The Prognostic Value of Previous Irradiation on Survival of Bladder Cancer Patients
Abstract
Background: Radiation exposure is an established risk factor for bladder cancer, however consensus is lacking on the survival characteristics of bladder cancer patients with a history of radiation therapy (RT). Confounding patient comorbidities and baseline characteristics hinders prior attempts at developing such a consensus.
Objective: To compare the survival characteristics of patients with suspected radiation-induced second primary cancer (RISPC) of the bladder to those with de novo bladder cancer, taking into account the patient comorbidities and baseline characteristics predictive of survival.
Methods: Retrospective analysis of patients with muscle-invasive (≥T2a) or BCG-refractory stage Tis-T1 urothelial bladder cancer. Patients were excluded if prior RT exposure was used as treatment for bladder cancer or if cause of death was due to post-operative complications. A digit matching propensity score algorithm was used to match patients with prior radiation treatment to those without prior treatment. Cox regression analysis for time until death was performed following creation of the propensity score matched sample.
Results: 29 patients with history of RT were matched with two controls each, resulting in a dataset of 87 observations in the event model. Results from the Cox model indicate a significantly increased hazard ratio for death at 2.22 (p = 0.047, 95% CI: 1.015–4.860) given a history of prior radiation therapy.
Conclusions: In a small cohort, bladder cancer patients who underwent cystectomy had a significantly higher risk of death in the face of prior pelvic RT. This effect was found to be independent of surgical complications, numerous established patient characteristics and comorbidities traditionally predictive of survival.
INTRODUCTION
Bladder cancer remains a prominent malignancy with 74,690 new cases in 2014 and 15,580 deaths in the United States, with those most at risk consisting of Caucasian men, smokers, and those with certain chemical and parasite exposures [1]. Another less frequent but well-established risk factor is prior radiation exposure, first evidenced in studies of post-nuclear survivors and later in occupational exposure and medical radiation analyses [2]. The energy of ionizing radiation causes damage to nearly all cellular components, however double stranded DNA breaks induced by radiation therapy in particular allow for abberant repair mechanisms and create potential for mutation and carcinogenesis. Different mechanisms for local RISPCs have been proposed, while the well described “bystander effect” may provide an explanation for distant RISPCs following radiation therapy (RT), however such evidence for systemic cytokine signaling and long distance intercellular communication following RT is largely limited to cellular and animal studies [3–6]. Nevertheless, an overwhelming amount of experimental and observational evidence in the scientific literature links high-dose ionizing radiation to carcinogenesis, and in studies of patients receiving high doses of radiation, a linear dose-response relationship has been described between radiation exposure and secondary cancer risk [7, 8].
Most frequently reported and well-substantiated by large studies are secondary cancers following RT to the pelvis [9–13]. The association of RT and secondary cancers is thought to be due to low-dose radiation scatter that is not completely effective in destroying cells in the periphery of the target field [14–16]. While the risk of radiation-induced second primary cancer (RISPC) is small, these carcinomas are often high grade and muscle-invasive, with evidence from histological studies linking radiation exposure to higher rates of p53 mutations and the in vitro progression from low-grade to high-grade tumors [17]. Whole genome mRNA expression profiling identified subtypes of muscle invasive bladder cancer (MIBC), among these was a neoadjuvant chemotherapy-resistant subtype which expresses an active p53 gene expression signature [18].
Still under debate, however, is the overall effect of RISPCs on disease-specific survival. There are variable and conflicting data supporting both a worse and equivalent survival rate for patients with a history of radiation treatment compared to those without a history of radiation [19]. The exact effect is difficult to discern due to variably long latency periods reported between RT and RISPC, the variation in treatment variables collected amongst large registry studies, and gaps of information in patient comorbidities. In the present investigation we seek to compare survival outcomes for patients undergoing cystectomy for bladder cancer between those with and without a history of pelvic RT, taking into account patient comorbidities and other characteristics determined to be independent predictors of survival as reported in the literature. We hypothesize that, among bladder cancer patients, those with a history of pelvic RT will exhibit lower survival rates when taking into account baseline patient demongraphics and comorbidities.
METHODS
Patient selection
From 2006 to 2014, 389 patients were seen at a tertiary care center for muscle-invasive or recurrent bladder cancer refractory to intravesical therapy. Inclusion criteria consisted of patients with muscle invasive urothelial bladder cancer, pathologic stage ≥T2a diagnosed on biopsy, or with recurrent (BCG-refractory) pathologic stage Tis-T1 bladder cancer. Review was approved by the Colorado Multiple Institutional Review Board (COMIRB). Records were retrospectively reviewed and divided into two groups: A case group consisting of cystectomy patients with a history of prior pelvic radiation, including electron beam radiation therapy (EBRT) or brachytherapy (BT) directed towards prostate, cervical or colorectal cancer, and a control group, consisting of cystectomy patients with no history of radiation therapy of any kind. We used a commonly employed criteria for suspected RISPC in our inclusion criteria [20].
1. The tumor arose in a previously irradiated field.
2. The new tumor is histologically different from the original condition.
3. There was no evidence of the new tumor at the time of radiation therapy.
4. A latency period existed between irradiation and the development of the new tumor.
Patients were excluded from review if they had a prior history of radiation therapy as treatment for bladder cancer or if death was determined to be due to immediate post-operative complications within 30 days, since the objective of this study was not to assess for death due to higher associated surgical complications. This retrospective review was performed with approval from the institutional review board.
Data collection
We included several covariates found in the literature to be independent predictors of survival, as well as perioperative risks and complications. Patient variables examined included age, TNM stage, Charlson Comorbidity Index (CCI), smoking history, gender, history of recurrent bladder cancer, intravesical BCG use, family history of cancer, chemical exposures or high risk occupations related to bladder cancer, (textile workers, tire workers, truck drivers, dye workers, leather workers, painters, hair dressers, chemical workers etc.), peripheral vascular disease and marital status. Perioperative variables included use of chemotherapy, pre-operative laboratory values (BUN, creatinine, hemoglobin, platelet count), hydronephrosis and post-operative complications (bowel obstruction, abscess, infection, diarrhea, anastomotic urine leak).
Treatment and follow-up
All patients were treated by one of three surgeons with cystectomy and ileal conduit, continent urinary diversion to the skin, or orthotopic neobladder using an open or robotic-assisted laparoscopic approach with bilateral lymph node dissection and/or chemotherapy when indicated. A standard lymph node dissection was performed in all cases where it did not expose the patient to unnecessary harm. The traditional limits of dissection include the bifurcation of the aorta superiorly, the endopelvic fascia or circumflex iliac vein inferiorly, the genitofemoral nerves laterally and the obturator nerve and vessels posteriorly. In cases where a complete dissection was not safe due to prior pelvic vascular surgery, high-dose radiation, or extreme age, only the nodes in this region that could be safely dissected were removed, and sent to pathology. Pathological specimens were reviewed by a histopathologist for neoplastic cell type and nodal involvement. All patients were treated within 3 months of diagnostic TURBT or BCG treatment failure and followed periodically in clinic throughout the course of their lives.
End points
The primary end-point was overall survival determined from death reports by hospital records and/or national social security death index.
Statistical analysis
A digit matching propensity score algorithm was used to match patients with prior radiation treatment to those without prior treatment wherein two controls were matched to the cases to provide a 1:2 ratio [21]. Propensity scores were computed using logistic regression for RT as the dependent variable. Due to the small sample size not all available variables could be entered into the propensity model. Selection was based on factors most likely to affect RT and convergence of the model. The independent variables included age, stage (1–4), use of chemotherapy, BCG use, preoperative lab values (BUN, serum creatinine, and hemoglobin), age-adjusted weighted CCI, hydronephrosis, smoking history, whether or not the patient was being treated for recurrent cancer at time of cystectomy, family history of non-prostatic cancer, and high-risk occupation. The distribution of propensity scores were plotted for the unmatched and matched cohorts. Balance of covariates between RT cohorts before and after matching were calculated using Fisher’s Exact 2 × 2 Test and Mann-Whitney U Test for dichotomous and continuous variables, respectively. After the propensity score matched sample was created, Cox regression analysis for time until death was performed using discrete and continuous variables: prior radiation therapy, stage, node status (N0-N3), age at time of cystectomy, platelet count, marital status (married, single, widowed), and post-operative complications (abscess, bowel obstruction, diarrhea, infection, urine leak). All analyses were performed using SAS 9.4 Copyright (c) 2002–2012 by SAS Institute Inc., Cary, NC, USA.
RESULTS
Study cohort
There were 340 patients included in the analytic data set treated with cystectomy, 307 patients had no RT of any kind, 33 patients had a history of pelvic radiation including EBRT (14), brachytherapy (11), or EBRT +brachytherapy (8) prior to cystectomy. 14 patients were excluded due to post-operative complications (Table 1). The median duration between radiation therapy and diagnosis of bladder cancer was 8 years, range 3 to 11 years. At the time of analysis, 145 of 340 patients were deceased, 27 had identifiable causes of death unrelated to bladder cancer. The median age of our cohort was 66 years (range 28–91 years), and median time from procedure to time of death for all patients in the study was 298 days (range 39–3557 days), with a median follow-up length of 303 days (range 39–3557 days).
Propensity score matching
Of the 33 cases, only 29 could be matched with two controls, resulting in a dataset of 87 observations in the event model. Figures 1 and 2 provide the distribution of propensity scores before and after matching, respectively. Although the four unmatched cases all terminated in death, their dates of death and time from procedure until death were within the respective ranges for those cases resulting in death in the matched data. Table 2 shows the Fisher Exact and Mann-Whitney U tests as measures of balance between covariates before and after matching. Standardized differences are not reported due to the small sample size and the increased leverage that imparts to extremeobservations.
Cox regression analysis
Outcomes of subsequent Cox Regression analysis of the matched sample are provided in Table 3. The Cox model for time until death included prior radiation therapy, complications arising after surgery, age at surgery, platelet count, nodes, and marital status. Complications included abscess, bowel obstruction, diarrhea, infection and urine leak. Complications that were single valued after matching and excluded from the event model included adhesions and enteric fistula.
Results from the Cox model indicate a significantly increased hazard ratio for death at 2.22 (p = 0.047) given a history of prior radiation therapy compared to patients who did not receive prior radiation. Other covariates with significant change in risk of death include platelet count (HR = 0.991, p = 0.001) (i.e. for every increase of 1,000 platelets per mcL there is a 0.8% decrease in the probability of dying), married versus single (HR = 4.04, p = 0.013), and node status when comparing TNM stages N0 and N2 (HR = 4.30, p = 0.011) (Fig. 3).
DISCUSSION
This is, to our knowledge, the first survival comparison of cystectomy patients to employ a propensity score for history of radiation therapy. Given the wealth of conflicting data on survival from RISPC compared to de novo cancers, analyses that seek to reduce selection bias and account for established survival factors are necessary to delineate the true magnitude of any such observed effects while controlling for potential confounders. For instance, a low preoperative hemoglobin (<12 g/dl) was found to be an independent predictor of cancer-specific survival, and while our data do not corroborate this, prior research suggests that thrombocytosis is not only associated with poor survival but is an indicator of other unfavorable hematologic abnormalities that confer worse prognosis [22, 23]. Social characteristics such as marital status also exert major impacts on survival, with one study finding increased hazard ratios for death following cystectomy of 1.20 (p < 0.001) and 1.4 (p = 0.005) for single and widowed patients, respectively, when compared to their married counterparts [24]. Analysis of SEER data for bladder cancer patients found an increased risk of death for male widows of 1.74 which rises to 2.1 if over the age of 70 [25]. Radiation can also cause perioperative complications such as fluid collections, pelvic abscesses, and other fibrotic changes, making post-operative complications essential variables as well [26, 27]. In addition, recent data suggests that a history of radiation creates a propensity for positive margin status and subsequent worse survival, and while the absolute proportion of patients with positive margins was higher in the radiated group, this was not determined to be significant [28]. Smoking, on the other hand, is a well known risk factor for the development of urothelial cancer but is an uncertain prognostic factor in MIBC, with only suggestive evidence that quitting may improve overall survival [29].
For these reasons, we attempted to include as many of these variables as possible. Patient comorbidities have been speculated to account for decreased survival in RT cohorts, and our baseline patient characteristics would appear to support this assumption with a discrepancy between calculated CCI scores for the case and control groups of 4.85 and 2.85, respectively. We feel the use of CCI was valuable in controlling for these factors as validated by Koppie et al. (2008) and recommended for this very type of investigation [30]. In addition, the aforementioned variable latency between RT and RISPC leads to speculation on the origin of second primary cancers, but consensus seems to exist that secondary cancers arise after at least 5 years [7] This was corroborated by Liauw et al. (2006) when following 348 patients for a median of 10.5 years following treatment with brachytherapy and/or EBRT, noting that while the incidence of bladder cancers 5 years after RT was higher than observed rates, the rate of secondary cancers in the RT group did not change appreciably with extended follow-up intervals [31]. Large registry studies also corroborate these findings, demonstrating increased risks for secondary cancers following radiation therapy for prostate cancer only after 5 years [16]. Our study is in agreement with these findings with an average onset of suspected RISPC of the bladder at 7 years and 9 months following RT.
It should be noted that non-invasive lesions as well as muscle-invasive lesions were included, and while each have different survival rates at baseline, the proportion of these in each group was very similar and comprised a small minority of each cohort. Furthermore, since cancer aggressiveness is the item of interest, we do not feel that this cystectomy population should be excluded from analysis.
Given the difficulty in obtaining data with a sufficient number of prior RT and deaths from single institution, this is an exploratory analysis only. The power of the study to detect significant risk for the 12 factors in the Cox regression is limited by the small sample size of 58 controls and 29 cases. In this hypothesis generating study, although the standard errorsare unstable due to the oversaturated model for the propensity scores and outcomes, the point estimates have been retained to locate possible significant hazard ratios. We recognize as a limitation to this study that future studies with greater number of outcomes and more power may refute these findings. With an overall count of events in the matched data of only 45 deaths, a more rigorous analysis would restrict the number of covariates on the right hand side of the predictive Cox model to no more than 4 or 5 covariates given the low number of events. However, since the 4 covariates that have significant hazard ratios were found even in the presence of the other covariates their effect on outcome is most probably not a spurious finding, though in future similar studies with a larger number of events the point estimate may vary considerably from those reported here. Furthermore, the sample size is most likely the basis for a lack of significant survival discrepancy amongst T stages in our cohort and the limited discrepancy between node statuses.
Due to such model constraints it was decided to evaluate chemotherapy collectively instead of separating it into neo-adjuvant versus adjuvant covariates. In our entire cohort approximately 51.9% of chemotherapy was delivered neo-adjuvantly, however future analyses with larger sample sizes should consider evaluating adjuvant versus neo-adjuvant as separate covariates.
Even though there are observed mortality differences between those who had radiotherapy and those who did not, its been suggested that this may be attributable to delay in bladder cancer diagnosis due to diagnostic complications resulting from non-urologists conducting radiation and follow up or from clinically significant hematuria being overlooked as a normal finding in post-radiotherapy patients [2]. While patients were treated within 3 months of diagnosis in the current analysis, it is possible that patient referrals were delayed for these reasons and patient reports of how long ago they first noticed hematuria varied widely between patients. While onset of hematuria as a marker for carcinogenesis was not felt to be a reliable data point, cystoscopy reports prior to radiation were also not available to determine whether or not pre-existing urothelial cancer was present. Radiation technology and approach has also certainly changed over the course of our study, and while taken together in form and location within the pelvic region there is a significant effect on survival, the results may differ when evaluating isolated forms and locations of RT applied within the pelvic field.
Radiation is also difficult to associate with a specific outcome because of changes in technology. Intensity-modulated radiation therapy (IMRT), for instance, is a newer form of radiotherapy that results in low dose normal tissue irradiation but carries an increased risk of leakage contributing to theoretical increased out-of-field RISPC risk. The RISPC risk is still held to be variable and small for IMRT, however the effect has only begun to be investigated in recent years [16]. Contemporary studies comparing newer forms of radiotherapy continue to find increased risk for RISPC for two-dimensional radiation therapy, most frequently of the bladder, however IMRT may have a reduced potential for RISPC despite conflicting experimental evidence [32, 33].
The analysis underscores the multitude of factors that underlie survival and the inherent complexity in estimating which patients will survive longer. These results add value to our understanding of patient characteristics that more strongly portend a worse survival. At this time, the results indicate a significantly increased risk of death given a history of radiation when taking into account several independent risk factors for survival and baseline patient characteristics, lending support to the idea that radiation may contribute to the development of cancers of a more aggressive nature than those arising de novo.
CONCLUSION
Previous reports on RISPC survival following cystectomy have been largely case based, observational, or of limited control of comorbidities. We sought to perform an exploratory analysis using increased number of regression analysis covariates that have been found to be associated with survival following cystectomy. These results demonstrate a significant association between history of RT and reduced survival rates after adjusting for patient demographics, comorbidities and surgical complications, however this study is limited by small sample size and lack of standardization of RT. Future studies should take into account surgeon experience, geographic variation, radiation type, amount, and route of administration, and the myriad of survival cofactors in order to refine the presentconclusions.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
ACKNOWLEDGMENTS
No external sources of funding were used in this analysis.
REFERENCES
1 | Siegel R, Ma J, Zou Z, Jemal A(2014) Cancer statistics, CA: A Cancer Journal for Clinicians64: 1929doi:10.3322/caac.21208 |
2 | Sountoulides P, Koletsas N, Kikidakis D, Paschalidis K, Sofikitis N(2010) Secondary malignancies following radiotherapy for prostate cancerTher Adv Urol2: 3119125doi:10.1177/1756287210374462 |
3 | Kumar S(2012) Second Malignant Neoplasms Following RadiotherapyInternational Journal of Environmental Research and Public Health9: 1247444759doi:10.3390/ijerph9124744 |
4 | Sachs RK, Brenner DJ(1304) Solid tumor risks after high dosesof ionizing radiationPNAS102: 371304013045doi: 10.1073/pnas.0506648102 |
5 | Mullenders L, Atkinson M, Paretzke H, Sabatier L, Bouffler S(2009) Assessing cancer risks of low-dose radiationNature Reviews Cancer9: 8596604doi:10.1038/nrc2603 |
6 | Prise KM, O’Sullivan JM(2009) Radiation-induced bystander signalling in cancer therapyNature Reviews Cancer9: 5351360doi:10.1158/0008-5472.CAN-10-0276 |
7 | Li CI, Nishi N, McDougall JA, Semmens EO, Sugiyama H, Soda M(7187) Relationship between Radiation Exposure and Risk ofSecond Primary Cancers among Atomic Bomb SurvivorsCancer Research70: 1871877198doi: 10.1158/0008-5472.CAN-10-0276 |
8 | Wakeford R(2004) The cancer epidemiology of radiationOncogene23: 3864046428doi:10.1038/sj.onc.1207896 |
9 | Boice JD, Engholm G, Kleinerman RA(1988) Radiation dose and second cancer risk in patients treated for cancer of the cervixRadiation Research116: 1355 |
10 | Neugut AI, Ahsan H, Robinson E, Ennis RD(1997) Bladder carcinoma and other second malignancies after radiotherapy for prostate carcinomaCancer79: 816001604doi:10.1002/cncr.28769 |
11 | Davis EJ, Beebe-Dimmer JL, Yee CL, Cooney KA(2735) Risk of secondprimary tumors in men diagnosed with prostate cancer: Apopulation-based cohort studyCancer120: 1727352741doi: 10.1002/cncr.28769 |
12 | Kleinerman RA, Boice JD, Storm HH(1995) Second primary cancer after treatment for cervical cancer. An international cancer registries studyCancer76: 3442452doi: 10.1002/1097-0142(19950801)76:3<442::AID-CNCR2820760315>3.0.CO;2-L |
13 | Kendal WS, Nicholas G(2007) A population-based analysis of second primary cancers after irradiation for rectal cancerAmerican Journal of Clinical Oncology30: 4333339 |
14 | Senkus E, Konefka T, Nowaczyk M, Jassem J(2001) Second lower genitaltract squamous cell carcinoma followingcervical cancerActa Obstetricia et Gynecologica Scandinavica79: 9765770doi: 10.1034/j.1600-0412.2000.079009765.x |
15 | Filatova E. Multiple primary vaginal tumors following the radiation treatment of cervical cancer. Vopr Onkol 1990; 36(6):726-9. http://eutils.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&id=2378094&retmode=ref&cmd=prlinks |
16 | Murray L, Henry A, Hoskin P, Siebert FA, Venselaar (2013) Second primary cancers after radiation for prostate cancer: A review of data from planning studiesRadiat Oncol8: 1172 |
17 | Sandhu JS, Vickers AJ, Bochner B, Donat SM, Herr HW, Dalbagni G(2006) Clinical characteristics of bladder cancer in patientspreviously treated with radiation for prostate cancerBJU Int98: 15962doi:10.1111/j.1464-410X.2006.06182.x |
18 | Choi W, Porten S, Kim S(2014) Identification of distinct basal and luminal subtypes of muscle-invasive bladder cancer with different sensitivities to frontline chemotherapyCancer Cell25: 2152165doi:10.1016/j.ccr.2014.01.009 |
19 | Murray L, Henry A, Hoskin P, Siebert F-A, Venselaar JPROBATE group of GEC ESTRO(2014) Second primary cancers after radiation forprostate cancer: A systematic review of theclinical dataand impact of treatment techniqueRadiother Oncol110: 2213228doi:10.1016/j.radonc.2013.12.012 |
20 | Cahan WG, Woodard HQ, Higinbotham NL, Stewart FW, Coley BL(1998) Sarcoma in irradiated bone. Report of eleven cases – 1948Cancer82: 1834doi: 10.1002/1097-0142(194805)1:1<3::AID-CNCR2820010103>3.0.CO;2-7 |
21 | Parsons LS(2004) Performing a1:N Case-Control Match on Propensity ScoreProceedings of the Twenty-Sixth Annual SAS Users Group International Conference |
22 | Todenhöfer T, Renninger M, Schwentner C, Stenzl A, Gakis G(2012) A new prognostic model for cancer-specific survival after radical cystectomy including pretreatment thrombocytosis and standard pathological risk factorsBJU IntE110: 11 Pt B533540doi:10.1111/j.1464-410X.2012.11231.x |
23 | Gakis G, Todenhöfer T, Stenzl A(2011) The prognostic value of hematological and systemic inflammatory disorders in invasive bladder cancerCurr Opin Urol21: 5428433doi:10.1097/MOU.0b013e32834966fa |
24 | Gore JL, Kwan L, Saigal CS, Litwin MS(2005) Marriage and mortality in bladder carcinomaCancer104: 611881194doi:10.1002/cncr.21295 |
25 | Nelles JL, Joseph SA, Konety BR(2009) The impact of marriage on bladder cancer mortalityUrol Oncol27: 3263267doi:10.1016/j.urolonc.2008.04.016 |
26 | Schuster TG, Marcovich R, Sheffield J, Montie JE, Lee CT. Radical cystectomy for bladder cancer after definitive prostate cancer treatment. Urology 2003;61(2):342- 7–discussion347. http://eutils.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&id=12597943&retmode=ref&cmd=prlinks |
27 | Tolhurst SR, Rapp DE, O’Connor RC, Lyon MB, Orvieto MA, Steinberg GD(2005) Complications after cystectomy and urinary diversion in patients previously treated for localized prostate cancerUrology66: 4824829doi:10.1016/j.urology.2005.04.046 |
28 | Luchey AM, Lin H-Y, Yue B(2015) Implications of Definitive Prostate Cancer Therapy on Soft Tissue Margins and Survival in Patients Undergoing Radical Cystectomy for Bladder Urothelial CancerJ Urol [Epub ahead of print] |
29 | Aveyard P, Adab P, Cheng KK, Wallace DMA, Hey K, Murphy MFG(2002) Does smoking status influence the prognosis of bladdercancer? A systematic reviewBJU Int90: 3228239doi: 10.1046/j.1464-410X.2002.02880.x |
30 | Koppie TM, Serio AM, Vickers AJ, Vora K, Dalbagni G, Donat SM(2008) Age-adjusted Charlson comorbidity score is associated with treatment decisions and clinical outcomes for patients undergoing radical cystectomy for bladder cancerCancer112: 1123842392doi:10.1002/cncr.23462 |
31 | Liauw SL, Sylvester JE, Morris CG, Blasko JC, Grimm PD(2006) Second malignancies after prostate brachytherapy: Incidence of bladder and colorectal cancers in patients with 15 years of potential follow-uInt J Radiat Oncol biol Phys66: 3669673doi:10.1016/j.ijrobp.2006.05.016 |
32 | Nieder AM, Porter MP, Soloway MS(2008) Radiation therapy forprostate cancer increases subsequent risk of bladder andrectal cancer: A population based cohort studyJ Urol180: 520052009discussion2009–10. doi:10.1016/j.juro.2008.07.038 |
33 | Huang J, Kestin LL, Ye H, Wallace M, Martinez AA, Vicini FA(2011) Analysis of second malignancies after modern radiotherapy versusprostatectomy for localized prostate cancerRadiotherapy and Oncology98: 18186doi:10.10.16/j.radonc.2010.09.012 |
Figures and Tables
Fig.1
Distribution of propensity scores before matching. No XRT: No history of radiation therapy. XRT: History of radiation therapy.
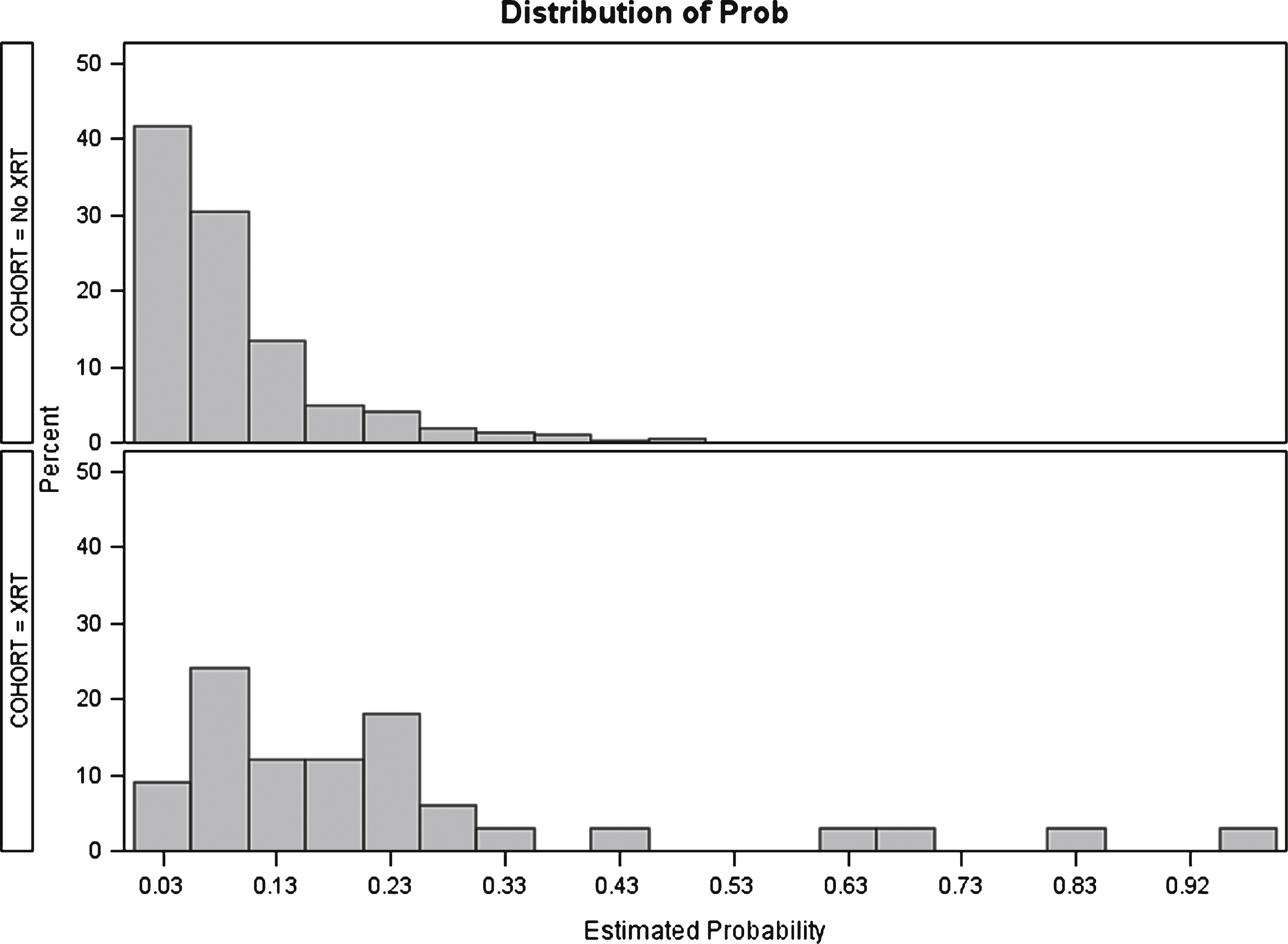
Fig.2
Distribution of propensity scores after matching. No XRT: No history of radiation therapy. XRT: History of radiation therapy.
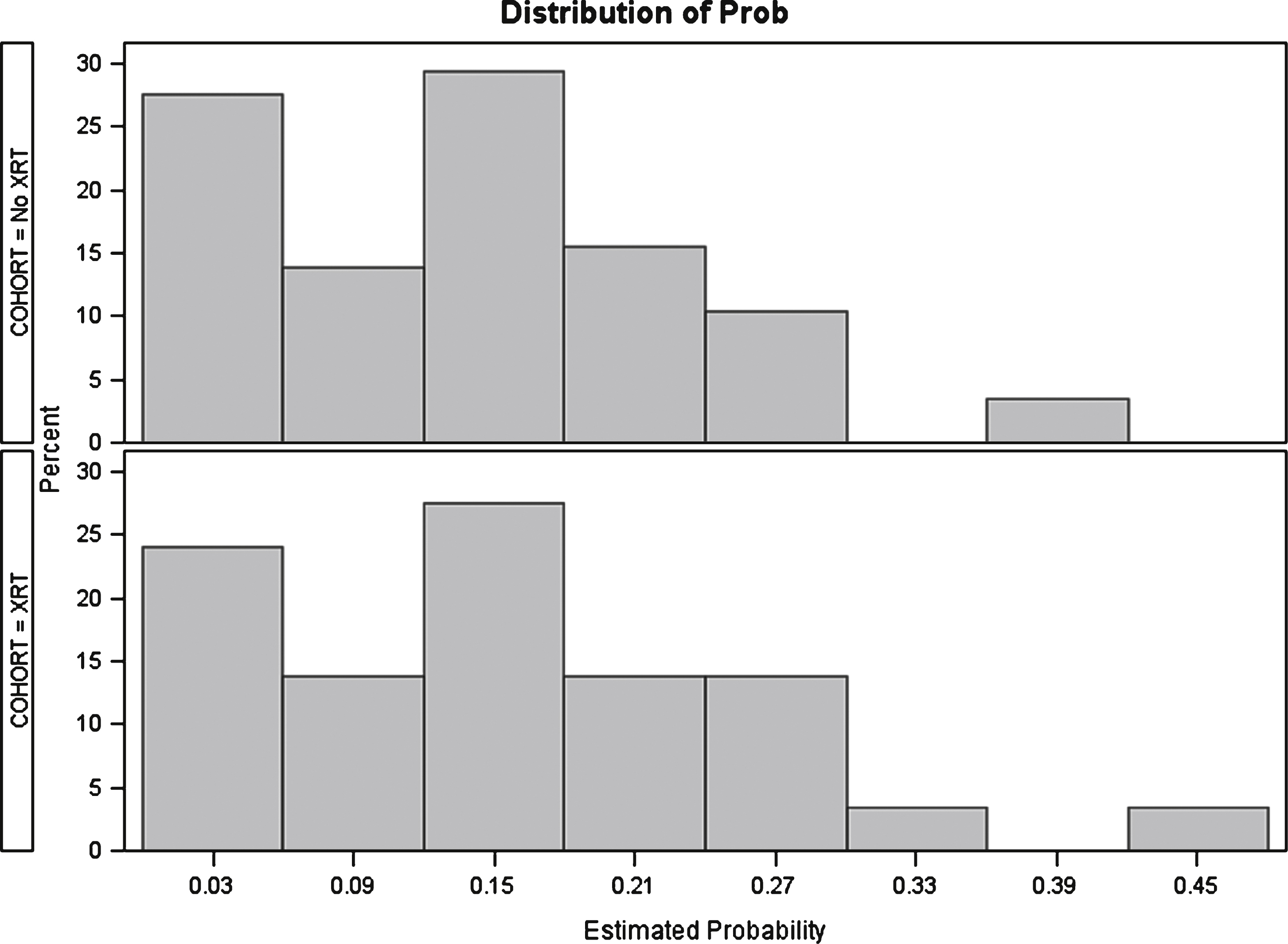
Fig.3
Significant cox regression variables. RT: Radiation therapy. UTI: Urinary tract infection.
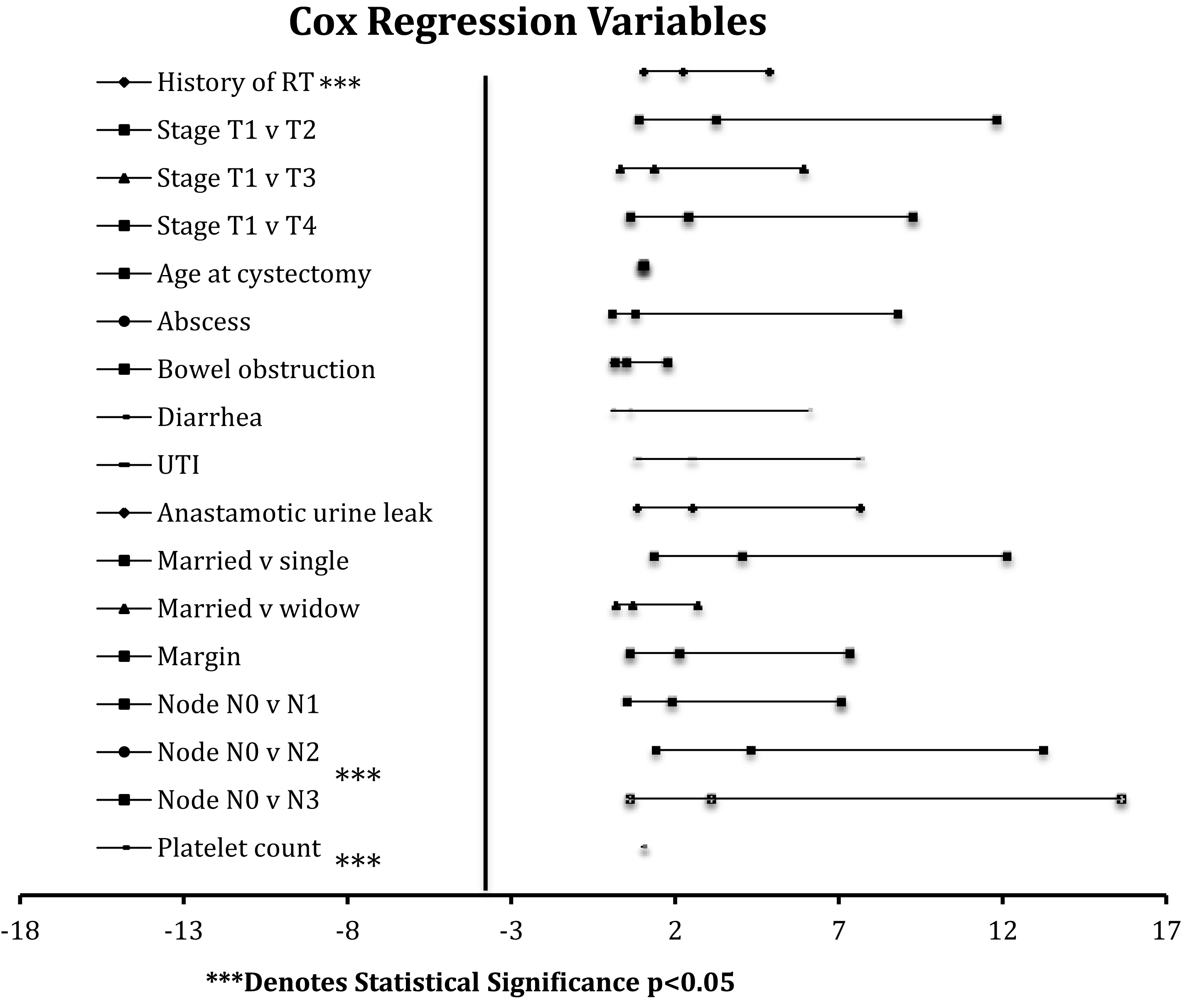
Table 1
Patient exclusion record
| Perioperative causes of death | Number of patients |
| Cardiopulmonary failure | 6 |
| Severe septic shock | 2 |
| Respiratory failure | 2 |
| Unknown causes | 1 |
| Intra-op DIC of unknown origin | 1 |
| PE/pneumonia | 1 |
| Saddle embolus and hemorrhagic shock | 1 |
| Intra-op cardiac arrest | 1 |
PE: Pulmonary embolism. DIC: Disseminated intravascularcoagulation.
Table 2
Fisher Exact and Mann-Whitney U tests as measures of balance between covariates before and after matching
| Fisher’s Exact 2 × 2 Test: count (proportion) | ||||||
| Covariate | Matched Cohort N = 87 | Unmatched Cohort N = 340 | ||||
| No Pelvic RT N = 58 | Pelvic RT N = 29 | P-VAL | No Pelvic RT N = 307 | Pelvic RT N = 33 | P-VAL | |
| Deaths * | 28 (48.28) | 17 (58.62) | 0.495 | 118 (38.44) | 20 (60.61) | 0.016 |
| Stage CIS | 1 (1.72) | 1 (3.45) | 1 | 35 (11.40) | 2 (6.06) | 0.556 |
| Stage T1 | 9 (15.52) | 4 (13.79) | 1 | 31 (10.10) | 4 (12.12) | 0.761 |
| Stage T2 | 18 (31.03) | 8 (27.59) | 0.808 | 91 (29.64) | 9 (27.27) | 0.843 |
| Stage T3 | 16 (27.59) | 9 (31.03) | 0.804 | 103 (33.55) | 9 (27.27) | 0.561 |
| Stage T4 | 14 (24.14) | 7 (24.14) | 1 | 47 (15.31) | 9 (27.27) | 0.086 |
| Node N0 | 44 (75.86) | 22 (75.86) | 1 | 235 (76.55) | 24 (72.73) | 0.668 |
| Node N1 | 4 (6.90) | 0 (0.00) | 0.296 | 23 (7.49) | 0 (0.00) | 0.147 |
| Node N2 | 8 (13.79) | 7 (24.14) | 0.243 | 37 (12.05) | 9 (27.27) | 0.028 |
| Node N3 | 2 (3.45) | 0 (0.00) | 0.55 | 12 (3.91) | 0 (0.00) | 0.615 |
| Margin | 5 (8.62) | 5 (17.24) | 0.291 | 24 (7.28) | 5 (15.15) | 0.181 |
| History of BCG use | 13 (22.41) | 7 (24.14) | 1 | 70 (22.80) | 9 (27.27) | 0.524 |
| Chemotherapy | 31 (53.45) | 17 (58.62) | 0.819 | 131 (42.67) | 19 (57.58) | 0.139 |
| Current cancer was a recurrence | 14 (24.14) | 6 (20.69) | 0.793 | 50 (16.29) | 8 (24.24) | 0.233 |
| Smoking history (Y, N) | 38 (65.52) | 18 (62.07) | 0.814 | 228 (74.27) | 20 (60.61) | 0.102 |
| High risk occupation | 10 (17.24) | 6 (20.69) | 0.772 | 43 (14.01) | 7 (21.21) | 0.298 |
| Family history of cancer | 25 (43.10) | 13 (44.83) | 1 | 102 (33.22) | 15 (45.45) | 0.179 |
| Male gender | 50 (86.21) | 24 (82.76) | 0.753 | 230 (74.92) | 27 (81.82) | 0.522 |
| Married | 44 (75.86) | 24 (82.76) | 0.586 | 206 (67.10) | 27 (81.82) | 0.113 |
| Single | 4 (6.90) | 4 (13.79) | 0.432 | 34 (11.07) | 5 (15.15) | 0.562 |
| Widow | 10 (17.24) | 1 (3.45) | 0.091 | 31 (10.10) | 1 (3.03) | 0.341 |
| PVD | 1 (1.72) | 1 (3.45) | 1 | 9 (2.93) | 1 (3.03) | 1 |
| Hydronephrosis | 9 (15.52) | 3 (10.34) | 0.743 | 31 (10.10) | 3 (9.09) | 1 |
| Bowel obstruction | 4 (6.90) | 4 (13.79) | 0.432 | 27 (8.79) | 4 (12.12) | 0.523 |
| Abscess | 1 (1.72) | 0 (0.00) | 1 | 6 (1.95) | 0 (0.00) | 1 |
| Diarrhea | 1 (1.72) | 0 (0.00) | 1 | 6 (1.95) | 0 (0.00) | 1 |
| Post-op UTI | 11 (18.97) | 4 (13.79) | 0.765 | 71 (23.13) | 5 (15.15) | 0.382 |
| Urine leak | 9 (15.52) | 2 (6.90) | 0.323 | 53 (17.26) | 3 (9.09) | 0.324 |
| Mann-Whitney U Test: median (IQR) | ||||||
| Covariate | Matched Cohort N = 87 | Unmatched Cohort N = 340 | ||||
| No Pelvic RT | Pelvic RT | P-VAL | No Pelvic RT | Pelvic RT | P-VAL | |
| Days from cystectomy to death (median (IQR)) * | 619.5 (159, 1229)00 | 158 (31, 579)00 | 0.002 | 456 (146, 1202) | 133 (30, 434)00 | 0.0001 |
| Age-adjusted CCI | 3.50 (2.00, 6.00)0 | 4.00 (2.00, 5.00)0 | 0.646 | 3.00 (1.00, 4.00)0 | 4.00 (2.00, 5.00)0 | 0.014 |
| Age at cystectomy | 72.50 (63.00, 78.00) | 71.00 (64.00, 76.00) | 0.459 | 66.00 (57.00, 74.00) | 73.00 (65.00, 78.00) | 0.014 |
| BUN | 16.50 (13.00, 23.00) | 19.00 (16.00, 23.00) | 0.402 | 15.00 (12.00, 20.00) | 20.00 (16.00, 24.00) | 0.001 |
| Creatinine | 1.10 (0.92, 1.48)0 | 1.11 (1.00, 1.40)0 | 0.663 | 1.10 (0.90, 1.30)0 | 1.17 (1.00, 1.60)0 | 0.020 |
| Hemoglobin | 13.60 (11.20, 15.20) | 13.30 (12.00, 15.30) | 0.829 | 14.00 (12.00, 15.50) | 13.20 (11.90, 15.20) | 0.181 |
| Platelet count | 237.50 (172.00, 286.00) | 228.00 (192.00, 282.00) | 0.815 | 262.00 (209.00, 321.00) | 228.00 (192.00, 283.00) | 0.029 |
CIS: Carcinoma in-situ. BCG: Bacillus Calmette-Guerin. PVD: Peripheral vascular disease. UTI: Urinary tract infection. CCI: Charlson Comorbidity Index. *Deaths and Days from cystectomy to death are placed in this table simply for convenience of indication, these do not serve as covariates in the matching process.
Table 3
Results from multivariable cox regression analysis of propensity score matched sample
| Parameter | Estimate | Standard Error | P-val | Hazard Ratio | Lower CI | Upper CI |
| History of Pelvic RT | 0.798 | 0.402 | 0.047 | 2.221 | 1.015 | 4.860 |
| Stage T1 vs. T2 | 1.175 | 0.663 | 0.076 | 3.239 | 0.889 | 11.804 |
| Stage T1 vs. T3 | 0.298 | 0.758 | 0.694 | 1.347 | 0.307 | 5.905 |
| Stage T1 vs. T4 | 0.869 | 0.695 | 0.211 | 2.386 | 0.616 | 9.243 |
| Age at cystectomy | 0.018 | 0.016 | 0.273 | 1.018 | 0.986 | 1.050 |
| Abscess | –0.261 | 1.248 | 0.835 | 0.771 | 0.068 | 8.777 |
| Bowel Obstruction | –0.705 | 0.649 | 0.278 | 0.494 | 0.139 | 1.753 |
| Diarrhea | –0.588 | 1.224 | 0.631 | 0.555 | 0.051 | 6.045 |
| UTI | 0.921 | 0.570 | 0.106 | 2.513 | 0.827 | 7.636 |
| Anastamotic urine leak | –0.312 | 0.622 | 0.616 | 0.732 | 0.217 | 2.463 |
| Married vs. single | 1.396 | 0.563 | 0.013 | 4.040 | 1.346 | 12.122 |
| Married vs. widow | –0.386 | 0.702 | 0.582 | 0.680 | 0.173 | 2.670 |
| Margin | 0.746 | 0.637 | 0.242 | 2.108 | 0.609 | 7.305 |
| Node N0 vs. N1 | 0.632 | 0.678 | 0.351 | 1.882 | 0.502 | 7.053 |
| Node N0 vs. N2 | 1.459 | 0.576 | 0.011 | 4.300 | 1.399 | 13.219 |
| Node N0 vs. N3 | 1.126 | 0.834 | 0.177 | 3.085 | 0.607 | 15.681 |
| Platelet count | –0.009 | 0.003 | 0.001 | 0.991 | 0.986 | 0.996 |
RT: Radiation therapy. UTI: Urinary tract infection.




