Implementation of a Reduced Opioid Utilization Protocol for Radical Cystectomy
Abstract
BACKGROUND:
Radical cystectomy (RC) often requires a prolonged course of opioid medications for postoperative pain management. We implemented a Reduced Opioid Utilization (ROU) protocol to decrease exposure to opioid medications.
OBJECTIVE:
To determine the impact of the ROU protocol on opioid exposure, pain control, inpatient recovery, and complication rates among patients who underwent RC.
METHODS:
The ROU protocol includes standardized recovery pathways, a multimodal opioid-sparing pain regimen, and improved patient and provider education regarding non-opioid medications. Opioid exposure was calculated as morphine equivalent dose (MED), and was compared between RC patients following the ROU protocol and patients who previously followed our traditional pathway. Opioid-related adverse drug events (ORADEs), pain scores, length of stay, and 90-day complications, readmission, and mortality were also compared between cohorts.
RESULTS:
104 patients underwent RC, 54 (52%) of whom followed the ROU protocol. ROU patients experienced a statistically significant decrease in opioid exposure in the post-anesthesia care unit (p = 0.003) and during their postoperative recovery (85.7±21.0 MED vs 352.6±34.4 MED, p < 0.001). The ROU protocol was associated with a statistically significant decrease in ORADEs after surgery. There was no significant difference in average pain scores, length of stay, readmissions, or 90-day complication or mortality rates.
CONCLUSIONS:
The ROU protocol decreased opioid use by 77% without compromising pain control or increasing the rate of complications. This study demonstrates the efficacy of non-opioid medications in controlling postoperative pain, and highlights the role providers can play to decrease patient exposure to opioids after RC surgery.
INTRODUCTION
Bladder cancer is the 6th most common cancer in the United States, with over 80,000 new cases diagnosed each year [1]. Radical cystectomy (RC) with urinary diversion is the gold standard treatment for non-metastatic, muscle-invasive bladder cancer, and is indicated in high-risk non-muscle invasive bladder cancer refractory to intravesical therapy [2]. This surgery involves the removal of the bladder, prostate, and seminal vesicles in men, and bladder, uterus, fallopian tubes, ovaries, and anterior vaginal wall in women. Despite a shift toward minimally-invasive approaches, RC still carries significant morbidity and mortality [3]. Patients require 5–10 hospital days to recover, and experience significant postoperative pain and functional impairment [4–8].
Patients are routinely exposed to opioids during their postoperative in-patient recovery for acute pain management. For many, this is their first encounter with these medications, which carry a high propensity for dependence and abuse. Previous reports have shown that between 3–6% of surgical patients become newly addicted to opioid medications after surgery [9]. Multimodal care pathways, including enhanced recovery after surgery (ERAS) protocols, have shown to improve postoperative care by reducing length of stay (LOS), time to return of bowel function, and decreasing readmission in several surgical specialties [10]. However, postoperative opioid use has proven more difficult to control [11].
To address this issue, we implemented a quality improvement initiative, the Reduced Opioid Utilization (ROU) protocol, specifically aimed to reduce patients’ opioid exposure. In this prospective observational study, we tracked opioid exposure, clinical milestones of recovery, pain scores, readmission rates, and 90-day complications between RC patients following the ROU protocol and patients treated with traditional care to assess the efficacy of this intervention.We hypothesize that RC patients following the ROU protocol will have similar post-operative pain scores and clinical outcomes compared to RC patients following our traditional care pathway, highlighting the feasibility of reducing opioid exposure in this population.
METHODS
Reduced opioid utilization (ROU) protocol
The ROU protocol was established to reduce opioid exposure, to improve preoperative patient education, and to set expectations regarding pain control after RC surgery. Physicians and APPs were encouraged to discuss the dangers of opioid use and the value of non-narcotic medications with patients during the preoperative visit. Providers were also educated on the use of opioid alternatives. This process included several in-service training sessions for PACU nursing staff and the inpatient urology floor team, posting of information about the ROU protocol on staff forums, and meetings with attending physicians in anesthesiology to encourage interdepartmental support. This study was approved by the Stanford University Institutional Review Board (IRB-48180).
A multi-modal pain regimen was established that utilized both pharmacologic and non-pharmacologic treatments. Patients presenting for RC were given a single dose of acetaminophen 1000 mg, gabapentin 300 mg, celecoxib 200 mg, and tramadol 100 mg by mouth in the preoperative holding area. Although tramadol is known opioid agonist, it was included in our protocol because it is far less potent than other commonly used analgesics [12]. Intraoperatively, surgeons were encouraged to utilize local anesthetic agents including liposomal bupivacaine administered in multiple layers of the lower midline incision. Postoperative analgesia included scheduled acetaminophen 1000 mg PO Q6H, ketorolac 15 mg/ml Q6H, gabapentin 100 mg PO TID, and Tramadol 50 mg PO PRN for breakthrough pain. Non-pharmacologic strategies such as ice packs and abdominal binders were used to supplement pain control, and patient-controlled analgesia (PCA) was avoided. Dosing of any narcotics was managed by trained pharmacists and was determined using patients’ renal and hepatic function, weight, and use of opioids prior to surgery. Individualized narcotic pain regimens were ordered as needed for patients who did not adequately respond to non-opioid regimens, and for patients with significant opioid exposure prior to admission. Alvimopan, a peripherally-acting μ-opioid receptor antagonist, was prescribed for all patients postoperatively receiving opioids until return of bowel function. Default order sets were changed in the electronic medical record (Epic, Wisconsin, USA) to encourage adherence and sustainabilityof the ROU protocol.
Study design
Patients who underwent RC between February 2017 and July 2018 were included in this study.There was no exclusion of patients on the basis of cancer stage or histology. Three fellowship-trained urologic oncology surgeons performed all RC surgeries. Patients receiving RC surgery underwent either open or robotic surgery, depending on surgeon preference and clinical indications, and the type of UD was determined prior to surgery through discussion and consultation with the patient. Patients following the ROU protocol (October 2017 –July 2018) were then retrospectively compared to patients receiving traditional care prior to the implementation of the intervention (February 2017 –September 2017). Demographic information and perioperative data were also collected and compared between cohorts.
Outcome measures
The primary outcome was patient opioid exposure, measured as oral morphine equivalent dose (MED). All opioid medications and doses were converted to morphine per os dose according to Stanford University Lexicomp. Medications not included in Stanford University Lexicomp were converted to morphine per os dose according to previously published equianalgesic dosing [13, 14]. Opioid exposure was calculated for the intraoperative procedure, recovery in the PACU, and during their postoperative in-patient admission. Secondary outcomes included ORADEs, postoperative pain scores, complications, in-patient recovery milestones, and all-cause mortality. ORADEs included nausea, emesis, constipation, sedation (± naloxone administration), pruritis, diaphoresis, and confusion, delirium, or agitation. Complications were captured from the date of RC up to and including 90-days after surgery, and were graded using Clavien-Dindo Classification [15, 16]. In-patient recovery milestones included days to first flatus, first bowel movement, first ambulation around the in-patient unit, and days to first tolerance of liquid diet and regular diet. Average pain at 24-, 48-, and 72-hours after surgery was measured using Visual Analogue Scale (VAS) scores collected by nursing inquiry.
Statistical analysis
Data were analyzed using Stata v15.1 (Stata Corp. 2013. College Station, TX) statistical software. Graphical analyses were performed using GraphPad Prism (Version 6.07. La Jolla, CA). Continuous variables were analyzed using Mann-Whitney U test when non-parametric or Student’s t test when normally distributed. Categorical variables were summarized in frequency tables with counts and percentages, and were analyzed using Pearson’s χ2 and Fisher’s exact test where appropriate. All tests were two-sided, and a p-value of <0.05 was deemed statistically significant. Measures of central tendency were reported as mean±standard error.
RESULTS
A total of 104 patients underwent RC during the study period and were included in the analyses. Fifty-four (52%) patients followed the ROU protocol, and 50 (48%) patients followed the traditional care pathway. Demographic information and patient comorbidities are shown in Tables 1 and 2. The mean age of all patients at time of RC was 70.2±0.9 years of age, and was similar between ROU and non-ROU patients (70.9±1.2 vs 69.4±1.4, p = 0.409). There was also no significant difference in sex, race, BMI, smoking status, or preoperative lab values between cohorts. Coincident with a surgeon trained in robotic cystectomy and enhanced recovery after surgery (ERAS) protocols joining the faculty shortly before implementation of the protocol, more patients on the ROU protocol had robotic surgery and fewer had epidural placement. ROU patients had significantly longer operative time compared to non-ROU patients (371.4±12.8 minutes vs 305.2±6.1 minutes, p < 0.001) (Table 3).
Table 1
Demographic information of included patients
| Preoperative characteristics | ROU pathway | Non-ROU pathway | p-value |
| n = 54 (%) | n = 50 (%) | (p < 0.05) | |
| Sex | 0.24 | ||
| Male | 46 (85.2) | 38 (72.0) | |
| Female | 8 (14.8) | 12 (24.0) | |
| Race/Ethnicity | 0.30 | ||
| Asian | 5 (9.3) | 4 (8.0) | |
| Black or African American | 2 (3.7) | 1 (2.0) | |
| Hispanic or Latino | 3 (5.6) | 10 (20.0) | |
| Native Hawaiian or Pacific Islander | 1 (1.9) | 0 (0.0) | |
| Other or Unknown | 2 (3.7) | 1 (2.0) | |
| White | 41 (75.9) | 34 (68.0) | |
| Age (years) | 70.9±1.2 | 69.4±1.4 | 0.41 |
| BMI | 27.4±0.6 | 29.1±0.7 | 0.07 |
Abbreviations: BMI = body mass index, ROU = reduced opioid utilization. Measures of central tendency are reported as mean±SEM. Continuous variables were analyzed using Student’s t test, categorical variables were analyzed using Chi Square test.
Table 2
Past medical history and comorbidities of included patients
| Preoperative characteristics | ROU pathway | Non-ROU pathway | p-value |
| n = 54 (%) | n = 50 (%) | (p < 0.05) | |
| Smoking Status | 0.28 | ||
| Never smoker | 22 (40.7) | 21 (42.0) | |
| Current smoker | 5 (9.3) | 1 (2.0) | |
| Former smoker | 27 (50.0) | 28 (56.0) | |
| Comorbidities | 0.55 | ||
| Arrhythmia | 8 (14.8) | 10 (20.0) | |
| Chronic pain syndrome | 4 (7.4) | 5 (10.0) | |
| Chronic obstructive pulmonary disease | 5 (9.3) | 2 (4.0) | |
| Coronary artery disease | 7 (13.0) | 6 (12.0) | |
| Diabetes mellitus | 11 (20.4) | 15 (30.0) | |
| Myocardial infarction | 1 (1.9) | 2 (4.0) | |
| Renal disease | 8 (14.8) | 9 (18.0) | |
| Stroke/CVA | 0 (0.0) | 4 (8.0) | |
| Preoperative Lab Values | |||
| WBC (K/μl) | 7.5±0.4 | 7.6±0.4 | 0.81 |
| Hgb (g/dl) | 12.7±0.2 | 12.2±0.3 | 0.17 |
| Platelets (K/μl) | 218.3±10.2 | 227.4±11.8 | 0.56 |
| Serum creatinine (mg/dl) | 1.05±0.05 | 1.15±0.08 | 0.26 |
| Serum albumin (g/dl) | 3.8±0.05 | 3.6±0.08 | 0.08 |
| ASA Classification | 0.44 | ||
| II | 19 (35.2) | 22 (44.0) | |
| III | 34 (63.0) | 28 (56.0) | |
| IV | 1 (1.9) | 0 (0.0) |
Abbreviations: ASA = American Society of Anesthesiologists, CVA = cerebrovascular accident, Hgb = hemoglobin, ROU = reduced opioid utilization, WBC = white blood cells. Measures of central tendency are reported as mean±SEM. Continuous variables were analyzed using Student’s t test, categorical variables were analyzed using Chi Square test.
Table 3
Surgical approach, urinary diversion, and operative characteristics
| ROU pathway | Non-ROU pathway | p-value | |
| n = 54 (%) | n = 50 (%) | (p < 0.05) | |
| Primary Surgery of Interest | 0.12 | ||
| Radical cystectomy | 14 (25.9) | 21 (42.0) | |
| Radical cystoprostatectomy | 39 (72.2) | 26 (52.0) | |
| Radical cystectomy and TAH (–BSO) | 1 (1.9) | 1 (2.0) | |
| Radical cystectomy and TAH (+BSO) | 0 (0.0) | 2 (4.0) | |
| Surgical Approach | <0.001 | ||
| Open | 34 (63.0) | 50 (100.0) | |
| Robotic (Robot-assisted) | 20 (37.0) | 0 (0.0) | |
| Urinary Diversion | 0.61 | ||
| Ileal conduit | 38 (70.4) | 37 (74.0) | |
| Ileal neobladder (Studer pouch) | 15 (27.8) | 13 (26.0) | |
| Ureterostomy | 1 (1.9) | 0 (0.0) | |
| Epidural | 1 (1.9) | 34 (68.0) | <0.001 |
| Operative Time (minutes) | 371.4±12.8 | 305.2±6.1 | <0.001 |
| Estimated Blood Loss (ml) | 766.7±87.4 | 888.6±81.3 | 0.31 |
Abbreviations: BSO = bilateral salpingo-oopherectomy, ROU = reduced opioid utilization, TAH = total abdominal hysterectomy. Measures of central tendency are reported as mean±SEM. Continuous variables were analyzed using Student’s t test, categorical variables were analyzed using Chi Square test.
We observed a significant reduction in opioid exposure in both the PACU and during patients’ in-patient admission for patients following the ROU protocol (Fig. 1). 48.1% (26/54) of ROU patients received no opioids in the PACU, compared to only 24% (12/50) of non-ROU patients (p = 0.011). While all non-ROU patients received opioids during their postoperative admission, 20.4% (11/54) of ROU patients received no opioid medications at all after their surgery. VAS pain scores were similar between both cohorts at 24-, 48-, and 72-hours after surgery (Fig. 2). No significant difference was found in opioid exposure between cohorts during their intraoperative procedure, or between opioid prescriptions at discharge (Table 4).
Fig. 1
Comparison of opioid exposure measured as morphine equivalent dose (MED) in both the (A) post-anesthesia care unit (PACU) and (8) during patients’ postoperative admission between ROU and Non-ROU patients. Graphical analysis using Tukey plot. Analysis was performed using unpaired student’s t test. p < 0.05 was deemed statistically significant.
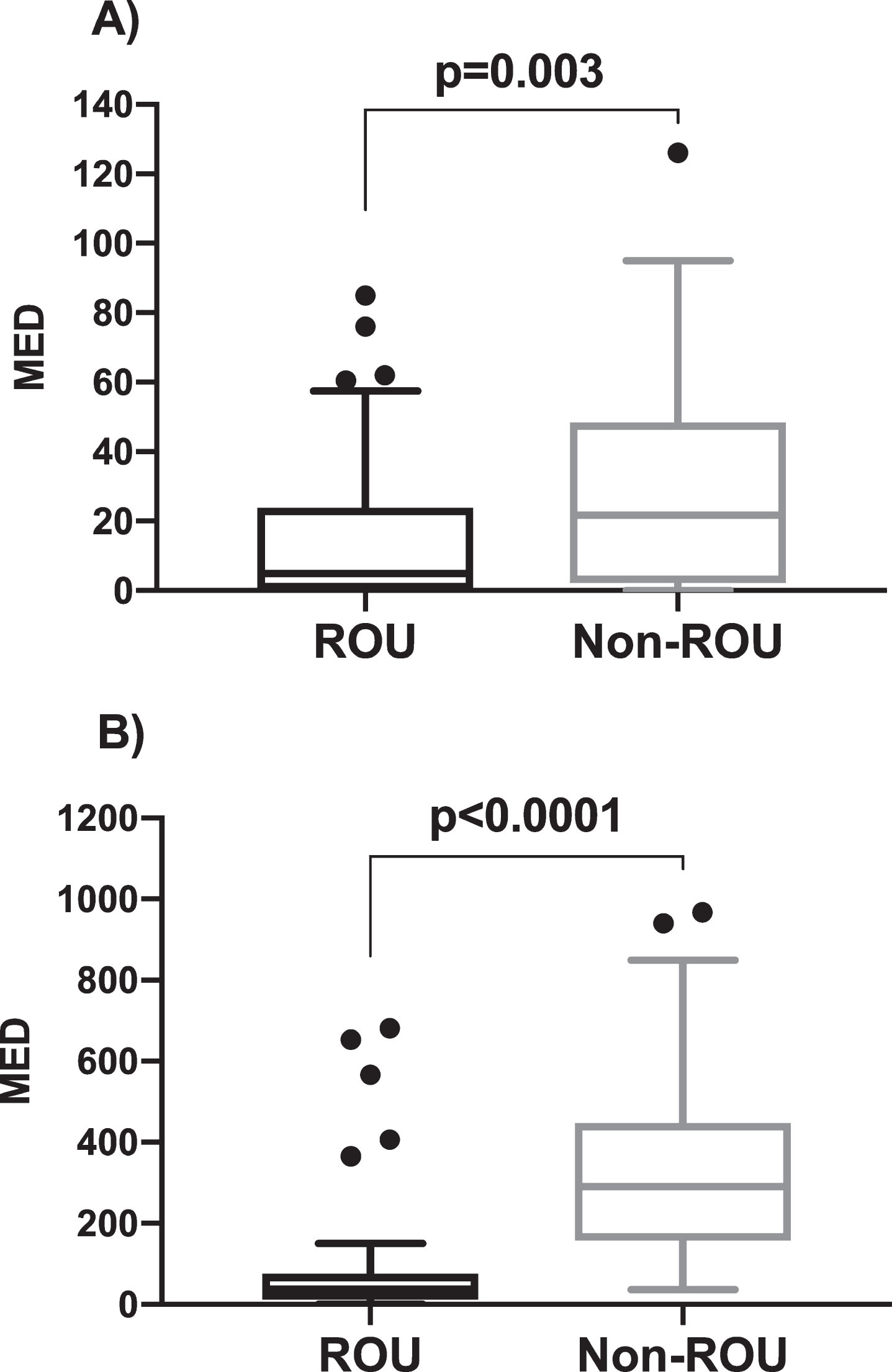
Fig. 2
Comparison of average pain scores measured using the visual analogue scale. Measure of central tendency are reported as mean ± SD. Analysis was performed using student’s t test. P < 0.05 was deemed statistically significant.
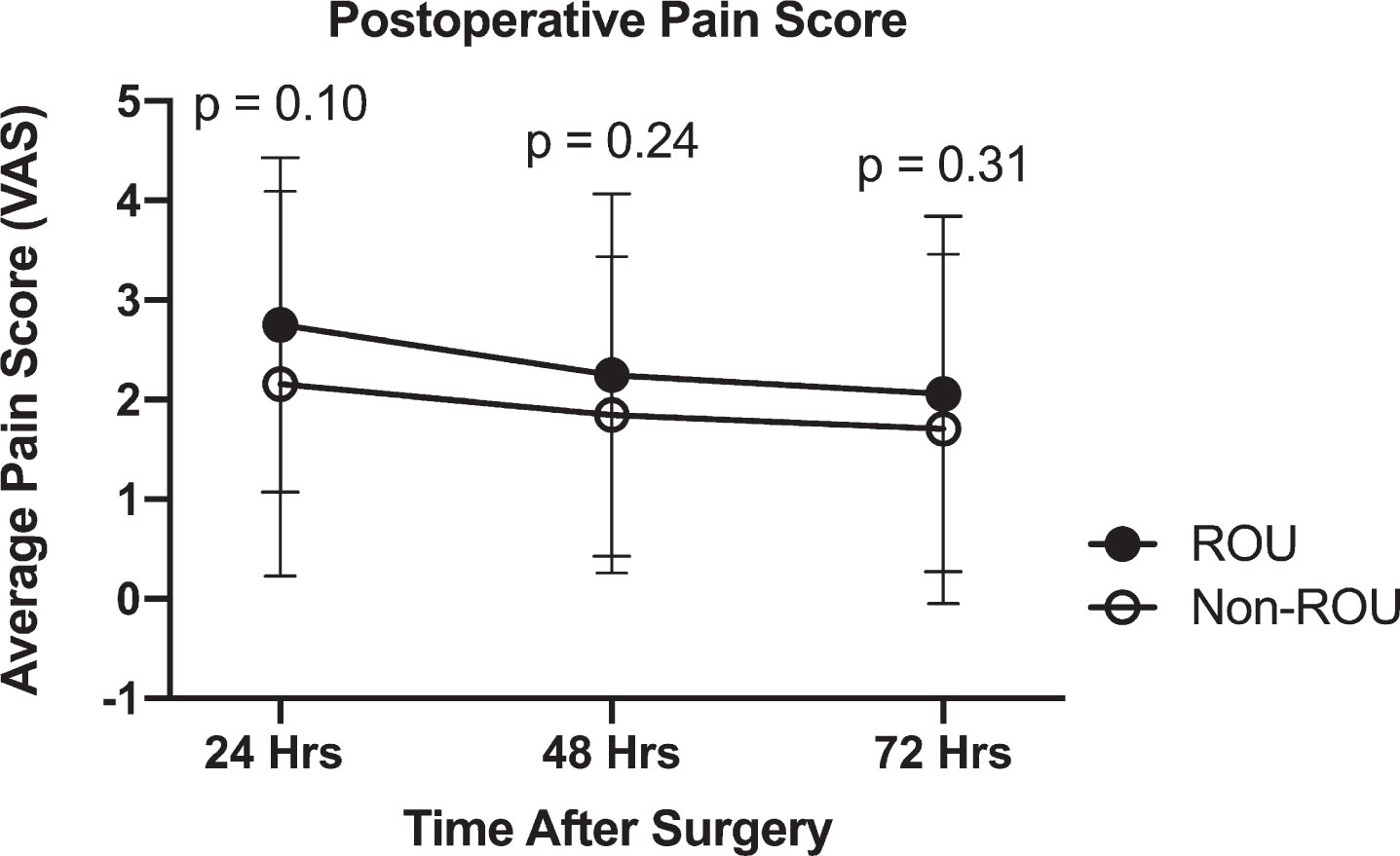
Table 4
Opioid use and opioid related adverse events during surgery, PACU, postoperative admission, and home recovery
| ROU pathway | Non-ROU pathway | p-value | |
| n = 54 (%) | n = 50 (%) | (p < 0.05) | |
| Intra-Op (MED) | 148.2±13.7 | 133.3±11.3 | 0.41 |
| PACU (MED) | 14.8±2.9 | 30.5±4.4 | <0.01 |
| No. of patients with ORADE (POD#0) | 4 (7.4) | 7 (14.0) | 0.28 |
| Nausea | 2 (3.7) | 3 (6.0) | |
| Vomiting | 3 (5.6) | 2 (4.0) | |
| Sedation (± naloxone) | 0 (0.0) | 2 (4.0) | |
| No. of patients without opioid use | 26 (48.1) | 12 (24.0) | 0.01 |
| Post-Op (MED) | 85.7±21.0 | 398.1±62.2 | <0.001 |
| MEDD during postoperative admission | 17.2±2.8 | 89.3±9.9 | <0.001 |
| No. of patients with ORADE | 7 (13.0) | 27 (54.0) | <0.001 |
| Nausea | 6 (11.1) | 13 (26.0) | |
| Vomiting | 3 (5.6) | 22 (44.0) | |
| Confusion/delirium/agitation | 0 (0.0) | 5 (10.0) | |
| Constipation | 2 (3.7) | 8 (16.0) | |
| Diaphoresis | 1 (1.9) | 0 (0.0) | |
| Pruritis | 0 (0.0) | 1 (2.0) | |
| No. of patients without opioid use | 11 (20.4) | 0 (0.0) | <0.001 |
| Home/Discharge (MED) | 282.1±23.1 | 279.1±37.0 | 0.94 |
| No. of patients with ORADE (reported) | 12 (22.2) | 14 (28.0) | 0.50 |
| Nausea | 2 (3.7) | 3 (6.0) | |
| Vomiting | 1 (1.9) | 2 (4.0) | |
| Confusion/delirium/agitation | 0 (0.0) | 2 (4.0) | |
| Constipation | 12 (22.2) | 7 (14.0) | |
| Pruritis | 0 (0.0) | 1 (2.0) | |
| No. of patients without opioid Rx | 9 (16.7) | 7 (14.0) | 0.71 |
Abbreviations: MED = morphine equivalent dose, MEDD = morphine equivalent daily dose, ORADE = opioid related adverse event, PACU = post anesthesia care unit, ROU = reduced opioid utilization. Measures of central tendency are reported as mean±SEM. Continuous variables were analyzed using Student’s t test, categorical variables were analyzed using Chi Square test.
Postoperative complications and ORADEs were compared during patients’ stay in the PACU, during in-hospital recovery, and up to 90-days following surgery. In all three areas, the most commonly reported ORADE was nausea. However, patients in the non-ROU group were more than twice as likely to report nausea during the immediate postoperative period (Table 4). The ROU Protocol was associated with a significantreduction in the number of patients experiencing ORADEs during in-patient recovery (13.0% vs 54.0%, p < 0.001), and did not increase the rate of complications overall. 33.7% of patients were readmitted within 90-days after RC, and all-cause mortality in this time period was 2.9%. There was no difference in readmission or all-cause mortality between ROU and non-ROU patients (Table 5).
Table 5
Complications, morbidity, and mortality up to 90-days following surgery
| ROU pathway | Non-ROU pathway | p-value | |
| n = 54 (%) | n = 50 (%) | (p < 0.05) | |
| Postoperative Intensive Care | |||
| No. of patients admitted to ICU | 6 (11.1) | 2 (4.0) | 0.17 |
| Avg. ICU admission (days) | 3.0±1.2 | 1.5±0.5 | 0.60 |
| No. of patients with NGT placed | 5 (9.3) | 8 (16.0) | 0.30 |
| No. of patients requiring reoperation | 4 (7.4) | 0 (0.0) | 0.12 |
| Postoperative Admission | |||
| No. of patients with a complication | 24 (44.4) | 29 (58.0) | 0.17 |
| Grade <3 | 27 | 38 | |
| Grade ≥3 | 8 | 2 | |
| Readmission (± ED Visit) | |||
| Avg. time of admission (days) | 3.1±0.5 | 4.1±0.8 | 0.32 |
| No. of patients with a complication | 18 (33.3) | 17 (34.0) | 0.94 |
| Grade <3 | 12 | 16 | |
| Grade ≥3 | 9 | 8 | |
| Outpatient Visit | |||
| No. of patients with a complication | 16 (29.6) | 18 (36.0) | 0.49 |
| Grade <3 | 17 | 18 | |
| Grade ≥3 | 0 | 1 | |
| All-Cause Mortality within 90 days | 1 (1.9) | 2 (4.0) | 0.51 |
Abbreviations: ED = emergency department, ICU = intensive care unit, NGT = nasogastric tube, ROU = reduced opioid utilization. Measures of central tendency are reported as mean±SEM. Continuous variables were analyzed using Student’s t test or Mann-Whitney U test, categorical variables were analyzed using Chi Square test or Fisher’s Exact test.
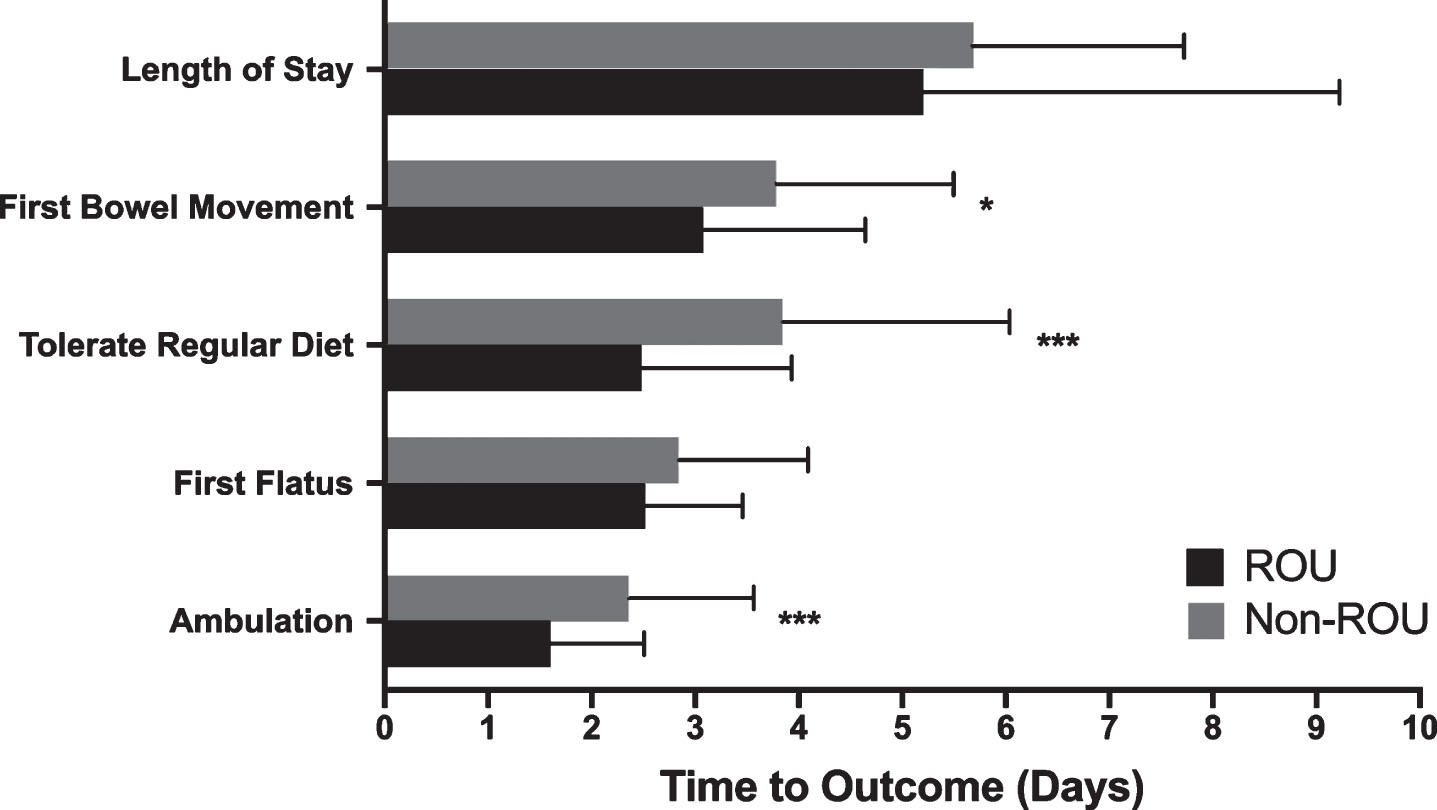
The ROU protocol was associated with a significant improvement in time to first ambulation around the patient unit (1.6±0.9 days vs 2.4±1.2 days, p < 0.001). They also experienced shorter time to return of bowel function (3.1±1.6 days vs 3.8±1.7 days, p = 0.027) and shorter time to tolerance of a regular diet after surgery (2.5±1.5 days vs 3.8±2.2 days, p < 0.001) (Fig. 3). The average LOS following RC was 5.4±0.3 days, with no significant difference observed between ROU and non-ROU patients. Although average days to first flatus was shorter in the ROU RC patients, this result was not statistically significant.
Fig. 3
Comparison of average time to reach inpatient clinical milestone of recovery between ROU and non-ROU patients. Measures of central tendency are reported as mean ± SD. Analysis was performed using unpaired student’s t test. *p < 0.05 was deemed statistically significant. *p < 0.05, **p < 0.01, ***p < 0.001.
Sub-group analysis of RC patients only undergoing open surgery showed that opioid exposure was still significantly reduced for patients following the ROU protocol in both the PACU (16.0±4.0 MED vs 30.5±4.4 MED, p = 0.02) and during postoperative admission (96.2±27.1 MED vs 345.6±34.4, p < 0.001). However, upon stratified analysis, operative time and time to regular diet were similar between cohorts. Lastly, the proportion of patients requiring reoperation was significantly higher in the ROU population when comparing only patients who underwent open RC (Table 6).
Table 6
Sub-Group analysis of open radical cystectomy patients
| ROU pathway | Non-ROU pathway | p-value | |
| n = 34 (%) | n = 50 (%) | (p < 0.05) | |
| Sex | 0.30 | ||
| Male | 29 (85.3) | 38 (72.0) | |
| Female | 5 (14.7) | 12 (24.0) | |
| Age | 71.9±1.4 | 69.4±1.4 | 0.23 |
| BMI | 27.7±0.8 | 29.1±0.7 | 0.20 |
| Comorbidities | 0.90 | ||
| Arrhythmia | 4 (11.8) | 10 (20.0) | |
| Chronic pain syndrome | 3 (8.8) | 5 (10.0) | |
| Chronic obstructive pulmonary disease | 1 (2.9) | 2 (4.0) | |
| Coronary artery disease | 3 (8.8) | 6 (12.0) | |
| Diabetes mellitus | 6 (17.6) | 15 (30.0) | |
| Myocardial infarction | 0 (0.0) | 2 (4.0) | |
| Renal disease | 4 (11.8) | 9 (18.0) | |
| Stroke/CVA | 0 (0.0) | 4 (8.0) | |
| Operative Time (min.) | 325.1±10.4 | 305.2±6.1 | 0.08 |
| Estimated Blood Loss (ml) | 902.1±87.3 | 888.6±81.3 | 0.91 |
| Opioid Exposure (MED) | |||
| Intra-op | 162.7±19.6 | 133.3±11.3 | 0.17 |
| PACU | 16.0±4.0 | 30.5±4.4 | 0.02 |
| Post-Op | 96.2±27.1 | 398.1±62.2 | <0.001 |
| Home/Discharge | 303.2±29.0 | 279.1±37.0 | 0.64 |
| Clinical Outcomes of Recovery (days) | |||
| Ambulation around inpatient unit | 1.45±0.12 | 2.36±0.17 | <0.001 |
| First flatus | 2.67±0.15 | 2.84±0.18 | 0.50 |
| First bowel movement | 3.12±0.17 | 3.78±0.24 | 0.04 |
| Toleration of regular diet | 3.06±0.26 | 3.84±0.31 | 0.07 |
| Length of stay | 5.56±0.58 | 5.68±0.29 | 0.84 |
| Complications (no. of patients) | |||
| Admitted to ICU post-op | 4 (11.8) | 2 (4.0) | 0,17 |
| Required NGT placement | 3 (8.8) | 8 (16.0) | 0.34 |
| Required reoperation | 3 (8.8) | 0 (0.0) | 0.03 |
| Postoperative admission | 15 (44.1) | 29 (58.0) | 0.21 |
| Readmission (± ED Visit) | 7 (20.6) | 17 (34.0) | 0.18 |
| Outpatient visit | 6 (17.6) | 18 (36.0) | 0.07 |
| All-Cause Mortality within 90 days | 1 (2.9) | 2 (4.0) | 0.80 |
Abbreviations: ASA = American Society of Anesthesiologists, CVA = cerebrovascular accident, Hgb = hemoglobin, ROU = reduced opioid utilization, WBC = white blood cells. Measures of central tendency are reported as mean±SEM. Continuous variables were analyzed using Student’s t test, categorical variables were analyzed using Chi Square test.
DISCUSSION
Chronic opioid use in the United States has become a national health concern, incurring significant costs and financial burden to the healthcare system while contributing to an increasing rate of fatal overdoses [17]. Similarly, opioid prescribing patterns by physicians have increased during this same period [17]. Surgeons and perioperative care teams face the difficult task of treating acute pain without contributing to patients’ opioid exposure, which has been shown to lead to chronic use in 5% of urologic surgery patients [18]. The rate of overdose, and opioid-related deaths has led to the declaration of a “national public health emergency” by the President of the United States in October of 2017 [19]. To address this national epidemic, public health advocates have sought to limit opioid prescriptions and promote access to the life-saving opioid antagonist, naloxone [20]. Healthcare providers therefore indirectly act as a gateway for potential narcotic abuse, highlighting the need for standardized recovery pathways that limit opioid exposure to improve patient safety.
The purpose of the ROU protocol was to reduce patient exposure to opioid analgesia following RC surgery. We were able to track patient opioid use throughout all phases of care, from preoperative exposure to discharge prescription patterns. We were also able to collect patient recovery data during the postoperative period, and identify complications following surgery up to 90 days. This pre-post study of our intervention, compared to traditional care pathways, demonstrates that improved patient and provider education, standardized recovery pathways, and increased use of non-opioid multimodal pain regimens can reduce opioid exposure without increasing pain. Furthermore, the ROU protocol significantly improved time to regular diet, time to ambulation, return of bowel function, and decreased the rate of ORADEs during postoperative recovery. However, there was a higher rate of re-intervention in the ROU cohort. While this difference was not statistically significant, the etiology of this remains unclear.
Previous standardized surgical recovery pathways, including ERAS, have sought to optimize and improve the quality of postoperative patient care. Originally designed for use in colorectal surgery, ERAS has been applied to urologic oncology surgery. However, adoption has been slow and implementation of the protocol has varied widely between institutions and surgical teams [21]. ERAS promotes preoperative Alvimopan and postoperative multimodal analgesia to minimize opioid use and reduce postoperative ileus. However, there is no standardized regimen that has been universally accepted. The primary aim of ERAS is not directly focused on reducing opioid use, but rather to decrease gastrointestinal complications, shorten LOS, and reduce readmissions [22, 23]. A study by Xu et al. compared outcomes of 205 RC patients, 124 of whom followed the ERAS protocol. ERAS patients used significantly less opioids per day (4.9 mg morphine equivalents vs 20.67 mg morphine equivalents, p < 0.001), but they reported greater pain during their postoperative recovery (visual analog scale 3.1 vs 1.1, p < 0.001) [4].
Only one previous study, by Audenet et al., has specifically addressed opioid use in RC surgery using a nonopioid (NOP) protocol [24]. NOP has similar aspects when compared to the ROU protocol including preoperative non-opioid analgesia and decreased reliance on PCA after surgery. In this study, 52 RARC patients following NOP were prospectively followed and retrospectively compared to 41 patients following conventional treatment. NOP patients experienced a significant decrease in postoperative opioid exposure (2.5 morphine milligram equivalents vs. 44 morphine milligram equivalents, p < 0.001), as well as a decrease in LOS (5 days vs. 7 days, p < 0.001) and time to regular diet. In our analyses, patients following the ROU protocol similarly experienced decreased time to regular diet, as well as faster return of bowel function and decreased time to ambulation. Given that increased opioid analgesic exposure is significantly correlated with postoperative ileus, increased time to first flatus, and time to tolerable oral diet, our result is not surprising [25]. Our LOS did not differ between cohorts, but was already 5.7±0.3 days prior to the implementation of our intervention. We also calculated opioid exposure at all phases of patient care, helping to identify strengths and areas for improvement of the ROU protocol.
The ROU protocol addresses an important aspect of postoperative pain management that has not been included in previous interventions –education and expectation-setting at patients’ preoperative clinic visit. While we cannot directly measure the impact of this intervention, patients with higher expectations of pain, ‘forecasted pain’, prior to surgery, have been shown to have higher ‘realized pain’ during their postoperative recovery [26–28]. Preoperative education aimed at increasing patient knowledge of pain has been shown to be an effective tool to aid in pain management [29]. Physicians and APPs can set expectations preoperatively to decrease pain and reduce patient opioid use. In the ROU protocol, education included ensuring the patient understood the nature of the surgery (i.e. ‘major’ or ‘invasive’ surgery), discussing the expectation of fatigue and muscle soreness after surgery, and emphasizing that symptoms improve with time. The side effects of narcotics, especially nausea and constipation, as well as the effectiveness of non-opioid analgesia were also discussed. Lastly, patients were provided with a handbook as a reference to facilitate understanding of their procedure.
Limitations
We acknowledge that our study has some important limitations. Patients were prospectively followed after implementation of the ROU protocol and retrospectively compared to a cohort of patients who underwent RC surgery prior to the invention. This may introduce temporal selection bias, and the Hawthorne effect may be observed among providers made aware of the study. While this may be a confounding factor when studying the independent effect of the ROU protocol, increasing mindfulness of opioid administration among healthcare providers can have an independent effect of decreasing patients’ opioid exposure. Since we relied on patient-reported symptoms and complications documented in clinical notes,we may not capture patients who were lost to follow-up or sought treatment at another institution. Lastly, the ROU protocol was introduced after the inclusion of robot-assisted RC at our institution. Although sub-group analysis of only patients undergoing open RC showed similar results to overall analysis, this limitation may confound our results.
CONCLUSION
Implementation of the ROU pathway for RC patients at our institution was associated with a 77% reduction in opioid exposure during the postoperative period, with no concomitant increase in pain. The ROU protocol improved clinical milestones of recovery, decreased ORADEs during inpatient admissions, and had comparable rates of complications, ED visits, readmissions, and all-cause mortality at 90-days compared to our traditional care pathway. Our study provides evidence that clinicians can rely on minimal, or even complete avoidance, of opioid analgesia for post-surgical pain management following RC. Future efforts will be directed at optimizing our protocol to reduce length of stay, to decrease intraoperative opioid exposure and opioid discharge prescriptions, and to collaborate with other institutions to promote the ROU protocol. This study highlights the role that urologists can play to decrease opioid exposure in our patient population.
ACKNOWLEDGMENTS
The authors have no acknowledgements.
FUNDING
The authors report no funding.
AUTHOR CONTRIBUTIONS
All authors contributed to the conception, performance, interpretation of data, draft preparation, and final review of this manuscript.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
REFERENCES
[1] | Cancer Stat Facts: Bladder Cancer. National Cancer Institute 2015. https://seer.cancer.gov/statfacts/html/urinb.html |
[2] | Alfred Witjes J , Lebret T , Compérat EM , et al. Updated 2016 EAU Guidelines on Muscle-invasive and Metastatic Bladder Cancer. Eur Urol. (2017) ;71: :462–75. doi: 10.1016/j.eururo.2016.06.020 |
[3] | Waingankar N , Mallin K , Smaldone M ,et al. Assessing the relative influence of hospital and surgeon volume on short-term mortality after radical cystectomy. BJU Int. (2017) ;120: :239–45. doi: 10.1111/bju.13804 |
[4] | Xu W , Daneshmand S , Bazargani ST ,et al. Postoperative pain management after radical cystectomy: Comparing traditional versus enhanced recovery protocol pathway. J Urol. (2015) ;194: :1209–13. doi: 10.1016/j.juro.2015.05.083 |
[5] | Shimko MS , Tollefson MK , Umbreit EC ,et al. Long-term complications of conduit urinary diversion. J Urol. (2011) ;185: :562–7. doi: 10.1016/j.juro.2010.09.096 |
[6] | Bhalla RG , Wang L , Chang SS ,et al. Association between preoperative albumin levels and length of stay after radical cystectomy. The Journal of Urology. (2017) ;198: :1039–45. doi: 10.1016/j.juro.2017.05.066 |
[7] | Patel HD , Ball MW , Cohen JE ,et al. Morbidity of urologic surgical procedures: An analysis of rates, risk factors, and outcomes. Urology. (2015) ;85: :552–9. doi: 10.1016/j.urology.2014.11.034 |
[8] | Parekh DJ , Reis IM , Castle EP ,et al. Robot-assisted radical cystectomy versus open radical cystectomy in patients with bladder cancer (RAZOR): An open-label, randomised, phase 3, non-inferiority trial. Lancet. (2018) ;391: :2525–36. doi: 10.1016/S0140-6736(18)30996-6 |
[9] | Brummett CM , Waljee JF , Goesling J ,et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surgery. (2017) ;152: :e170504. doi: 10.1001/jamasurg.2017.0504 |
[10] | Tyson MD , Chang SS . Enhanced recovery pathways versus standard care after cystectomy: A meta-analysis of the effect on perioperative outcomes. European Urology. (2016) ;70: :995–1003. doi: 10.1016/j.eururo.2016.05.031 |
[11] | Brandal D , Keller MS , Lee C ,et al. Impact of enhanced recovery after surgery and Opioid-Free anesthesia on opioid prescriptions at discharge from the hospital: A historical-prospective study. Anesth Analg. (2017) ;125: :1784–92. doi: 10.1213/ANE.0000000000002510 |
[12] | Opioid Morphine Equivalent Conversion Factors. Center for Medicare and Medicaid Services 2016. |
[13] | Anderson R , Saiers JH , Abram S ,et al. Accuracy in equianalgesic dosing. conversion dilemmas. J Pain Symptom Manage. (2001) ;21: :397–406. |
[14] | Glass PS , Hardman D , Kamiyama Y ,et al. Preliminary pharmacokinetics and pharmacodynamics of an ultra-short-acting opioid: Remifentanil (GI87084B). Anesth Analg. (1993) ;77: :1031–40. |
[15] | Clavien PA , Barkun J , de Oliveira ML ,et al. The Clavien-Dindo classification of surgical complications: Five-year experience. Ann Surg. (2009) ;250: :187–96. doi: 10.1097/SLA.0b013e3181b13ca2 |
[16] | Dindo D , Demartines N , Clavien P-A . Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. (2004) ;240: :205–13. |
[17] | Rudd RA , Aleshire N , Zibbell JE ,et al. Increases in Drug and Opioid Overdose Deaths–United States, 2000–2014. MMWR Morb Mortal Wkly Rep. (2016) ;64: :1378–82. doi: 10.15585/mmwr.mm6450a3 |
[18] | Jiang X , Orton M , Feng R ,et al. Chronic opioid usage in surgical patients in a large academic center. Ann Surg. (2017) ;265: :722–7. doi: 10.1097/SLA.0000000000001780 |
[19] | Christie C , Baker C , Cooper R ,et al. The President’s Commission on Combating Drug Addiction and the Opioid Crisis. The White House 2017. https://www.whitehouse.gov/sites/whitehouse.gov/files/images/Final_Report_Draft_11-1-2017.pdf (accessed 12 Mar 2019) |
[20] | Rutkow L , Vernick JS . Emergency legal authority and the opioid crisis. New England Journal of Medicine. (2017) ;377: :2512–4. doi: 10.1056/NEJMp1710862 |
[21] | Danna BJ , Wood EL , Baack Kukreja JE ,et al. The future of enhanced recovery for radical cystectomy: Current evidence, barriers to adoption, and the next steps. Urology. (2016) ;96: :62–8. doi: 10.1016/j.urology.2016.04.038 |
[22] | Rawlinson A , Kang P , Evans J ,et al. A systematic review of enhanced recovery protocols in colorectal surgery. The Annals of The Royal College of Surgeons of England. (2011) ;93: :583–8. doi: 10.1308/147870811X605219 |
[23] | Zhuang C-L , Ye X-Z , Zhang X-D ,et al. Enhanced recovery after surgery programs versus traditional care for colorectal surgery: A meta-analysis of randomized controlled trials. Dis Colon Rectum. (2013) ;56: :667–78. doi: 10.1097/DCR.0b013e3182812842 |
[24] | Audenet F , Attalla K , Giordano M ,et al. Prospective implementation of a nonopioid protocol for patients undergoing robot-assisted radical cystectomy with extracorporeal urinary diversion. Urologic Oncology: Seminars and Original Investigations Published Online First: February 2019. doi:10.1016/j.urolonc.2019.02.002 |
[25] | Koo KC , Yoon YE , Chung BH ,et al. Analgesic opioid dose is an important indicator of postoperative ileus following radical cystectomy with ileal conduit: Experience in the robotic surgery era. Yonsei Medical Journal. (2014) ;55: :1359. doi: 10.3349/ymj.2014.55.5.1359 |
[26] | Bayman EO , Parekh KR , Keech J ,et al. Preoperative patient expectations of postoperative pain are associated with moderate to severe acute pain after VATS. Pain Medicine. (2019) ;20: :543–54. doi: 10.1093/pm/pny096 |
[27] | Sipilä RM , Haasio L , Meretoja TJ ,et al. Does expecting more pain make it more intense? Factors associated with the first week pain trajectories after breast cancer surgery: PAIN. (2017) ;158: :922–30. doi: 10.1097/j.pain.0000000000000859 |
[28] | Alokozai A , Eppler SL , Lu LY ,et al. Can patients forecast their postoperative disability and pain? Clin Orthop Relat Res. (2019) ;477: :635–43. doi: 10.1097/CORR.0000000000000627 |
[29] | Louw A , Diener I , Butler DS ,et al. Preoperative education addressing postoperative pain in total joint arthroplasty: Review of content and educational delivery methods. Physiotherapy Theory and Practice. (2013) ;29: :175–94. doi: 10.3109/09593985.2012.727527 |




