Aquaporin 3 Expression Loss in Urothelial Carcinoma: Association with Tumor Invasion Depth, but not with Grading?
Abstract
We have previously provided molecular evidence of expression of aquaporin 3 (AQP3) in normal human urothelium and in UBC of various stages. Whereas former studies demonstrated that loss of AQP3 was associated with invasive and high-grade disease and worse progression-free and cancer-specific survival, this report investigates the expression of AQP3 in associated CIS. Contrary to what we had expected, all CIS specimens were shown to exhibit strong AQP3 expression, suggesting loss of AQP3 in UBC is primarily associated with the ability of tumor cells for invasion but not with grading as sign of dedifferentiation.
FIRST ANALYSIS OF AQP3 IN UROTHELIAL BLADDER CARCINOMA: ASSOCIATION WITH STAGE AND GRADING
The significance of aquaporin (AQP) water channels in UBC has been little investigated to date. AQPs are transmembrane pore-forming proteins that play a fundamental role in numerous physiological processes, most notably in fluid absorption and secretion. We have previously demonstrated that normal human urothelium expresses several AQPs, suggesting a potential role in water and urea transport across the urothelial layer [1]. Predominant expression of AQP3 is supportive of the hypothesis that the urothelium may be able to regulate osmolality and composition of the urine [2].
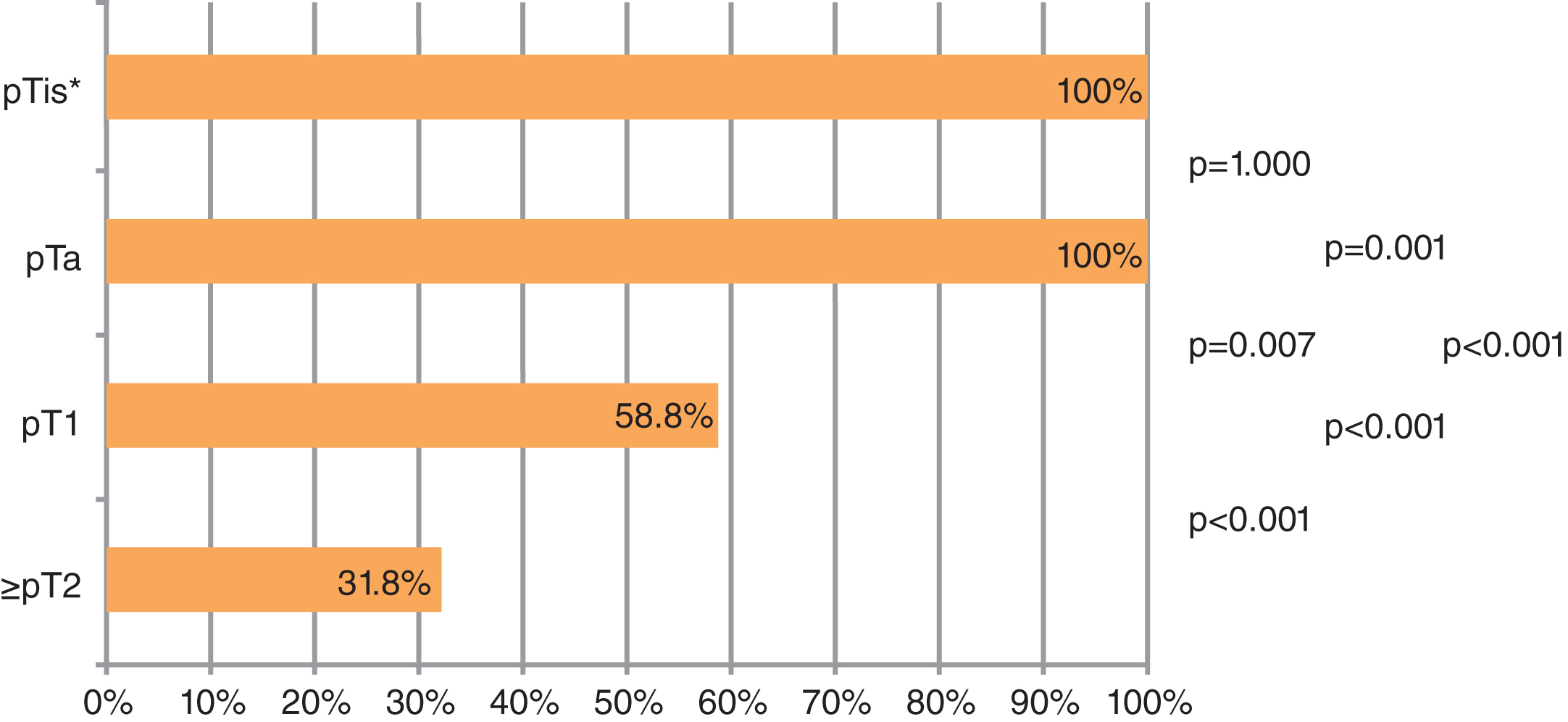
On the basis of these findings, we investigated the expression, localization and potential clinical significance of aquaporin water channels both in urothelial cancer cell lines and in human bladder carcinoma specimens of different stage and grade. In a first analysis, we performed immunohistochemical analysis of AQP3 in a small collective of 10 patients each with pTa, pT1 and pT2 UBC. We were able to demonstrate a strong AQP3 expression of 86% in stage pTa UBC, whereas it was expressed in only 66% and 33% of specimens of stage pT1 and pT2 tumors, respectively (p < 0.001). In terms of grading according to WHO 1973 classification, 100% of G1 tumors were AQP3 positive, however, only 89% and 28% of G2 and G3 tumors, respectively, were found to express AQP3 (p = 0.003). We concluded that AQPs may play a role in the progression of UBC and, in particular, that this could be of prognostic value [3].
AQP3 LOSS AS PROGNOSTIC PARAMETER FOR BLADDER CANCER IN EARLY- INVASIVE AND MUSCLE-INVASIVE STAGES
In support of this, we studied the potential prognostic role of AQP3 expression for early-invasive and muscle-invasive UBC. It could be demonstrated that loss of AQP3 protein expression was associated with worse progression-free survival in a subset of patients with pT1 bladder carcinoma (20% vs. 72%, p = 0.020). This finding was confirmed by multivariate Cox regression analysis (HR 7.58, CI 1.29 – 44.68; p = 0.025), where AQP3 was the only predictive parameter for PFS [4]. An independent analysis of patients with muscle-invasive UBC revealed a predictive value of loss or weak expression of AQP3 for worse PFS (19% vs. 75%, p = 0.043) and cancer-specific survival (CSS, 18% vs. 75%, p = 0.030). In multivariate Cox regression analysis AQP3 expression status was an independent prognostic parameter for PFS (HR 2.87, CI 1.06 – 7.7, p = 0.037) and CSS (HR 3.25, CI 1.20 – 8.77, p = 0.019), in addition to lymph node status and a positive surgical margin [5].
In our first studies into the role of AQP3 expression in urothelial bladder carcinoma we could demonstrate that loss of AQP3 was associated with tumor stage and grade and worse oncologic outcomes in patients with both early- and muscle-invasive UBC.
UNEXPECTED RESULTS IN A PRELIMINARY ANALYSIS OF AQP3 EXPRESSION IN ASSOCIATED CARCINOMA IN SITU
To date, the expression of AQP3 in CIS has not been investigated. First described by Melicow and Hollow in 1952, it is generally accepted that the presence of CIS is an unfavorable prognostic parameter despite the lack of invasive potential [6]. The aim of the present study was to investigate the expression and localization of AQP3 in a small series of CIS specimens associated with muscle-invasive UBC.
After informed consent was obtained from the patients, immunohistochemistry (IHC) was used to assess AQP3 protein expression in 14 patients having undergone cystectomy due to invasive UBC and associated CIS. Tumor stages were as follows: pT1 (n = 3), pT2 (n = 4), pT3 (n = 2) and pT4 (n = 5). All surgical samples were fixed in 10% formalin, dehydrated and embedded in paraffin wax. Dewaxed 4μm tissue sections were subjected to antigen retrieval by boiling for 10 min in tris-ethylenediaminetetraacetic acid (Tris-EDTA; pH 9) before labelling with titrated primary antibody (polyclonal anti-AQP3, host: rabbit, antigen: human AQP3, dilution 1 : 2000, Abcam, USA) for 16 h at 4°C.
All 14 specimens showed intense, homogeneous AQP3 expression in CIS whereas the marker was absent in adjacent (early) invasive tumors, as shown in Fig. 1. Retained expression of AQP3 by CIS is surprising given that CIS is commonly regarded as precursor of the poorly-differentiated non-papillary UBC phenotype. Furthermore, CIS was previously demonstrated to exhibit already many of the molecular aspects found in invasive UBC. The presence of CIS was also shown to be associated with specific genetic alterations found in patients with poor prognosis [7]. Taken together with the results from our previous studies, AQP3 appears to be consistently expressed in non-invasive UBC, irrespective of growth pattern and differentiation (see Fig. 2).
We conclude that loss of AQP3 expression in UBC is involved in mechanisms associated with tumor cell invasion beyond the basement membrane into the submucosa and muscle layers irrespective of tumor differentiation. Our findings provide a valid platform for further studies into the biological and prognostic significance of the expression of AQP3 in muscle-invasive urothelial UBC.
CONFLICT OF INTEREST
The authors declare that there are no conflicts of interest or imbursement with their submission. There was no funding by any institution not being associated to the affiliations of the authors. The study was undertaken according to local ethics committee approval.
ACKNOWLEDGMENTS
The authors thank Mrs. Stefanie Götz und Dr. Fabian Eder for their assistance.
REFERENCES
[1] | Rubenwolf PC , Georgopoulos NT , Clements LA , Feather S , Holland P , Thomas DF , Southgate J . Expression and localisation of aquaporin water channels in human urothelium in situ and in vitro. Eur Urol (2009) ;56: (6):1013–23. |
[2] | Rubenwolf PC , Georgopoulos NT , Kirkwood LA , Baker SC , Southgate J . Aquaporin expression contributes to human transurothelial permeability in vitro and is modulated by NaCl. PLoS One (2012) ;7: (9):e45339. |
[3] | Rubenwolf PC , Otto W , Denzinger S . Expression of aquaporin water channels in human urothelial carcinoma: correlation of AQP3 expression with tumour grade and stage? Eur Urol (2012) ;61: (3):627–8. |
[4] | Otto W , Rubenwolf PC , Burger M , Fritsche HM , Rößler W , May M , Hartmann A , Hofstädter F , Wieland WF , Denzinger S . Loss of aquaporin 3 protein expression constitutes an independent prognostic factor for progression-free survival: An immunohistochemical study on stage pT1 urothelial bladder cancer. BMC Cancer (2012) ;12: :459. |
[5] | Rubenwolf P , Thomas C , Denzinger S , Hartmann A , Burger M , Georgopoulos NT , Otto W . Loss of AQP3 protein expression is associated with worse progression-free and cancer-specific survival in patients with muscle-invasive bladder cancer. World J Urol (2015) ;33: (12):1959–64. |
[6] | Babjuk M , Burger M , Zigeuner R , Shariat SF , van Rhijn BW , Compérat E , Sylvester RJ , Kaasinen E , Böhle A , Palou Redorta J , Rouprêt M . EAU guidelines on non-muscle-invasive urothelial carcinoma of the bladder: Update 2013. Eur Urol (2013) ;64: (4):639–53. |
[7] | Burger M , Oosterlinck W , Konety B , Chang S , Gudjonsson S , Pruthi R , Soloway M , Solsona E , Sved P , Babjuk M , Brausi MA , Cheng C , Comperat E , Dinney C , Otto W , Shah J , Thürof J , Witjes JA . ICUD-EAU International Consultation on Bladder Cancer 2012: Non-muscle-invasive urothelial carcinoma of the bladder. Eur Urol (2013) ;63: (1):36–44. |
Figures and Tables
Fig.1
(A-C) Carcinama in situ showing intense AQP3 expression (10 fold mognification, A; 40 fold magnification, B). Note the complete loss of AQP3 in the associated adjacent muscle-inasive tumor (C).
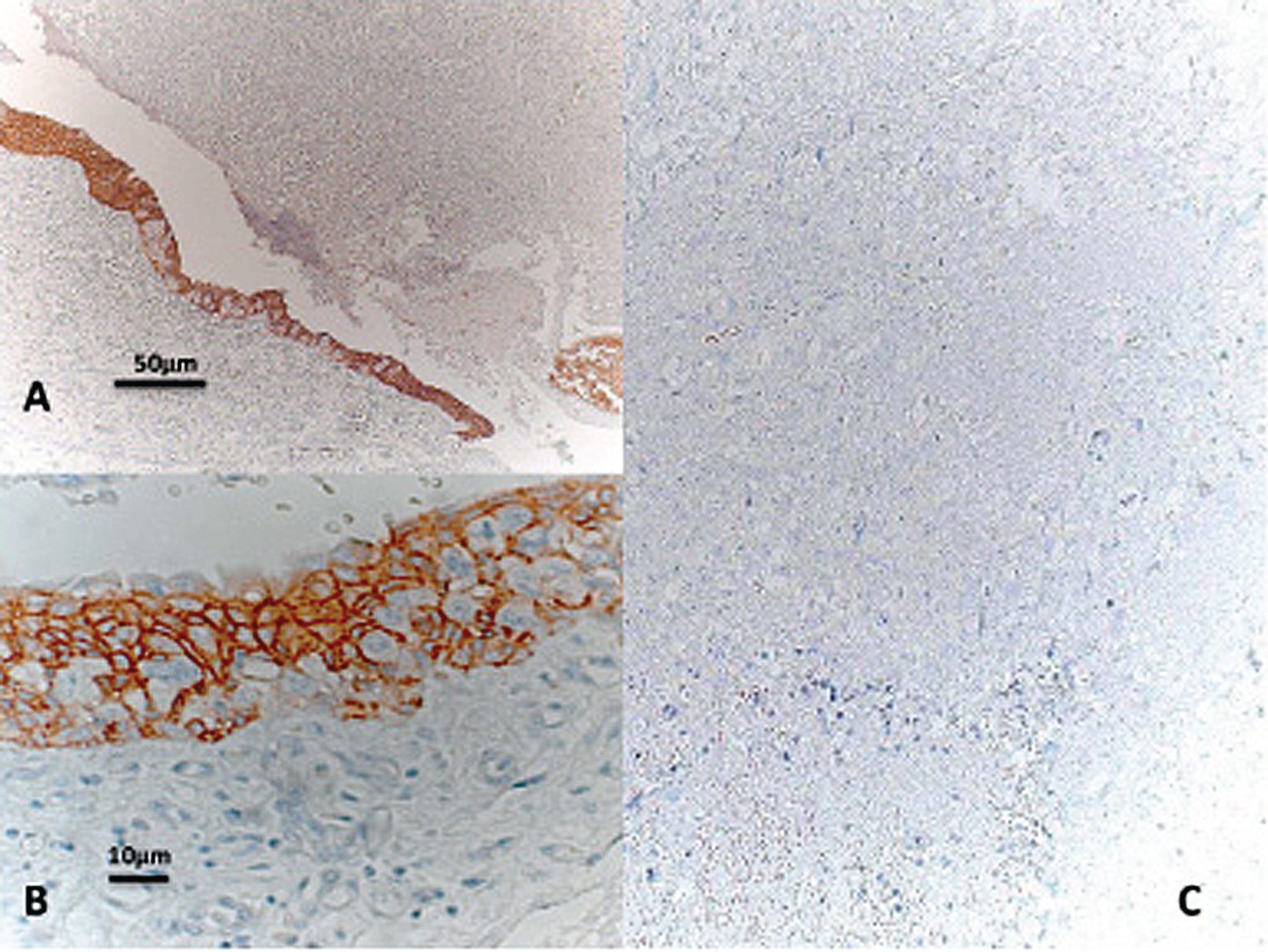
Fig.2
Rates of aquaporin 3 (AQP3) expression in urothelial bladder cancer (UBC) of various stages. *Associated carcinoma in situ.