Impact of Intestinal Urinary Diversion on the Risk of Fracture and Loss of Bone Mass: A Systematic Review
Abstract
BACKGROUND:
Patients undergoing intestinal urinary diversion (IUD) may have a higher risk of osteoporosis and risk of fractures due to metabolic acidosis and decrease of intestinal absorption surface.
OBJECTIVE:
We performed a systematic review of the available literature on the impact of IUD on bone demineralization.
METHODS:
We systematically searched PubMed®, for original articles published before April 2020. Primary end points were the risk of fracture and loss of bone density. Secondary outcomes were the metabolic changes in biochemical and urine parameters related to calcium metabolism and histological changes.
RESULTS:
Our electronic search identified a total of 2417 articles. After a detailed review, we selected 11 studies that addressed the impact of IUD on bone health in 10369 patients. The risk of bone fracture was studied in 3 articles, showing a higher risk in the IUD population. Of the 9 articles evaluating the relation between intestinal urinary diversion and bone density, 5 did find a positive association. One article evaluated the bone metabolism at a cellular level after IUD showing a decrease in bone turnover in this population. Three of the eight studies reporting data on serum parameters related to calcium and phosphate metabolism showed differences. Finally, a correlation between concentration of pyridolines in urine and loss of bone density was found in two of the three studies.
CONCLUSIONS:
Although published data on BMD are contradictory, patients undergoing IUD seem to be at higher risk of bone fractures. Our finding support the need to implement accessible strategies on osteoporosis screening and prevention in IUD patients.
INTRODUCTION
Urinary tract reconstructions using intestinal segments are performed in patients undergoing cystectomy for bladder cancer (BCa) or benign terminal bladder dysfunction. The majority of intestinal urinary diversion (IUD) are created using the terminal ileum as it has the advantage of a lower rate of intra- and postoperative adverse events than other intestinal segments [1, 2]. This implies the exclusion of this part from the gastrointestinal tract affecting the absorption of certain nutrients [1, 3].
The contact of urine with the absorptive surface of the intestinal mucosa in IUD cause metabolic derangements owing to the reabsorption of urine solutes [4]. In normal situations, the ileum and the colon absorb urinary chloride and excrete bicarbonate into the intestinal lumen. When exposed to urine, bicarbonate moves into the lumen of the IUD while chloride (Cl) and ammonium are absorbed leading to chronic hyperchloremic acidosis [5]. In response to metabolic acidosis, bone buffers the excess of protons and release calcium, resulting in hypercalciuria, without a concomitant increase on intestinal calcium absorption: acidosis suppresses the activity of osteoblasts at the same time that the synthesis of the receptor activator of nuclear factor kappa B ligand (RANKL) increases, which stimulates osteoclastic activity and recruitment of new osteoclasts promoting bone resorption to buffer the proton load [6]. Metabolic acidosis, therefore, is associated with an increase in urine calcium excretion [7]. Moreover, ileal resection compromises calcium absorption because calcium is mostly absorbed in the ileum. Therefore, patients undergoing an IUD are at considerable risk of bone demineralization.
Also, age-related bone loss is progressive and can lead to osteoporosis [8] increasing the risk for fractures. It is known that patients experiencing low impact fractures secondary to osteoporosis are at significantly higher risk for death than their non-osteoporotic counterparts, and this persists for several years post-fracture [9, 10].
The current literature in this field is scarce with only a few studies assessing the impact of IUD on bone metabolism [11–13]. Most of the publications focused on different outcomes related to bone health with inconsistent results. Two recent big population-based studies, however, suggested that patients undergoing IUD are at higher risk of fracture [14, 15]. Therefore, the purpose of this systematic review is to provide an analysis of the available literature on the impact of IUD on bones including the risk of fracture, bone mineral density and other indirect measurements of bone demineralization.
MATERIAL AND METHODS
Evidence acquisition
The systematic review was performed according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [16]. The detailed PICO (Population, Intervention, Comparison and Outcomes) design is provided in the Section Inclusion Criteria.
Literature Search
PubMed database was searched in April 2020 to identify reports on the impact of IUD on bone health published between January 1990 and April 2020. The keywords used in our search strategy were: ((urinary diversion) OR (ileal conduit) OR (neobladder) OR (urinary reservoir)) AND ((fracture) OR (osteomalacia) OR (osteopenia) OR (osteoporosis) OR (bone mass) OR (bone mineral density) OR (bone mineralization) OR (bone metabolism) OR (complication)).
The primary outcomes of interest were the risk of fracture and loss of bone mass density (BMD). The secondary outcomes were the metabolic changes in laboratory parameters related to calcium metabolism as well as in urine and histomorphometric changes in bone architecture histological changes, such as trabecular bone volume (TBV).
Initial screening was performed independently by two investigators (CP and KR) based on the titles and abstracts to identify ineligible reports, and reasons for exclusions were noted. Potentially relevant reports were subjected to a full-text review and the relevance of the reports was also confirmed after the data extraction process. Disagreements were resolved via consensus with a third investigator (BP).
Inclusion and exclusion criteria
Studies were included if they investigated adult patients with trauma, neurogenic bladder or bladder cancer (Patients) undergoing IUD (Intervention) as compared with those not having that surgery (Comparators) to assess the impact on bone fractures or loss of bone mineral density (Outcome) in nonrandomized observational or cohort studies. Animal models, children undergoing urinary diversion were excluded. We also excluded reviews, letters, editorials, meeting abstracts, replies from authors, and case reports. No language restrictions were applied. In cases of duplicate publications, the most recent publication was selected. References of included manuscripts were further scanned for additional studies of interest.
Data extraction
Two investigators (CP and KR) independently extracted the following information from the included articles: first authors name, publication year, recruitment country, period of patient recruitment, number of patients, age, study design, the reason for urinary diversion, type of urinary diversion, control group in case there is one, follow-up duration, median age and type of test to determine bone health. All discrepancies regarding data extraction were resolved by consensus with a third investigator (BP).
Quality assessment
The Newcastle-Ottawa Scale (NOS) was used to assess the quality of the included studies following the Cochrane Handbook for systematic reviews of interventions for included non-randomized studies [17, 18]. The scale rates following three factors: Selection (1–4 points), Comparability (1–2 points) and Exposure (1–3 points), with total scores ranging from 0 (lowest) to 9 (highest). Studies with scores of more than 6 were identified as “high-quality” choices.
EVIDENCE SYNTHESIS
Literature search and characteristics of the included studies
Our initial search identified 2417 records, and after elimination of duplicates, 2411 articles were available (Fig. 1). A total of 2392 articles were excluded after screening the titles and abstracts, and a full-text review was performed for 19 articles. After applying the selection criteria, we identified 11 articles with 10369 patients for qualitative evidence. Table 1 summarizes the extracted data from the 11 studies, including the type of urinary diversion and number of patients. The median age and follow-up duration ranged from 50 to 72 years, and from 28.4 months to 8 years, respectively. Seven out of the 11 studies included only male patients. The studies had a median NOS score of 7 (4–7), as shown in Appendix 1.
Fig. 1
PRISMA Flow Chart of the studies included.
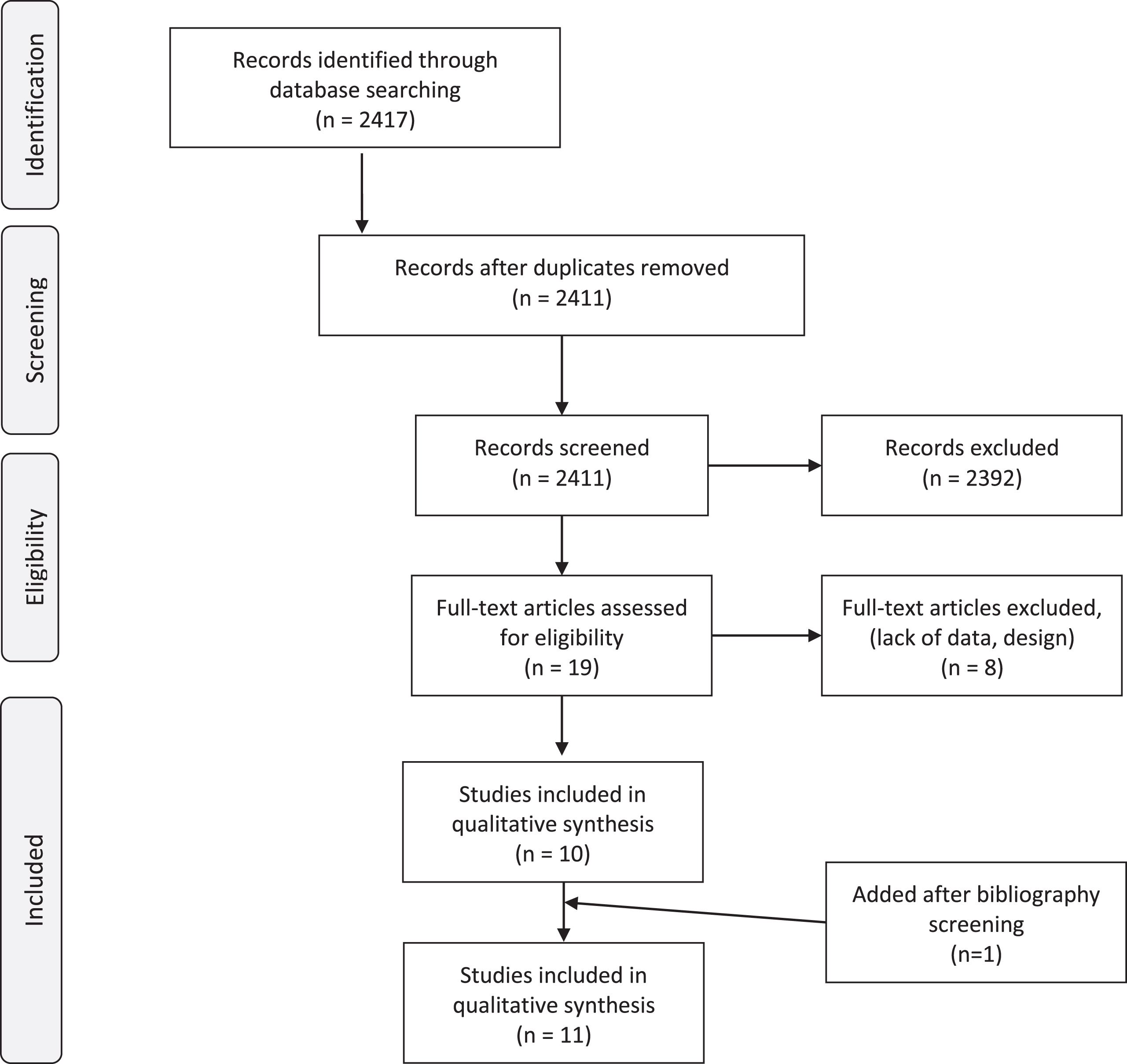
Table 1
Characteristics of the included studies
| Author | Journal | Year Publication | Country | Study type | Recruitment | n Total | ♂:♀ | n Study Cohort | Urinary diversion | n Diversion Group | Control cohort | n Control | Media age study cohort (years) | Median Follow up (years) | Outcome: Risk of Fracture | Outcome: BMD tests | Outcome: Serum markers | Outcome: Urin markers | Outcome: Histoiogical findings |
| Campanello M. | J Urol | 1996 | Swedan | Longitudinal prospective | NA | 48 | 48 | NA | NA | ✓ | ✓ | ✓ | |||||||
| 16:8 | Kock reservoir | 34 | 56 | 6,5 | |||||||||||||||
| 3:11 | Bricker | 14 | 72 | 6,1 | |||||||||||||||
| Davidsson T. | Urology | 1995 | Swedan | Longitudinal prospective | NA | 39 | 39 | NA | ✓ | ✓ | ✓ | ||||||||
| 15:5 | lleal or colonic conduit | 20 | 52 | 15,0 | |||||||||||||||
| 16:3 | cecal reservoir | 19 | 50 | 9,0 | |||||||||||||||
| Fujisawa M. | J Urol | 2000 | Japan | Longitudinal prospective | NA | 33 | 100% male | 33 | Orthotopic neobladder | 23 | NA | 62.8 | 2,4 | ✓ | ✓ | ✓ | |||
| sigmold | 23 | ||||||||||||||||||
| lleocolic | 8 | ||||||||||||||||||
| asc colon | 2 | ||||||||||||||||||
| Gannini S. | J amor Soc Nephrol | 1997 | Italy | Longitudinal prospective | 1987– 1995 | 41 | 100% male | 25 | Aged matched | 16 | 62.7 | 3,9 | ✓ | ✓ | ✓ | ||||
| Padovana | 25 | ||||||||||||||||||
| Gupta A. | JCO | 2014 | USA | population based-cohort | 2000– 2007 | 50520 | 37633:12887 | 4878 | Bca w/o IUD | 45642 | NA*> 66 | 3,4 | ✓ | ||||||
| continent | 721 | ||||||||||||||||||
| incontinent | 3892 | ||||||||||||||||||
| unknown | 265 | ||||||||||||||||||
| Incel N. | Int Urol Nephrol | 2006 | Turkey | Longitudinal retrospective | 1994– 2001 | 38 | 100% male | 19 | Matched control | 19 | 64 | ✓ | ✓ | ||||||
| Standford pouch | 11 | 60 | 4,9 | ||||||||||||||||
| lleal conduit | 8 | 68 | 3,0 | ||||||||||||||||
| Kawakita M. | J. Urol | 1996 | Japan | Longitudinal retrospective | 1984– 1993 | 359 | 100% male | 46 | Matched control | 313 | ✓ | ✓ | ✓ | ||||||
| Knock pouch | 20 | 58 | 5,2 | ||||||||||||||||
| indiana | 15 | 63 | 3,0 | ||||||||||||||||
| lleal conduit | 11 | 61 | 5,3 | ||||||||||||||||
| Poulsen A. | BJU | 1997 | Denmark | Longitudinal prospective | NA | 48 | 100% male | 32 | Healthy | 16 | 67 | 2,0 | ✓ | ||||||
| kock reservoir | 32 | 62 | |||||||||||||||||
| Richard P.O. | J. Urol | 2019 | Canada | population basedcohort | 1994– 2014 | 25894 | 5208 | No IUD | 20686 | ✓ | |||||||||
| 1020:3281//76:24 | Bca&IUD | 4301 | Bca&No IUD | 16772 | (1,79– 6,69 | ||||||||||||||
| 363:544//40:60 | IUD &No Bca | 907 | Healthy | 3914 | (5,57– 7,8) | ||||||||||||||
| Sevin G. | Eur Urol | 2002 | Turkey | Longitudinal retrospective | NA | 41 | 100% male | 27 | 14 | ✓ | |||||||||
| Indiana | 10 | 59.5 | 4,7 | ||||||||||||||||
| lleal neobladder | 17 | 58.6 | 2,8 | ||||||||||||||||
| Tschopp A. | J Urol | 1995 | Switzerland | Longitudinal retrospective | 1985– 1988 | 14 | 100% male | 14 | NA | 72.4 | (5– 8) | ✓ | |||||||
| Neobladder | 14 |
BMD: Bone mineral density; NA: not available; ✓: present.
Outcome 1: Risk of fracture
Three studies reported data on bone fractures in 10116 patients who underwent IUD [14, 15, 19]. Table 2 summarizes the results of these studies.
Table 2
Outcome 1: Risk of fracture on IUD patients
| Author | Cohort | n | Follow-up (years) | n events | %events | Median time to fracture (months) | Incidence/100 person-year | HR fracture any site |
| Gupta 2014 | IUD | 792 | 16 | 16,6 | 6,55 | 1,21 | ||
| Control | 10080 | 22 | 21,8 | 6,39 | REF | |||
| Richard 2019 | Bca + IUD | 4301 | 1,79 | 647 | 15,04 | 4,41 | 1,48 | |
| Bca w/o IUD | 16772 | 6,69 | 3373 | 20,11 | 2,63 | REF | ||
| IUD | 907 | 5,57 | 336 | 37,05 | 5,57 | 1,48 | ||
| healthy | 3914 | 7,80 | 1140 | 29,13 | 3,51 | REF | ||
| Campanello 1995 | Kock | 20 | 1 | |||||
| Bricker | 10 | 2 |
IUD: Intestinal urinary diversion; Bca: bladder cancer; w/o: without; REF: Reference.
Two population-based studies reported a significant increase in the risk of fractures in a population with BCa who had IUD in comparison with their counterparts without IUD [14, 15]. Additionally, Richard et al. reported a higher risk of fracture not only in a BCa population but also in non-BCa patients who also had an IUD [15].
Although it was not the primary objective of the study, Campanello et al. reported three patients with IUD who had a lumbar spine fracture out of 30 patients who underwent a plain X-Ray of the thoracolumbar spine [19].
In those studies, IUD seemed to be associated with an increase in risk fracture. Moreover, there was no statistical difference regarding the indication for the cystectomy.
Outcome 2: Bone mineral density tests
Nine studies, comprising a total of 267 patients undergoing IUD, analyzed the potential loss of bone mass in comparison with those without intervention [12, 13, 19–25].
Four studies reporting on 117 patients with IUD, did not find significant differences on BMD when compared with matched healthy population [13, 19–21]. Two of these groups reported no significant differences in bone mineral content (g/cm) using single-photon absorptiometry (SPA) and dual-energy x-ray absorptiometry (DEXA) [13, 21]. Similarly, the other two groups, did not find significant differences in bone mass density (g/cm2), between patients undergoing IUD and control, using either SPA or DEXA [19, 20].
On the contrary, five studies, comprising 150 patients, found an association of bone loss after IUD [12, 22–25]. Sevin et al compared a group of patients with neobladder using exclusively ileum segment (n = 17) and a group undergoing Indiana pouch, using ascendant colon (n = 10) with healthy controls (n = 14) [12]. They concluded that metabolic changes occur whenever bowel segments are interpoused in the urinary tract, as alkaline phosphatase levels were elevated in both neobladder groups compared to the control, but only the ileal neo-bladder group had a significant decrease bone density. A study including Stanford pouch (n = 9) and ileal conduit (n = 8) showed no differences in the Stanford group compared to the control group (n = 19), whereas patients with ileal conduit were osteopenic after IUD revealing a significant difference with the control group (Table 3) [25]. No difference in BMD or bone metabolism parameters were observed between the two diversion groups. Another study reporting on 46 patients with different types of IUD, found that patients with IUD and metabolic acidosis presented a significantly lower BMD [22]. The type of diversion did not result in a significant difference in metabolic acidosis or bone resorption. Patients undergoing Vesical Ileal Padovana reconstruction (n = 25) presented a significantly lower BMD in the femoral region compared to the control group[23]. Fujisawa et al. also found demineralization in spine and femur in men undergoing orthotopic neobladder using different intestinal segments (n = 23) as compared with the age-matched population [24].
Table 3
Oucome 2: Bone mineral density on IUD patients
| Author | Cohort | n Cohort analysed | SPA | DPA | DEXA | BMD Forearm | BMD Femoral | BMD Ward | BMD Lumbar | BMD whole body | Z-score Forearm | Z-score Femur | Z-score Lumber | T score Femur | T Score Lumber | T score < 2.5 Femur | T score < 2.5 Lumber |
| Campanello 1996 | ✓ | ✓ | |||||||||||||||
| Knock reservoir | 34 | 98±19% | 92±15% | 101±17% | 98±6% | –0.28±0.25 | –0.17±0.26 | ||||||||||
| lleal Conduit | 11* | 105±20% | 104±14% | 101±17% | 101±9% | –0.21±0.34 | 0,62±0.47 | ||||||||||
| Davidsson 1995 | ✓ | ✓ | |||||||||||||||
| Conduit (ileal and colonic) | 18 | 0.08±1.145 | –0.217±0.93 | –0.089±0.68 | |||||||||||||
| cecal reservoir | 15 | –0.157±0.839 | –0.485±1.065 | –0.399±1.058 | |||||||||||||
| Fujisawa 2000 | orthotopic neobladder | ✓ | –.82±0.99 | –0.35±1.031 | |||||||||||||
| sigmoid | 23 | ||||||||||||||||
| ileocolic | 8 | ||||||||||||||||
| ascendent colon | 2 | ||||||||||||||||
| Giannini 1997 | Padovana | 25 | ✓ | ||||||||||||||
| Padovana | 25 | 0.74±0.03* | 0.52±0.03 | 36% | 32% | ||||||||||||
| Control | 0.84±0.03 | 0.61±0.04 | |||||||||||||||
| Incel 2006 | ✓ | ||||||||||||||||
| Standford pouch | 11 | 0.99±0.11 | 1.11±0.14 | –0.63±0.82 | –0.9±1.18 | ||||||||||||
| lleal conduit | 8 | 0.90±0.11* | 1.02±0.13* | –1.43±0.86* | –1.76±1.14* | ||||||||||||
| control | 19 | 1.02±0.25 | 1.19±0.16 | –0.21±1.04 | –0.27±1.30 | ||||||||||||
| Kawakita 1996 | ✓ | ||||||||||||||||
| knock pouch | 20 | 0.52±0.19 | 0.19±0.22 | ||||||||||||||
| indiana | 15 | 0.61±0.37 | 0.30±0.30 | ||||||||||||||
| ileal conduit | 11 | 0.22±0.20 | 0.31±0.36 | ||||||||||||||
| Poulsen 1997 | ✓ | ✓ | |||||||||||||||
| ileal conduit | 25 | NA | NA | ||||||||||||||
| control | 16 | ||||||||||||||||
| Sevin 2002 | ✓ | ||||||||||||||||
| control | 14 | 0.9±0.1 | 1.2±0.2 | –0.6±0.5 | 0.6±1 | ||||||||||||
| ileal neobladder | 17 | 0.77±0.2* | 1±0.1* | –1.3±0.5* | –1.5±1.1* | ||||||||||||
| Indiana pouch | 10 | 0.8±0.2 | 1.1±0.4 | –0.8±0.9 | –0.9±1.1 | ||||||||||||
| Tschopp 1995 | ✓ | ||||||||||||||||
| ileal neobladder | 14 | 0.24±0.255 | 0.3±0.27 |
Outcome 3: Histology
Only one study analyzed bone metabolism at a cellular level after IUD [21]. Histomorphometric analysis was carried out after bone biopsy in 33 patients with IUD and compared with biopsies of 70 healthy patients. Trabecular bone volume (TBV), osteoid volume, osteoid surface, resorption surface and appositional rate were assessed. The analysis revealed greater TBV compared to normal values in the patient with a continent reservoir, but not in the conduit group. The mineral appositional rate (a measurement of the linear rate of new bone deposition), was significantly less in the two patients group than in the normal subjects, although there were no differences between both groups. These results may indicate a decrease in bone turnover in this population, with resulting reduction of osteoid formation. Osteoid volume and surface in this study tended to be rather on the low side, although within the normal range.
Outcome 4: Serum markers for bone metabolism
Eight studies including 239 patients with UID, reported data on serum parameters related to calcium and phosphate metabolism, such as osteocalcin, PTH, vitamin D, calcium or alkaline phosphatase [12, 19–25]. The mean values were within the normal ranges in five of the studies, as summarized in Table 4.
Table 4
Outcome 4: Serum markers for bone metabolism
| Study | Cohort | n Cohort analysed | Osteocalcin | 1.25 (OH)2 D3 | 25 (OH)2 D3 | PTH | PTH intact | FA | FA-bone specific | Ca total | Ionized Calcium |
| Campanello 1996 | μe/L | pg/mL | ng/mL | ng/L | μg/L | mmol/L | |||||
| Kock reservoir | 27 | 9.2±0.5 | 35±3 | 24±2 | 39±5 | 10.8±0.9 | 1.28±0.01 | ||||
| Bricker | 13 | 8,6±0.6 | 27±3 | 27±3 | 48.6±7.1 | 10±1.4 | 1.25±0.01 | ||||
| Davidsson T | μg/L | pmol/L | nmol/L | pmol/L | μcat/L | mmol/L | mmol/L | ||||
| Conduit | 17 | 3.2±1.6 | 53±12 | 63±18 | 4.4±3.0 | 3.5±0.8 | 2.33±0.08 | 1.26±0.04 | |||
| Reservoir | 18 | 3.7±1.1 | 69±29 | 57±24 | 2.9±1.1 | 3.0±0,7 | 2.31±0.09 | 1.24±0.05 | |||
| Fujisawa | NA | NA | U/L | NA | mg/L | NA | |||||
| Orthotopic neobladder | 33 | NA(↑2) | NA(↓2) | NA (normal in 33) | NA | 237±76 | NA | 9.3±0.4 | |||
| Giannini S | NA | NA | NA | pg/mL | NA | U/L | mmol/L | mmol/L | |||
| Padovana | 25 | 33.09±2.5 | 33.3±2.7 | 45.4±4.4 | 2.25±0.02 | 1.27±0.01 | |||||
| Control | 16 | 35.8±1.6 | 28.3±1.4 | 20.7±1.5 | 2.27±0.02 | 1.24±0.01 | |||||
| Incel et al | NA | NA | NA | pmol/L | U/L | mg/dL | |||||
| Standford pouch | 11 | 4.56±2.67 | 98.27±53.69 | 9.15±0.51 | |||||||
| Ileal conduit | 8 | 4.00±1,79 | 110.37±112.36 | 9.26±0.61 | |||||||
| Control | 19 | 3.03±0,96 | 71.05±15.90 | 9.25±0.55 | |||||||
| Kawakita | NA | NA | NA | NA | NA | IU/L | NA | mg/dL | |||
| kock pouch | 20 | 227±9.6 | 8.7±0.06 | ||||||||
| Indiana | 15 | 264±53.5 | 8.7±0.11 | ||||||||
| Ileal conduit | 11 | 216±22.9 | 8.6±22.9 | ||||||||
| Sevin et al | NA | NA | NA | pe/dL | NA | U/L | NA | mg/dL | |||
| Indiana | 10 | 47.3±9.5 | 121.4±43.6 | 9.3±0.6 | |||||||
| Ileal | 17 | 44.7±18.1 | 113.4±79.2 | 9.4±0.5 | |||||||
| control | 14 | 59.1±25.5 | 84.8±15.1 | 9.3±0.5 | |||||||
| Tschopp et al | NA | pg/mL | ng/mL | NA | pg/mL | U/L | mmol/L | ||||
| Ileal neobladder | 14 | 34.6(13.4–60.8) | 20.8 (8.7–27.3) | 37 (32–43) | 81(52–96) | 2.4 (2.09–2.54) |
Subgroup analysis showed significant differences in three of the studies [12, 23, 25]. Incel et al. found statistically significant higher PTH values in patients with Stanford pouch (which were not found in the ileal conduit group) compared to control subjects [25]. Alkaline phosphate or bone alkaline phosphate levels were significantly higher in patients undergoing IUD [12], as well as reported by Incel et al. in the subgroup of Stanford pouch (and not in the ileal conduit subgroup) [25].
Outcome 5: Urinary markers
Pyridinoline and deoxypyridinoline are pyridinium crosslinks which stabilize the collagen chains within the extracellular matrix. They are released during the breakdown of mature collagen and excreted in the urine, and therefore, are used as specific bone turnover markers [26]. However, the clinical role is still controversial, due to the high biological variability [27, 28].
Three studies comprising 120 patients, analyzed the relation between the BMD and pyridolines in patients undergoing IUD [22–24]. Correlation between concentration in urine of these markers and loss of BMD was found in two of them, suggesting that acidosis may be attributed to the decrease in bone mineral density [22, 24].
However, in the study by Giannini et al. these differences were found when these urine parameters were evaluated at different time after surgery [23]. Particularly, patients with a shorter time after surgery (< 36 months) presented higher urine hydroxyproline values, corresponding with the period in which a more severe metabolic acidosis.
DISCUSSION
Patients experiencing low impact fractures secondary to osteoporosis are at significantly higher risk for death than their counterparts, and this persists for several years post-fracture [9, 10]. Nonhip, nonvertebral fractures also were associated with premature mortality [10]. The main interest to analyze the fractures after urinary diversion, is that such a surgery is mostly performed in patients with BCa, an aged population who is already at higher risk of bone fractures due to aging and an add risk of pathological fractures in case of developing bone metastases [29].
In the current literature, just two studies, addressed this question using the risk of fractures in patients undergoing urinary diversion as the endpoint [14, 15]. Regarding the findings, the studies looking at the risk of fracture showed a clear association to the presence of IUD, whatever the indication (oncological or benign condition). The risk of fracture was around 21–48%higher in patients undergoing a IUD compared with their health counterparts.
However, most of the articles included in this review used the subrogated variable BMD as their primary outcome. Although the measurement of BMD is an indirect risk of fracture marker, the use of tools which can help to screen patients at risk of fracture is of utmost importance [30]. Therefore, the FRAX (Fracture Risk Assessment Tool) score, which includes BMD, estimates the individualized 10-year probability of hip and major osteoporotic fracture and can identify patients at high risk. Due to the large timeline of the included studies and the change in the techniques used to assess BMD, the results were heterogeneous and could not be compared. The results of current literature on the effect of IUD on BMD are insufficient to provide clear and robust conclusions.
Moreover, other bone resorption parameters as pyridiniums, serum parameters as well as histological bone architecture, have been studied with no strong repercussion in this field. The lack of robust literature using these variables and the contradictory results, complicates to make any statement related to them.
Regarding the kind of diversion, there are studies based on small simple size, which reported higher metabolic acidosis rates in patients with continent diversion compared to those with ileal conduit. It is postulated that patients are at especially high risk for metabolic acidosis when the intestinal segment is exposed to urine for long periods since longer contact with mucosa leads to more reabsorption [2]. However, in our systematic review, several studies found no difference between conduit, continent bladders and control groups related to BMD [19, 21]. Despite the expectations due to the lower reabsorption period and ratio, one study found significant differences in the ileal conduit group but not in the continent group compared to the control [25]. However, age difference is considered a potential explanation for these results.
Several studies assessed the difference between the intestinal segment used (colonic or ileal), founding no significant difference in metabolic acidosis and bone demineralization [21, 22]. On the contrary, Sevin et al. reported a decrease BMD in patients with ileal bladder and no changes in those with an Indiana pouch [12].
It is to note, that patients with renal insufficiency are less likely to eliminate de acid load and therefore this could be a confusion factor. Nevertheless, most of the studies excluded patients with renal insufficiency.
Unfortunately, the quality and heterogeneity of the reported data, as previously mentioned (number of patients, primary outcomes, diagnostic tests, measurements...) made not possible to meta-analyze the results.
LIMITATIONS
This systematic review presents relevant and important findings. Additionally, no language restriction was applied in order to prevent the language bias. However, the study also presents several limitations. First of all, inherent limitations associated to the population studies based on administrative databases (SEER Incidence database, Ontario Cancer Registry, Registered Person database, Canadian Institute for health Health Information, Discharge Abstract Database and Ontario Health Insurance Plan). Second, the use of a BMD as surrogate variable for bone fracture in most of the studies. Although BMD is an indirect risk of fracture marker, tools for predicting risk of fracture, like FRAX score (in which BMD is also included) are of utmost importance [30]. Third, most of the article included had a small sample size with retrospective design and limited follow-up duration. Fourth, in a high number of publications, female patients were not included because of the potential effect of menopause and hormonal changes on bone mineral metabolism. Likewise, some studies exclude patients with renal impairments due to the effect on osteoporosis introducing significant selection bias, whereas others include them.
To conclude, patients undergoing IUD seem to be at higher risk of bone fractures than their counterparts. Although data concerning BMD are not conclusive, the higher risk of fracture on these patients support the need to implement strategies for the prevention of osteoporosis in patients with IUD. Fracture risk algorithms which combine BMD and clinical risk factors such as FRAX score can be used to guide treatment decisions. However, a wise strategy to detect patients at high risk of bone fractures and who benefit the most from a prophylactic treatment still need to be explored.
ACKNOWLEDGMENTS
The authors have no acknowledgments.
FUNDING
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors
AUTHOR CONTRIBUTIONS
Carmen Pozo: conception, performance of work; interpretation or analysis of data; writing the article. Benjamin Pradere: performance of work; interpretation or analysis of data; writing the article. Katharina Rebhan: performance of work; interpretation or analysis of data; writing the article. Cao Chao: performance of work, writing the article. Lin Yang: performance of work, writing the article. Mohammad Abufaraj: interpretation or analysis of data; writing the article. Shahrokh F. Shariat: conception, interpretation or analysis of data, writing the article.
ETHICAL CONSIDERATIONS
This study, as a literature review, is exempt from any requirement for Institutional Review Board approval. No human or animal research was involved in the elaboration of this manuscript.
CONFLICT OF INTEREST
Shahrokh F. Shariat owns or co-owns the following patents: Methods to determine prognosis after therapy for prostate cancer, granted 2002-09-06; Methods to determine prognosis after therapy for bladder cancer, granted 2003-06-19; Prognostic methods for patients with prostatic disease, granted 2004-08-05; Soluble Fas: urinary marker for the detection of bladder transitional cell carcinoma, granted 2010-07-20. He is also an advisory board member of Astellas, Astra Zeneca, Bayer, BMS, Cepheid, Ferring, Ipsen, Janssen, Lilly, MSD, Olympus, Pfizer, Pierre Fabre, Richard Wolf, Roche, Sanochemia, Sanofi, Takeda and Urogen.
Carmen Pozo, Benjamin Pradere, Katharina Rebhan, Cao Chao, Lin Yang and Mohammad Abufaraj have no conflicts of interest to declare.
REFERENCES
[1] | Lee RK , Abol-Enein H , Artibani W , Bochner B , Dalbagni G , Daneshmand S , et al. Urinary diversion after radical cystectomy for bladder cancer: options, patient selection, and outcomes. BJU Int. (2014) ;113: (1):11–23. |
[2] | Van der Aa F , Joniau S , Van Den Branden M , Van Poppel H . Metabolic changes after urinary diversion. Adv Urol. (2011) ;2011: :764325. |
[3] | McDougal WS . Metabolic complications of urinary intestinal diversion. J Urol. (1992) ;147: (5):1199–208. |
[4] | Mills RD , Studer UE . Metabolic consequences of continent urinary diversion. J Urol. (1999) ;161: (4):1057–66. |
[5] | Boyd JD . Chronic Acidosis Secondary To Ureteral Transplantation. American Journal of Diseases of Children. (1931) ;42: (2):366–71. |
[6] | Alexander RT , Cordat E , Chambrey R , Dimke H , Eladari D . Acidosis and Urinary Calcium Excretion: Insights from Genetic Disorders. Journal of the American Society of Nephrology: JASN. (2016) ;27: (12):3511–20. |
[7] | Krieger NS , Frick KK , Bushinsky DA . Mechanism of acid-induced bone resorption. Curr Opin Nephrol Hypertens. (2004) ;13: (4):423–36. |
[8] | Pattanaungkul S , Riggs BL , Yergey AL , Vieira NE , O’Fallon WM , Khosla S . Relationship of intestinal calcium absorption to 1,25-dihydroxyvitaminD[1,25(OH)2D] levels in young versus elderly women: evidence for age-related intestinal resistance to 1,25(OH)2D action. J Clin Endocrinol Metab. (2000) ;85: (11):4023–7. |
[9] | Abrahamsen B , van Staa T , Ariely R , Olson M , Cooper C . Excess mortality following hip fracture: a systematic epidemiological review. Osteoporos Int. (2009) ;20: (10):1633–50. |
[10] | Bliuc D , Nguyen ND , Milch VE , Nguyen TV , Eisman JA , Center JR Mortality Risk Associated With Low-Trauma Osteoporotic Fracture and Subsequent Fracture in Men and Women. JAMA. (2009) ;301: (5):513–21. |
[11] | Campanello M , Herlitz H , Lindstedt G , Mellstrom D , Wilske J , Akerlund S , et al. Determinants of bone loss in patients with Kock ileal urinary reservoir. Scand J Urol Nephrol. (1999) ;33: (5):312–6. |
[12] | Sevin G , Kosar A , Perk H , Serel TA , Gurbuz G . Bone mineral content and related biochemical variables in patients with ileal bladder substitution and colonic Indiana pouch. Eur Urol. (2002) ;41: (6):655–9. |
[13] | Poulsen AL , Overgaard K , Steven K . Bone metabolism following bladder substitution with the ileal urethral Kock reservoir. Br J Urol. (1997) ;79: (3):339–47. |
[14] | Gupta A , Atoria CL , Ehdaie B , Shariat SF , Rabbani F , Herr HW , et al. Risk of fracture after radical cystectomy and urinary diversion for bladder cancer. J Clin Oncol. (2014) ;32: (29):3291–8. |
[15] | Richard PO , Bashir S , Riverin BD , Alibhai SMH , Kim SJ , Ahmad A , et al. Risk of Bone Fractures Following Urinary Intestinal Diversion: A Population Based Study. J Urol. (2019) ;202: (2):319–25. |
[16] | Liberati A , Altman DG , Tetzlaff J , Mulrow C , Gotzsche PC , Ioannidis JP , et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. (2009) ;6: (7):e1000100. |
[17] | Stang A . Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. (2010) ;25: (9):603–5. |
[18] | Deeks JJ , Dinnes J , D’Amico R , Sowden AJ , Sakarovitch C , Song F , et al. Evaluating non-randomised intervention studies. Health Technol Assess. (2003) ;7: (27):iii-x, 1–173. |
[19] | Campanello A , Herlitz H , Lindstedt G , Mellstrom D , Wilske J , Akerland S , et al. Bone mineral and related biochemical variables in patients with Kock ileal reservoir or Bricker conduit for urinary diversion. J Urol. (1996) ;155: (4):1209–13. |
[20] | Tschopp AB , Lippuner K , Jaeger P , Merz VW , Danuser H , Studer UE . No evidence of osteopenia 5 to 8 years after ileal orthotopic bladder substitution. J Urol.. (1996) ;155: (1):71–5. |
[21] | Davidsson T , Lindergard B , Obrant K , Mansson W . Long-term metabolic effects of urinary diversion on skeletal bone: histomorphometric and mineralogic analysis. Urology. (1995) ;46: (3):328–33. |
[22] | Kawakita M , Arai Y , Shigeno C , Terai A , Okada Y , Takeuchi H , et al. Bone demineralization following urinary intestinal diversion assessed by urinary pyridinium cross-links and dual energy x-ray absorptiometry. J Urol.. (1996) ;156: (2 Pt 1):355–9. |
[23] | Giannini S , Nobile M , Sartori L , Aragona F , Ruffato A , Dalle Carbonare L , et al. Bone density and skeletal metabolism in patients with orthotopic ileal neobladder. J Am Soc Nephrol. (1997) ;8: (10):1553–9. |
[24] | Fujisawa M , Nakamura I , Yamanaka N , Gotoh A , Hara I , Okada H , et al. Changes in calcium metabolism and bone demineralization after orthotopic intestinal neobladder creation. J Urol. (2000) ;163: (4):1108–11;quiz 295. |
[25] | Incel N , Incel NA , Uygur MC , Tan O , Erol D . Effect of stanford pouch and ileal conduit urinary diversions on bone mineral density and metabolism. Int Urol Nephrol. (2006) ;38: (3-4):447–51. |
[26] | Uebelhart D , Gineyts E , Chapuy M-C , Delmas PD . Urinary excretion of pyridinium crosslinks: a new marker of bone resorption in metabolic bone disease. Bone and Mineral. (1990) ;8: (1):87–96. |
[27] | Colwell A , Russell RGG , Eastell R . Factors affecting the assay of urinary 3–hydroxy pyridiniurn crosslinks of collagen as markers of bone resorption. European Journal of Clinical Investigation. (1993) ;23: (6):341–9. |
[28] | Blumsohn A , Eastell R . The performance and utility of biochemical markers of bone turnover: do we know enough to use them in clinical practice? Ann Clin Biochem. (1997) ;34: (Pt 5):449–59. |
[29] | Tsuda Y , Nakagawa T , Shinoda Y , Kanatani A , Kawai T , Taguchi S , et al. Skeletal-related events and prognosis in urothelial cancer patients with bone metastasis. Int J Clin Oncol. (2017) ;22: (3):548–53. |
[30] | Marshall D , Johnell O , Wedel H . Meta-analysis of how well measures of bone mineral density predict occurrence of osteoporotic fractures. Bmj. (1996) ;312: (7041):1254–9. |
Appendices
Appendix 1
NOS Score of 11 studies included
| Title | Selection | Comparability | Outcome/Exposure | |
| Campanello M., 1996 | *** | ** | ** | 7 |
| Davidsson T., 1995 | *** | * | *** | 7 |
| Fujisawa M., 2000 | ** | * | * | 4 |
| Giannini S., 1996 | *** | * | *** | 7 |
| Gupta A., 2014 | *** | * | ** | 6 |
| Incel N., 2006 | **** | * | ** | 7 |
| Kawakita M., 1996 | ** | * | *** | 6 |
| Poulsen A., 1997 | *** | * | ** | 6 |
| Richard O., 2019 | **** | * | ** | 7 |
| Sevin G., 2002 | *** | * | * | 5 |
| Tschopp A., 1995 | *** | * | *** | 7 |




