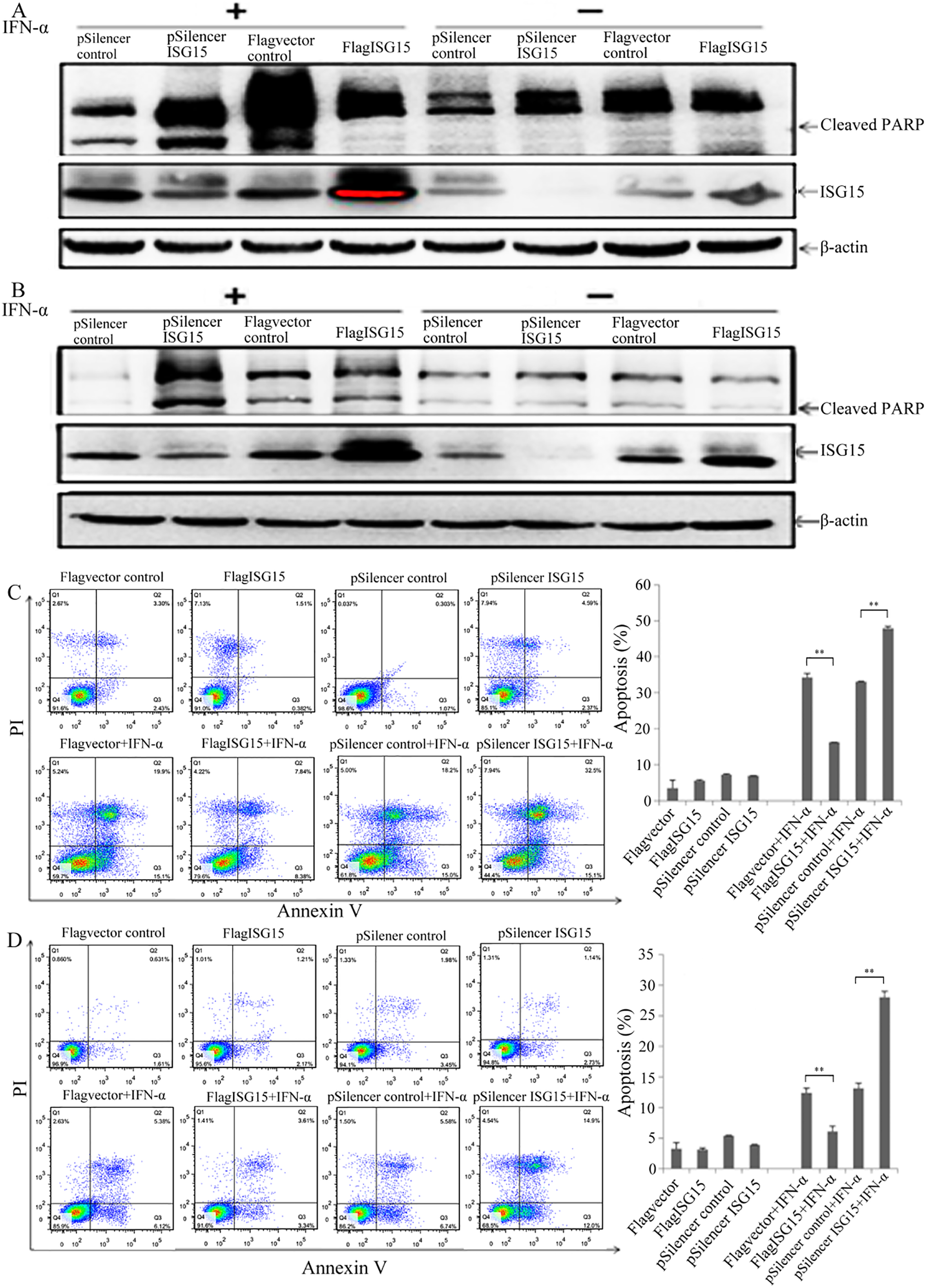
To investigate the role of ISG15 expression in IFN-
Figure 2.
ISG15 mediates IFN-
3.3ISG15 expression is regulated by interaction between miR-370 and ISG15 mRNA
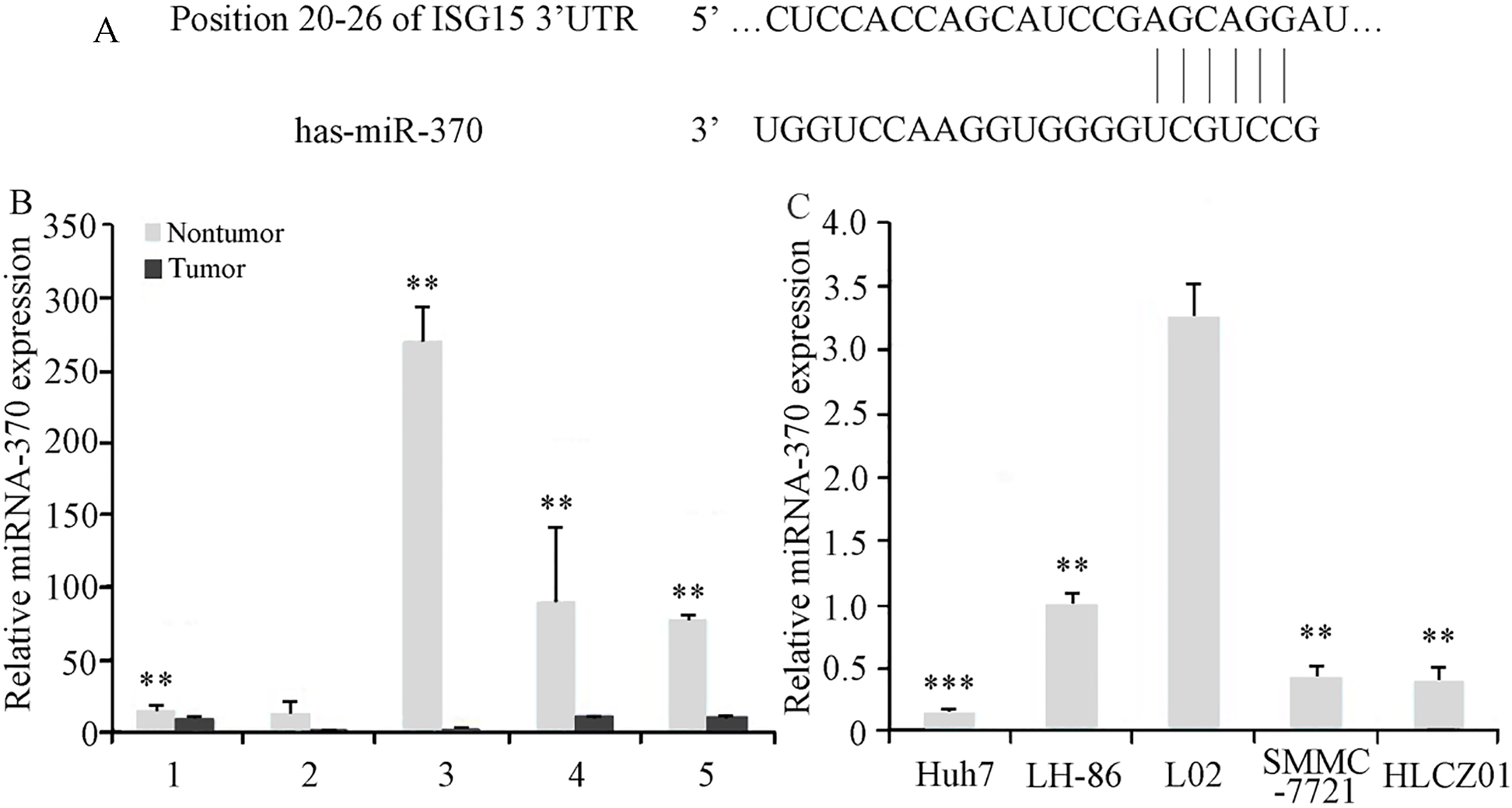
To investigate the factors involved in the regulation of ISG15 expression in HCC cells, we analyzed the ISG15 mRNA sequence to identify possible miRNA target sites. Our sequence analysis revealed a high level of complementarity between human miR-370 and a 7-bp sequence in the 3’-UTR of human ISG15 mRNA (Fig. 3A). Our analysis of miR-370 expression showed that the level of miR-370 was significantly lower in 4 of the 5 HCC tumor samples tested, compared to those in adjacent noncancerous tissues (Fig. 3B). The levels of miR-370 expression in the HCC cell lines, HLCZ01, LH86, Huh7, and SMMC7721, were also reduced, compared with the level of miR-370 in the irradiation immortalized, normal human liver cell line, LO2 (Fig. 3C). These results suggested that reduced miR-370 expression contributes to HCC progression.
Figure 3.
Identification of miR-370 target sequence in ISG15 mRNA, and analysis of miR-370 expression in HCC tumor tissues and cell lines. (A) The 3
Western blot analysis showed that transfection with miR-370 reduced ISG15 protein expression in Huh7 and LH86 cells without IFN-
Figure 4.
Effects of miR-370 on ISG15 expression and PARP cleavage. (A) Huh7 and LH86 cells were transfected with 25, 50 or 100 nmol miR-370 or anti-miR-370 siRNA for 24 h before assessing the levels of ISG15 and actin (loading control) proteins by western blotting. (B) After transfection with 100 nmol miR-370 and/or pFLAG-ISG15 for 24 h, LH86 and Huh7 cells were subjected to western blotting to assess the levels of cleaved PARP, ISG15, and actin proteins. (C and D) After treatment with or without 2,000 IU/mL IFN-
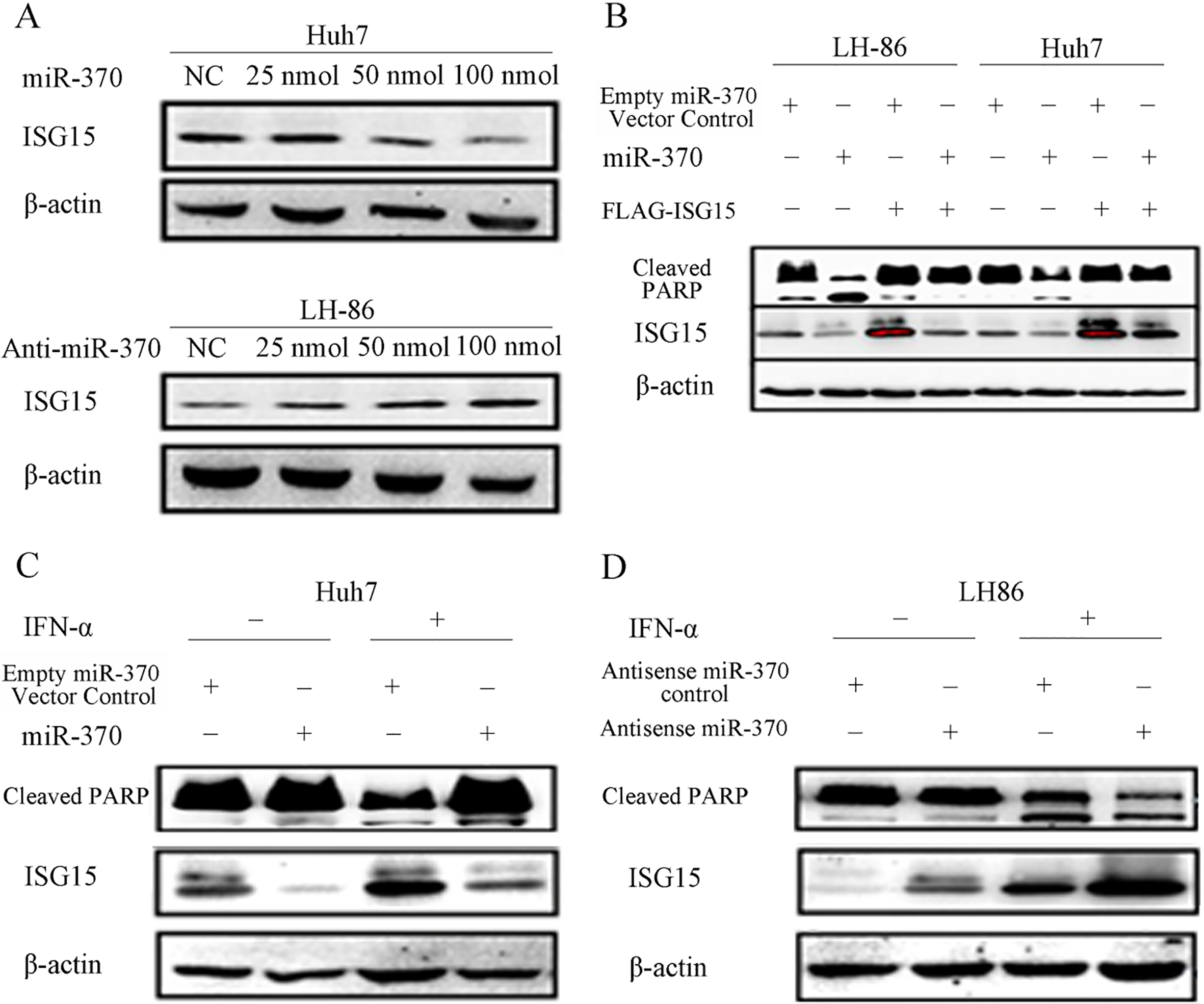
The combined results of our bioinformatic (Table S1) and western blot analyses suggested that miR-370 downregulates ISG15 expression by targeting the 3’-UTR of the ISG15 mRNA.
Figure 5.
The sensitivity of cancer cells to IFN-
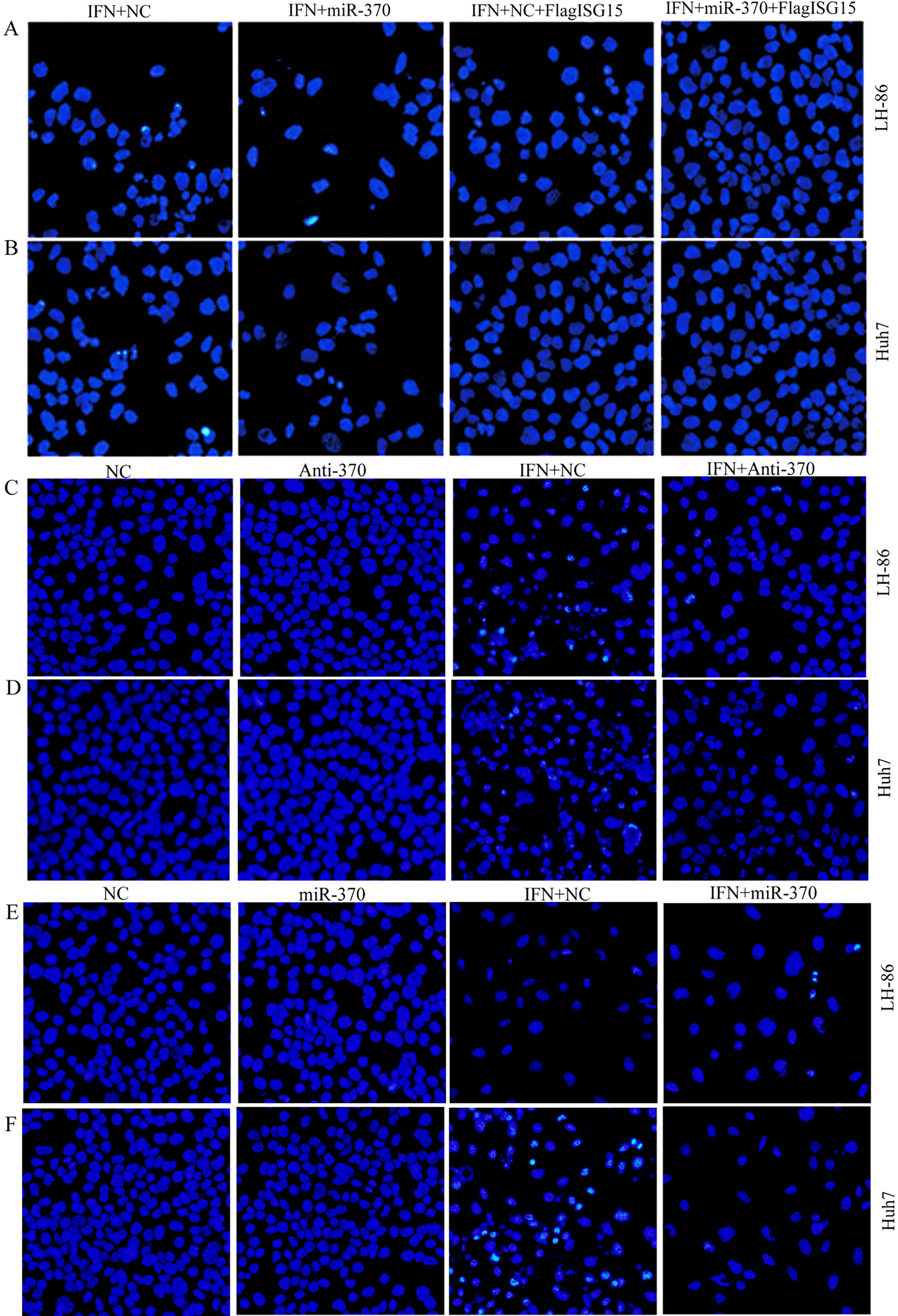
3.4Targeting of ISG15 by miR-370 modulates the IFN-α
The role of miR-370-mediated regulation of ISG15 expression in its sensitivity to IFN-
3.5miR-370 modulates IFN-α
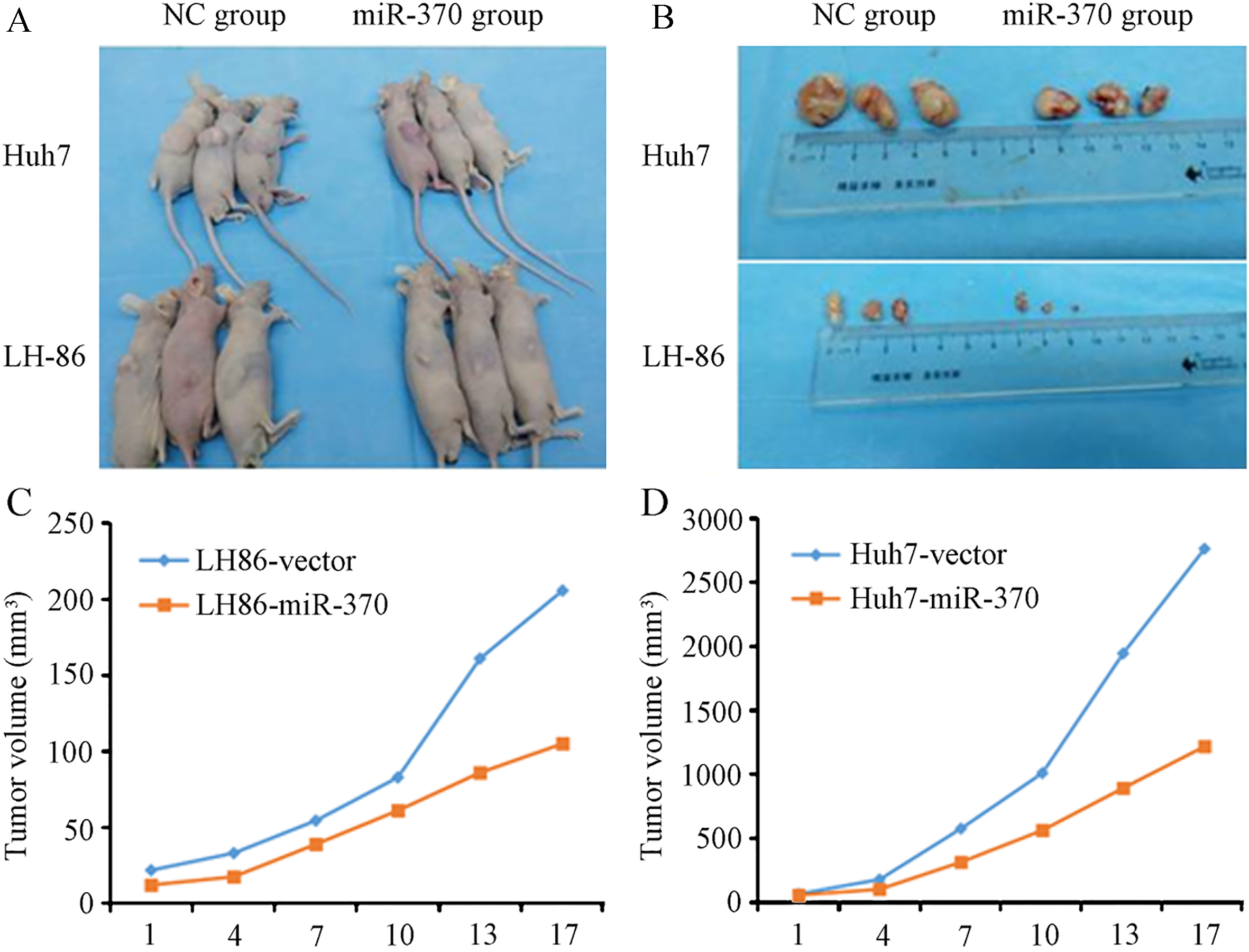
We performed HCC xenograft experiments in immunodeficient NOD/SCID mice to determine whether the effects of miR-370 on ISG15-mediated IFN-
Figure 6.
MiR-370 modulates the sensitivity of cancer cells to IFN-
4.Discussion
The pleiotropic cytokine IFN-
We found that IFN-
Our bioinformatic analysis suggested that miR-370 downregulated ISG15 expression in Huh7 and LH86 cells (Fig. 4) through a putative interaction between miR-370 and the 3’-UTR of ISG15 mRNA (Fig. 3). Xu et al. showed that miR-370 expression gradually decreased over the course of HCC tumorigenesis in rats, and they also showed that reduced expression of miR-370 in tumor tissue samples was associated with tumor progression, angiogenesis, microsatellite tumors, capsular invasion, and poor prognosis in HCC patients [41]. We observed significantly lower levels of miR-370 in all the HCC cell lines examined and in most of the HCC tumor samples, compared with LO2 cells (normal hepatocytes) and noncancerous adjacent tissues, respectively (Fig. 3). We also showed that miR-370 expression reduced the expression of ISG15 and increased apoptosis in Huh7 and LH86 cells treated with IFN-
Li et al. have also shown that increased ISG15 expression was associated with tumor progression, metastasis and poor survival in HCC patients without IFN-
Figure 7.
Scheme of the study findings. INF-
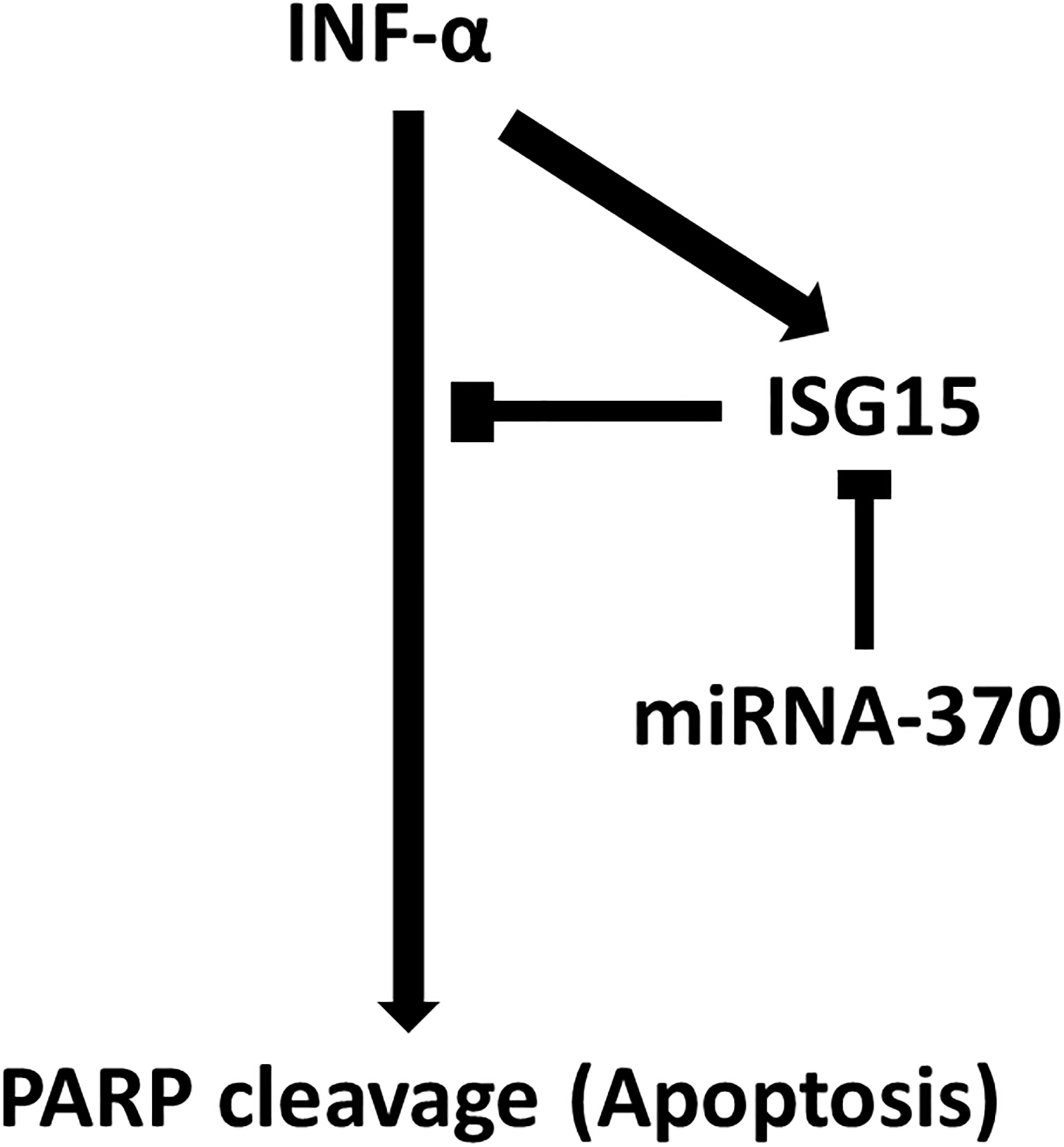
In conclusion, our results showed that miR-370 downregulates IFN-
Our findings suggest that the miR-370 functions as a tumor suppressor in HCC cells, and might be a useful prognostic marker for HCC, especially in patients with IFN-
Acknowledgments
This work was supported by a grant from the Changsha City Third Science and Technology Project (grant number K1403380-31), and the National Natural Science Foundation of China (grant number 816701030).
Conflict of interest
The authors have no conflicts of interest.
References
[1] | L. Torre, F. Bray, R. Siegel, J. Ferlay, J. Lortet-Tieulent and A. Jemal, Global cancer statistics, 2012, CA: A Cancer Journal for Clinicians 65: ((2015) ), 87. |
[2] | S.T. Fan, C. Mau Lo, R.T. Poon, C. Yeung, C. Leung Liu, W.K. Yuen, C. Ming Lam, K.K. Ng and S. Ching Chan, Continuous improvement of survival outcomes of resection of hepatocellular carcinoma: a 20-year experience, Ann Surg 253: ((2011) ), 745–58. |
[3] | M. Durante, N. Reppingen and K. Held, Immunologically augmented cancer treatment using modern radiotherapy, Trends in Molecular Medicine 19: ((2013) ), 565. |
[4] | C.A. Barker and M.A. Postow, Combinations of Radiotherapy and Immunotherapy for Melanoma: A Review of Clinical Outcomes, International Journal of Radiation Oncology, Biology, Physics 88: ((2014) ), 986. |
[5] | F. Rocha, Y. Hashimoto, L. Traverso, R. Dorer, R. Kozarek, W. Helton and V. Picozzi, Interferon-based Adjuvant Chemoradiation for Resected Pancreatic Head Cancer: Long-term Follow-up of the Virginia Mason Protocol, Annals of Surgery 263: ((2016) ), 376. |
[6] | D. Lamm, M. Brausi, M.A. O’Donnell and J.A. Witjes, Interferon alfa in the treatment paradigm for non-muscle-invasive bladder cancer, Urol Oncol 32: ((2014) ), 35 e21–30. |
[7] | H. Sun, Z. Tang, L. Wang, L. Qin, Z. Ma, Q. Ye, B. Zhang, Y. Qian, Z. Wu and J. Fan, Postoperative interferon alpha treatment postponed recurrence and improved overall survival in patients after curative resection of HBV-related hepatocellular carcinoma: a randomized clinical trial, Journal of Cancer Research and Clinical Oncology 132: ((2006) ), 458. |
[8] | V. Mazzaferro, R. Romito, M. Schiavo, L. Mariani, T. Camerini, S. Bhoori, L. Capussotti, F. Calise, R. Pellicci and G. Belli, Prevention of hepatocellular carcinoma recurrence with alpha-interferon after liver resection in HCV cirrhosis, Hepatology (Baltimore, Md.) 44: ((2006) ), 1543. |
[9] | A.G. Miamen, H. Dong and L.R. Roberts, Immunotherapeutic approaches to hepatocellular carcinoma treatment, Liver Cancer 1: ((2012) ), 226–37. |
[10] | I.M. Pedersen, G. Cheng, S. Wieland, S. Volinia, C.M. Croce, F.V. Chisari and M. David, Interferon modulation of cellular microRNAs as an antiviral mechanism, Nature 449: ((2007) ), 919. |
[11] | S.P. Nana-Sinkam and C.M. Croce, Clinical applications for microRNAs in cancer, Clin Pharmacol Ther 93: ((2013) ), 98–104. |
[12] | H. Fan, P. Lv, J. Lv, X. Zhao, M. Liu, G. Zhang and H. Tang, miR-370 suppresses HBV gene expression and replication by targeting nuclear factor IA, Journal of Medical Virology ((2016) ). |
[13] | C. Zhang, C. Wang, X. Chen, C. Yang, K. Li, J. Wang, J. Dai, Z. Hu, X. Zhou and L. Chen, Expression profile of microRNAs in serum: a fingerprint for esophageal squamous cell carcinoma, Clinical Chemistry 56: ((2010) ), 1871. |
[14] | W. Yungang, L. Xiaoyu, T. Pang, L. Wenming and X. Pan, miR-370 targeted FoxM1 functions as a tumor suppressor in laryngeal squamous cell carcinoma (LSCC), Biomed Pharmacother 68: ((2014) ), 149–154. |
[15] | C. Fan, S. Liu, Y. Zhao, Y. Han, L. Yang, G. Tao, Q. Li and L. Zhang, Upregulation of miR-370 contributes to the progression of gastric carcinoma via suppression of FOXO1, Biomed Pharmacother 67: ((2013) ), 521–526. |
[16] | Z. Wu, H. Sun, W. Zeng, J. He and X. Mao, Upregulation of MircoRNA-370 induces proliferation in human prostate cancer cells by downregulating the transcription factor FOXO1, PLoS One 7: ((2012) ), e45825. |
[17] | F. Meng, H. Wehbe-Janek, R. Henson, H. Smith and T. Patel, Epigenetic regulation of microRNA-370 by interleukin-6 in malignant human cholangiocytes, Oncogene 27: ((2008) ), 378. |
[18] | A.V. Olaru, G. Ghiaur, S. Yamanaka, D. Luvsanjav, F. An, I. Popescu, S. Alexandrescu, S. Allen, T.M. Pawlik and M. Torbenson, A microRNA downregulated in human cholangiocarcinoma controls cell cycle through multiple targets involved in the G1/S checkpoint, Hepatology (Baltimore, Md.) 54: ((2011) ), 2089. |
[19] | C.M. Tracy, A.E. Epstein, D. Darbar, J.P. Dimarco, S.B. Dunbar, N.A. Estes, 3rd, T.B. Ferguson, Jr., S.C. Hammill, P.E. Karasik, M.S. Link, J.E. Marine, M.H. Schoenfeld, A.J. Shanker, M.J. Silka, L.W. Stevenson, W.G. Stevenson and P.D. Varosy, 2012 ACCF/AHA/HRS focused update of the 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, J Am Coll Cardiol 60: ((2012) ), 1297–313. |
[20] | A. Dixon-McIver, P. East, C.A. Mein, J.B. Cazier, G. Molloy, T. Chaplin, T. Andrew Lister, B.D. Young and S. Debernardi, Distinctive patterns of microRNA expression associated with karyotype in acute myeloid leukaemia, PLoS One 3: ((2008) ), e2141. |
[21] | A. Hadjivasiliou, ISG15 implicated in cytoskeleton disruption and promotion of breast cancer, Expert Rev Proteomics 9: ((2012) ), 7. |
[22] | L.M. Wood, Z.K. Pan, M.M. Seavey, G. Muthukumaran and Y. Paterson, The ubiquitin-like protein, ISG15, is a novel tumor-associated antigen for cancer immunotherapy, Cancer Immunol Immunother 61: ((2012) ), 689–700. |
[23] | J.B. Andersen, M. Aaboe, E.C. Borden, O.G. Goloubeva, B.A. Hassel and T.F. Orntoft, Stage-associated overexpression of the ubiquitin-like protein, ISG15, in bladder cancer, Br J Cancer 94: ((2006) ), 1465–71. |
[24] | V.K. Vincent-Chong, S.M. Ismail, Z.A. Rahman, N.A. Sharifah, A. Anwar, P.J. Pradeep, A. Ramanathan, L.P. Karen-Ng, T.G. Kallarakkal, W.M. Mustafa, M.T. Abraham, K.K. Tay and R.B. Zain, Genome-wide analysis of oral squamous cell carcinomas revealed over expression of ISG15, Nestin and WNT11, Oral Dis 18: ((2012) ), 469–76. |
[25] | J. Tao, P. Hua, J. Wen, Y. Hu, H. Yang and X. Xie, Prognostic value of ISG15 mRNA level in drinkers with esophageal squamous cell cancers, International Journal of Clinical and Experimental Pathology 8: ((2015) ), 10975. |
[26] | S. Yang, H.-C. Jeung, H.J. Jeong, Y.H. Choi, J.E. Kim, J.-J. Jung, S.Y. Rha, W.I. Yang and H.C. Chung, Identification of genes with correlated patterns of variations in DNA copy number and gene expression level in gastric cancer, Genomics 4: ((2007) ), 451–459. |
[27] | B. Sainz, B. Martín, M. Tatari, C. Heeschen and S. Guerra, ISG15 Is a Critical Microenvironmental Factor for Pancreatic Cancer Stem Cells, Cancer Research ((2014) ). |
[28] | S.D. Desai, A.L. Haas, L.M. Wood, Y.-C. Tsai, S. Pestka, E.H. Rubin and A. Saleem, Elevated expression of ISG15 in tumor cells interferes with the ubiquitin/26S proteasome pathway, Cancer Research 66: ((2006) ), 921–928. |
[29] | C. Li, J. Wang, H. Zhang, M. Zhu, F. Chen, Y. Hu, H. Liu and H. Zhu, Interferon-stimulated Gene 15 (ISG15) is a trigger for tumorigenesis and metastasis of hepatocellular carcinoma, Oncotarget 5: ((2014) ), 8429. |
[30] | N.V. Giannakopoulos, J.K. Luo, V. Papov, W. Zou, D.J. Lenschow, B.S. Jacobs, E.C. Borden, J. Li, H.W. Virgin and D.E. Zhang, Proteomic identification of proteins conjugated to ISG15 in mouse and human cells, Biochem Biophys Res Commun 336: ((2005) ), 496–506. |
[31] | C. Zuo, X. Sheng, M. Ma, M. Xia and L. Ouyang, ISG15 in the tumorigenesis and treatment of cancer: An emerging role in malignancies of the digestive system, Oncotarget 7: ((2016) ), 74393–74409. |
[32] | H. Zhu, H. Zhao, C. Collins, S. Eckenrode, Q. Run, R. McIndoe, J. Crawford, D. Nelson, J. She and C. Liu, Gene expression associated with interferon alfa antiviral activity in an HCV replicon cell line, Hepatology (Baltimore, Md.) 37: ((2003) ), 1180. |
[33] | U.F. Cajee, R. Hull and M. Ntwasa, Modification by ubiquitin-like proteins: significance in apoptosis and autophagy pathways, Int J Mol Sci 13: ((2012) ), 11804–31. |
[34] | X. Qiu, Y. Hong, D. Yang, M. Xia, H. Zhu, Q. Li, H. Xie, Q. Wu, C. Liu and C. Zuo, ISG15 as a novel prognostic biomarker for hepatitis B virus-related hepatocellular carcinoma, Int J Clin Exp Med 8: ((2015) ), 17140–50. |
[35] | K. Herzer, T. Hofmann, A. Teufel, C. Schimanski, M. Moehler, S. Kanzler, H. Schulze-Bergkamen and P. Galle, IFN-alpha-induced apoptosis in hepatocellular carcinoma involves promyelocytic leukemia protein and TRAIL independently of p53, Cancer research 69: ((2009) ), 855. |
[36] | W. Jiang, C. Zhang, Z. Tian and J. Zhang, hIFN-alpha gene modification augments human natural killer cell line anti-human hepatocellular carcinoma function, Gene Ther 20: ((2013) ), 1062–1069. |
[37] | L. Wang, W. Wu, H. Sun, X. Wu, L. Qin, Y. Liu, K. Liu and Z. Tang, Mechanism of interferon alpha on inhibition of metastasis and angiogenesis of hepatocellular carcinoma after curative resection in nude mice, Journal of Gastrointestinal Surgery: Official Journal of the Society for Surgery of the Alimentary Tract 7: ((2003) ), 587. |
[38] | T. Leung, Y. Patt, W. Lau, S. Ho, S. Yu, A. Chan, T. Mok, W. Yeo, C. Liew and N. Leung, Complete pathological remission is possible with systemic combination chemotherapy for inoperable hepatocellular carcinoma, Clinical cancer Research: An Official Journal of the American Association for Cancer Research 5: ((1999) ), 1676. |
[39] | T. Urabe, S. Kaneko, E. Matsushita, M. Unoura and K. Kobayashi, Clinical pilot study of intrahepatic arterial chemotherapy with methotrexate, 5-fluorouracil, cisplatin and subcutaneous interferon-alpha-2b for patients with locally advanced hepatocellular carcinoma, Oncology 55: ((1998) ), 39. |
[40] | S. Kaneko, T. Urabe and K. Kobayashi, Combination chemotherapy for advanced hepatocellular carcinoma complicated by major portal vein thrombosis, Oncology 62: ((2002) ), 69. |
[41] | W.P. Xu, M. Yi, Q.Q. Li, W.P. Zhou, W.M. Cong, Y. Yang, B.F. Ning, C. Yin, Z.W. Huang, J. Wang, H. Qian, C.F. Jiang, Y.X. Chen, C.Y. Xia, H.Y. Wang, X. Zhang and W.F. Xie, Perturbation of MicroRNA-370/Lin-28 homolog A/nuclear factor kappa B regulatory circuit contributes to the development of hepatocellular carcinoma, Hepatology 58: ((2013) ), 1977–91. |
Appendices
Supplementary data
Table S1
The bioinformatic results of miR-370 downregulates ISG15 expression by targeting the 3’-UTR of the ISG15
| Predicted consequential pairing of | Seed | Site-type | 3’pairing | Local AU | Position | TA | SPS | Context | Context | Conserved | Per | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| target region (top) and miRNA (bottom) | match | contribution | contribution | contribution | contribution | contribution | contribution | score | percentile | branch length | ||
| Position 3–9 of ISG15 has-miR-650 | 5’ NNNNNNNNNNNNNNG 3’ CAGGACUCUCGCG | 7mer-1A | NA | NA | NA | NA | NA | NA | Too close to ORF | 0.073 | NA | |
| Position 3–9 of ISG15 has-miR-4443 | 5’ NNNNNNNNNNNNNNG 3’ UUUUGGGU | 7mer-m8 | NA | NA | NA | NA | NA | NA | Too close to ORF | 0.073 | NA | |
| Position 3–9 of ISG15 has-miR-3612 | 5’NNNNNNNNNNNNNNG 3’AGGUAAAGAGUUCU | 7mer-1A | NA | NA | NA | NA | NA | NA | Too close to ORF | 0.073 | NA | |
| Position 6–12 of ISG15 has-miR-3615 | 5’NNNNNNNNNNNGGGC 3’ACUCCAGUGUAACG | 7mer-1A | NA | NA | NA | NA | NA | NA | Too close to ORF | 0.073 | NA | |
| Position 7–13 of ISG15 has-miR-4456 | 5’NNNNNNNNNNGGGCC 3’UUUUCCUU | 7mer-m8 | NA | NA | NA | NA | NA | NA | Too close to ORF | 0.343 | NA | |
| Position 8–15 of ISG15 has-miR-138 | 5’ NNNNNNNNNGG 3’GCCGGACUAA | 8mer | NA | NA | NA | NA | NA | NA | Too close to ORF | 0.343 | ||
| Position 17–23 of ISG15 has-miR-423-3p | 5’GGCCUCCA 3’UGACUCCCCGGAGUC | 7mer-1A | 0.034 | 71 | 0.013 | NA | ||||||
| Position 20–26 of ISG15 has-miR-370 | 5’CUCCACCAG 3’UGGUCCAAG | 7mer-1A | 0.042 | 0.02 | 59 | 0.013 | NA | |||||
| Position 24–30 of ISG15 has-miR-640 | 5’ACCAGCAUCCGAGCA 3’UCUCCGUCCAAGGA | 7mer-1A | 0.053 | 52 | 0.368 | NA | ||||||
| Position 27–33 of ISG15 has-miR-4780 | 5’AGCAUCCGAGCAGGA 3’CGAUCCCUAGUCCG | 7mer-m8 | 0.048 | 76 | 0.073 | NA | ||||||
| Position 33–40 of ISG15 has-miR-1180 | 5’CGAGCAGGAUCAA 3’UGUGUGGGUGCG | 8mer | 0.003 | 0.043 | 97 | 0.013 | NA | |||||
| Position 34–40 of ISG15 has-miR-4664-3p | 5’GAGCAGGAUCAAGGG 3’CUGCCCCGAGUGUCU | 7mer-1A | 0.013 | 67 | 0.013 | NA | ||||||
| Position 54–61 of ISG15 has-miR-4753-3p | 5’AAUAAAGGCUGUUGU 3’UGUGUUCCGAUUUC | 8mer | 0.054 | 0.078 | 99 | 0.406 | NA |



